Seasonal Effects of Glucosinolate and Sugar Content Determine the Pungency of Small-Type (Altari) Radishes (Raphanus sativus L.)
Abstract
:1. Introduction
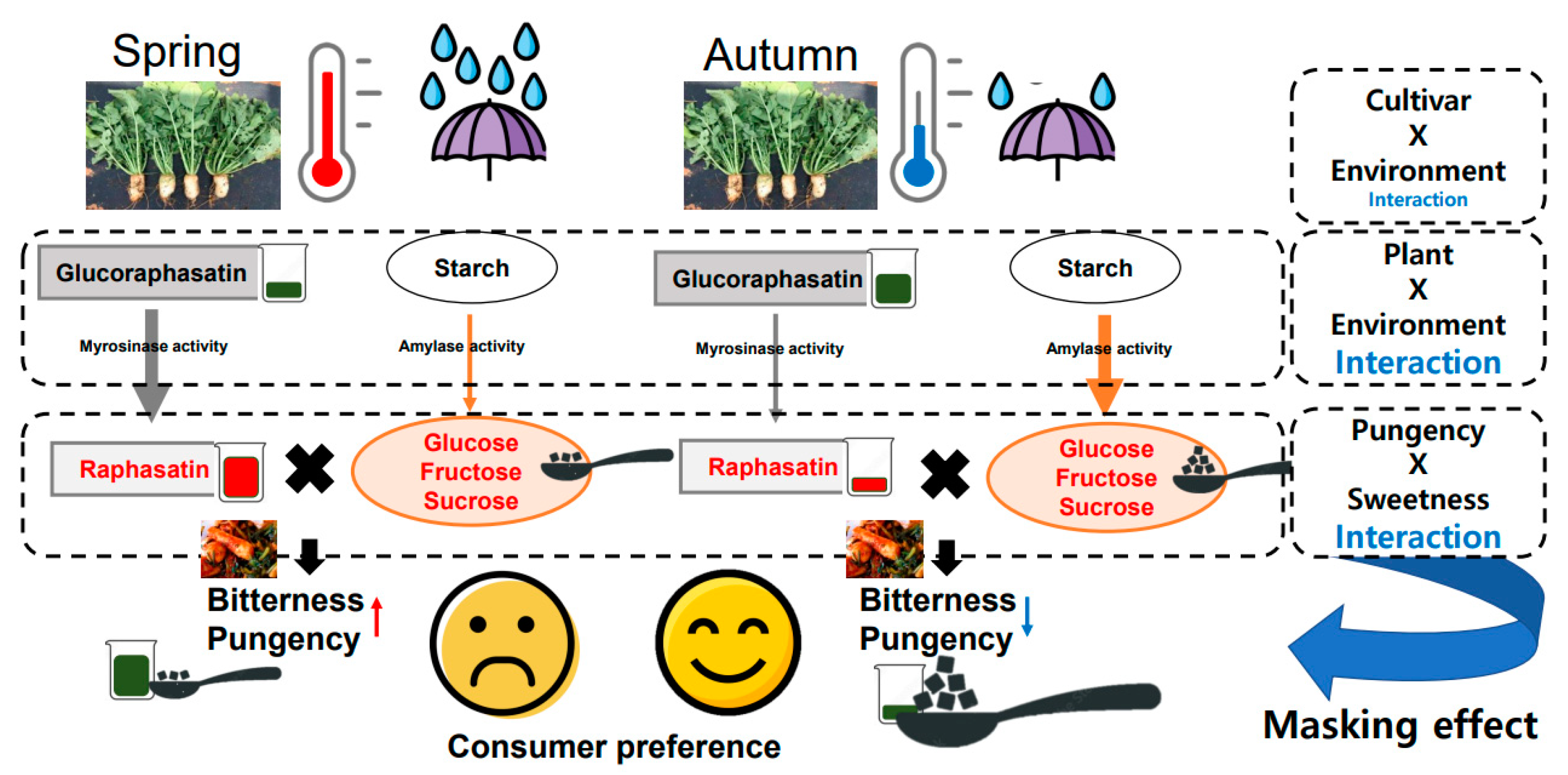
2. Results and Discussion
2.1. Growing Environmental Factors
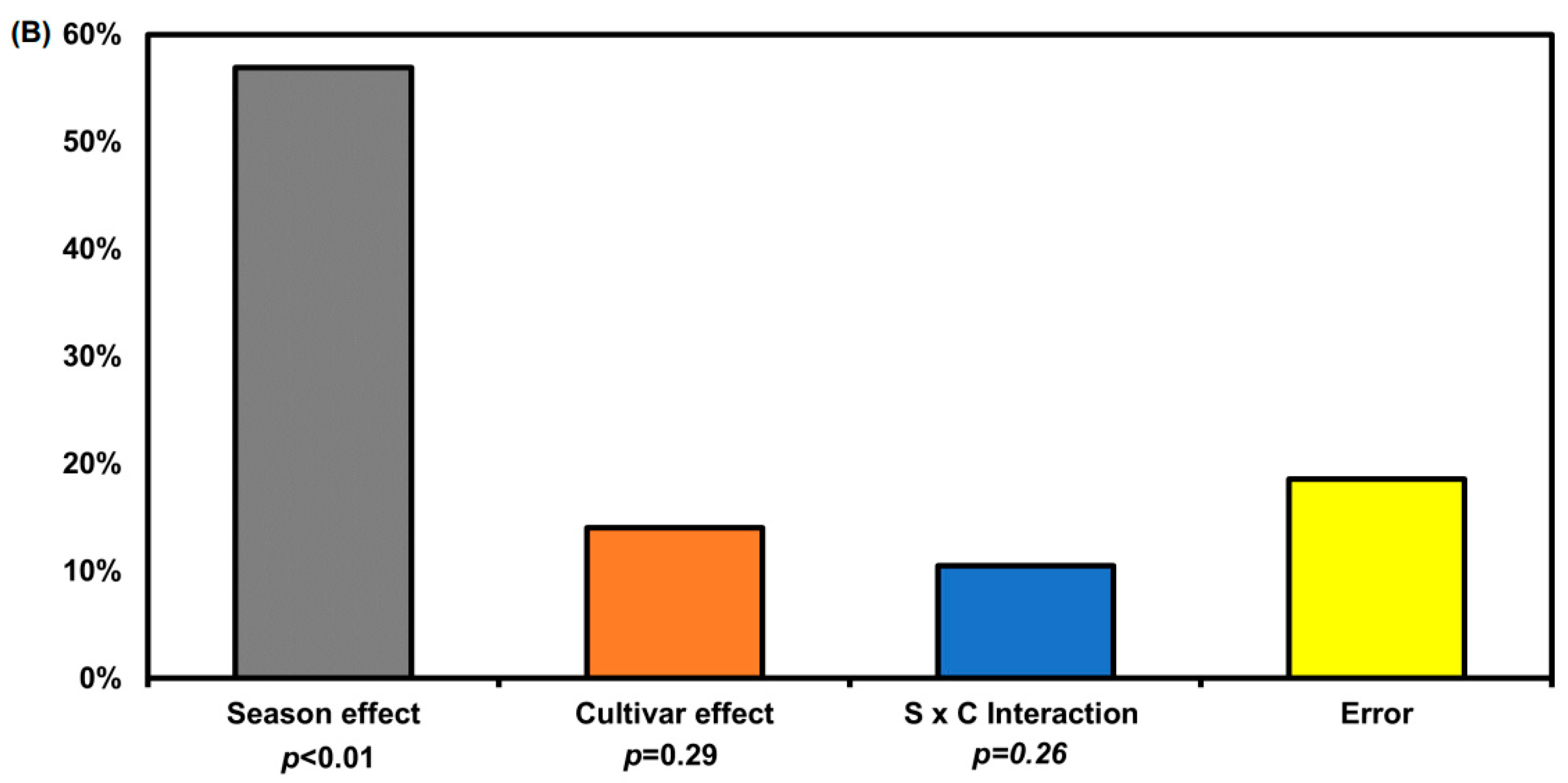
2.2. Quantification of Glucosinolate
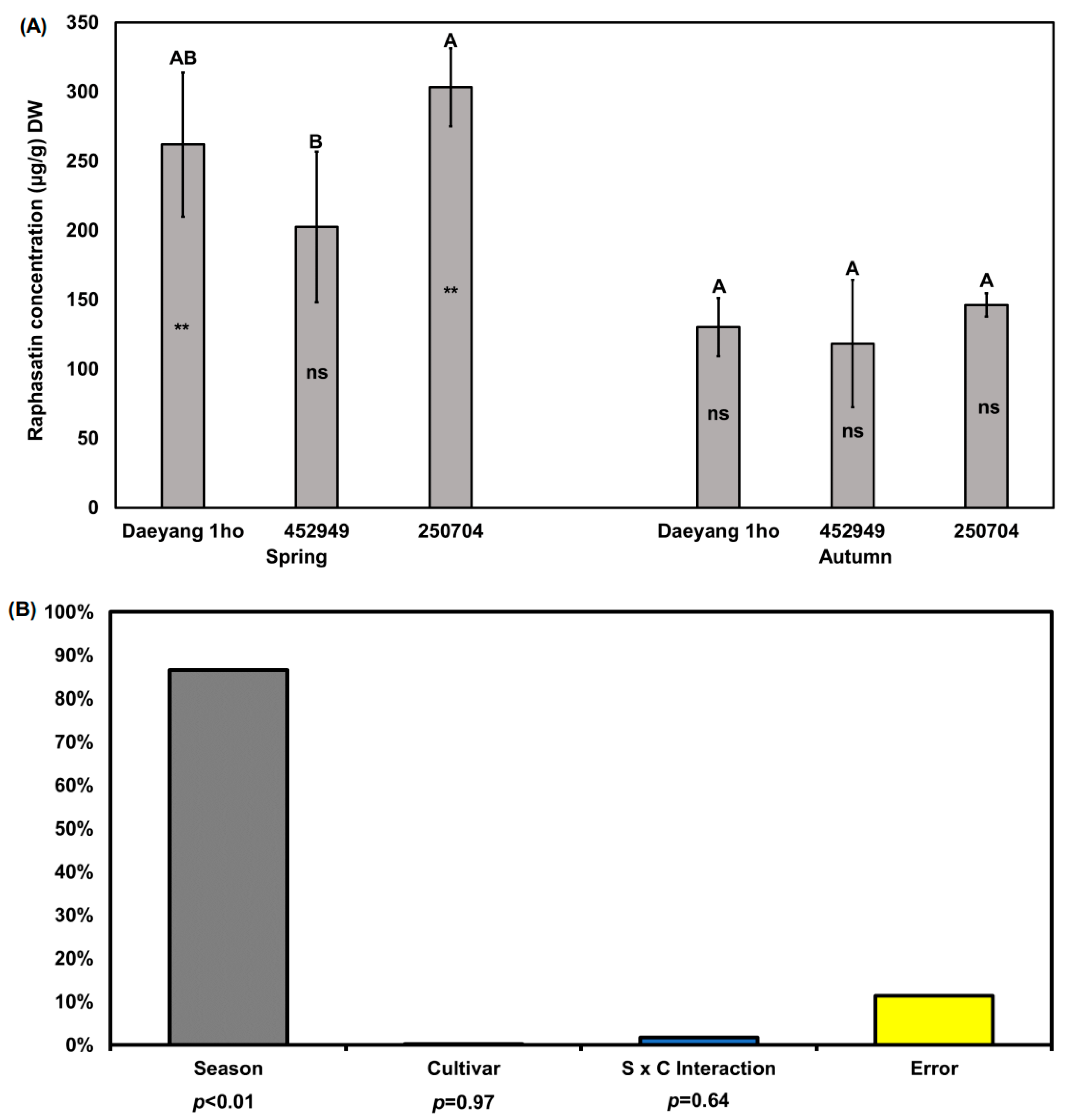
2.3. Glucosinolate Hydrolysis Products
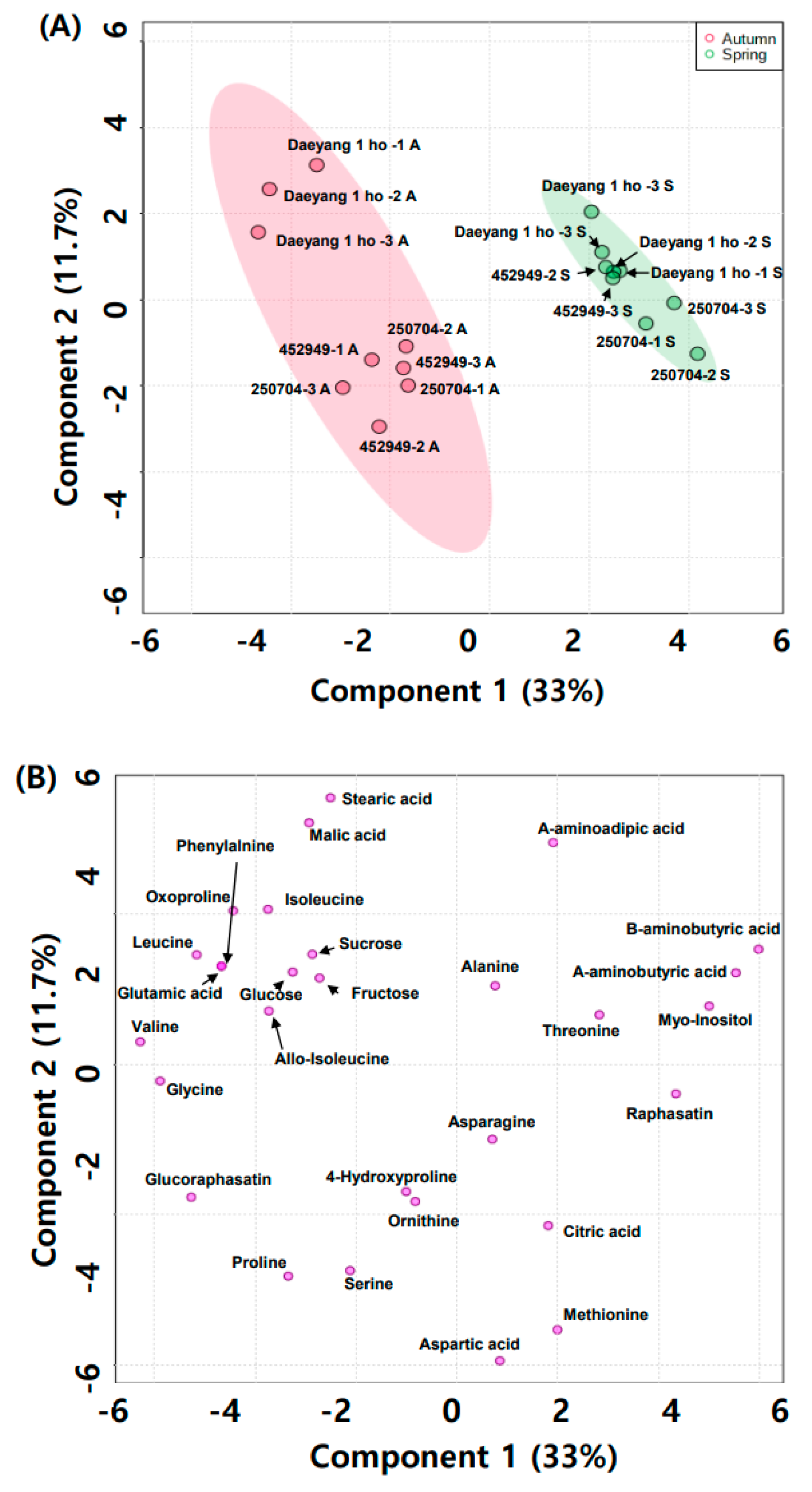
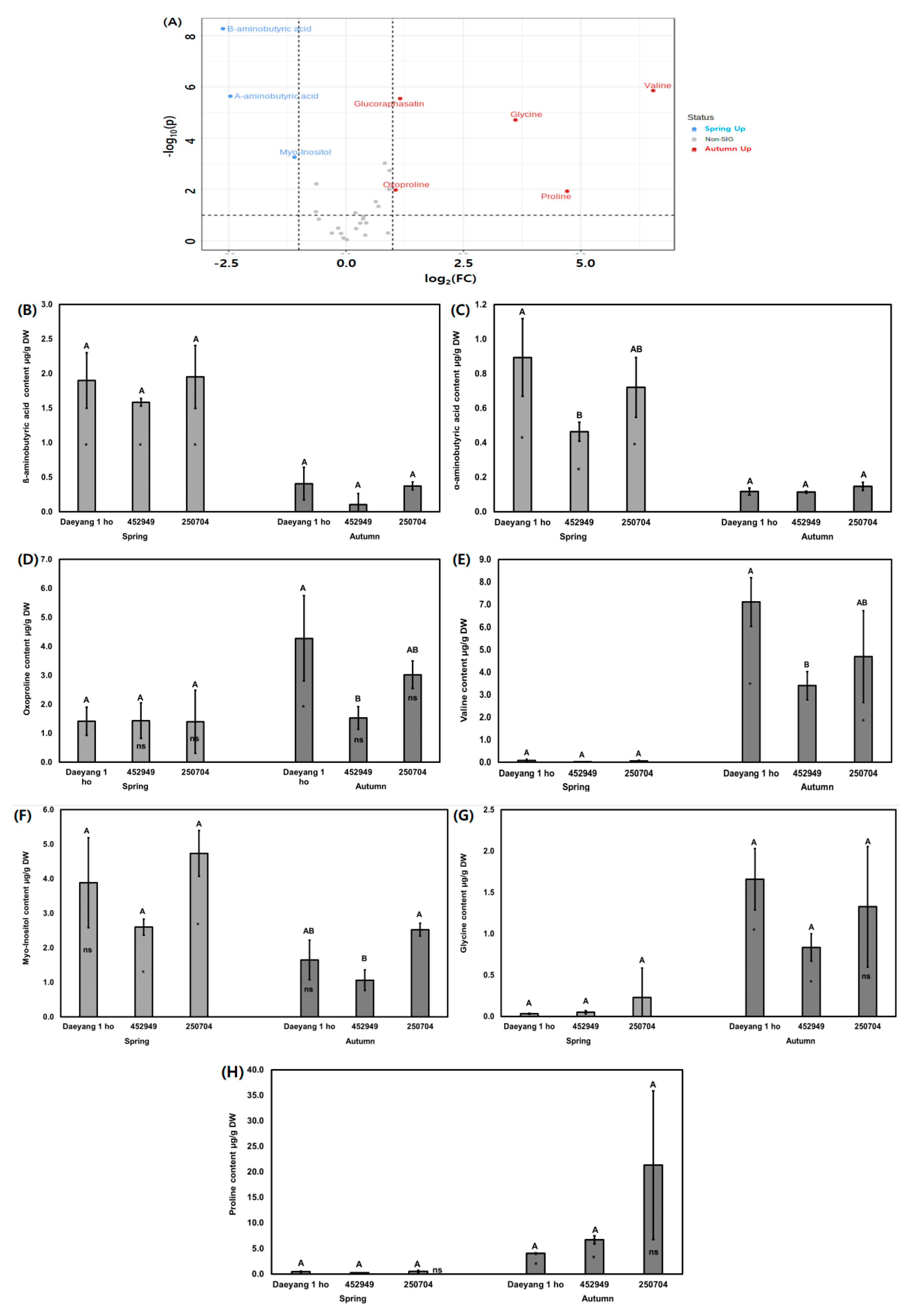
2.4. Water-Soluble Primary Metabolite Changes with Season
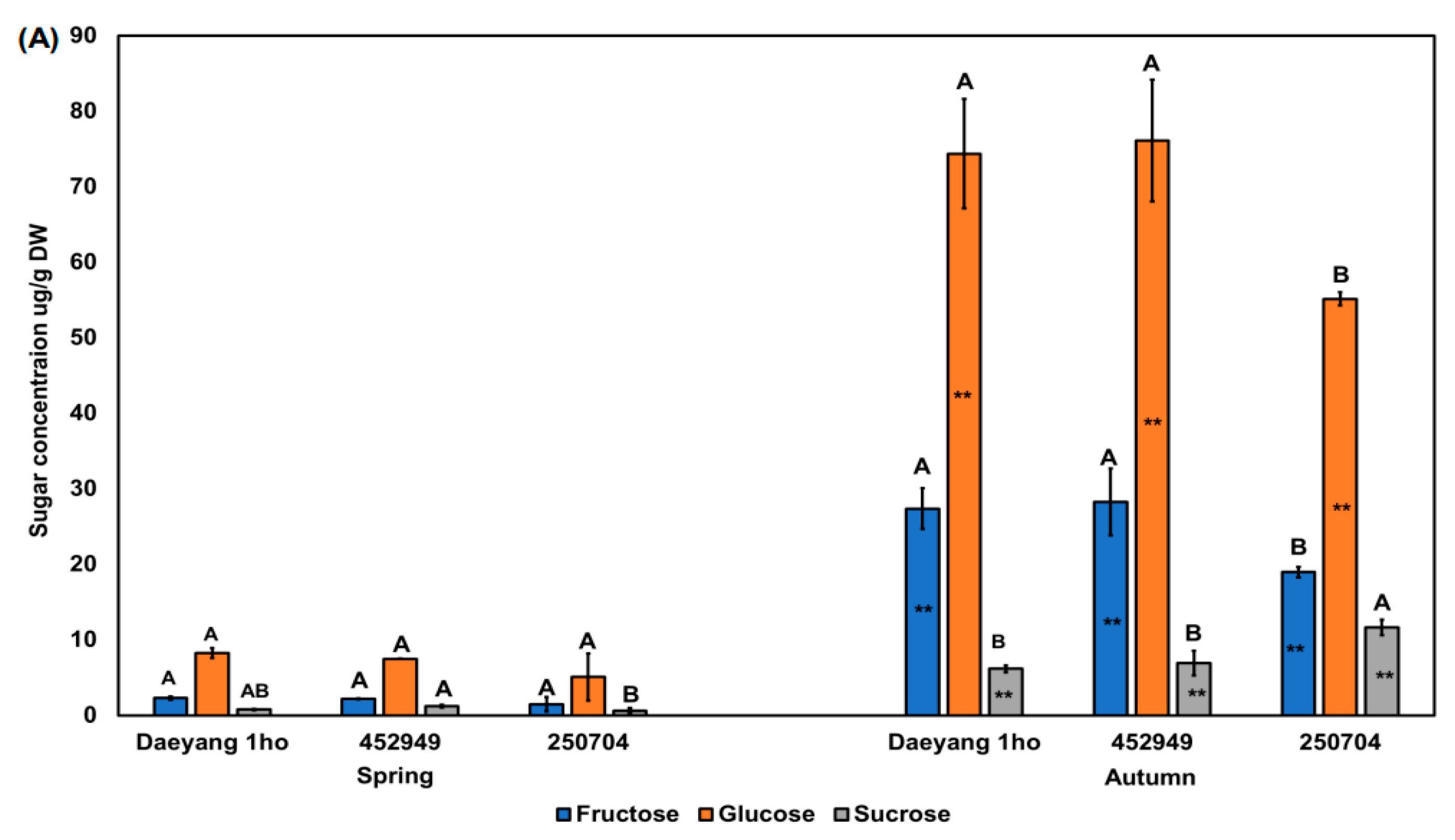
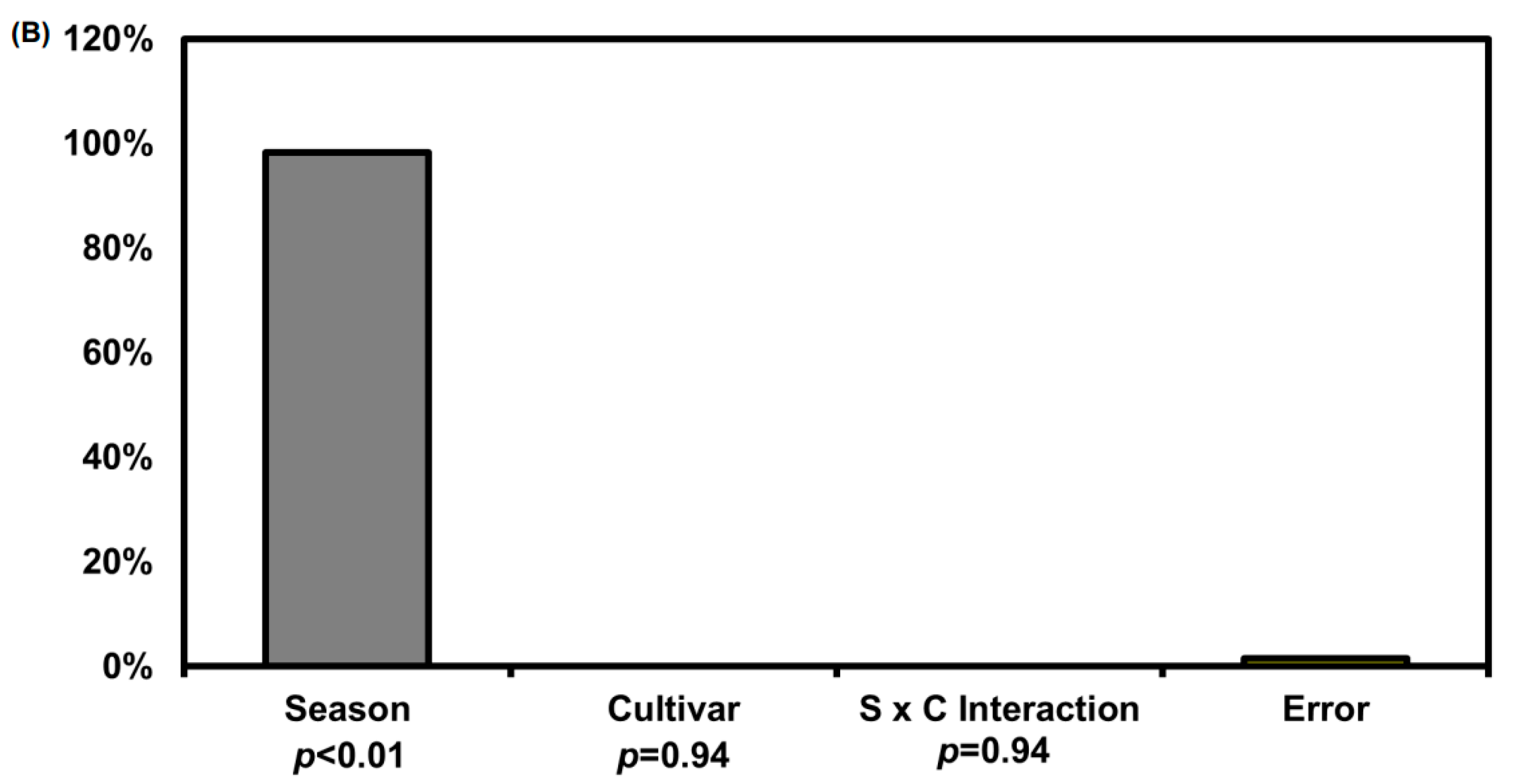
2.5. Sweetness Value Based on Major Sugar Contents
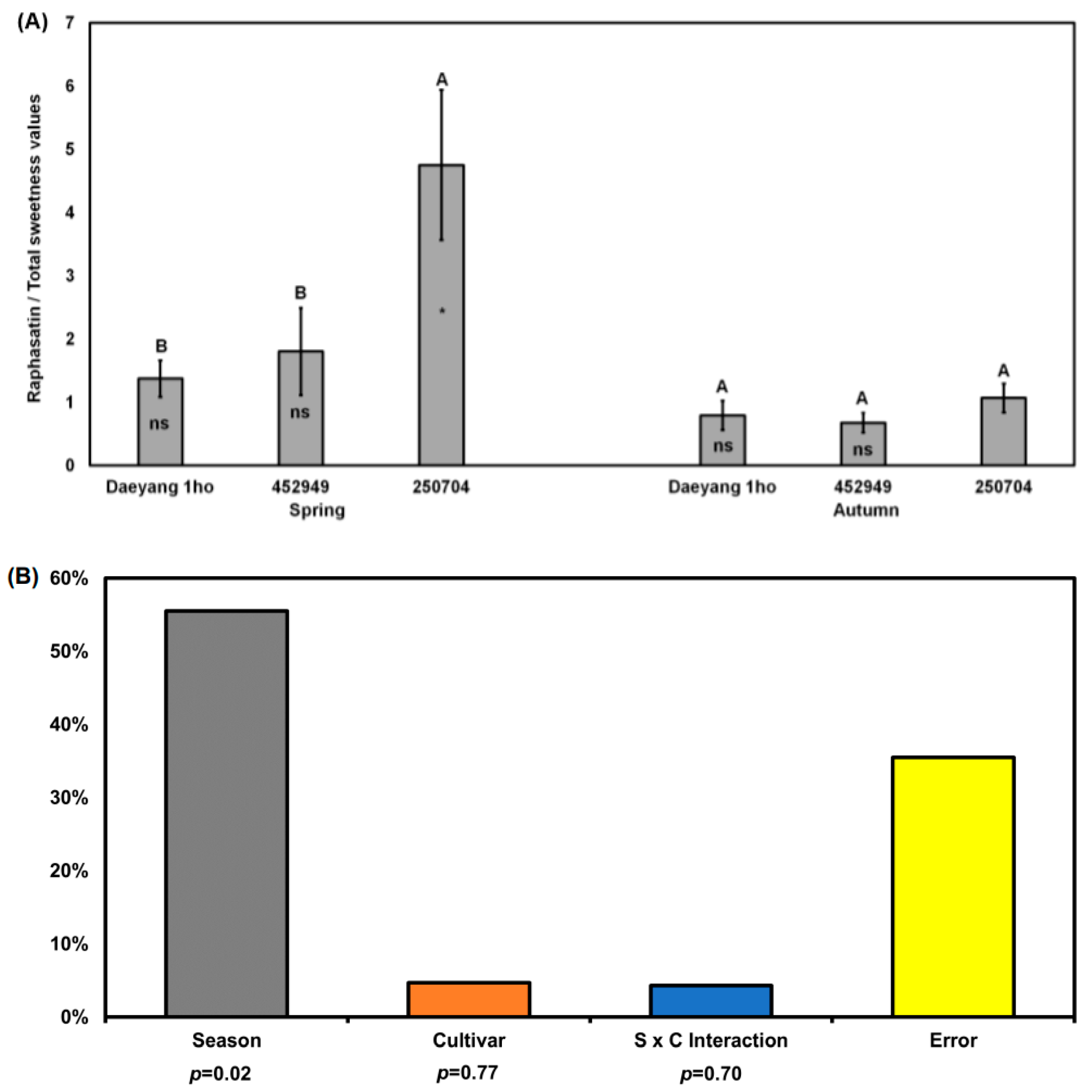
2.6. Ratio between Raphasatin and Total Sweetness Values from Sugars
3. Materials and Methods
3.1. Cultivation and Growing Conditions
3.2. Quantification of Glucosinolate
3.3. Glucosinolate Hydrolysis Products Measurement
3.4. Water-Soluble Primary Metabolite and Sugar Contents
3.5. Raphasatin Content/Total Sweetness Value from Sugars Ratio
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Carlson, D.G.; Daxenbichler, M.E.; VanEtten, C.H.; Hill, C.B.; Williams, P.H. Glucosinolates in Radish Cultivars. J. Am. Soc. Hortic. Sci. 1985, 110, 634–638. [Google Scholar]
- Lee, O.N.; Park, H.Y. Assessment of genetic diversity in cultivated radishes (Raphanus sativus) by agronomic traits and SSR markers. Sci. Hortic. 2017, 223, 19–30. [Google Scholar] [CrossRef]
- Ryu, K.D.; Chung, D.H.; Kim, J.K. Comparison of Radish Cultivars for Physicochemical Properties and Kakdugi Preparation. Korean J. Food Sci. Technol. 2000, 32, 681–690. [Google Scholar]
- Takeshi, N.; Kitashiba, H. The Radish Genome; Springer: New York, NY, USA, 2017. [Google Scholar]
- Lee, Y.C. Kimchi: The famous fermented vegetable product in Korea. Food Rev. Int. 1991, 7, 399–415. [Google Scholar] [CrossRef]
- Han, E.S. Progress of Korea Kimchi Industry. Food Sci. Ind. 2020, 53, 422–434. [Google Scholar]
