2.1. Canopy Development
Plant shoot growth pattern and shape is commonly manipulated by growers worldwide by numerous techniques including planting density, trellising, plant hormones, and by the physical removal of plant organs.
Figure 1 depicts the response of large cannabis plants to seven architecture-manipulating treatments compared to the natural growth habit of the non-treated ("Control") plants. The treatments are detailed in
Section 3.2. Plant structure was not altered visually by “Defoliation” and by removal of 2° branches (“2° Branch removal”), and in both "BBLR" (Removal of leaves and branches from the bottom part of the plant) treatments, only the lower part of the canopy differed in form from the non-treated control. Plants of the “Double prune” treatment had similar shape to the “Control” plants, with a shorter stature. Two treatments that visibly altered shoot structure considerably were “1° Branch removal” that is composed of a single elongated straight stem with no branches, and “Single prune”, which induced development of two stem-like branches. These results for the large plants evaluated in the present study correlate with responses we received for two other varieties of medical cannabis under short-plant cultivation [
20], with the exception that the shape of the plants of the pruning treatments was less elongated, likely representing genetic differences in the response to alterations of apical dominance, or in growing conditions that promote elongation.
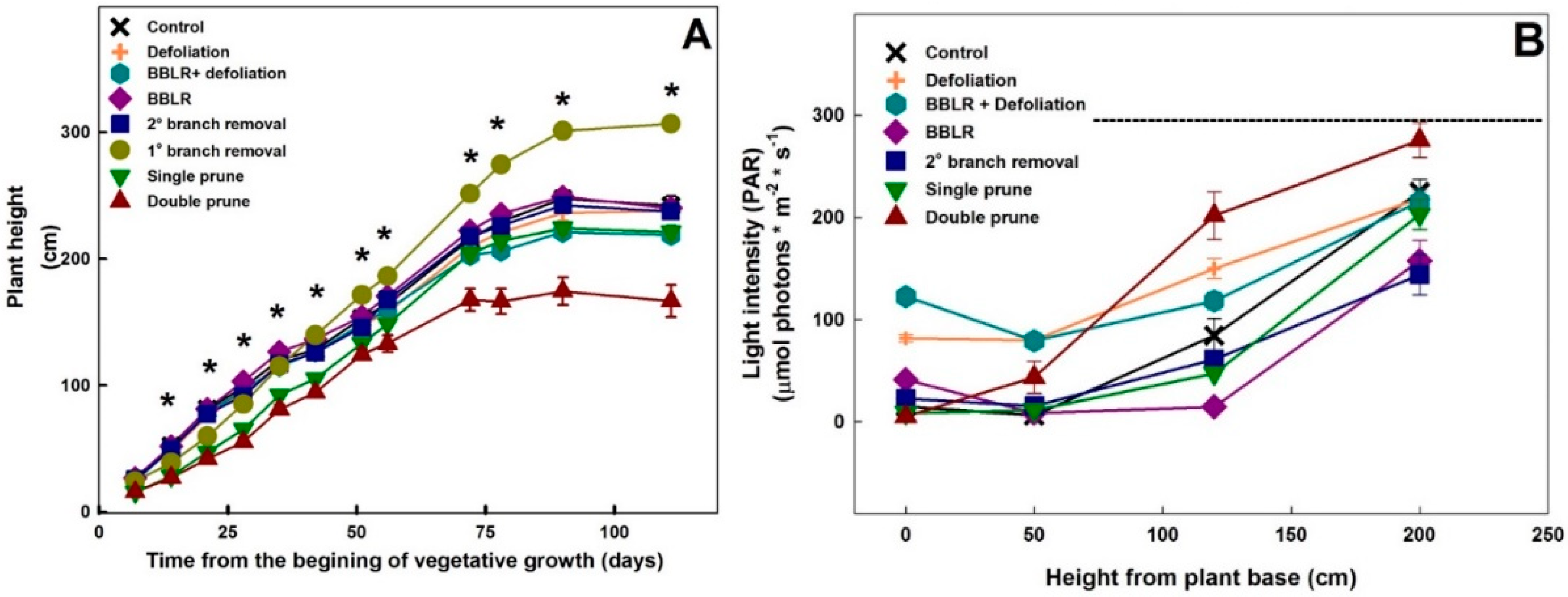
Plant height varied between treatments (
Figure 2A). As early as 7 days after the initiation of the treatments, a statistically significant reduction in height was measured in the pruned plants. Approximately 67 days after the initiation of the treatments, which was 7 days after the transfer to the short-day regime and the second pruning event, the stretched in height of the “Single prune” plants, compensating for the reduction in height imposed by the pruning and the plants reached the height of the “Control” plants.
