Vanilla beyond Vanilla planifolia and Vanilla × tahitensis: Taxonomy and Historical Notes, Reproductive Biology, and Metabolites
Abstract
1. Introduction
2. Historical and Taxonomic Notes
3. Reproductive Biology
3.1. Vanilla Flowers
3.2. Breeding System and Pollination
3.3. Pollinators and Visitors
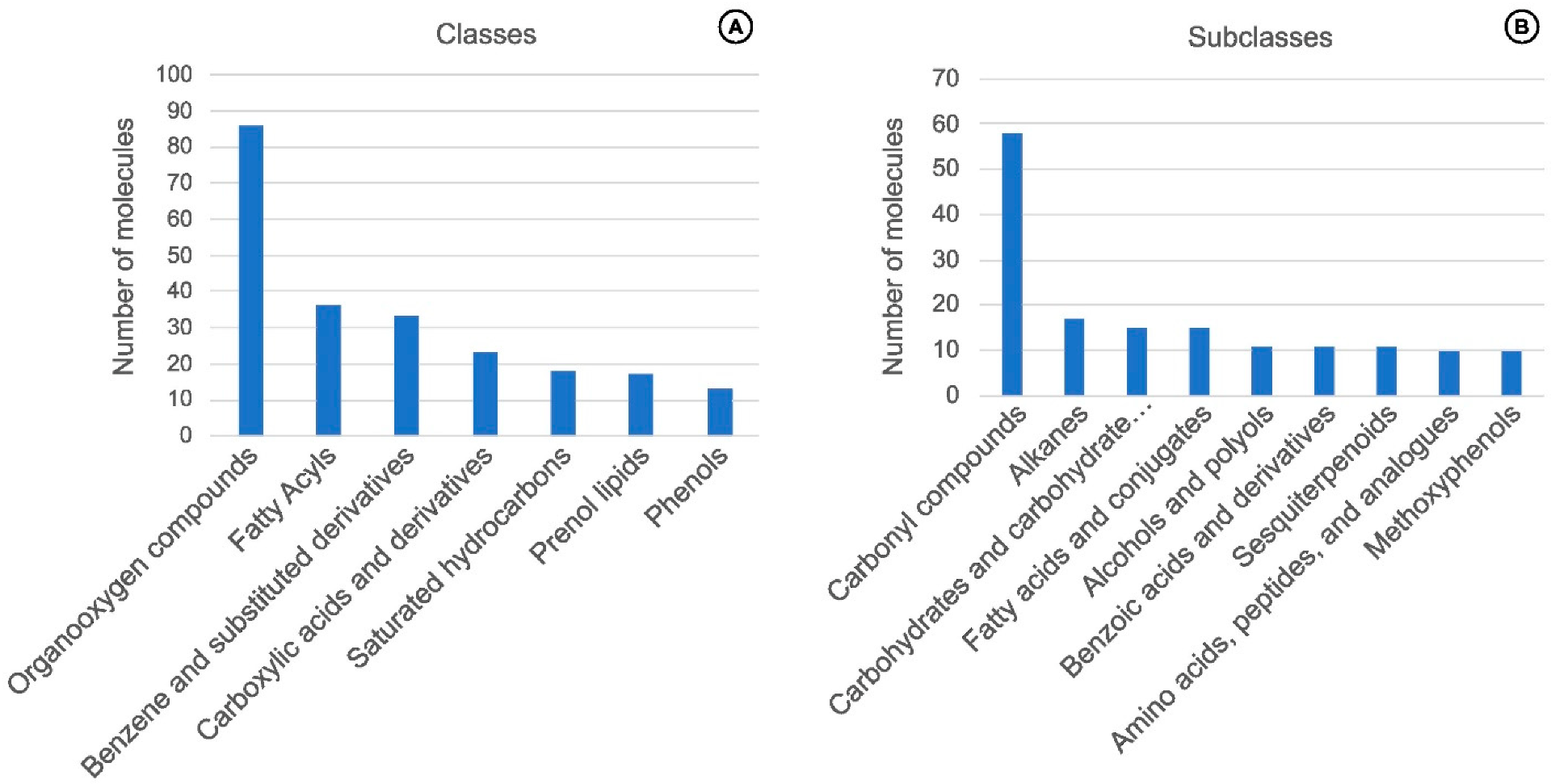
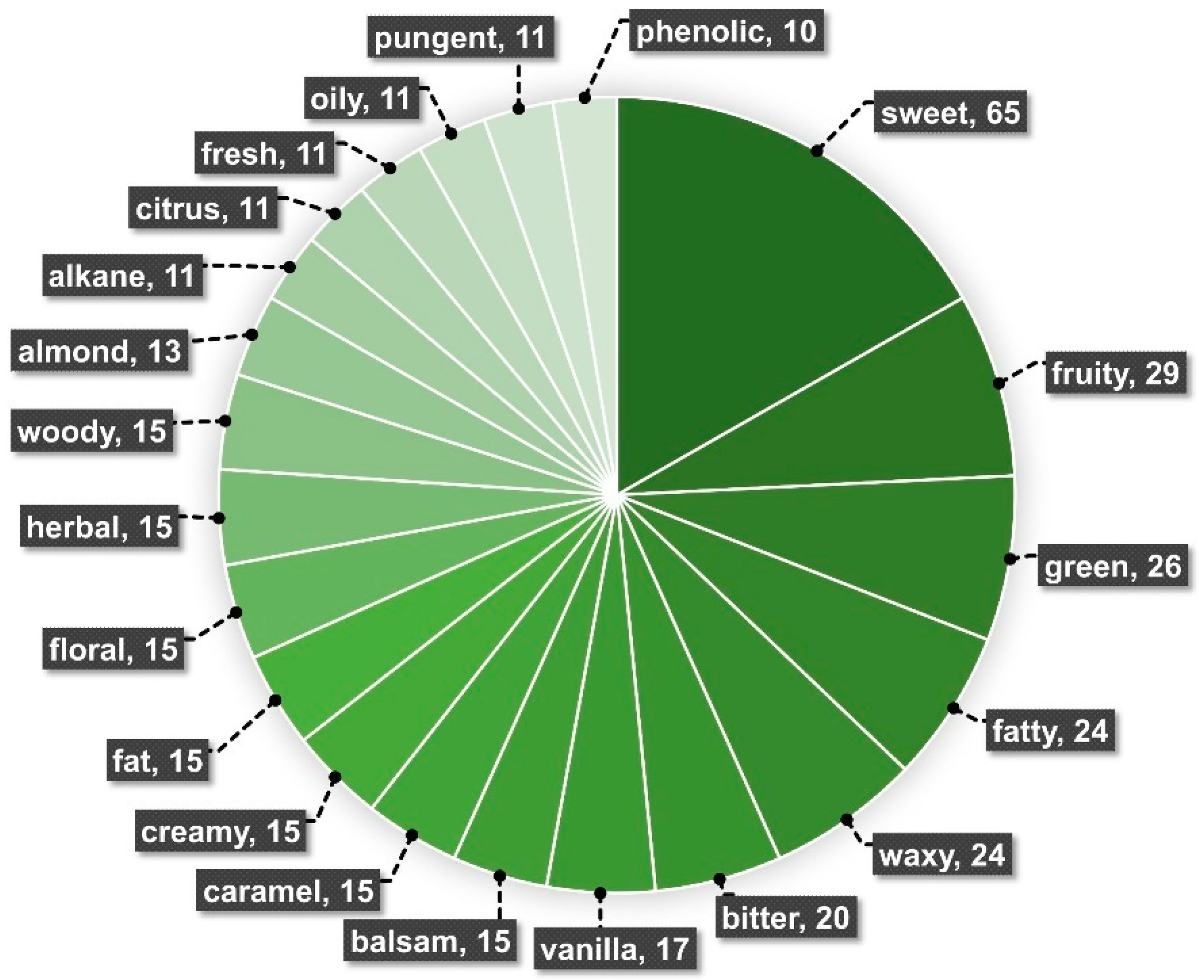
4. Chemical Traits from Vanilla Crop WRs
Molecules, Chemical Classes, and Potential Flavor Descriptors in Vanilla Wild-Relative Species
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Gallage, N.J.; Møller, B.L. Vanilla: The Most Popular Flavour. In Biotechnology of Natural Products; Schwab, W., Lange, B.M., Wüst, M., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 3–24. ISBN 978-3-319-67902-0. [Google Scholar]
- Karremans, A.P.; Chinchilla, I.F.; Rojas-Alvarado, G.; Cedeño-Fonseca, M.; Damián, A.; Léotard, G. A Reappraisal of Neotropical Vanilla. With a Note on Taxonomic Inflation and the Importance of Alpha Taxonomy in Biological Studies. Lankesteriana 2020, 20, 395–497. [Google Scholar] [CrossRef]
- Fraga, C.N.D.; Couto, D.R.; Pansarin, E.R. Two New Species of Vanilla (Orchidaceae) in the Brazilian Atlantic Forest. Phytotaxa 2017, 296, 63. [Google Scholar] [CrossRef]
- Andriamihaja, C.F.; Ramarosandratana, A.V.; Grisoni, M.; Jeannoda, V.; Besse, P. The Leafless Vanilla Species-Complex from the South-West Indian Ocean Region: A Taxonomic Puzzle and a Model for Orchid Evolution and Conservation Research. Diversity 2020, 12, 443. [Google Scholar] [CrossRef]
- Ranadive, A.S. Vanillin and Related Flavor Compounds in Vanilla Extracts Made from Beans of Various Global Origins. J. Agric. Food Chem. 1992, 40, 1922–1924. [Google Scholar] [CrossRef]
- Pérez-Silva, A.; Nicolás-García, M.; Petit, T.; Dijoux, J.B.; de los Ángeles Vivar-Vera, M.; Besse, P.; Grisoni, M. Quantification of the Aromatic Potential of Ripe Fruit of Vanilla planifolia (Orchidaceae) and Several of Its Closely and Distantly Related Species and Hybrids. Eur. Food Res. Technol. 2021, 247, 1489–1499. [Google Scholar] [CrossRef]
- Berenstein, N. Making a Global Sensation: Vanilla Flavor, Synthetic Chemistry, and the Meanings of Purity. Hist. Sci. 2016, 54, 399–424. [Google Scholar] [CrossRef]
