Insecticidal Activity of Four Essential Oils Extracted from Chilean Patagonian Plants as Potential Organic Pesticides
Abstract
:1. Introduction
2. Results
2.1. Chemical Characterization of Essential Oils
2.2. Insecticidal Activity
2.2.1. Insecticidal Activity against Houseflies
2.2.2. Insecticidal Activity against Moths
2.2.3. Insecticidal Activity against Mosquitoes
3. Discussion
4. Materials and Methods
4.1. Plant Collection
4.2. Isolation of Essential Oils
4.3. Essential Oils Chemical Characterization
4.4. Insecticidal Activity
4.4.1. Insecticidal Activity against Houseflies
4.4.2. Insecticidal Activity against Moths
4.4.3. Insecticidal Activity against Mosquitoes
4.5. Data Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Molares, S.; Ladio, A. Mapuche perceptions and conservation of Andean. Nothofagus forests and their medicinal plants: A case study from a rural community in Patagonia, Argentina. Biodivers. Conserv. 2012, 21, 1079–1093. [Google Scholar] [CrossRef]
- Molares, S.; Ladio, A. Medicinal plants in the cultural landscape of a Mapuche-Tehuelche community in arid Argentine Patagonia: An eco-sensorial approach. J. Ethnobiol. Ethnomed. 2014, 10, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Martínez Crovetto, R. Apuntes sobre la vegetación de los alrededores del Lago Cholila. Publicación Técnica De La Fac. De Cienc. Agrar. 1980, 1, 1–22. [Google Scholar]
- Silva, F.; Ullrich, T.; Hartman, P.; Medina, H.; Moraga, L.; Saini, G. Plantas Medicinales de la región de Aysen-Chile. B Lat. Caribe Pl. 2004, 3, 36–45. [Google Scholar]
- Montes, M.; Wilkomirsky, T. Medicina Tradicional Chilena; Editorial de la Universidad de Concepción: Concepción, Chile, 1987; pp. 72–73. [Google Scholar]
- González, S.B.; Houghton, P.J.; Hoult, J.R.S. The activity against leukocyte eicosanoid generation of essential oil and polar fractions of Adesmia boronioides Hook. f. Phytother. Res. 2003, 3, 290–293. [Google Scholar] [CrossRef] [PubMed]
- González, S.B.; Ladio, A.H.; Gastaldi, B.; Silva Sofrás, F.M.; Mazzoni, A.; Sánchez, G. Paramela (Adesmia boronioides Hook.f.): From popular uses to commercialization. In Ethnobotany, 1st ed.; Martinez, J.L., Muñoz-Acevedo, A., Rai, M., Eds.; CRC Press: Boca Raton, FL, USA, 2018; pp. 89–104. [Google Scholar]
- Mølgaard, P.; Holler, J.G.; Asar, B.; Liberna, I.; Rosenbæk, L.B.; Jebjerg, C.P.; Jørgensen Jeanette, L.; Guzman, A.; Adsersen, A.; Simonsen, H.T. Antimicrobial evaluation of Huilliche plant medicine used to treat wounds. J. Ethnopharmacol. 2011, 138, 219–227. [Google Scholar] [CrossRef]
- Rodríguez, R.; Matthei, O.; Quezada, M. Flora Arbórea de Chile; Editorial de la Universidad de Concepción: Concepción, Chile, 1983; p. 408. [Google Scholar]
- Bittner, M.; Aguilera, M.A.; Hernández, V.; Arbert, C.; Becerra, J.; Casanueva, M.E. Fungistatic activity of essential oils extracted from Peumus boldus Mol., Laureliopsis philippiana (Looser) Schodde and Laurelia sempervirens (Ruiz & Pav.) Tul. (Chilean Monimiaceae). Chil. J. Agric. Res. 2009, 69, 30–37. [Google Scholar] [CrossRef] [Green Version]
- Toledo, D.; Mutis, A.; Hormazabal, E.; Palma, R.; Parada, M.; Scheuermann, E.; Quiroz, A. Chemical composition and antibacterial activity of Laureliopsis philippiana (Looser) essential oil. BLACPMA 2014, 13, 117–125. [Google Scholar]
