Antibacterial Potential of Extracts and Phytoconstituents Isolated from Syncarpia hillii Leaves In Vitro
Abstract
:1. Introduction
2. Results
2.1. Antibacterial Effects of Primary Methanolic Extracts
2.2. Minimum Inhibitory Concentration (MIC) of Primary Extract
2.3. Minimum Bactericidal Concentration (MBC) of Primary Extracts
2.4. Biofilm Eradication Activity of S. hillii Extracts
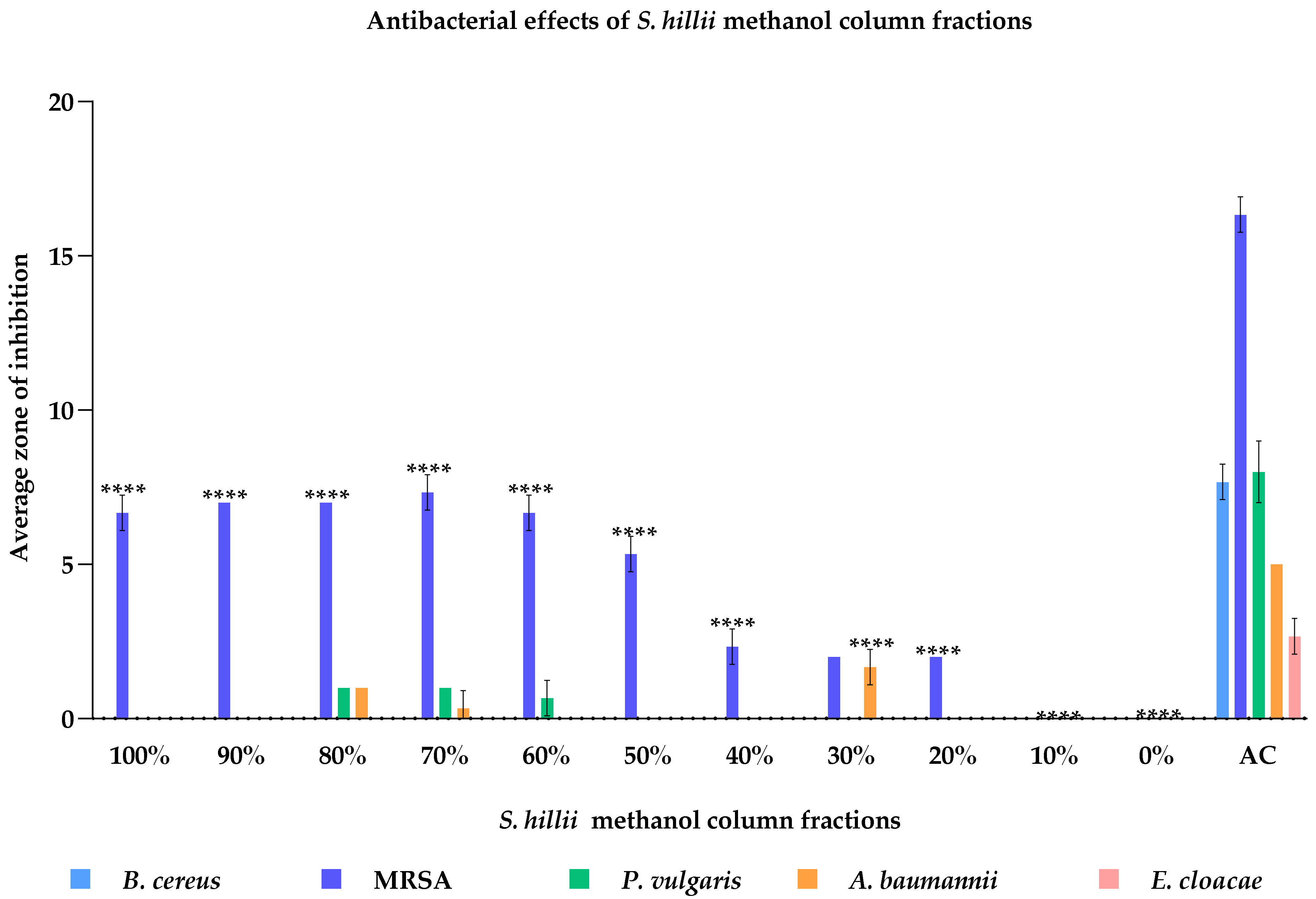
2.5. Antimicrobial Activity of Primary Column Fractions of S. hillii-Derived Methanolic Extract
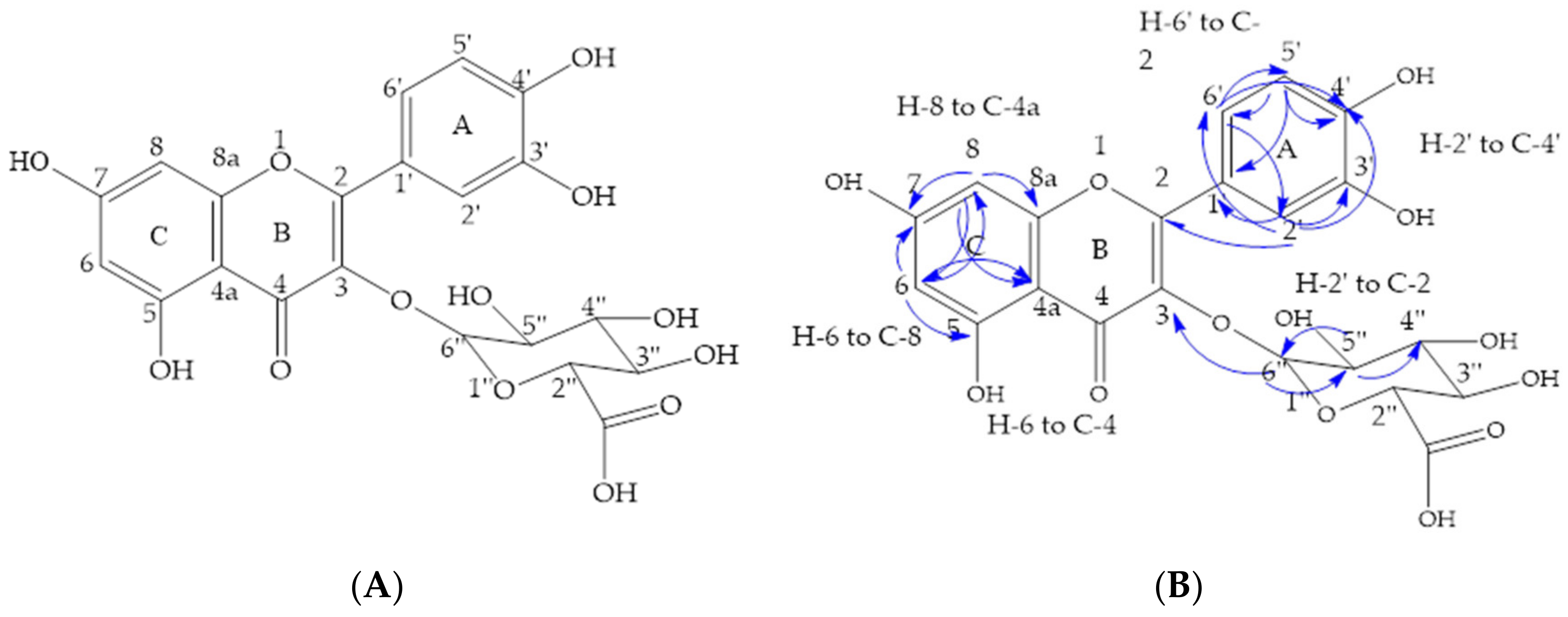
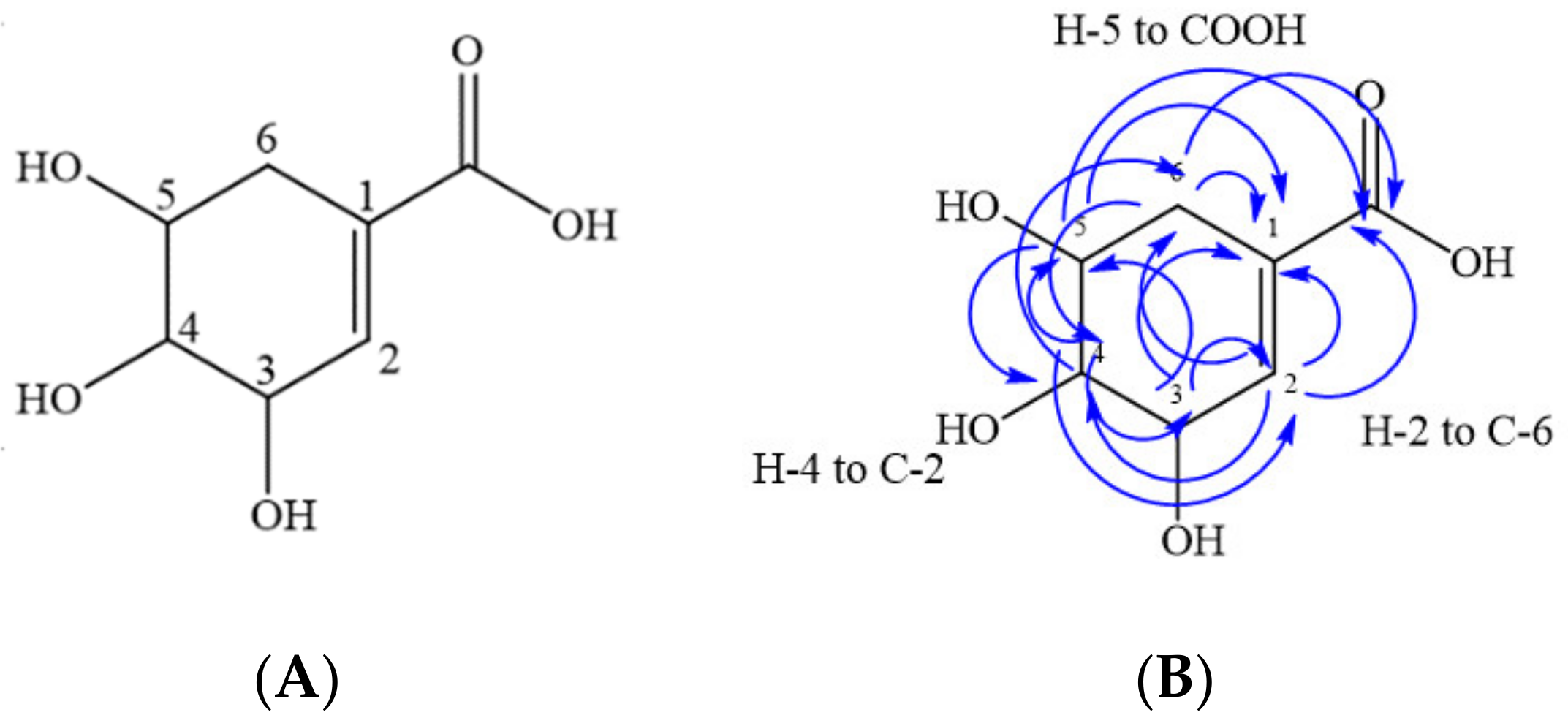
2.6. Isolation of Compounds from Primary Methanolic Fractions
2.7. Antimicrobial Activity of Isolated Compounds from the S. hillii Methanolic Extract
2.8. Predicted ADME Properties of the Isolated Compounds
3. Discussion
4. Materials and Methods
4.1. Collection and Preparation of the Primary Leaf Extracts
4.2. Bacterial Cultures
4.3. Well Diffusion Assay (WDA)
4.4. Minimum Inhibitory Concentration (MIC)
4.5. Minimum Bactericidal Concentration (MBC)
4.6. Direct Enumeration Method for Biofilm Eradication
4.7. Antimicrobial Activity of S. hillii Methanol Extract Column Fractions
4.8. Isolation of Bioactive Compounds from S. hillii Leaf Extracts
4.9. Antimicrobial Activity of the Isolated Compounds
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Attia, R.; Messaoud, C.; Arraki, K.; Zedet, A.; Demougeot, C.; Boussaïd, M.; Girard, C. Phytochemical screening and arginase inhibitory activity of extracts from several Tunisian medicinal plants. S. Afr. J. Bot. 2018, 120, 313–318. [Google Scholar] [CrossRef]
- Cos, P.; Vlietinck, A.J.; Berghe, D.V.; Maes, L. Anti-infective potential of natural products: How to develop a stronger in vitro ‘proof-of-concept’. J. Ethnopharmacol. 2006, 106, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Chaves, J.O.; de Souza, M.C.; da Silva, L.C.; Lachos-Perez, D.; Torres-Mayanga, P.C.; Machado, A.P.d.F.; Forster-Carneiro, T.; Vázquez-Espinosa, M.; González-de-Peredo, A.V.; Barbero, G.F.; et al. Extraction of Flavonoids From Natural Sources Using Modern Techniques. Front. Chem. 2020, 8, 507887. [Google Scholar] [CrossRef] [PubMed]
