Phytochemical Screening, Phenolic Compounds and Antioxidant Activity of Biomass from Lychnis flos-cuculi L. In Vitro Cultures and Intact Plants
Abstract
1. Introduction
2. Results
2.1. Plant Material Obtained from In Vitro Cultures
2.2. Preliminary TLC Analysis
2.3. UHPLC-MS Analysis
2.4. Total Phenolic, Total Phenolic Acid and Total Flavonoid Content
2.5. Preliminary Determination of Antioxidant Activity and Radical Scavenging Activity
2.6. The Correlation between Phenolic Content and Antioxidant Activity
3. Discussion
4. Materials and Methods
4.1. Plant Material and In Vitro Cultures
4.2. Preliminary TLC Analysis
4.3. UHPLC-MS Analysis
4.3.1. Plant Material and Preparation of Samples
4.3.2. UHPLC Conditions
4.4. Determination of the Total Phenolic (TP), Total Phenolic Acid (TPA) and Total Flavonoid (TF) Content
4.5. Determination of the Antioxidant Capacity
4.6. Determination of the Radical Scavenging Activity
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Maliński, M.P.; Michalska, A.D.; Tomczykowa, M.; Tomczyk, M.; Thiem, B. Ragged Robin (Lychnis flos-cuculi)—A plant with potential medicinal value. Rev. Bras. Farmacogn. 2014, 24, 722–730. [Google Scholar] [CrossRef]
- Tiţă, I.; Mogoşanu, G.D.; Tiţă, M.G. Ethnobotanical inventory of medicinal plants from the South-West of Romania. Farmacia 2009, 57, 141–156. [Google Scholar]
- Chandra, S.; Rawat, D.S. Medicinal plants of the family Caryophyllaceae: A review of ethno-medicinal uses and pharmacological properties. Integr. Med. Res. 2015, 4, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Bucharow, W.G.; Chirva, V.J.; Bucharowa, I.L. Triterpene glycosides from Coronaria flos-cuculi. Pharmazie 1974, 29, 540. [Google Scholar]
- Báthori, M.; Lafont, R.; Girault, J.P.; Máthé, I. Structural diversity of ecdysteroids of Lychnis flos-cuculi. Acta Pharm. Hung. 2001, 71, 157–167. [Google Scholar]
- Tomczyk, M. Preliminary phytochemical investigation of Lychnis flos-cuculi herbs. J. Nat. Med. 2008, 62, 473–475. [Google Scholar] [CrossRef]
- Costea, T.; Nencu, I.; Gîrd, C.E.; Popescu, M.L. Ragged robin (Lychnis flos cuculi L.) aerial parts—Botanical characterization, phytochemical screening and antioxidant activity. Studia Univ. Vasile Goldis Arad Seria Stiintele Vietii 2017, 27, 231–238. [Google Scholar]
- Mamadalieva, N.Z.; Egamberdieva, D.; Lafont, R.; Girault, J.P. Phytoecdysteroids and antibacterial activity of the plant Coronaria flos-cuculi. Chem. Nat. Comp. 2008, 44, 404–406. [Google Scholar] [CrossRef]