- Januszewska, R.; Giret, E.; Clement, F.; Van Leuven, I.; Goncalves, C.; Vladislavleva, E.; Pradal, P.; Nåbo, R.; Landuyt, A.; D’Heer, G.; et al. Impact of Vanilla Origins on Sensory Characteristics of Chocolate. Food Res. Int. 2020, 137, 109313. [Google Scholar] [CrossRef]
- Khoyratty, S.; Kodja, H.; Verpoorte, R. Vanilla Flavor Production Methods: A Review. Ind. Crops Prod. 2018, 125, 433–442. [Google Scholar] [CrossRef]
- Brunschwig, C.; Collard, F.X.; Bianchini, J.-P.; Raharivelomanana, P. Evaluation of Chemical Variability of Cured Vanilla Beans (Vanilla tahitensis and Vanilla planifolia). Nat. Prod. Commun. 2009, 4, 1934578X0900401. [Google Scholar] [CrossRef]
- Pérez-Silva, A.; Odoux, E.; Brat, P.; Ribeyre, F.; Rodriguez-Jimenes, G.; Robles-Olvera, V.; García-Alvarado, M.A.; Günata, Z. GC–MS and GC–Olfactometry Analysis of Aroma Compounds in a Representative Organic Aroma Extract from Cured Vanilla (Vanilla planifolia G. Jackson) Beans. Food Chem. 2006, 99, 728–735. [Google Scholar] [CrossRef]
- Watteyn, C.; Fremout, T.; Karremans, A.P.; Huarcaya, R.P.; Azofeifa Bolaños, J.B.; Reubens, B.; Muys, B. Vanilla Distribution Modeling for Conservation and Sustainable Cultivation in a Joint Land Sparing/Sharing Concept. Ecosphere 2020, 11, e03056. [Google Scholar] [CrossRef]
- Ellestad, P.; Pérez-Farrera, M.A.; Buerki, S. Genomic Insights into Cultivated Mexican Vanilla planifolia Reveal High Levels of Heterozygosity Stemming from Hybridization. Plants 2022, 11, 2090. [Google Scholar] [CrossRef]
- Rabary, L.; Holland, H. Madagascar Vanilla Crop Quality Suffers as Thieves Spark Violence. Reuters, 18 July 2019. [Google Scholar]
- Ferreira Filho, R.L.; Barberena, F.F.V.A.; Costa, J.M. Orchidaceae in Floodplains of the Islands of Abaetetuba, Amazonian Brazil: A Flora Threatened by Intensive Management for Açaí Palm (Euterpe Oleracea). Brittonia 2021, 73, 1–24. [Google Scholar] [CrossRef]
- Grand View Research Vanillin Market Size, Share & Trends Analysis Report by End-Use (Food & Beverage, Fragrance, Pharmaceutical), by Region (North America, Europe, Asia Pacific, Central & South America, MEA), and Segment Forecasts, 2020–2025. Available online: https://www.grandviewresearch.com/industry-analysis/vanillin-market (accessed on 23 November 2022).
- Navarro, C. Mexican Producers of Vanilla Beans Face Tightening Supplies, Increased Demand. Available online: https://digitalrepository.unm.edu/sourcemex/6372 (accessed on 23 November 2022).