- Herrera-Rodríguez, C.; Ramírez-Mendoza, C.; Becerra-Morales, I.; Silva-Aguayo, G.; Urbina-Parra, A.; Figueroa-Cares, I.; Martínez-Bolaños, L.; Rodríguez-Maciel, J.C.; Lagunes-Tejeda, A.; Pastene-Navarrete, E.; et al. Bioactivity of Peumus boldus Molina, Laurelia sempervirens (Ruiz & Pav.) Tul. and Laureliopsis philippiana (Looser) Schodde (Monimiaceae) essential oils against Sitophilus zeamais Motschulsky). Chil. J. Agric. Res. 2015, 75, 334–340. [Google Scholar] [CrossRef] [Green Version]
- Norambuena, C.; Silva, G.; Urbina, A.; Figueroa, I.; Rodríguez-Maciel, J.C. Insecticidal activity of Laureliopsis philippiana (Looser) Schodde (Atherospermataceae) essential oil against Sitophilus spp. (Coleoptera Curculionidae). Chil. J. Agric. Res. 2016, 76, 330–336. [Google Scholar] [CrossRef] [Green Version]
- Cordero, S.; Abello, L.; Galvez, F. Plantas Silvestres Comestibles y Medicinales de Chile y Otras Partes del Mundo, 1st ed.; Corporación Chilena de la Madera: Concepción, Chile, 2017; 292p. [Google Scholar]
- Zapata, N.; Lognay, G.; Smagghe, G. Bioactivity of essential oils from leaves and bark of Laurelia sempervirens and Drimys winteri against Acyrthosiphon pisum. Pest. Manag. 2010, 66, 1324–1331. [Google Scholar] [CrossRef]
- Tampe, J.; Espinoza, J.; Chacón-Fuentes, M.; Quiroz, A.; Rubilar, M. Evaluation of Drimys winteri (Canelo) Essential Oil as Insecticide against Acanthoscelides obtectus (Coleoptera: Bruchidae) and Aegorhinus superciliosus (Coleoptera: Curculionidae). Insects 2020, 11, 335. [Google Scholar] [CrossRef] [PubMed]
- Cruz Madariaga, G.; Lara Aguilar, A. Tipificación, Cambio de Estructura y Normas de Manejo para Ciprés de las Guaytecas (Pilgerodendron uvifera (D. Don) Florin.); en la Isla Grande de Chiloé: Castro, Chile, 1981. [Google Scholar]
- Espinoza, J.; Urzúa, A.; Tampe, J.; Parra, L.; Quiroz, A. Repellent activity of the essential oil from the heartwood of Pilgerodendron uviferum (D. Don) Florin against Aegorhinus superciliosus (Coleoptera: Curculionidae). Molecules 2016, 21, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Espinoza, J.; Urzúa, A.; Bardehle, L.; Quiroz, A.; Echeverría, J.; González-Teuber, M. Antifeedant effects of essential oil, extracts, and isolated sesquiterpenes from Pilgerodendron uviferum (D. Don) florin heartwood on red clover borer Hylastinus obscurus (Coleoptera: Curculionidae). Molecules 2018, 23, 1282. [Google Scholar] [CrossRef] [Green Version]
- Di Giovanni, F.; Wilke, A.B.B.; Beier, J.C.; Pombi, M.; Mendoza-Roldan, J.A.; Desneux, N.; Canale, A.; Lucchi, A.; Dantas-Torres, F.; Otranto, D.; et al. Parasitic strategies of arthropods of medical and veterinary importance. Entomol. Gen. 2021, 41, 511–522. [Google Scholar] [CrossRef]
- Bass, C.; Denholm, I.; Williamson, M.S.; Nauen, R. The global status of insect resistance to neonicotinoid insecticides. Pestic Biochem. Phys. 2015, 121, 78–87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benelli, G.; Wilke, A.B.; Bloomquist, J.R.; Desneux, N.; Beier, J.C. Overexposing mosquitoes to insecticides under global warming: A public health concern? Sci. Total Environ. 2021, 762, 143069. [Google Scholar] [CrossRef]