- Jang, D.-J.; Chung, K.R.; Yang, H.J.; Kim, K.-S.; Kwon, D.Y. Discussion on the origin of kimchi, representative of Korean unique fermented vegetables. J. Ethn. Foods 2015, 2, 126–136. [Google Scholar] [CrossRef] [Green Version]
- Sadowski, J.; Chittaranjan, K. Genetics, Genomics and Breeding of Vegetable Brassicas; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Giamoustaris, A.; Mithen, R. Genetics of aliphatic glucosinolates. IV. Side-chain modification in Brassica oleracea. Theor. Appl. Genet. 1996, 93, 1006–1010. [Google Scholar] [CrossRef]
- Ishida, M.; Nagata, M.; Ohara, T.; Kakizaki, T.; Hatakeyama, K.; Nishio, T. Small variation of glucosinolate composition in Japanese cultivars of radish (Raphanus sativus L.) requires simple quantitative analysis for breeding of glucosinolate component. Breed. Sci. 2012, 62, 63–70. [Google Scholar] [CrossRef] [Green Version]
- Yi, G.; Lim, S.; Chae, W.B.; Park, J.E.; Park, H.R.; Lee, E.J.; Huh, J.H. Root Glucosinolate Profiles for Screening of Radish (Raphanus sativus L.) Genetic Resources. J. Agric. Food Chem. 2016, 64, 61–70. [Google Scholar] [CrossRef]
- Kim, M.-R.; Rhee, H.-S. Purification of 4-Methylthio-3-Butenyl Isothiocyanate the Pungent Principle in Radish Roots by Rp-Hplc. Korean J. Food Cook. Sci. 1986, 2, 16–20. [Google Scholar]
- Ku, K.-M.; Jeffery, E.H.; Juvik, J.A.; Kushad, M.M. Correlation of Quinone Reductase Activity and Allyl Isothiocyanate Formation Among Different Genotypes and Grades of Horseradish Roots. J. Agric. Food Chem. 2015, 63, 2947–2955. [Google Scholar] [CrossRef]
- Nakamura, Y.; Nakamura, K.; Asai, Y.; Wada, T.; Tanaka, K.; Matsuo, T.; Okamoto, S.; Meijer, J.; Kitamura, Y.; Nishikawa, A.; et al. Comparison of the Glucosinolate−Myrosinase Systems among Daikon (Raphanus sativus, Japanese White Radish) Varieties. J. Agric. Food Chem. 2008, 56, 2702–2707. [Google Scholar] [CrossRef]
- Wang, J.; Qiu, Y.; Wang, X.; Yue, Z.; Yang, X.; Chen, X.; Zhang, X.; Shen, D.; Wang, H.; Song, J.; et al. Insights into the species-specific metabolic engineering of glucosinolates in radish (Raphanus sativus L.) based on comparative genomic analysis. Sci. Rep. 2017, 7, 16040. [Google Scholar] [CrossRef] [Green Version]