- Sosa, A. Meet the Long Island Lawyer Pursuing Nearly 100 Lawsuits Over Products Labeled as “Vanilla”. Available online: https://www.insideedition.com/meet-the-long-island-lawyer-pursuing-nearly-100-lawsuits-over-products-labeled-as-vanilla-63329 (accessed on 23 November 2022).
- Chattopadhyay, P.; Banerjee, G.; Sen, S.K. Cleaner Production of Vanillin through Biotransformation of Ferulic Acid Esters from Agroresidue by Streptomyces Sannanensis. J. Clean. Prod. 2018, 182, 272–279. [Google Scholar] [CrossRef]
- Flanagan, N.S.; Navia-Samboni, A.; González-Pérez, E.N.; Mendieta-Matallana, H. Distribution and Conservation of Vanilla Crop Wild Relatives: The Value of Local Community Engagement for Biodiversity Research. Neotrop. Biol. Conserv. 2022, 17, 205–227. [Google Scholar] [CrossRef]
- Flanagan, N.S.; Mosquera-Espinosa, A.T. An Integrated Strategy for the Conservation and Sustainable Use of Native Vanilla Species in Colombia. Lankesteriana 2016, 16, 201–208. [Google Scholar] [CrossRef]
- Hu, Y.; Resende, M.F.R.; Bombarely, A.; Brym, M.; Bassil, E.; Chambers, A.H. Genomics-Based Diversity Analysis of Vanilla Species Using a Vanilla planifolia Draft Genome and Genotyping-By-Sequencing. Sci. Rep. 2019, 9, 3416. [Google Scholar] [CrossRef]
- Rain, P. Vanilla: The Cultural History of the World’s Most Popular Flavor and Fragrance; Jeremy, P. Tarcher/Penguin: New York, NY, USA, 2004; ISBN 978-1-58542-363-7. [Google Scholar]
- Linares, V.; Adams, M.J.; Cradic, M.S.; Finkelstein, I.; Lipschits, O.; Martin, M.A.S.; Neumann, R.; Stockhammer, P.W.; Gadot, Y. First Evidence for Vanillin in the Old World: Its Use as Mortuary Offering in Middle Bronze Canaan. J. Archaeol. Sci. Rep. 2019, 25, 77–84. [Google Scholar] [CrossRef]
- Nielsen, L.R.; Siegismund, H.R. Interspecific Differentiation and Hybridization in Vanilla Species (Orchidaceae). Heredity 1999, 83, 560–567. [Google Scholar] [CrossRef]
- Grisoni, M.; Nany, F. The Beautiful Hills: Half a Century of Vanilla (Vanilla planifolia Jacks. Ex Andrews) Breeding in Madagascar. Genet. Resour. Crop Evol. 2021, 68, 1691–1708. [Google Scholar] [CrossRef]
- The IUCN Red List of Threatened Species. Available online: https://www.iucnredlist.org/en (accessed on 23 November 2022).
