Diversity of Useful Plants in Cabo Verde Islands: A Biogeographic and Conservation Perspective
Abstract
:1. Introduction
2. Results
2.1. The First Reported Useful Species—A Brief Historical Note
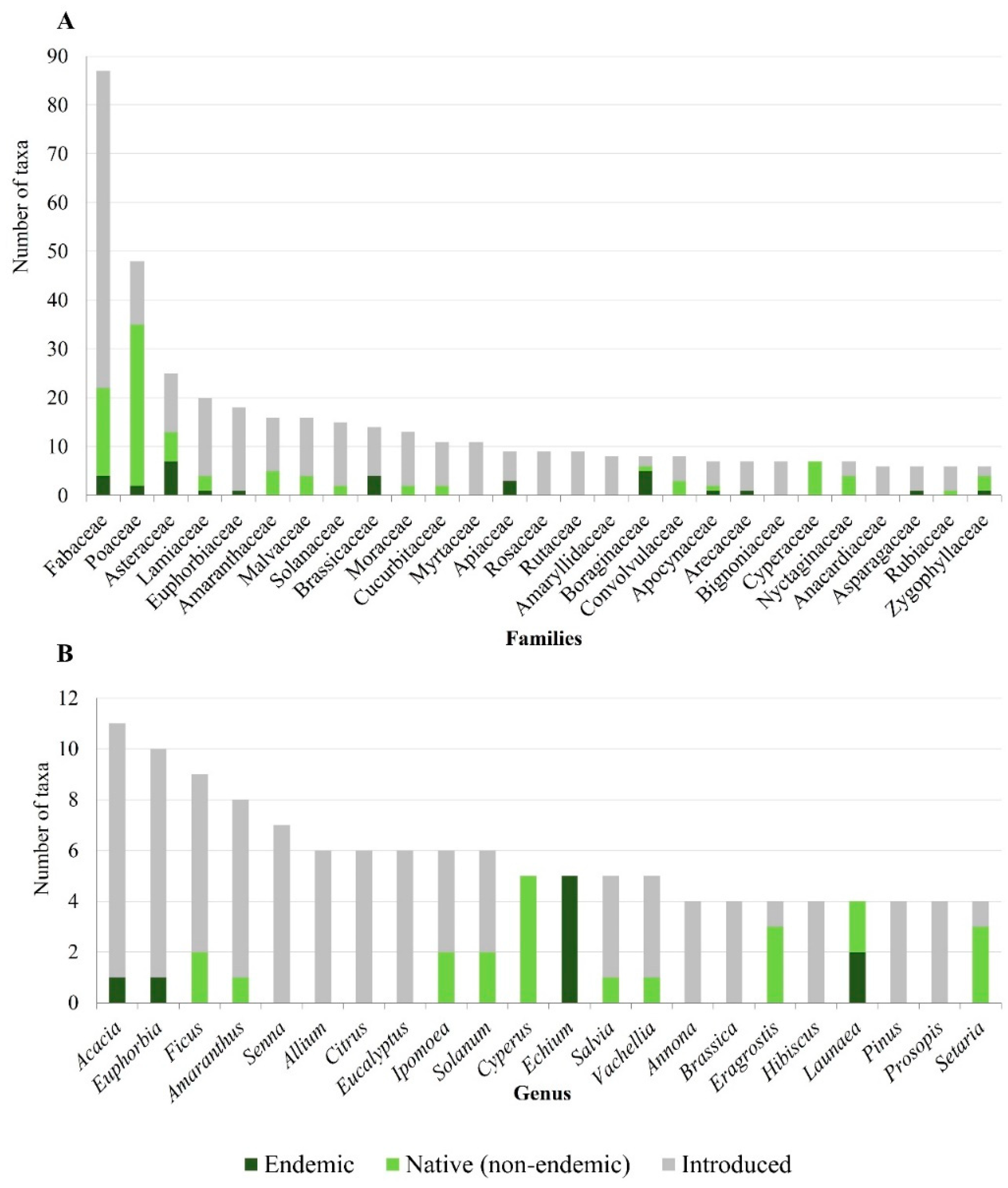
2.2. Taxonomic Diversity
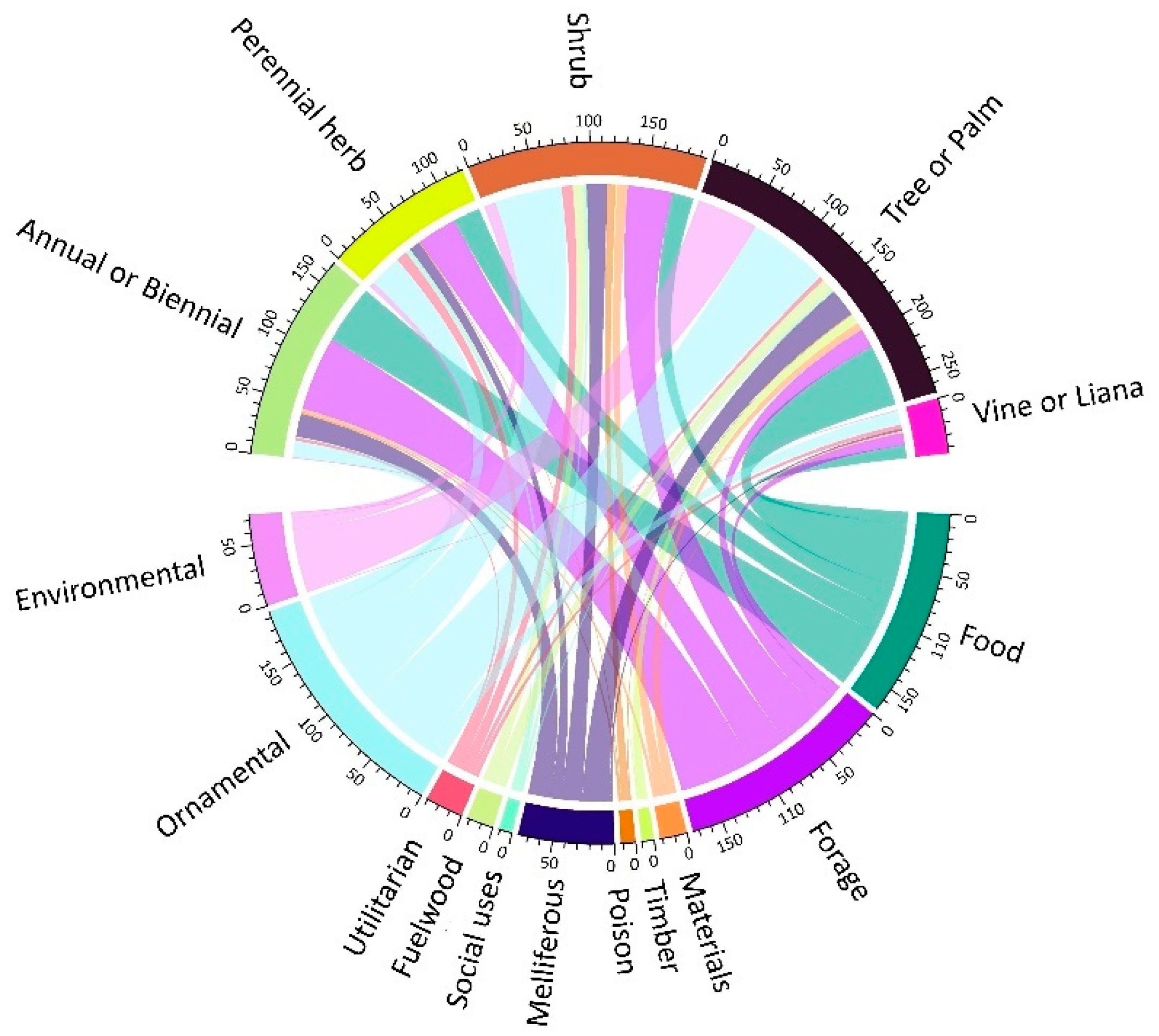
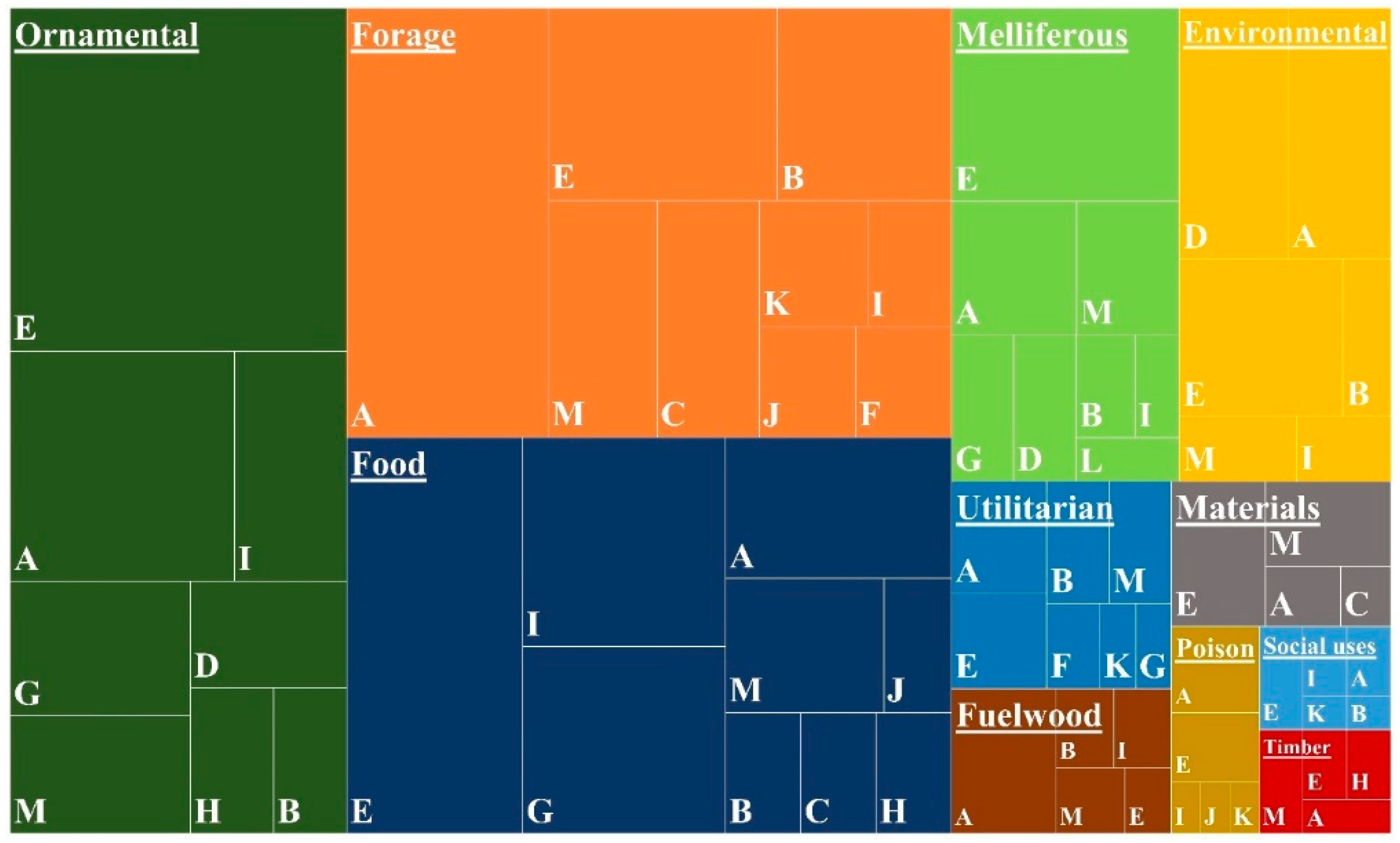
2.3. Main Uses of Cabo Verdean Flora
2.4. Growth form Diversity and Uses
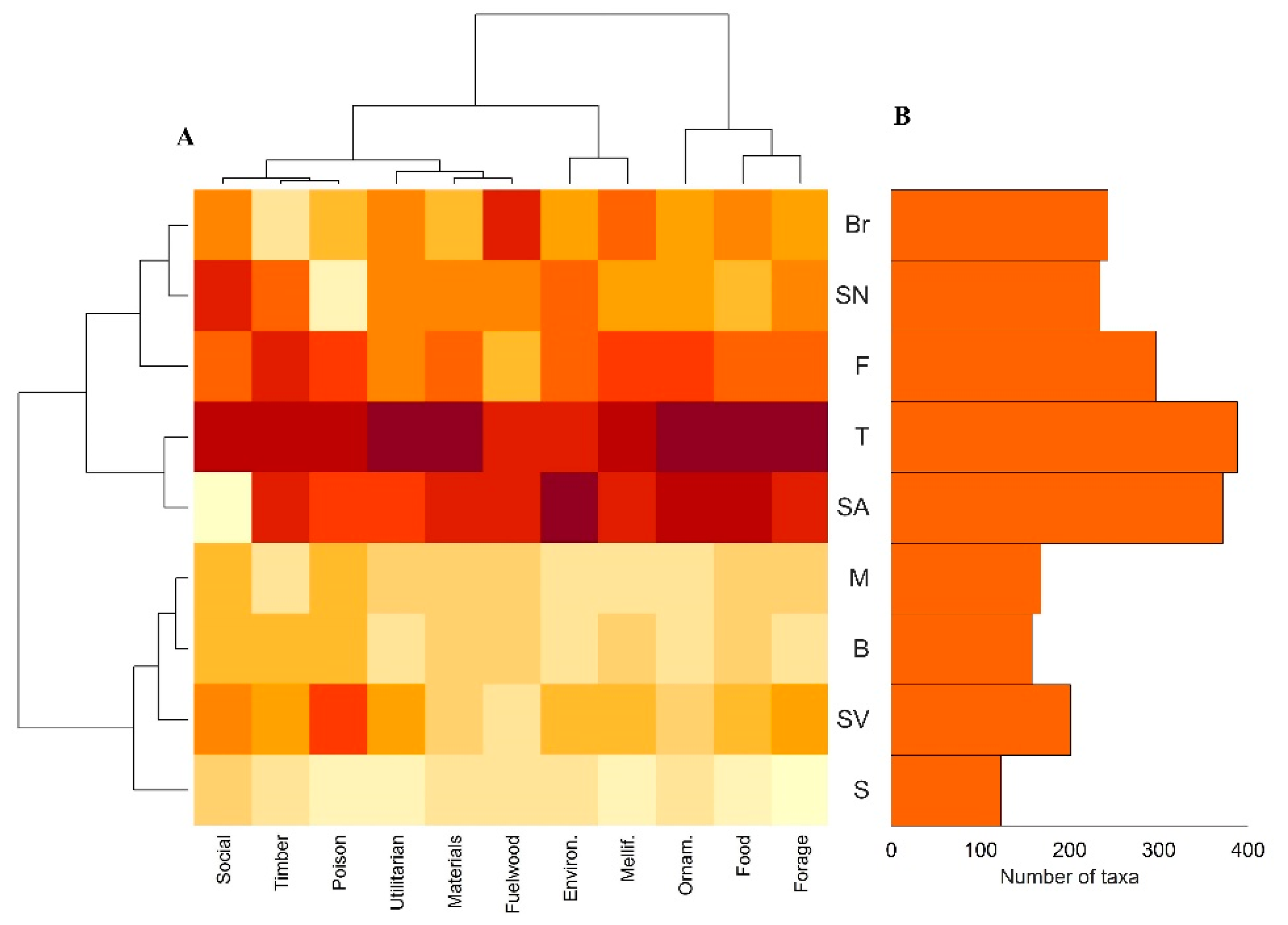
2.5. Distribution across Cabo Verde Archipelago
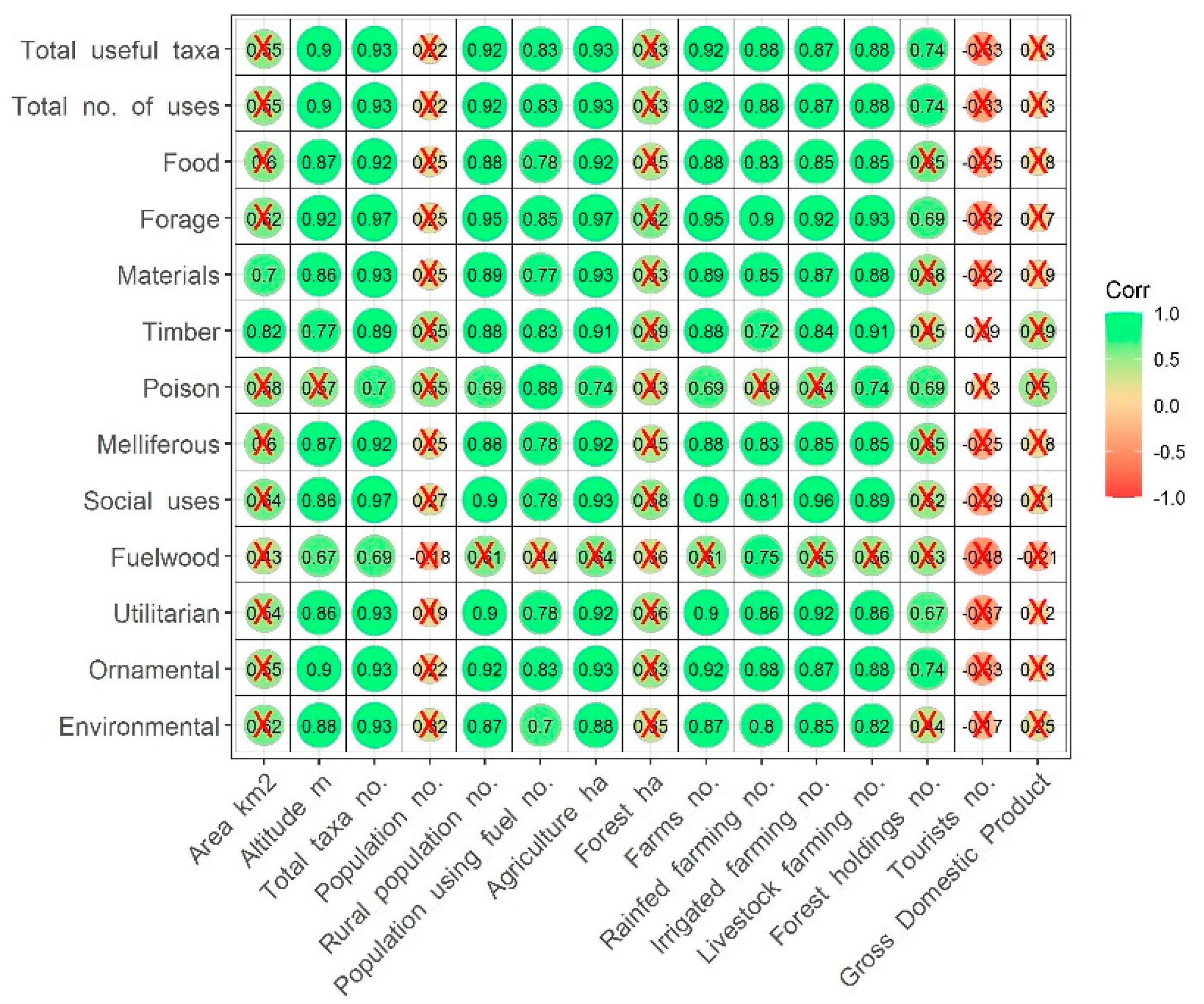
2.6. Uses vs. Biogeographic Origin
2.7. Native Species Conservation
2.8. Agrobiodiversity and Traditional Knowledge
3. Discussion
3.1. Taxonomic Diversity
3.2. History behind Diversity
3.3. Plant Uses and Sustainability
4. Materials and Methods
4.1. The Study Area
4.2. Listing Useful Plants
4.3. Geographic and Socio-Economic Data
4.4. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ceballos, G.; Ehrlich, P.R.; Dirzo, R. Biological annihilation via the ongoing sixth mass extinction signaled by vertebrate population losses and declines. Proc. Natl. Acad. Sci. USA 2017, 114, E6089–E6096. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kier, G.; Kreft, H.; Lee, T.M.; Jetz, W.; Ibisch, P.L.; Nowicki, C.; Mutke, J.; Barthlott, W. A global assessment of endemism and species richness across island and mainland regions. Proc. Natl. Acad. Sci. USA 2009, 106, 9322–9327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Florencio, M.; Patiño, J.; Nogué, S.; Traveset, A.; Borges, P.A.; Schaefer, H.; Amorim, I.R.; Arnedo, M.; Ávila, S.P.; Cardoso, P.; et al. Macaronesia as a fruitful arena for ecology, evolution, and conservation biology. Front. Ecol. Evol. 2021, 9, 718169. [Google Scholar] [CrossRef]
- Monteiro, F.; Fortes, A.; Ferreira, V.; Essoh, A.P.; Gomes, I.; Correia, A.M.; Romeiras, M.M. Current Status and Trends in Cabo Verde Agriculture. Agronomy 2020, 10, 74. [Google Scholar] [CrossRef] [Green Version]
- Varela, D.; Monteiro, F.; Vidigal, P.; Silva, L.; Romeiras, M.M. Mechanisms implemented for the sustainable development of agriculture: An overview of Cabo Verde performance. Sustainability 2020, 12, 5855. [Google Scholar] [CrossRef]
