Oil Palm Breeding in the Modern Era: Challenges and Opportunities
Abstract
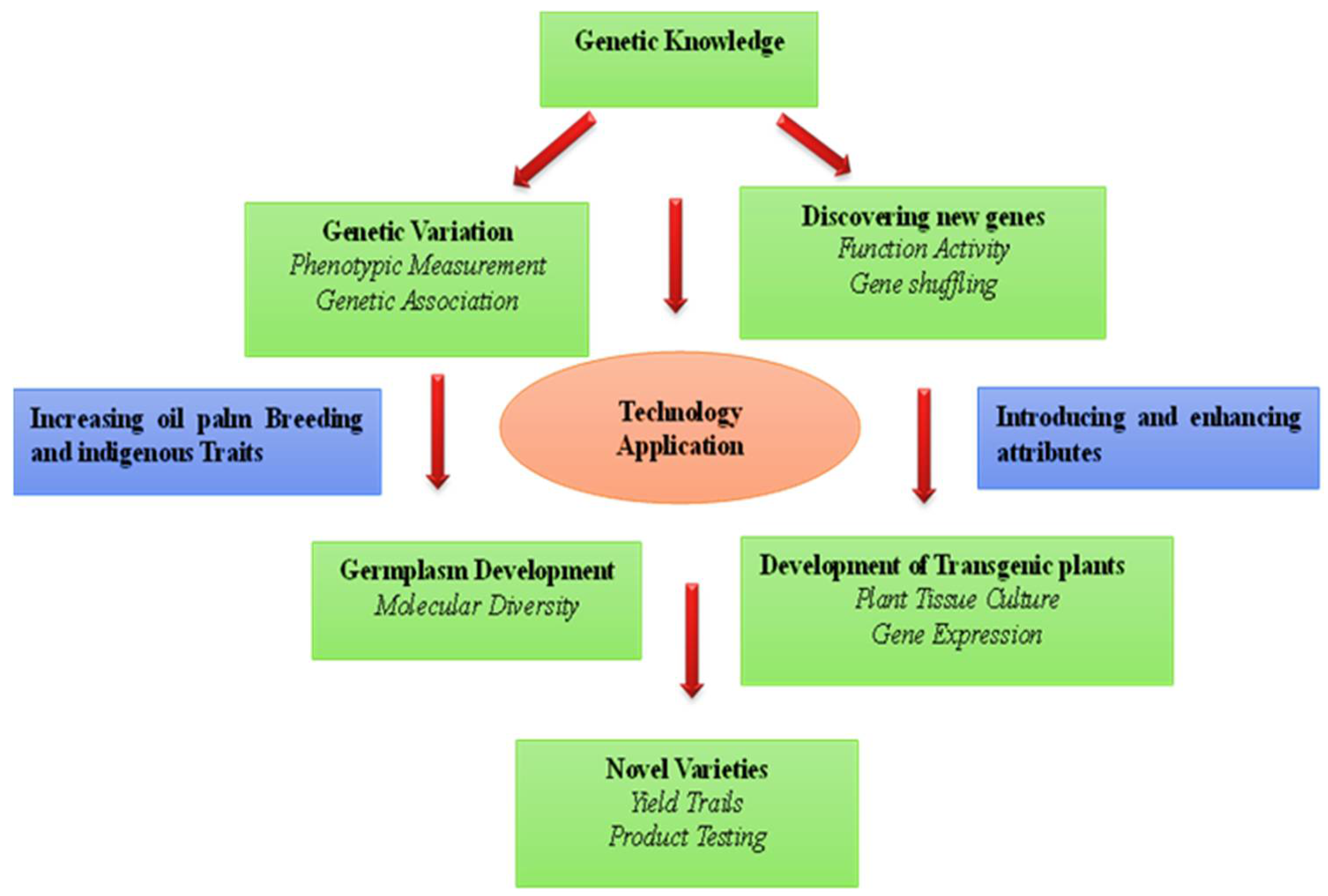
:1. Introduction
2. Genetic Diversity of Oil Palm Germplasm
2.1. Oil Palm Germplasm Characterization
2.2. Morphological Characterization of Oil Palm Germplasm
2.3. Molecular Characterization of Oil Palm Germplasm
2.4. Characterization of Oil Palm Germplasm Using Biochemical Techniques
3. Role of Conventional Breeding in Oil Palm
4. Importance of Tissue Culture in Oil Palm
5. Genetic Engineering of Oil Palm
| Explants |
Transformation/
Strain | Vector | Selection Marker | Promoter Used | Reporter Gene |
Transgene/
Expression | Studied Parameters | References |
|---|---|---|---|---|---|---|---|---|
| Young Leaves | LBA4404 | pUBA | bar | Ubi1 | Gus | Southern Bolt | Study glufosinate-ammonium- resistant transgenic oil palm | [41] |
| Embryonic Calli | LBA4404 | pBIDOG | DOGR1, | CaMV35S | Gus | PCR/ Southern Bolt | Introduction of new selection agent | [49] |
| Immature Embryo | LBA4404 | pCAMBIA 1310 | nptII | CaMV35S | Gus | Gus Assay | In vitro culture of IE for direct plant regeneration | [42] |
| Protoplast, Basidiospore, mycelium | LBA4404 | pCAMBIA 1300 | hyg | gpd fungal promoter | Gus/gfp | PCR | Understanding pathogenicity factor associated with G.boninense | [53] |
| Calli Clump | Electroporation | pCAMBIA 1303 | hptII | CaMV35S | GusA/ mgfp5 | PCR | An efficient electroporation-mediated transformation method for oil palm calli | [52] |
| Embryonic Calli | Biolistic | pMI11, pMI11G, pMI3, pMI3G | pmi gene | Ubi1, CaMV35S | GusA | PCR/ Southern Bolt | To produce transgenic oil palm by using mannose as a selectable agent | [38] |
| Immature Embryo | Biolistic | pBI121 | npt II | CaMV35S | Gus | Gus Assay | In vitro culture of IE for indirect plant regeneration | [42] |
| Embryonic Calli | Biolistic | pBINPLUS | hpt | 35S (2XCaMV35S) | GFP | PCR/ Southern Bolt | Increase the level of transgene expression and transformation efficiencies in oil palm | [54] |
| Immature Embryo | Biolistic | pPSP’AP-VF6 | HYG | CaMV35S, MSP | - | PCR | Successful integration of antiPATE driven by MSP in American oil palm plantlets | [43] |
6. Association Mapping for QTL Identification
7. Functional Markers and Their Application
Genomic Selection of Oil Palm
8. Genome Editing Technology
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Montúfar, R.; Louise, C.; Tranbarger, T. Elaeis oleifera (Kunth) Cortés: A neglected palm from the Ecuadorian Amazon. Rev. Ecuat. De Med. Y Cienc. Biol. 2018, 39, 11–18. [Google Scholar] [CrossRef]
- Montoya, C.; Lopes, R.; Flori, A.; Cros, D.; Cuellar, T.; Summo, M.; Espeout, S.; Rivallan, R.; Risterucci, A.M.; Bittencourt, D.; et al. Quantitative trait loci (QTLs) analysis of palm oil fatty acid composition in an interspecific pseudo-backcross from Elaeis oleifera (H.B.K.) Cortés and oil palm (Elaeis guineensis Jacq.). Tree Genet. Genomes 2013, 9, 1207–1225. [Google Scholar] [CrossRef] [Green Version]