- Kevan, P.G. Plant Breeding Systems and Pollination. In The Pollination of Cultivated Plants: A Compendium for Practitioners; David Ward, R., Ed.; Food and Agriculture Organization of the United Nations: Rome, Italy, 2018; pp. 164–172. ISBN 978-92-5-130506-5. [Google Scholar]
- Lubinsky, P.; Van Dam, M.; Van Dam, A. Pollination of Vanilla and Evolution in Orchidaceae. Lindleyana 2006, 75, 926–929. [Google Scholar]
- Ramachandra Rao, S.; Ravishankar, G. Vanilla Flavour: Production by Conventional and Biotechnological Routes. J. Sci. Food Agric. 2000, 80, 289–304. [Google Scholar] [CrossRef]
- Do Nascimento, T.A.; da Furtado, M.S.C.; Pereira, W.C.; Barberena, F.F.V.A. Vanilla Bahiana Hoehne (Orchidaceae): Studies on Fruit Development and New Perspectives into Crop Improvement for the Vanilla planifolia Group. Biota Neotrop. 2019, 19, e20180696. [Google Scholar] [CrossRef]
- Pansarin, E.R. Vanilla Flowers: Much More than Food-Deception. Bot. J. Linn. Soc. 2022, 198, 57–73. [Google Scholar] [CrossRef]
- Van Dam, A.R.; Householder, J.E.; Lubinsky, P. Vanilla Bicolor Lindl. (Orchidaceae) from the Peruvian Amazon: Auto-Fertilization in Vanilla and Notes on Floral Phenology. Genet. Resour. Crop Evol. 2010, 57, 473–480. [Google Scholar] [CrossRef]
- Reis, C.A.M.; Brondani, G.E.; de Almeida, M. Biologia floral, reprodutiva e propagação vegetativa de baunilha. Sci. Agrar. Parana. 2011, 10, 69. [Google Scholar] [CrossRef]
- Gigant, R.L.; De Bruyn, A.; Church, B.; Humeau, L.; Gauvin-Bialecki, A.; Pailler, T.; Grisoni, M.; Besse, P. Active Sexual Reproduction but No Sign of Genetic Diversity in Range-Edge Populations of Vanilla Roscheri Rchb. f. (Orchidaceae) in South Africa. Conserv. Genet. 2014, 15, 1403–1415. [Google Scholar] [CrossRef]
- Pansarin, E.R.; Pansarin, L.M. Floral Biology of Two Vanilloideae (Orchidaceae) Primarily Adapted to Pollination by Euglossine Bees. Plant Biol. 2014, 16, 1104–1113. [Google Scholar] [CrossRef]
- Petersson, L. Pollination Biology of the Endemic Orchid Vanilla Bosseri in Madagascar. Master’s Thesis, Uppsala University, Uppsala, Sweden, 2015. [Google Scholar]
- Gigant, R.L.; Rakotomanga, N.; Goulié, C.; Da Silva, D.; Barre, N.; Citadelle, G.; Silvestre, D.; Grisoni, M.; Besse, P. Microsatellite Markers Confirm Self-Pollination and Autogamy in Wild Populations of Vanilla Mexicana Mill. (Syn. V. Inodora) (Orchidaceae) in the Island of Guadeloupe. In Microsatellite Markers; Abdurakhmonov, I.Y., Ed.; InTech: London, UK, 2016; ISBN 978-953-51-2797-0. [Google Scholar]
- Gigant, R.L.; De Bruyn, A.; M’sa, T.; Viscardi, G.; Gigord, L.; Gauvin-Bialecki, A.; Pailler, T.; Humeau, L.; Grisoni, M.; Besse, P. Combining Pollination Ecology and Fine-Scale Spatial Genetic Structure Analysis to Unravel the Reproductive Strategy of an Insular Threatened Orchid. S. Afr. J. Bot. 2016, 105, 25–35. [Google Scholar] [CrossRef]
- Anjos, A.; Barberena, F.; Pigozzo, C. Biologia Reprodutiva de Vanilla Bahiana Hoehne (Orchidaceae). Orquidário 2017, 30, 3–4. [Google Scholar]
- Watteyn, C.; Scaccabarozzi, D.; Muys, B.; Van Der Schueren, N.; Van Meerbeek, K.; Guizar Amador, M.F.; Ackerman, J.D.; Cedeño Fonseca, M.V.; Chinchilla Alvarado, I.F.; Reubens, B.; et al. Trick or Treat? Pollinator Attraction in Vanilla Pompona (Orchidaceae). Biotropica 2022, 54, 268–274. [Google Scholar] [CrossRef]
- Pansarin, E.R.; Ferreira, A.W.C. Evolutionary Disruption in the Pollination System of Vanilla (Orchidaceae). Plant Biol. 2022, 24, 157–167. [Google Scholar] [CrossRef]