- Oliver, S.J.J. The role of traditional medicine practice in primary health care within Aboriginal Australia: A review of the literature. J. Ethnobiol. Ethnomed. 2013, 9, 46. [Google Scholar] [CrossRef] [Green Version]
- Akter, K.; Barnes, E.C.; Brophy, J.J.; Harrington, D.; Community Elders, Y.; Vemulpad, S.R.; Jamie, J.F. Phytochemical Profile and Antibacterial and Antioxidant Activities of Medicinal Plants Used by Aboriginal People of New South Wales, Australia. J. Evid.-Based Complementary Altern. Med. 2016, 2016, 14. [Google Scholar] [CrossRef] [Green Version]
- Abe, R.; Ohtani, K. An ethnobotanical study of medicinal plants and traditional therapies on Batan Island, the Philippines. J. Ethnopharmacol. 2013, 145, 554–565. [Google Scholar] [CrossRef]
- Brophy, J.J.; Craig, D.C.; Goldsack, R.J.; Fookes, C.J.R. Hilly acetate, a keto-acetate from the leaf steam volatiles of Syncarpia hillii. Phyfochenustry 1994, 37, 1645–1647. [Google Scholar]
- Southwell, I.A.; Brophy, J. Essential Oil Isolates from the Australian Flora. Part 3. J. Essent. Oil Res. 2000, 12, 267–278. [Google Scholar] [CrossRef]
- Brophy, J.J.; Goldsack, R.J.; Bean, A.R.; Forster, P.I.; Fookes, C.J.R. The Leaf Essential Oils of the Genus Syncarpia Ten. (Myrtaceae). Flavour Fragr. J. 1996, 11, 361–366. [Google Scholar] [CrossRef]
- Setzer, M.C.; Schmidt, J.M.; Irvine, A.K.; Jackes, B.; Setzer, W. Biological activity of rainforest plant extracts from Far North Queensland, Australia. Aust. J. Med. Herbal. 2006, 18, 6–20. [Google Scholar]
- Bayramov, D.; Li, Z.; Patel, E.; Izadjoo, M.; Kim, H.; Neff, J. A Novel Peptide-Based Antimicrobial Wound Treatment is Effective Against Biofilms of Multi-Drug Resistant Wound Pathogens. Mil. Med. 2018, 183, 481–486. [Google Scholar] [CrossRef] [Green Version]
- Ayaz, M.; Junaid, M.; Ullah, F.; Sadiq, A.; Ovais, M.; Ahmad, W.; Ahmad, S.; Zeb, A. Chemical profiling, antimicrobial and insecticidal evaluations of Polygonum hydropiper L. BMC Complement. Altern. Med. 2016, 16, 502. [Google Scholar] [CrossRef] [Green Version]
- Ayaz, M.; Subhan, F.; Sadiq, A.; Ullah, F.; Ahmed, J.; Sewell, R.D.E. Cellular efflux transporters and the potential role of natural products in combating efflux mediated drug resistance. Front. Biosci. Landmark 2017, 22, 732–756. [Google Scholar] [CrossRef]