Architectural manipulations that involve wounding and removal of plant organs alter endogenous developmental programs by affecting apical dominance or sink/source relations and hence also the hormonal profile [
24]. Three developmental changes identified in the plants point to involvement of hormonal activity. First, the two semi-main branches developed under the “Single prune” treatment could be attributed to increased amounts of gibberellic acid, which stimulates both plant elongation and inhibition of lateral bud development [
24]. Such an increase in active gibberellin production was previously seen in different perennials [
25] and could also explain why no reduction in plant height was observed in this treatment. Second, the second pruning event was implemented at the transition to the short day regime, at which time the compact compound inflorescences of cannabis start to develop by restriction of branch elongation and development of short branchlets [
26]. It is possible that this developmental shift involves gibberellin catabolism, which could also explain the shorter plants of the “Double prune” treatment, and the increased branching, resulting in the development of “bushier” plants in this treatment. A lack of gibberellin-induced bud dormancy at the switch to short photoperiod might also explain the stimulation of axillary bud growth and development throughout the plant. Third, the “1° Branch removal” treatment caused the main stem to elongate more than in all other treatments. When this treatment was imposed on smaller medical cannabis plants, the same phenotypic response was found in one of the two genotypes studied [
20]. Cytokinin is a phytohormone that induces cell division and is highly related to plant branching [
27]. The root-derived molecules are transported acropetally in the shoot causing axillary bud development and branching. The removal of branches from the plant removed sink locations, possibly resulting in higher deposition of cytokinin to the apical meristem, inducing enhanced meristem activity and plant elongation.
In cannabis folklore, a “stretching period” of rapid growth is considered to occur at the two weeks following the transition to a short-day photoperiod, prior to the termination of plant elongation and the formation of inflorescences. In the present study, no such “stretching” was seen, but rather a continuation of the pre-short-day growth rate. It is possible that this tale originates from “indoor” growers that conventionally change the cultivation light source from metal-halide (MH) to high pressure sodium (HPS) at the onset of the short photoperiod. These light sources differ in spectrum and intensity. The HPS light spectra is poor in the blue light fraction and rich in the far-red fraction, and both lack of blue light and enrichment in far-red are known elicitors of plant elongation [
9]. These results debunk the concept of an endogenous “stretching period” and point at spectral properties as inducers of the growth enhancement.
As the light travels through the plant canopy it is absorbed by both leaves and branches, changing both in intensity and spectrum [
23]. To evaluate light abundance within the canopy, light intensity was measured at four different heights, and the results are presented in
Figure 2B. Light intensity gradually reduced with the decrease in height towards the bottom of the plants (
Figure 2B), and the extent of reduction and the intensity level along the vertical profile differed between treatments. The highest intensities at the plant base were obtained for both defoliation treatments, demonstrating the potential of defoliation for reducing micro-climate gradients.
Surprisingly, both “BBLR” treatments had higher light intensity at the bottom of the plant than at the height of 50 cm aboveground (
Figure 2B). This is likely caused by horizontal introduction of light to this part of the plants, which is sparse in vegetation due to the treatment, by reflectance from side plants, or by light penetrating from the space between plant rows.
Light intensity at the location 50 cm aboveground was similar for both “Defoliation” treatments (
Figure 2B). This indicates that light absorbed by branches and inflorescences was similar in both treatments, and that in spite of the removal of bottom branches, shoot density was similar in both treatments, as will also be demonstrated by the effects of the treatments on the plant biomass. The difference in light intensity at the plant base between the “BBLR + defoliation” and the “Defoliation” treatments was similar to the difference between the “BBLR” and “Control” treatments and was small in both cases, ranging from 25 to 41 μmol m
−2 s
−1. This quantifies the amount of light reflected from the sides at the bottom of the plot to be between 25 and 41 μmol m
−2 s
−1.
Light penetration to the shoot affects plant development in numerous ways. First, increased light intensity at the lower area of the shoot goes hand-in-hand with a warmer and drier micro-climate [
14]. Both increased light exposure and reduced relative humidity promote transpiration and photosynthesis rate [
28], supporting accelerated growth. This was demonstrated in numerous cropping systems. In eggplant (
Solanum melongena), for example, increased photosynthesis was recorded in plants pruned to increase light penetration to the canopy, resulting in increased content of carbon assimilates [
29]. Such improved plant function is desired in intensive agriculture, and plant canopy manipulation is therefore often used to increase light penetration. In
Camellia oleifera Abel., an open-center shape increased light penetration and temperature at the bottom of the canopy and reduced relative humidity. This altered microclimate increased seed yield and oil contents at the lower part of the canopy compared to a round, closed canopy shape [
14]. In apple trees, light penetration positively correlated with flower bud density, fruit yield, fruit skin color, soluble solids, and fruit firmness [
30]. With the potential for improved chemical composition due to increased light penetration, the cannabinoid profile of the plants was examined next.
