Climatic Factors Determine the Distribution Patterns of Leaf Nutrient Traits at Large Scales
Abstract
:1. Introduction
2. Results
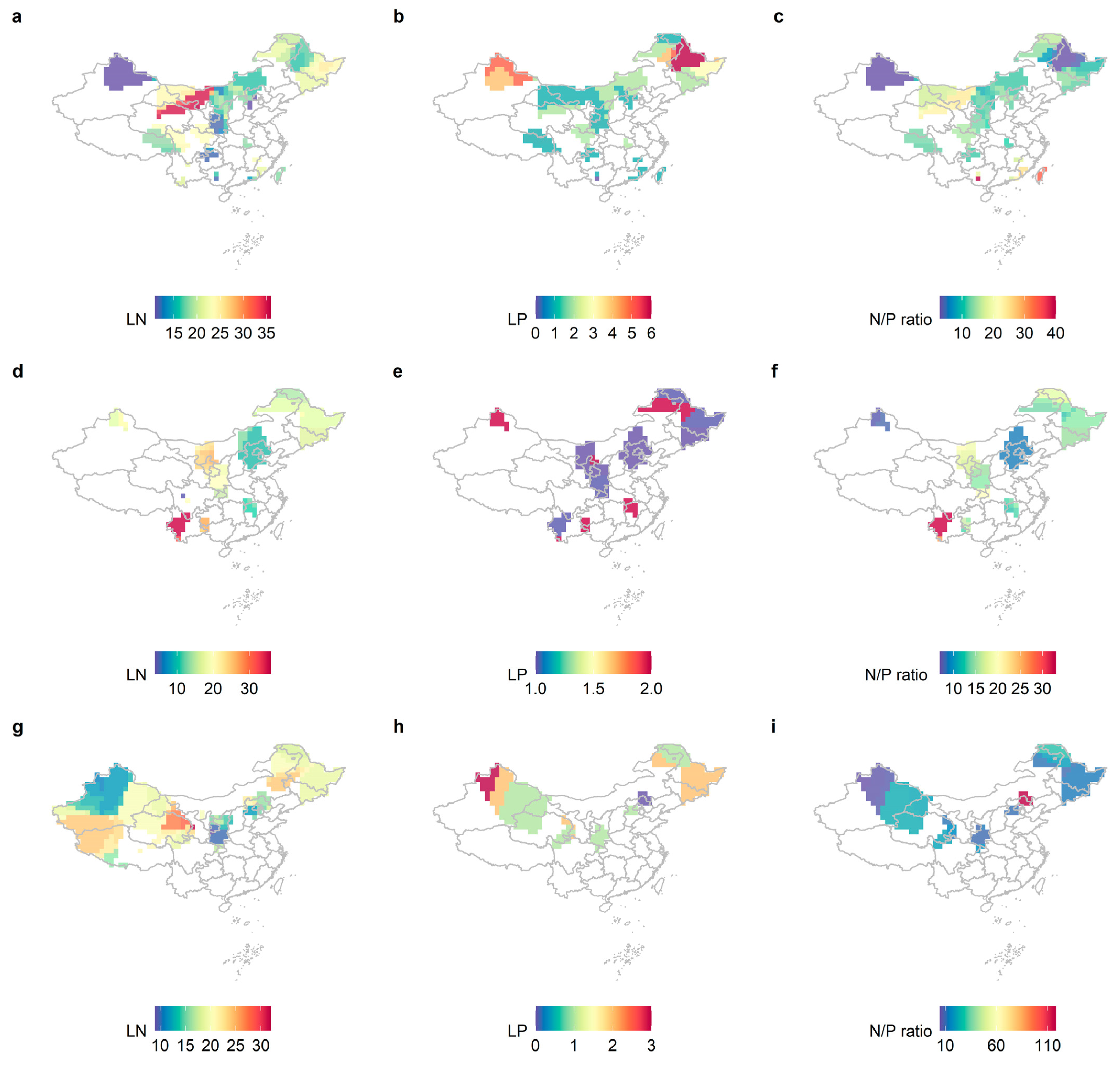
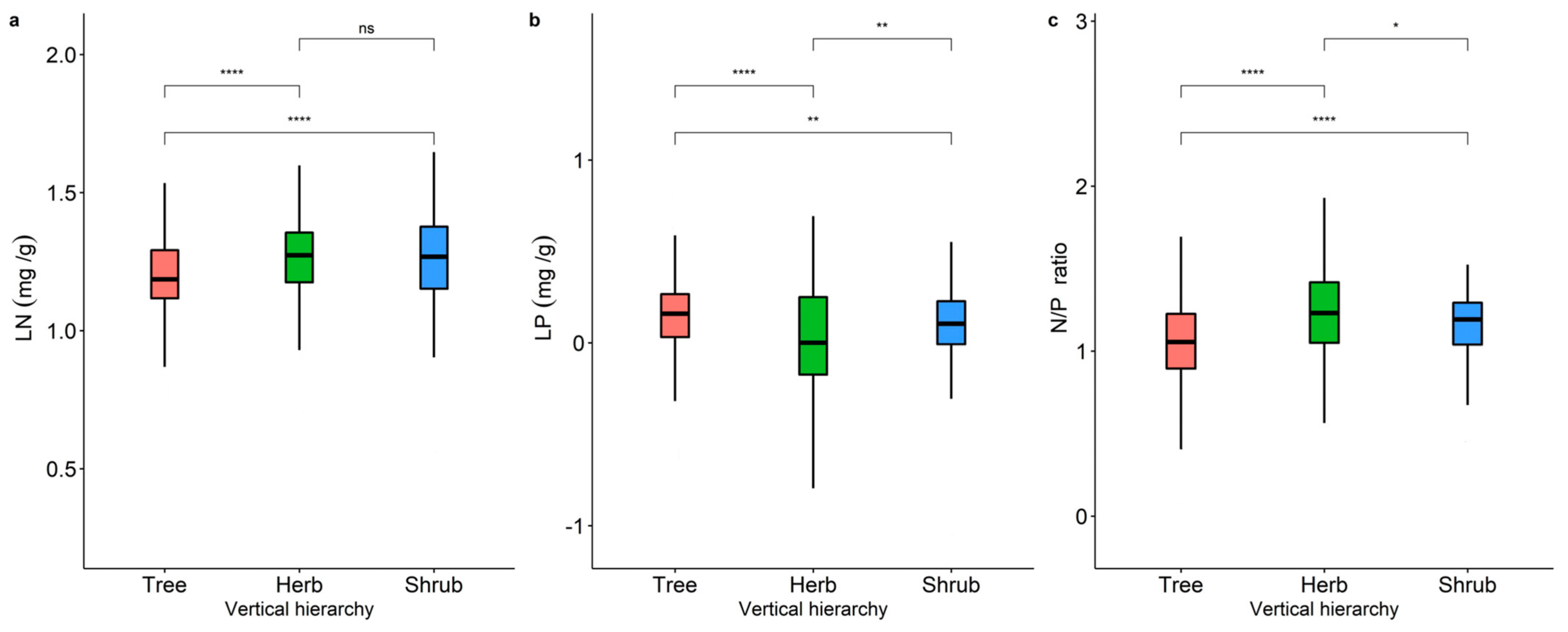
2.1. Variability in LN, LP, and N/P of Different Life Forms and Distribution Patterns in China
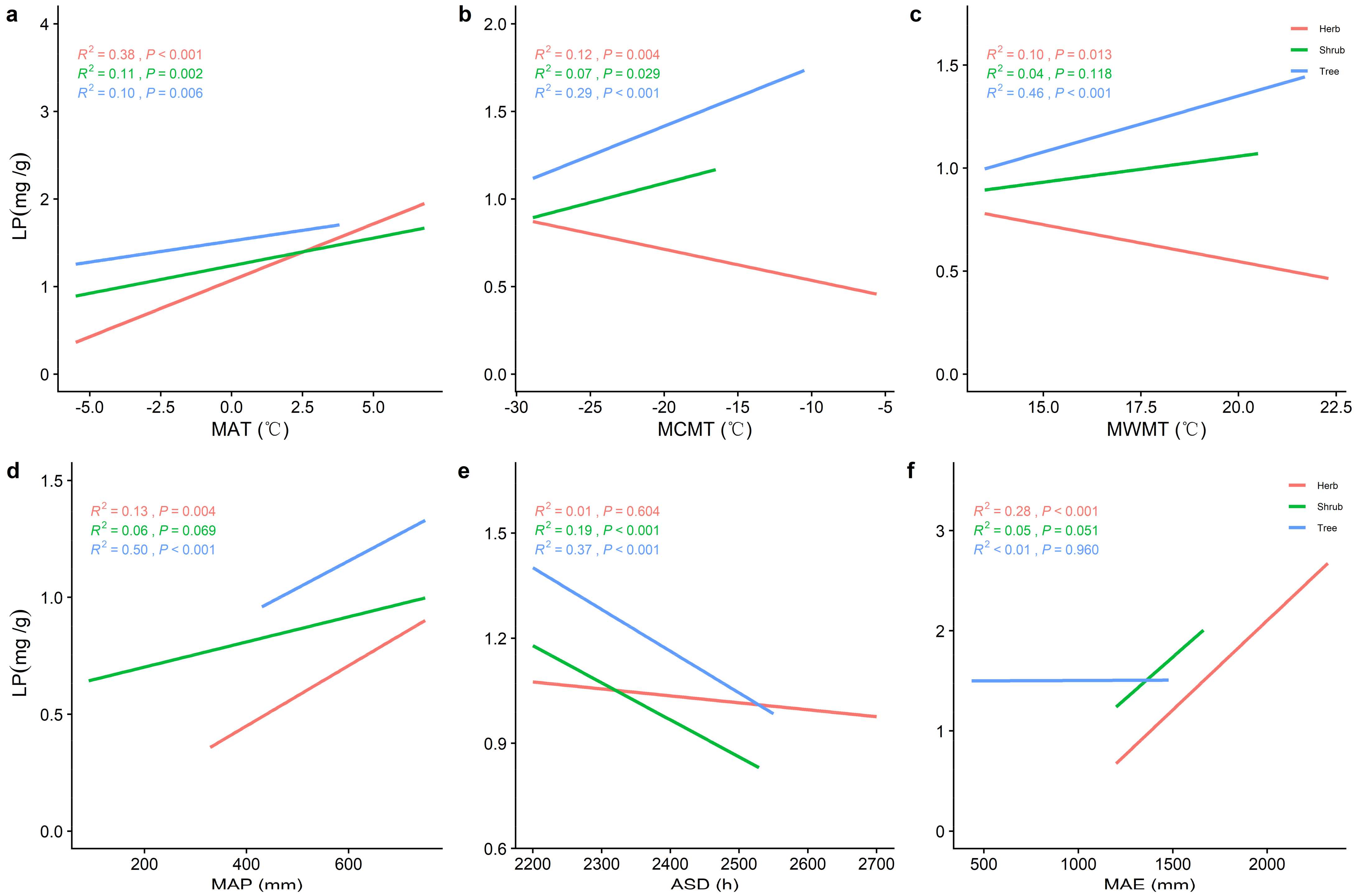
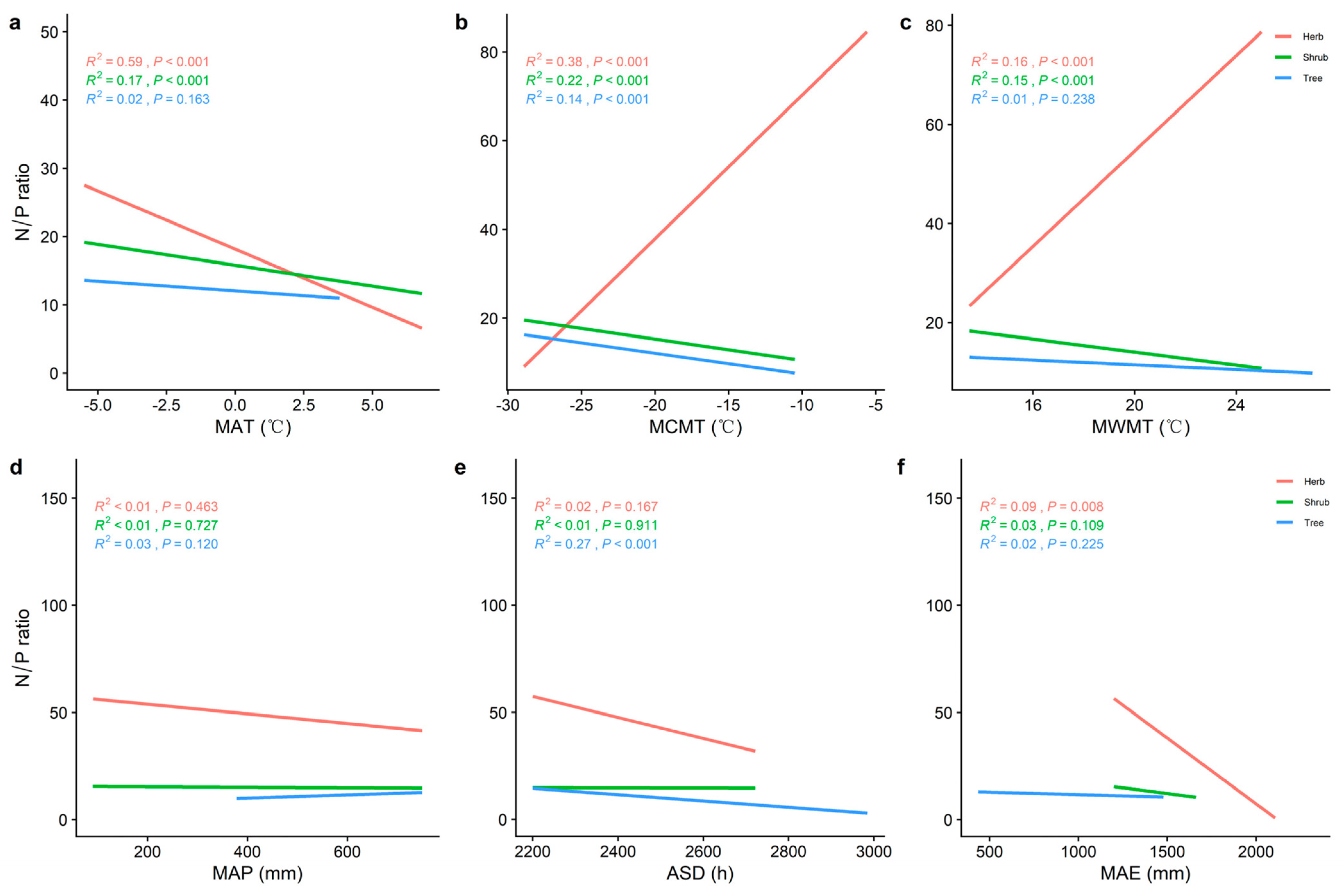
2.2. Correlations between Climatic Factors and LN, LP, and N/P at Different Life Forms
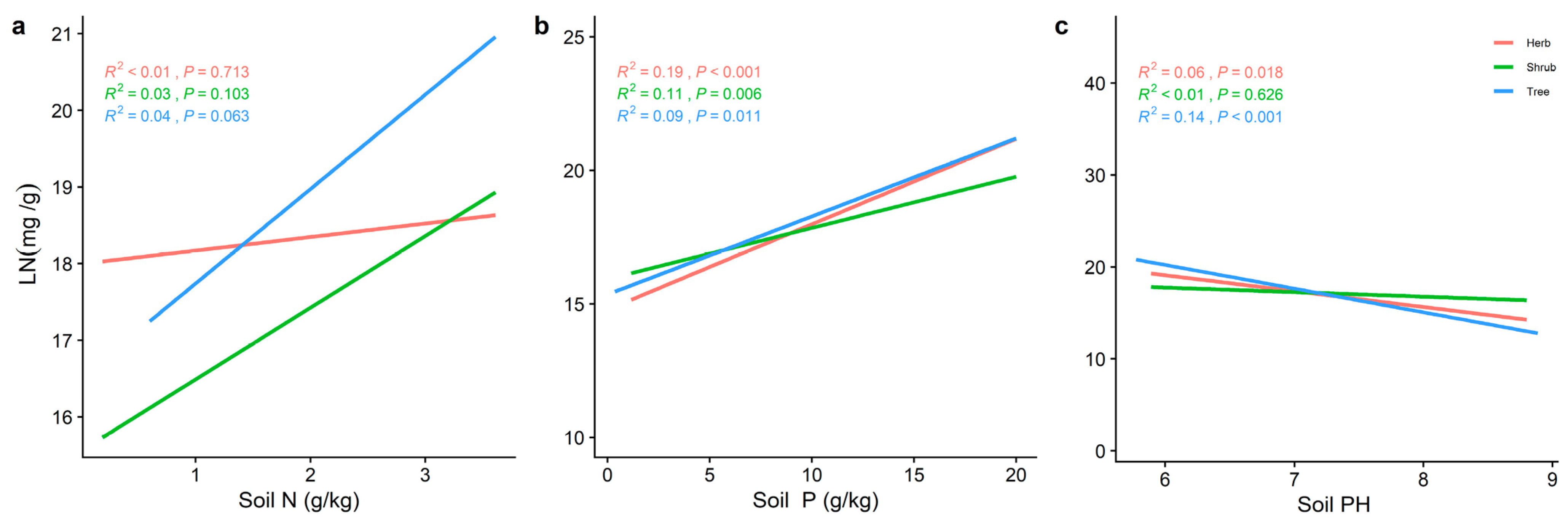
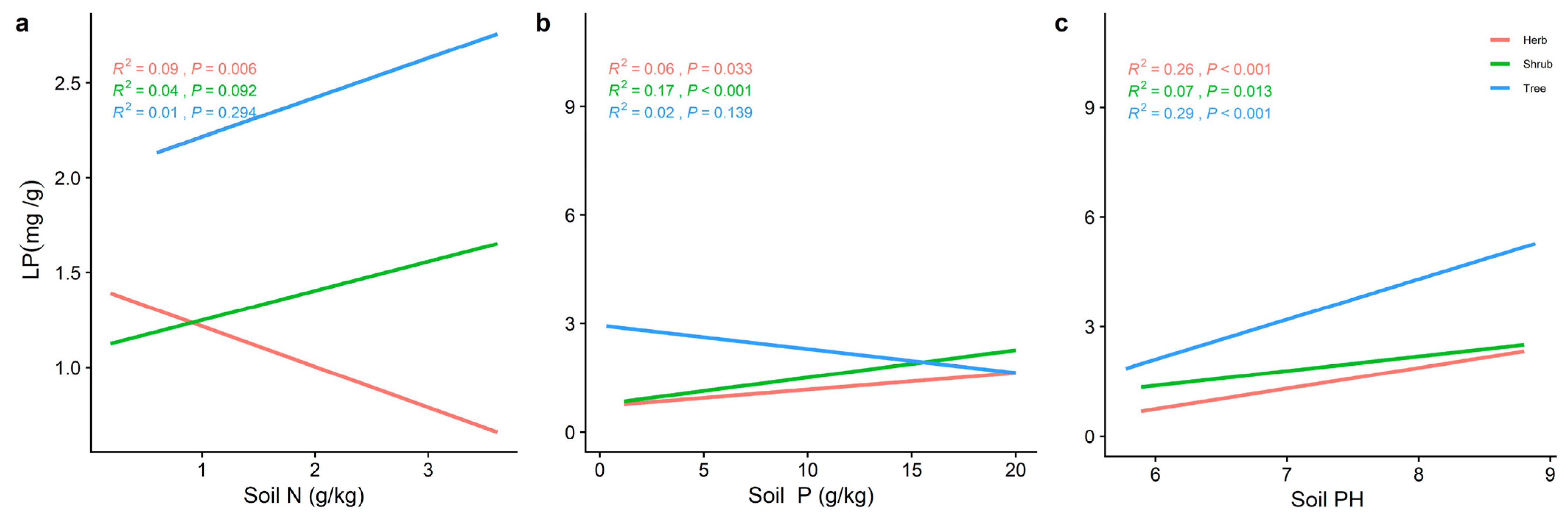
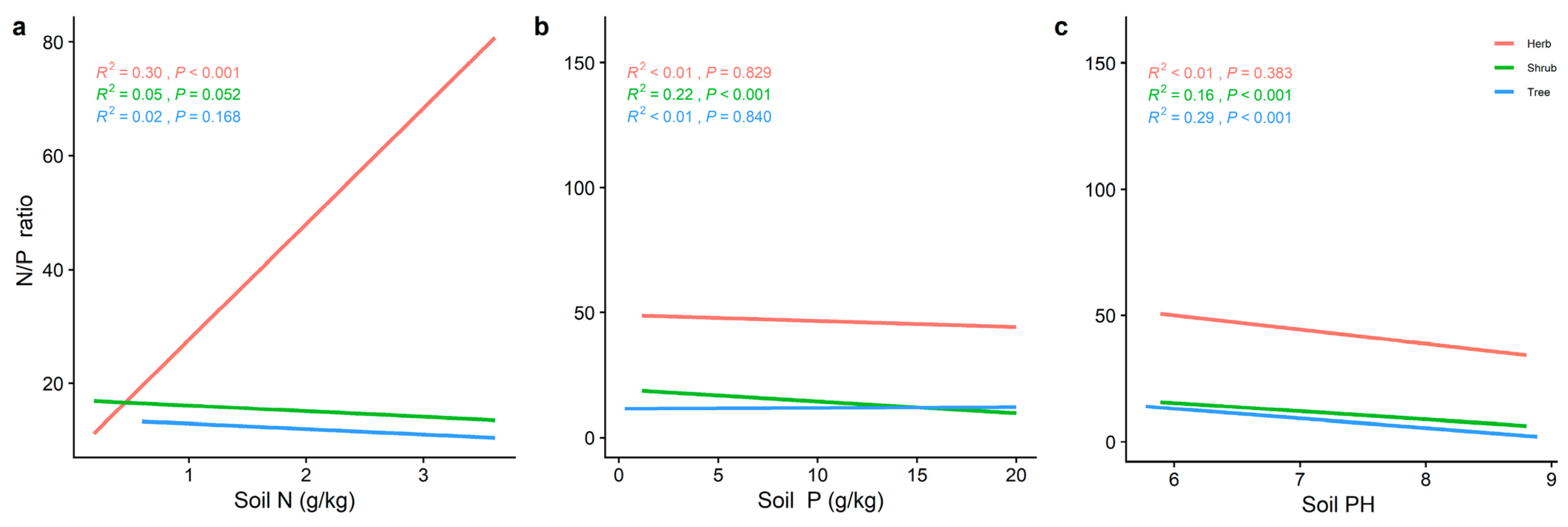
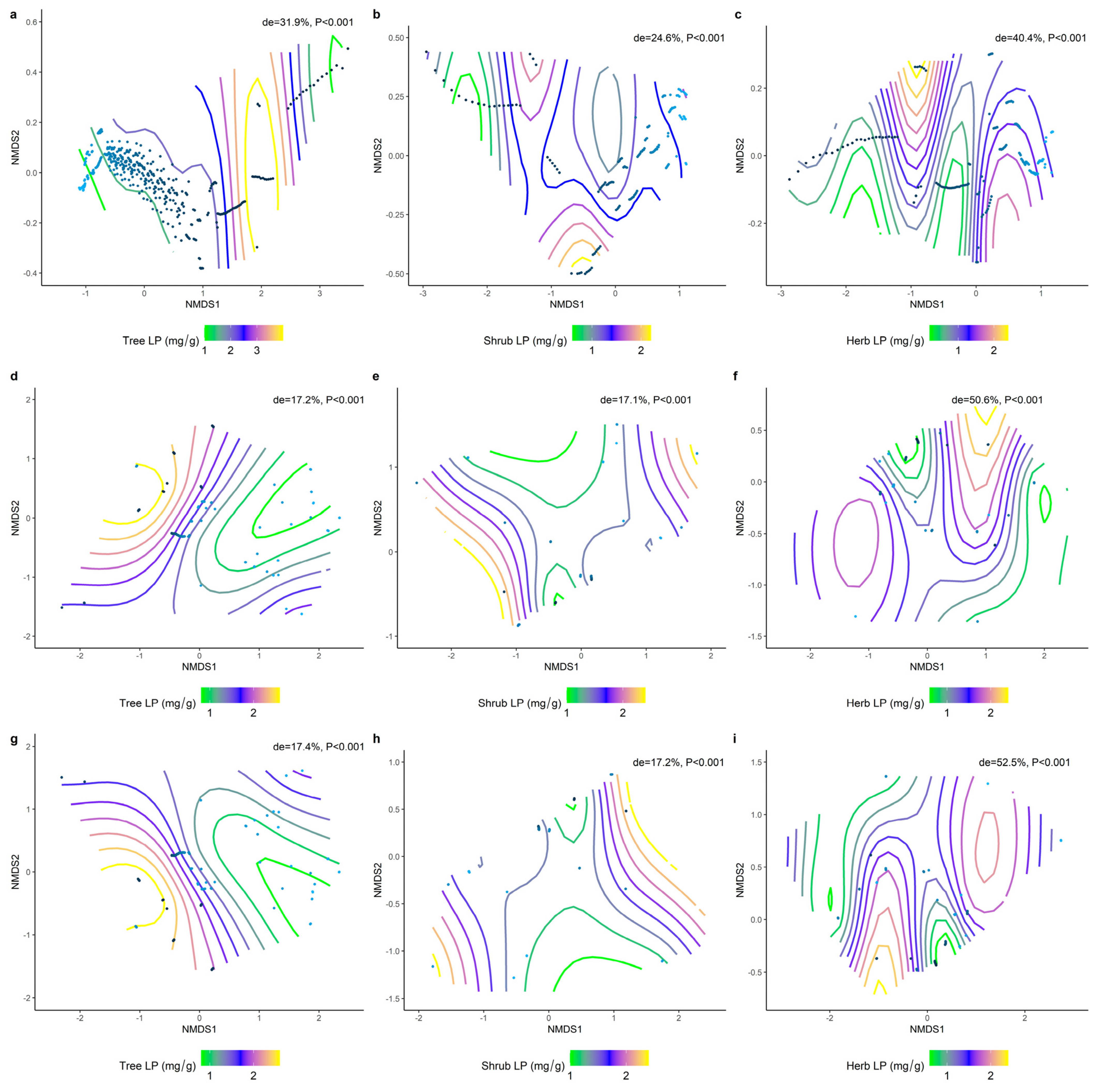
2.3. Effect of Soil Factors on the Relationship between LN, LP, and Leaf N/P at Different Life Forms
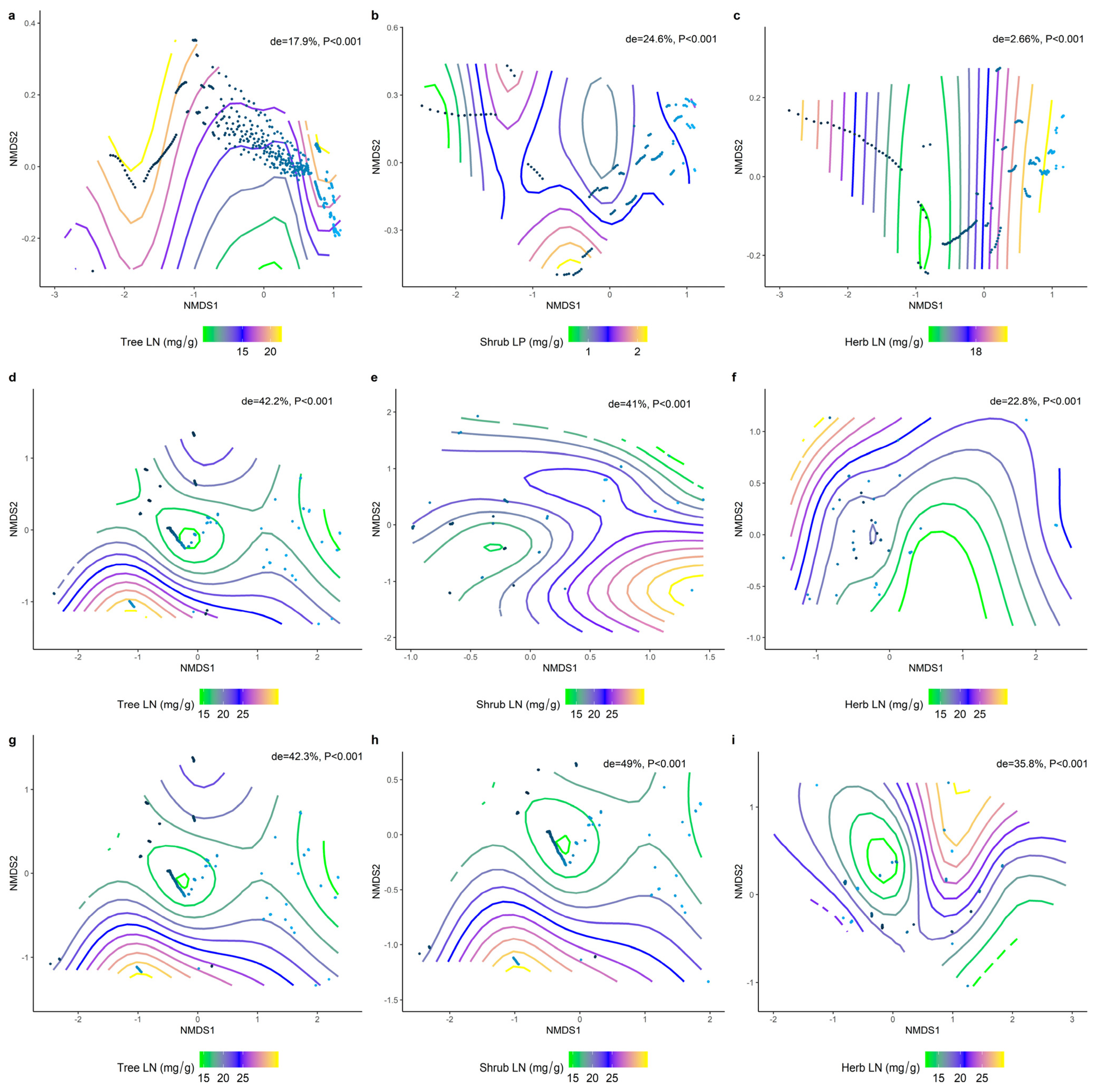
2.4. Climatic and Soil Factors Dominate Changes in the Functional Traits of Different Communities
3. Discussion
4. Materials and Methods
4.1. Study Area and Sample Data
4.2. Functional Data
4.3. Environmental Data
4.4. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bello, F.D.; Lavorel, S.; Díaz, S.; Berg, M.P.; Cipriotti, P.; Feld, C.K.; Hering, D. Towards an assessment of multiple ecosystem processes and services via functional traits. Biodivers. Conserv. 2010, 19, 2873–2893. [Google Scholar] [CrossRef]
- Ma, J.; Niklas, K.J.; Liu, L.; Fang, Z.D.; Li, Y.R.; Shi, P.J. TTree size influences leaf shape but does not affect the proportional relationship between leaf area and the product of length and width. Front. Plant Sci. 2022, 13, 850203. [Google Scholar] [CrossRef]
- Huang, C.; Xu, Y.; Zang, R. Variation patterns of functional trait moments along geographical gradients and their environmental determinants in the subtropical evergreen broadleaved forests. Front. Plant Sci. 2021, 12, 1414. [Google Scholar] [CrossRef]