- Dinan, L.; Lafont, R. Effects and applications of arthropod steroid hormones (ecdysteroids) in mammals. J. Endocrinol. 2006, 191, 1–8. [Google Scholar] [CrossRef]
- Moses, T.; Papadopoulou, K.K.; Osbourn, A. Metabolic and functional diversity of saponins, biosynthetic intermediates and semi-synthetic derivatives. Crit. Rev. Biochem. Mol. Biol. 2014, 49, 439–462. [Google Scholar] [CrossRef]
- Li, A.N.; Li, S.; Zhang, Y.J.; Xu, X.R.; Chen, Y.M.; Li, H.B. Resources and Biological Activities of Natural Polyphenols. Nutrients 2014, 6, 6020–6047. [Google Scholar] [CrossRef] [PubMed]
- Maliński, M.P.; Kikowska, M.; Kruszka, D.; Napierała, M.; Florek, E.; Sliwinska, E.; Thiem, B. Various in vitro systems of Ragged Robin (Lychnis flos-cuculi L.): A new potential source of phytoecdysteroids? Plant Cell Tiss. Organ Cult. 2019, 139, 39–52. [Google Scholar] [CrossRef]
- Matkowski, A. Plant in vitro culture for the production of antioxidants—A review. Biotechnol. Adv. 2008, 26, 548–560. [Google Scholar] [CrossRef] [PubMed]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.-Y.; Li, Q.; Bi, K.-S. Bioactive flavonoids in medicinal plants: Structure, activity and biological fate. Asian J. Pharm. Sci. 2018, 13, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. The antioxidant paradox: Less paradoxical now? Br. J. Clin. Pharmacol. 2012, 75, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Alfa, H.H.; Arroo, R.R.J. Over 3 decades of research on dietary flavonoid antioxidants and cancer prevention: What have we achieved? Phytochem. Rev. 2019, 18, 989–1004. [Google Scholar] [CrossRef]
- Smetanska, I. Production of secondary metabolites using plant cell cultures. Adv. Biochem. Eng. Biotechnol. 2008, 111, 187–228. [Google Scholar] [CrossRef]
- Chandana, B.C.; Kumari Nagaveni, H.C.; Lakshmana, D.; Shashikala, S.K.; Heena, M.S. Role of plant tissue culture in micropropagation, secondary metabolite production and conservation of some endangered medicinal crops. J. Pharmacogn. Phytochem. 2018, SP3, 246–251. [Google Scholar]
- Thiem, B.; Kikowska, M.; Maliński, M.P.; Kruszka, D.; Napierała, M.; Florek, E. Ecdysteroids: Production in plant in vitro cultures. Phytochem. Rev. 2017, 16, 603–622. [Google Scholar] [CrossRef]