2.2. Chemical Response
The difference between cultivation of large vs. small plants entails differences in plant physiology and larger variations in micro-climate conditions along the plant [
31,
32]. A longer distance between the shoot apex and the plant base entails larger hormonal and micro-environment gradients and lower values at the plant base [
31]. In addition, in taller plants, more plant organs (at the center and bottom of the plants) suffer from shading, as more leaves and branches above absorb light [
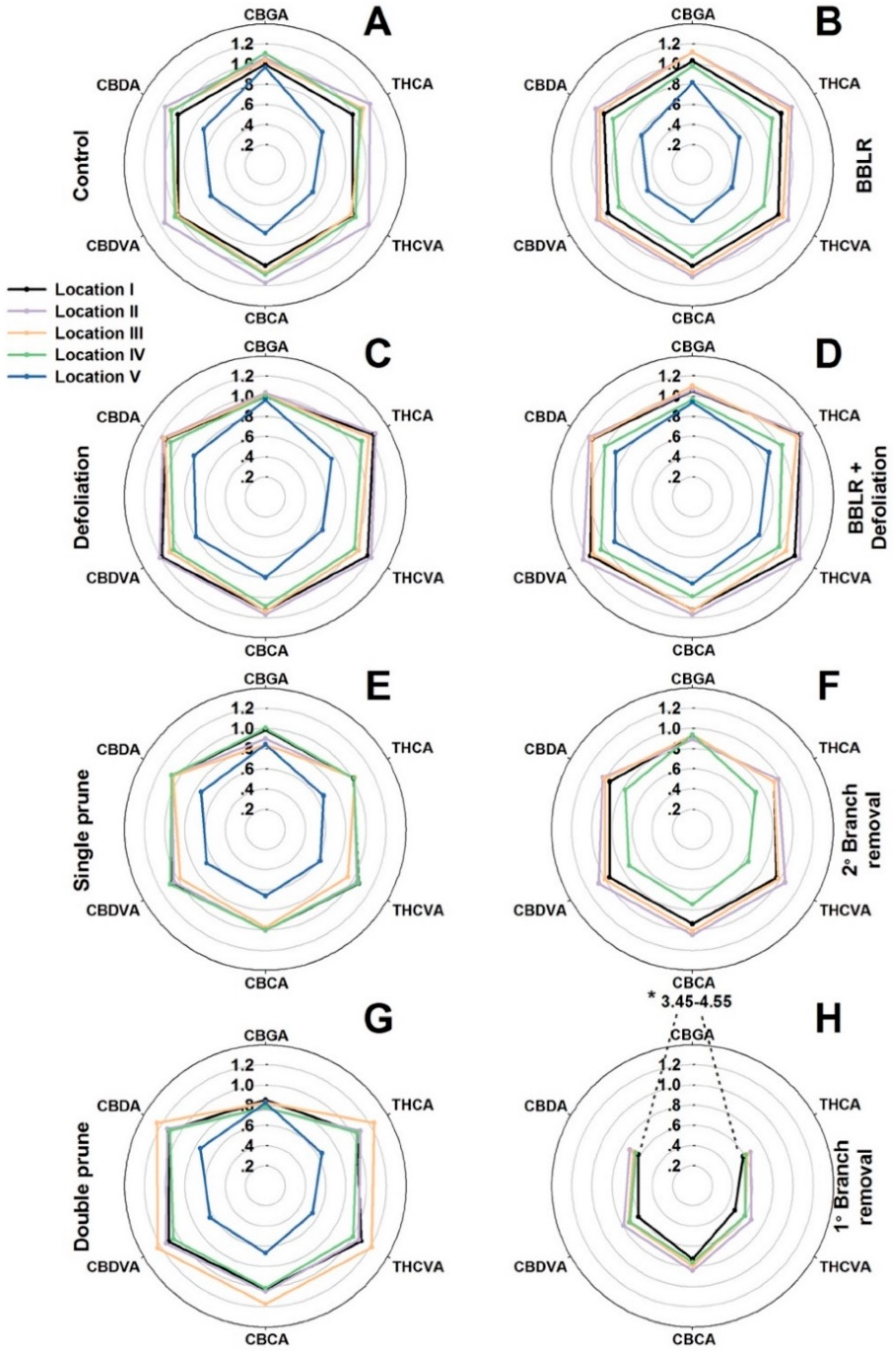
33]. As cannabis inflorescence development is affected by both endogenous and exogenous factors, it is likely that bigger plants will suffer from increased spatial variability compared with smaller plants. To compare variations along the plant and between treatments, the average concentration of each cannabinoid in each sampled location was compared to the concentration in the primary, apical inflorescence (location I) of the “Control” treatment (
Figure 3). In
Figure 3, the further a data point is from the center, the higher is the concentration of the cannabinoid in the specific location compared to the concentration at location I of the control plants. The absolute values represented by this figure can be found in
Supplemental Figures S1–S5.
In the major cannabinoid biosynthetic pathway, cannabigerolic acid (CBGA) is the first cannabinoid formed, which serves as a precursor for an enzymatic catalyzed biosynthesis of the primary cannabinoids ∆
9-tetrahydrocannabinolic acid (THCA), cannabidiolic acid (CBDA), and cannabichromenic acid (CBCA(. Similarly, in a parallel minor pathway, ∆
9-tetrahydrocannabivarinic acid (THCVA), cannabidivarinic acid (CBDVA), and cannabichromevarinic acid (CBCVA) are formed from cannabigerovarinic acid (CBGVA). The concentration of CBGA was considerably affected by the treatments and demonstrated a treatment-dependent response. It was highest in the “1° Branch removal” treatment (
Figure 3H) and reached a level 3.7–4.6-times higher than in the “Control” plants (
Figure 3A). However, in all other treatments and locations, except for location II and III in both “BBLR” treatments (
Figure 3B,D), CBGA concentration was lower than the “Control” concentration. Since CBGA is the precursor of all other cannabinoids, its concentration in the plant is dynamic and reflects the net activities of its biosynthesis and further transformation down the cannabinoid pathways. A question arises as to whether the high level of CBGA in the “1° Branch removal” plants is due to intensified biosynthesis, or rather a reduction in its enzymatic transformation to other cannabinoids. As the concentrations of all other cannabinoids in the “1° Branch removal” treatment were reduced considerably (by 25–40%) compared to the “Control”, it implies that the CBGA “enrichment” in this treatment is a result of inhibition of metabolic activity down the cannabinoid pathway.