- Brilhante, M.; Varela, E.; Essoh, A.P.; Fortes, A.; Duarte, M.C.; Monteiro, F.; Ferreira, V.; Correia, A.M.; Duarte, M.P.; Romeiras, M.M. Tackling Food Insecurity in Cabo Verde Islands: The Nutritional, Agricultural and Environmental Values of the Legume Species. Foods 2021, 10, 206. [Google Scholar] [CrossRef]
- Rocha, V.; Duarte, M.C.; Catarino, S.; Duarte, I.; Romeiras, M.M. Cabo Verde’s Poaceae flora: A reservoir of Crop Wild Relatives diversity for crop improvement. Front. Plant Sci. 2021, 12, 630217. [Google Scholar] [CrossRef]
- Essoh, A.P.; Silva, J.; Lima, K.; Catarino, S.; Duarte, M.C.; Gomes, S.; Varela, E.; Moura, M.; Duarte, M.P.; Romeiras, M.M. Pharmacological potential of native plants used in traditional medicine in Santiago Island (Cabo Verde). Front. Pharmacol. 2022. submitted. [Google Scholar]
- Varela, D.; Romeiras, M.M.; Silva, L. Implications of climate change on the distribution and conservation of Cabo Verde endemic trees. Glob. Ecol. Conserv. 2022, 34, e02025. [Google Scholar] [CrossRef]
- Romeiras, M.M.; Duarte, M.C.; Santos-Guerra, A.; Carine, M.; Francisco-Ortega, J. Botanical exploration of the Cape Verde Islands: From the pre-Linnaean records and collections to late 18th century floristic accounts and expeditions. Taxon 2014, 63, 625–640. [Google Scholar] [CrossRef]
- Romeiras, M.M.; Catarino, L.; Torrão, M.M.; Duarte, M.C. Diversity and origin of medicinal exotic flora in Cape Verde Islands. Plant Ecol. Evol. 2011, 142, 214–225. [Google Scholar] [CrossRef] [Green Version]
- Duarte, M.C.; Romeiras, M.M. Cabo Verde. In Encyclopedia of Islands (No. 2); Gillespie, R., Gillespie, R.G., Clague, D.A., Eds.; University of California Press: Oakland, CA, USA, 2009; pp. 143–148. [Google Scholar]
- Neto, C.; Costa, J.C.; Figueiredo, A.; Capelo, J.; Gomes, I.; Vitória, S.; Semedo, J.M.; Lopes, A.; Dinis, H.; Correia, E.; et al. The role of climate and topography in shaping the diversity of plant communities in Cabo Verde Islands. Diversity 2020, 12, 80. [Google Scholar] [CrossRef] [Green Version]
- Instituto Nacional de Estatística, Anuário Estatístico de 2019. Available online: https://ine.cv/fr/publicacoes/anuario-estatistico-2019/ (accessed on 15 February 2022).