- Liu, M.; Seo, H.Y.; Min, S.; Ku, K.M. Phytonutrients and Metabolism Changes in Topped Radish Root and Its Detached Leaves During 1 °C Cold Postharvest Storage. Horticulturae 2022, 8, 42. [Google Scholar] [CrossRef]
- Wei, J.; Miao, H.; Wang, Q. Effect of glucose on glucosinolates, antioxidants and metabolic enzymes in Brassica sprouts. Sci. Hortic. 2011, 129, 535–540. [Google Scholar] [CrossRef]
- Charron, C.S.; Saxton, A.M.; Sams, C.E. Relationship of climate and genotype to seasonal variation in the glucosinolate-myrosinase system. II. Myrosinase activity in ten cultivars of Brassica oleracea grown in fall and spring seasons. J. Sci. Food Agric. 2005, 85, 682–690. [Google Scholar] [CrossRef]
- Charron, C.S.; Sams, C.E. Glucosinolate Content and Myrosinase Activity in Rapid-cycling Brassica oleracea Grown in a Controlled Environment. J. Am. Soc. Hortic. Sci. 2004, 129, 321–330. [Google Scholar] [CrossRef] [Green Version]
- Rosa, E.A.; Rodrigues, P.M.F. The effect of light and temperature on glucosinolate concentration in the leaves and roots of cabbage seedlings. J. Sci. Food Agric. 1998, 78, 208–212. [Google Scholar] [CrossRef]
- Rosa, E.; Rodrigues, A.S. Total and Individual Glucosinolate Content in 11 Broccoli Cultivars Grown in Early and Late Seasons. HortScience 2001, 36, 56–59. [Google Scholar] [CrossRef]
- Padilla, G.; Cartea, M.E.; Velasco, P.; De Haro, A.; Ordás, A. Variation of glucosinolates in vegetable crops of Brassica rapa. Phytochemistry 2007, 68, 536–545. [Google Scholar] [CrossRef]
- del Carmen Martínez-Ballesta, M.; Moreno, D.A.; Carvajal, M. The Physiological Importance of Glucosinolates on Plant Response to Abiotic Stress in Brassica. Int. J. Mol. Sci. 2013, 14, 11607–11625. [Google Scholar] [CrossRef] [Green Version]
- Schreiner, M.; Huyskens-Keil, S.; Peters, P.; Schonhof, I.; Krumbein, A.; Widell, S. Seasonal climate effects on root colour and compounds of red radish. J. Sci. Food Agric. 2002, 82, 1325–1333. [Google Scholar] [CrossRef]