Contrary to the common belief in the cannabis industry that consider the cannabinoid concentrations of the primary inflorescence (the “cola”) to be highest in the plant, we report here that for most treatments (e.g., “Control”, “2° Branch removal”, “Double prune”, and both “BBLR” treatments), the concentrations of most cannabinoids were higher in locations II and III than in the apical meristem of location I. Furthermore, in both the “Control” and “Single prune” plants, location IV also had higher concentrations than location I. This result is supported by results of Danziger and Bernstein [
20] that reported as well that cannabinoid concentrations in the primary inflorescence were not always the highest in the plant. As cannabinoid concentrations in an inflorescence are affected by exogenous (environmental) factors, as well as endogenous developmental/location effects, the difference between treatments in this phenomenon should be evaluated taking into consideration both these aspects. From a microclimate standpoint, locations I, II, and IV are at the top of the plant and are highly exposed to sunlight with little to no shading by other plants or branches. Location III on the other hand is also highly exposed but could be shaded by adjacent plants within the row or by plants from parallel rows at certain hours of the photoperiod. Similar to light intensity, air circulation as well is expected to be prevalent in these locations, maintaining similar temperature and humidity at locations I and II and to a lesser extent at locations III and IV. Hence, environmental variations alone cannot explain the identified variations in concentrations.
As micro-climate was partially contradictive as the cause of the observed variation in concentrations, developmental/location effects should be considered. Within the plant, changes in the timing of chemical maturation between inflorescences of different developmental orders may have been reflected as a variability in the chemical profile. A possible explanation is that the primary apical meristem of location I, as the dominant growing zone of the plant, is slower to transition to the reproductive phase, or slower to cease inflorescence growth, thus delaying the maturation of the cannabinoid chemical profile, resulting in lower abundance of secondary metabolites at the time of harvest.
In all treatments, location V (IV in the “2° Branch removal” treatment) was significantly lower down the plant than the remaining locations. Even though at all other locations some variation in light intensity caused little to no effect on secondary metabolism, the cannabinoid concentrations at location V were reduced considerably in all treatments by up to 40% (
Figure 3). As at this location light levels were very low, light appears to be a limiting factor for secondary metabolism. The treatments that suffered least from light scarcity at the location of the bottom-most inflorescence were both “Defoliation” treatments, which also had the smallest decline in cannabinoids in this location. The ability of these treatments to mitigate cannabinoids loss, even though the defoliating was imposed only three weeks before harvest, indicates the high importance of light throughout the process up to chemical maturation.
There was a spatial difference between the ratios of THCA:THCVA and CBDA:CBDVA. For example, in the “BBLR + Defoliation” treatment, the relative increase in CBDVA between locations II and IV was higher than the relative increase in CBDA, but the increase in THCA was higher than THCVA. The biosynthetic pathway of these cannabinoids is intertwined, as both THCA and CBDA originate from the CBGA precursor and both THCVA and CBDVA originate from CBGVA. However, both THCVA and THCA are synthesized by the tetrahydrocannabinolic acid synthase (THCA synthase) enzyme, while CBDA and CBDVA are synthesized by the cannabidiolic acid synthase (CBDA synthase) enzyme [
34]. The difference in ratios between the two pairs (THCA:THCVA and CBDVA:CBDA) suggests differences in substrate selectivity between the enzymes, as well as some environmental effects on their activity or biosynthesis, which should be further explored. Environmental conditions such as light are known to effect some enzymes’ activities [
35] and could explain the previously-described reduction of metabolites in location V.
Secondary metabolism is influenced by environmental conditions such as light [
36], temperature, humidity, mineral nutrition, and water availability [
37], as well as intrinsic factors such as tissue age and location [
38]. Several studies examined the influence of light quality on chemical quality in cannabis and identified variations in the cannabinoid profile [
8,
9,
39], which may explain in part the considerable reduction in cannabinoid content at the bottom of the plant. According to Ma et al. [
40], flavonoid concentrations decreased with the reduction in light intensity in
Anoectochilus formosanus, and similarly, a decrease in phenolic compounds with lower daily light integrals was detected in sweet basil (
Ocimum basilicum) [
41]. The reduced cannabinoid levels can be partially explained by light quality alteration and lowered light intensity at the bottom of the plants.
