β-Ionone: Its Occurrence and Biological Function and Metabolic Engineering
Abstract
1. Introduction
2. Occurrence of β-Ionone
3. Biosynthesis, Extraction, or Formation of β-Ionone during Processing and Storage of Foods
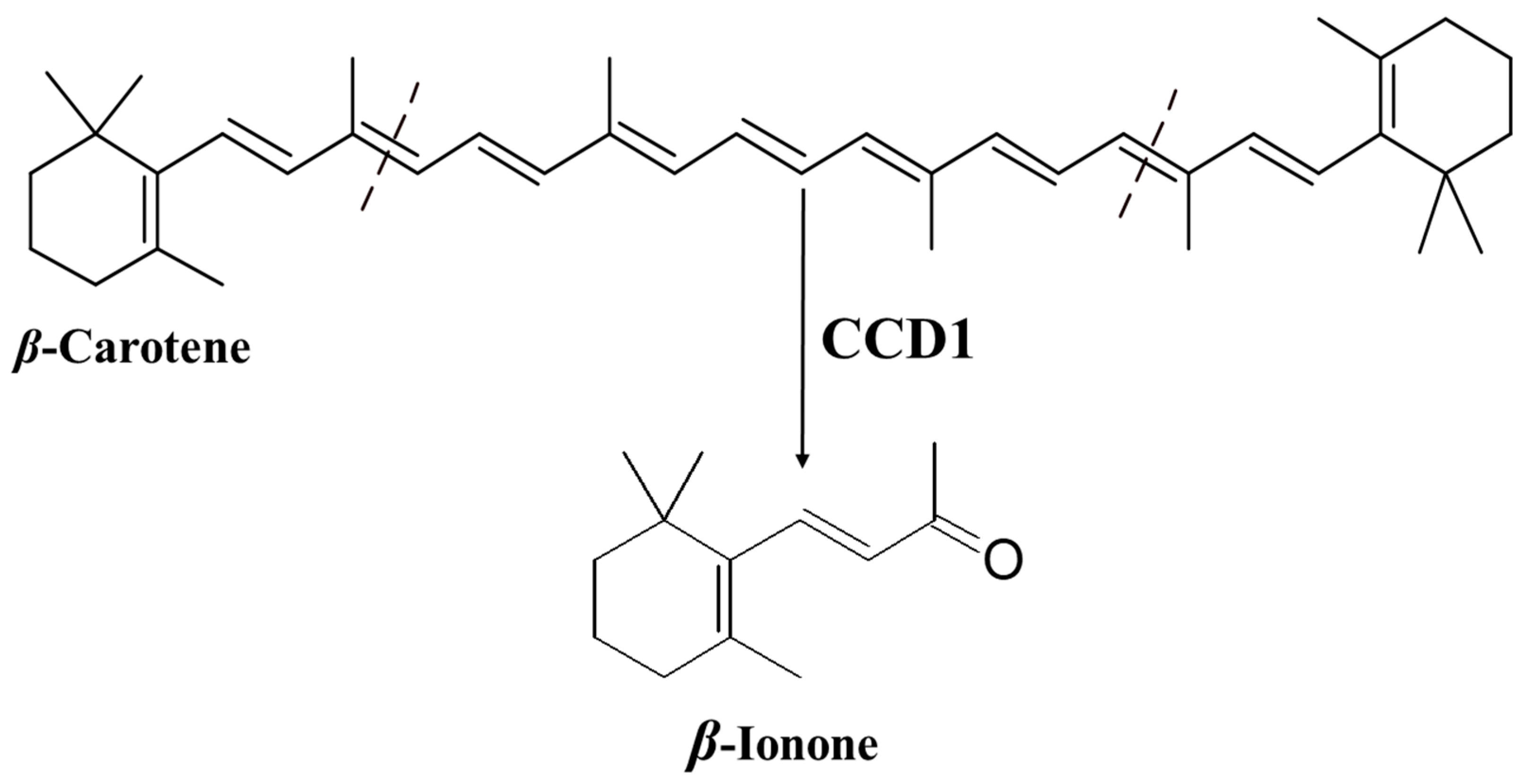
3.1. Biosynthesis of β-Ionone
3.2. Extraction or Formation of β-Ionone during Processing and Storage of Foods
4. Biological Activities of β-Ionone
4.1. Role of β-Ionone in Plants
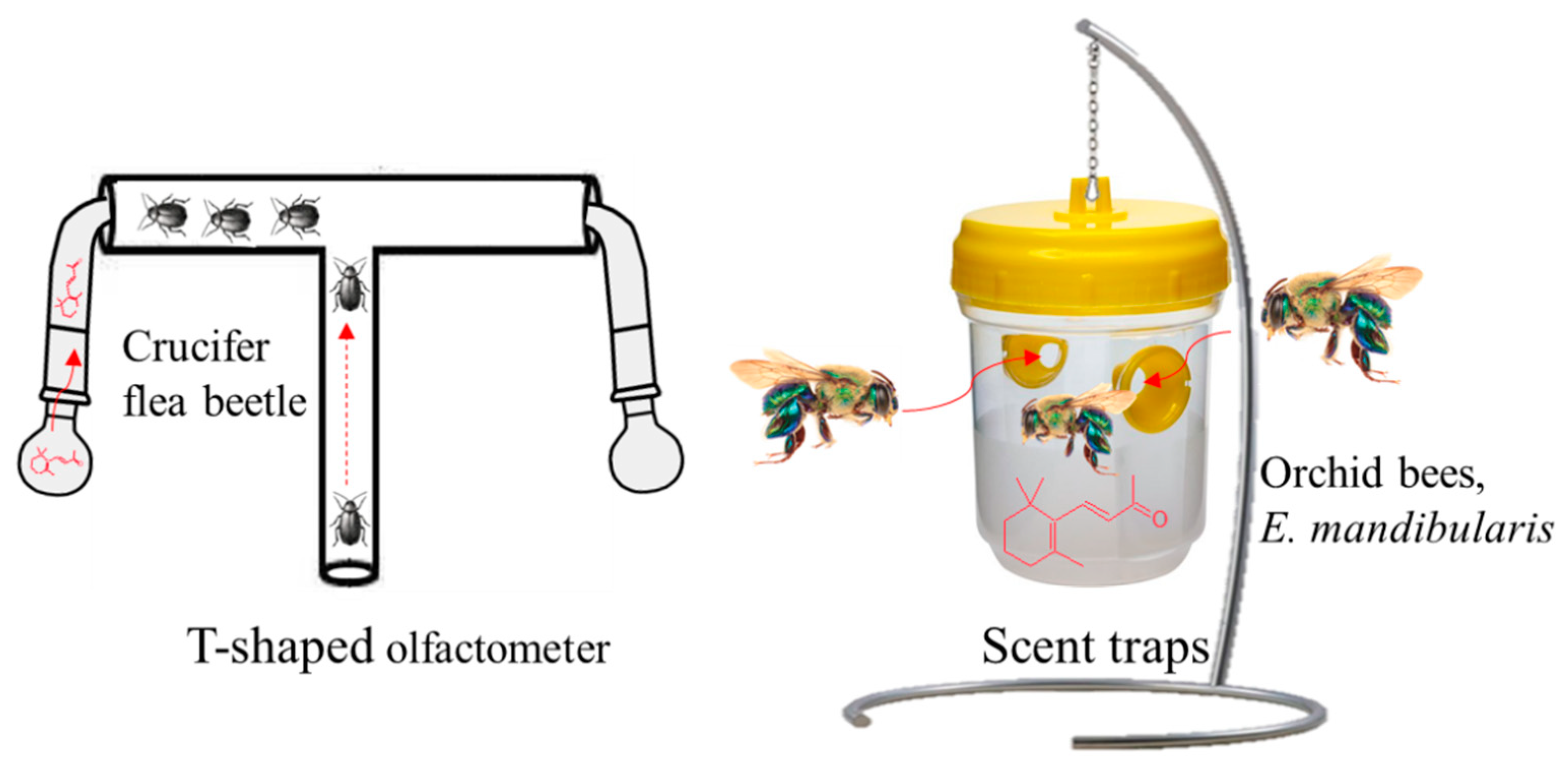
4.2. β-Ionone as an Insect Attractant/Repellant Volatile Compound
4.3. Potential of β-Ionone in Human Health
5. Microbial Production and Biotransformation of β-Ionone
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dudareva, N.; Negre, F.; Nagegowda, D.A.; Orlova, I. Plant volatiles: Recent advances and future perspectives. Crit. Rev. Plant Sci. 2006, 25, 417–440. [Google Scholar] [CrossRef]
- Knudsen, J.; Gershenzon, J. The chemical diversity of floral scent. In Biology of Floral Scent; Pichersky, N.D.A.E., Ed.; Taylor & Francis: Boca Raton, FL, USA, 2006. [Google Scholar]
- Dudareva, N.; Pichersky, E.; Gershenzon, J. Biochemistry of plant volatiles. Plant Physiol. 2004, 135, 1893–1902. [Google Scholar] [CrossRef]
- Böttger, A.; Vothknecht, U.; Bolle, C.; Wolf, A. Plant secondary metabolites and their general function in plants. In Lessons on Caffeine, Cannabis & Co: Plant-Derived Drugs and Their Interaction with Human Receptors; Springer: Cham, Switzerland, 2018; pp. 3–17. [Google Scholar]
- Kessler, A.; Kalske, A. Plant secondary metabolite diversity and species interactions. Annu. Rev. Ecol. Evol. Syst. 2018, 49, 115–138. [Google Scholar] [CrossRef]
- Reinhard, J.; Srinivasan, M.V.; Zhang, S. Scent-triggered navigation in honeybees. Nature 2004, 427, 411. [Google Scholar] [CrossRef]
- Pichersky, E.; Gershenzon, J. The formation and function of plant volatiles: Perfumes for pollinator attraction and defense. Curr. Opin. Plant Biol. 2002, 5, 237–243. [Google Scholar] [CrossRef]
- Rosenkranz, M.A.S.; Schnitzler, J. Plant volatiles. eLS 2016, 1–9. [Google Scholar] [CrossRef]
- Ayseli, M.T.; Ayseli, Y.İ. Flavors of the future: Health benefits of flavor precursors and volatile compounds in plant foods. Trends Food Sci. Technol. 2016, 48, 69–77. [Google Scholar] [CrossRef]
- Goff, S.A.; Klee, H.J. Plant volatile compounds: Sensory cues for health and nutritional value? Science 2006, 311, 815–819. [Google Scholar] [CrossRef] [PubMed]
- Baldermann, S.; Yamamoto, M.; Yang, Z.; Kawahashi, T.; Kuwano, K.; Watanabe, N. C13-Apocarotenoids: More than flavor compounds? In Carotenoid Cleavage Products; ACS Publications: Washington, DC, USA, 2013; pp. 73–80. [Google Scholar] [CrossRef]
- Walter, M.H.; Strack, D. Carotenoids and their cleavage products: Biosynthesis and functions. Nat. Prod. Rep. 2011, 28, 663–692. [Google Scholar] [CrossRef] [PubMed]
- Auldridge, M.E.; McCarty, D.R.; Klee, H.J. Plant carotenoid cleavage oxygenases and their apocarotenoid products. Curr. Opin. Plant Biol. 2006, 9, 315–321. [Google Scholar] [CrossRef]
- Ibdah, M.; Azulay, Y.; Portnoy, V.; Wasserman, B.; Bar, E.; Meir, A.; Burger, Y.; Hirschberg, J.; Schaffer, A.A.; Katzir, N. Functional characterization of CmCCD1, a carotenoid cleavage dioxygenase from melon. Phytochemistry 2006, 67, 1579–1589. [Google Scholar] [CrossRef]