- Astorkia, M.; Hernández, M.; Bocs, S.; Ponce, K.; León, O.; Morales, S.; Quezada, N.; Orellana, F.; Wendra, F.; Sembiring, Z.; et al. Detection of significant SNP associated with production and oil quality traits in interspecific oil palm hybrids using RARSeq. Plant Sci. 2020, 291, 110366. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Sun, Y.; Qu, J.; Syah, R.; Lim, C.H.; Alfiko, Y.; Rahman, N.E.B.; Suwanto, A.; Yue, G.; Wong, L.; et al. Transcriptome and functional analysis reveals hybrid vigor for oil biosynthesis in oil palm. Sci. Rep. 2017, 7, 439. [Google Scholar] [CrossRef] [Green Version]
- Tisserat, B. Clonal propagation of palms. In Plant Tissue Culture Manual: Supplement 7; Lindsey, K., Ed.; Springer: Dordrecht, The Netherlands, 1997; pp. 473–486. [Google Scholar]
- Rajesh, M.K.; Radha, E.; Karun, A.; Parthasarathy, V.A. Plant regeneration from embryo-derived callus of oil palm—The effect of exogenous polyamines. Plant Cell Tissue Organ Cult. 2003, 75, 41–47. [Google Scholar] [CrossRef]
- Borisjuk, N.; Kishchenko, O.; Eliby, S.; Schramm, C.; Anderson, P.; Jatayev, S.; Kurishbayev, A.; Shavrukov, Y. Genetic Modification for Wheat Improvement: From Transgenesis to Genome Editing. BioMed Res. Int. 2019, 2019, 6216304. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Matamoros, M.F.; Villanueva, M.A.; Islas-Flores, T. Genetic transformation of cell-walled plant and algae cells: Delivering DNA through the cell wall. Brief. Funct. Genom. 2018, 17, 26–33. [Google Scholar] [CrossRef]
- Yadava, P.; Abhishek, A.; Singh, R.; Singh, I.; Kaul, T.; Pattanayak, A.; Agrawal, P.K. Advances in Maize Transformation Technologies and Development of Transgenic Maize. Front. Plant Sci. 2017, 7, 1949. [Google Scholar] [CrossRef] [Green Version]
- Khatodia, S.; Bhatotia, K.; Passricha, N.; Khurana, S.M.P.; Tuteja, N. The CRISPR/Cas Genome-Editing Tool: Application in Improvement of Crops. Front. Plant Sci. 2016, 7, 506. [Google Scholar] [CrossRef] [Green Version]
- Okoye, M.; Uguru, M.; Bakoumé, C.; Singh, R.; Okwuagwu, C. Assessment of Genetic Diversity of NIFOR Oil Palm Main Breeding Parent Genotypes Using Microsatellite Markers. Am. J. Plant Sci. 2016, 7, 218–237. [Google Scholar] [CrossRef] [Green Version]
- Barcelos, E.; Rios, S.D.A.; Cunha, R.N.V.; Lopes, R.; Motoike, S.Y.; Babiychuk, E.; Skirycz, A.; Kushnir, S. Oil palm natural diversity and the potential for yield improvement. Front. Plant Sci. 2015, 6, 190. [Google Scholar] [CrossRef] [PubMed]
- Rajanaidu, N. Institiut Penyelidikan Minyak Kelapa Sawit M. PORIM Oil Palm Genebank: Collection, Evaluation, Utilization and Conservation of Oil Palm Genetic Resources; Palm Oil Research Institute of Malaysia: Kuala Lumpur, Malaysia, 1994. [Google Scholar]