- Sarwar, M.; Kirkegaard, J.A. Biofumigation potential of brassicas II: Effect of environment and ontogeny on glucosinolate production and implications for screening. Plant Soil 1998, 201, 91–101. [Google Scholar] [CrossRef]
- Zhang, H.; Schonhof, I.; Krumbein, A.; Gutezeit, B.; Li, L.; Stützel, H.; Schreiner, M. Water supply and growing season influence glucosinolate concentration and composition in turnip root (Brassica rapa ssp. rapifera L.). J. Plant Nutr. Soil Sci. 2008, 171, 255–265. [Google Scholar] [CrossRef]
- Cartea, M.E.; Velasco, P.; Obregón, S.; Padilla, G.; de Haro, A. Seasonal Variation in Glucosinolate Content in Brassica Oleracea Crops Grown in Northwestern Spain. Phytochemistry 2008, 69, 403–410. [Google Scholar] [CrossRef]
- Sánchez-Pujante, P.J.; Borja-Martínez, M.; Pedreño, M.Á.; Almagro, L. Biosynthesis and Bioactivity of Glucosinolates and Their Production in Plant in Vitro Cultures. Planta 2017, 246, 19–32. [Google Scholar] [CrossRef]
- Mérillon, J.M.; Ramawat, K.G. Glucosinolates; Springer: New York, NY, USA, 2017. [Google Scholar]
- Hayata, Y.; Shinohara, Y.; Suzuki, Y. The Effect of High Temperature on the Growth and Endogenous Substances of Radish Root. Jpn. Soc. Hortic. Sci. 1986, 55, 51–55. [Google Scholar] [CrossRef]
- Beck, T.K.; Jensen, S.; Bjoern, G.K.; Kidmose, U. The Masking Effect of Sucrose on Perception of Bitter Compounds in Brassica Vegetables. J. Sens. Stud. 2014, 29, 190–200. [Google Scholar] [CrossRef]
- Kim, M.R.; Rhee, H.S. Decrease of Pungency in “Radish Kimchi” during Fermentation. J. Food Sci. 1993, 58, 128–131. [Google Scholar] [CrossRef]
- Coogan, R.; Wills, R.; Nguyen, V. Pungency levels of white radish (Raphanus sativus L.) grown in different seasons in Australia. Food Chem. 2001, 72, 1–3. [Google Scholar] [CrossRef]
- Chorol, S.; Angchok, D.; Stobdan, T. Irrigation timing as a glucosinolate alteration factor in radish (Raphanus sativus L.) (Gya Labuk and Tsentay Labuk) in the Indian Trans-Himalayan region of Ladakh. J. Food Compos. Anal. 2021, 100, 103904. [Google Scholar] [CrossRef]
