Flavonoid Accumulation in an Aseptic Culture of Summer Savory (Satureja hortensis L.)
Abstract
:1. Introduction
2. Results and Discussion
2.1. Flavonoid Accumulation in Aseptic Plants
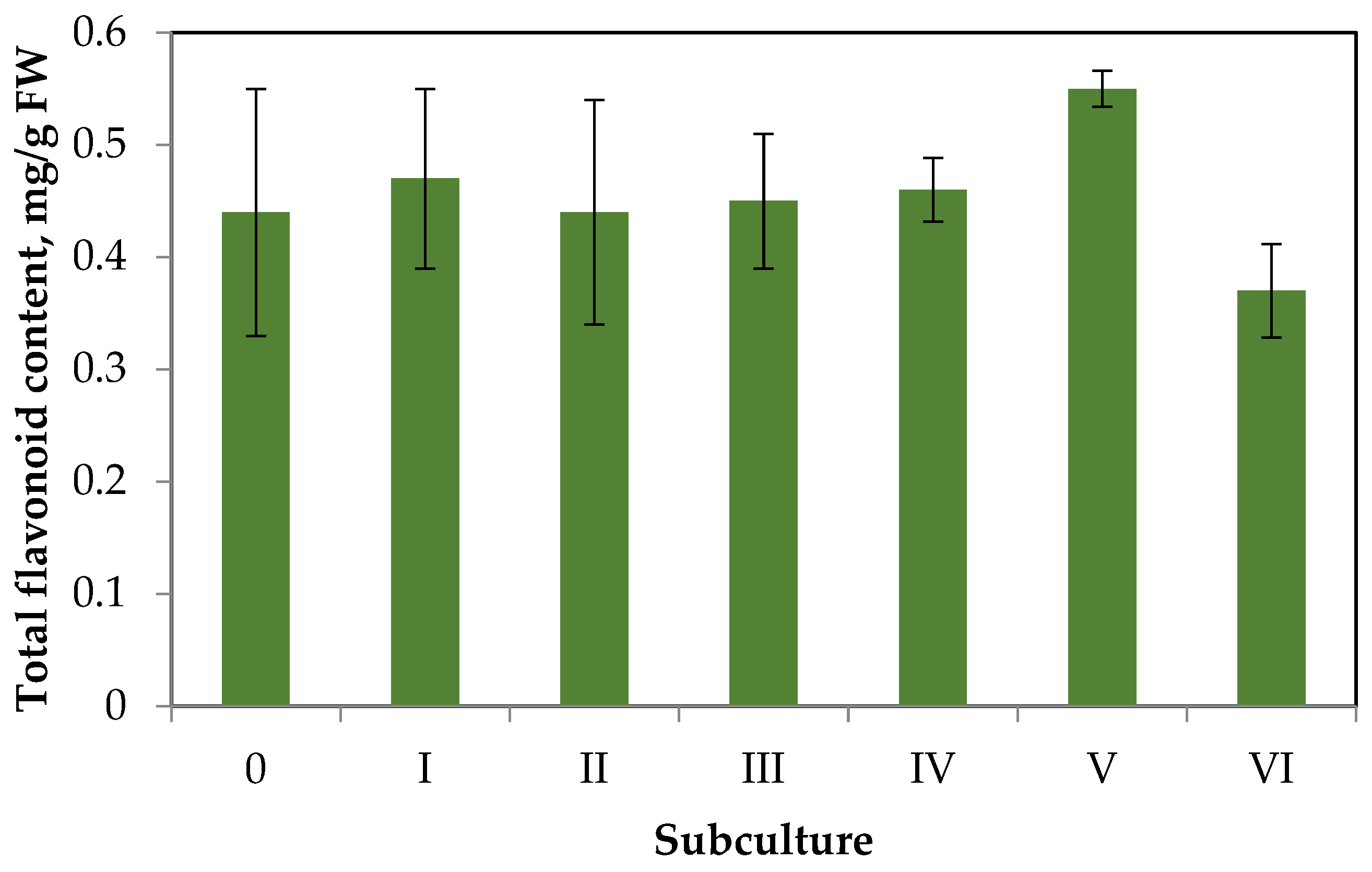
2.2. Flavonoid Accumulation in Callus Tissue
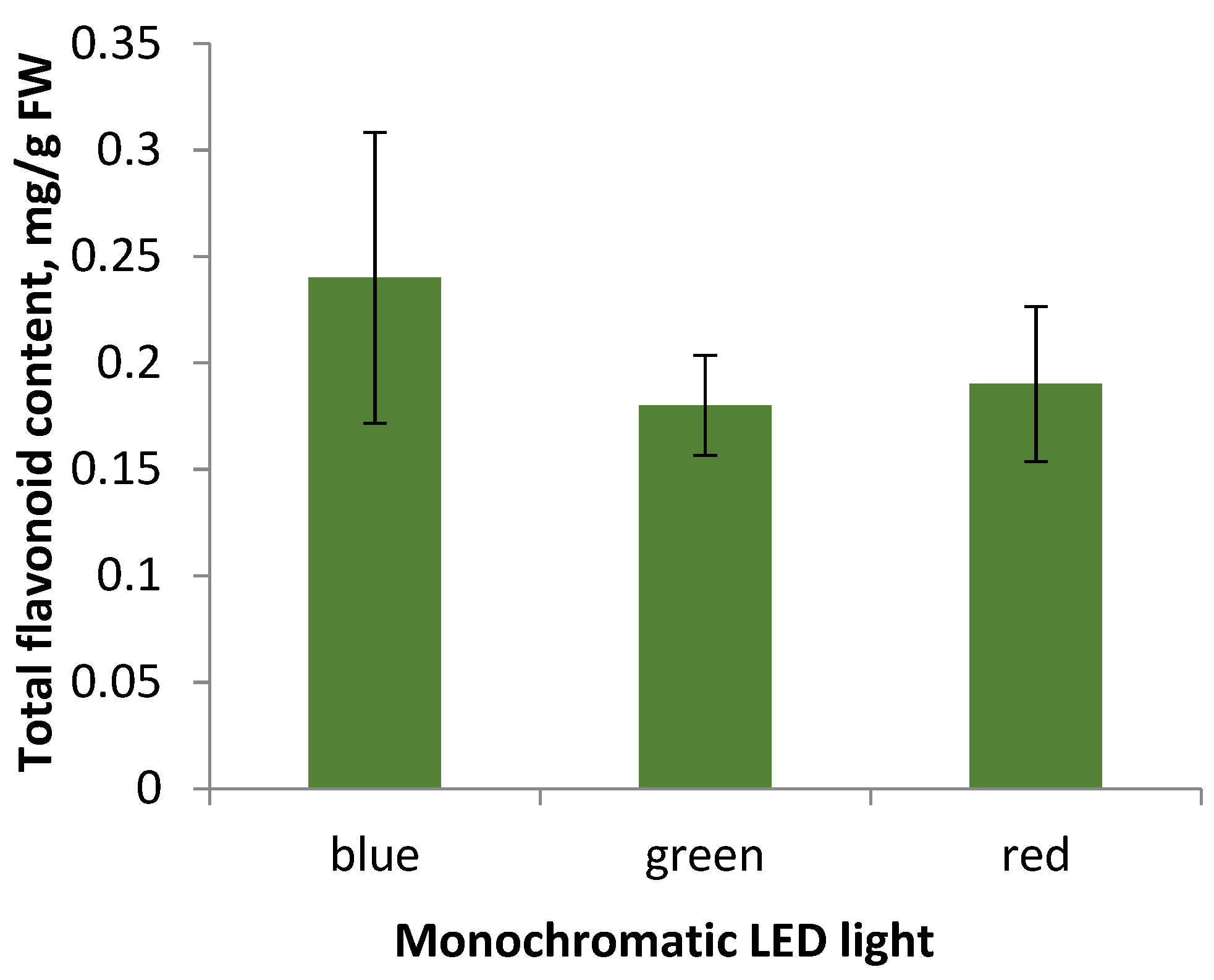
2.3. Effect of Monochromatic Light on Callus Tissue Characteristics and Flavonoid Content
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dias, D.A.; Urban, S. A historical overview of natural products in drug discovery. Metabolites 2012, 2, 303–336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giri, C.C.; Zaheer, M. Chemical elicitors versus secondary metabolite production in vitro using plant cell, tissue and organ cultures: Recent trends and a sky eye view appraisal. Plant Cell Tissue Organ Cult. 2016, 126, 1–18. [Google Scholar] [CrossRef]
- British Broadcasting Corporation (BBC) Research. Plant-Derived Drugs: Global Markets. Available online: https://www.bccresearch.com/market-research/biotechnology/botanical-and-plant-derived-drugs-global-markets-bio022h.html (accessed on 22 December 2021).
