Impact of Storage Condition on Chemical Composition and Antifungal Activity of Pomelo Extract against Colletotrichum gloeosporioides and Anthracnose in Post-harvest Mango
Abstract
:1. Introduction
2. Results
2.1. Chemical Composition of Pomelo Extract
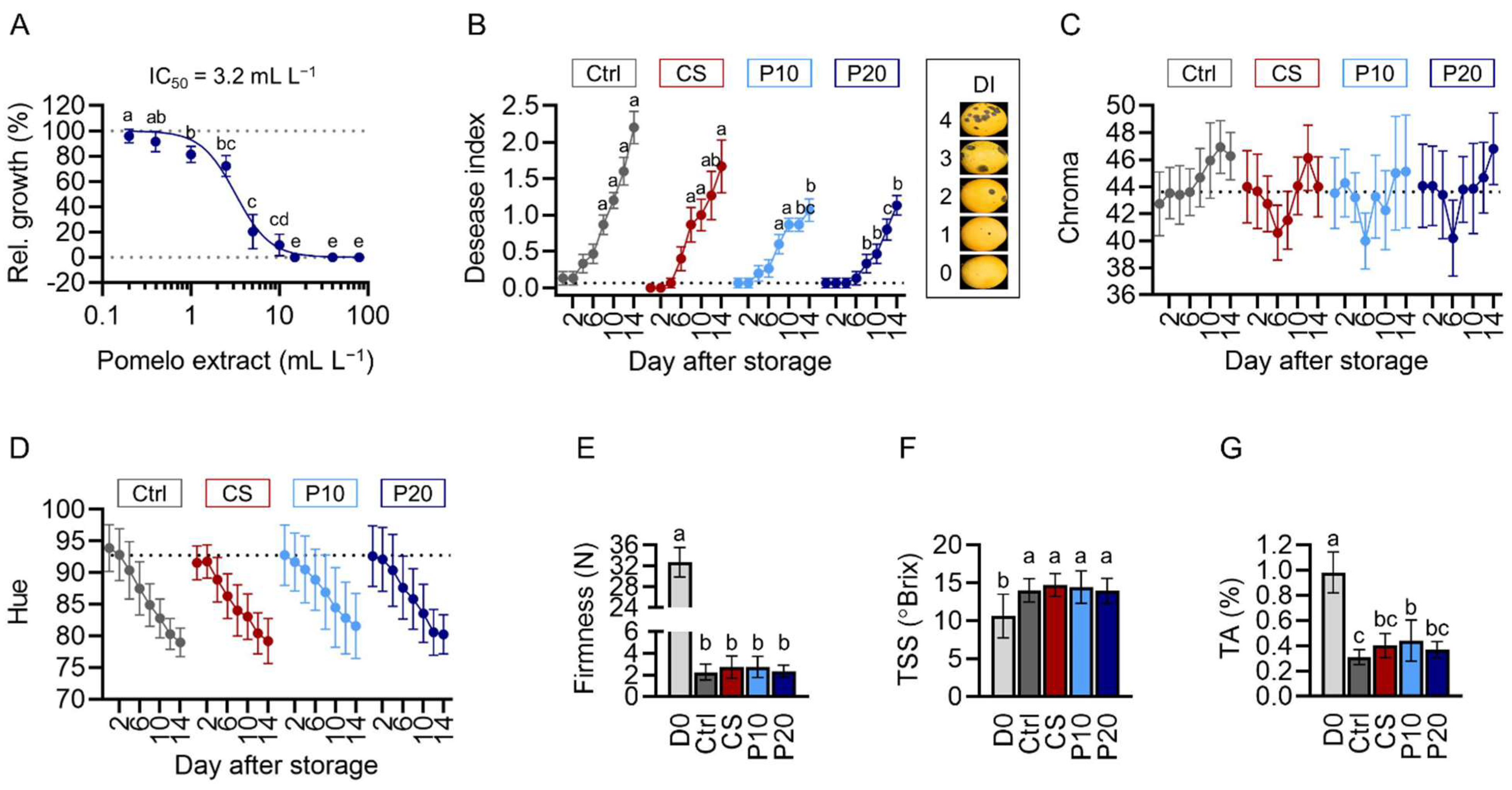
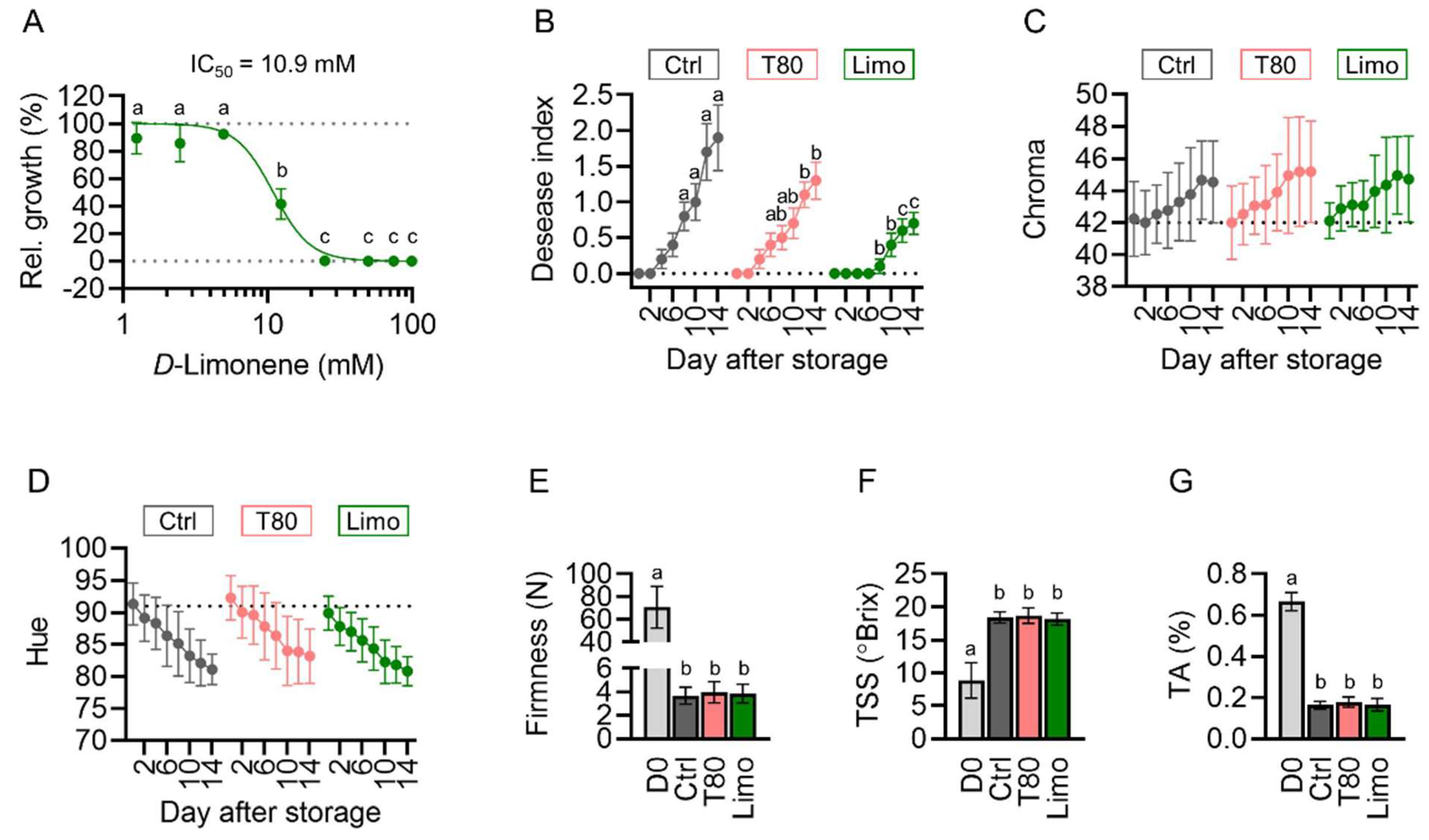
2.2. Pomelo Extract Inhibits C. gloeosporioides In Vitro and in ‘Keitt’ Mango
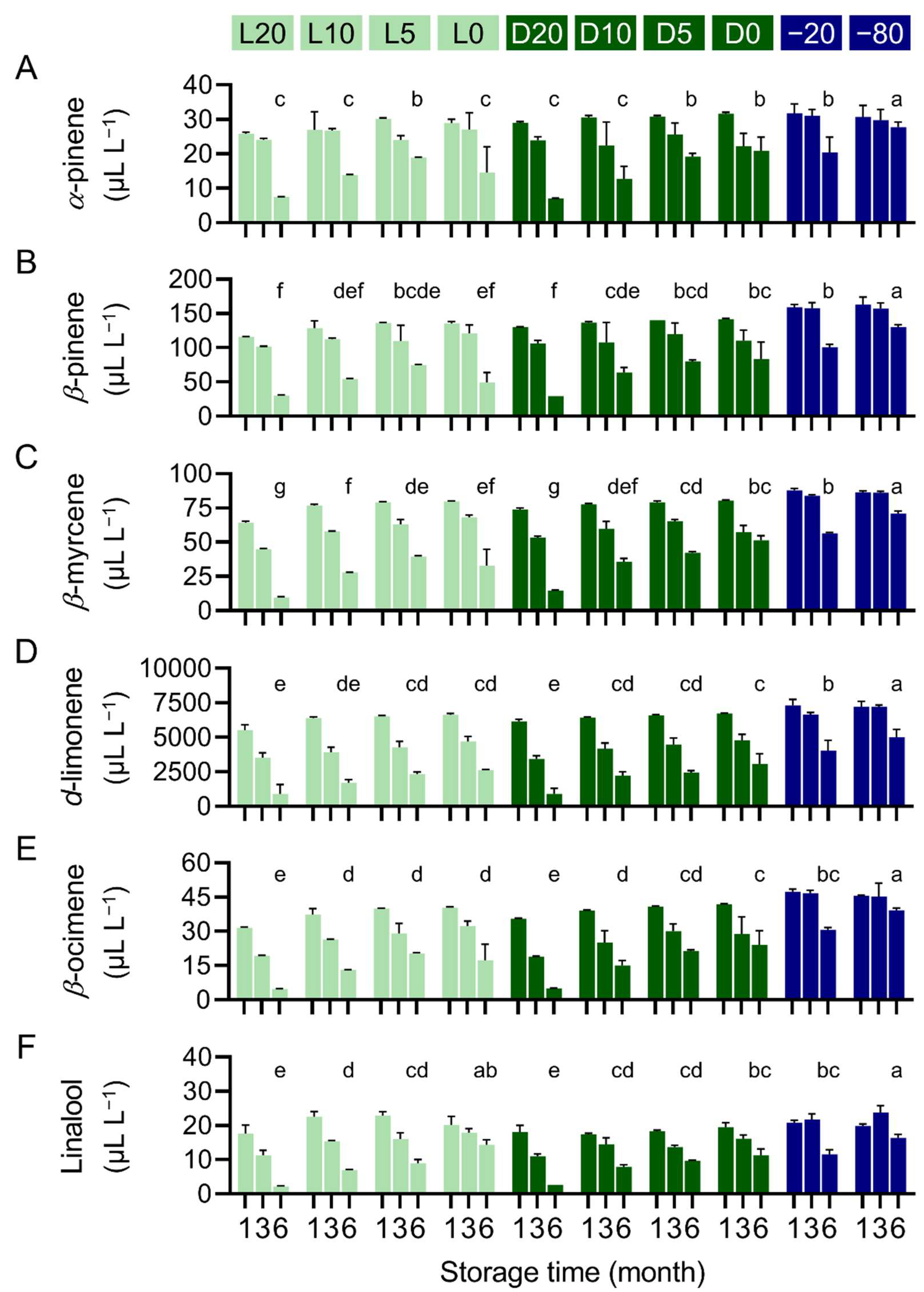
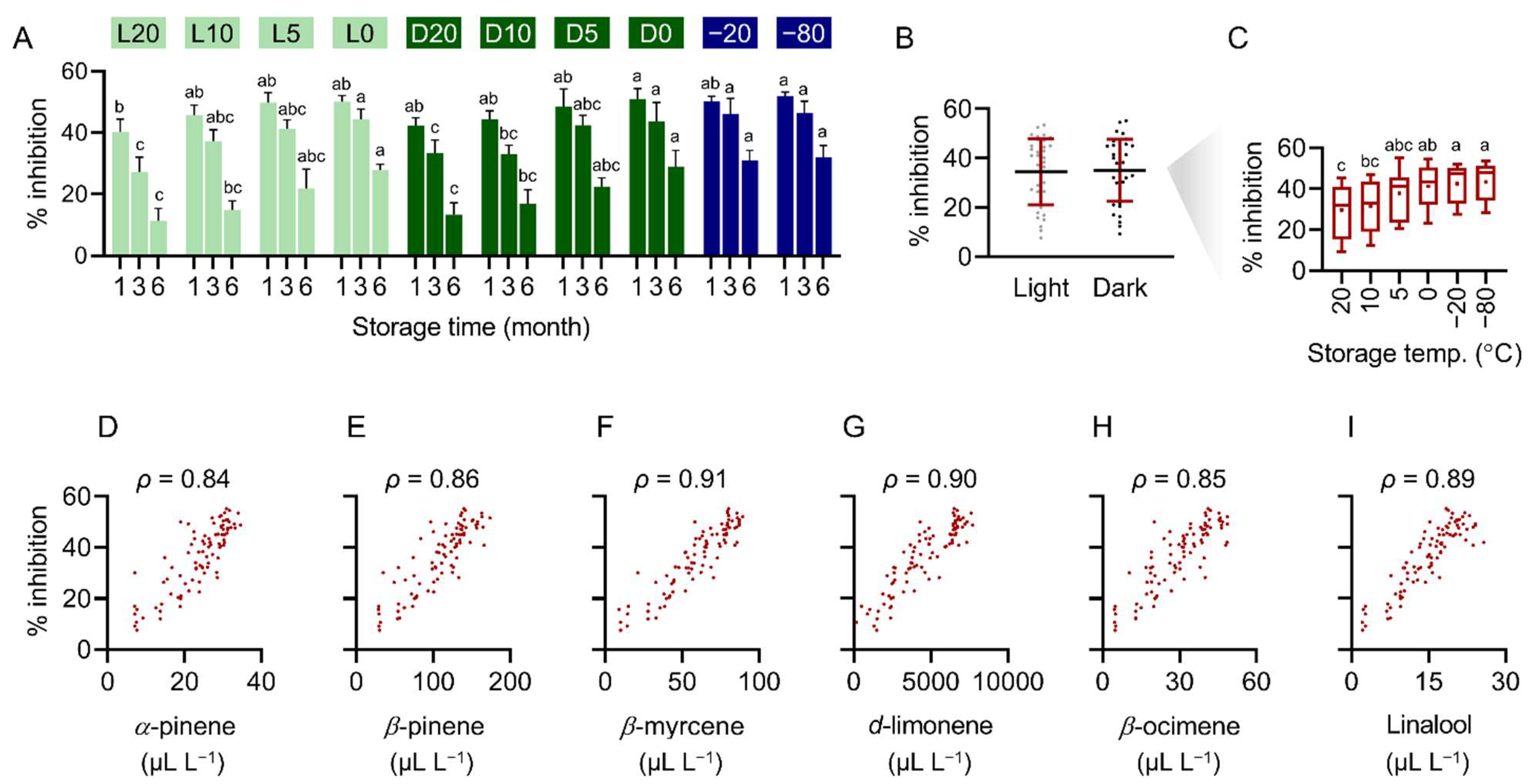
2.3. Change in Main Constituents of Pomelo Extract Stored under Different Conditions
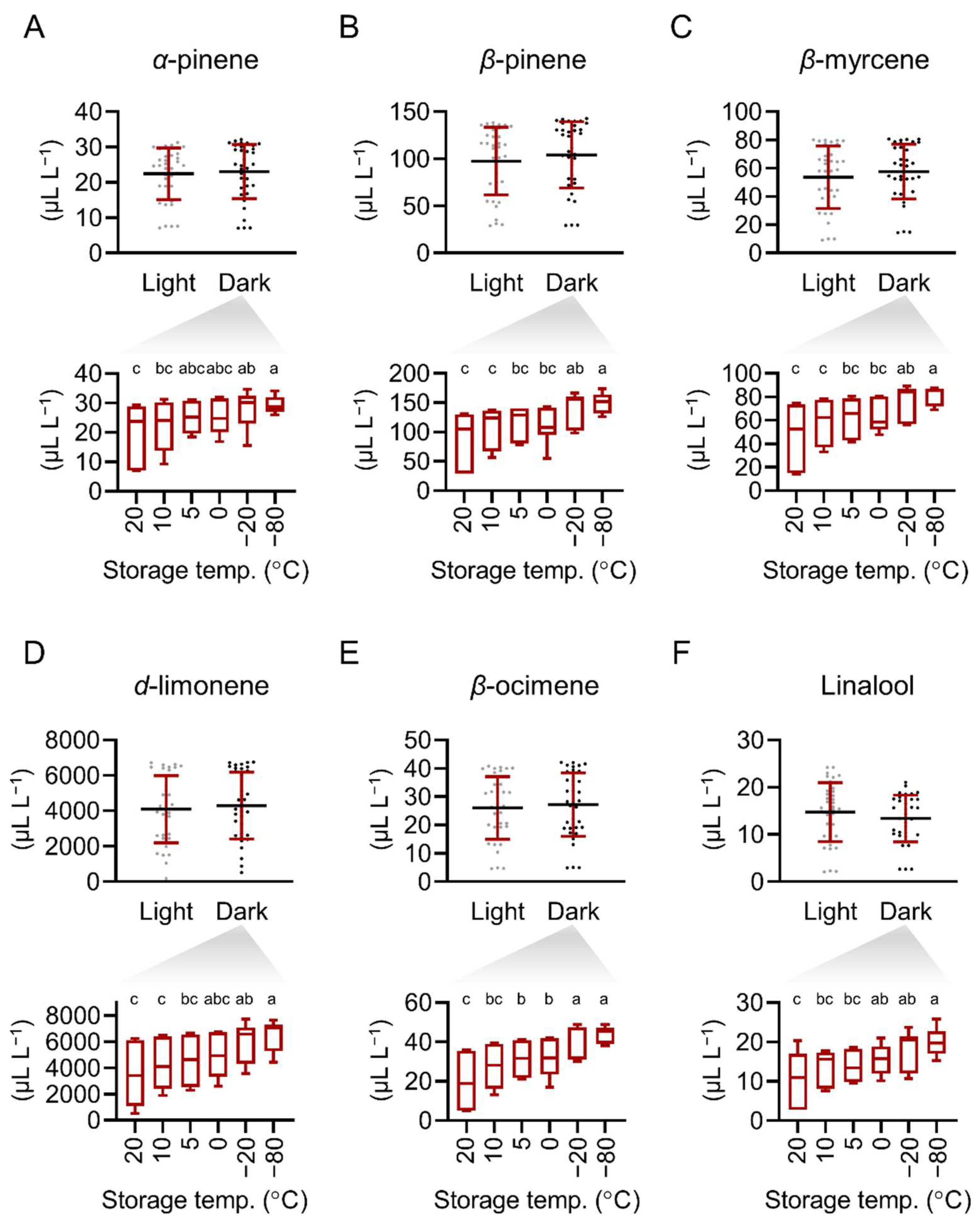
2.4. Refrigeration and Freeze Storage Retain the Main Constituents in Pomelo Extract
2.5. Antifungal Activity of Pomelo Extract Correlates to the Abundance of Main Constituents
2.6. D-Limonene Acts as the Key Component against Mango Anthracnose
3. Discussion
4. Material and Methods
4.1. Plant Materials
4.2. Preparation and Preservation Tests of Pomelo Extract
4.3. Gas Chromatography-Mass Spectrometry Analysis
4.4. Isolation of Colletotrichum gloeosporioides and In Vitro Fungicidal Assay
4.5. Preparation of Chitosan Coating Incorporated with Pomelo Extract and d-limonene Emulsion
4.6. Coating Treatment and Storage Experiments of Mango
4.7. Determination of Disease Index, Firmness, Total Soluble Solids, and Titratable Acids
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Xiao, L.; Ye, F.; Zhou, Y.; Zhao, G. Utilization of Pomelo Peels to Manufacture Value-Added Products: A Review. Food Chem. 2021, 351, 129247. [Google Scholar] [CrossRef] [PubMed]