- Barbosa, L.A.G. Subsídios para um dicionário utilitário e glossário dos nomes vernáculos das plantas de arquipélago de Cabo Verde. Garcia Orta 1961, 9, 37–91. [Google Scholar]
- Gomes, I.; Gomes, S.; Vera-Cruz, M.T.; Kilian, N.; Leyens, T.; Lobin, W. Plantas Endémicas e Árvores Indígenas de Cabo Verde; INIDA: Praia, Cabo Verde, 1995; pp. 1–33. [Google Scholar]
- Varela, J.M. Contribuição para a utilização terapêutica de plantas medicinais de Cabo Verde & de plantas medicinais comuns a Angola, Cabo Verde, Guiné-Bissau e Moçambique. Anais Academia de Estudos de Culturas Comparadas 2001, 3, 57–74. [Google Scholar]
- Monod, T.; Mota, A.T.; Mauny, R. Description de la côte occidentale d’Afrique (Sénégal au Cap de Monte, Archipels), par Valentim Fernandes (1506–1510). Mem. Centro Estudos Guiné Portug. 1951, 11, 1–223. [Google Scholar]
- Sauvageot, S. Navigation de Lisbonne â l‘île de Sao Tome par un pilote portugais anonyme (vers 1545). Garcia de Orta 1961, 9, 123–138. [Google Scholar]
- Frutuoso, G. As Saudades da Terra; Tipografia Funchalense: Funchal, Portugal, 1873; pp. 1–919. [Google Scholar]
- Teixeira-da-Mota, A.; Carreira, A. “Milho Zaburro” and “Milho Maçaroca” in Guinea and in the Islands of Cabo Verde. J. Int. Afr. Inst. 1966, 36, 73–84. [Google Scholar] [CrossRef]
- Roberts, G.; Defoe, D. The Four Years Voyages of Capt. George Roberts. Available online: https://archive.org/details/fouryearsvoyage00robegoog/page/n14/mode/2up (accessed on 14 January 2022).
- Forster, J. Plantae Atlanticae ex insulis Madeira, Sti Jacobi, Adscencionis, Stae Helenae et Fayal reportatae. Comment. Soc. Regiae Sci. Gott. 1789, 9, 36–64. [Google Scholar]
- Feijó, J.S. Ensaio económico sobre as Ilhas de Cabo Verde em 1797. Mem. Econ. Acad. Real Sci. Lisboa 1815, 5, 172–193. [Google Scholar]
- Gardère, M.L.; Duarte, M.C.; Moraes, P.L.R.; Muller, S.; Romeiras, M.M. The scientific expedition of João da Silva Feijó to the Cabo Verde Islands (1783–1796) and the tribulations of his herbarium. Adansonia 2019, 41, 101–175. [Google Scholar] [CrossRef]
- Chelmicki, J.C.C.; Varnhagen, F.A. Corografia Cabo-Verdiana ou Descripção Geographico-Historica da Província das Ilhas de Cabo Verde e Guiné; Typographia, L. C. da Cunha: Lisboa, Portugal, 1841; Volume 1, p. 304. [Google Scholar]
- Chelmicki, J.C.C.; Varnhagen, F.A. Corografia Cabo-Verdiana ou Descripção Geographico-Historica da Província das Ilhas de Cabo Verde e Guiné; Typographia, L. C. da Cunha: Lisboa, Portugal, 1841; Volume 2, p. 511. [Google Scholar]
- Schmidt, J.A. Beiträge zur Flora der Cap Verdischen Inseln; Akademische Buchhandlung: Heidelberg, Germany, 1852; pp. 1–378. [Google Scholar]
- Palma, A.; David, A. Catálogo de Boas Práticas Agroecológicas nas Áreas Protegidas de Santo Antão, Santiago e São Nicolau; Centro de Estudios Rurales y de Agricultura Internacional: Praia, Cabo Verde, 2020; pp. 1–88. [Google Scholar]
- Plants of the World Online. Facilitated by the Royal Botanic Gardens, Kew. Available online: http://www.plantsoftheworldonline.org/ (accessed on 11 March 2022).
- Brochmann, C.; Rustan, Ø.H.; Lobin, W.; Kilian, N. The endemic vascular plants of the Cape Verde Islands, W Africa. Sommerfeltia 1997, 24, 1–356. [Google Scholar] [CrossRef]
- Romeiras, M.M.; Carine, M.; Duarte, M.C.; Catarino, S.; Dias, F.S.; Borda-de-Água, L. Bayesian methods to analyze historical collections in time and space: A case study using Cabo Verde Endemic Flora. Front. Plant Sci. 2020, 11, 278. [Google Scholar] [CrossRef] [PubMed]
- Rivas-Martínez, S.; Lousã, M.; Costa, J.C.; Duarte, M.C. Geobotanical survey of Cabo Verde Islands (West Africa). Int. J. Geobot. Res. 2017, 7, 1–103. [Google Scholar] [CrossRef]
- World Flora Online. Available online: http://www.worldfloraonline.org/ (accessed on 24 January 2022).
- Kalita, B.; Roy, A.; Annamalai, A.; Lakshmi, P.T.V. A molecular perspective on the taxonomy and journey of Citrus domestication. Perspect. Plant Ecol. Evol. Syst. 2021, 53, 125644. [Google Scholar] [CrossRef]
- The IUCN Red List of Threatened Species. Version 2021-3. Available online: https://www.iucnredlist.org (accessed on 28 February 2022).
- Romeiras, M.M.; Catarino, S.; Gomes, I.; Fernandes, C.; Costa, J.C.; Caujapé-Castells, J.; Duarte, M.C. IUCN Red List assessment of the Cape Verde endemic flora: Towards a global strategy for plant conservation in Macaronesia. Bot. J. Linn. Soc. 2016, 180, 413–425. [Google Scholar] [CrossRef] [Green Version]
- Cámara-Leret, R.; Paniagua-Zambrana, N.; Balslev, H.; Macía, M.J. Ethnobotanical knowledge is vastly under-documented in northwestern South America. PLoS ONE 2014, 9, e85794. [Google Scholar] [CrossRef] [Green Version]
- Vu, D.T.; Nguyen, T.A. The neglected and underutilized species in the Northern mountainous provinces of Vietnam. Genet. Resour. Crop Evol. 2017, 64, 1115–1124. [Google Scholar] [CrossRef]
- Nortje, J.M.; van Wyk, B.-E. Useful plants of Namaqualand, South Africa: A checklist and analysis. S. Afr. J. Bot. 2019, 122, 120–135. [Google Scholar] [CrossRef]
- Welcome, A.K.; Van Wyk, B.-E. An inventory and analysis of the food plants of southern Africa. S. Afr. J. Bot. 2019, 122, 136–179. [Google Scholar] [CrossRef]
- Diazgranados, M.; Allkin, B.; Black, N.; Cámara-Leret, R.; Canteiro, C.; Carretero, J.; Eastwood, R.; Hargreaves, S.; Hudson, A.; Milliken, W.; et al. World Checklist of Useful Plant Species; Royal Botanical Gardens: Kew, UK, 2020; pp. 1–689. [Google Scholar] [CrossRef]
- State of the World’s Plants. Available online: https://stateoftheworldsplants.org/2017/report/SOTWP_2017_1_naming_and_counting_the_world_s_plant_families.pdf/ (accessed on 11 January 2022).
- Linder, H.P.; Lehmann, C.E.R.; Archibald, S.; Osborne, C.P.; Richardson, D.M. Global grass (Poaceae) success underpinned by traits facilitating colonization, persistence and habitat transformation. Biol. Rev. Camb. Philos. Soc. 2018, 93, 1125–1144. [Google Scholar] [CrossRef]
- Duarte, M.C.; Rego, F.; Moreira, I. Distribution patterns of plant communities on Santiago Island, Cape Verde. J. Veg. Sci. 2005, 16, 283–292. [Google Scholar] [CrossRef]
- Beleza, S.; Campos, J.; Lopes, J.; Araújo, I.I.; Hoppfer Almada, A.; Correia e Silva, A.; Parra, E.J.; Rocha, J. The admixture structure and genetic variation of the archipelago of Cape Verde and its implications for admixture mapping studies. PLoS ONE 2012, 7, e51103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santana-Pérez, J.M. The African Atlantic islands in maritime history during the Ancien Régime. Int. J. Marit. Hist. 2018, 30, 634–648. [Google Scholar] [CrossRef]
- Torrão, M.M. Dietas Alimentares: Transferências e Adaptaçöes nas Ilhas de Cabo Verde, 1460–1540; Instituto de Investigação Científica Tropical: Lisbon, Portugal, 1995; pp. 1–108. [Google Scholar]
- Wieczorek, J.-P. Programme de Sauvegarde du Site de Cidade Velha; Technical Report; The United Nations Educational, Scientific and Cultural Organization: Paris, France, 1990. [Google Scholar]
- Ferrão, J.E.M. A Aventura das Plantas e os Descobrimentos Portugueses, 2nd ed.; Instituto de Investigação Científica Tropical; Fundação Berardo: Lisboa, Portugal, 1993; pp. 1–228. [Google Scholar]
- Khoshbakht, K.; Hammer, K. How many plant species are cultivated? Genet. Resour. Crop. Evol. 2008, 55, 925–928. [Google Scholar] [CrossRef]
- Fortes, A.R.; Ferreira, V.; Barbosa Simões, E.; Baptista, I.; Grando, S.; Sequeira, E. Food Systems and Food Security: The Role of Small Farms and Small Food Businesses in Santiago Island, Cabo Verde. Agriculture 2020, 10, 216. [Google Scholar] [CrossRef]
- Gomes, I.; Gomes, S. Arvores Centenárias de Cabo Verde (Fichas técnicas); INIDA: São Jorge dos Órgãos, Cabo Verde, 2018. [Google Scholar]
- Garí, J.A. Review of the African millet diversity. In Proceedings of the International Workshop on Fonio, Food Security and Livelihood among the Rural Poor in West Africa, Bamako, Mali, 19–22 November 2001; pp. 1–9. [Google Scholar]
- Carreira, A. Migrações nas Ilhas de Cabo Verde, 2nd ed.; Instituto Cabo Verdiano do Livro: Praia, Cabo Verde, 1983; p. 322. [Google Scholar]
- Trucco, F.; Tranel, P.J. Amaranthus. In Wild Crop Relatives: Genomic and Breeding Resources; Kole, C., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 11–21. [Google Scholar] [CrossRef]
- Senna, M.R.L. Dissertação Sobre as Ilhas de Cabo Verde, 1818. Anotações e Comentários de A. Carreira; Instituto Caboverdiano do Livro: Praia, Cabo Verde, 1987. [Google Scholar]
- Ficalho, C. Plantas úteis da África Portuguesa; Imprensa Nacional: Lisbon, Portugal, 1884; pp. 1–279. [Google Scholar]
- Rocha, A. Subsídios para a História de Santo Antão (1462/1983); Imprensa Nacional de Cabo Verde: Praia, Cabo Verde, 1990; pp. 1–131. [Google Scholar]
