Elevated Carbon Dioxide and Chronic Warming Together Decrease Nitrogen Uptake Rate, Net Translocation, and Assimilation in Tomato
Abstract
1. Introduction
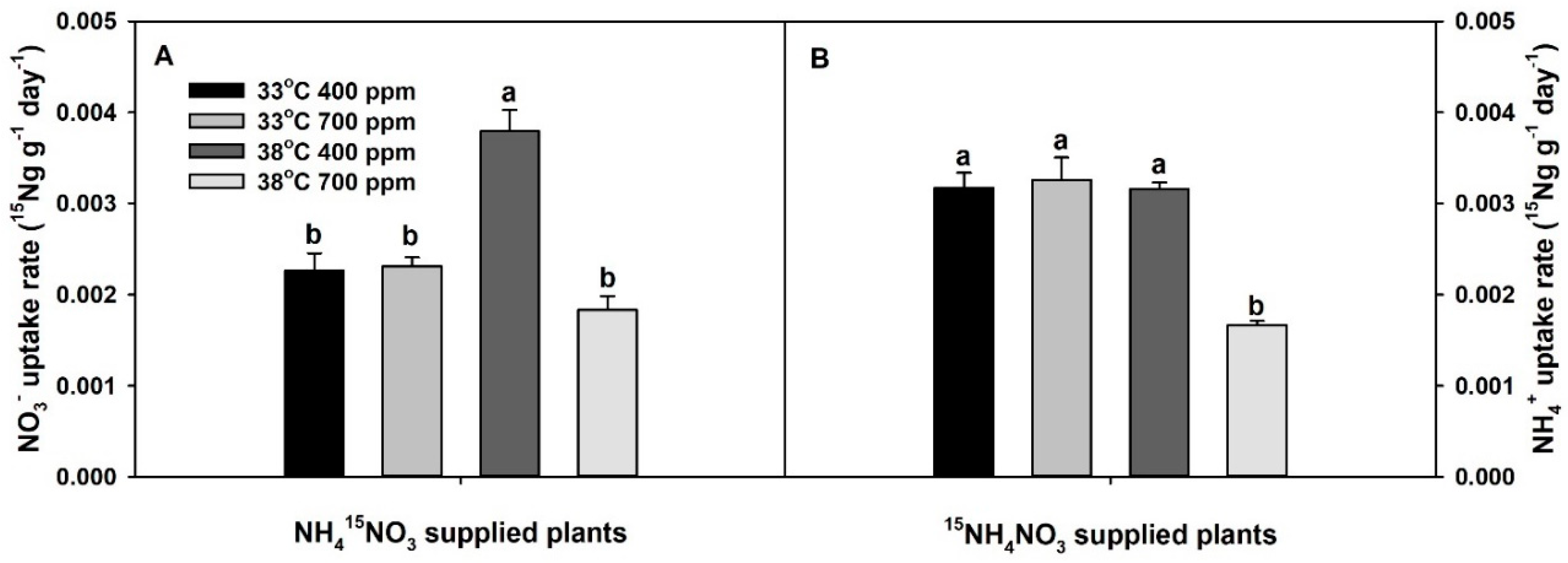
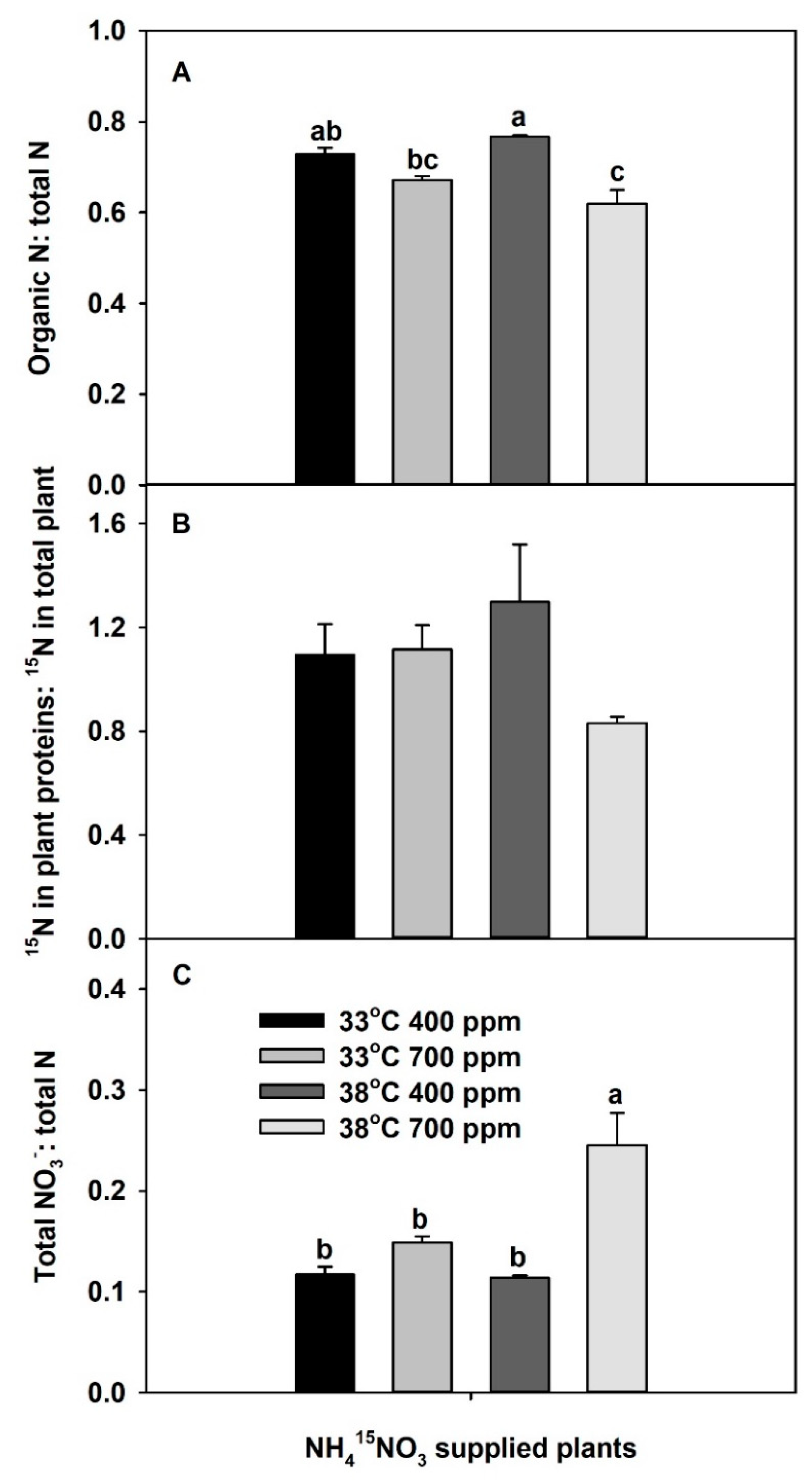
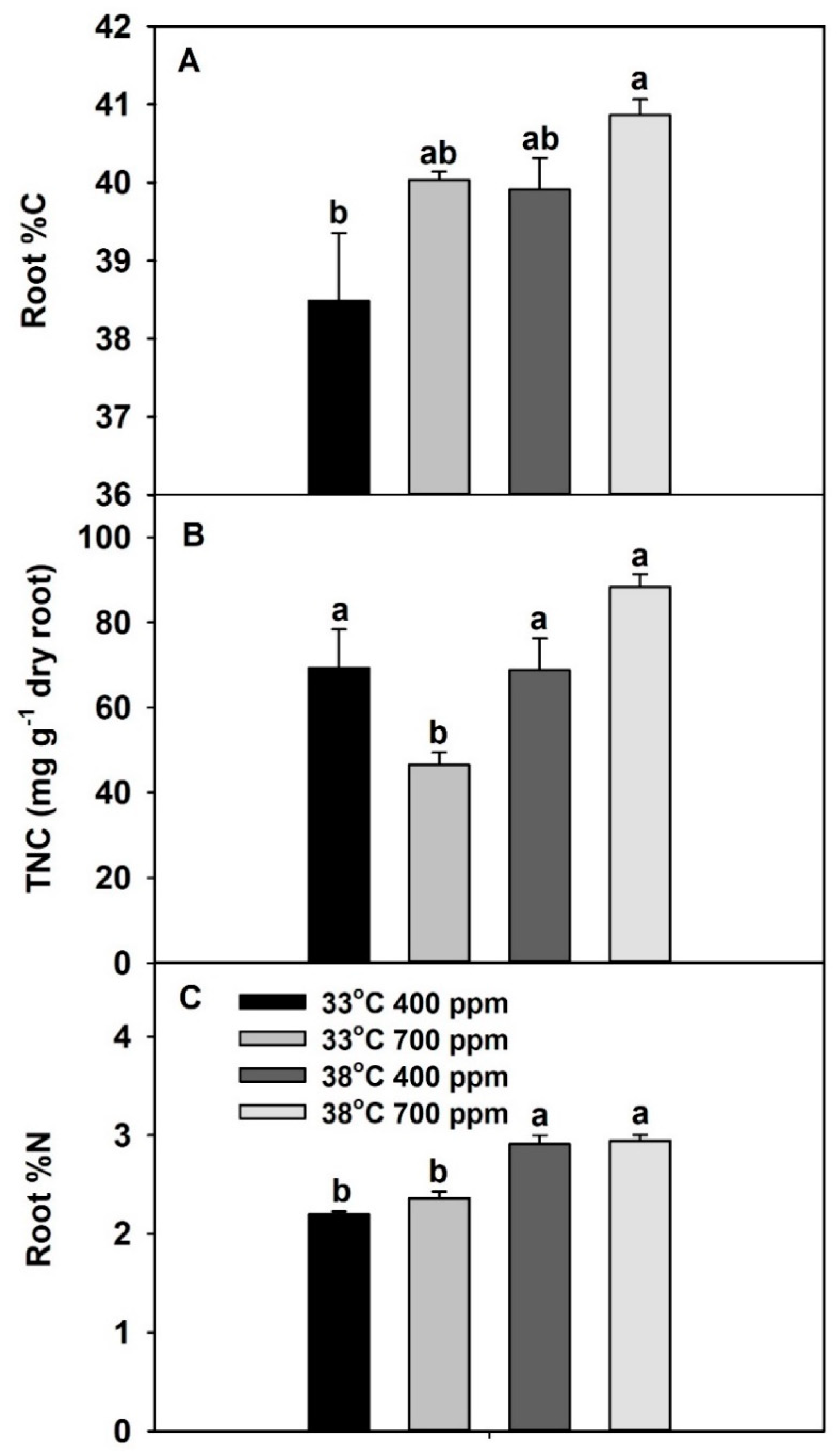
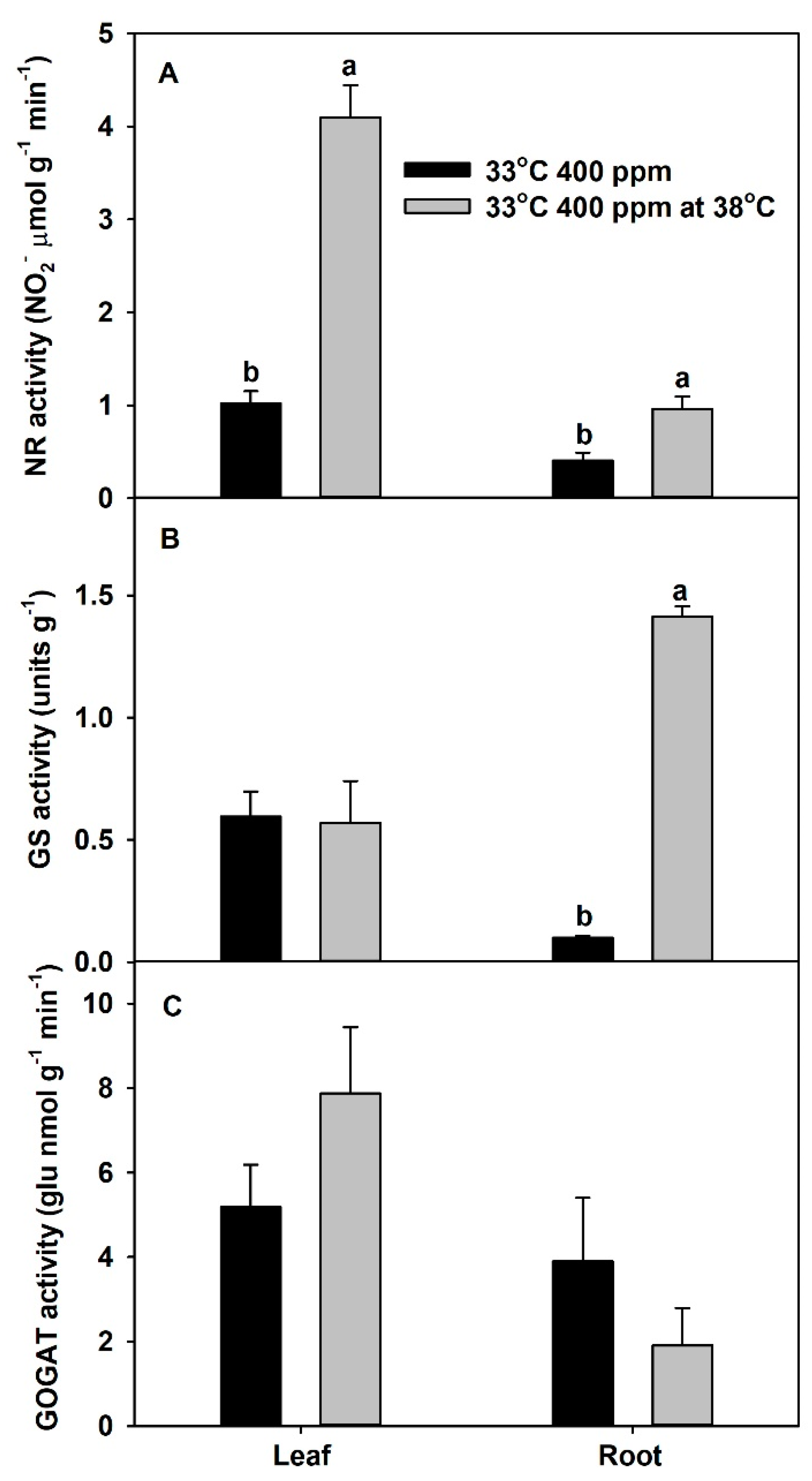
2. Results
3. Discussion
4. Materials and Methods
4.1. Plant Material, Growth Conditions, and Treatments
4.2. Plant N and Protein Analysis
4.3. Total Nonstructural Carbohydrate Assay
4.4. In Vitro N-Assimilatory-Protein Activity Assays
4.5. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- IPCC. Climate Change 2007: The Scientific Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Solomon, S., Qin, D., Manning, M., Chen, Z., Marquis, M., Averyt, K., Tignor, M., Miller, H., Eds.; Cambridge University Press: Cambridge, UK, 2007.
- IPCC. Climate Change 2014: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Field, C., Barros, V., Dokken, D., Mach, K., Mastrandrea, M., Bilir, T., Chatterjee, M., Ebi, K., Estrada, Y., Genova, R., et al., Eds.; Cambridge University Press: Cambridge, UK, 2014.
- Heckathorn, S.A.; Giri, A.; Mishra, S.; Bista, D. Heat stress and roots. In Climate Change and Plant Abiotic Stress Tolerance; Tuteja, N., Gill, S., Eds.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2014; pp. 109–136. [Google Scholar]
- Jauregui, I.; Aroca, R.; Garnica, M.; Zamarreño, Á.M.; García-Mina, J.M.; Serret, M.D.; Parry, M.; Irigoyen, J.J.; Aranjuelo, I. Nitrogen assimilation and transpiration: Key processes conditioning responsiveness of wheat to elevated [CO2] and temperature. Physiol. Plant. 2015, 155, 338–354. [Google Scholar] [CrossRef] [PubMed]
- Jayawardena, D.M.; Heckathorn, S.A.; Bista, D.R.; Mishra, S.; Boldt, J.K.; Krause, C.R. Elevated CO2 plus chronic warming reduce nitrogen uptake and levels or activities of nitrogen-uptake and -assimilatory proteins in tomato roots. Physiol. Plant. 2017, 159, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Shaw, M.R.; Zavaleta, E.S.; Chiariello, N.R.; Cleland, E.E.; Mooney, H.A.; Field, C.B. Grassland responses to global environmental changes suppressed by elevated CO2. Science 2002, 298, 1987–1990. [Google Scholar] [CrossRef]