- Tian, D.; Yan, Z.; Ma, S.; Ding, Y.; Luo, Y.; Chen, Y.; Du, E.; Han, W.; Kovacs, E.D.; Shen, H.; et al. Family-level leaf nitrogen and phosphorus stoichiometry of global terrestrial plants. Sci. China Life Sci. 2019, 62, 1047–1057. [Google Scholar] [CrossRef] [Green Version]
- Hofhansl, F.; Chacón-Madrigal, E.; Brännström, Å.; Dieckmann, U.; Franklin, O. Mechanisms driving plant functional trait variation in a tropical forest. Ecol. Evol. 2021, 11, 3856–3870. [Google Scholar] [CrossRef]
- Wang, Z.; Zheng, R.; Yang, L.; Tan, T.; Li, H.; Liu, M. Elevation gradient distribution of indices of tree population in a montane forest: The role of leaf traits and the environment. For. Ecosyst. 2022, 9, 100012. [Google Scholar] [CrossRef]
- Taylor, P.G.; Cleveland, C.C.; Wieder, W.R.; Sullivan, B.W.; Doughty, C.E.; Dobrowski, S.Z.; Townsend, A.R. Temperature and rainfall interact to control carbon cycling in tropical forests. Ecol. Lett. 2017, 20, 779–788. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; Zhang, Y.; Xiong, K.; Yu, Y.; Min, X. Changes of leaf functional traits in karst rocky desertification ecological environment and the driving factors. Glob. Ecol. Conserv. 2020, 24, e01381. [Google Scholar] [CrossRef]
- Guo, Y.; Yan, Z.; Zhang, Y.W.; Zhou, G.; Xie, Z.; Tang, Z. Environmental constraints on the inter-genus variation in the scaling relationship between leaf nitrogen and phosphorus concentrations. J. Plant Ecol. 2021, 14, 616–627. [Google Scholar] [CrossRef]
- Biswas, D.; Ma, B.; Morrison, M.J. Changes in leaf nitrogen and phosphorus, photosynthesis, respiration, growth and resource use efficiency of a rapeseed cultivar as affected by drought and high temperature. Can. J. Plant Sci. 2019, 99, 413–419. [Google Scholar] [CrossRef]