- Bozzano, M.; Jalonen, R.; Thomas, E.; Boshier, D.; Gallo, L.; Cavers, S.; Bordács, S.; Smith, P.; Loo, J. Genetic Considerations in Ecosystem Restoration Using Native Tree Species. State of the World’s Forest Genetic Resources—Thematic Study; FAO and Bioversity International: Rome, Italy, 2014; pp. 1–282. [Google Scholar]
- Krigas, N.; Tsoktouridis, G.; Anestis, I.; Khabbach, A.; Libiad, M.; Megdiche-Ksouri, W.; Ghrabi-Gammar, Z.; Lamchouri, F.; Tsiripidis, I.; Tsiafouli, M.A.; et al. Exploring the potential of neglected local endemic plants of three Mediterranean regions in the ornamental sector: Value chain feasibility and readiness timescale for their sustainable exploitation. Sustainability 2021, 13, 2539. [Google Scholar] [CrossRef]
- Romeiras, M.M.; Pena, A.R.; Menezes, T.; Vasconcelos, R.; Monteiro, F.; Paulo, O.S.; Moura, M. Shortcomings of phylogenetic studies on recent radiated insular groups: A meta-analysis using Cabo Verde biodiversity. Int. J. Mol. Sci. 2019, 20, 2782. [Google Scholar] [CrossRef] [Green Version]
- Sánchez-Pinto, L.; Rodríguez, M.L.; Rodríguez, S.; Martín, K.; Cabrera, A.; Marrero, M.C. Pteridophyta, Spermatophyta. In Lista Preliminar de Especies Silvestres de Cabo Verde (Hongos, Plantas y Animales Terrestres): 38–57; Arechavaleta, M., Zurita, N., Marrero, M.C., Martín, J.L., Eds.; Consejería de Medio Ambiente y Ordenación Territorial, Gobierno de Canarias: Canarias, Spain, 2005; pp. 38–57. [Google Scholar]
- Costa, F.L. Contribuições para o conhecimento dos processos erosivos em Cabo Verde. GeoINov 2004, 9, 215–244. [Google Scholar]
- Castilla-Beltrán, A.; Nascimento, L.; Fernández-Palacios, J.M.; Fonville, T.; Whittaker, R.J.; Edwards, M.E.; Nogué, S. Late Holocene environmental change and the anthropization of the highlands of Santo Antão Island, Cabo Verde. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2019, 524, 101–117. [Google Scholar] [CrossRef]
- Paiva, J.; Martins, E.; Diniz, M.A.; Moreira, I.; Gomes, I.; Gomes, S. (Eds.) Flora de Cabo Verde; Fascicles 2, 3, 9, 10, 12, 13, 16, 19, 23, 25, 27, 30, 31, 33, 37, 38, 46, 49, 51, 52, 56, 66,69, 74, 80, 88, 59; Instituto de Investigação Científica Tropical: Lisboa, Portugal; Instituto Nacional de Investigação e Desenvolvimento Agrário: Praia, Cape Verde, 1995. [Google Scholar]
- Paiva, J.; Martins, E.; Diniz, M.A.; Moreira, I.; Gomes, I.; Gomes, S. (Eds.) Flora de Cabo Verde; Fascicles 17, 22, 24, 32, 43, 54, 55, 63a, 64, 72, 77, 78, 85, 67; Instituto de Investigação Científica Tropical: Lisboa, Portugal; Instituto Nacional de Investigação e Desenvolvimento Agrário: Praia, Cape Verde, 1996. [Google Scholar]
- Paiva, J.; Martins, E.; Diniz, M.A.; Moreira, I.; Gomes, I.; Gomes, S. (Eds.) Flora de Cabo Verde; Fascicles 5, 8, 11, 14, 20, 21, 28, 29, 34, 41, 44, 47, 61, 62, 63, 65, 70, 70a, 71, 75, 83, 87, 94, 96, 101, 102, 103, 104, 106; Instituto de Investigação Científica Tropical: Lisboa, Portugal; Instituto Nacional de Investigação e Desenvolvimento Agrário: Praia, Cape Verde, 2002. [Google Scholar]
- Chevalier, A. Les îles du Cap Vert. Géographie, biogéographie, agriculture. Flore Archipel. Rev. Bot. Appl. Agric. Trop. 1935, 15, 733–1090. [Google Scholar]
- Teixeira, A.J.S.; Barbosa, L.A.G. A agricultura do arquipélago de Cabo Verde. Cartas agrícolas. Problemas agrários. Memórias da Junta de Investigações do Ultramar 1958, 2, 1–178. [Google Scholar]
- Nogueira, I. Plantas colhidas pelo Eng. L. A. Grandvaux Barbosa no Arquipélago de Cabo Verde—II. Spermatophyta (Annonaceae-Moringaceae). Garcia Orta Sér. Bot. 1975, 2, 89–106. [Google Scholar]
- Nogueira, I. Plantas colhidas pelo Eng. L. A. Grandvaux Barbosa no Arquipélago de Cabo Verde—III. Spermatophyta (Rubiaceae—Gentianaceae). Garcia Orta Sér. Bot. 1976, 3, 19–32. [Google Scholar]
- Nogueira, I. Plantas colhidas pelo Eng. L. A. Grandvaux Barbosa no Arquipélago de Cabo Verde—VI. Spermatophyta (Nyctaginaceae—Casuarinaceae). Garcia Orta Sér. Bot. 1977, 3, 85–98. [Google Scholar]
- Nogueira, I. Plantas colhidas pelo Eng. L. A. Grandvaux Barbosa no Arquipélago de Cabo Verde—VII. Spermatophyta (Cannaceae—Cyperaceae). Garcia Orta Sér. Bot. 1978–1979, 4, 1–6. [Google Scholar]
- Fernandes, R.B. Cucurbitaceae de Cabo Verde. Garcia Orta Sér. Bot. 1976, 3, 1–14. [Google Scholar]
- Ormonde, J. Plantas colhidas pelo Eng. L. A. Grandvaux Barbosa no Arquipélago de Cabo Verde. IV. Spermatophyta (Leguminosae). Garcia Orta Sér. Bot. 1976, 3, 33–48. [Google Scholar]
- Ormonde, J. Plantas colhidas pelo Eng. L. A. Grandvaux Barbosa no Arquipélago de Cabo Verde. V. Spermatophyta (Rosaceae—Umbelliferae). Garcia Orta Sér. Bot. 1977, 3, 73–80. [Google Scholar]
- Ormonde, J. Plantas colhidas pelo Eng. L. A. Grandvaux Barbosa no Arquipélago de Cabo Verde. V. Spermatophyta (Boraginaceae—Plantaginaceae). Garcia Orta Sér. Bot. 1980, 4, 171–188. [Google Scholar]
- Lewejohann, K.; Lobin, W. Ergänzungen und Anmerkungen zu: O. Eriksson, A. Hansen & P. Sunding—Flora of Macaronesia. Checklist of vascular plants, 2nd revised edition. Garcia Orta Sér. Bot. 1981, 5, 9–11. [Google Scholar]
- Sunding, P. Additions to the vascular flora of the Cape Verde islands II, Botanical Garden and Museum, University of Oslo. Garcia Orta Sér. Bot. 1981, 5, 31–42. [Google Scholar]
- Lobin, W. Additions and corrections to: O. Eriksson, A. Hansen & P. Sunding—Flora of Macaronesia. Checklist of vascular plants, 2nd revised edition—II. Garcia Orta Sér. Bot. 1982, 5, 213–224. [Google Scholar]
- Carter, S.; Lobin, W.; Radcliffe-Smith, A. A collection of and notes on Euphorbiaceae from the Cape Verde Islands (Dicotyledonae). Senckenberg. Biol. 1984, 64, 429–451. [Google Scholar]
- Hansen, A.; Sunding, P. Flora of Macaronesia. Checklist of vascular plants. 3rd rev. ed. Sommerfeltia 1985, 1, 1–167. [Google Scholar] [CrossRef]
- Nogueira, I.; Ormonde, J. Plantas colhidas pelo Eng. L. A. Grandvaux Barbosa no Arquipelago de Cabo Verde—X. Aditamentos e correcções às partes I e VII. Garcia Orta Sér. Bot. 1985, 6, 163–176. [Google Scholar]
- Diniz, C.; Matos, C. Carta de zonagem agro-ecológica e da vegetação. I. Ilha de Santiago. Garcia Orta Sér. Bot. 1986, 8, 39–82. [Google Scholar]
- Diniz, C.; Matos, C. Carta de zonagem agro-ecológica e da vegetação. II. Ilha do Fogo. Garcia Orta Sér. Bot. 1987, 9, 35–70. [Google Scholar]
- Hiemstra, F. Vegetation and range-land of the Planalto Leste area (Santo Antão, Rep. de Cabo Verde). Cour. Forsch.-Inst. Senckenberg 1986, 81, 165–177. [Google Scholar]
- Zizka, G. Chromosomenzählungen bei einigen kapverdischen Pflanzen. Cour. Forsch.-Inst. Senckenberg 1986, 81, 181–182. [Google Scholar]
- Basto, M.F.P. Aditamentos à flora vascular de Cabo Verde. Garcia Orta Sér. Bot. 1987, 9, 1–15. [Google Scholar]
- Basto, M.F.P. Aditamentos à flora vascular de Cabo Verde II. Garcia Orta Sér. Bot. 1993, 11, 75–82. [Google Scholar]
- Hiemstra, F.; Kamminga, P.; Wouters, F. Forest and range management for rural development. ‘Planalto Leste’ Project, Santo Antão, Cape Verde Islands. Cour. Forschungsinst. Senckenberg 1987, 95, 51–55. [Google Scholar]
- Lobin, W.; Zizka, G. Einteilung der Flora (Phanerogamae) der Kapverdischen Inseln nach ihrer Einwanderungsgeschichte. Cour. Forsch. Senckenberg 1987, 95, 127–153. [Google Scholar]
- Zur Strassen, R. Wildpflanzen-Fluren als natürliche Reservoire für potentielle Pflanzenschädlinge unter den kapverdischen Fransenflüglern (Insecta: Thysanoptera). Cour. Forsch. Senckenberg 1987, 95, 65–70. [Google Scholar]
- Rustan, Ø.H.; Brochmann, C. Additions to the vascular flora of Cabo Verde—III. Garcia Orta Sér. Bot. 1993, 11, 31–62. [Google Scholar]
- Duarte, M.C.; Moreira, I. A vegetação de Santiago (Cabo Verde). Apontamento histórico. Garcia Orta Sér. Bot. 2002, 16, 51–80. [Google Scholar]
- Instituto Nacional de Investigação e Desenvolvimento Agrário. Lista de Variedades Hortícolas Recomendadas em Cabo Verde. 2012. Available online: http://www.portaldoconhecimento.gov.cv/bitstream/10961/1920/1/Lista%20de%20Variedades%20Hort%C3%ADculas%20Recomendadas%20%20em%20Cabo%20Verde.pdf (accessed on 24 February 2022).
- Cook, F.E.M. Economic Botany Data Collection Standard; Royal Botanic Gardens: Kew, UK, 1995; p. 146. [Google Scholar]
- Morrone, J.J. Biogeographical Regions under Track and Cladistic Scrutiny. J. Biogeogr. 2002, 29, 149–152. [Google Scholar] [CrossRef] [Green Version]
- Instituto Nacional de Estatística. Estatísticas do Ambiente 2016. Available online: https://ine.cv/publicacoes/estatisticas-do-ambiente-2016/ (accessed on 14 February 2022).
- Instituto Nacional de Estatística. Avaliação de Impacto da Pandemia da COVID-19—Inquérito Rápido às Empresas 2º Trimestre 2020. Available online: https://ine.cv/publicacoes/avaliacao-impacto-da-pandemia-da-covid-19-inquerito-rapido-as-empresas-2o-trimestre/ (accessed on 15 February 2022).
- Ministério da Agricultura e Ambiente. V. Recenseamento Geral da Agricultura 2015. Principais Resultados Definitivos; Ministério da Agricultura e Ambiente: Praia, Cabo Verde.