- Tocmo, R.; Pena-Fronteras, J.; Calumba, K.F.; Mendoza, M.; Johnson, J.J. Valorization of Pomelo (Citrus grandis Osbeck) Peel: A Review of Current Utilization, Phytochemistry, Bioactivities, and Mechanisms of Action. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1969–2012. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, D.; Bautista-Baños, S. A Review on the Use of Essential Oils for Postharvest Decay Control and Maintenance of Fruit Quality during Storage. Crop Prot. 2014, 64, 27–37. [Google Scholar] [CrossRef]
- Lima, N.B.; Batista, M.V.d.A.; De Morais, M.A.; Barbosa, M.A.G.; Michereff, S.J.; Hyde, K.D.; Câmara, M.P.S. Five Colletotrichum Species are Responsible for Mango Anthracnose in Northeastern Brazil. Fungal Divers. 2013, 61, 75–88. [Google Scholar] [CrossRef]
- Swart, S.H.; Serfontein, J.J.; Swart, G.; Labuschagne, C. Chemical Control of Post-harvest Diseases of Mango: The Effect of Fludioxonil and Prochloraz on Soft Brown Rot, Stem-End Rot and Anthracnose. Acta Hortic. 2009, 820, 503–510. [Google Scholar] [CrossRef]
- Henriod, R.; Diczbalis, Y.; Sole, D.; Stice, K.N.; Tora, L. Investigation into Various Fungicides and Alternative Solutions for Controlling Postharvest Diseases in Papaya Fruit. Acta Hortic. 2016, 1111, 113–119. [Google Scholar] [CrossRef]
- Shimshoni, J.A.; Bommuraj, V.; Chen, Y.; Sperling, R.; Barel, S.; Feygenberg, O.; Maurer, D.; Alkan, N. Postharvest Fungicide for Avocado Fruits: Antifungal Efficacy and Peel to Pulp Distribution Kinetics. Foods 2020, 9, 124. [Google Scholar] [CrossRef] [Green Version]
- De la Rosa-García, S.C.; Martínez-Torres, P.; Gómez-Cornelio, S.; Corral-Aguado, M.A.; Quintana, P.; Gómez-Ortíz, M. Antifungal activity of ZnO and MgO nanomaterials and their mixtures against Colletotrichum gloeosporioides strains from Tropical Fruit. J. Nanomater. 2018, 2018, 3498527. [Google Scholar] [CrossRef] [Green Version]
- Meena, M.; Pilania, S.; Pal, A.; Mandhania, S.; Bhushan, B.; Kumar, S.; Gohari, G.; Saharan, V. Cu-chitosan Nano-net Improves Keeping Quality of Tomato by Modulating Physio-Biochemical Responses. Sci. Rep. 2020, 10, 21914. [Google Scholar] [CrossRef]