- Yahyaa, M.; Bar, E.; Dubey, N.K.; Meir, A.; Davidovich-Rikanati, R.; Hirschberg, J.; Aly, R.; Tholl, D.; Simon, P.W.; Tadmor, Y. Formation of norisoprenoid flavor compounds in carrot (Daucus carota L.) roots: Characterization of a cyclic-specific carotenoid cleavage dioxygenase 1 gene. J. Agric. Food Chem. 2013, 61, 12244–12252. [Google Scholar] [CrossRef]
- Yahyaa, M.; Berim, A.; Isaacson, T.; Marzouk, S.; Bar, E.; Davidovich-Rikanati, R.; Lewinsohn, E.; Ibdah, M. Isolation and functional characterization of carotenoid cleavage dioxygenase-1 from Laurus nobilis L. (Bay Laurel) fruits. J. Agric. Food Chem. 2015, 63, 8275–8282. [Google Scholar] [CrossRef] [PubMed]
- Simkin, A.J.; Schwartz, S.H.; Auldridge, M.; Taylor, M.G.; Klee, H.J. The tomato carotenoid cleavage dioxygenase 1 genes contribute to the formation of the flavor volatiles β-ionone, pseudoionone, and geranylacetone. Plant J. 2004, 40, 882–892. [Google Scholar] [CrossRef] [PubMed]
- Tandon, K.; Baldwin, E.; Shewfelt, R. Aroma perception of individual volatile compounds in fresh tomatoes (Lycopersicon esculentum, Mill.) as affected by the medium of evaluation. Postharvest Biol. Technol. 2000, 20, 261–268. [Google Scholar] [CrossRef]
- Sewenig, S.; Bullinger, D.; Hener, U.; Mosandl, A. Comprehensive authentication of (E)-α (β)-ionone from raspberries, using constant flow MDGC-C/P-IRMS and enantio-MDGC-MS. J. Agric. Food Chem. 2005, 53, 838–844. [Google Scholar] [CrossRef]
- Pino, J.A.; Quijano, C.E. Study of the volatile compounds from plum (Prunus domestica L. cv. Horvin) and estimation of their contribution to the fruit aroma. Food Sci. Technol. 2012, 32, 76–83. [Google Scholar] [CrossRef]
- Nawade, B.; Shaltiel-Harpaz, L.; Yahyaa, M.; Bosamia, T.C.; Kabaha, A.; Kedoshim, R.; Zohar, M.; Isaacson, T.; Ibdah, M. Analysis of apocarotenoid volatiles during the development of Ficus carica fruits and characterization of carotenoid cleavage dioxygenase genes. Plant Sci. 2020, 290, 110292. [Google Scholar] [CrossRef]
- Belsito, D.; Bickers, D.; Bruze, M.; Calow, P.; Greim, H.; Hanifin, J.M.; Rogers, A.E.; Saurat, J.H.; Sipes, I.G.; Tagami, H. A toxicologic and dermatologic assessment of ionones when used as fragrance ingredients. Food Chem. Toxicol. 2007, 45, S130–S167. [Google Scholar] [CrossRef]
- Ansari, M.; Emami, S. β-Ionone and its analogs as promising anticancer agents. Eur. J. Med. Chem. 2016, 123, 141–154. [Google Scholar] [CrossRef]
- Balbi, A.; Anzaldi, M.; Mazzei, M.; Miele, M.; Bertolotto, M.; Ottonello, L.; Dallegri, F. Synthesis and biological evaluation of novel heterocyclic ionone-like derivatives as anti-inflammatory agents. Bioorg. Med. Chem. 2006, 14, 5152–5160. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.-W.; Wang, K.; Chang, X.-X.; Jin, F.-F.; Wang, Q.; Jiang, X.-F.; Liu, J.-R.; Wu, Y.-H.; Yang, C. Beta-ionone-inhibited proliferation of breast cancer cells by inhibited COX-2 activity. Arch. Toxicol. 2019, 93, 2993–3003. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Singh, G.; Kaur, H.; Saxena, A.K.; Ishar, M.P.S. Synthesis of β-ionone derived chalcones as potent antimicrobial agents. Bioorg. Med. Chem. Lett. 2012, 22, 6343–6346. [Google Scholar] [CrossRef] [PubMed]