- Tremblay, R.L.; Ackerman, J.D.; Zimmerman, J.K.; Calvo, R.N. Variation in Sexual Reproduction in Orchids and Its Evolutionary Consequences: A Spasmodic Journey to Diversification: Evolutionary Processes in Orchids. Biol. J. Linn. Soc. 2004, 84, 1–54. [Google Scholar] [CrossRef]
- Cameron, K. Vanilla Orchids: Natural History and Cultivation; Timber Press: Portland, OR, USA, 2012; ISBN 978-1-60469-321-8. [Google Scholar]
- Cameron, K.M. Vanilla Phylogeny and Classification. In Handbook of Vanilla Science and Technology; Havkin-Frenkel, D., Belanger, F.C., Eds.; John Wiley & Sons Ltd.: Chichester, UK, 2018; pp. 375–390. ISBN 978-1-119-37732-0. [Google Scholar] [CrossRef]
- Rodolphe, G.; Severine, B.; Michel, G.; Pascale, B. Biodiversity and Evolution in the Vanilla Genus. In The Dynamical Processes of Biodiversity—Case Studies of Evolution and Spatial Distribution; Grillo, O., Ed.; InTech: London, UK, 2011; ISBN 978-953-307-772-7. [Google Scholar]
- Flanagan, N.S.; Chavarriaga, P.; Mosquera-Espinosa, A.T. Conservation and Sustainable Use of Vanilla Crop Wild Relatives in Colombia. In Handbook of Vanilla Science and Technology; Havkin-Frenkel, D., Belanger, F.C., Eds.; John Wiley & Sons Ltd.: Chichester, UK, 2018; pp. 85–109. ISBN 978-1-119-37732-0. [Google Scholar] [CrossRef]
- Soto Arenas, M.A.; Dressler, R.L. A Revision of the Mexican and Central American Species of Vanilla Plumier Ex Miller with a Characterization of Their Its Region of the Nuclear Ribosomal DNA. Lankesteriana 2013, 9, 285–354. [Google Scholar] [CrossRef]
- Householder, E.; Janovec, J.; Mozambite, A.B.; Maceda, J.H.; Wells, J.; Valega, R.; Maruenda, H.; Christenson, E. Diversity, Natural History, and Conservation of Vanilla (Orchidaceae) in Amazonian Wetlands of Madre De Dios, Peru. J. Bot. Res. Inst. Tex. 2010, 4, 227–243. [Google Scholar]
- Soto Arenas, M.A.; Cribb, P. A New Infrageneric Classification and Synopsis of the Genus Vanilla Plum. Ex Mill. (Orchidaceae: Vanillinae). Lankesteriana 2013, 9, 355–398. [Google Scholar] [CrossRef]
- Chaipanich, V.; Wanachantararak, P.; Hasin, S. Original Article Floral Morphology and Potential Pollinator of Vanilla Siamensis Rolfe Ex Downie (Orchidaceae: Vanilloideae) in Thailand. Thail. Nat. Hist. Mus. J. 2020, 14, 1–14. [Google Scholar]
- Damian Parizaca, L.A. Taxonomía del género Vanilla Plum. ex Mill. (Orchidaceae: Vanilleae) en el Perú. Master’s Thesis, Universidad Nacional Mayor de San Marcos, Lima, Perú, 2020. [Google Scholar]
- Pansarin, E.R.; Aguiar, J.M.R.B.V.; Pansarin, L.M. Floral Biology and Histochemical Analysis of Vanilla edwallii Hoehne (Orchidaceae: Vanilloideae): An Orchid Pollinated by Epicharis (Apidae: Centridini): Floral Biology of Vanilla edwallii. Plant Species Biol. 2014, 29, 242–252. [Google Scholar] [CrossRef]
- Bory, S.; Brown, S.; Duval, M.-F.; Besse, P. Evolutionary Processes and Diversification in the Genus Vanilla. In Vanilla; Odoux, E., Grisoni, M., Eds.; CRC Press: Boca Raton, FL, USA, 2010; pp. 15–28. ISBN 978-0-429-19197-8. [Google Scholar]
- Childers, N.F.; Cibes, H.R. Vanilla Culture in Puerto Rico; U.S. Department of Agriculture: Washington, DC, USA, 1948. [Google Scholar]
- Soto Arenas, M.Á. Filogeografía y Recursos Genéticos de Las Vainillas de México. Available online: https://www.snib.mx/iptconabio/resource?r=SNIB-J101 (accessed on 23 November 2022).