- Wang, D.; Heckathorn, S.A.; Wang, X.; Philpott, S.M. A meta-analysis of plant physiological and growth responses to temperature and elevated CO2. Oecologia 2012, 169, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, J.I.; Delhaize, E.; Frommer, W.B.; Guerinot, M.L.; Harrison, M.J.; Herrera-Estrella, L.; Horie, T.; Kochian, L.V.; Munns, R.; Nishizawa, N.K.; et al. Using membrane transporters to improve crops for sustainable food production. Nature 2013, 497, 60–66. [Google Scholar] [CrossRef]
- Taub, D.R.; Wang, X. Why are nitrogen concentrations in plant tissues lower under elevated CO2? A critical examination of the hypotheses. J. Integr. Plant Biol. 2008, 50, 1365–1374. [Google Scholar] [CrossRef] [PubMed]
- Nacry, P.; Bouguyon, E.; Gojon, A. Nitrogen acquisition by roots: Physiological and developmental mechanisms ensuring plant adaptation to a fluctuating resource. Plant Soil 2013, 370, 1–29. [Google Scholar] [CrossRef]
- Näsholm, T.; Kielland, K.; Ganeteg, U. Uptake of organic nitrogen by plants. New Phytol. 2009, 182, 31–48. [Google Scholar] [CrossRef]
- Bassirirad, H. Kinetics of nutrient uptake by roots: Responses to global change. New Phytol. 2000, 147, 155–169. [Google Scholar] [CrossRef]
- Rubio-Asensio, J.S.; Bloom, A.J. Inorganic nitrogen form: A major player in wheat and Arabidopsis responses to elevated CO2. J. Exp. Bot. 2016, 68, 2611–2625. [Google Scholar] [CrossRef]
- Atkin, O.; Cummins, W.R. The effect of root temperature on the induction of nitrate reductase activities and nitrogen uptake rates in arctic plant species. Plant Soil 1994, 159, 187–197. [Google Scholar] [CrossRef]
- Clarkson, D.T.; Warner, A.J. Relationships between root temperature and the transport of ammonium and nitrate ions by Italian and perennial ryegrass (Lolium multiflorum and Lolium perenne). Plant Physiol. 1979, 64, 557–561. [Google Scholar] [CrossRef]
- Cruz, C.; Lips, S.H.; Martins-Loucao, M.A. Effect of root temperature on carob growth: Nitrate versus ammonium nutrition. J. Plant Nutr. 1993, 16, 1517–1530. [Google Scholar] [CrossRef]