- Marques, K.M.; Galati, V.C.; Fernandes, J.D.R.; Guimarães, J.E.R.; Silva, J.P.; Mattiuz, B.H.; Mattiuz, C.F.M. Use of Chitosan for the Control of Postharvest Anthracnose and Quality in Avocados. Acta Hortic. 2016, 1120, 225–232. [Google Scholar] [CrossRef]
- Sarkhosh, A.; Schaffer, B.; Vargas, A.I.; Palmateer, A.J.; Lopez, P.; Soleymani, A.; Farzaneh, M. Antifungal Activity of Five Plant-Extracted Essential Oils against Anthracnose in Papaya Fruit. Biol. Agric. Hortic. 2018, 34, 18–26. [Google Scholar] [CrossRef]
- Dias, B.L.; Costa, P.F.; Dakin, M.S.; de Souza Carlos Mou rao, D.; Dias, F.R.; de Sousa, R.R.; Ped ro Raym undo Argiielles, O.; Talita Pereira de Souza, F.; Fabricio, S.C.; Gil Rod rigues Dos, S. Control of Papaya Fruits Anthracnose by Essential Oils of Medicinal Plants Associated to Different Coatings. J. Med. Plants Res. 2020, 14, 239–246. [Google Scholar] [CrossRef]
- Ali, A.; Wee Pheng, T.; Mustafa, M.A. Application of Lemongrass Oil in Vapour Phase for the Effective Control of Anthracnose of ‘Sekaki’ Papaya. J. Appl. Microbiol. 2015, 118, 1456–1464. [Google Scholar] [CrossRef] [PubMed]
- Chillet, M.; Minier, J.; Ducrog, M.; Meile, J.C. Postharvest Treatment of Mango: Potential Use of Essential Oil with Thymol to Control Anthracnose Development. Fruits 2018, 73, 153–157. [Google Scholar] [CrossRef]
- Esquivel-Chávez, F.; Colín-Chávez, C.; Virgen-Ortiz, J.J.; Martínez-Téllez, M.A.; Avena-Bustillos, R.J.; Peña-Madrigal, G.; Miranda-Ackermanf, M.A. Control of Mango Decay Using Antifungal Sachets Containing of Thyme Oil/Modified Starch/Agave Fructan Microcapsules. Future Foods 2021, 3, 100008. [Google Scholar] [CrossRef]