- Eibl, R.; Meier, P.; Stutz, I.; Schildberger, D.; Hühn, T.; Eibl, D. Plant cell culture technology in the cosmetics and food industries: Current state and future trends. Appl. Microbiol. Biotechnol. 2018, 102, 8661–8675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tepe, B.; Cilkiz, M. Pharmacological and phytochemical overview on Satureja. Pharm. Biol. 2016, 54, 375–412. [Google Scholar] [PubMed] [Green Version]
- Kemertelidze, E.; Sagareishvili, T.; Syrov, V.; Khushbaktova, Z.; Tsutskiridze, L.; Kurashvili, R. Saturin-effective vegetative remedy in treatment of type 2 diabetes mellitus. Georgian Med. News 2012, 2, 47–52. [Google Scholar]
- Mchedlishvili, D.; Mchedlishvili, D.; Kuchukashvili, Z.; Tabatadze, T. Influence of flavonoids isolated from Satureja hortensis L. on hypercholesterolemic rabbits. Indian J. Pharmacol. 2005, 37, 259–260. [Google Scholar]
- Kemertelidze, E.; Sagareishvili, T.; Syrov, V.; Khushbaktova, Z. Chemical compounds and pharmacological activity of the leaves of summer savory growing in Georgia. Pharm. Chem. J. 2004, 38, 33–36. [Google Scholar] [CrossRef]
- Exarchou, V.; Nenadis, N.; Tsimidou, M.; Gerothanassis, I.P.; Troganis, A.; Boskou, D. Antioxidant activities and phenolic composition of extracts from Greek oregano, Greek sage, and Summer savory. J. Agric. Food Chem. 2002, 50, 5294–5299. [Google Scholar] [CrossRef]
- Kosar, M.; Dorman, H.J.D.; Hiltunen, R. Effect of an acid treatment on the phytochemical and antioxidant characteristics of extracts from selected Lamiaceae species. Food Chem. 2005, 91, 525–533. [Google Scholar] [CrossRef]
- Chkhikvishvili, I.; Sanikidze, T.; Gogia, N.; Mchedlishvili, T.; Enukidze, M.; Machavariani, M.; Vinokur, Y.; Rodov, V. Rosmarinic acid-rich extracts of summer savory (Satureja hortensis L.) protect Jurkat T cells against oxidative stress. Oxidative Med. Cell. Longev. 2013, 2013, 456253. [Google Scholar] [CrossRef] [Green Version]
- Lung, I.; Soran, M.L.; Tudoran, C.; Măruţoiu, C. Effect of microwave irradiation on polyphenolic compounds from Satureja hortensis L. Cent. Eur. J. Chem. 2013, 11, 535–541. [Google Scholar] [CrossRef]
- Mašković, P.; Veličković, V.; Mitić, M.; Đurović, S.; Zeković, Z.; Radojković, M.; Cvetanović, A.; Švarc-Gajić, J.; Vujić, J. Summer savory extracts prepared by novel extraction methods resulted in enhanced biological activity. Ind. Crops Prod. 2017, 109, 875–881. [Google Scholar] [CrossRef]
- Boroja, T.; Katanić, G.; Rosić, D.; Selaković, J.; Joksimović, D.; Mišić, V.; Stanković, N.; Jovičić, V.; Mihailović, J. Summer savory (Satureja hortensis L.) extract: Phytochemical profile and modulation of cisplatin-induced liver, renal and testicular toxicity. Food Chem. Toxicol. 2018, 118, 252–263. [Google Scholar] [CrossRef] [PubMed]
- Malankina, E.L.; Romanova, N.G.; Solopov, S.G.; Tkacheva, T.N. Garden savory—A promising spicy culture for the Moscow region. Kartof. I Ovoshi 2018, 11, 25–27. [Google Scholar]
- Plánder, S.; Gontaru, L.; Blazics, B.; Veres, K.; Kéry, Á.; Kareth, S.; Simándi, B. Major antioxidant constituents from Satureja hortensis L. extracts obtained with different solvents. Eur. J. Lipid Sci. Technol. 2012, 114, 772–779. [Google Scholar] [CrossRef]