- Okwuagwu, C.O.; Ataga, C.D.; Okoye, M.; Okolo, E.C. Germplasm Collection of Highland Palms of Afikpo in Eastern Nigeria. Bayero J. Pure Appl. Sci. 2011, 4, 112–114. [Google Scholar] [CrossRef]
- Bakoumé, C.; Wickneswari, R.; Siju, S.; Rajanaidu, N.; Kushairi, A.; Billotte, N. Genetic diversity of the world’s largest oil palm (Elaeis guineensis Jacq.) field genebank accessions using microsatellite markers. Genet. Resour. Crop Evol. 2015, 62, 349–360. [Google Scholar] [CrossRef]
- Okwuagwu, C.O.; Okoye, M.; Okolo, E.C.; Ataga, C.D.; Uguru, M. Genetic variability of fresh fruit bunch yield in Deli/dura×tenera breeding populations of oil palm (Elaeis guineensis Jacq.) in Nigeria. J. Trop. Agric. 2008, 46, 40–45. [Google Scholar]
- Rajanaidu, N.; Kushairi, A.; Rafii, M.; Mohd Din, A.; Maizura, I.; Jalani, B.S. Oil palm breeding and genetic resources. In Advances in Oil Palm Research; Malaysian Palm Oil Board: Kuala Lumpur, Malaysia, 2000; pp. 171–237. [Google Scholar]
- Hayati, A.; Wickneswari, R.; Ithnin, M.; Rajanaidu, N. Genetic diversity of oil palm (Elaeis guineensis Jacq.) germplasm collections from Africa: Implications for improvement and conservation of genetic resources. TAG Theor. Appl. Genet. Theor. Und Angew. Genet. 2004, 108, 1274–1284. [Google Scholar] [CrossRef]
- Noh, A.; Rafii, M.Y.; Saleh, G.; Kushairi, A.; Latif, M.A. Genetic Performance and General Combining Ability of Oil Palm Deli dura x AVROS pisifera Tested on Inland Soils. Sci. World J. 2012, 2012, 792601. [Google Scholar] [CrossRef] [Green Version]
- van de Wiel, C.; Schaart, J.; Niks, R.; Visser, R. Traditional Plant Breeding Methods; Wageningen UR Plant Breeding: Wageningen, The Netherlands, 2010. [Google Scholar]
- Madon, M. Short communication: Genomic constitution of oil palm interspecific hybrid crosses monitored by genomic in situ hybridisation (gish). J. Oil Palm Res. 2018, 30, 338–344. [Google Scholar] [CrossRef] [Green Version]
- Camillo, J.; Leão, A.P.; Alves, A.A.; Formighieri, E.F.; Azevedo, A.L.; Nunes, J.D.; de Capdeville, G.; de A Mattos, J.K.; Souza, M.T., Jr. Reassessment of the Genome Size in Elaeis guineensis and Elaeis oleifera, and Its Interspecific Hybrid. Genom. Insights 2014, 7, 13–22. [Google Scholar] [CrossRef] [Green Version]
- Maria, M.; Clyde, M.M.; Cheah, S.C. Cytological analysis of Elaeis guineensis (tenera) chromosomes. Elaeis 1995, 7, 122–131. [Google Scholar]
- Singh, R.; Ong-Abdullah, M.; Low, E.-T.L.; Manaf, M.A.A.; Rosli, R.; Nookiah, R.; Ooi, L.C.-L.; Ooi, S.-E.; Chan, K.-L.; Halim, M.A.; et al. Oil palm genome sequence reveals divergence of interfertile species in Old and New worlds. Nature 2013, 500, 335–339. [Google Scholar] [CrossRef] [Green Version]