- Sohn, U.-D.; Whang, W.-K.; Ham, I.-H.; Min, Y.-S.; Bae, K.-L.; Yim, S.-H.; Park, S.-Y. Process for Preparing Quercetin-3-O-β-D-Glucuronide(qgc) Isolated from Rumex Aquaticus. WO/03/099307 A1, 4 December 2003. [Google Scholar]
- Zeng, W.-C.; Zhang, Z.; Gao, H.; Jia, L.-R.; He, Q. Chemical Composition, Antioxidant, and Antimicrobial Activities of Essential Oil from Pine Needle (Cedrus deodara). J. Food Sci. 2012, 77, C824–C829. [Google Scholar] [CrossRef]
- Frost, J.W.; Frost, K.M.; Knop, D.R. Biocatalytic Synthesis of Shikimc Acid; United States Patent and Trademark Office, Ed.; Board of Trustees operating Michigan State University: East Lansing, MI, USA, 2003. [Google Scholar]
- Yang, L.; Wen, K.-S.; Ruan, X.; Zhao, Y.-X.; Wei, F.; Wang, Q. Response of Plant Secondary Metabolites to Environmental Factors. Molecules 2018, 23, 762. [Google Scholar] [CrossRef] [Green Version]
- Steigbigel, R.T.; Steigbigel, N.H. Static vs Cidal Antibiotics. Clin. Infect. Dis. 2018, 68, 351–352. [Google Scholar] [CrossRef]
- Friedel, J.K.; Mölter, K.; Fischer, W.R.J.B.; Soils, F.o. Comparison and improvement of methods for determining soil dehydrogenase activity by using triphenyltetrazolium chloride and iodonitrotetrazolium chloride. Biol. Fertil. Soils 1994, 18, 291–296. [Google Scholar] [CrossRef]
- Alvarado-Gomez, E.; Perez-Diaz, M.; Valdez-Perez, D.; Ruiz-Garcia, J.; Magana-Aquino, M.; Martinez-Castanon, G.; Martinez-Gutierrez, F. Adhesion forces of biofilms developed in vitro from clinical strains of skin wounds. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 82, 336–344. [Google Scholar] [CrossRef]
- Mihai, M.M.; Preda, M.; Lungu, I.; Gestal, M.C.; Popa, M.I.; Holban, A.M. Nanocoatings for Chronic Wound Repair-Modulation of Microbial Colonization and Biofilm Formation. Int. J. Mol. Sci. 2018, 19, 1179. [Google Scholar] [CrossRef] [Green Version]
- Stathopoulou, M.K.; Banti, C.N.; Kourkoumelis, N.; Hatzidimitriou, A.G.; Kalampounias, A.G.; Hadjikakou, S.K. Silver complex of salicylic acid and its hydrogel-cream in wound healing chemotherapy. J. Inorg. Biochem. 2018, 181, 41–55. [Google Scholar] [CrossRef]
- Shetty, B.S.; Udupa, S.L.; Udupa, A.L.; Somayaji, S.N. Effect of Centella asiatica L (Umbelliferae) on normal and dexamethasone-suppressed wound healing in Wistar Albino rats. Int. J. Low. Extrem. Wounds 2006, 5, 137–143. [Google Scholar] [CrossRef]