- Borer, E.T.; Bracken, M.E.; Seabloom, E.W.; Smith, J.E.; Cebrian, J.; Cleland, E.E.; Elser, J.J.; Fagan, W.F.; Gruner, D.S.; Harpole, W.S. Global biogeography of autotroph chemistry: Is insolation a driving force? Oikos 2013, 122, 1121–1130. [Google Scholar] [CrossRef] [Green Version]
- Miatto, R.C.; Wright, I.J.; Batalha, M.A. Relationships between soil nutrient status and nutrient-related leaf traits in Brazilian cerrado and seasonal forest communities. Plant Soil. 2016, 404, 13–33. [Google Scholar] [CrossRef]
- Wright, I.J.; Dong, N.; Maire, V.; Prentice, I.C.; Westoby, M.; Díaz, S. Global climatic drivers of leaf size. Nature 2017, 357, 917–921. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, H.; Gong, Y.; Zuo, X. Precipitation variability affects aboveground biomass directly and indirectly via plant functional traits in the desert steppe of inner Mongolia, Northern China. Front. Plant Sci. 2021, 12, 674527. [Google Scholar] [CrossRef] [PubMed]
- Picotte, J.J.; Rhode, J.M.; Cruzan, M.B. Leaf morphological responses to variation in water availability for plants in the Piriqueta caroliniana complex. Plant Ecol. 2009, 200, 267–275. [Google Scholar] [CrossRef]
- Fang, Z.; Han, X.Y.; Xie, M.Y.; Jiao, F. Spatial Distribution Patterns and Driving Factors of Plant Biomass and Leaf N, P Stoichiometry on the Loess Plateau of China. Plants. 2021, 10, 2420. [Google Scholar] [CrossRef] [PubMed]
- Faucon, M.P.; Houben, D.; Lambers, H. Plant Functional Traits: Soil and Ecosystem Services. Trends Plant Sci. 2017, 20, 385–394. [Google Scholar] [CrossRef]
- Zhu, J. Abiotic Stress Signaling and Responses in Plants. Cell. 2016, 167, 313–324. [Google Scholar] [CrossRef] [Green Version]