- Schmutz, J.; Cannon, S.B.; Schlueter, J.; Ma, J.; Mitros, T.; Nelson, W.; Hyten, D.L.; Song, Q.; Thelen, J.J.; Cheng, J.; et al. Genome sequence of the palaeopolyploid soybean. Nature 2010, 463, 178–183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chalhoub, B.; Denoeud, F.; Liu, S.; Parkin, I.A.; Tang, H.; Wang, X.; Chiquet, J.; Belcram, H.; Tong, C.; Samans, B.; et al. Plant genetics. Early allopolyploid evolution in the post-Neolithic Brassica napus oilseed genome. Science 2014, 345, 950–953. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schnable, P.S.; Ware, D.; Fulton, R.S.; Stein, J.C.; Wei, F.; Pasternak, S.; Liang, C.; Zhang, J.; Fulton, L.; Graves, T.A.; et al. The B73 maize genome: Complexity, diversity, and dynamics. Science 2009, 326, 1112–1115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiao, Y.; Xu, P.; Fan, H.; Baudouin, L.; Xia, W.; Bocs, S.; Xu, J.; Li, Q.; Guo, A.; Zhou, L.; et al. The genome draft of coconut (Cocos nucifera). Gigascience 2017, 6, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Denis, M.; Bouvet, J.-M. Efficiency of genomic selection with models including dominance effect in the context of Eucalyptus breeding. Tree Genet. Genomes 2013, 9, 37–51. [Google Scholar] [CrossRef]
- Rival, A.; Bertrand, L.; Beulé, T.; Combes, M.C.; Trouslot, P.; Lashermes, P. Suitability of RAPD analysis for the detection of somaclonal variants in oil palm (Elaeis guineensis Jacq). Plant Breed. 1998, 117, 73–76. [Google Scholar] [CrossRef]
- Rohani, P.; Wearing, H.J.; Cameron, T.; Sait, S.M. Natural enemy specialization and the period of population cycles. Ecol. Lett. 2003, 6, 381–384. [Google Scholar] [CrossRef] [Green Version]
- Rival, A.; Ilbert, P.; Labeyrie, A.; Torres, E.; Doulbeau, S.; Personne, A.; Dussert, S.; Beulé, T.; Durand-Gasselin, T.; Tregear, J.W.; et al. Variations in genomic DNA methylation during the long-term in vitro proliferation of oil palm embryogenic suspension cultures. Plant Cell Rep. 2013, 32, 359–368. [Google Scholar] [CrossRef]
- Ooi, S.-E.; Choo, C.-N.; Ishak, Z.; Ong-Abdullah, M. A candidate auxin-responsive expression marker gene, EgIAA9, for somatic embryogenesis in oil palm (Elaeis guineensis Jacq.). Plant Cell. Tissue Organ Cult. 2012, 110, 201–212. [Google Scholar] [CrossRef]
- Yunus, A.M.M.; Fischer, R. Development of a Protoplast based Transformation System for Genetic Engineering of Oil Palm; Publikationsserver der RWTH Aachen University: Aachen, Germany, 2013. [Google Scholar]
- Bahariah, B.; Mat Yunus, A.M.; Rasid, O.; Ghulam Kadir, A.P. Multiplex CRISPR/Cas9-mediated genome editing of the FAD2 gene in rice: A model genome editing system for oil palm. J. Genet. Eng. Biotechnol. 2021, 19, 86. [Google Scholar] [CrossRef]
- Sambanthamurthi, R.; Abdullah, S.; Ghulam Kadir, A.P. Genetic manipulation of the oil palm—Challenges and prospects. Planter 2002, 78, 547–562. [Google Scholar]
- Ghulam Kadir, A.P.; Rasid, O.; Hashim, A.; Ishak, Z.; Rosli, S.; Sambanthamurthi, R. Tissue Culture and Genetic Engineering of Oil Palm. In Palm Oil: Production, Processing, Characterization, and Uses; Science Direct; AOCS Press: Urbana, IL, USA, 2012; pp. 87–135. [Google Scholar] [CrossRef]