- Yang, X.; Wei, X.; Yang, J.; Du, T.; Yin, C.; Fu, B.; Huang, M.; Liang, J.; Gong, P.; Liu, S.; et al. Epitranscriptomic regulation of insecticide resistance. Sci. Adv. 2021, 7, eabe5903. [Google Scholar] [CrossRef]
- Malizia, R.A.; Cardell, D.A.; Molli, J.S.; González, S.; Guerra, P.E.; Grau, R.J. Volatile constituents of leaf oils from the Cupressacea family: Part II. Austrocedrus chilensis, Fitzroya cupressoides and Pilgerodendron uviferum species growing in Argentina. J. Essent. 2000, 12, 233–237. [Google Scholar] [CrossRef]
- Oyarzún, M.L.; Garbarino, J.A. Sesquiterpenoids from Pilgerodendron uvífera. Phytochemistry 1988, 27, 1121–1123. [Google Scholar] [CrossRef]
- Madrid, A.; Godoy, P.; González, S.; Zaror, L.; Moller, A.; Werner, E.; Cuellar, M.; Villena, J.; Montenegro, I. Chemical characterization and anti-oomycete activity of Laureliopsis philippianna essential oils against Saprolegnia parasitica and S. australis. Molecules 2015, 20, 8033–8047. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barrero, A.F.; Herrador, M.M.; Arteaga, P.; Lara, A.; Cortes, M. Chemical composition of the essential oil from Drimys winteri Forst. wood. J. Essent. 2000, 12, 685–688. [Google Scholar] [CrossRef]
- Monsalvez, M.; Zapata, N.; Vargas, M.; Berti, M.; Bittner, M.; Hernández, V. Antifungal effects of n-hexane extract and essential oil of Drimys winteri bark against Take-All disease. Ind. Crop. Prod. 2010, 31, 239–244. [Google Scholar] [CrossRef]
- Muñoz, O.; Christen, P.; Cretton, S.; Barrero, A.F.; Lara, A.; Herrador, M.M. Comparison of the essential oils of leaves and stem bark from two different populations of Drimys winteri a Chilean herbal medicine. Nat. Prod. Commun. 2011, 6, 879–882. [Google Scholar] [CrossRef] [Green Version]
- Verdeguer, M.; García-Rellán, D.; Boira, H.; Pérez, E.; Gandolfo, S.; Blázquez, M.A. Herbicidal Activity of Peumus boldus and Drimys winterii Essential Oils from Chile. Molecules 2011, 16, 403–411. [Google Scholar] [CrossRef] [Green Version]
- González, S.B.; Bandoni, A.L.; van Baren, C.; Lira, P.D.L.; Cerda-García-Rojas, C.M.; Joseph-Nathan, P. Structure, conformation and absolute configuration of novel bisnorsesquiterpenes from the Adesmia boronioides essential oil. Tetrahedron 2002, 58, 3065–3071. [Google Scholar] [CrossRef]
- González, S.B.; Bandoni, A.L.; Van Baren, C.; Di Leo Lira, P.; Cerda-García-Rojas, C.M.; Joseph-Nathan, P. The Essential Oil of the Aerial Parts of Adesmia boronioides Hook. F. J. Essent. Oil Res. 2004, 16, 513–516. [Google Scholar] [CrossRef]
- Pavela, R.; Benelli, G. Essential oils as ecofriendly biopesticides? Challenges and constraints. Trends Plant. Sci. 2016, 21, 1000–1007. [Google Scholar] [CrossRef]
- Pavela, R. Acute toxicity and synergistic and antagonistic effects of the aromatic compounds of some essential oils against Culex quinquefasciatus Say larvae. Parasitol. Res. 2015, 114, 3835–3853. [Google Scholar] [CrossRef]
- Pavela, R. Acute, synergistic and antagonistic effects of some aromatic compounds on the Spodoptera littoralis Boisd. (Lep., Noctuidae) larvae. Ind. Crop. Prod. 2014, 60, 247–258. [Google Scholar] [CrossRef]