- Kikowska, M.; Thiem, B.; Szopa, A.; Klimek-Szczykutowicz, M.; Rewers, M.; Sliwinska, E.; Ekiert, H. Comparative analysis of phenolic acids and flavonoids in shoot cultures of Eryngium alpinum L.: An endangered and protected species with medicinal value. Plant Cell Tiss. Organ Cult. 2019, 139, 167–175. [Google Scholar] [CrossRef]
- Miguel-Chávez, R. Phenolic Antioxidant Capacity: A Review of the State of the Art. In Phenolic Compounds: Biological Activity; Soto-Hernandez, M., Ed.; InTechOpen: London, UK, 2017. [Google Scholar] [CrossRef]
- Simirgiotis, M.J.; Schmeda-Hirschmann, G.; Borguez, J.; Kennelly, E.J. The Passiflora tripartita (Banana Passion) fruit: A source of bioactive flavonoid C-glycosides isolated by HSCCC and characterized by HPLC–DAD–ESI/MS/MS. Molecules 2013, 18, 1672–1692. [Google Scholar] [CrossRef] [PubMed]
- Wilson, I.D.; Morgan, E.D.; Lafont, R.; Shockcor, J.P.; Lindon, J.C.; Nicholson, J.K.; Wright, B. High performance liquid chromatography coupled to nuclear magnetic resonance spectroscopy and mass spectrometry applied to plant products: Identification of ecdysteroids from Silene otites. Chromatographia 1999, 49, 374–378. [Google Scholar] [CrossRef]
- Meng, Y.; Whiting, P.; Zibareva, L.; Bertho, G.; Girault, J.P.; Lafont, R.; Dinan, L. Identification and quantitative analysis of the phytoecdysteroids in Silene species (Caryophyllaceae) by high-performance liquid chromatography—Novel ecdysteroids from S. pseudotites. J. Chromatogr. A 2001, 935, 309–319. [Google Scholar] [CrossRef]
- Novozhilova, E.; Rybin, V.; Gorovoy, P.; Gavrilenko, I.; Doudkin, R. Phytoecdysteroids of the East Asian Caryophyllaceae. Pharmacogn. Mag. 2015, 11, 225–230. [Google Scholar] [CrossRef][Green Version]
- Mamadalieva, N.Z.; Lafont, R.; Wink, M. Diversity of secondary metabolites in the genus Silene L. (Caryophyllaceae)—Structures, distribution, and biological properties. Diversity 2014, 6, 415–499. [Google Scholar] [CrossRef]
- Zibareva, L.N.; Seliverstova, A.A.; Suksamrar, A.; Morozov, S.V.; Chernyak, E.I. Phytoecdysteroids from the aerial part of Silene colpophylla. Chem. Nat. Comp. 2014, 50, 571–572. [Google Scholar] [CrossRef]
- Wang, Y.H.; Avula, B.; Jadhav, A.N.; Smillie, T.J.; Khan, I.A. Structural characterization and identification of ecdysteroids from Sida rhombifolia L. in positive electrospray ionization by tandem mass spectrometry. Rapid Comm. Mass Spectrom. 2008, 22, 2413–2422. [Google Scholar] [CrossRef]
- Zibareva, L.N.; Saatov, Z.; Abubakirov, N.K. Stachysterone D, viticosterone E and α-ecdysone from Lychnis chalcedonica. Chem. Nat. Comp. 1991, 27, 514–515. [Google Scholar] [CrossRef]