- Bahariah, B.; Ghulam Kadir, A.P.; Mat Yunus, A.M.; Subhi, S.; Khalid, N.; Othman, D.R. Biolistic transformation of oil palm using the phosphomannose isomerase (pmi) gene as a positive selectable marker. Biocatal. Agric. Biotechnol. 2013, 2, 295–304. [Google Scholar] [CrossRef]
- Masani, M.Y.A.; Noll, G.A.; Parveez, G.K.A.; Sambanthamurthi, R.; Prüfer, D. Efficient Transformation of Oil Palm Protoplasts by PEG-Mediated Transfection and DNA Microinjection. PLoS ONE 2014, 9, e96831. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mat Yunus, A.M.; Ghulam Kadir, A.P. Development of transformation vectors for the production of potentially high oleate transgenic oil palm. Electron. J. Biotechnol. 2008, 11, 23–31. [Google Scholar] [CrossRef]
- Izawati, A.M.; Parveez, G.K.; Masani, M.Y. Transformation of oil palm using Agrobacterium tumefaciens. In Transgenic Plants; Methods in Molecular Biology; Dunwell, J., Wetten, A., Eds.; Humana Press: Totowa, NJ, USA, 2012; Volume 847, pp. 177–188. [Google Scholar] [CrossRef]
- Abdullah, R.; Zainal, A.; Heng, W.; Li, L.; Beng, Y.; Phing, L.; Abdullah Sirajuddin, S.; Yap, W.; Joseph, J.; Jusoh, S.A.; et al. Immature embryo: A useful tool for oil palm (Elaeis guineensis Jacq.) genetic transformation studies. Electron. J. Biotechnol. 2005, 8, 8. [Google Scholar] [CrossRef] [Green Version]
- Bhore, S.; Shah, F.H. Genetic Transformation of the American Oil Palm (Elaeis oleifera) Immature Zygotic Embryos with Antisense Palmitoyl-Acyl Carrier Protein Thioesterase (PATE) Gene. World Appl. Sci. J. 2012, 16, 362–369. [Google Scholar]
- Lee, M.-P.; Yeun, L.-H.; Abdullah, R. Expression of Bacillus thuringiensis insecticidal protein gene in transgenic oil palm. Electron. J. Biotechnol. 2006, 9, 117–126. [Google Scholar] [CrossRef] [Green Version]
- Ismail, I.; Fong, S.L.; Abdullah, R.; Fei, C.; Zainal, Z.; Sidik, N. Molecular and Expression Analysis of Cowpea Trypsin Inhibitor (CpTI) Gene in Transgenic Elaeis guineensis Jacq Leaves. Aust. J. Crop Sci. 2010, 4, 37–48. [Google Scholar]
- Ariffin, N.; Abdullah, R.; Muad, M.; Lourdes, J.; Emran, N.; Ismail, M.; Ismail, I.; Fadzil, M.; Kong, L.; Siddiqui, Y.; et al. Construction of expression vectors of polyhydroxybutyrate-co-hydroxyvalerate (PHBV) and transient expression of transgenes in immature oil palm embryos. Plasmid 2011, 66, 136–143. [Google Scholar] [CrossRef]
- Bahariah, B.; Rasid, O.; Rahmah, S.; Ghulam Kadir, A.P.; Mat Yunus, A.M. Genetic transformation of oil palm based on selection with hygromycin. J. Oil Palm Res. 2020, 33, 577–587. [Google Scholar]