- Stepanycheva, E.; Petrova, M.; Chermenskaya, T.; Pavela, R. Fumigant effect of essential oils on mortality and fertility of thrips Frankliniella occidentalis Perg. Environ. Sci. Pollut. Res. 2019, 26, 30885–30892. [Google Scholar] [CrossRef] [PubMed]
- Pavela, R.; Žabka, M.; Bednář, J.; Tříska, J.; Vrchotová, N. New knowledge for yield, composition and insecticidal activity of essential oils obtained from the aerial parts or seeds of fennel (Foeniculum vulgare Mill.). Ind. Crop. Prod. 2016, 83, 275–282. [Google Scholar] [CrossRef]
- Espinoza, J.; Medina, C.; Aniñir, W.; Escobar-Bahamondes, P.; Ungerfeld, E.; Urzúa, A.; Quiroz, A. Insecticidal, Repellent and Antifeedant Activity of Essential Oils from Blepharocalyx cruckshanksii (Hook. & Arn.) Nied. Leaves and Pilgerodendron uviferum (D. Don) Florin Heartwood against Horn Flies, Haematobia irritans (Diptera: Muscidae). Molecules 2021, 26, 6936. [Google Scholar] [CrossRef] [PubMed]
- Paz, C.; Burgos, V.; Iturra, A.; Rebolledo, R.; Ortiz, L.; Baggio, R.; Cespedes-acuña, C.L. Industrial Crops & Products Assessment of insecticidal responses of extracts and compounds of Drimys winteri, Lobelia tupa, Viola portalesia and Vestia foetida against the granary weevil Sitophilus granarius. Ind. Crop. Prod. 2018, 122, 232–238. [Google Scholar] [CrossRef]
- Zapata, N.; Smagghe, G. Repellency and toxicity of essential oils from the leaves and bark of Laurelia sempervirens and Drimys winteri against Tribolium castaneum. Ind. Crop. Prod. 2010, 32, 405–410. [Google Scholar] [CrossRef]
- Govindarajan, M.; Rajeswary, M.; Benelli, G. δ-Cadinene, calarene and δ-4-carene from Kadsura heteroclita essential oil as novel larvicides against malaria, dengue and filariasis mosquitoes. Comb. Chem High. Throughput Screen 2016, 19, 565–571. [Google Scholar] [CrossRef]
- Pavela, R.; Morshedloo, M.R.; Mumivand, H.; Khorsand, G.J.; Karami, A.; Maggi, F.; Desneux, N.; Benelli, G. Phenolic monoterpene-rich essential oils from Apiaceae and Lamiaceae species: Insecticidal activity and safety evaluation on non-target earthworms. Entomol. Gen. 2020, 40, 421–435. [Google Scholar] [CrossRef]
- Sánchez-Gómez, S.; Pagán, R.; Pavela, R.; Mazzara, E.; Spinozzi, E.; Marinelli, O.; Zeppa, L.; Morshedloog, M.R.; Maggi, F.; Canale, A.; et al. Lethal and sublethal effects of essential oil-loaded zein nanocapsules on a zoonotic disease vector mosquito, and their non-target impact. Ind. Crop. Prod. 2022, 176, 114413. [Google Scholar] [CrossRef]
- Benelli, G.; Pavela, R.; Giordani, C.; Casettari, L.; Curzi, G.; Cappellacci, L.; Petrelli, R.; Maggi, F. Acute and sub-lethal toxicity of eight essential oils of commercial interest against the filariasis mosquito Culex quinquefasciatus and the housefly Musca domestica. Ind. Crop. Prod. 2018, 112, 668–680. [Google Scholar] [CrossRef]
- Benelli, G.; Pavoni, L.; Zeni, V.; Ricciardi, R.; Cosci, F.; Cacopardo, G.; Gendusa, S.; Spinozzi, E.; Petrelli, R.; Cappellacci, L.; et al. Developing a highly stable Carlina acaulis essential oil nanoemulsion for managing Lobesia botrana. Nanomaterials 2020, 10, 1867. [Google Scholar] [CrossRef]
- WHO. Report of the WHO Informal Consultation on the Evaluation and Testing of Insecticides; CTD/WHOPES/IC/96.1; WHO: Geneva, Switzerland, 1996. [Google Scholar]