- Bros, I.; Soran, M.L.; Briciu, R.D.; Cobzac, S.C. HPTLC quantification of some flavonoids in extracts of Satureja hortensis L. obtained by use of different techniques. J. Planar Chromatogr. Mod. TLC 2009, 22, 25–28. [Google Scholar] [CrossRef]
- Gupta, S.D.; Jatothu, B. Fundamentals and applications of light-emitting diodes LEDs in in vitro plant growth and morphogenesis. Plant Biotechnol. Rep. 2013, 7, 211–220. [Google Scholar] [CrossRef]
- Okamoto, K.; Yanagi, T.; Takita, S. Development of plant growth apparatus using blue and red LED as artificial light source. Acta Hortic. 1996, 440, 111–116. [Google Scholar] [CrossRef]
- Jao, R.C.; Fang, W. Growth of potato plantlets in vitro is different when provided concurrent versus alternating blue and red light photoperiods. HortScience 2004, 39, 380–382. [Google Scholar] [CrossRef] [Green Version]
- Jao, R.-C.; Lai, C.-C.; Fang, W.; Chang, S.-F. Effects of red light on the growth of Zantedschia plantlets in vitro and tuber formation using light-emitting diodes. HortScience 2005, 40, 436–438. [Google Scholar] [CrossRef] [Green Version]
- Gam, D.T.; Khoi, P.H.; Ngoc, P.B.; Linh, L.K.; Hung, N.K.; Anh, P.T.L.; Thu, N.T.; Hien, N.T.T.; Khanh, T.D.; Ha, C.H. LED Lights Promote Growth and Flavonoid Accumulation of Anoectochilus roxburghii and Are Linked to the Enhanced Expression of Several Related Genes. Plants 2020, 9, 1344. [Google Scholar] [CrossRef] [PubMed]
- Shulgina, A.A.; Kalashnikova, E.A.; Tarakanov, I.G.; Kirakosyan, R.N.; Cherednichenko, M.Y.; Polivanova, O.B.; Baranova, E.N.; Khaliluev, M.R. Influence of Light Conditions and Medium Composition on Morphophysiological Characteristics of Stevia rebaudiana Bertoni In Vitro and In Vivo. Horticulturae 2021, 7, 195. [Google Scholar] [CrossRef]
- Zhang, X.H.; Zheng, X.T.; Sun, B.Y.; Peng, C.L.; Chow, W.S. Over-expression of the CHS gene enhances resistance of Arabidopsis leaves to high Light. Environ. Exp. Bot. 2018, 154, 33–43. [Google Scholar] [CrossRef] [Green Version]
- Ma, Z.H.; Li, W.F.; Mao, J.; Li, W.; Zuo, C.W.; Zhao, X.; Dawuda, M.M.; Shi, X.Y.; Chen, B.H. Synthesis of light-inducible and light-independent anthocyanins regulated by specific genes in grape ‘Marselan’ (V. vinifera L.). PeerJ 2019, 7, e6521. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Su, M.; Li, H.; Zeng, B.; Chang, Q.; Lai, Z. Effects of supplemental lighting with different light qualities on growth and secondary metabolite content of Anoectochilus roxburghii. PeerJ 2018, 6, e5274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, H.K.; Chen, Y.Y.; Hu, T.T.; Zhang, S.J.; Zhang, Y.H.; Zhao, T.Y.; Yu, H.E.; Kang, Y.F. The influence of light-emitting diodes on the phenolic compounds and antioxidant activities in pea sprouts. J. Funct. Foods 2016, 25, 459–465. [Google Scholar] [CrossRef]
- Azad, M.O.K.; Kim, W.W.; Park, C.H.; Cho, D.H. Effect of Artificial LED light and far infrared irradiation on phenolic compound, isoflavones and antioxidant capacity in soybean (Glycine max L.) Sprout. Foods 2018, 7, 174. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Chen, Q.; Zhuang, S.; Wen, Y.; Cheng, W.; Zeng, Z.; Jiang, T.; Tang, C. Effect of Anoectochilus roxburghii flavonoids extract on H2O2—Induced oxidative stress in LO2 cells and D-gal induced aging mice model. J. Ethnopharmacol. 2020, 254, 112670. [Google Scholar] [CrossRef] [PubMed]