- Tindall, J.A.; Mills, H.A.; Radcliffe, D.E. The effect of root zone temperature on nutrient uptake of tomato. J. Plant Nutr. 1990, 13, 939–956. [Google Scholar] [CrossRef]
- Bassirirad, H.; Caldwell, M.M.; Bilbrough, C. Effects of soil temperature and nitrogen status on kinetics of 15NO3− uptake by roots of field-grown Agropyron desertorum (Fisch. ex Link) Schult. New Phytol. 1993, 123, 485–489. [Google Scholar] [CrossRef]
- Delucia, E.H.; Heckathorn, S.A.; Day, T.A. Effects of soil temperature on growth, biomass allocation and resource acquisition of Andropogon gerardii Vitman. New Phytol. 1992, 120, 543–549. [Google Scholar] [CrossRef]
- Giri, A.; Heckathorn, S.; Mishra, S.; Krause, C. Heat stress decreases levels of nutrient-uptake and -assimilation proteins in tomato roots. Plants 2017, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- Mainali, K.P.; Heckathorn, S.A.; Wang, D.; Weintraub, M.N.; Frantz, J.M.; Hamilton, E.W. Impact of a short-term heat event on C and N relations in shoots vs. roots of the stress-tolerant C4 grass, Andropogon gerardii. J. Plant Physiol. 2014, 171, 977–985. [Google Scholar] [CrossRef]
- Coleman, J.S.; Bazzaz, F.A. Effects of CO2 and temperature on growth and resource use of co-occurring C3 and C4 annuals. Ecology 1992, 73, 1244–1259. [Google Scholar] [CrossRef]
- Arndal, M.F.; Schmidt, I.K.; Kongstad, J.; Beier, C.; Michelsen, A. Root growth and N dynamics in response to multi-year experimental warming, summer drought and elevated CO2 in a mixed heathland-grass ecosystem. Funct. Plant Biol. 2014, 41, 1–10. [Google Scholar] [CrossRef]
- Dijkstra, F.A.; Blumenthal, D.; Morgan, J.A.; Pendall, E.; Carrillo, Y.; Follett, R.F. Contrasting effects of elevated CO2 and warming on nitrogen cycling in a semiarid grassland. New Phytol. 2010, 187, 426–437. [Google Scholar] [CrossRef]
- Bassirirad, H.; Griffin, K.L.; Strain, B.R.; Reynolds, J.F. Effects of CO2 enrichment on growth and root 15NH4+ uptake rate of loblolly pine and ponderosa pine seedlings. Tree Physiol. 1996, 16, 957–962. [Google Scholar] [CrossRef]