- Gong, H.; Li, Y.; Yu, T.; Zhang, S.; Sun, D. Soil and climate effects on leaf nitrogen and phosphorus stoichiometry along elevational gradients. Glob. Ecol. Conserv. 2020, 23, e01138. [Google Scholar] [CrossRef]
- Zhu, B.; Izuta, T.; Watanabe, M. Nitrogen use efficiency of Quercus serrata seedlings under different soil nitrogen and phosphorus supplies. J. Agric. Meteorol. 2020, 76, 11–18. [Google Scholar]
- Zhang, Y.W.; Guo, Y.; Tang, Z.; Feng, Y.; Fang, J. Patterns of nitrogen and phosphorus pools in terrestrial ecosystems in China. Earth Syst. Sci. Data 2021, 13, 5337–5351. [Google Scholar] [CrossRef]
- Gao, J.; Song, Z.; Liu, Y. Response mechanisms of leaf nutrients of endangered plant (Acer catalpifolium) to environmental factors varied at different growth stages. Glob. Ecol. Conserv. 2019, 17, e00521. [Google Scholar] [CrossRef]
- Latham, P.A.; Zuuring, H.R.; Coble, D.W. A method for quantifying vertical forest structure. For. Ecol. Manag. 1998, 104, 157–170. [Google Scholar] [CrossRef]
- Cheng, X.; Ping, T.; Li, Z.; Wang, T.; Han, H.; Epstein, H.E. Effects of environmental factors on plant functional traits across different plant life forms in a temperate forest ecosystem. New For. 2022, 53, 125–142. [Google Scholar] [CrossRef]
- Poorter, H.; Niinemets, Ü.; Poorter, L.; Wright, I.J.; Villar, R. Causes and consequences of variation in leaf mass per area (LMA): A meta-analysis. New Phytol. 2009, 182, 565–588. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.; Asner, G.; Dahlin, K.; Anderson, C.; Knapp, D.; Martin, R.; Mascaro, J.; Chazdon, R.; Cole, R.; Wanek, W. Landscape-Scale Controls on Aboveground Forest Carbon Stocks on the Osa Peninsula, Costa Rica. PLoS ONE 2015, 10, e0126748. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, D.; Yan, Z.; Niklas, K.J.; Han, W.; Kattge, J.; Reich, P.B.; Luo, Y.; Chen, Y.; Tang, Z.; Hu, H. Global leaf nitrogen and phosphorus stoichiometry and their scaling exponent. Natl. Sci. Rev. 2018, 5, 728–739. [Google Scholar] [CrossRef] [Green Version]
- He, M.S.; Luo, Y.; Peng, Q.W.; Yang, S.Q.; Li, K.H.; Han, W.X. Leaf C: N: P stoichiometry of 67 plant species and its relations with climate factors across the deserts in Xinjiang, China. J. Appl. Ecol. 2019, 30, 2171–2180. [Google Scholar]
- Jing, Y.R.; Deng, X.W.; Hui, W.; Li, Y.Q.; Xiang, W.H. Leaf nitrogen and phosphorus stoichiometry of shrubland plants in the rocky desertification area of Southwestern Hunan, China. J. Appl. Ecol. 2017, 28, 415–422. [Google Scholar]
- Guo, X.C.; Shi, P.J.; Niinemets, Ü.; Wang, R.; Liu, M.; Li, Y.R.; Dong, L. “Diminishing returns” for leaves of five age-groups of Phyllostachys edulis culms. Am. J. Bot. 2021, 108, 1662–1672. [Google Scholar] [CrossRef]
- Jiang, P.P.; Yang, C.; Chen, Y.M.; Aamp, N. C, N, P stoichiometric characteristics of tree, shrub, herb leaves and litter in forest community of Shaanxi Province, China. J. Appl. Ecol. 2016, 27, 365–372. [Google Scholar]
- Wei, D.P.; Zhang, J.; Zhang, D.J.; Li, C.B.; Zhao, Y.B.; Zhang, J. Leaf carbon, nitrogen, and phosphorus resorption and the stoichiometry in Pinus massoniana plantations with various canopy densities. Chin J Appl Environ Biol. 2017, 23, 560–569. [Google Scholar]
- Tan, Q.Q.; Jia, Y.F.; Wang, G.A. Decoupling of soil nitrogen and phosphorus dynamics along a temperature gradient on the Qinghai-Tibetan Plateau. Geoderma. 2021, 396, 115084. [Google Scholar] [CrossRef]