The results in
Figure 3 reveal variability in concentrations of cannabinoids between locations in the plant in all treatments. To evaluate if the investigated treatments affected the extent of variability and have the potential for standardization of the chemical potential, we analyzed the effect of the treatments on uniformity of the chemical profile within the plant. A new score, “Plant Uniformity score” (PUS) that was recently developed by us [
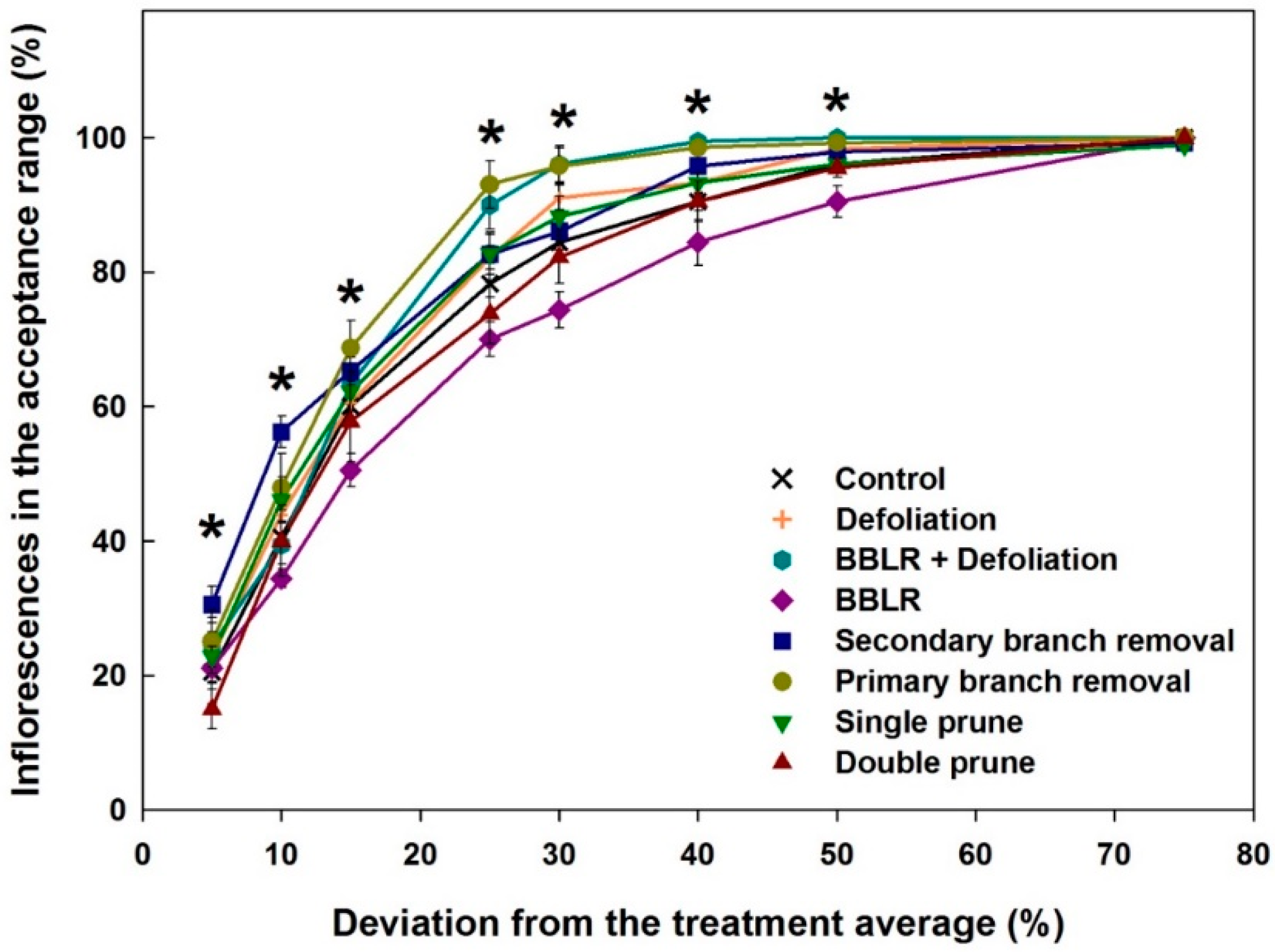
20] was applied for the analysis. The “Plant Uniformity score” (PUS) integrates the variabilities of individual cannabinoids to generate an integrated plant uniformity value. This score allows to calculate the percentage of inflorescences in a treatment which their chemical profile deviates from the treatment average by a pre-defined percentage, for example, a deviation by more than ±5%, 15%, or 30% from the treatment average, etc.
Figure 4 presents the plant uniformity score starting at a strict 5% deviation restriction, and up to an inclusive 75% deviation limit. High uniformity score is achieved by chemically similar inflorescences regardless of the treatment average concentration of each cannabinoid. The analysis revealed a few treatment-related effects: (i) All treatments except “Double prune” and “BBLR” improved chemical uniformity compared to the “Control”; (ii) BBLR was the least uniform treatment in the 10–50% range; (iii) in the strict 5–10% deviation range, "2° Branch removal” was the most uniform. but for the more accepting terms, “1° Branch removal” was more uniform; (iv) “BBLR + Defoliation” was the most uniform treatment (when excluding the branch removal treatments); (v) at all treatments, a “perfect score” (>95 uniformity) was achieved in the 50% deviation rate except for “BBLR”, which reached this goal only at the 75% level of deviation restriction. These results are similar to results reported by Danziger and Bernstein [
20] for smaller cannabis plants. In both studies, a perfect score was reached at the 50% restriction level by all treatments except for “1° Branch removal” and “2° Branch removal”, and a general increase in uniformity was achieved by most treatments compared to the “Control". This identified similarity between plants of different genetic background and size suggests that a maximum of 50% chemical variation from the average concentrations is a universal characteristic expected for cannabis plants regardless of their size. On the other hand, under a more restrictive levels of variance, the smaller plants grown by Danziger and Bernstein [
20] had a plant uniformity score higher by 5–15 points, demonstrating that a higher uniformity was achieved by cultivating smaller plants, though this should be tested under similar growing conditions and varieties.