- Ciska, E.; Martyniak-Przybyszewska, B.; Kozlowska, H. Content of Glucosinolates in Cruciferous Vegetables Grown at the Same Site for Two Years under Different Climatic Conditions. J. Agric. Food Chem. 2000, 48, 2862–2867. [Google Scholar] [CrossRef] [PubMed]
- Bell, L.; Oloyede, O.O.; Lignou, S.; Wagstaff, C.; Methven, L. Taste and Flavor Perceptions of Glucosinolates, Isothiocyanates, and Related Compounds. Mol. Nutr. Food Res. 2018, 62, e1700990. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiu, Y.-C.; Matak, K.; Ku, K.-M. Methyl jasmonate treated broccoli: Impact on the production of glucosinolates and consumer preferences. Food Chem. 2019, 299, 125099. [Google Scholar] [CrossRef] [PubMed]
- Bones, A.; Rossiter, J. The enzymic and chemically induced decomposition of glucosinolates. Phytochemistry 2006, 67, 1053–1067. [Google Scholar] [CrossRef]
- Becker, T.M.; Juvik, J.A. The Role of Glucosinolate Hydrolysis Products from Brassica Vegetable Consumption in Inducing Antioxidant Activity and Reducing Cancer Incidence. Diseases 2016, 4, 22. [Google Scholar] [CrossRef] [Green Version]
- Oliviero, T.; Verkerk, R.; Van Boekel, M.; Dekker, M. Effect of water content and temperature on inactivation kinetics of myrosinase in broccoli (Brassica oleracea var. italica). Food Chem. 2014, 163, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Din, J.; Khan, S.U.; Ali, I.; Gurmani, A.R. Physiological and Agronomic Response of Canola Varieties to Drought Stress. J. Anim. Plant Sci. 2011, 21, 78–82. [Google Scholar]
- Akram, N.A.; Iqbal, M.; Muhammad, A.; Ashraf, M.; Al-Qurainy, F.; Shafiq, S. Aminolevulinic acid and nitric oxide regulate oxidative defense and secondary metabolisms in canola (Brassica napus L.) under drought stress. Protoplasma 2018, 255, 163–174. [Google Scholar] [CrossRef]
- Akram, N.A.; Waseem, M.; Ameen, R.; Ashraf, M. Trehalose pretreatment induces drought tolerance in radish (Raphanus sativus L.) plants: Some key physio-biochemical traits. Acta Physiol. Plant. 2016, 38, 1–10. [Google Scholar] [CrossRef]