- Courts, F.L.; Williamson, G. The occurrence, fate and biological activities of C-glycosyl flavonoids in the human diet. Crit. Rev. Food Sci. Nutr. 2015, 55, 1352–1367. [Google Scholar] [CrossRef]
- Böttger, S.; Melzig, M.F. Triterpenoid saponins of the Caryophyllaceae and Illecebraceae family. Phytochem. Lett. 2011, 4, 59–68. [Google Scholar] [CrossRef]
- Karamian, R.; Ghasemlou, F. Screening of total phenol and flavonoid content, antioxidant and antibacterial activities of the methanolic extracts of three Silene species from Iran. Int. J. Agric. Crop Sci. 2015, 5, 205–312. [Google Scholar]
- Kikowska, M.A.; Chmielewska, M.; Włodarczyk, A.; Studzińska-Sroka, E.; Żuchowski, J.; Stochmal, A.; Kotwicka, M.; Thiem, B. Effect of Pentacyclic Triterpenoids-Rich Callus Extract of Chaenomeles japonica (Thunb.) Lindl. Ex Spach on Viability, Morphology, and Proliferation of Normal Human Skin Fibroblasts. Molecules 2018, 23, 3009. [Google Scholar] [CrossRef] [PubMed]
- Báthori, M.; Zupko, I.; Hunyadi, A.; Gacsne-Baitz, E.; Dinya, Z.; Forgo, P. Monitoring the antioxidant activity of extracts originated from various Serratula species and isolation of flavonoids from Serratula coronata. Fitoterapia 2004, 75, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Miliauskas, G.; van Beek, T.A.; de Waard, P.; Venskutonis, R.P.; Sudhölter, E.J.R. Identification of radical scavenging compounds in Rhaponticum carthamoides by means of LC-DAD-SPE-NMR. J. Nat. Prod. 2005, 68, 168–172. [Google Scholar] [CrossRef] [PubMed]
- Das, N.; Mishra, S.K.; Bishayee, A.; Ali, E.S.; Bishayee, A. The phytochemical, biological, and medicinal attributes of phytoecdysteroids: An updated review. Acta Pharm. Sin. B 2020. in Press. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin–Ciocalteu reagent. Meth. Enzymol. 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Polish Pharmaceutical Society. Polish Pharmacopoeia VI; Polish Pharmaceutical Society: Warsaw, Poland, 2002. [Google Scholar]
- Meda, A.; Lamien, C.E.; Romito, M.; Milogo, J.; Nacoulma, O.G. Determination of the total phenolic, flavonoid and proline contents in Burkina Fasan honey, as well as their radical scavenging activity. Food Chem. 2005, 91, 571–577. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Annegowda, H.V.; Anwar, L.N.; Mordi, M.N.; Ramanathans, S.; Mansor, S.M. Influence of sonication on the phenolic content and antioxidant activity of Terminalia catappa L. leaves. Pharmacogn. Res. 2010, 2, 368–373. [Google Scholar] [CrossRef] [PubMed]