- Yarra, R.; Jin, L.; Zhao, Z.; Cao, H. Progress in Tissue Culture and Genetic Transformation of Oil Palm: An Overview. Int. J. Mol. Sci. 2019, 20, 5353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Izawati, A.M.D.; Masani, M.Y.A.; Ismanizan, I.; Parveez, G.K.A. Evaluation on the effectiveness of 2-deoxyglucose-6-phosphate phosphatase (DOGR1) gene as a selectable marker for oil palm (Elaeis guineensis Jacq.) embryogenic calli transformation mediated by Agrobacterium tumefaciens. Front. Plant Sci. 2015, 6, 727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, P.; Nagarajan, A.; Uchil, P.D. Electroporation. Cold Spring Harb. Protoc. 2019, 2019, 519–525. [Google Scholar] [CrossRef] [Green Version]
- Mariani, T.S.; Ermavitalini, D.; Mitsutaka, T.; Chia, T.F.; Miyake, H. GUS gene expression in somatic embryo of oil palm (Elaeis guineensis Jacq.). Asian J. Appl. Sci. 2015, 3, 649–650. [Google Scholar]
- Darmawan, C.; Wiendi, N.; Utomo, C.; Liwang, T. Electroporation-mediated genetic transformation of oil palm (Elaeis guineensis). Biodiversitas J. Biol. Divers. 2020, 21, 3720–3726. [Google Scholar] [CrossRef]
- Govender, N.; Wong, M.-Y. Detection of Oil Palm Root Penetration by Agrobacterium-Mediated Transformed Ganoderma boninense, Expressing Green Fluorescent Protein. Phytopathology 2016, 107, 483–490. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bahariah, B.; Mat Yunus, A.M.; Rasid, O.; Ghulam Kadir, A.P. Construction of a vector containing hygromycin (HPT) gene driven by double 35S (2XCaMV35S) promoter for oil palm transformation. J. Oil Palm Res. 2017, 29, 180–188. [Google Scholar] [CrossRef] [Green Version]
- Varshney, R.K.; Graner, A.; Sorrells, M.E. Genic microsatellite markers in plants: Features and applications. Trends Biotechnol. 2005, 23, 48–55. [Google Scholar] [CrossRef]
- Singh, R.; Tan, S.G.; Panandam, J.M.; Rahman, R.A.; Ooi, L.C.; Low, E.T.; Cheah, S.C. Mapping quantitative trait loci (qtls) for fatty acid composition in an interspecific cross of oil palm. BMC Plant Biol. 2009, 9, 114. [Google Scholar] [CrossRef] [Green Version]
- Gan, Y.L.; Fu, J.Y.; Lai, O.M.; Chew, B.H.; Yuen, K.H.; Teng, K.T.; Nesaretnam, K.; Selvaduray, K.R.; Meganathan, P. Effect of palm-based tocotrienols and tocopherol mixture supplementation on platelet aggregation in subjects with metabolic syndrome: A randomized controlled trial. Sci. Rep. 2017, 7, 11542. [Google Scholar] [CrossRef] [Green Version]
- Teh, C.K.; Ong, A.L.; Kwong, Q.B.; Apparow, S.; Chew, F.T.; Mayes, S.; Mohamed, M.; Appleton, D.; Kulaveerasingam, H. Genome-wide association study identifies three key loci for high mesocarp oil content in perennial crop oil palm. Sci. Rep. 2016, 6, 19075. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sambanthamurthi, R.; Singh, R.; Kadir, A.P.G.; Abdullah, M.O.; Kushairi, A. Opportunities for the Oil Palm via Breeding and Biotechnology. In Breeding Plantation Tree Crops: Tropical Species; Jain, S.M., Priyadarshan, P.M., Eds.; Springer: New York, NY, USA, 2009; pp. 377–421. [Google Scholar] [CrossRef]