The chronic lack of light at the base of the plant that we found to be inherent in the cannabis plants, induces chemical variation, and most likely also reduces floral yield. This restriction of light availability down the canopy could be mitigated by local supplementation of light by sub-canopy or intra-canopy illumination. Under such illumination, the lighting systems give light to the middle and bottom canopy, which can lead to higher whole plant photosynthesis and better crop productivity. Many studies have shown that supplemental lighting within the canopy can enhance yield of crop plants [
42,
43,
44]. A previous study assessed the possibility to increase cannabis yield by sub-canopy LED lights, and the results showed a 5% increase in THCA + THC concentrations, but not other cannabinoids, as well as a 13–17% increase in inflorescence yield [
22]. Effects on chemical standardization were not evaluated. The supplemented light within the canopy could be used not only to increase light intensity, but also to compensate for the change in spectral quality as it passes through leaf tissue [
23].
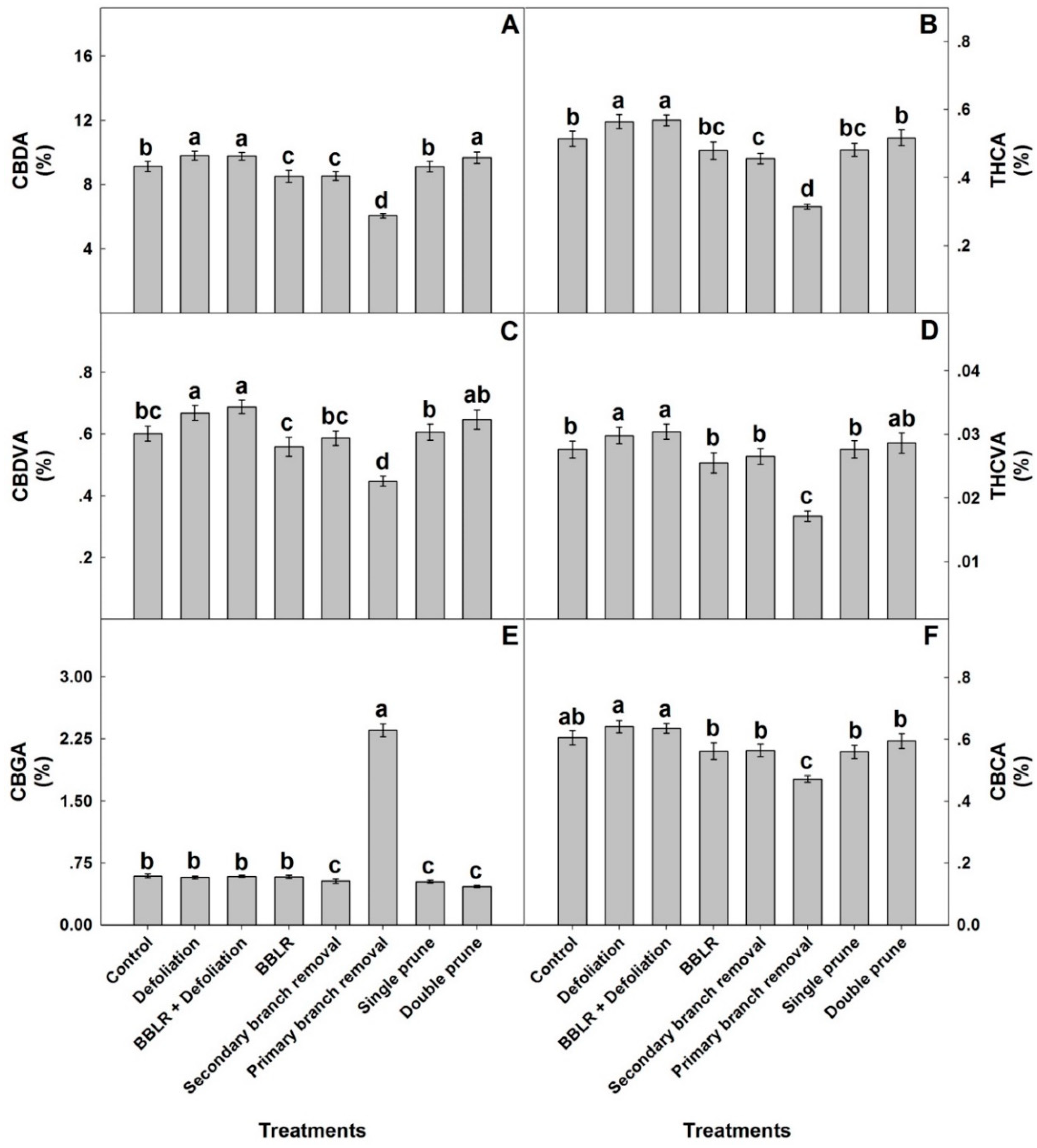
To compare responses of individual cannabinoids to the architecture manipulation treatments, the plant average concentrations of each cannabinoid are compared in
Figure 5. The plant average concentrations of most identified cannabinoids (CBDA, THCA, CBDVA, THCVA, and CBCA) had a similar yet not identical response to the plant architecture treatments, while the response of CBGA differed considerably. A plant-average concentration was affected by all in-planta variations, and an important finding is that it was similar to the concentration at the primary inflorescence at location I the “cola”), which is the industry standard, and therefore demonstrated less variability between treatments than was found for some of the individual locations. The plant average concentrations are the relevant concentrations for industrial production of extraction-based products, whereas concentrations at individual locations and their in-planta variability are crucial for direct consumption of dry inflorescences by smoking and vaporizing. Results for all individual locations can be found in
Supplemental Figures S1–S5.