- Nishimura, T.; Kato, H. Taste of free amino acids and peptides. Food Rev. Int. 1988, 4, 175–194. [Google Scholar] [CrossRef]
- Kirimura, J.; Shimizu, A.; Kimizuka, A.; Ninomiya, T.; Katsuya, N. Contribution of peptides and amino acids to the taste of foods. J. Agric. Food Chem. 1969, 17, 689–695. [Google Scholar] [CrossRef]
- Park, S.; Arasu, M.V.; Lee, M.-K.; Chun, J.-H.; Seo, J.M.; Lee, S.-W.; Al-Dhabi, N.A.; Kim, S.-J. Quantification of glucosinolates, anthocyanins, free amino acids, and vitamin C in inbred lines of cabbage (Brassica oleracea L.). Food Chem. 2014, 145, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Zabaras, D.; Roohani, M.; Krishnamurthy, R.; Cochet, M.; Delahunty, C.M. Characterisation of taste-active extracts from raw Brassica oleracea vegetables. Food Funct. 2013, 4, 592–601. [Google Scholar] [CrossRef] [PubMed]
- Engel, E.; Baty, C.; le Corre, D.; Souchon, I.; Martin, N. Flavor-Active Compounds Potentially Implicated in Cooked Cauliflower Acceptance. J. Agric. Food Chem. 2002, 50, 6459–6467. [Google Scholar] [CrossRef]
- Jones, R.B.; Faragher, J.D.; Winkler, S. A review of the influence of postharvest treatments on quality and glucosinolate content in broccoli (Brassica oleracea var. italica) heads. Postharvest Biol. Technol. 2006, 41, 1–8. [Google Scholar] [CrossRef]
- Levine, L.H.; Bisbee, P.A.; Richards, J.T.; Birmele, M.N.; Prior, R.L.; Perchonok, M.; Dixon, M.; Yorio, N.C.; Stutte, G.W.; Wheeler, R.M. Quality characteristics of the radish grown under reduced atmospheric pressure. Adv. Space Res. 2008, 41, 754–762. [Google Scholar] [CrossRef]
- Hara, M.; Torazawa, D.; Asai, T.; Takahashi, I. Variations in the soluble sugar and organic acid contents in radish (Raphanus sativus L.) cultivars. Int. J. Food Sci. Technol. 2011, 46, 2387–2392. [Google Scholar] [CrossRef] [Green Version]
- Seo, M.S.; Chung, J.H.; Park, B.S.; Kim, J.S. Analysis of Sugars Content by Genotypes in 82 Radish (Raphanus Sativus L.). Korean J. Plant Resour. 2018, 31, 453–465. [Google Scholar]