- Ministério do Desenvolvimento Rural. Inventário Florestal Nacional de Cabo Verde. 2012. Available online: http://www.caboverdeifn.ifer.cz/?page_id=8 (accessed on 10 February 2022).
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna. Available online: https://www.R-project.org/ (accessed on 14 January 2022).

| Taxa | Common Names | Uses | Hist. |
|---|---|---|---|
| Acanthaceae | |||
| Dicliptera verticillata | Joelho, orelha-de-rato, rapazinho | Fr | |
| Aizoaceae | |||
| Zaleya pentandra | Erva-de-rapé | S,O | |
| Amaranthaceae | |||
| Aerva javanica | Florinha, panasco | U | |
| Amaranthus graecizans subsp. graecizans | Bredo, bredo-femba | Fr | |
| Arthrocaulon franzii | Murraça, murraça-rosa-crioula, murraçona | Fu | |
| Celosia trigyna | Fd | ||
| Patellifolia procumbens | Selga | Fr | |
| Apiaceae | |||
| *Tornabenea annua [a] | Funcho, funtcho, futcho-bravo | Fr | |
| *Tornabenea tenuissima [a] | Aipo, funtcho | Fr | |
| *Tornabenea bischoffii [a] | Funcho | Fr | |
| Apocynaceae | |||
| Calotropis procera | Bombardeiro | Fr,Fu,U | ● |
| *Periploca chevalieri | Corcabra, curcabra, lantisco, lentisco | Fr,Mt | ● |
| Arecaceae | |||
| *Phoenix atlantica | Tamareira | Fd,Fr,U,O,E | |
| Asparagaceae | |||
| *Dracaena caboverdeana [b] | Dragoeiro | Mt,O,E | ● |
| Asteraceae | |||
| *Artemisia gorgonum | Losna | P | |
| *Asteriscus daltonii subsp. vogelii | Macela, marcela, marcela-lenha | P | |
| Blainvillea gayana | Barba-bodi, cachacinho, erva-moura, loura, targa, targa-branco | Fr | |
| *Conyza feae [c] | Losna-brava, marcela, marcelinha, mato-contrário, palha-santa | Fr | |
| Launaea arborescens | Carqueja, craquejo | Fu | |
| Launaea intybacea | Algodão-de-garça, paja-leite, palha-de-leite, serralha | Fd | |
| *Launaea picridioides | Marê-tope, serragem, serralha, tortolhinha, tortolhinho | Fr | |
| *Launaea thalassica | Serralha, tortolhinha | Fr | |
| Pseudoconyza viscosa | Butra, talga, vampiro | Fr | |
| *Pulicaria diffusa | Losna | P | |
| *Sonchus daltonii | Coroa-de-rei | Fr | |
| Sonchus oleraceus | Algodão-de-graça, leituga, palha-leite, sarralha, serralha | Fr | |
| Vernonia colorata | Catchiça | Fr | |
| Boraginaceae | |||
| *Echium hypertropicum | Língua-de-vaca, língua-di-baca | Fr,Fu | |
| *Echium stenosiphon subsp. glabrescens | Língua-de-vaca | Fr | |
| *Echium stenosiphon subsp. lindbergii | Língua-de-vaca | Fr | |
| *Echium stenosiphon subsp. stenosiphon | Língua-de-vaca | Fr | |
| *Echium vulcanorum | Língua-de-vaca | Fr,Fu | |
| Heliotropium ramosissimum | Alfavaca, alfavaca-da-achada, erva-das-sete-sangrias, furtaragem, mama-de-bitcho, tchero-fede, três-marias | Fr | |
| Brassicaceae | |||
| *Diplotaxis glauca | Matona, mostarda, mostarda-braba | Fr | |
| *Diplotaxis varia | Mostarda-braba | Ml | |
| *Lobularia canariensis subsp. spathulata | O | ||
| *Lobularia canariensis subsp. fruticosa | Sempre-noivinha | Ml,O | |
| Caryophyllaceae | |||
| *Polycarpaea gayi | Cidreira-de-rocha, mato-branco, palha-bidião, palha-de-bidion, talim, talinho, telim | U | |
| Cistaceae | |||
| *Helianthemum gorgoneum | Matinho, piorno-de-flor-amarela | Fr | |
| Commelinaceae | |||
| Commelina benghalensis | Grande-rato, orelha-de-rato, palha-de-água | Fr | |
| Convolvulaceae | |||
| Distimake aegyptius | Maraganha, n’onhen’onhe, palha-corda | Fr | |
| Ipomoea eriocarpa | Cordinha, jejé-calabaceira, lagaço-cozinho, legação-cabecinho, monhe-monhe, monho-monho | Fr | |
| Ipomoea pes-caprae subsp. brasiliensis | Lacacã, lacacã-grande, lacacan-de-vaca, legação-de-rocha | E | |
| Crassulaceae | |||
| *Aeonium gorgoneum | Ceilão, mata-sede, saião, seilão, sião | O | |
| Cucurbitaceae | |||
| Citrullus colocynthis | Balancia-brabo, melancia-brava, melão-bravo, olho-de-boi, olho-de-vaca | Fr | |
| Momordica charantia | Aboboreira-de-são-caetano, banana-rato, erva-de-são-caetano, palha-de-são-caetano, sancaetano, são caetano | Fd,Ml,Mt | |
| Cyperaceae | |||
| Bulbostylis barbata | Soldinha | Fr,Ml | |
| Cyperus alternifolius subsp. flabelliformis | Chapeudisol, goia, junco | Fr,U | |
| Cyperus articulatus | Goia, junco | Fr,U | |
| Cyperus esculentus | Djunça, junça, vista | Fd,Fr | |
| Cyperus hortensis | Fr,U | ||
| Cyperus rotundus | Grama, guel, injunça, junça, junco | Fd,Fr | |
| Fimbristylis ferruginea | Junco, junquinho | Fr | |
| Equisetaceae | |||
| Equisetum ramosissimum | Carsim, cavalinha, talim | S | |
| Euphorbiaceae | |||
| *Euphorbia tuckeyana | Tira-olho, tortilho, tortodjo, tortolho | Fu,Mt | ● |
| Fabaceae | |||
| Abrus precatorius subsp. africanus | Jequeriti, santa-clara | Fr,U | |
| *Acacia caboverdeana [b] | Espinheiro-branco, neu-neu (fruits) | Fr,Fu,E | ● |
| Alysicarpus ovalifolius | Fr | ||
| Clitoria ternatea | Bachinha-de-cordoniz, palha-lopes | Fr | |
| Crotalaria senegalensis | Ovos-de-rato, ovos-de-rato-pequeno | Fr | |
| Desmodium ospriostreblum | Crioulinha | Fr | |
| Dichrostachys cinerea | Espinheiro, espinheiro-branco, espinheiro-cachupa, espigo-de-cachupa, espinho-cachupa, espinho-catchupa | Fu | |
| Genista stenopetala | E | ||
| Grona hirta | Maratchinga, marquinha | Fr,E | |
| Lablab purpureus subsp. purpureus | Creca, feijão-branco-de-vagem-branca, feijão-caqui, feijão-careca, feijão-cutelinho, feijão-pedra, feijão-pedra-bombone, feijão-vaca | Fd,Fr | ● |
| *Lotus brunneri | Cabritagem, cafetalha, cafetagem, piorno-amarelo, piorno-preto | Fr,P | |
| *Lotus jacobaeus | Piorno, piorno-preto | Fr | |
| *Lotus purpureus | Piorno, piorno-amarelo | Fr | |
| Macrotyloma daltonii | Corda-lopes, cordeirinha-preta, favalinha, feijoeiro-de-lagartiga | Fr | |
| Rhynchosia minima var. memnonia | Feijoeiro-de-lagartixa | Fr | |
| Sesbania pachycarpa | Acácia-sizinanthe, sesinanthe, ticome-se | Fr | |
| Stylosanthes fruticosa | Fr | ||
| Tephrosia linearis | Fr | ||
| Tephrosia purpurea | Mt | ||
| Teramnus labialis subsp. arabicus | Caransaqui, corda-lopes-pequena, cordeirinha-branca | Fr | |
| Vachellia nilotica subsp. adstringens | Acácia | Ml,O,E | |
| Vigna unguiculata subsp. unguiculata | Bongolon-d’angola, feijão-bezugo, feijão-bongolon, feijão-bongolon-amarelo, feijão-bongolon-com-boca-preta, feijão-congo | Fd,Fr | ● |
| Frankeniaceae | |||
| *Frankenia caboverdeana [b] | Mato-de-engodo, mato-de-sargaço, palha-engodo | U | |
| Lamiaceae | |||
| Lavandula coronopifolia | Marmulano-da-terra, risco, risque | Fr | |
| *Lavandula rotundifolia | Aipo, alfazema-brava, gilbon | Fr | |
| Ocimum americanum | Fd | ||
| Salvia aegyptiaca | Alfazema, alfazema-da-terra,bálsamo-de-pastor, ermofassima, malfazema, marcelina, rosmaninho | Fr | |
| Malvaceae | |||
| Grewia villosa | Balneda, barnadeiro, barnedo, barneldo, barnelo | Fd,Ml,U | |
| Melhania ovata | Lolo-branco, mato-branco, salva-vidas | Fd | |
| Sida rhombifolia | Lolo, loulo, loulo-preto-grande | Fr,U | |
| Urena lobata | Bassago | U | |
| Moraceae | |||
| Ficus sur | Figueira, figueira-brava, figueira-preta | Fd,Fr,T | ● |
| Ficus sycomorus | Figueira-branca, figueira-brava, figueira-de-figos-grandes | Fd,Fr,T,E | ● |
| Nyctaginaceae | |||
| Boerhavia coccinea | Albeza, batata-de-asno, batata-de-burro, batata-de-oze, cordeira, costa-branca, costa-branca-fêmea, mato-branco | Fr | |
| Boerhavia diffusa | Albeza, batata-de-burro, costa-branca, costa-branca-fêmea | Fr,Ml | |
| Boerhavia repens | Costa-branca, costa-branca-fina, costa-branca-miúda, folha-branca, palha-branca, palha-costa, palha-seca | Fr | |
| Commicarpus helenae | Albeça-branca, albéza-branco, butra, costa-branca, costa-branca-bastarda, folha-branca, mato-branco, mato-lagarto | Fr | |
| Plantaginaceae | |||
| *Globularia amygdalifolia | Argueiro, mato-botão, medronho | Fr | |
| Poaceae | |||
| Andropogon gayanus var. tridentatus | Palha-ladeira, touça, touça-fêmea | Fr | |
| Bothriochloa bladhii | Touça, touça-macho, palha-cavalo | Fr,E | |
| Cenchrus ciliaris | Balanco, palha-branca, palha-grossa, rabo-de-gato | Fr | |
| Cenchrus pedicellatus subsp. pedicellatus | Balanco-branco | Fr | |
| Cenchrus pedicellatus subsp. unispiculus | Balanco-branco | Fr | |
| Chloris gayana | Fr | ||
| Chloris pilosa | Fr | ||
| Dactyloctenium aegyptium | Djinguilano, jéjé-jiuguilam, palha-de-boi-fraca, pé-de-galinha | Fd,Fr | |
| Dichanthium annulatum | Palha-soca, soca, touça-fêmea | Fr,U | |
| Dichanthium foveolatum | Palha-fina, palha-minha, palha-sisuda, sisuda | Fr | |
| Digitaria ciliaris | Djé-djé-cinha, djé-djé-pequeno, djeiezinho | Fr | |
| Digitaria horizontalis | Gé-gé, jéjézinho | Fr | |
| Digitaria nodosa | Palha-carriço, palha-grossa | Fr | |