- Pfalzgraff, A.; Brandenburg, K.; Weindl, G. Antimicrobial Peptides and Their Therapeutic Potential for Bacterial Skin Infections and Wounds. Front. Pharmacol. 2018, 9, 281. [Google Scholar] [CrossRef]
- Li, H.-W.; Xiang, Y.-Z.; Zhang, M.; Jiang, Y.-H.; Zhang, Y.; Liu, Y.-Y.; Lin, L.-B.; Zhang, Q.-L. A novel bacteriocin from Lactobacillus salivarius against Staphylococcus aureus: Isolation, purification, identification, antibacterial and antibiofilm activity. LWT 2021, 140, 110826. [Google Scholar] [CrossRef]
- Narayanan, K.B.; Park, G.T.; Han, S.S. Biocompatible, antibacterial, polymeric hydrogels active against multidrug-resistant Staphylococcus aureus strains for food packaging applications. Food Control 2021, 123, 107695. [Google Scholar] [CrossRef]
- Jaśkiewicz, M.; Janczura, A.; Nowicka, J.; Kamysz, W. Methods Used for the Eradication of Staphylococcal Biofilms. Antibiotiocs 2019, 8, 174. [Google Scholar] [CrossRef] [Green Version]
- Abdar, M.H.; Taheri-Kalani, M.; Taheri, K.; Emadi, B.; Hasanzadeh, A.; Sedighi, A.; Pirouzi, S.; Sedighi, M. Prevalence of extended-spectrum beta-lactamase genes in Acinetobacter baumannii strains isolated from nosocomial infections in Tehran, Iran. GMS Hyg Infect. Control 2019, 14, Doc02. [Google Scholar] [CrossRef]
- Barrientos, S.; Stojadinovic, O.; Golinko, M.S.; Brem, H.; Tomic-Canic, M. Growth factors and cytokines in wound healing. Wound Repair Regen. 2008, 16, 585–601. [Google Scholar] [CrossRef]
- da Costa Júnior, S.D.; de Oliveira Santos, J.V.; de Almeida Campos, L.A.; Pereira, M.A.; Magalhães, N.S.S.; Cavalcanti, I.M.F. Antibacterial and antibiofilm activities of quercetin against clinical isolates of Staphyloccocus aureus and Staphylococcus saprophyticus with resistance profile. Int. J. Environ. Agric. Biotechnol. 2018, 3, 266213. [Google Scholar] [CrossRef] [Green Version]
- Amin, M.U.; Khurram, M.; Khattak, B.; Khan, J. Antibiotic additive and synergistic action of rutin, morin and quercetin against methicillin resistant Staphylococcus aureus. BMC complementary Altern. Med. 2015, 15, 59. [Google Scholar] [CrossRef] [Green Version]