- Pavela, R.; Sedlák, P. Post-application temperature as a factor influencing the insecticidal activity of essential oil from Thymus vulgaris. Ind. Crop. Prod. 2018, 113, 46–49. [Google Scholar] [CrossRef]
- Abbott, W.S. A method of computing the effectiveness of an insecticide. J. Econ. Entomol. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- Finney, D.J. Statistical logic in the monitoring of reactions to therapeutic drugs. Methods Inf. Med. 1971, 10, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Kenis, M.; Benelli, G.; Biondi, A.; Calatayud, P.A.; Day, R.; Desneux, N.; Harrison, R.D.; Kriticos, D.; Rwomushana, I.; van den Berg, J.; et al. Invasiveness, biology, ecology, and management of the fall armyworm, Spodoptera frugiperda. Entomol. Gen. 2022; in press. [Google Scholar] [CrossRef]
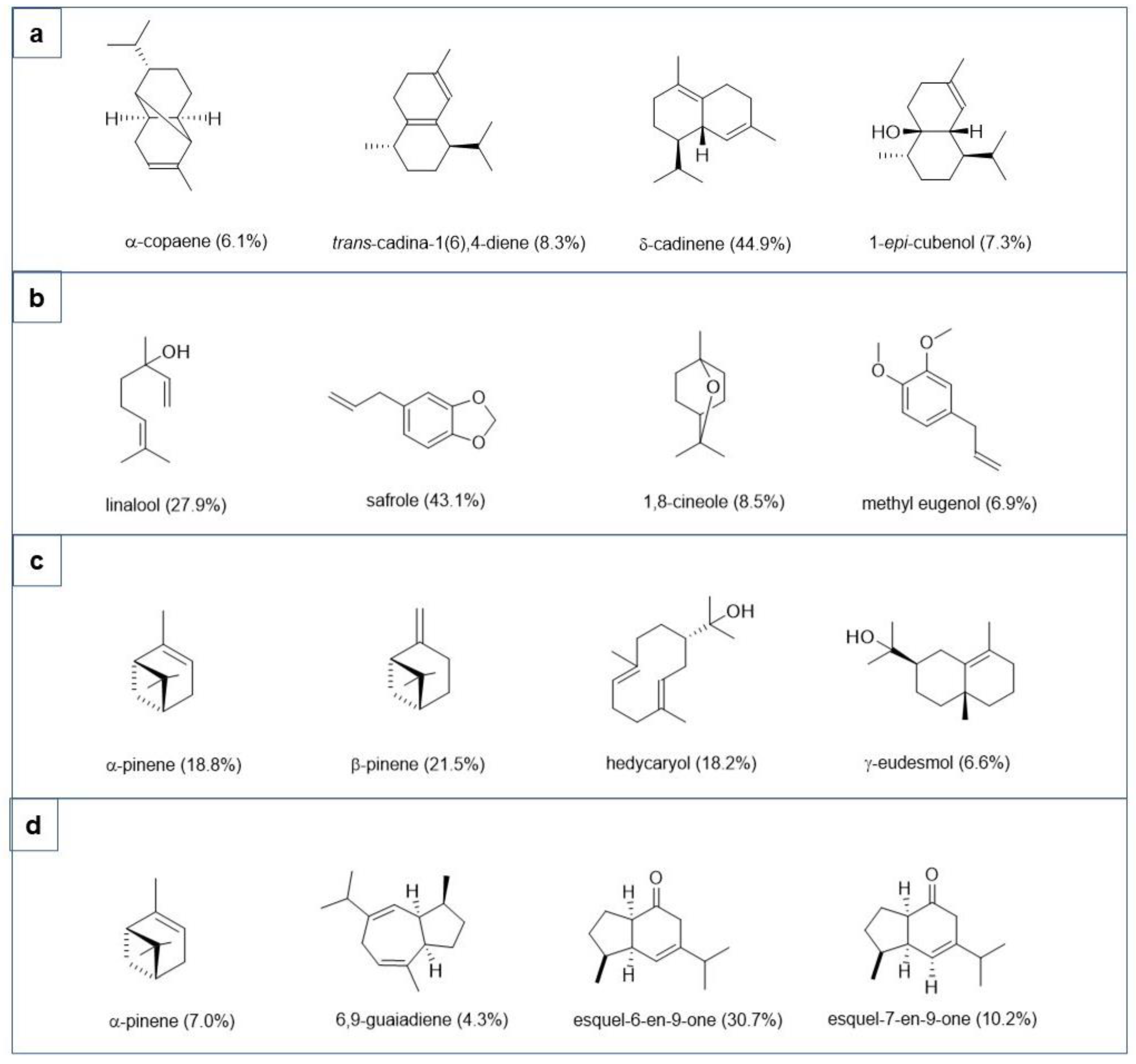
| No | Component a | RI b | RI Lit. c | Essential Oil | ID e | |||
|---|---|---|---|---|---|---|---|---|
| Ciprés % d | Tepa % | Canelo % | Paramela% | |||||
| 1 | 2-heptanone | 893 | 889 | Tr f | RI, MS | |||
| 2 | 2-heptanol | 902 | 894 | Tr | RI, MS | |||
| 3 | isobutyl isobutyrate | 912 | 908 | Tr | RI, MS | |||
| 4 | α-thujene | 921 | 924 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | RI, MS | |
| 5 | α-pinene | 926 | 932 | Tr | 1.0 ± 0.2 | 18.8 ± 2.8 | 7.0 ± 1.3 | Std, RI, MS |
| 6 | ethyl tiglate | 934 | 929 | Tr | RI, MS | |||
| 7 | Camphene | 939 | 946 | Tr | 0.5 ± 0.2 | Tr | Std, RI, MS | |
| 8 | thuja-2,4(10)-diene | 945 | 953 | 0.5 ± 0.1 | RI, MS | |||
| 9 | Benzaldehyde | 955 | 952 | Tr | Std, RI, MS | |||
| 10 | Sabinene | 966 | 969 | 0.9 ± 0.2 | 1.8 ± 0.4 | 0.1 ± 0.0 | Std, RI, MS | |
| 11 | β-pinene | 968 | 974 | 1.3 ± 0.3 | 21.5 ± 3.5 | 1.2 ± 0.3 | Std, RI, MS | |
| 12 | 3-p-menthene | 977 | 984 | Tr | RI, MS | |||
| 13 | 3-octanone | 986 | 979 | Tr | RI, MS | |||
| 14 | Myrcene | 989 | 988 | 0.4 ± 0.2 | 0.9 ± 0.2 | Std, RI, MS | ||
| 15 | 2-pentyl-furan | 990 | 990 | 0.1 ± 0.0 | RI, MS | |||
| 16 | 3-octanol | 997 | 988 | 0.1 ± 0.0 | RI, MS | |||
| 17 | α-phellandrene | 1003 | 1002 | 3.0 ± 0.6 | 0.4 ± 0.1 | 0.2 ± 0.0 | Std, RI, MS | |
| 18 | δ-3-carene | 1008 | 1008 | 0.1 ± 0.0 | 0.1 ± 0.0 | Std, RI, MS | ||
| 19 | α-terpinene | 1014 | 1014 | 0.1 ± 0.0 | 0.4 ± 0.1 | 0.2 ± 0.1 | Std, RI, MS | |
| 20 | p-cymene | 1022 | 1020 | 1.1 ± 0.2 | 0.1 ± 0.0 | 1.5 ± 0.3 | Std, RI, MS | |
| 21 | limonene | 1025 | 1024 | 0.8 ± 0.2 | 2.6 ± 0.5 | 2.1 ± 0.4 | Std, RI, MS | |
| 22 | 1,8-cineole | 1027 | 1026 | 8.5 ± 1.1 | 0.5 ± 0.1 | 0.1 ± 0.0 | Std, RI, MS | |
| 23 | (Z)-β-ocimene | 1037 | 1032 | 0.1 ± 0.0 | 0.6 ± 0.2 | Tr | Std, RI, MS | |
| 24 | benzene acetaldehyde | 1043 | 1036 | Tr | RI, MS | |||
| 25 | (E)-β-ocimene | 1047 | 1044 | 0.2 ± 0.0 | Tr | Std, RI, MS | ||
| 26 | γ-terpinene | 1055 | 1054 | 0.1 ± 0.0 | 0.7 ± 0.2 | 0.4 ± 0.1 | Std, RI, MS | |
| 27 | acetophenone | 1065 | 1059 | Tr | RI, MS | |||
| 28 | cis-linalool oxide | 1071 | 1067 | Tr | RI, MS | |||
| 29 | terpinolene | 1085 | 1086 | 0.3 ± 0.1 | 0.3 ± 0.0 | 0.1 ± 0.0 | Std, RI, MS | |
| 30 | p-cymenene | 1086 | 1089 | 0.1 ± 0.0 | RI, MS | |||
| 31 | trans-linalool oxide | 1087 | 1084 | Tr | RI, MS | |||
| 32 | 6-camphenone | 1092 | 1095 | Tr | RI, MS | |||
| 33 | 2-nonanone | 1094 | 1087 | Tr | RI, MS | |||