- Correa-Pacheco, Z.N.; Bautista-Baños, S.; Valle-Marquina, M.Á.; Hernández-López, M. The Effect of Nanostructured Chitosan and Chitosan-Thyme Essential Oil Coatings on Colletotrichum gloeosporioides Growth in vitro and on cv Hass avocado and Fruit Quality. J. Phytopathol. 2017, 165, 297–305. [Google Scholar] [CrossRef]
- Chen, G.-W.; Lin, Y.-H.; Lin, C.-H.; Jen, H.-C. Antibacterial Activity of Emulsified Pomelo (Citrus grandis Osbeck) Peel Oil and Water-Soluble Chitosan on Staphylococcus aureus and Escherichia coli. Molecules 2018, 23, 840. [Google Scholar] [CrossRef] [Green Version]
- Deng, W.; Liu, K.; Cao, S.; Sun, J.; Zhong, B.; Chun, J. Chemical Composition, Antimicrobial, Antioxidant, and Antiproliferative Properties of Grapefruit Essential Oil Prepared by Molecular Distillation. Molecules 2020, 25, 217. [Google Scholar] [CrossRef] [Green Version]
- Abdel-Aziz, M.M.; Emam, T.M.; Elsherbiny, E.A. Effects of Mandarin (Citrus reticulata) Peel Essential Oil as a Natural Antibiofilm Agent against Aspergillus niger in Onion Bulbs. Postharvest Biol. Technol. 2019, 156, 110959. [Google Scholar] [CrossRef]
- Bhandari, D.P.; Poudel, D.K.; Satyal, P.; Khadayat, K.; Dhami, S.; Aryal, D.; Chaudhary, P.; Ghimire, A.; Parajuli, N. Volatile Compounds and Antioxidant and Antimicrobial Activities of Selected Citrus Essential Oils Originated from Nepal. Molecules 2021, 26, 6683. [Google Scholar] [CrossRef]
- Hosni, K.; Zahed, N.; Chrif, R.; Abid, I.; Medfei, W.; Kallel, M.; Brahim, N.B.; Sebei, H. Composition of Peel Essential Oils from Four Selected Tunisian Citrus Species: Evidence for the Genotypic Influence. Food Chem. 2010, 123, 1098–1104. [Google Scholar] [CrossRef]
- Regnier, T.; du Plooy, W.; Combrinck, S.; Botha, B. Fungitoxicity of Lippia scaberrima essential oil and selected terpenoid components on two mango postharvest spoilage pathogens. Postharvest Biol. Technol. 2008, 48, 254–258. [Google Scholar] [CrossRef]