CBGA levels of the plant average concentrations were highest in the “1° Branch removal” plants, reaching 2.38% of the inflorescence dry weight, while in all other treatments the concentration range was 0.45–0.6% (
Figure 5). Both pruning treatments and “2° Branch removal” had statistically lower CBGA concentrations than the “Control”, “BBLR”, and both defoliation treatments. For all other identified cannabinoids (except CBCA), both defoliation treatments had significantly higher concentrations than the “Control”, while “1° Branch removal” was always lower. In the cannabis plant, CBGA is synthesized from olivetolic acid and geranyl diphosphate. It is the precursor for most cannabinoids and is converted in the plant by CBCA synthase, CBDA synthase, and THCA synthase to the three primary cannabinoids, namely CBCA, CBDA, and THCA, respectively. The primary cannabinoids are then converted downstream to numerous cannabinoid families [
1]. The considerably higher concentrations of CBGA in the “1° Branch removal” treatment may therefore result from reduced biosynthesis of CBGA or from inhibited metabolic conversion of CBGA to the three primary cannabinoids. The results, which demonstrated lower concentrations of the primary cannabinoids in the “1° Branch removal” treatment compared to all other treatments, point at inhibition of the conversion of GBGA to the three primary cannabinoids in this treatment, likely due to restricted energy availability in these suppressed plants.
In general, the plant average concentrations of all cannabinoids except CBGA followed the trend of “Defoliation” = “BBLR + Defoliation” > “Double prune” > “Control” = “Single prune” > “BBLR” = “2° Branch removal” > “1° Branch removal”.
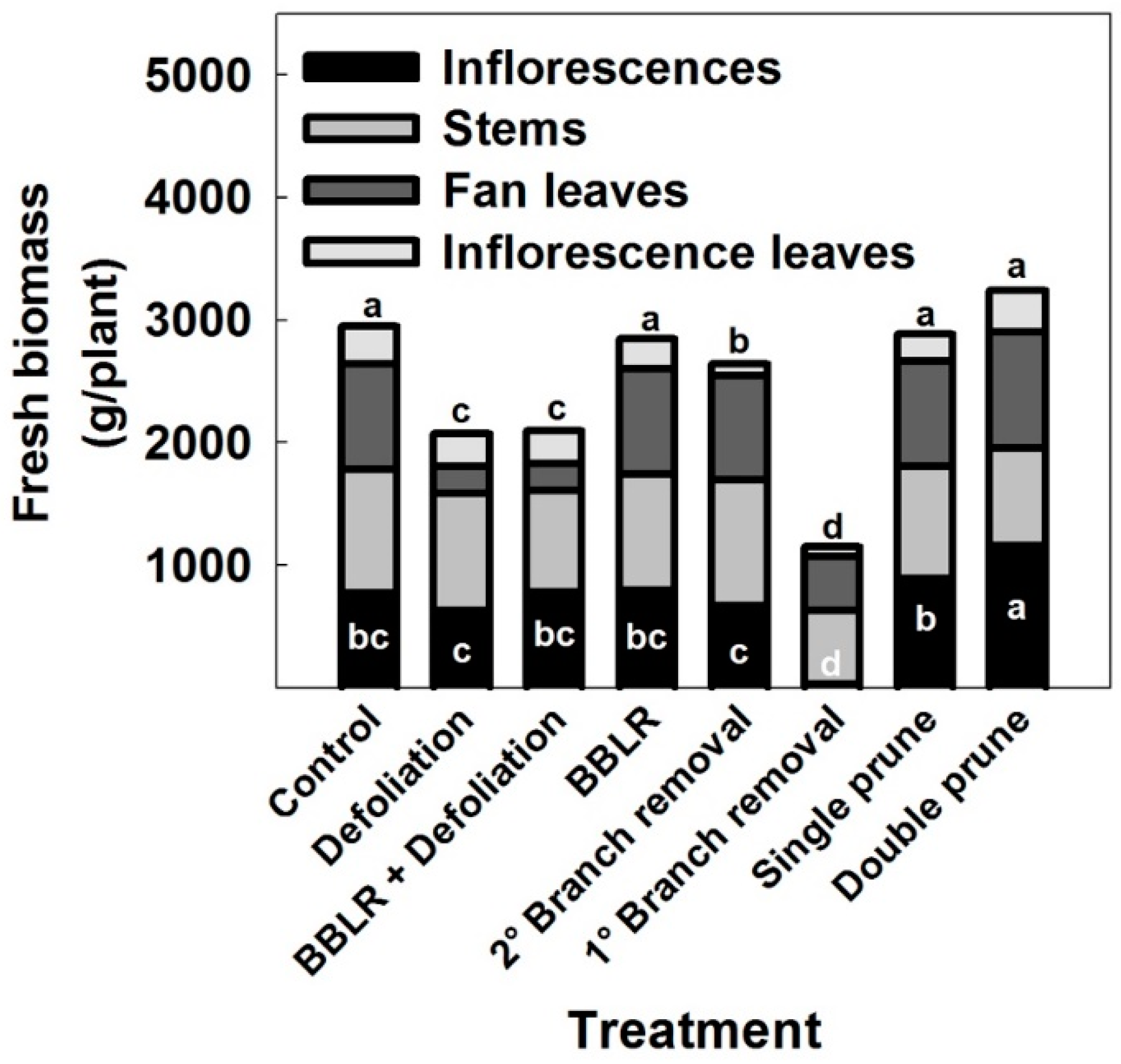
2.3. Inflorescence and Cannabinoid Yield
In many crops, shoot architectural manipulations induce not only morphological changes but also affect yield. In cannabis, yield could be described as either inflorescence biomass (per plant or cultivation area), or as cannabinoid production (per plant or cultivation area). In some industrial cannabis production schemes, vegetative plant biomass is also referred to as yield, as it can be utilized for example in the textile or construction industries [
45]. Fresh biomass accumulation by different plant organs in the medical cannabis plants is presented in
Figure 6. Inflorescence yield production per plant was affected by the treatments. Plants of the “Double prune” treatment produced higher yield (
p < 0.05) than the “Control”, and the “1° Branch removal” plants had lower yield than the control (
p < 0.05). All other treatments did not affect significantly yield biomes (
Figure 6), demonstrating developmental plasticity of the reproductive growth in response to the invasive changes in shoot architecture.
The “1° Branch removal” treatment reduced inflorescence yield considerably, by up to 99%. This dramatic reduction in yield is a result of the absence of active meristems in the plants due to the removal of branches and branchlets by the severe pruning, which prevented inflorescence development along the stem, as well as by a reduction in photosynthetic leaf area per plant, which reduced energy production. This result is supported by a similar decline in inflorescence yield that was reported for smaller cannabis plant cultivation [
20].