- Bai, J.; Wu, Y.; Liu, X.; Zhong, K.; Huang, Y.; Gao, H. Antibacterial Activity of Shikimic Acid from Pine Needles of Cedrus deodara against Staphylococcus aureus through Damage to Cell Membrane. Int. J. Mol. Sci. 2015, 16, 27145–27155. [Google Scholar] [CrossRef] [Green Version]
- Higuita, N.I.A.; Huycke, M.M. Enterococcal Disease, Epidemiology, and Implications for Treatment. In Enterococci: From Commensals to Leading Causes of Drug Resistant Infection; Gilmore, M.S., Clewell, D.B., Ike, Y., Shankar, N., Eds.; Massachusetts Eye and Ear Infirmary: Boston, MA, USA, 2014. [Google Scholar]
- Anderson, A.C.; Jonas, D.; Huber, I.; Karygianni, L.; Wölber, J.; Hellwig, E.; Arweiler, N.; Vach, K.; Wittmer, A.; Al-Ahmad, A. Enterococcus faecalis from Food, Clinical Specimens, and Oral Sites: Prevalence of Virulence Factors in Association with Biofilm Formation. Front Microbiol. 2016, 6, 1534. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Esmail, M.A.M.; Abdulghany, H.M.; Khairy, R.M. Prevalence of Multidrug-Resistant Enterococcus faecalis in Hospital-Acquired Surgical Wound Infections and Bacteremia: Concomitant Analysis of Antimicrobial Resistance Genes. Infect. Dis. Res. Treat. 2019, 12, 1178633719882929. [Google Scholar] [CrossRef] [PubMed]
- Merritt, J.H.; Kadouri, D.E.; O’Toole, G.A. Growing and analyzing static biofilms. Curr. Protoc. Microbiol. 2005, 1, 1B.1.1–1B.1.18. [Google Scholar] [CrossRef] [Green Version]

| Bacterial Strain | Antibiotic Standard ZOI (mm) | Average ZOI (mm) for S. hillii Extracts (100 mg/mL) | ||||
|---|---|---|---|---|---|---|
| Aqueous | Methanol | Ethanol | Isopropanol | Hexane | ||
| Gram-positive bacteria | ||||||
| Bacillus cereus (ATCC 14579) | 7.00 ± 0.00 | 3.56 ± 0.50 | 5.00 ± 0.00 | 5.28 ± 0.42 | 3.22 ± 0.63 | 1.56 ± 0.68 |
| MSSA (NCTC 6571) | 11.33 ± 1.89 | 4.44 ± 0.50 | 5.89 ± 0.54 | 6.00 ± 0.82 | 4.22 ± 0.63 | 3.89 ± 0.74 |
| MRSA (QUT 1113) | 8.33 ± 0.24 | 6.44 ± 0.50 | 7.33 ± 0.45 | 6.67 ± 0.47 | 5.78 ± 0.92 | 3.56 ± 0.50 |
| MRSA (ATCC 33591) | 7.00 ± 0.00 | 6.56 ± 0.83 | 6.33 ± 0.45 | 5.89 ± 0.57 | 5.78 ± 0.92 | 2.89 ± 0.87 |
| Bacillus subtilis (QUT 0535) | 9.00 ± 0.00 | NA | 4.33 ± 0.45 | 4.00 ± 0.00 | 1.89 ± 0.57 | 1.67 ± 0.47 |
| Staphylococcus epidermidis (QUT 0613) | 9.00 ± 0.00 | 3.67 ± 0.47 | 5.67 ± 0.45 | 5.17 ± 0.33 | 2.67 ± 0.67 | 0.56 ± 0.50 |