- Hernández-Hernández, J. Mexican Vanilla Production. In Handbook of Vanilla Science and Technology; Havkin-Frenkel, D., Belanger, F.C., Eds.; John Wiley & Sons Ltd.: Chichester, UK, 2018; pp. 1–26. ISBN 978-1-119-37732-0. [Google Scholar] [CrossRef]
- Wurz, A.; Grass, I.; Tscharntke, T. Hand Pollination of Global Crops—A Systematic Review. Basic Appl. Ecol. 2021, 56, 299–321. [Google Scholar] [CrossRef]
- Dressler, R.L. The Orchids: Natural History and Classification; Harvard University Press: Cambridge, MA, USA, 1981; ISBN 978-0-674-87525-8. [Google Scholar]
- Pijl, L.; van der Pijl, L.; Dodson, C.H. Orchid Flowers: Their Pollination and Evolution; Published Jointly by the Fairchild Tropical Garden and the University of Miami Press: Miami, FL, USA, 1966; ISBN 978-0-87024-069-0. [Google Scholar]
- Vega, M.; Solís-Montero, L.; Alavez, V.; Rodríguez-Juárez, P.; Gutiérrez-Alejo, M.; Wegier, A. Vanilla Cribbiana Soto Arenas Vanilla Hartii Rolfe Vanilla Helleri A.D. Hawkes Vanilla Inodora Schiede Vanilla Insignis Ames Vanilla Odorata C. Presl Vanilla Phaeantha Rchb. f. Vanilla planifolia Jacks Ex. Andrews Vanilla Pompona Schiede Orchidaceae. In Ethnobotany of the Mountain Regions of Mexico; Casas, A., Blancas Vázquez, J.J., Eds.; Ethnobotany of Mountain Regions; Springer International Publishing: Cham, Germany, 2022; pp. 1–21. ISBN 978-3-319-77089-5. [Google Scholar]
- Bory, S.; Grisoni, M.; Duval, M.-F.; Besse, P. Biodiversity and Preservation of Vanilla: Present State of Knowledge. Genet. Resour. Crop Evol. 2008, 55, 551–571. [Google Scholar] [CrossRef]
- Ranadive, A.S. Quality Control of Vanilla Beans and Extracts. In Handbook of Vanilla Science and Technology; Havkin-Frenkel, D., Belanger, F.C., Eds.; John Wiley & Sons Ltd.: Chichester, UK, 2018; pp. 237–260. ISBN 978-1-119-37732-0. [Google Scholar] [CrossRef]
- Gliessman, S. Preserving the Wildness of Crop Wild Relatives. Agroecol. Sustain. Food Syst. 2022, 46, 1–2. [Google Scholar] [CrossRef]
- Hasing, T.; Tang, H.; Brym, M.; Khazi, F.; Huang, T.; Chambers, A.H. A Phased Vanilla planifolia Genome Enables Genetic Improvement of Flavour and Production. Nat. Food 2020, 1, 811–819. [Google Scholar] [CrossRef]
- Belanger, F.C.; Havkin-Frenkel, D. Molecular Analysis of a Vanilla Hybrid Cultivated in Costa Rica. In Handbook of Vanilla Science and Technology; Havkin-Frenkel, D., Belanger, F.C., Eds.; John Wiley & Sons Ltd.: Chichester, UK, 2018; pp. 391–401. ISBN 978-1-119-37732-0. [Google Scholar] [CrossRef]
- Quirós, E.V. Vanilla Production in Costa Rica. In Handbook of Vanilla Science and Technology; Havkin-Frenkel, D., Belanger, F.C., Eds.; John Wiley & Sons Ltd: Chichester, UK, 2018; pp. 41–51. ISBN 978-1-119-37732-0. [Google Scholar] [CrossRef]
- Chambers, A.H. Vanilla (Vanilla spp.) Breeding. In Advances in Plant Breeding Strategies: Industrial and Food Crops; Al-Khayri, J.M., Jain, S.M., Johnson, D.V., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 707–734. ISBN 978-3-030-23264-1. [Google Scholar]
- Djoumbou Feunang, Y.; Eisner, R.; Knox, C.; Chepelev, L.; Hastings, J.; Owen, G.; Fahy, E.; Steinbeck, C.; Subramanian, S.; Bolton, E.; et al. ClassyFire: Automated Chemical Classification with a Comprehensive, Computable Taxonomy. J. Cheminformatics 2016, 8, 61. [Google Scholar] [CrossRef] [PubMed]
- Garg, N.; Sethupathy, A.; Tuwani, R.; Nk, R.; Dokania, S.; Iyer, A.; Gupta, A.; Agrawal, S.; Singh, N.; Shukla, S.; et al. FlavorDB: A Database of Flavor Molecules. Nucleic Acids Res. 2018, 46, D1210–D1216. [Google Scholar] [CrossRef] [PubMed]