- Suryawanshi, S.N.; Bhat, B.A.; Pandey, S.; Chandra, N.; Gupta, S. Chemotherapy of leishmaniasis. Part VII: Synthesis and bioevaluation of substituted terpenyl pyrimidines. Eur. J. Med. Chem. 2007, 42, 1211–1217. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, S.E.A.J.K. The accumulation of 3-ionone and 3-hydroxy esters of 3-ionone in tobacco immunized by foliar inoculation with tobacco mosaic virus. Phytopathology 1992, 82, 580–582. [Google Scholar] [CrossRef]
- Wang, L.-M.; Li, M.-T.; Jin, W.-W.; Li, S.; Zhang, S.-Q.; Yu, L.-J. Variations in the components of Osmanthus fragrans Lour. essential oil at different stages of flowering. Food Chem. 2009, 114, 233–236. [Google Scholar] [CrossRef]
- Goodner, K.; Mahattanatawee, K.; Plotto, A.; Sotomayor, J.; Jordan, M. Aromatic profiles of Thymus hyemalis and Spanish T. vulgaris essential oils by GC–MS/GC–O. Ind. Crops Prod. 2006, 24, 264–268. [Google Scholar] [CrossRef]
- Oyedeji, A.O.; Ekundayo, O.; Koenig, W.A. Essential oil composition of Lawsonia inermis L. leaves from Nigeria. J. Essent. Oil Res. 2005, 17, 403–404. [Google Scholar] [CrossRef]
- Verma, R.S.; Padalia, R.C.; Chauhan, A. Chemical composition of essential oil and rose-water extract of Himalayan Musk Rose (Rosa brunonii Lindl.) from Kumaon region of western Himalaya. J. Essent. Oil Res. 2016, 28, 332–338. [Google Scholar] [CrossRef]
- Huang, F.-C.; Horváth, G.; Molnár, P.; Turcsi, E.; Deli, J.; Schrader, J.; Sandmann, G.; Schmidt, H.; Schwab, W. Substrate promiscuity of RdCCD1, a carotenoid cleavage oxygenase from Rosa damascena. Phytochemistry 2009, 70, 457–464. [Google Scholar] [CrossRef]
- Basu, S.; Zandi, P.; Cetzal-Ix, W.; Sengupta, R. The genus Rosa: An aristocrat from the plant family with class, color and fragrance. IRSEN 2015, 1–9. Available online: https://www.academia.edu/download/38410616/Basu_et_al_Roses_2015.pdf (accessed on 15 March 2021).
- Honarvar, M.; Javidnia, K.; Khosh-Khui, M. Essential oil composition of fresh and dried flowers of Rosa moschata from Iran. Chem. Nat. Compd. 2011, 47, 826–828. [Google Scholar] [CrossRef]
- Anca, T.; Philippe, V.; Ilioara, O.; Mircea, T. Composition of essential oils of Viola tricolor and V. arvensis from Romania. Chem. Nat. Compd. 2009, 45, 91–92. [Google Scholar] [CrossRef]
- Flamini, G.; Luigi Cioni, P.; Morelli, I.; Ceccarini, L.; Andolfi, L.; Macchia, M. Composition of the essential oil of Medicago marina L. from the coastal dunes of Tuscany, Italy. Flavour Fragr. J. 2003, 18, 460–462. [Google Scholar] [CrossRef]
- Viuda-Martos, M.; Sendra, E.; Pérez-Alvarez, J.A.; Fernández-López, J.; Amensour, M.; Abrini, J. Identification of flavonoid content and chemical composition of the essential oils of Moroccan herbs: Myrtle (Myrtus communis L.), rockrose (Cistus ladanifer L.) and Montpellier cistus (Cistus monspeliensis L.). J. Essent. Oil Res. 2011, 23, 1–9. [Google Scholar] [CrossRef]
- Tellez, M.R.; Khan, I.A.; Kobaisy, M.; Schrader, K.K.; Dayan, F.E.; Osbrink, W. Composition of the essential oil of Lepidium meyenii (Walp.). Phytochemistry 2002, 61, 149–155. [Google Scholar] [CrossRef]