| Enterococcus faecalis (QUT code 1105) | 3.00 ± 0.00 | NA | 2.33 ± 0.45 | 2.33 ± 0.47 | NA | 1.89 ± 0.31 |
| Enterococcus faecium (QUT code 1101) | 6.00 ± 0.00 | NA | 2.33 ± 0.45 | 2.78 ± 0.42 | 2.00 ± 0.82 | 2.33 ± 0.47 |
| Enterococcus gallinarum (ATCC 49573) | 4.00 ± 0.00 | 0.89 ± 0.74 | 1.33 ± 0.45 | 2.67 ± 0.67 | NA | 2.67 ± 0.47 |
| Enterococcus casseliflavus (ATCC 25788) | 4.00 ± 0.00 | NA | 2.00 ± 0.00 | 2.00 ± 0.67 | NA | 0.67 ± 0.47 |
| Staphylococcus saprophyticus (QUT 0703) | 8.00 ± 0.00 | 4.22 ± 0.42 | 5.00 ± 0.00 | 4.78 ± 0.42 | 3.78 ± 0.42 | 2.22 ± 0.42 |
| Gram-negative bacteria | ||||||
| Klebsiella pneumoniae (ATCC 27736) | 3.00 ± 0.00 | NA | NA | NA | NA | NA |
| Pseudomonas aeruginosa (ATCC 27853) | 5.00 ± 0.00 | NA | NA | NA | NA | NA |
| Escherichia coli (ATCC 25922) | 6.00 ± 0.00 | NA | NA | NA | NA | NA |
| Proteus vulgaris (ATCC 6380) | 6.33 ± 0.47 | NA | 1.67 ± 0.45 | 1.44 ± 0.50 | 0.11 ± 0.31 | NA |
| Proteus mirabilis (ATCC 7002) | 6.00 ± 0.00 | NA | 2.00 ± 0.00 | 1.78 ± 0.42 | NA | NA |
| Acinetobacter baumannii (ATCC 19606) | 4.00 ± 0.00 | NA | 1.00 ± 0.00 | 0.89 ± 0.57 | NA | NA |
| Enterobacter aerogenes (ATCC 13048) | 3.00 ± 0.00 | NA | NA | NA | NA | NA |
| Enterobacter cloacae (ATCC 13047) | 3.00 ± 0.00 | NA | 0.44 ± 0.47 | 0.22 ±0.42 | NA | NA |
| Bacterial Strain | Antibiotic Standard (µg/mL) | S. hillii Extracts (mg/mL) | ||||
|---|---|---|---|---|---|---|
| Methanol | Ethanol | Aqueous | Isopropanol | Hexane | ||
| Gram-positive bacteria | ||||||
| Bacillus cereus (ATCC 14579) | 5.00 | 1.25 | 2.50 | 10.00 | 5.00 | >10.00 |
| MSSA (NCTC 6571) | 5.00 * | 1.25 | 1.25 | 5.00 | 5.00 | >10.00 |
| MRSA (QUT 1113) | 5.00 * | 0.63 | 1.25 | 2.50 | 2.50 | >10.00 |
| MRSA (ATCC 33591) | 5.00 * | 0.63 | 0.63 | 2.50 | 2.50 | >10.00 |
| Bacillus subtilis (QUT 0535) | 2.50 | 5.00 | 5.00 | ND | 10.00 | >10.00 |
| Staphylococcus epidermidis (QUT 0613) | 4.00 * | 0.63 | 0.63 | 2.50 | 2.50 | ND |
| Staphylococcus saprophyticus (QUT 0703) | 30.00 * | 1.25 | 1.25 | 5.00 | ND | >10.00 |
| Gram-negative bacteria | ||||||
| Proteus vulgaris (ATCC 6380) | 5.00 | 1.25 | 1.25 | ND | ND | ND |
| Proteus mirabilis (ATCC 7002) | 5.00 | 1.25 | 2.50 | ND | ND | ND |
| Acinetobacter baumannii (ATCC 19606) | 10.00 * | 2.50 | 2.50 | ND | ND | ND |