| Echinochloa colonum | Djé-djé-pequeno | Fr | |
| Eleusine indica subsp. indica | Barba-de-bode, palha-boi, palha-grossa | Fr | |
| Eragrostis cilianensis | Djé-djézinho | Fr | |
| Eragrostis ciliaris | Padja-do-menino-jesus, palhinha | Fr,U | |
| Eragrostis minor | Fr | ||
| Hackelochloa granularis | Fr | ||
| Heteropogon contortus | Azagaia, rabo-de-asno, soca-mansa, touça-mansa, toussa-matcho | Fr,E | |
| Heteropogon melanocarpus | Zagaia | Fr | |
| *Hyparrhenia caboverdeana [b] | Palha-de-guiné, palha-negra, touça, touça-fêmea | Fr | |
| Imperata cylindrica | Palha-carga | Fr,U | ● |
| Melinis minutiflora | Palha-governo, palha-mafe, palha-mafó | Fr | |
| Paspalum scrobiculatum | Patacho | Fr | |
| Polypogon viridis | Graminho, palha-de-água | Fr | |
| Rottboellia cochinchinensis | Palha-grossa | Fr | |
| Schizachyrium brevifolium | Fr | ||
| Setaria barbata | Djé-djé-palha-de-água, jéjé | Fd,Fr | |
| Setaria pumila | Gé-gé-pequeno | Fr | |
| Setaria verticillata | Pega-saia | Fr | |
| Tricholaena teneriffae | Palha-branca, palha-de-vassoura | Fr | |
| *Urochloa caboverdiana | Dje-dje, jé-jé | Fd,Fr | |
| Urochloa ramosa | Djé-djé, jé-jé, jé-jé-folha-larga | Fd,Fr | |
| Urochloa xantholeuca | Djé-djé | Fr | |
| Portulacaceae | |||
| Portulaca oleracea | Beldroega, bordulega, brêdo-fêmea, sangue-sangria | Fd,Fr,Ml | |
| Pteridaceae | |||
| Adiantum capillus-veneris | Aibenca, avenca | O | |
| Resedaceae | |||
| Caylusea hexagyna | Campa, laca-laca, laga-laga, palha-lagada, piorno | Fr,Ml | |
| Rhamnaceae | |||
| Ziziphus mauritiana | Simbrom, zimbrão, zimbreiro-da-índia | Fd,Fr,Ml,Fu,T,E | |
| Rubiaceae | |||
| Oldenlandia corymbosa var. corymbosa | Fr | ||
| Sapindaceae | |||
| Cardiospermum halicacabum | Conta-de-cavalo | O | |
| Dodonaea viscosa | O | ||
| Sapotaceae | |||
| *Sideroxylon marginatum | Figueira-de-macaco, marmulano, marmolano | Fd,Fr,Fu | ● |
| Solanaceae | |||
| Solanum nigrum | Malagueta-de-galinha, uva-catchorro, uva-de-santa-maria | Fd | |
| Solanum scabrum | Fd | ||
| Tamaricaceae | |||
| Tamarix senegalensis | Tarafe, tarrafe, tamargueira | Fu,O,E | ● |
| Typhaceae | |||
| Typha domingensis | Palha-das-esteiras, tabúa | U | |
| Urticaceae | |||
| *Forsskaolea procridifolia | Língua-de-vaca-branca, mato-gonçalo, ortiga, palha-renda, pega-saia, rafa-saia, rapa-saia, urtiga | Fr | |
| Zygophyllaceae | |||
| Fagonia cretica | Arroz-de-pardal, cabritaia-do-campo, matinho-de-agulhas | Fr,P | |
| Fagonia latifolia [c] | Cabaceira, matinho | Fr | |
| Tetraena gaetula subsp. waterlotii [c] | Acelga-de-água, bidion, fuminga, morraça-branca, murraça | P,Fu,E | |
| *Tetraena vicentina | E |
| Taxa | Common Names | Uses | Hist. |
|---|---|---|---|
| Acanthaceae | |||
| Eranthemum pulchellum | Flor-viúva | O | |
| Pseuderanthemum maculatum | Dakarense | Ml | |
| Aizoaceae | |||
| Carpobrotus edulis | Bálsamo | O,E | |
| Tetragonia tetragonioides | Espinafre-da-nova-zelândia | Fd | |
| Amaranthaceae | |||
| Alternanthera sessilis | Abri-olho, abrodjo, arre-porra, mão-na-pé, mon-na-pé | Fr,O | |
| Amaranthus blitum | Bredo | Fd | |
| Amaranthus caudatus | Bredo-macho | Fd,Fr,O | |
| Amaranthus cruentus | Crista-de-perú | Fd,Fr | |
| Amaranthus hybridus subsp. hybridus | Bredo-macho, rabo-de-galo | Fr | |
| Amaranthus spinosus | Bredo, bredo-com-espinhos, bredo-espinhoso, bredo-macho | Fd | |
| Amaranthus tortuosus | Bredo, bredo-macho | Fr | |
| Amaranthus viridis | Bredo-sem-espinhos | Fd,Fr | |
| Atriplex halimus | Fr,O,E | ||
| Beta vulgaris | Beterraba | Fd,Fr,Mt | |
| Gomphrena globosa | O | ||
| Amaryllidaceae | |||
| Allium ampeloprasum | Alho-francês | Fd | |
| Allium ascalonicum | Chalota | Fd | |
| Allium cepa | Cebola | Fd | |
| Allium fistulosum | Cebolinha | Fd | |
| Allium sativum | Alho | Fd | |
| Allium schoenoprasum | Cebolinha-miúda | Fd | |
| Hymenocallis littoralis | Lírio | Ml,O | |
| Scadoxus multiflorus | O | ||
| Anacardiaceae | |||
| Anacardium occidentale | Cadju, cajueiro, cajuleiro | Fd,Ml,T,E | |
| Mangifera indica | Mangue, mangueira | Fd,Fr,Ml | |
| Schinus molle | Pimenteira, pimenteira-bastarda, pimenta-rosa | Fd,P,O,E | |
| Schinus terebinthifolia | Pimenteira | O | |
| Sclerocarya birrea subsp. caffra | Ocanho | Fd | |
| Spondias mombin | Mamipreiro, manipo | Fd,Ml | ● |
| Annonaceae | |||
| Annona cherimola | Cherimolia | Fd | |
| Annona muricata | Pinha, pinhão, pinhão-azedo, sap-sap | Fd,O | |
| Annona reticulata | Anoneira, coração-de-boi | Fd,O | |
| Annona squamosa | Pinha, pinho | Fd | ● |
| Apiaceae | |||
| Anethum graveolens | Endro, ente, entro, erva-doce | Fd,Fr | |
| Apium graveolens | Aipo | Fd | |
| Coriandrum sativum | Coentro, cuentro | Fd | |
| Daucus carota | Cenoura | Fd | |
| Foeniculum vulgare | Erva-doce, funcho, funcho-gomado | Fd | ● |
| Petroselinum crispum | Salsa | Fd | |
| Apocynaceae | |||
| Asclepias curassavica | Cravo, pitchula-de-leite | Ml,O | |
| Cascabela thevetia | Chapéu-de-napoleão, mundium | Ml,O | |
| Catharanthus roseus | Bigalo, flor-de-anjo, flor-de-finado, sempre-noiva | Ml,O | |
| Nerium oleander | Cevadilha, loendro, loureiro-rosa, rosa, rosa-loira, roseira-branca-singela, sempre-noiva-branca, sevadilha | P,O | |
| Plumeria rubra | O | ||
| Araceae | |||
| Caladium bicolor | O | ||
| Colocasia esculenta | Inhame, mafafa, malanca, muncoco | Fd | |
| Xanthosoma sagittifolium | Inhame, mafafa-preta, malanca, mincoco | Fd | |
| Arecaceae | |||
| Borassus flabellifer | Cibe | O | |
| Cocos nucifera | Coqueiro | Fd,Ml,O,E | ● |
| Elaeis guineensis | Coconote, dem-dem, palmeira-do-azeite | Ml,O | |
| Phoenix canariensis | O | ||
| Phoenix dactylifera | Palmeira-do-saará, tamareira, tamareira-do-saará | Fd,O,E | ● |
| Washingtonia filifera | Palmeira-leque | O | |
| Aristolochiaceae | |||
| Aristolochia littoralis | O | ||
| Asparagaceae | |||
| Agave americana | Ml | ||
| Agave sisalana | Carapate-manila, carrapato-de-lisboa, pita, sisal | P,U,O | |
| Asparagus officinalis | Espargo | Ml | |
| Dracaena hyacinthoides | O | ||
| Furcraea foetida | Carapate, carrapato, carrapato-da-terra, piteira-de-cabo-verde | P,U,O,E | ● |
| Asphodelaceae | |||
| Aloe vera | Aloés, babosa | Ml,S,O,E | |
| Asteraceae | |||
| Bidens bipinnata | Gúia, seta, seta-branca, seta-preta | Ml | |
| Bidens pilosa | Agulha, gua, palha-agulha, seta, seta-preta, setinha | Fr,Ml | |
| Calendula arvensis | O | ||
| Cichorium endivia | Endivia | Fd | |
| Cichorium intybus | Chicória | S,O | |
| Helianthus annuus | Girassol | Ml | |
| Lactuca sativa | Alface | Fd | |
| Synedrella nodiflora | Targa | Fr,Ml | |
| Tagetes erecta | Cravo, cravo-branco, cravo-de-burro | Ml,O | |
| Tanacetum parthenium | Altamires | O | |
| Urospermum picroides | Palha-leite, palha-leite-amarga, raposade, serralha | Fr,Ml | |
| Zinnia peruviana | Cravo, cravo-branco, zinha, zinia | Fr,O | |
| Basellaceae | |||
| Anredera cordifolia | O | ||
| Basella alba | Tinta-de-macaca, tinta-de-macaco | Mt,O | |
| Bignoniaceae | |||
| Crescentia cujete | Cabaceira, calabaceira | U | |
| Dolichandra unguis-cati | Unha-de-gato | O | |
| Handroanthus impetiginosus | Pau-d’arco | S,Fu,Mt,O | |
| Jacaranda mimosifolia | Jacandrão | O,E | |
| Kigelia africana subsp. africana | O | ||
| Spathodea campanulata | Árvore-da-chama, tulipeira-do-gabão, tulipa-do-gabão | O | |
| Tabebuia rosea | Farroba | O,E | |
| Tecoma stans | Ervilha-de-flor | O | |
| Boraginaceae | |||
| Cordia sebestena | O | ||
| Heliotropium arborescens | Baunilha, baunilha-de-cheiro | O | |
| Brassicaceae | |||
| Barbarea verna | Agrião-de-terra | Fd | |
| Brassica juncea | Mostarda | Fd | |
| Brassica nigra | Mostarda, mostarda-branca, mostarda-brava, mostarda-preta | Fd,Ml | |
| Brassica oleracea | Couve | Fd,Fr | |
| Brassica rapa | Couve-chinesa, nabo | Fd | |
| Eruca vesicaria | Rúcula | Fd | |
| Lobularia maritima | Sempre-noiva | O | |
| Matthiola maderensis | O | ||
| Nasturtium officinale | Agrião, agrião-de-água, agrião-vulgar | Fd | |
| Raphanus raphanistrum subsp. sativus | Rábano, rabanete | Fd | |
| Bromeliaceae | |||
| Ananas comosus | Ananaseiro | Fd | ● |
| Cactaceae | |||
| Opuntia ficus-indica | Figueira-da-índia, figueira-do-inferno, tabaibo | Fd,Fr,Ml,O | ● |
| Pereskia aculeata | O | ||
| Selenicereus undatus | Barse, pilahayo | Fd,O | |
| Calophyllaceae | |||
| Mammea americana | Abricó-do-pará, mamão, mamoeiro | Fd | ● |
| Cannaceae | |||
| Canna indica | Cana-da-índia, coqueirinho, coqueirinho-de-jardim, lírio | O | |
| Caprifoliaceae | |||
| Lonicera confusa | Madressilva, madressilva-de-cheiro | O | |