- Zatla, A.T.; Dib, M.E.A.; Djabou, N.; Tabti, B.; Meliani, N.; Costa, J.; Muselli, A. Chemical variability of essential oil of Daucus carota subsp. sativus from Algeria. J. Herbs Spices Med. Plants 2017, 23, 216–230. [Google Scholar] [CrossRef]
- Guillot, S.; Peytavi, L.; Bureau, S.; Boulanger, R.; Lepoutre, J.-P.; Crouzet, J.; Schorr-Galindo, S. Aroma characterization of various apricot varieties using headspace–solid phase microextraction combined with gas chromatography–mass spectrometry and gas chromatography–olfactometry. Food Chem. 2006, 96, 147–155. [Google Scholar] [CrossRef]
- Solís-Solís, H.M.; Calderón-Santoyo, M.; Schorr-Galindo, S.; Luna-Solano, G.; Ragazzo-Sánchez, J.A. Characterization of aroma potential of apricot varieties using different extraction techniques. Food Chem. 2007, 105, 829–837. [Google Scholar] [CrossRef]
- Gokbulut, I.; Karabulut, I. SPME–GC–MS detection of volatile compounds in apricot varieties. Food Chem. 2012, 132, 1098–1102. [Google Scholar] [CrossRef]
- Chairote, G.; Rodriguez, F.; Crouzet, J. Characterization of additional volatile flavor components of apricot. J. Food Sci. 1981, 46, 1898–1901. [Google Scholar] [CrossRef]
- Lewinsohn, E.; Sitrit, Y.; Bar, E.; Azulay, Y.; Meir, A.; Zamir, D.; Tadmor, Y. Carotenoid pigmentation affects the volatile composition of tomato and watermelon fruits, as revealed by comparative genetic analyses. J. Agric. Food Chem. 2005, 53, 3142–3148. [Google Scholar] [CrossRef] [PubMed]
- Nunes, C.; Coimbra, M.A.; Saraiva, J.; Rocha, S.M. Study of the volatile components of a candied plum and estimation of their contribution to the aroma. Food Chem. 2008, 111, 897–905. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, N.; Zhao, M.; Jing, T.; Jin, J.; Wu, B.; Wan, X.; Schwab, W.; Song, C. Carotenoid cleavage dioxygenase 4 catalyzes the formation of carotenoid-derived volatile β-ionone during tea (Camellia sinensis) Withering. J. Agric. Food Chem. 2020, 68, 1684–1690. [Google Scholar] [CrossRef] [PubMed]
- Eyres, G.T.; Marriott, P.J.; Dufour, J.-P. Comparison of odor-active compounds in the spicy fraction of hop (Humulus lupulus L.) essential oil from four different varieties. J. Agric. Food Chem. 2007, 55, 6252–6261. [Google Scholar] [CrossRef] [PubMed]
- Cooper, C.M.; Davies, N.W.; Menary, R.C. C-27 apocarotenoids in the flowers of Boronia megastigma (Nees). J. Agric. Food Chem. 2003, 51, 2384–2389. [Google Scholar] [CrossRef] [PubMed]
- Simkin, A.J.; Underwood, B.A.; Auldridge, M.; Loucas, H.M.; Shibuya, K.; Schmelz, E.; Clark, D.G.; Klee, H.J. Circadian regulation of the PhCCD1 carotenoid cleavage dioxygenase controls emission of β-ionone, a fragrance volatile of petunia flowers. Plant Physiol. 2004, 136, 3504–3514. [Google Scholar] [CrossRef]
- Wong, K.; Teng, Y. Volatile components of Lawsonia inermis L. flowers. J. Essent. Oil Res. 1995, 7, 425–428. [Google Scholar] [CrossRef]
- Han, Y.; Wang, H.; Wang, X.; Li, K.; Dong, M.; Li, Y.; Zhu, Q.; Shang, F. Mechanism of floral scent production in Osmanthus fragrans and the production and regulation of its key floral constituents, β-ionone and linalool. Hortic. Res. 2019, 6, 1–12. [Google Scholar] [CrossRef]
- Baldermann, S.; Kato, M.; Fleischmann, P.; Watanabe, N. Biosynthesis of α-and β-ionone, prominent scent compounds, in flowers of Osmanthus fragrans. Acta Biochim. Pol. 2012, 59, 79–81. [Google Scholar] [CrossRef]