- Bungau, S.; Behl, T.; Aleya, L.; Bourgeade, P.; Aloui-Sossé, B.; Purza, A.L. Expatiating the impact of anthropogenic aspects and climatic factors on long-term soil monitoring and management. Environ. Sci. Pollut. Res. 2021, 28, 30528–30550. [Google Scholar] [CrossRef]
- Mondal, S.; Ghosal, S.; Barua, R. Impact of elevated soil and air temperature on plants growth, yield and physiological interaction: A critical review. Zhongguo Nong Ye Ke Xue 2016, 14, 293–305. [Google Scholar]
- Gong, H.D.; Wang, H.; Jiao, D.Y.; Cai, Z.Q. Phenotypic plasticity of seedlings of five tropical tree species in response to different light and nutrient availability. Trop. Ecol. 2016, 57, 727–737. [Google Scholar]
- Wang, C.X.; He, G.X.; Song, Z.B.; Fan, B.; Shi, L.T. Effects of soil nitrogen and phosphorus levels on leaf nitrogen and phosphorus contents and photosynthesis of tamarindus indica l. In yuanjiang and yuanmou dry-hot valley. Chin. J. Ecol. 2019, 38, 710–718. [Google Scholar]
- Jiang, D.; Geng, Q.; Li, Q.; Luo, Y.; Vogel, J.; Shi, Z.; Ruan, H.; Xu, X. Nitrogen and Phosphorus Resorption in Planted Forests Worldwide. Forests 2019, 10, 201. [Google Scholar] [CrossRef] [Green Version]
- Song, W.; Chen, S.; Zhou, Y.; Lin, G. Rainfall amount and timing jointly regulate the responses of soil nitrogen transformation processes to rainfall increase in an arid desert ecosystem. Geoderma 2020, 364, 114197. [Google Scholar] [CrossRef]
- Gao, X.L.; Li, X.G.; Zhao, L. Shrubs magnify soil phosphorus depletion in Tibetan meadows: Conclusions from C:N:P stoichiometry and deep soil profiles. Sci. Total Environ. 2021, 785, 147320. [Google Scholar] [CrossRef]
- Samuel, A.D.; Bungau, S.; Tit, D.M.; Melinte, C.E.; Purza, L.; Badea, G.E. Effects of long term application of organic and mineral fertilizers on soil enzymes. Rev. Chim. 2018, 69, 2608–2612. [Google Scholar] [CrossRef]