- Mayes, S.; Jack, P.; Corley, R. The use of molecular markers to investigate the genetic structure of an oil palm breeding programme. Heredity 2000, 85 Pt 3, 288–293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sathish, D.; Mohankuma, R.C. RAPD markers for identifying oil palm (Elaeis guineensis Jacq.) parental varieties (dura & pisifera) and the hybrid tenera. Indian J. Biotechnol. 2007, 6, 354–358. [Google Scholar]
- Singh, R.; Low, E.T.L.; Ooi, L.C.L.; Ong-Abdullah, M.; Ting, N.C.; Nagappan, J.; Nookiah, R.; Amiruddin, M.D.; Rosli, R.; Manaf, M.A.A.; et al. The oil palm SHELL gene controls oil yield and encodes a homologue of SEEDSTICK. Nature 2013, 500, 340–344. [Google Scholar] [CrossRef] [Green Version]
- Moretzsohn, M.C.; Ferreira, M.A.; Amaral, Z.; Coelho, P.; Grattapaglia, D.; Ferreira, M.E. Genetic diversity of brazilian oil palm (elaeis oleifera hbk) germplasm collected in the amazon forest. Euphytica 2002, 124, 35–45. [Google Scholar] [CrossRef]
- Moretzsohn, M.; Nunes, C.; Ferreira, M.; Grattapaglia, D. RAPD linkage mapping of the shell thickness locus in oil palm ( Elaeis guineensis Jacq.). Theor. Appl. Genet. 2000, 100, 63–70. [Google Scholar] [CrossRef]
- Dou, Y.; Xia, W.; Mason, A.S.; Huang, D.; Sun, X.; Fan, H.; Xiao, Y. Developing functional markers for vitamin E biosynthesis in oil palm. PLoS ONE 2021, 16, e0259684. [Google Scholar] [CrossRef]
- Xiao, Y.; Zhou, L.; Xia, W.; Mason, A.S.; Yang, Y.; Ma, Z.; Peng, M. Exploiting transcriptome data for the development and characterization of gene-based ssr markers related to cold tolerance in oil palm (elaeis guineensis). BMC Plant Biol. 2014, 14, 384. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Williams, R.; Prakash, C.S.; He, G. Identification and characterization of gene-based ssr markers in date palm (phoenix dactylifera l). BMC Plant Biol. 2012, 12, 237. [Google Scholar] [CrossRef] [Green Version]
- Billotte, N.; Marseillac, N.; Risterucci, A.M.; Adon, B.; Brottier, P.; Baurens, F.C.; Singh, R.; Herrán, A.; Asmady, H.; Billot, C.; et al. Microsatellite-based high density linkage map in oil palm (Elaeis guineensis Jacq.). Theor. Appl. Genet. 2005, 110, 754–765. [Google Scholar] [CrossRef]
- Babu, B.K.; Mathur, R.K.; Kumar, P.N.; Ramajayam, D.; Ravichandran, G.; Venu, M.V.B.; Babu, S.S. Development, identification and validation of CAPS marker for SHELL trait which governs dura, pisifera and tenera fruit forms in oil palm (Elaeis guineensis Jacq.). PLoS ONE 2017, 12, e0171933. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bai, B.; Wang, L.; Zhang, Y.J.; Lee, M.; Rahmadsyah, R.; Alfiko, Y.; Yue, G.H. Developing genome-wide snps and constructing an ultrahigh-density linkage map in oil palm. Sci. Rep. 2018, 8, 691. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grattapaglia, D.; Silva-Junior, O.B.; Resende, R.T.; Cappa, E.P.; Müller, B.S.F.; Tan, B.; Isik, F.; Ratcliffe, B.; El-Kassaby, Y.A. Quantitative Genetics and Genomics Converge to Accelerate Forest Tree Breeding. Front. Plant Sci. 2018, 9, 1693. [Google Scholar] [CrossRef] [PubMed]