- Cohen, I.; Halpern, M.; Yermiyahu, U.; Bar-Tal, A.; Gendler, T.; Rachmilevitch, S. CO2 and nitrogen interaction alters root anatomy, morphology, nitrogen partitioning and photosynthetic acclimation of tomato plants. Planta 2019, 250, 1423–1432. [Google Scholar] [CrossRef] [PubMed]
- Jauregui, I.; Aparicio-Tejo, P.M.; Avila, C.; Cañas, R.; Sakalauskiene, S.; Aranjuelo, I. Root-shoot interactions explain the reduction of leaf mineral content in Arabidopsis plants grown under elevated [CO2] conditions. Physiol. Plant. 2016, 158, 65–79. [Google Scholar] [CrossRef] [PubMed]
- Kruse, J.; Hetzger, I.; Hänsch, R.; Mendel, R.R.; Walch-Liu, P.; Engels, C.; Rennenberg, H. Elevated pCO2 favours nitrate reduction in the roots of wild-type tobacco (Nicotiana tabacum cv. Gat.) and significantly alters N-metabolism in transformants lacking functional nitrate reductase in the roots. J. Exp. Bot. 2002, 53, 2351–2367. [Google Scholar] [CrossRef][Green Version]
- van Ginkel, J.H.; Gorissen, A.; van Veen, J.A. Carbon and nitrogen allocation in Lolium perenne in response to elevated atmospheric CO2 with emphasis on soil carbon dynamics. Plant Soil 1997, 188, 299–308. [Google Scholar] [CrossRef]
- Bigot, J.; Boucaud, J. Short-term responses of Brassica rapa plants to low root temperature: Effects on nitrate uptake and its translocation to the shoot. Physiol. Plant. 1996, 96, 646–654. [Google Scholar] [CrossRef]
- Rufty, T.W.; Raper, C.D.; Jackson, W.A. Nitrogen assimilation, root growth and whole plant responses of soybean to root temperature, and to carbon dioxide and light in the aerial environment. New Phytol. 1981, 88, 607–619. [Google Scholar] [CrossRef]
- Sattelmacher, B.; Marschner, H.; Kühne, R. Effects of root zone temperature on root activity of two potato (Solanum tuberosum L.) clones with different adaptation to high temperature. J. Agron. Crop Sci. 1990, 165, 131–137. [Google Scholar] [CrossRef]
- Tachibana, S. The influence of root temperature on nitrate assimilation by cucumber and figleaf gourd. J. Jpn. Soc. Hortic. Sci. 1998, 57, 440–447. [Google Scholar] [CrossRef]