- Danh, L.T.; Giao, B.T.; Duong, C.T.; Nga, N.T.T.; Tien, D.T.K.; Tuan, N.T.; Huong, B.T.C.; Nhan, T.C.; Trang, D.T.X. Use of Essential Oils for the Control of Anthracnose Disease Caused by Colletotrichum acutatum on Post-Harvest Mangoes of Cat Hoa Loc Variety. Membranes 2021, 11, 719. [Google Scholar] [CrossRef] [PubMed]
- Quintana-Rodriguez, E.; Rivera-Macias, L.E.; Adame-Alvarez, R.M.; Torres, J.M.; Heil, M. Shared Weapons in Fungus-fungus and Fungus-plant Interactions? Volatile Organic Compounds of Plant or Fungal Origin Exert Direct Antifungal Activity in vitro. Fungal Ecol. 2018, 33, 115–121. [Google Scholar] [CrossRef]
- Feng, J.; Wang, R.; Chen, Z.; Zhang, S.; Yuan, S.; Cao, H.; Jafari, S.M.; Yang, W. Formulation Optimization of D-limonene-loaded Nanoemulsions as a Natural and Efficient Biopesticide. Colloids Surf. A Physicochem. Eng. Asp. 2020, 596, 124746. [Google Scholar] [CrossRef]
- Scariot, F.J.; Foresti, L.; Delamare, A.P.L.; Echeverrigaray, S. Activity of Monoterpenoids on the in vitro Growth of two Colletotrichum species and the Mode of Action on C. acutatum. Pestic. Biochem. Physiol. 2020, 170, 104698. [Google Scholar] [CrossRef]
- Hong, J.K.; Yang, H.J.; Jung, H.; Yoon, D.J.; Sang, M.K.; Jeun, Y.C. Application of Volatile Antifungal Plant Essential Oils for Controlling Pepper Fruit Anthracnose by Colletotrichum gloeosporioides. Plant Pathol. J. 2015, 31, 269–277. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Liu, T.; Zhou, C.; Huang, Y.; Liu, X.; Yuan, H. Antifungal Activity of Essential Oils from Three Artemisia Species Against Colletotrichum gloeosporioides of Mango. Antibiotics 2021, 10, 1331. [Google Scholar] [CrossRef]