- Rafii, M.; Sukaimi, J.; Din, A.; Jalloh, M.; Swaray, S.; Yusuff, O.; Chukwu, S. Genetic Improvement of Oil Palm Through Recurrent Selection. In The Oil Palm Genome; Compendium of Plant Genomes; Springer: Cham, Switzerland, 2020; pp. 35–46. [Google Scholar] [CrossRef]
- Kadam, D.C.; Potts, S.M.; Bohn, M.O.; Lipka, A.E.; Lorenz, A.J. Genomic Prediction of Single Crosses in the Early Stages of a Maize Hybrid Breeding Pipeline. G3 2016, 6, 3443–3453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cros, D.; Bocs, S.; Riou, V.; Ortega-Abboud, E.; Tisné, S.; Argout, X.; Pomiès, V.; Nodichao, L.; Lubis, Z.; Cochard, B.; et al. Genomic preselection with genotyping-by-sequencing increases performance of commercial oil palm hybrid crosses. BMC Genom. 2017, 18, 839. [Google Scholar] [CrossRef] [Green Version]
- El-Mounadi, K.; Morales-Floriano, M.L.; Garcia-Ruiz, H. Principles, Applications, and Biosafety of Plant Genome Editing Using CRISPR-Cas9. Front. Plant Sci. 2020, 11, 56. [Google Scholar] [CrossRef]
- Arora, L.; Narula, A. Gene Editing and Crop Improvement Using CRISPR-Cas9 System. Front. Plant Sci. 2017, 8, 1932. [Google Scholar] [CrossRef] [Green Version]
- Yarra, R.; Cao, H.; Jin, L.; Mengdi, Y.; Zhou, L. CRISPR/Cas mediated base editing: A practical approach for genome editing in oil palm. 3 Biotech 2020, 10, 306. [Google Scholar] [CrossRef]
- Kalyana Babu, B.; Mary Rani, K.L.; Sarika Sahu, R.K.; Mathur Naveen Kumar, P.; Ravichandran, G.; Anitha, P.; Bhagya, H.P. Development andvalidation of whole genome-wide and genic microsatellite markers in oil palm (Elaeis guineensis Jacq.): First microsatellite database (OpSatdb). Sci. Rep. 2019, 9, 1899. [Google Scholar]
- Haun, W.; Coffman, A.; Clasen, B.; Demorest, Z.; Lowy, A.; Ray, E.; Retterath, A.; Stoddard, T.; Juillerat, A.; Cedrone, F.; et al. Improved soybean oil quality by targeted mutagenesis of the fatty acid desaturase 2 gene family. Plant Biotechnol. J. 2014, 12, 934–940. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
John Martin, J.J.; Yarra, R.; Wei, L.; Cao, H. Oil Palm Breeding in the Modern Era: Challenges and Opportunities. Plants 2022, 11, 1395. https://doi.org/10.3390/plants11111395
John Martin JJ, Yarra R, Wei L, Cao H. Oil Palm Breeding in the Modern Era: Challenges and Opportunities. Plants. 2022; 11(11):1395. https://doi.org/10.3390/plants11111395
Chicago/Turabian StyleJohn Martin, Jerome Jeyakumar, Rajesh Yarra, Lu Wei, and Hongxing Cao. 2022. "Oil Palm Breeding in the Modern Era: Challenges and Opportunities" Plants 11, no. 11: 1395. https://doi.org/10.3390/plants11111395
APA StyleJohn Martin, J. J., Yarra, R., Wei, L., & Cao, H. (2022). Oil Palm Breeding in the Modern Era: Challenges and Opportunities. Plants, 11(11), 1395. https://doi.org/10.3390/plants11111395