| 34 | linalool | 1100 | 1095 | 27.9 ± 3.1 | 2.8 ± 0.4 | 0.3 ± 0.1 | Std, RI, MS | |
| 35 | ethyl heptanoate | 1101 | 1097 | Tr | RI, MS | |||
| 36 | 2-methyl butyl-2-methyl butyrate | 1106 | 1100 | Tr | RI, MS | |||
| 37 | 1,3,8-p-menthatriene | 1109 | 1108 | Tr | RI, MS | |||
| 38 | trans-thujone | 1113 | 1112 | Tr | RI, MS | |||
| 39 | 3-methyl-3-butenyl 3-methyl butanoate | 1115 | 1112 | Tr | RI, MS | |||
| 40 | α-campholenal | 1122 | 1122 | 0.7 ± 0.2 | RI, MS | |||
| 41 | allo-ocimene | 1129 | 1128 | Tr | RI, MS | |||
| 42 | trans-pinocarveol | 1133 | 1135 | 0.2 ± 0.0 | Std, RI, MS | |||
| 43 | camphor | 1138 | 1141 | 0.1 ± 0.0 | Std, RI, MS | |||
| 44 | trans-verbenol | 1141 | 1140 | 0.1 ± 0.0 | RI, MS | |||
| 45 | 1,4-dimethyl-4-acetyl-1-cyclohexene | 1147 | 1152 | Tr | RI, MS | |||
| 46 | isobutyl hexanoate | 1154 | 1149 | Tr | RI, MS | |||
| 47 | trans-pinocamphone | 1155 | 1158 | Tr | RI, MS | |||
| 48 | pinocarvone | 1157 | 1160 | Tr | RI, MS | |||
| 49 | borneol | 1161 | 1165 | Tr | Std, RI, MS | |||
| 50 | δ-terpineol | 1164 | 1162 | Tr | RI, MS | |||
| 51 | cis-pinocamphone | 1169 | 1172 | Tr | RI, MS | |||
| 52 | ethyl benzoate | 1170 | 1169 | Tr | RI, MS | |||
| 53 | terpinen-4-ol | 1172 | 1174 | 0.2 ± 0.0 | 0.6 ± 0.2 | 1.0 ± 0.2 | Std, RI, MS | |
| 54 | p-cymen-8-ol | 1183 | 1179 | Tr | 0.1 ± 0.0 | RI, MS | ||
| 55 | α-terpineol | 1187 | 1186 | 2.1 ± 0.4 | 0.4 ± 0.1 | 0.3 ± 0.1 | Std, RI, MS | |
| 56 | myrtenal | 1190 | 1195 | 0.2 ± 0.0 | Std, RI, MS | |||
| 57 | methyl chavicol | 1196 | 1195 | Tr | RI, MS | |||
| 58 | verbenone | 1204 | 1204 | 0.1 ± 0.0 | RI, MS | |||
| 59 | trans-carveol | 1218 | 1215 | 0.1 ± 0.0 | RI, MS | |||
| 60 | carvone | 1240 | 1239 | 0.1 ± 0.0 | Std, RI, MS | |||
| 61 | bornyl acetate | 1281 | 1287 | Tr | Std, RI, MS | |||
| 62 | safrole | 1284 | 1285 | 43.1 ± 3.9 | Tr | RI, MS | ||
| 63 | theaspirane | 1290 | 1298 | Tr | RI, MS | |||
| 64 | indane derivative | 1336 | 0.2 ± 0.0 | MS | ||||
| 65 | α-cubebene | 1343 | 1345 | Tr | RI, MS | |||
| 66 | eugenol | 1355 | 1356 | 1.1 | Tr | Std, RI, MS | ||
| 67 | α-copaene | 1367 | 1374 | 6.1 ± 0.9 | Tr | RI, MS | ||
| 68 | β-bourbonene | 1376 | 1387 | Tr | RI, MS | |||
| 69 | β-elemene | 1385 | 1389 | Tr | 1.2 ± 0.3 | Std, RI, MS | ||
| 70 | α-gurjunene | 1400 | 1409 | Tr | RI, MS | |||
| 71 | methyl eugenol | 1406 | 1403 | 6.9 ± 1.1 | Tr | RI, MS | ||
| 72 | (E)-caryophyllene | 1409 | 1417 | 4.9 ± 0.8 | 0.2 ± 0.0 | 1.4 ± 0.3 | Std, RI, MS | |
| 73 | 4,8-α-epoxy-caryophyllane | 1412 | 1415 | Tr | RI, MS | |||
| 74 | α-guaiene | 1431 | 1437 | 0.2 ± 0.0 | RI, MS | |||
| 75 | 6,9-guaiadiene | 1436 | 1442 | 4.3 ± 0.6 | RI, MS | |||
| 76 | aromadendrene | 1440 | 1439 | 0.7 ± 0.2 | RI, MS | |||
| 77 | α-humulene | 1443 | 1452 | 3.9 ± 0.7 | Tr | 0.2 ± 0.0 | Std, RI, MS | |
| 78 | allo-aromadendrene | 1450 | 1458 | Tr | RI, MS | |||
| 79 | (E)-β-farnesene | 1452 | 1454 | 0.1 ± 0.0 | Std, RI, MS | |||
| 80 | sesquisabinene | 1456 | 1457 | Tr | RI, MS | |||
| 81 | trans-cadina-1(6),4-diene | 1466 | 1475 | 8.3 ± 1.2 | RI, MS | |||
| 82 | γ-muurolene | 1469 | 1478 | 0.8 ± 0.2 | RI, MS | |||
| 83 | germacrene D | 1471 | 1484 | 0.1 ± 0.0 | Tr | RI, MS | ||
| 84 | β-selinene | 1475 | 1489 | 0.1 ± 0.0 | 0.3 ± 0.1 | RI, MS | ||