- Grande Tovar, C.D.; Delgado-Ospina, J.; Navia Porras, D.P.; Peralta-Ruiz, Y.; Cordero, A.P.; Castro, J.I.; Chaur Valencia, M.N.; Mina, J.H.; Chaves López, C. Colletotrichum gloesporioides Inhibition in situ by Chitosan-Ruta graveolens Essential Oil Coatings: Effect on Microbiological, Physicochemical, and Organoleptic Properties of Guava (Psidium guajava L.) during Room Temperature Storage. Biomolecules 2019, 9, 399. [Google Scholar] [CrossRef] [Green Version]
- Peralta-Ruizm, Y.; Grande Tovar, C.; Sinning-Mangonez, A.; Bermont, D.; Pérez Cordero, A.; Paparella, A.; Chaves-López, C. Colletotrichum gloesporioides Inhibition Using Chitosan-Ruta graveolens L Essential Oil Coatings: Studies In Vitro and In Situ on Carica papaya Fruit. Int. J. Food Microbiol. 2020, 326, 108649. [Google Scholar] [CrossRef]
- Landi, L.; Peralta-Ruiz, Y.; Chaves-López, C.; Romanazzi, G. Chitosan Coating Enriched with Ruta graveolens L. Essential Oil Reduces Postharvest Anthracnose of Papaya (Carica papaya L.) and Modulates Defense-Related Gene Expression. Front. Plant Sci. 2021, 12, 765806. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, J.E.; Rossi, D.C.P.; Jabes, D.L.; Barbosa, D.A.; Cunha, F.F.M.; Nunes, L.R.; Arruda, D.C.; Pelleschi Taborda, C. In Vitro and In Vivo Inhibitory Activity of Limonene against Different Isolates of Candida spp. J. Fungi 2020, 6, 183. [Google Scholar] [CrossRef]
- Yu, H.; Lin, Z.-X.; Xiang, W.-L.; Huang, M.; Tang, J.; Lu, Y.; Zhao, Q.-H.; Zhang, Q.; Rao, Y.; Liu, L. Antifungal Activity and Mechanism of D-Limonene against Foodborne Opportunistic Pathogen Candida tropicalis. LWT Food Sci. Technol. 2022, 159, 113144. [Google Scholar] [CrossRef]
- Silva, R.R.; da Silva, A.C.; Rodella, R.A.; Marques, M.O.M.; Zanuncio, A.J.V.; Soares, M.A.; Serrão, J.E.; Zanuncio, J.C.; Furtado, E.L. Limonene, a Chemical Compound Related to the Resistance of Eucalyptus Species to Austropuccinia psidii. Plant Dis. 2020, 104, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Umagiliyage, A.L.; Becerra-Mora, N.; Kohli, P.; Fisher, D.J.; Choudhary, R. Antimicrobial Efficacy of Liposomes Containing D-Limonene and its Effect on the Storage Life of Blueberries. Postharvest Biol. Technol. 2017, 128, 130–137. [Google Scholar] [CrossRef]