- Galeas, M.D.P. Gas Chromatography—Mass Spectrometry and Gas Chromatography-Olfactometry Analysis of Aroma Compounds of Vanilla Pompona Schiede. 2015. Available online: https://rucore.libraries.rutgers.edu/rutgers-lib/46341/ (accessed on 23 November 2022). [CrossRef]
- Toth, S. Volatile Compounds in Vanilla. In Handbook of Vanilla Science and Technology; Havkin-Frenkel, D., Belanger, F.C., Eds.; John Wiley & Sons Ltd.: Chichester, UK, 2018; pp. 285–347. ISBN 978-1-119-37732-0. [Google Scholar] [CrossRef]
- Ehlers, D.; Pfister, M. Compounds of Vanillons (Vanilla Pompona Schiede). J. Essent. Oil Res. 1997, 9, 427–431. [Google Scholar] [CrossRef]
- Palama, T.L.; Grisoni, M.; Fock-Bastide, I.; Jade, K.; Bartet, L.; Choi, Y.H.; Verpoorte, R.; Kodja, H. Metabolome of Vanilla planifolia (Orchidaceae) and Related Species under Cymbidium Mosaic Virus (CymMV) Infection. Plant. Physiol. Biochem. 2012, 60, 25–34. [Google Scholar] [CrossRef]
- Maruenda, H.; del Vico, M.L.; Householder, J.E.; Janovec, J.P.; Cañari, C.; Naka, A.; Gonzalez, A.E. Exploration of Vanilla Pompona from the Peruvian Amazon as a Potential Source of Vanilla Essence: Quantification of Phenolics by HPLC-DAD. Food Chem. 2013, 138, 161–167. [Google Scholar] [CrossRef]
- Leyva, V.E.; Lopez, J.M.; Zevallos-Ventura, A.; Cabrera, R.; Cañari-Chumpitaz, C.; Toubiana, D.; Maruenda, H. NMR-Based Leaf Metabolic Profiling of V. Planifolia and Three Endemic Vanilla Species from the Peruvian Amazon. Food Chem. 2021, 358, 129365. [Google Scholar] [CrossRef]
- Da Silva Oliveira, J.P.; Garrett, R.; Bello Koblitz, M.G.; Furtado Macedo, A. Vanilla Flavor: Species from the Atlantic Forest as Natural Alternatives. Food Chem. 2022, 375, 131891. [Google Scholar] [CrossRef]
- Kundu, A. Vanillin Biosynthetic Pathways in Plants. Planta 2017, 245, 1069–1078. [Google Scholar] [CrossRef]
- Brumano, C.A.N. A trajetória social da baunilha do Cerrado na cidade de Goiás/GO. Master’s Thesis, Universidade de Brasília, Brasília, Brazil, 2019. [Google Scholar]
- Vieira, R.F.; Camillo, J.; Coradin, L. Espécies Nativas da Flora Brasileira de Valor Econômico Atual ou Potencial: Plantas Para o Futuro: Região Centro-Oeste; Ministério do Meio Ambiente: Brasília, Brazil, 2016; ISBN 978-85-7738-309-2. [Google Scholar]
- Singletary, K.W. Vanilla: Potential Health Benefits. Nutr. Today 2020, 55, 186–196. [Google Scholar] [CrossRef]



| Species | Breeding System | Natural Fruit Set | Manual Self-Pollination Fruit Set | Manual Cross-Pollination Fruit Set | Spontaneous Self-Pollination Fruit Set | Potential Pollinators | References |
|---|---|---|---|---|---|---|---|
| V. bahiana | Outcrossing | 2.35% | 11.11–71.43% | 24.44–83.33% | 0 | Eulaema sp. bees | [40] |
| V. barbellata Rchb. f. | Outcrossing | 18.2% | 100% | - | - | Bees | [43] |
| V. bicolor | Autogamy | 43% | - | - | 71% | - | [33,49] |
| V. bosseri | Outcrossing | 3.96% | 86% | 43% | 0 | Bees (Macrogalea ellioti, Liotrigona modecassa, Liotrigona mahafalya) | [37] |
| V. claviculata | Outcrossing | 15% | 100% | - | - | Bees | [43] |
| V. chamissonis | Outcrossing | 21.21% | 78.78% | 75.76% | 6.06% | - | [34] |
| V. cristato-callosa | Outcrossing | 6.6% | - | - | - | Euglossine (Euglossa sp.) bees | [49] |
| V. dilloniana | Outcrossing | 14.5% | 100% | - | - | Bees | [43] |
| V. edwallii | Outcrossing | <15% | - | - | 0 | Epicharis affinis bees | [53] |
| V. guianensis | Autogamy | 78% | - | - | - | - | [49] |
| V. humblotii | Outcrossing | 0.62–1.2% | 90.9% | 86.7% | 6.7% | Allodape obscuripennis bees and sunbird (Nectarinia coquerelli) | [39] |
| V. martinezii | Autogamy | up to 53% in a clone | - | - | - | - | [48] |
| V. mexicana (syn. V. inodora) | Autogamy | 2.5–53.7% | - | - | 53.9% | Carpenter bees (Xylocopa sp.) | [38,48] |