| 85 | β-dihydroagarofuran | 1487 | 1496 | 1.8 ± 0.4 | RI, MS | |||
| 86 | bicyclogermacrene | 1487 | 1500 | 0.1 ± 0.0 | 2.6 ± 0.4 | RI, MS | ||
| 87 | epizonarene | 1491 | 1501 | 0.6 ± 0.2 | RI, MS | |||
| 88 | α-muurolene | 1493 | 1500 | 2.9 ± 0.6 | RI, MS | |||
| 89 | esquel-6-en-9-one | 1494 | 30.7 ± 3.1 | MS | ||||
| 90 | epishyobunone | 1502 | 1498 | 0.2 ± 0.0 | RI, MS | |||
| 91 | γ-cadinene | 1505 | 1513 | 0.1 ± 0.0 | RI, MS | |||
| 92 | trans-calamenene | 1519 | 1521 | 3.4 ± 0.6 | RI, MS | |||
| 93 | δ-cadinene | 1521 | 1522 | 44.9 ± 2.9 | 0.1 ± 0.0 | RI, MS | ||
| 94 | trans-cadina-1,4-diene | 1525 | 1533 | 1.6 ± 0.3 | RI, MS | |||
| 95 | γ-dehydro-ar-himachalene | 1533 | 1530 | 0.6 ± 0.2 | RI, MS | |||
| 96 | α-agarofuran | 1533 | 1540 | 0.6 ± 0.2 | RI, MS | |||
| 97 | α-calacorene | 1535 | 1542 | 2.4 ± 0.2 | RI, MS | |||
| 98 | furopelargone A | 1536 | 1538 | 1.1 ± 0.3 | RI, MS | |||
| 99 | hedycaryol | 1543 | 1546 | 0.3 ± 0.1 | 18.2 ± 2.9 | RI, MS | ||
| 100 | β-calacorene | 1555 | 1564 | 0.1 ± 0.0 | RI, MS | |||
| 101 | cryolan-8-ol | 1558 | 1573 | Tr | RI, MS | |||
| 102 | (E)-nerolidol | 1562 | 1561 | 0.1 ± 0.0 | Std, RI, MS | |||
| 103 | spathulenol | 1567 | 1577 | Tr | 0.1 ± 0.0 | RI, MS | ||
| 104 | caryophyllene oxide | 1571 | 1583 | Tr | 0.7 ± 0.2 | Std, RI, MS | ||
| 105 | gleenol | 1578 | 1586 | 1.5 ± 0.2 | RI, MS | |||
| 106 | allo-hedycariol | 1579 | 1580 | 0.2 ± 0.0 | RI, MS | |||
| 107 | furopelargone B | 1583 | 1588 | 7.0 ± 1.1 | RI, MS | |||
| 108 | esquel-7-en-9-one | 1589 | 10.2 ± 1.4 | MS | ||||
| 109 | humulol | 1591 | 1609 | Tr | RI, MS | |||
| 110 | 5-epi-7-epi-α-eudesmol | 1596 | 1607 | 0.1 ± 0.0 | 0.4 ± 0.1 | RI, MS | ||
| 111 | α-corocalene | 1615 | 1622 | 0.2 ± 0.0 | RI, MS | |||
| 112 | 1-epi-cubenol | 1620 | 1627 | 7.3 ± 0.9 | RI, MS | |||
| 113 | 10-epi-γ-eudesmol | 1608 | 1622 | 0.4 ± 0.1 | 0.5 ± 0.1 | 2.2 ± 0.4 | RI, MS | |
| 114 | eremoligenol | 1619 | 1629 | 0.6 ± 0.2 | RI, MS | |||
| 115 | γ-eudesmol | 1622 | 1630 | 6.6 ± 1.1 | RI, MS | |||
| 116 | hinesol | 1629 | 1640 | 0.3 ± 0.0 | RI, MS | |||
| 117 | cubenol | 1634 | 1645 | 5.4 ± 1.0 | RI, MS | |||
| 118 | 4-α-hydroxy-dihydro agarofuran | 1634 | 1651 | 2.2 ± 0.5 | RI, MS | |||
| 119 | α-muurolol | 1639 | 1644 | 1.4 ± 0.3 | RI, MS | |||
| 120 | β-eudesmol | 1639 | 1649 | 5.2 ± 1.0 | RI, MS | |||
| 121 | α-eudesmol | 1643 | 1652 | 0.6 ± 0.2 | 6.3 ± 1.1 | RI, MS | ||
| 122 | α-cadinol | 1645 | 1652 | 0.3 ± 0.1 | RI, MS | |||
| 123 | 7-epi-α-eudesmol | 1646 | 1662 | 0.2 ± 0.0 | RI, MS | |||
| 124 | bulnesol | 1656 | 1670 | 0.5 ± 0.1 | RI, MS | |||
| 125 | cadalene | 1665 | 1675 | 0.3 ± 0.1 | RI, MS | |||
| 126 | kaurene | 2039 | 2042 | 1.3 ± 0.3 | RI, MS | |||
| 127 | n-heneicosane | 2100 | 2100 | 0.1 ± 0.0 | Std, RI, MS | |||
| 128 | n-tricosane | 2300 | 2300 | Tr | Std, RI, MS | |||
| Total identified (%) | 98.3 | 99.9 | 99.4 | 79.9 | ||||
| Grouped compounds (%) | ||||||||
| Monoterpene hydrocarbons | Tr | 18.0 | 48.7 | 13.6 | ||||
| Oxygenated monoterpenes | 30.3 | 4.4 | 3.5 | |||||
| Sesquiterpene hydrocarbons | 81.2 | 0.4 | 5.9 | 5.3 | ||||
| Oxygenated sesquiterpenes | 17.2 | Tr | 39.0 | 57.2 | ||||
| Phenylpropanoids | 51.1 | Tr | Tr | |||||
| Others | 0.2 | 1.4 | 0.6 | |||||