- Du Plooy, W.; Regnier, T.; Combrinck, S. Essential Oil Amended Coatings as Alternatives to Synthetic Fungicides in Citrus Postharvest Management. Postharvest Biol. Technol. 2009, 53, 117–122. [Google Scholar] [CrossRef]
- Da Silva, A.C.R.; Lopes, P.M.; Azevedo, M.M.B.D.; Costa, D.C.M.; Alviano, C.S.; Alviano, D.S. Biological Activities of α-Pinene and β-Pinene Enantiomers. Molecules 2012, 17, 6305–6316. [Google Scholar] [CrossRef] [Green Version]
- Guimarães, A.C.; Meireles, L.M.; Lemos, M.F.; Guimarães, M.C.C.; Endringer, D.C.; Fronza, M.; Scherer, R. Antibacterial Activity of Terpenes and Terpenoids Present in Essential Oils. Molecules 2019, 24, 2471. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Zhao, C.; Tian, G.; Lu, C.; Zhao, S.; Bao, Y.; McClements, D.J.; Xiao, H.; Zheng, J. Effects of Preheating and Storage Temperatures on Aroma Profile and Physical Properties of Citrus-Oil Emulsions. J. Agric. Food Chem. 2017, 65, 7781–7789. [Google Scholar] [CrossRef]
- Zahi, M.R.; Wan, P.; Liang, H.; Yuan, Q. Formation and Stability of D-Limonene Organogel-Based Nanoemulsion Prepared by a High-Pressure Homogenizer. J. Agric. Food Chem. 2014, 62, 12563–12569. [Google Scholar] [CrossRef]
- He, F.; Qian, Y.L.; Qian, M.C. Flavor and Chiral Stability of Lemon-Flavored Hard Tea during Storage. Food Chem. 2018, 239, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Van den Dool, H.; Kratz, P.D. A Generalization of the Retention Index System Including Linear Temperature Programmed Gas-Liquid Partition Chromatography. J. Chromatogr. A 1963, 11, 463–471. [Google Scholar] [CrossRef]
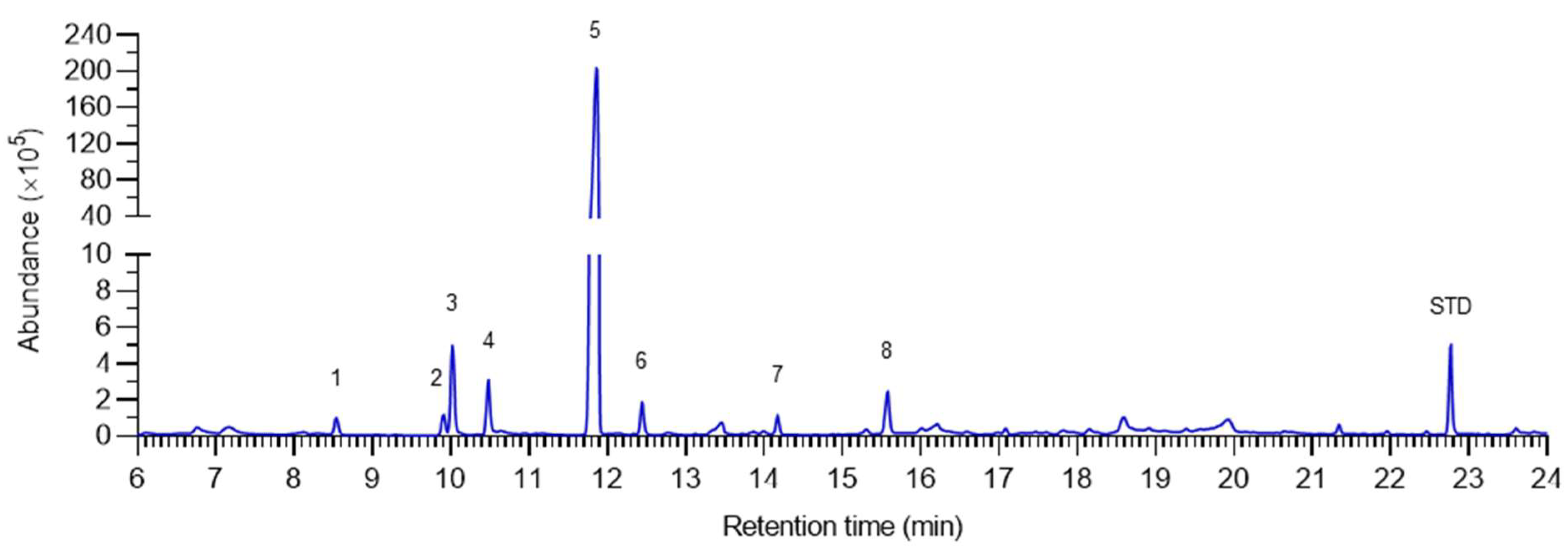