- Pistelli, L.; Noccioli, C.; D’Angiolillo, F.; Pistelli, L. Composition of volatile in micropropagated and field grown aromatic plants from Tuscany Islands. Acta Biochim. Pol. 2013, 60, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Bharati, A.J.; Bansal, Y.K. In vitro production of flavonoids: A review. World J. Pharm. Pharm. Sci. 2014, 3, 508–533. [Google Scholar]
- Yeoman, M.M.; Yeoman, C.L. Manipulating secondary metabolism in cultured plant cells. New Phytol. 1996, 134, 553–569. [Google Scholar] [CrossRef]
- Del Valle, J.C.; Buide, M.L.; Casimiro-Soriguer, I.; Whittall, J.B.; Narbona, E. On flavonoid accumulation in different plant parts: Variation patterns among individuals and populations in the shore campion (Silene littorea). Front. Plant Sci. 2015, 6, 939. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Lin, Y.; Chen, X.; Bai, Y.; Wang, C.; Xu, X.; Wang, Y.; Lai, Z. Effects of blue light on flavonoid accumulation linked to the expression of miR393, miR394 and miR395 in longan embryogenic calli. PLoS ONE 2018, 13, e0191444. [Google Scholar] [CrossRef] [Green Version]
- Zhao, D.; Li, M.; Xing, J.; Tong, Z. Effects of light on cell growth and flavonoids biosynthesis in callus cultures of Saussurea medusa Maxim. Acta Photophysiol. Sin. 1999, 25, 127–132. [Google Scholar]
- Deng, Y.; Yao, J.; Wang, X.; Guo, H.; Duan, D. Transcriptome sequencing and comparative analysis of Saccharina japonica (Laminariales, Phaeophyceae) under blue light induction. PLoS ONE 2012, 7, e39704. [Google Scholar] [CrossRef]
- Son, K.H.; Oh, M.M. Leaf shape, growth, and antioxidant phenolic compounds of two lettuce cultivars grown under various combinations of blue and red light-emitting diodes. Hortscience 2013, 48, 988–995. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]




| Plant Material | Subculture | Total Flavonoid Content, mg/g FW |
|---|---|---|
| Leaf | 0 | 8.17 ± 1.07 |
| Stem | 0 | 0.34 ± 0.04 |
| Leaf | 1 | 8.20 ± 0.78 |
| Stem | 1 | 0.93 ± 0.15 |
| Leaf | 2 | 8.35 ± 0.17 |
| Stem | 2 | 1.50 ± 0.22 |
| Flower bud | 2 | 7.55 ± 0.29 |
| Leaf | 3 | 7.02 ± 0.90 |
| Stem | 3 | 1.58 ± 0.76 |
| Corolla | 3 | 0.78 ± 0.12 |
| Calyx | 3 | 5.27 ± 0.28 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khlebnikova, D.A.; Efanova, E.M.; Danilova, N.A.; Shcherbakova, Y.V.; Rivera Sidorova, I. Flavonoid Accumulation in an Aseptic Culture of Summer Savory (Satureja hortensis L.). Plants 2022, 11, 533. https://doi.org/10.3390/plants11040533
Khlebnikova DA, Efanova EM, Danilova NA, Shcherbakova YV, Rivera Sidorova I. Flavonoid Accumulation in an Aseptic Culture of Summer Savory (Satureja hortensis L.). Plants. 2022; 11(4):533. https://doi.org/10.3390/plants11040533
Chicago/Turabian StyleKhlebnikova, Darya A., Evgeniya M. Efanova, Nina A. Danilova, Yaroslava V. Shcherbakova, and Irina Rivera Sidorova. 2022. "Flavonoid Accumulation in an Aseptic Culture of Summer Savory (Satureja hortensis L.)" Plants 11, no. 4: 533. https://doi.org/10.3390/plants11040533
APA StyleKhlebnikova, D. A., Efanova, E. M., Danilova, N. A., Shcherbakova, Y. V., & Rivera Sidorova, I. (2022). Flavonoid Accumulation in an Aseptic Culture of Summer Savory (Satureja hortensis L.). Plants, 11(4), 533. https://doi.org/10.3390/plants11040533