- Ezeta, F.N.; Jackson, W.A. Nitrate translocation by detopped corn seedlings. Plant Physiol. 1975, 56, 148–156. [Google Scholar] [CrossRef]
- Hungria, M.; Kaschuk, G. Regulation of N2 fixation and NO3−/NH4+ assimilation in nodulated and N-fertilized Phaseolus vulgaris L. exposed to high temperature stress. Environ. Exp. Bot. 2014, 98, 32–39. [Google Scholar] [CrossRef]
- Bloom, A.; Burger, M.A.; Kimball, B.J.; Pinter, J.P. Nitrate assimilation is inhibited by elevated CO2 in field-grown wheat. Nat. Clim. Chang. 2014, 4, 477–480. [Google Scholar] [CrossRef]
- Bloom, A.J.; Asensio, J.S.R.; Randall, L.; Rachmilevitch, S.; Cousins, A.B.; Carlisle, E.A. CO2 enrichment inhibits shoot nitrate assimilation in C3 but not C4 plants and slows growth under nitrate in C3 plants. Ecology 2012, 93, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Bloom, A.J.; Burger, M.; Asensio, J.S.R.; Cousins, A.B. Carbon dioxide enrichment inhibits nitrate assimilation in wheat and Arabidopsis. Science 2010, 328, 899–903. [Google Scholar] [CrossRef] [PubMed]
- Bloom, A.J.; Smart, D.R.; Nguyen, D.T.; Searles, P.S. Nitrogen assimilation and growth of wheat under elevated carbon dioxide. Proc. Natl. Acad. Sci. USA 2002, 99, 1730–1735. [Google Scholar] [CrossRef] [PubMed]
- Andrews, M.; Condron, L.M.; Kemp, P.D.; Topping, J.F.; Lindsey, K.; Hodge, S.; Raven, J.A. Elevated CO2 effects on nitrogen assimilation and growth of C3 vascular plants are similar regardless of N-form assimilated. J. Exp. Bot. 2019, 70, 683–690. [Google Scholar] [CrossRef]
- Bloom, A.J.; Kasemsap, P.; Rubio-Asensio, J.S. Rising atmospheric CO2 concentration inhibits nitrate assimilation in shoots but enhances it in roots of C3 plants. Physiol. Plant. 2020, 168, 963–972. [Google Scholar] [CrossRef]
- Amos, J.A.; Scholl, R.L. Effect of growth temperature on leaf nitrate reductase, glutamine synthetase and NADH-glutamate dehydrogenase of juvenile maize genotypes. Crop Sci. 1977, 17, 445–448. [Google Scholar] [CrossRef]
- Chopra, R.K. Effects of temperature on the in vivo assay of nitrate reductase in some C3 and C4 species. Ann. Bot. 1983, 51, 617–620. [Google Scholar] [CrossRef]