| Peak | Compound | RT | Abundance | Conc. | RI a |
|---|---|---|---|---|---|
| (min) | (% total) | (μL L−1) | |||
| 1 | α-pinene | 8.54 | 0.37 | 36.0 | 931 |
| 2 | Sabinene | 9.91 | 0.37 | 36.3 | 973 |
| 3 | β-pinene | 10.03 | 1.79 | 175.3 | 977 |
| 4 | β-myrcene | 10.48 | 0.91 | 89.2 | 991 |
| 5 | d-limonene | 11.88 | 76.11 | 7445.9 | 1032 |
| 6 | β-ocimene | 12.45 | 0.51 | 49.5 | 1049 |
| 7 | Linalool | 14.17 | 0.31 | 30.8 | 1101 |
| 8 | DDMP * | 15.58 | 0.78 | 76.5 | 1146 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, Y.-J.; Wu, Y.-J.; Lee, F.-W.; Ou, L.-Y.; Chen, C.-N.; Chu, Y.-Y.; Kuan, Y.-C. Impact of Storage Condition on Chemical Composition and Antifungal Activity of Pomelo Extract against Colletotrichum gloeosporioides and Anthracnose in Post-harvest Mango. Plants 2022, 11, 2064. https://doi.org/10.3390/plants11152064
Cheng Y-J, Wu Y-J, Lee F-W, Ou L-Y, Chen C-N, Chu Y-Y, Kuan Y-C. Impact of Storage Condition on Chemical Composition and Antifungal Activity of Pomelo Extract against Colletotrichum gloeosporioides and Anthracnose in Post-harvest Mango. Plants. 2022; 11(15):2064. https://doi.org/10.3390/plants11152064
Chicago/Turabian StyleCheng, Yu-Jung, Ying-Jou Wu, Fang-Wei Lee, Ling-Yi Ou, Chi-Nan Chen, Yu-Ying Chu, and Yen-Chou Kuan. 2022. "Impact of Storage Condition on Chemical Composition and Antifungal Activity of Pomelo Extract against Colletotrichum gloeosporioides and Anthracnose in Post-harvest Mango" Plants 11, no. 15: 2064. https://doi.org/10.3390/plants11152064
APA StyleCheng, Y.-J., Wu, Y.-J., Lee, F.-W., Ou, L.-Y., Chen, C.-N., Chu, Y.-Y., & Kuan, Y.-C. (2022). Impact of Storage Condition on Chemical Composition and Antifungal Activity of Pomelo Extract against Colletotrichum gloeosporioides and Anthracnose in Post-harvest Mango. Plants, 11(15), 2064. https://doi.org/10.3390/plants11152064