- Azuma, H.; Toyota, M.; Asakawa, Y.; Takaso, T.; Tobe, H. Floral scent chemistry of mangrove plants. Int. J. Plant Res. 2002, 115, 47–53. [Google Scholar] [CrossRef]
- Evangelia, P.; Constantinos, V.; Maria, C.; Olga, T. Study of volatile components of Acacia farnesiana Willd. Flowers. Rec. Nat. Prod. 2017, 11, 474–478. [Google Scholar] [CrossRef]
- Maia, J.G.S.; Zoghbi, M.D.G.B.; Andrade, E.H.A.; Carreira, L.M.M. Volatiles from flowers of Thevetia peruviana (Pers.) K. Schum. and Allamanda cathartics Linn. (Apocynaceae). J. Essent. Oil Res. 2000, 12, 322–324. [Google Scholar] [CrossRef]
- Shi, J.; Cao, C.; Xu, J.; Zhou, C. Research advances on biosynthesis, regulation, and biological activities of apocarotenoid aroma in horticultural plants. J. Chem. 2020, 2020. [Google Scholar] [CrossRef]
- Aloum, L.; Alefishat, E.; Adem, A.; Petroianu, G. Ionone Is More than a Violet’s Fragrance: A Review. Molecules 2020, 25, 5822. [Google Scholar] [CrossRef] [PubMed]
- García-Limones, C.; Schnäbele, K.; Blanco-Portales, R.; Luz Bellido, M.; Caballero, J.L.; Schwab, W.; Muñoz-Blanco, J. Functional Characterization of FaCCD1: A Carotenoid Cleavage Dioxygenase from Strawberry Involved in Lutein Degradation during Fruit Ripening. J. Agric. Food Chem. 2008, 56, 9277–9285. [Google Scholar] [CrossRef] [PubMed]
- Ohmiya, A. Carotenoid cleavage dioxygenases and their apocarotenoid products in plants. Plant Biotechnol. 2009, 26, 351–358. [Google Scholar] [CrossRef]
- Huang, F.-C.; Molnár, P.; Schwab, W. Cloning and functional characterization of carotenoid cleavage dioxygenase 4 genes. J. Exp. Bot. 2009, 60, 3011–3022. [Google Scholar] [CrossRef]
- Rubio, A.; Rambla, J.L.; Santaella, M.; Gómez, M.D.; Orzaez, D.; Granell, A.; Gómez-Gómez, L. Cytosolic and plastoglobule-targeted carotenoid dioxygenases from Crocus sativus are both involved in β-ionone release. J. Biol. Chem. 2008, 283, 24816–24825. [Google Scholar] [CrossRef]
- Pénicaud, C.; Achir, N.; Dhuique-Mayer, C.; Dornier, M.; Bohuon, P. Degradation of β-carotene during fruit and vegetable processing or storage: Reaction mechanisms and kinetic aspects: A review. Fruits 2011, 66, 417–440. [Google Scholar] [CrossRef]
- Limbo, S.; Torri, L.; Piergiovanni, L. Light-induced changes in an aqueous β-carotene system stored under halogen and fluorescent lamps, affected by two oxygen partial pressures. J. Agric. Food Chem. 2007, 55, 5238–5245. [Google Scholar] [CrossRef]
- Nguyen, M.; Francis, D.; Schwartz, S. Thermal isomerisation susceptibility of carotenoids in different tomato varieties. J. Sci. Food Agric. 2001, 81, 910–917. [Google Scholar] [CrossRef]
- Gong, H.; Yang, Z.; Liu, M.; Shi, Z.; Li, J.; Chen, W.; Qiao, X. Time-dependent categorization of volatile aroma compound formation in stewed Chinese spicy beef using electron nose profile coupled with thermal desorption GC–MS detection. Food Sci. Hum. Well 2017, 6, 137–146. [Google Scholar] [CrossRef]
- Malowicki, S.M.; Martin, R.; Qian, M.C. Comparison of sugar, acids, and volatile composition in raspberry bushy dwarf virus-resistant transgenic raspberries and the wild type ‘Meeker’ (Rubus Idaeus L.). J. Agric. Food Chem. 2008, 56, 6648–6655. [Google Scholar] [CrossRef]