- Magalhães, A.C.; Peters, D.B.; Hageman, R.H. Influence of temperature on nitrate metabolism and leaf expansion in soybean (Glycine max L. Merr.) seedlings. Plant Physiol. 1976, 58, 12–16. [Google Scholar] [CrossRef]
- Rachmilevitch, S.; Huang, B.; Lambers, H. Assimilation and allocation of carbon and nitrogen of thermal and nonthermal Agrostis species in response to high soil temperature. New Phytol. 2006, 170, 479–490. [Google Scholar] [CrossRef]
- Vicente, R.; Pérez, P.; Martínez-Carrasco, R.; Usadel, B.; Kostadinova, S.; Morcuende, R. Quantitative RT–PCR platform to measure transcript levels of C and N metabolism-related genes in durum wheat: Transcript profiles in elevated [CO2] and high temperature at different levels of N supply. Plant Cell Physiol. 2015, 56, 1556–1573. [Google Scholar] [CrossRef] [PubMed]
- Masclaux-Daubresse, C.; Daniel-Vedele, F.; Dechorgnat, J.; Chardon, F.; Gaufichon, L.; Suzuki, A. Nitrogen uptake, assimilation and remobilization in plants: Challenges for sustainable and productive agriculture. Ann. Bot. 2010, 105, 1141–1157. [Google Scholar] [CrossRef]
- Jayawardena, D.M.; Heckathorn, S.A.; Bista, D.R.; Boldt, J.K. Elevated carbon dioxide plus chronic warming causes dramatic increases in leaf angle in tomato, which correlates with reduced plant growth. Plant Cell Environ. 2019, 42, 1247–1256. [Google Scholar] [CrossRef] [PubMed]
- Dieleman, W.I.J.; Vicca, S.; Dijkstra, F.A.; Hagedorn, F.; Hovenden, M.J.; Larsen, K.S.; Morgan, J.A.; Volder, A.; Beier, C.; Dukes, J.S.; et al. Simple additive effects are rare: A quantitative review of plant biomass and soil process responses to combined manipulations of CO2 and temperature. Glob. Chang. Biol. 2012, 18, 2681–2693. [Google Scholar] [CrossRef] [PubMed]
- McGrath, J.M.; Lobell, D.B. Reduction of transpiration and altered nutrient allocation contribute to nutrient decline of crops grown in elevated CO2 concentrations. Plant Cell Environ. 2013, 36, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Pate, J.S. Uptake, assimilation and transport of nitrogen compounds by plants. Soil Biol. Biochem. 1973, 5, 109–119. [Google Scholar] [CrossRef]