| Enterobacter cloacae (ATCC 13047) | 20.00 * | 1.25 | 1.25 | ND | ND | ND |
| Bacterial Strain | Tested Concentration of the Standard Antibiotic (mg/mL) | MBC of S. hillii Extracts (mg/mL) | |
|---|---|---|---|
| Methanol | Ethanol | ||
| Gram-positive bacteria | |||
| Bacillus cereus (ATCC 14579) | 1.0 | 15.0 | 20.0 |
| MSSA (NCTC 6571) | 1.0 | 5.0 | 5.0 |
| MRSA (ATCC 33591) | 1.0 | 7.5 | 7.5 |
| MRSA (QUT 1113) | 1.0 | 7.5 | 5.0 |
| Bacillus subtilis (QUT 0535) | 1.0 | 15.0 | 15.0 |
| Staphylococcus epidermidis (QUT 0613) | 1.0 | 5.0 | 5.0 |
| Enterococcus faecalis (QUT 1105) | 1.0 | >20.0 | >20.0 |
| Enterococcus faecium (QUT 1101) | 1.0 | >20.0 | >20.0 |
| Enterococcus gallinarum (ATCC 13048) | 1.0 | >20.0 | >20.0 |
| Enterococcus casseliflavus (ATCC 25788) | 1.0 | >20.0 | >20.0 |
| Staphylococcus saprophyticus (QUT 0703) | 1.0 | 5.0 | 5.0 |
| Gram-negative Bacteria | |||
| Proteus vulgaris (ATCC 7002) | 1.0 | >20.0 | >20.0 |
| Proteus mirabilis (ATCC 6380) | 1.0 | >20.0 | >20.0 |
| Acinetobacter baumannii (ATCC 19606) | 1.0 | >20.0 | >20.0 |
| Enterobacter cloacae (ATCC 13047) | 1.0 | >20.0 | >20.0 |
| Carbon Number | 1H | 13C | HMBC (13C) |
|---|---|---|---|
| 1 | |||
| 2 | 157.6 | ||
| 3 | 134.0 | ||
| 4 | 177.8 | ||
| 4a | 104.2 | ||
| 5 | 161.6 | ||
| 6 | H (d) 6.10 | 98.5 | C-5, C-7, C-8 and C-4a |
| 7 | 164.6 | ||
| 8 | H (d) 6.29 | 93.3 | C-6, C-4a, C-8a, and C-7 |
| 8a | 157.0 | ||
| 1′ | 121.4 | ||
| 2′ | H; (s) 7.58 | 115.9 | C-2, C-1′, C-6′, C-3′ and C-4′ |
| 3′ | 144.5 | ||
| 4′ | 148.5 | ||
| 5′ | H; (d) 6.74 | 114.6 | C-1′, C-6′, C-4′ and C-3′ |
| 6′ | H; (d) 7.50 | 121.9 | C-2, C-2′, C-4′ and C-5′ |
| 1′′ | |||
| 2′′ | H; (d) 3.64 | 76.2 | |
| 3′′ | H; (t) 3.47 | 71.5 | |
| 4′′ | H; (t) 3.35 | 76.2 | |
| 5′′ | H; (t) 3.42 | 74.0 | C-4′′, and C-6′′ |
| 6′′ | H; (d) 5.23 | 102.8 | C-3 and C-5′′ |
| Carbon Number | 1H | 13C | HMBC (13C) |
|---|---|---|---|
| 1 | 129.5 | ||
| 2 | H (s) 6.58 | 138.4 | C-1, C-4, C-6 and COOH |
| 3 | H (s) 4.20 | 65.9 | C-1, C-2 and C-5 |
| 4 | H (m) 3.52–3.54 | 70.9 | C-2, C-3, C-5 and C-6 |
| 5 | H (m) 3.81–3.84 | 67.2 | C-1, C-3, C-4 and COOH |
| 6a | H (d) 2.40 | 30.6 | C-1, C-2, C-4, C-5 and COOH |
| 6b | H (d) 1.99 | 30.6 | C-1, C-2, C-4, C-5 and COOH |
| COOH | - | 168.8 | - |