- Konsula, Z.; Liakopoulou-Kyriakides, M. Hydrolysis of starches by the action of an α-amylase from Bacillus subtilis. Process. Biochem. 2004, 39, 1745–1749. [Google Scholar] [CrossRef]
- Jahan, M.S.; Pervin, M.S.; Rana, A.M.; Shovon, M.S.; Sharma, S.D.; Karim, M.R.; Rahman, M.H. Correlation between Β-Amylase Activity and Starch Content in Different Cultivars of Radish (Raphanus sativus L.). BioTechnology Indian J. 2014, 9, 298–302. [Google Scholar]
- Smith, A.M.; Zeeman, S.C.; Smith, S.M. Starch Degradation. Annu. Rev. Plant Biol. 2005, 56, 73–98. [Google Scholar] [CrossRef] [PubMed]
- Derkx, A.P.; Mares, D.J. Late-maturity α-amylase expression in wheat is influenced by genotype, temperature and stage of grain development. Planta 2020, 251, 51. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, C.M. Sucrose Accumulation in Sugar Beet Under Drought Stress. J. Agron. Crop Sci. 2010, 196, 243–252. [Google Scholar] [CrossRef]
- Sharma, A.; Shahzad, B.; Kumar, V.; Kohli, S.K.; Sidhu, G.P.S.; Bali, A.S.; Handa, N.; Kapoor, D.; Bhardwaj, R.; Zheng, B. Phytohormones Regulate Accumulation of Osmolytes Under Abiotic Stress. Biomology 2019, 9, 285. [Google Scholar] [CrossRef] [Green Version]
- Wieczorek, M.N.; Dunkel, A.; Szwengiel, A.; Czaczyk, K.; Drożdżyńska, A.; Wojtasiak, R.Z.; Jeleń, H.H. The relation between phytochemical composition and sensory traits of selected Brassica vegetables. LWT 2021, 156, 113028. [Google Scholar] [CrossRef]
- Colonna, W.J.; Samaraweera, U.; Clarke, M.A.; Cleary, M.; Godshall, M.A.; White, J.S. Sugar; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar]
- Kosemura, S.; Yamamura, S.; Hasegawa, K. Chemical studies on 4-methylthio-3-butenyl isothiocyanate from roots of japanese radish(Raphanus sativus L.) in connection with raphanusanins, phototropism-regulating substances of radish hypocotyls. Tetrahedron Lett. 1993, 34, 481–484. [Google Scholar] [CrossRef]
- Kim, M.R.; Rhee, H.S. Volatile Sulfur Components from Fresh Radishes of Korean Origin. Korean J. Food Cook. Sci. 1985, 1, 33–39. [Google Scholar]