| Caricaceae | |||
| Carica papaya | Bijagó-preta, papaeira | Fd | ● |
| Caryophyllaceae | |||
| Dianthus caryophyllus | O | ||
| Casuarinaceae | |||
| Allocasuarina verticillata | E | ||
| Casuarina equisetifolia | Casuarina | E | |
| Combretaceae | |||
| Terminalia catappa | Amendoeira, amendoeira-da-índia | Fd,T,O | |
| Commelinaceae | |||
| Tradescantia zebrina | O | ||
| Convolvulaceae | |||
| Argyreia nervosa | O | ||
| Ipomoea batatas | Batata, batata-belém, batata-doce, batata-doce-preta, batata-malevinho, batata-quarenta-dias, corda-copo, cordinha, giginha-muralha, nhá-júlia, pau-de-vinho, quirino, temerosa | Fd,Fr,Ml | ● |
| Ipomoea carnea | O | ||
| Ipomoea muricata | Calabaceira | O | |
| Ipomoea tuberculata | Rosas-de-madeira | Fr,O | |
| Crassulaceae | |||
| Kalanchoe daigremontiana | Bálsamo | O | |
| Kalanchoe pinnata | Bálsamo, figueirinha | O | |
| Cucurbitaceae | |||
| Citrullus lanatus | Melancia | Fd | ● |
| Cucumis anguria | Pepino-bravo, pepino-de-macaco, pepino-sanjo, pepino-santcho | Fr | |
| Cucumis melo | Melão | Fd | ● |
| Cucumis sativus | Pepino | Fd | |
| Cucurbita maxima | Abóbora-roca, aboboreira, roca | Fd,Ml | |
| Cucurbita moschata | Abóbora-de-sequeiro-de-porco, abóbora-jarda, aboboreira | Fd,Ml | |
| Cucurbita pepo | Aboboreira, aboboreira-vulgar | Fd,Ml | ● |
| Lagenaria siceraria | Abobreira-de-cabaça, buli, cabaça, cabaceira | Fd,U | |
| Luffa aegyptiaca | Bobra | U | |
| Cupressaceae | |||
| Cupressus sempervirens | Cupressus | Fu,E | |
| Hesperocyparis arizonica | E | ||
| Hesperocyparis lusitanica | Cedro-português, cedro-do-buçaco | Fu,E | |
| Hesperocyparis macrocarpa | E | ||
| Dioscoreaceae | |||
| Dioscorea japonica | Fd | ||
| Euphorbiaceae | |||
| Acalypha wilkesiana | O | ||
| Breynia disticha | Groselhinha | O | |
| Euphorbia chamaesyce | Solda-inglesa | Fr,P | |
| Euphorbia heterophylla | Travador | Ml | |
| Euphorbia hirta | Desfamador, erva-santa-luzia, itervina, marcelinha, marcelintra, palha-pico, solda-inglesa, solda-inglesa-grande | Fr,Ml | |
| Euphorbia hyssopifolia | Padja-lete, palha-leite, palha-soda, solda-inglesa | Fr | |
| Euphorbia milii | Coroa-de-cristo | Ml | |
| Euphorbia pulcherrima | O | ||
| Euphorbia splendens | O | ||
| Euphorbia tirucalli | Borracha, mato-leitoso | O | |
| Euphorbia tithymaloides | O | ||
| Jatropha curcas | Purga, purgueira | Mt,E | ● |
| Jatropha gossypiifolia | Chagas-velhas, purgueira, purgueira-da-guiné | Fr | |
| Jatropha multifida | Purgueira-da-guiné | O | |
| Manihot carthagenensis subsp. glaziovii | Borracheira, mandioqueira-borracha | Mt,O | |
| Manihot esculenta | Mandioca, mandioca-borracha, mandioca-branca, mandioca-brasil, mandioca-mulata, mandioca-pau-de-terra | Fd,Fr,Ml | ● |
| Ricinus communis | Bafureira, djague, djague-djague, jag-jag, mamona, rícino | Fr,Ml | ● |
| Fabaceae | |||
| Acacia bivenosa | E | ||
| Acacia brachystachya | E | ||
| Acacia cyclops | E | ||
| Acacia holosericea | Alosericia, oredjona | Ml,E | |
| Acacia longifolia | E | ||
| Acacia mearnsii | E | ||
| Acacia pycnantha | E | ||
| Acacia salicina | E | ||
| Acacia saligna | E | ||
| Acacia victoriae | E | ||
| Adenanthera pavonina | Coral | O | |
| Albizia lebbeck | Pau-feijão | Ml,O,E | |
| Arachis hypogaea | Amendoim, mancarra | Fd,Fr | ● |
| Bauhinia galpinii | O | ||
| Bauhinia monandra | O | ||
| Caesalpinia pulcherrima | Barbas-de-barata, brinco-de-princesa | O | ● |
| Cajanus cajan | Congo, feijão-congo, feijão-ervilha, feijão-figueira | Fd,Fr | ● |
| Canavalia ensiformis | Fava-rica | Fd,Fr | |
| Cassia fistula | Canafístula, canafrista, jardim | O | ● |
| Ceratonia siliqua | Alfarrobeira | Fd,Fr,E | ● |
| Chamaecytisus prolifer | Fr | ||
| Colophospermum mopane | O | ||
| Crotalaria retusa var. retusa | Bons-dias, flor-de-lagartixa, gaivé, ovos-de-rato | Fr | |
| Delonix regia | Acácia-rubra | Ml,O | |
| Desmanthus virgatus | Bencaiumba, bom-de-caimbra, caiumbra, quintinha, sementinha | Fr,Ml | |
| Desmodium tortuosum | Crioula, crioula-fina, crioula-pequena, marquinha | Fr | |
| Erythrina variegata | O | ● | |
| Erythrina velutina | Fabatera | O | |
| Erythrostemon gilliesii | Barbas-de-barata | Ml,O | |
| Gliricidia sepium | Fr,O | ||
| Guilandina bonduc | Ouri, uri, uriseira | U | |
| Indigofera tinctoria | Tinta | Mt | ● |
| Leucaena leucocephala | Acácia, acácia-leucena, linhaça, linhacho, sementinha-da-terra | Fr,Ml | |
| Libidibia coriaria | Crisalpina | Mt,O | |
| Lonchocarpus sericeus | O | ||
| Medicago sativa | Anafe, luzerna | Fr | |
| Millettia thonningii | O | ||
| Mucuna pruriens | Canhoma, feijão-de-bitcho, feijão-de-lagartixa, ganhoma | Fr | |
| Parkia biglobosa | Alfarroba-da-guiné | O | |
| Parkinsonia aculeata | Acácia, acácia-espinheiro, acácia-martins, espinho-branco | Fr,Ml,E | |
| Phaseolus lunatus | Banjona, bonjinho, fava, fava-terra, favona, feijão, feijão-bombone-branco, feijão-bonge, feijão-espadinha, feijão-fava | Fd | ● |
| Phaseolus vulgaris | Bonje, favona, feijão, sapatinha | Fd | ● |
| Pithecellobium dulce | Mampisa, roseira | Fd,O | |
| Prosopis chilensis | E | ||
| Prosopis juliflora | Acacia-americana, algaroba | Fr,Ml,O,E | |
| Prosopis pallida | E | ||
| Prosopis tamarugo | E | ||
| Samanea saman | Pau-feijão | Fd,Fr | |
| Senegalia senegal | E | ||
| Senna artemisioides nothosubsp. sturtii | E | ||
| Senna bicapsularis | Beijinho, canafístula, flor, jardim, jardineira | O | |
| Senna corymbosa | O | ||
| Senna obtusifolia | O | ||
| Senna occidentalis | Baguinha, canafista, empincheira, fedegosa, munhanóca, pincheira, trincheira | Ml,S | |
| Senna septemtrionalis | O | ||
| Senna siamea | O | ||
| Sesbania grandiflora | Cacia-japónica | Fd,Ml,O | |
| Tamarindus indica | Tamarindeiro, tamarindo, tambarindo, tambarino | Fd,Fr,Fu,O,E | ● |
| Tara spinosa | Tara-do-chile | O | |
| Tipuana tipu | O | ||
| Trifolium glomeratum | Bonança, trevo | Fr | |
| Vachellia farnesiana | Acácia-esponja, aroma, espinheiro-branco, espinheiro-preto, espinho-branco, espinho-preto, esponjeira, imbulda, perfume | Ml,Mt,O,E | ● |
| Vachellia nilotica subsp. indica | Acácia, espinheira, espinheiro-preto, espinho-preto | Fr,Fu,Mt,O,E | |
| Vachellia seyal | E | ||
| Vachellia tortilis | E | ||
| Geraniaceae | |||
| Pelargonium × hybridum | O | ||
| Pelargonium inquinans | O | ||
| Pelargonium zonale | Malva-sardinheira | O | |
| Iridaceae | |||
| Iris florentina | Lírio-branco, tulipa-branca | O | |
| Lamiaceae | |||
| Clerodendrum speciosissimum | Rosaquina, rosa-quina | O | |
| Clerodendrum umbellatum | O | ||
| Lavandula dentata | Rosmaninho | O | |
| Leonurus sibiricus | O | ||
| Mentha × wirtgeniana | Bergamota, hortolô, hortelã | Fd | |
| Mentha pulegium | Poeijos | Fd | |
| Mentha x piperita | Ortelã-pimenta | Fd | |
| Ocimum basilicum | Mangericão, mangerona, mangirão, mangirona | Fd | ● |
| Ocimum gratissimum | Fd | ||
| Origanum vulgare | Mangerona-selvagem | Fd | |
| Salvia coccinea | Trepadeira-de-lisboa | O | |
| Salvia eriocalyx | Salva | O | |
| Salvia leucantha | O | ||
| Salvia rosmarinus | Alecrim, alecrim-de-portugal | Fd | |
| Tectona grandis | Teca | T | |
| Volkameria aculeata | O | ||
| Lauraceae | |||
| Cinnamomum burmanni | Caneleira | Fd | |
| Cinnamomum camphora | Árvore-de-cânfora, canforeira | O | |
| Cinnamomum verum | Caneleira | Fd,O | |
| Laurus nobilis | Loureiro | Fd,P | |
| Persea americana | Abacate, abacateiro | Fd,Ml | |
| Loasaceae | |||
| Mentzelia aspera | Lapadeira, pega-saia, rapo-tchapo, rato-tchapo | Ml | |
| Lythraceae | |||
| Punica granatum | Romã, romangeira, romanzeira, romãzeira | Fd,Mt,O | ● |
| Malvaceae | |||
| Abelmoschus esculentus | Quiabo | Fd | ● |
| Abutilon grandifolium | Vara-de-lobo | Mt | |
| Adansonia digitata | Calabaceira, caxabuceira, imbondeiro | Fd,Ml | ● |
| Ceiba pentandra | Poilão, polon | Fd,Ml,U,E | |
| Cola lateritia | Amoreira, maria-cujá, moreira | Fd,O | |
| Gossypium hirsutum | Algodão, algodoeiro, algodoeiro-vulgar | U | ● |
| Hibiscus cannabinus | Malva-brava | Fd,U | |
| Hibiscus rosa-sinensis | Cardeal, cardiais | Fr,Ml,O | |
| Hibiscus sabdariffa | Bissap | Fd | ● |
| Hibiscus surattensis | O | ||
| Sida salviifolia | Lol-branco, lôlo-preto | Fr,U | |
| Thespesia populnea | Bela-sombra | O | |
| Meliaceae | |||
| Azadirachta indica | Primo-de-morôdjo | P,E | |
| Khaya senegalensis | Mogno | Ml,E | |
| Melia azedarach | Intendente, tendente, tindint, viúva | T,U,O,E | |
| Trichilia emetica | Mafureira, mafurra, mafurreira, mufurreira | Fd,O | |
| Moraceae | |||
| Artocarpus altilis | Fruta-pão | Fd | |