| Essential Oil | M. domestica Female | M. domestica Male | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| LC50 (µg adult−1) | CI95 | LC90 (µg adult−1) | CI95 | χ2 (df = 3) | p-Value | LC50 (µg adult−1) | CI95 | LC90 (µg adult−1) | CI95 | χ2 (df = 3) | p-Value | |
| Canelo | 76.7 | 60.1–96.5 | 296.5 | 259.7–312.5 | 1.766 | 0.622 ns | 18.3 | 13.3–25.1 | 140.8 | 121.5–165.9 | 0.697 | 0.705 ns |
| Tepa | 88.7 | 81.3–94.2 | 128.6 | 118.5–139.7 | 1.564 | 0.457 ns | 24.6 | 18.5–29.7 | 119.3 | 89.4–139.7 | 1.096 | 0.777 ns |
| Paramela | 65.2 | 51.7–78.1 | 195.1 | 156.5–220.1 | 2.583 | 0.273 ns | 11.1 | 8.5–21.7 | 113.1 | 98.7–120.5 | 5.958 | 0.113 ns |
| Ciprés | 68.6 | 58.2–75.8 | 183.7 | 152.5–211.1 | 1.782 | 0.257 ns | 11.3 | 8.4–15.5 | 75.1 | 48.9–95.5 | 3.893 | 0.273 ns |
| Essential Oil | LC50 (µg larva−1) | CI95 | LC90 (µg larva−1) | CI95 | χ2 (df = 3) | p-Value |
|---|---|---|---|---|---|---|
| Canelo | 39.7 | 28.5–51.7 | 110.1 | 82.5–128.7 | 0.505 | 0.917 ns |
| Tepa | 35.2 | 29.1–40.6 | 72.3 | 59.5–93.2 | 0.569 | 0.966 ns |
| Paramela | 66.7 | 55.1–77.2 | 124.5 | 104.6–142.8 | 2.676 | 0.444 ns |
| Ciprés | 33.8 | 26.5–41.7 | 106.3 | 89.7–127.6 | 1.861 | 0.761 ns |
| Essential Oil | LC50 (µL L−1) | CI95 | LC90 (µL L−1) | CI95 | χ2 | p-Level | df |
|---|---|---|---|---|---|---|---|
| Canelo | 48.6 | 33.5–62.8 | 111.2 | 98.5–126.9 | 3.129 | 0.536 ns | 4 |
| Tepa | 52.2 | 39.8–61.1 | 81.5 | 71.8–92.7 | 1.452 | 0.325 ns | 4 |
| Paramela | 77.3 | 72.5–82.1 | 110.6 | 101.5–124.3 | 1.762 | 0.623 ns | 3 |
| Ciprés | 261.7 | 232.8–287.6 | 685.1 | 601.2–723.5 | 5.497 | 0.241 ns | 4 |
| Plant Species | L. philippiana | D. winteri | A. boronioides | P. uviferum |
|---|---|---|---|---|
| Yield | 0.06 | 0.04 | 0.04 | 0.07 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giordani, C.; Spinozzi, E.; Baldassarri, C.; Ferrati, M.; Cappellacci, L.; Santibañez Nieto, D.; Pavela, R.; Ricciardi, R.; Benelli, G.; Petrelli, R.; et al. Insecticidal Activity of Four Essential Oils Extracted from Chilean Patagonian Plants as Potential Organic Pesticides. Plants 2022, 11, 2012. https://doi.org/10.3390/plants11152012
Giordani C, Spinozzi E, Baldassarri C, Ferrati M, Cappellacci L, Santibañez Nieto D, Pavela R, Ricciardi R, Benelli G, Petrelli R, et al. Insecticidal Activity of Four Essential Oils Extracted from Chilean Patagonian Plants as Potential Organic Pesticides. Plants. 2022; 11(15):2012. https://doi.org/10.3390/plants11152012
Chicago/Turabian StyleGiordani, Cristiano, Eleonora Spinozzi, Cecilia Baldassarri, Marta Ferrati, Loredana Cappellacci, Daniela Santibañez Nieto, Roman Pavela, Renato Ricciardi, Giovanni Benelli, Riccardo Petrelli, and et al. 2022. "Insecticidal Activity of Four Essential Oils Extracted from Chilean Patagonian Plants as Potential Organic Pesticides" Plants 11, no. 15: 2012. https://doi.org/10.3390/plants11152012
APA StyleGiordani, C., Spinozzi, E., Baldassarri, C., Ferrati, M., Cappellacci, L., Santibañez Nieto, D., Pavela, R., Ricciardi, R., Benelli, G., Petrelli, R., & Maggi, F. (2022). Insecticidal Activity of Four Essential Oils Extracted from Chilean Patagonian Plants as Potential Organic Pesticides. Plants, 11(15), 2012. https://doi.org/10.3390/plants11152012