- Kliebenstein, D.J.; Kroymann, J.; Brown, P.; Figuth, A.; Pedersen, D.; Gershenzon, J.; Mitchell-Olds, T. Genetic Control of Natural Variation in Arabidopsis Glucosinolate Accumulation. Plant Physiol. 2001, 126, 811–825. [Google Scholar] [CrossRef] [Green Version]
- Ku, K.M.; Jeffery, E.H.; Juvik, J.A. Optimization of methyl jasmonate application to broccoli florets to enhance health-promoting phytochemical content. J. Sci. Food Agric. 2014, 94, 2090–2096. [Google Scholar] [CrossRef]
- Chakraborty, I.; Bodurtha, K.J.; Heeder, N.J.; Godfrin, M.P.; Tripathi, A.; Hurt, R.H.; Shukla, A.; Bose, A. Massive Electrical Conductivity Enhancement of Multilayer Graphene/Polystyrene Composites Using a Nonconductive Filler. ACS Appl. Mater. Interfaces 2014, 6, 16472–16475. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Lee, M.Y.; Shon, J.C.; Kwon, Y.S.; Liu, K.-H.; Lee, C.H.; Ku, K.-M. Untargeted and targeted metabolomics analyses of blackberries—Understanding postharvest red drupelet disorder. Food Chem. 2019, 300, 125169. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chae, S.-H.; Lee, O.N.; Park, H.Y.; Ku, K.-M. Seasonal Effects of Glucosinolate and Sugar Content Determine the Pungency of Small-Type (Altari) Radishes (Raphanus sativus L.). Plants 2022, 11, 312. https://doi.org/10.3390/plants11030312
Chae S-H, Lee ON, Park HY, Ku K-M. Seasonal Effects of Glucosinolate and Sugar Content Determine the Pungency of Small-Type (Altari) Radishes (Raphanus sativus L.). Plants. 2022; 11(3):312. https://doi.org/10.3390/plants11030312
Chicago/Turabian StyleChae, Seung-Hun, O New Lee, Han Yong Park, and Kang-Mo Ku. 2022. "Seasonal Effects of Glucosinolate and Sugar Content Determine the Pungency of Small-Type (Altari) Radishes (Raphanus sativus L.)" Plants 11, no. 3: 312. https://doi.org/10.3390/plants11030312
APA StyleChae, S.-H., Lee, O. N., Park, H. Y., & Ku, K.-M. (2022). Seasonal Effects of Glucosinolate and Sugar Content Determine the Pungency of Small-Type (Altari) Radishes (Raphanus sativus L.). Plants, 11(3), 312. https://doi.org/10.3390/plants11030312