- Liu, Y.; Evans, S.E.; Friesen, M.L.; Tiemann, L.K. Root exudates shift how N mineralization and N fixation contribute to the plant-available N supply in low fertility soils. Soil Biol. Biochem. 2022, 165, 108541. [Google Scholar] [CrossRef]
- Shen, J.; Yuan, L.; Zhang, J.; Li, H.; Bai, Z.; Chen, X.; Zhang, W.; Zhang, F. Phosphorus Dynamics: From Soil to Plant. Plant Physiol. 2011, 156, 997–1005. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hg, A.; Qc, A.; Jie, G.B. Latitudinal, soil and climate effects on key leaf traits in northeastern China. Glob. Ecol. Conserv. 2020, 22, e00904. [Google Scholar]
- He, M.; Dijkstra, F.A.; Zhang, K.; Li, X.; Tan, H.; Gao, Y.; Li, G. Leaf nitrogen and phosphorus of temperate desert plants in response to climate and soil nutrient availability. Sci. Rep. 2014, 4, 6932. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muscarella, R.; Uriarte, M. Do community-weighted mean functional traits reflect optimal strategies? Proc. R. Soc. B Biol. Sci. 2016, 283, 20152434. [Google Scholar] [CrossRef]
- Potarzycki, J. Effect of magnesium or zinc supplementation at the background of nitrogen rate on nitrogen management by maize canopy cultivated in monoculture. Plant Soil Environ. 2011, 57, 19–25. [Google Scholar] [CrossRef] [Green Version]
- Walash, M.I.; Metwally, E.S.; Eid, M.I.; El-Shaheny, R.N. Spectrophotometric determination of risedronate and etidronate in pharmaceutical formulations via the molybdovanadate method. Anal. Lett. 2009, 42, 1571–1587. [Google Scholar] [CrossRef]
- Wang, L.Q.; Ali, A. Climate regulates the functional traits–aboveground biomass relationships at a community-level in forests: A global meta-analysis. Sci. Total Environ. 2021, 761, 143238. [Google Scholar] [CrossRef]