As both inflorescence yield and chemical profile were altered by the architecture manipulation treatments, it should not be surprising that the cannabinoid yields per plant were also affected. In
Table 1 the results of cannabinoid yield (g/plant) are presented. In spite of the four-times higher concentration of CBGA in the “1° Branch removal” plants compared with all other treatments (
Figure 5), CBGA production per plant in this treatment was significantly lower (
p < 0.05;
Table 1). Dramatically lower production of cannabinoids per plant in this treatment was apparent also for all other identified cannabinoids, which accumulated to only ~3% of the cannabinoid yield produced by other treatments in terms of g/plant.
For all cannabinoids, higher yields (g/plant) were produced by the “Double prune” plants compared to all other treatments (for CBGA the difference was not statistically different;
Table 1). The “Defoliation” and both “BBLR” treatments, as well as the “2° Branch removal” and “Single prune” treatments did not affect significantly (
p > 0.05) cannabinoid yields (as compared with the non-treated control;
Table 1). The highest cannabinoid yield was achieved by the increased inflorescence yield in the “Double prune” treatment, more so than by the increase in cannabinoid concentrations in the defoliation treatments. This demonstrates that in spite of the expected diluting effect due to growth, the additional inflorescence tissue benefited whole plant cannabinoid production more so than the induced increases in tissue concentrations. This conclusion can be utilized for optimization of production schemes for specific production goals.
The increase in inflorescence biomass in the “Double prune” treatment compared to the control contributed more to the cannabinoid yield than the increase in cannabinoid concentrations at the “bottom” inflorescences by light penetration in the defoliation treatments. In different medicinal plants, the essential oil is the desired component, and efforts to increase its concentration as well as total yield include optimization of light intensity as well as moderate water stress. For rosemary (
Rosmarinus officinalis L.), higher light intensity with no water stress resulted in increased essential oil yield in spite of the reduction in tissue concentration. On the other hand, decreased light intensity coupled with moderate water stress resulted in a yield similar to the high light intensity treatment due to significantly higher essential oil concentration [
46].
Eucalyptus citriodora, on the other hand, produced less essential oil under full sun, probably due to light-induced stress [
47], while Chaste tree (
Vitex agnus-castus L.) and sweet basil (
Ocimum basilicum) responded to decreased light intensity and decreased water availability with decreased essential oil production [
48,
49]. In most reported studies, the change in biomass was more influential on total secondary metabolites yield than changes in metabolites concentrations, most likely because the range of changes of secondary metabolite concentrations is smaller than effects on biomass accumulation. It is thus suggested that maximizing inflorescence yield weight should be the first line strategy in cannabis, to be fine-tuned by optimization of cannabinoid concentrations. It should however be considered that the longitudinal light gradient down the plant has a considerable effect on cannabinoid uniformity, with fluctuations of up to 50% between locations.