- Andrews, M. The partitioning of nitrate assimilation between root and shoot of higher plants. Plant Cell Environ. 1986, 9, 511–519. [Google Scholar] [CrossRef]
- Marschner, H. Mineral Nutrition of Higher Plants; Academic Press: London, UK, 1995. [Google Scholar]
- Alexandratos, N.; Bruinsma, J. World Agriculture towards 2030/2050: The 2012 Revision; ESA Working Paper, No. 12-03; Food and Agriculture Organization of the United Nations: Rome, Italy, 2012. [Google Scholar]
- Sung, J.; Lee, S.; Lee, Y.; Ha, S.; Song, B.; Kim, T.; Waters, B.M.; Krishnan, H.B. Metabolomic profiling from leaves and roots of tomato (Solanum lycopersicum L.) plants grown under nitrogen, phosphorus or potassium-deficient condition. Plant Sci. 2015, 241, 55–64. [Google Scholar] [CrossRef]
- Padilla, F.M.; Peña-Fleitas, M.T.; Gallardo, M.; Thompson, R.B. Threshold values of canopy reflectance indices and chlorophyll meter readings for optimal nitrogen nutrition of tomato. Ann. Appl. Biol. 2015, 166, 271–285. [Google Scholar] [CrossRef]
- Smart, D.R.; Ferro, A.; Ritchie, K.; Bugbee, B.G. On the use of antibiotics to reduce rhizoplane microbial populations in root physiology and ecology investigations. Physiol. Plant. 1995, 95, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Heckathorn, S.; Frantz, J.; Yu, F.; Gray, J. Effects of boron deficiency on geranium grown under different nonphotoinhibitory light levels. J. Am. Soc. Hortic. Sci. 2009, 134, 183–193. [Google Scholar] [CrossRef]
- Pilbeam, D. Nitrogen. In Handbook of Plant Nutrition; Barker, A., Pilbeam, D., Eds.; CRC Press: Boca Raton, FL, USA, 2015; pp. 17–64. [Google Scholar]
- Cataldo, D.A.; Maroon, M.; Schrader, L.E.; Youngs, V.L. Rapid colorimetric determination of nitrate in plant tissue by nitration of salicylic acid1. Commun. Soil Sci. Plant Anal. 1975, 6, 71–80. [Google Scholar] [CrossRef]
- Bloom, A.J.; Randall, L.; Taylor, A.R.; Silk, W.K. Deposition of ammonium and nitrate in the roots of maize seedlings supplied with different nitrogen salts. J. Exp. Bot. 2012, 63, 1997–2006. [Google Scholar] [CrossRef] [PubMed]