- Hansen, A.M.S.; Frandsen, H.L.; Fromberg, A. Authenticity of raspberry flavor in food products using SPME-chiral-GC-MS. Food Sci. Nutr. 2016, 4, 348–354. [Google Scholar] [CrossRef]
- Plotto, A.; Barnes, K.; Goodner, K. Specific anosmia observed for β-ionone, but not for α-ionone: Significance for flavor research. J. Food Sci. 2006, 71, S401–S406. [Google Scholar] [CrossRef]
- Giuliano, G.; Al-Babili, S.; von Lintig, J. Carotenoid oxygenases: Cleave it or leave it. Trends Plant Sci. 2003, 8, 145–149. [Google Scholar] [CrossRef]
- Winterhalter, P.; Rouseff, R.L. Carotenoid-Derived Aroma Compounds; ACS Publications: Washington, DC, USA, 2001. [Google Scholar]
- Bouvier, F.; Isner, J.-C.; Dogbo, O.; Camara, B. Oxidative tailoring of carotenoids: A prospect towards novel functions in plants. Trends Plant Sci. 2005, 10, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Havaux, M. Carotenoid oxidation products as stress signals in plants. Plant J. 2014, 79, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Faria, L.R.; Zanella, F.C. Beta-ionone attracts Euglossa mandibularis (Hymenoptera, Apidae) males in western Paraná forests. Rev. Bras. Entomol. 2015, 59, 260–264. [Google Scholar] [CrossRef]
- Wei, S.; Hannoufa, A.; Soroka, J.; Xu, N.; Li, X.; Zebarjadi, A.; Gruber, M. Enhanced β-ionone emission in Arabidopsis over-expressing AtCCD1 reduces feeding damage in vivo by the crucifer flea beetle. Environ. Entomol. 2011, 40, 1622–1630. [Google Scholar] [CrossRef] [PubMed]
- Cáceres, L.; Lakshminarayan, S.; Yeung, K.-C.; McGarvey, B.; Hannoufa, A.; Sumarah, M.; Benitez, X.; Scott, I. Repellent and attractive effects of α-, β-, and dihydro-β-ionone to generalist and specialist herbivores. J. Chem. Ecol. 2016, 42, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ghisalberti, E.L.; Ridsdill-Smith, J. Volatiles from Trifolium as feeding deterrents of redlegged earth mites. Phytochemistry 1999, 52, 601–605. [Google Scholar] [CrossRef]
- Ômura, H.; Honda, K.; Hayashi, N. Floral scent of Osmanthus fragrans discourages foraging behavior of cabbage butterfly, Pieris rapae. J. Chem. Ecol. 2000, 26, 655–666. [Google Scholar] [CrossRef]
- Gruber, M.; Xu, N.; Grenkow, L.; Li, X.; Onyilagha, J.; Soroka, J.; Westcott, N.; Hegedus, D. Responses of the crucifer flea beetle to Brassica volatiles in an olfactometer. Environ. Entomol. 2009, 38, 1467–1479. [Google Scholar] [CrossRef]
- Cappellari, S.C.; Harter-Marques, B.; Aumeier, P.; Engels, W. Mecardonia tenella (Plantaginaceae) attracts oil-, perfume-, and pollen-gathering bees in southern Brazil. Biotropica 2009, 41, 721–729. [Google Scholar] [CrossRef]
- Wang, S.; Ridsdill-Smith, T.; Ghisalberti, E. Chemical defenses of Trifolium glanduliferum against redlegged earth mite Halotydeus destructor. J. Agric. Food Chem. 2005, 53, 6240–6245. [Google Scholar] [CrossRef]
- Lwande, W.; Ndakala, A.J.; Hassanali, A.; Moreka, L.; Nyandat, E.; Ndungu, M.; Amiani, H.; Gitu, P.M.; Malonza, M.M.; Punyua, D.K. Gynandropsis gynandra essential oil and its constituents as tick (Rhipicephalus appendiculatus) repellents. Phytochemistry 1999, 50, 401–405. [Google Scholar] [CrossRef]