| Bacterial Strain | Antibiotic Standard | Compounds (µg/mL) | ||
|---|---|---|---|---|
| Name | MIC (µg/mL) | Quercetin | Shikimic Acid | |
| Gram-positive bacteria | ||||
| Bacillus cereus (ATCC 14579) | Erythromycin | 5.0 | >200.0 | >200.0 |
| MRSA (ATCC 33591) | SXT | 5.0 * | >200.0 | >200.0 |
| Enterococcus faecalis (QUT 1105) | Teicoplanin | 30.0 * | 0.78 | 200.0 |
| Gram-negative bacteria | ||||
| Proteus vulgaris (ATCC 6380) | SXT | 5.0 | 200.0 | >200.0 |
| Acinetobacter baumannii (ATCC 19606) | Gentamicin | 10.0 * | >200.0 | >200.0 |
| Enterobacter cloacae (ATCC 13047) | Gentamicin | 20.0 * | 200.0 | 200.0 |
| Klebsiella pneumoniae (ATCC 27736) | Gentamicin | 2.0 * | >200.0 | >200.0 |
| Pseudomonas aeruginosa (ATCC 27853) | Gentamicin | 10.0 * | 200.0 | 200.0 |
| Descriptors | Values | Recommended Range |
|---|---|---|
| Molecular weight | 478.36 | 150–500 g/mol |
| Num. H-bond acceptors | 13 | 10 |
| Num. H-bond donors | 8 | 5 |
| Molar Refractivity | 110.77 | 40 to 130 |
| TPSA | 227.58 A2 | 20–130 Å2 |
| Log Po/w | 1.13 | <5 |
| GI absorption | Low | - |
| CYP1A2 inhibitor | No | - |
| CYP2C19 inhibitor | No | - |
| CYP2C9 inhibitor | No | - |
| CYP2D6 inhibitor | No | - |
| CYP3A4 inhibitor | No | - |
| Lipinski rule violation | 2 | Maximum 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perera, M.M.N.; Dighe, S.N.; Katavic, P.L.; Collet, T.A. Antibacterial Potential of Extracts and Phytoconstituents Isolated from Syncarpia hillii Leaves In Vitro. Plants 2022, 11, 283. https://doi.org/10.3390/plants11030283
Perera MMN, Dighe SN, Katavic PL, Collet TA. Antibacterial Potential of Extracts and Phytoconstituents Isolated from Syncarpia hillii Leaves In Vitro. Plants. 2022; 11(3):283. https://doi.org/10.3390/plants11030283
Chicago/Turabian StylePerera, Muthukuttige M. N., Satish N. Dighe, Peter L. Katavic, and Trudi A. Collet. 2022. "Antibacterial Potential of Extracts and Phytoconstituents Isolated from Syncarpia hillii Leaves In Vitro" Plants 11, no. 3: 283. https://doi.org/10.3390/plants11030283
APA StylePerera, M. M. N., Dighe, S. N., Katavic, P. L., & Collet, T. A. (2022). Antibacterial Potential of Extracts and Phytoconstituents Isolated from Syncarpia hillii Leaves In Vitro. Plants, 11(3), 283. https://doi.org/10.3390/plants11030283