| V. palmarum | Autogamy | 67.3–71.4% | 76.7–80.0% | 80–83.3% | 66.6–73.3% | Hummingbirds (Amazilia fimbriata) | [42,49] |
| V. paulista | Outcrossing | 0.4–6.9% | 100% | - | 0 | Eulaema nigrita and Eufriesea violacea | [36] |
| V. poitaei | Outcrossing | 6.4% | 100% | - | - | Bees | [43] |
| V. pompona | Outcrossing | 2.42% and 5% | - | - | 0 | Eulaema cingulata bees | [41,57] |
| V. riberoi | Outcrossing | 1.1% | - | - | - | Euglossine bees | [49] |
| V. roscheri | Outcrossing | 26.3% | 64% | 71% | 0 | Allodapine bees (Allodapula variegata and Allodape rufogastra) | [35] |
| V. siamensis | Outcrossing | 3.6% | - | - | - | Thrinchostoma spp. bees | [51] |
| First Authors | Year of Publication | Species | Plant Organs |
|---|---|---|---|
| Ehlers and Pfister [73] | 1997 | V. pompona | fruits |
| Palama et al. [74] | 2012 | V. pompona | leaves |
| Maruenda et al. [75] | 2013 | V. pompona | fruits |
| Galeas [71] | 2015 | V. pompona | fruits |
| Toth et al. [72] | 2018 | V. pompona (from Madagascar), V. pompona (origin unknown), Wild Type (from Peru) | fruits |
| Leyva et al. [76] | 2021 | V. pompona, V. palmarum, V. ribeiroi | leaves |
| Pérez-Silva et al. [6] | 2021 | V. pompona subsp grandiflora, V. pompona subsp pittieri, V. sotoarenasii, V. crenulata, V. imperialis, V. bahiana, V. lindmaniana, V. planifolia × V. pompona, V. planifolia × V. phaeantha | fruits |
| da Silva Oliveira et al. [77] | 2022 | V. bahiana, V. chamissonis | fruits |
| Watteyn et al. [41] | 2022 | V. pompona | flowers |
| Species | p-Anisyl Alcohol | p-Hydroxybenzaldehyde | p-Hydroxybenzoic Acid | p-Hydroxybenzyl Alcohol | Vanillic Acid | Vanillin | Vanillyl Alcohol |
|---|---|---|---|---|---|---|---|
| V. bahiana | [6] | [6,77] | [6,77] | [6] | [6,77] | [77] | [6,77] |
| V. chamissonis | [77] | [77] | [77] | [77] | [77] | ||
| V. crenulata | [6] | [6] | |||||
| V. imperialis | [6] | [6] | |||||
| V. planifolia × V. phaeantha | [6] | [6] | [6] | [6] | [6] | [6] | [6] |
| V. planifolia × V. pompona | [6] | [6] | [6] | [6] | [6] | [6] | [6] |
| V. pompona (from Madagascar) | [72] | [72] | [72] | [72] | |||
| V. pompona (origin unknown) | [72] | [72] | [72] | [72] | [72] | [72] | |
| V. pompona Shiede (cured) | [71,73] | [71,73] | [73] | [71] | [73] | [71,73] | |
| V. pompona subsp grandiflora | [6,75] | [6,75] | [6,75] | [6,75] | [6,75] | [6,75] | [6,75] |
| V. pompona subsp pittieri | [6] | [6] | [6] | [6] | [6] | [6] | |
| V. sotoarenasii | [6] | [6] | [6] | [6] | [6] | [6] | |
| Wild Type (from Peru) | [72] | [72] | [72] | [72] | [72] | [72] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Oliveira, R.T.; da Silva Oliveira, J.P.; Macedo, A.F. Vanilla beyond Vanilla planifolia and Vanilla × tahitensis: Taxonomy and Historical Notes, Reproductive Biology, and Metabolites. Plants 2022, 11, 3311. https://doi.org/10.3390/plants11233311
de Oliveira RT, da Silva Oliveira JP, Macedo AF. Vanilla beyond Vanilla planifolia and Vanilla × tahitensis: Taxonomy and Historical Notes, Reproductive Biology, and Metabolites. Plants. 2022; 11(23):3311. https://doi.org/10.3390/plants11233311
Chicago/Turabian Stylede Oliveira, Renatha Tavares, Joana Paula da Silva Oliveira, and Andrea Furtado Macedo. 2022. "Vanilla beyond Vanilla planifolia and Vanilla × tahitensis: Taxonomy and Historical Notes, Reproductive Biology, and Metabolites" Plants 11, no. 23: 3311. https://doi.org/10.3390/plants11233311
APA Stylede Oliveira, R. T., da Silva Oliveira, J. P., & Macedo, A. F. (2022). Vanilla beyond Vanilla planifolia and Vanilla × tahitensis: Taxonomy and Historical Notes, Reproductive Biology, and Metabolites. Plants, 11(23), 3311. https://doi.org/10.3390/plants11233311