- Kandeler, E.; Gerber, H. Short-term assay of soil urease activity using colorimetric determination of ammonium. Biol. Fertil. Soils 1988, 6, 68–72. [Google Scholar] [CrossRef]
- Chow, P.S.; Landhäusser, S.M. A method for routine measurements of total sugar and starch content in woody plant tissues. Tree Physiol. 2004, 24, 1129–1136. [Google Scholar] [CrossRef]
- Berteli, F.; Corrales, E.; Guerrero, C.; Ariza, M.J.; Pliego, F.; Valpuesta, V. Salt stress increases ferredoxin-dependent glutamate synthase activity and protein level in the leaves of tomato. Physiol. Plant. 1995, 93, 259–264. [Google Scholar] [CrossRef]
- Lutts, S.; Majerus, V.; Kinet, J.M. NaCl effects on proline metabolism in rice (Oryza sativa) seedlings. Physiol. Plant. 1999, 105, 450–458. [Google Scholar] [CrossRef]
- Darmaun, D.; Manary, M.J.; Matthews, D.E. A method for measuring both glutamine and glutamate levels and stable isotopic enrichments. Anal. Biochem. 1985, 147, 92–102. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jayawardena, D.M.; Heckathorn, S.A.; Rajanayake, K.K.; Boldt, J.K.; Isailovic, D. Elevated Carbon Dioxide and Chronic Warming Together Decrease Nitrogen Uptake Rate, Net Translocation, and Assimilation in Tomato. Plants 2021, 10, 722. https://doi.org/10.3390/plants10040722
Jayawardena DM, Heckathorn SA, Rajanayake KK, Boldt JK, Isailovic D. Elevated Carbon Dioxide and Chronic Warming Together Decrease Nitrogen Uptake Rate, Net Translocation, and Assimilation in Tomato. Plants. 2021; 10(4):722. https://doi.org/10.3390/plants10040722
Chicago/Turabian StyleJayawardena, Dileepa M., Scott A. Heckathorn, Krishani K. Rajanayake, Jennifer K. Boldt, and Dragan Isailovic. 2021. "Elevated Carbon Dioxide and Chronic Warming Together Decrease Nitrogen Uptake Rate, Net Translocation, and Assimilation in Tomato" Plants 10, no. 4: 722. https://doi.org/10.3390/plants10040722
APA StyleJayawardena, D. M., Heckathorn, S. A., Rajanayake, K. K., Boldt, J. K., & Isailovic, D. (2021). Elevated Carbon Dioxide and Chronic Warming Together Decrease Nitrogen Uptake Rate, Net Translocation, and Assimilation in Tomato. Plants, 10(4), 722. https://doi.org/10.3390/plants10040722