| Artocarpus heterophyllus | Jaqueira | Fd | |
| Artocarpus integer | Jaqueira | Fd | |
| Ficus benjamina | Figueira-brava-da-índia | O | |
| Ficus carica | Figueira, figueira-de-portugal | Fd | ● |
| Ficus elastica | Borracheira | O | |
| Ficus leonensis | O | ||
| Ficus lutea | Lemba-lemba | O | |
| Ficus religiosa | Figueira-de-goa, figueira-da-índia | O | |
| Ficus thonningii | Fr,O | ||
| Morus nigra | Amoreira, morreira | Fd | |
| Moringaceae | |||
| Moringa oleifera | Acácia-blanco, acácia-branca, moringa | Fd,Fr,Mt,O | |
| Musaceae | |||
| Musa × paradisiaca | Banana-pão, bananeira | Fd,Ml | ● |
| Myrtaceae | |||
| Corymbia citriodora | O,E | ||
| Eucalyptus camaldulensis subsp. camaldulensis | Calipe, calipo, calipto, eucalipto | Ml,O,E | |
| Eucalyptus globulus | Calipe, calipo, calipto, eucalipto | P,O | |
| Eucalyptus gomphocephala | Calipe, calipo, calipto, eucalipto | E | |
| Eucalyptus pruinosa | Calipe, calipo, calipto, eucalipto | O | |
| Eucalyptus tereticornis | Calipe, calipo, calipto, eucalipto | E | |
| Eucalyptus viminalis | Calipe, calipo, calipto, eucalipto | O | |
| Eugenia uniflora | Pitangueira | Fd | |
| Psidium cattleyanum | Araçá, goiavinha | Fd | |
| Psidium guajava | Goiabeira | Fd,Fr | ● |
| Syzygium jambos | Jamboeiro, jambre | Fd,O | |
| Nyctaginaceae | |||
| Bougainvillea glabra | Bongavilia, buganvílea | O | |
| Bougainvillea spectabilis | Buganvil, buganvila, buganvílea, mungavi | Ml,O | |
| Mirabilis jalapa | Batata-de-burro, batata-de-porco, gasimi, jesimi, maravilhas | Fr,O | |
| Olacaceae | |||
| Ximenia americana | Ameixieira, ameixeira-brava | Fd | ● |
| Oleaceae | |||
| Jasminum officinale | O | ||
| Jasminum sambac | Jasmineiro | O | |
| Olea europaea subsp. europaea | Oliveira, oliveira-brava, zambujeiro, zambujo | O | |
| Oxalidaceae | |||
| Averrhoa bilimbi | Fd | ||
| Oxalis debilis | O | ||
| Oxalis latifolia | Azedinha | O | |
| Papaveraceae | |||
| Argemone mexicana | Cardo, cardo-santo | Ml,Mt | ● |
| Passifloraceae | |||
| Passiflora edulis | Maracujá-pequeno | Fd | |
| Passiflora quadrangularis | Maracujá-grande | Fd | |
| Petiveriaceae | |||
| Rivina humilis | Uva-de-macaco | Mt | |
| Phyllanthaceae | |||
| Phyllanthus acidus | Azedinha, groselheira, groselha | Fd,O | |
| Phytolaccaceae | |||
| Phytolacca americana | Capa-rosa, uva-macaco | Fd | |
| Phytolacca dioica | Bela-sombra | O,E | |
| Pinaceae | |||
| Pinus canariensis | Fu,E | ||
| Pinus halepensis | Fu,E | ||
| Pinus pinaster | Fu,E | ||
| Pinus radiata | Fu,E | ||
| Plantaginaceae | |||
| Antirrhinum majus | Boca-de-lobo-pequena, boca-dilobo, mataquim | Ml | |
| Cymbalaria muralis | O | ||
| Plantago major | Fedegosa, tanchagem, tantchas | Fr | |
| Plumbaginaceae | |||
| Plumbago zeylanica | Fogo-da-serra, joelho-de-cabra, mato-gonçalves, pega-cabrito | O | |
| Poaceae | |||
| Arundo donax | Caniço, cariço | U | |
| Avena sativa | Palha-de-trigo | Fr | |
| Bambusa vulgaris | Bambu-grande, carisso-da-guiné | U | |
| Coix lacryma-jobi | O | ||
| Cymbopogon citratus | Belgata, capim-limão, chá-de-príncipe, chali, xali | Fd | |
| Eragrostis tenella | Fr | ||
| Paspalum vaginatum | Fr,O | ||
| Saccharum officinarum | Cana-de-açúcar, cana-doce-preta | Fd,Fr | ● |
| Setaria parviflora | Balanco, gôgô, rabo-de-gato, rabo-de-raposa | Fr | |
| Sorghum bicolor | Bimberim, sorgo | Fd,Fr,Ml | ● |
| Sorghum halepense | Achada-carreira, sololo | Fr,U | |
| Stenotaphrum secundatum | Fr,O | ||
| Zea mays | Midjo, milho, milho-de-capa-preta | Fd,Fr,Ml | ● |
| Polygonaceae | |||
| Antigonon leptopus | Fátima, rosa-di-campo, rosa-di-fátima, trepadeira-de-fátima | Ml,O | |
| Coccoloba uvifera | Bela-sombra, mogno | O | |
| Proteaceae | |||
| Grevillea robusta | Carvalho-prateado, grevilia | Ml,T,O,E | |
| Rosaceae | |||
| Cydonia oblonga | Gamboeiro, marmeleiro | Fd | ● |
| Fragaria × ananassa | Morangueiro | Fd | |
| Malus domestica | Macieira | Fd | ● |
| Prunus persica | Pessegueiro | Fd,O | |
| Pyrus communis | Pereira, pereira-mansa | Fd | ● |
| Rhaphiolepis bibas | Nespereira, nespereira-do-japão | Fd | ● |
| Rosa × centifolia | Roseira | O | |
| Rosa moschata | Roseira | O | |
| Rosa sempervirens | Roseira | O | |
| Rubiaceae | |||
| Cinchona pubescens | Quineira | O | |
| Coffea arabica | Cafeeiro, cafezeiro | Fd,S | ● |
| Mitracarpus hirtus | Beitece, beio-teso, beiteso, locotém | Fr,P | |
| Morinda citrifolia | Noni | Fd | |
| Spermacoce verticillata | Bedjo-teso, biteso, lactane, locotane, locotano, velho-teso | Fr | |
| Rutaceae | |||
| Chloroxylon swietenia | Pau-setim | O | |
| Citrus × aurantium | Laranjeira, laranjeira-azeda, laranjeira-doce | Fd,Ml | ● |
| Citrus × limon var. bergamia [a] | Bergamo, bergamota | Fd | |
| Citrus × limon var. limon | Limoeiro | Fd,Ml | ● |
| Citrus maxima | Toranjeira | Fd | |
| Citrus medica | Cidreira, limoeiro | Fd | ● |
| Citrus x aurantiifolia | Limeira, limeira-azeda, limoeiro-pequeno | Fd | ● |
| Ruta chalepensis | Arruda, aruda | Ml,S | |
| Triphasia trifolia | O | ||
| Salicaceae | |||
| Salix x fragilis | O | ||
| Sapindaceae | |||
| Melicoccus bijugatus | O | ||
| Sapindus saponaria | Aveleira, aveloa, avelon, boa-madeira, saboeira, sapodilha | P,T,Mt | |
| Sapotaceae | |||
| Manilkara zapota | Nispere | Fd | |
| Scrophulariaceae | |||
| Myoporum tenuifolium | Pitosporum | O | |
| Simmondsiaceae | |||
| Simmondsia chinensis | Jojoba | Mt | |
| Solanaceae | |||
| Alkekengi officinarum | Fd | ||
| Capsicum annuum | Malagueta, malagueta-arredondada, malaguetona, pimentão | Fd | |
| Capsicum baccatum | Pimento | Fd,O | |
| Capsicum frutescens | Malagueta, malagueta-pontiaguda, malaguetinha, piripiri | Fd,P | ● |
| Datura innoxia | Barbiaca-preta, barbiaca-preta, barbidjaca, berbiaca, berbilhaca, burbilhaca, cardo-preto, padja-fede, palha-fede | Ml,O | |
| Nicotiana glauca | Chaluteiro, charroteira, charuteiro, tabaco-bravo, tabamqueira | O | |
| Nicotiana tabacum | Erba, erva-brava, erva-santa, tabaco | S,O | ● |
| Petunia axillaris | Petunia | O | |
| Physalis peruviana | Capucha, caputcha, uva-caneca, uva-canela, uva-madeira | Fd | ● |
| Solanum betaceum | Tomate-arbóreo | Fd | |
| Solanum lycopersicum | Camacho, tomate, tomateiro, tomatinho | Fd | |
| Solanum melongena | Beringela, beringelo, bringela | Fd | |
| Solanum tuberosum | Batata, batata-inglesa, batateira | Fd | |
| Talinaceae | |||
| Talinum paniculatum | Laranjeirinha, limãozinho | Fd,O | |
| Tamaricaceae | |||
| Tamarix canariensis | Tarafe, tarrafe, tamargueira | E | |
| Tropaeolaceae | |||
| Tropaeolum majus | Chagas | O | |
| Verbenaceae | |||
| Aloysia citrodora | Lúcia-lima | O | |
| Lantana camara | Freira, kambara, lantana-cor-de-rosa, lantuna, lantuna-amarela | Fr,O,E | |
| Verbena officinalis | Agibon-da-terra, gibon, gilbom, verbena | Fd | |
| Verbena tweedieana | O | ||
| Vitaceae | |||
| Vitis vinifera | Uveira, vinha | Fd | ● |
| Zingiberaceae | |||
| Etlingera elatior | Rosa-de-porcelana | O | |
| Hedychium gardnerianum | O | ||
| Zingiber officinale | Gengibre | Fd | |
| Zygophyllaceae | |||
| Balanites aegyptiaca | E | ||
| Tribulus cistoides | Abreodjo, abre-olho, abriolha, abrochona, abroio, abrolho | Fr,Ml |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duarte, M.C.; Gomes, I.; Catarino, S.; Brilhante, M.; Gomes, S.; Rendall, A.; Moreno, Â.; Fortes, A.R.; Ferreira, V.S.; Baptista, I.; et al. Diversity of Useful Plants in Cabo Verde Islands: A Biogeographic and Conservation Perspective. Plants 2022, 11, 1313. https://doi.org/10.3390/plants11101313
Duarte MC, Gomes I, Catarino S, Brilhante M, Gomes S, Rendall A, Moreno Â, Fortes AR, Ferreira VS, Baptista I, et al. Diversity of Useful Plants in Cabo Verde Islands: A Biogeographic and Conservation Perspective. Plants. 2022; 11(10):1313. https://doi.org/10.3390/plants11101313
Chicago/Turabian StyleDuarte, Maria Cristina, Isildo Gomes, Silvia Catarino, Miguel Brilhante, Samuel Gomes, Aline Rendall, Ângela Moreno, Arlindo Rodrigues Fortes, Vladmir Silves Ferreira, Isaurinda Baptista, and et al. 2022. "Diversity of Useful Plants in Cabo Verde Islands: A Biogeographic and Conservation Perspective" Plants 11, no. 10: 1313. https://doi.org/10.3390/plants11101313
APA StyleDuarte, M. C., Gomes, I., Catarino, S., Brilhante, M., Gomes, S., Rendall, A., Moreno, Â., Fortes, A. R., Ferreira, V. S., Baptista, I., Dinis, H., & Romeiras, M. M. (2022). Diversity of Useful Plants in Cabo Verde Islands: A Biogeographic and Conservation Perspective. Plants, 11(10), 1313. https://doi.org/10.3390/plants11101313