- Wang, J.F.; Wang, X.X.; Ji, Y.H.; Gao, J. Climate factors determine the utilization strategy of forest plant resources at large scales. Front. Plant Sci. 2022, 13, 990441. [Google Scholar] [CrossRef]
- Kramer, P.J. Carbon dioxide concentration, photosynthesis, and dry matter production. Bioscience 1981, 31, 29–33. [Google Scholar] [CrossRef]
- Zou, B.; Chen, J.; Zhai, L.; Fang, X.; Zheng, Z. Satellite based mapping of ground PM2.5 concentration using generalized additive modeling. Remote Sens. 2017, 9, 1. [Google Scholar] [CrossRef] [Green Version]
- Sweeney, C.J.; Vries, F.T.; Dongen, B.E. Root traits explain rhizosphere fungal community composition among temperate grassland plant species. New Phytol. 2021, 229, 1492–1507. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Wang, J.; Zhang, L.; Lv, C.; Liu, L.; Zhao, H.; Gao, J. Climatic Factors Determine the Distribution Patterns of Leaf Nutrient Traits at Large Scales. Plants 2022, 11, 2171. https://doi.org/10.3390/plants11162171
Wang X, Wang J, Zhang L, Lv C, Liu L, Zhao H, Gao J. Climatic Factors Determine the Distribution Patterns of Leaf Nutrient Traits at Large Scales. Plants. 2022; 11(16):2171. https://doi.org/10.3390/plants11162171
Chicago/Turabian StyleWang, Xianxian, Jiangfeng Wang, Liuyang Zhang, Chengyu Lv, Longlong Liu, Huixin Zhao, and Jie Gao. 2022. "Climatic Factors Determine the Distribution Patterns of Leaf Nutrient Traits at Large Scales" Plants 11, no. 16: 2171. https://doi.org/10.3390/plants11162171
APA StyleWang, X., Wang, J., Zhang, L., Lv, C., Liu, L., Zhao, H., & Gao, J. (2022). Climatic Factors Determine the Distribution Patterns of Leaf Nutrient Traits at Large Scales. Plants, 11(16), 2171. https://doi.org/10.3390/plants11162171