- Phoonan, W.; Deowanish, S.; Chavasiri, W. Food attractant from mulberry leaf tea and its main volatile compounds for the biocontrol of Lasioderma serricorne F. (Coleoptera: Anobiidae). J. Stored Prod. Res. 2014, 59, 299–305. [Google Scholar] [CrossRef]
- Höckelmann, C.; Moens, T.; Jüttner, F. Odor compounds from cyanobacterial biofilms acting as attractants and repellents for free-living nematodes. Limnol. Oceanogr. 2004, 49, 1809–1819. [Google Scholar] [CrossRef]
- Yang, G.; Zhang, Y.-N.; Gurr, G.M.; Vasseur, L.; You, M.-S. Electroantennogram and behavioral responses of Cotesia plutellae to plant volatiles. Insect Sci. 2016, 23, 245–252. [Google Scholar] [CrossRef]
- Asokkumar, S.; Naveenkumar, C.; Raghunandhakumar, S.; Kamaraj, S.; Anandakumar, P.; Jagan, S.; Devaki, T. Antiproliferative and antioxidant potential of beta-ionone against benzo (a) pyrene-induced lung carcinogenesis in Swiss albino mice. Mol. Cell. Biochem. 2012, 363, 335–345. [Google Scholar] [CrossRef]
- Liu, J.R.; Sun, X.R.; Dong, H.W.; Sun, C.H.; Sun, W.G.; Chen, B.Q.; Song, Y.Q.; Yang, B.F. β-Ionone suppresses mammary carcinogenesis, proliferative activity and induces apoptosis in the mammary gland of the Sprague-Dawley rat. Int. J. Oncol. 2008, 122, 2689–2698. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Liu, T.; Chen, J.; Yang, Z.; Xu, S.; Fan, Y.; Zeng, J.; Chen, Y.; Ma, Z.; Gao, Y.; et al. Activation of PSGR with β-ionone suppresses prostate cancer progression by blocking androgen receptor nuclear translocation. Cancer Lett. 2019, 453, 193–205. [Google Scholar] [CrossRef]
- Lu, Y.; Yang, Q.; Lin, Z.; Yang, X. A modular pathway engineering strategy for the high-level production of β-ionone in Yarrowia lipolytica. Microb. Cell Factories 2020, 19, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Czajka, J.J.; Nathenson, J.A.; Benites, V.T.; Baidoo, E.E.; Cheng, Q.; Wang, Y.; Tang, Y.J. Engineering the oleaginous yeast Yarrowia lipolytica to produce the aroma compound β-ionone. Microb. Cell Fact. 2018, 17, 136. [Google Scholar] [CrossRef] [PubMed]
- Ryan, O.W.; Skerker, J.M.; Maurer, M.J.; Li, X.; Tsai, J.C.; Poddar, S.; Lee, M.E.; DeLoache, W.; Dueber, J.E.; Arkin, A.P. Selection of chromosomal DNA libraries using a multiplex CRISPR system. eLife 2014, 3, e03703. [Google Scholar] [CrossRef] [PubMed]
- Strucko, T.; Maury, J.; Borodina, I. EasyClone: Method for iterative chromosomal integration of multiple genes in Saccharomyces cerevisiae. FEMS Yeast Res. 2014, 14, 238–248. [Google Scholar]
- Amen, T.; Kaganovich, D. Integrative modules for efficient genome engineering in yeast. Microbial Cell 2017, 4, 182. [Google Scholar] [CrossRef]
- Jessop-Fabre, M.M.; Jakočiūnas, T.; Stovicek, V.; Dai, Z.; Jensen, M.K.; Keasling, J.D.; Borodina, I. EasyClone-MarkerFree: A vector toolkit for marker-less integration of genes into Saccharomyces cerevisiae via CRISPR-Cas9. Biotechnol. J. 2016, 11, 1110–1117. [Google Scholar] [CrossRef]
- López, J.; Bustos, D.; Camilo, C.; Arenas, N.; Saa, P.A.; Agosin, E. Engineering Saccharomyces cerevisiae for the overproduction of β-ionone and its precursor β-carotene. Front. Bioeng. Biotechnol. 2020, 8. [Google Scholar] [CrossRef]
- Jullesson, D.; David, F.; Pfleger, B.; Nielsen, J. Impact of synthetic biology and metabolic engineering on industrial production of fine chemicals. Biotechnol. Adv. 2015, 33, 1395–1402. [Google Scholar] [CrossRef] [PubMed]
- Ko, Y.-S.; Kim, J.W.; Lee, J.A.; Han, T.; Kim, G.B.; Park, J.E.; Lee, S.Y. Tools and strategies of systems metabolic engineering for the development of microbial cell factories for chemical production. Chem. Soc. Rev. 2020, 49, 4615–4636. [Google Scholar] [CrossRef] [PubMed]
- Navarrete, C.; Jacobsen, I.H.; Martínez, J.L.; Procentese, A. Cell factories for industrial production processes: Current issues and emerging solutions. Processes 2020, 8, 768. [Google Scholar] [CrossRef]
- Gambacorta, F.V.; Dietrich, J.J.; Yan, Q.; Pfleger, B.F. Rewiring yeast metabolism to synthesize products beyond ethanol. Curr. Opin. Chem. Biol. 2020, 59, 182–192. [Google Scholar] [CrossRef]
- Liu, Y.; Nielsen, J. Recent trends in metabolic engineering of microbial chemical factories. Curr. Opin. Biotechnol. 2019, 60, 188–197. [Google Scholar] [CrossRef]
- Abdel-Mawgoud, A.M.; Markham, K.A.; Palmer, C.M.; Liu, N.; Stephanopoulos, G.; Alper, H.S. Metabolic engineering in the host Yarrowia lipolytica. Metab. Eng. 2018, 50, 192–208. [Google Scholar] [CrossRef]
- Beekwilder, J.; van Rossum, H.M.; Koopman, F.; Sonntag, F.; Buchhaupt, M.; Schrader, J.; Hall, R.D.; Bosch, D.; Pronk, J.T.; van Maris, A.J. Polycistronic expression of a β-carotene biosynthetic pathway in Saccharomyces cerevisiae coupled to β-ionone production. J. Biotechnol. 2014, 192, 383–392. [Google Scholar] [CrossRef]
- López, J.; Essus, K.; Kim, I.-k.; Pereira, R.; Herzog, J.; Siewers, V.; Nielsen, J.; Agosin, E. Production of β-ionone by combined expression of carotenogenic and plant CCD1 genes in Saccharomyces cerevisiae. Microb. Cell Factories 2015, 14, 84. [Google Scholar] [CrossRef]
- Werner, N.; Ramirez-Sarmiento, C.A.; Agosin, E. Protein engineering of carotenoid cleavage dioxygenases to optimize β-ionone biosynthesis in yeast cell factories. Food Chem. 2019, 299, 125089. [Google Scholar] [CrossRef]
- López, J.; Cataldo, V.F.; Peña, M.; Saitua, F.; Ibaceta, M.; Saa, P.A.; Agosin, E. Build your bioprocess on a solid strain-β-carotene production in recombinant Saccharomyces cerevisiae. Front. Bioeng. Biotechnol. 2019, 7, 171. [Google Scholar] [CrossRef]
- Zhang, C.; Chen, X.; Lindley, N.D.; Too, H.P. A “plug-n-play” modular metabolic system for the production of apocarotenoids. Biotechnol. Bioeng. 2018, 115, 174–183. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paparella, A.; Shaltiel-Harpaza, L.; Ibdah, M. β-Ionone: Its Occurrence and Biological Function and Metabolic Engineering. Plants 2021, 10, 754. https://doi.org/10.3390/plants10040754
Paparella A, Shaltiel-Harpaza L, Ibdah M. β-Ionone: Its Occurrence and Biological Function and Metabolic Engineering. Plants. 2021; 10(4):754. https://doi.org/10.3390/plants10040754
Chicago/Turabian StylePaparella, Antonello, Liora Shaltiel-Harpaza, and Mwafaq Ibdah. 2021. "β-Ionone: Its Occurrence and Biological Function and Metabolic Engineering" Plants 10, no. 4: 754. https://doi.org/10.3390/plants10040754
APA StylePaparella, A., Shaltiel-Harpaza, L., & Ibdah, M. (2021). β-Ionone: Its Occurrence and Biological Function and Metabolic Engineering. Plants, 10(4), 754. https://doi.org/10.3390/plants10040754