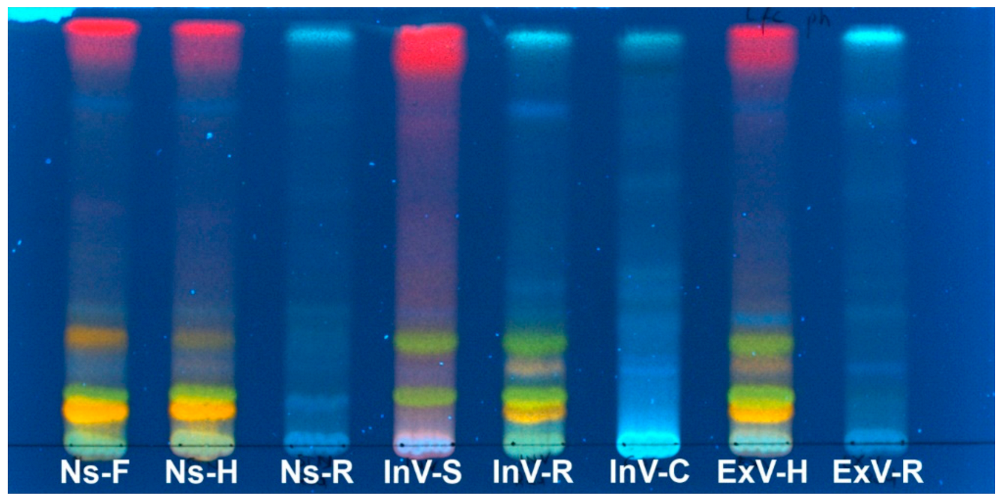
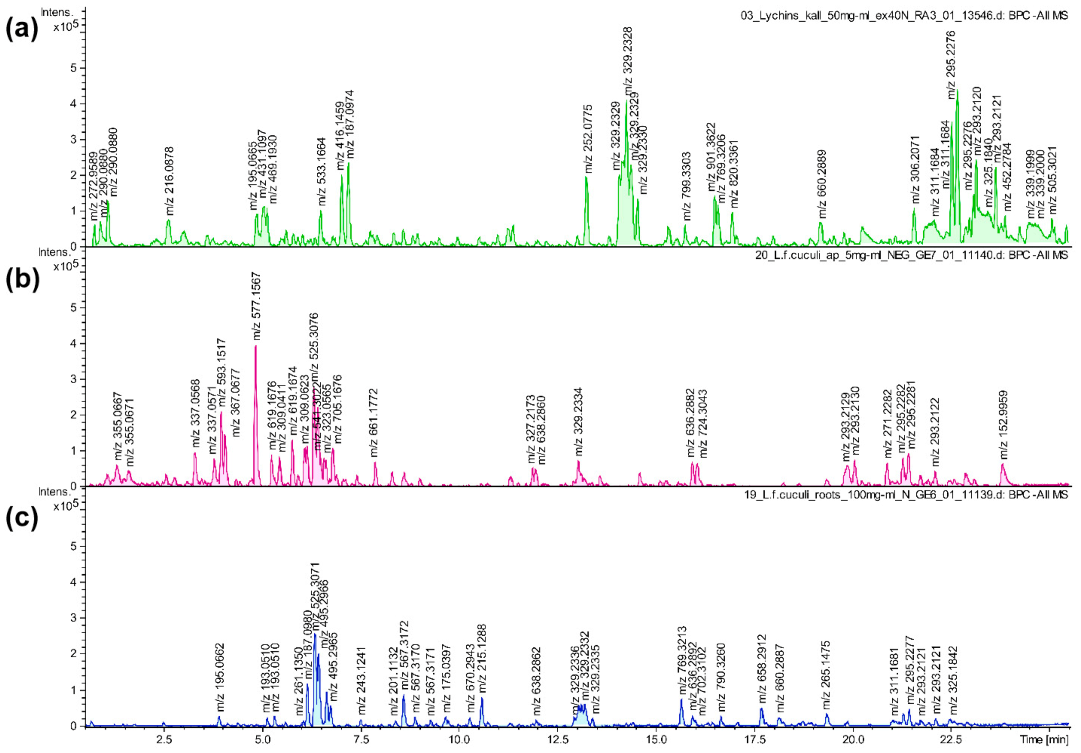
| No. | Rt (min) | Tentative Identification | UV (nm) | [M + H]+ m/z | [M − H]− m/z | MS2 | Compound Class | Present in: | References |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 0.66 | quinic acid derivative | 197 | 191 | 377, 191 | PA | R | not found | |
| 2 | 0.68 | quinic acid derivative | 197 | 377 | 377, 191 | PA | H | not found | |
| 3 | 0.87 | quinic acid | 195 | 191 | 191 | PA | H, R | not found | |
| 4 | 2.46 | quinic acid derivative | 355 | 355, 191 | PA | H | not found | ||
| 5 | 3.01 | quinic acid derivative | 355 | 355, 209, 191 | PA | H | not found | ||
| 6 | 4.8 | ferulic acid derivative | 198, 282 | 195 | 136, 195 | PA | C | not found | |
| 7 | 5.04 | apigenin derivative | 218, 290, 350 | 433 | 253, 271, 433 | F | C | not found | |
| 8 | 5.39 | benzoic acid glycoside | 198, 224 | 253 | 253, 121 | PA | R | not found | |
| 9 | 6.05 | dalpanin | 202, 222, 327 | 533 | 267, 353, 533 | F | C | not found | |
| 10 | 6.5 | apigenin-6,8-di-C-β-D-glucopyranoside (vicenin II) | 212, 269, 350 | 595 | 595, 449, 431, 329 | F | H | [6,21] | |
| 11 | 7.48 | vitexin rhamnoside (vitexin derivative) | 214, 270, 339 | 579 | 579, 433, 415, 313 | F | H | [6,21] | |
| 12 | 8 | luteolin-8-C-β-D-glucopyranoside derivative (orientin derivative) | 215, 269, 341 | 619 | 619, 607, 447, 323 | F | H | [6,21] | |
| 13 | 8.14 | apigenin-5-O-β-D-glucopyranosyl, 8-C-(6″acetyl)-β-D-glucopyranoside | 213, 269, 350 | 635 | 635, 619, 607, 329 | F | H | [6,21] | |
| 14 | 8.42 | 20-hydroxyecdysone | 247 | 479 | 525, 479 | E | H, R | [22,23,24] | |
| 15 | 8.47 | polypodine B | 228 | 495 | 541, 495 | E | H, R | [22,23] | |
| 16 | 8.83 | 20-hydroxyecdysone derivative (ajugasterone C) | 220 | 479 | 525, 479 | E | R | [22,23,24] | |
| 17 | 8.91 | integristerone A | 220 | 495 | 541, 495 | E | R | [22,23,24] | |
| 18 | 9.16 | luteolin-8-C-β-D-glucopyranoside derivative (orientin derivative) isomer I | 214, 270, 338 | 619 | 619, 577, 495, 315 | F | H | [6,21] | |
| 19 | 9.51 | unknown C-glycosyl derivative | 215, 270, 339 | 661 | 661, 619, 523 | F | H | [21] | |
| 20 | 9.7 | unknown C-glycosyl derivative | 215, 271, 330 | 705 | 705, 661, 619, 309 | F | H | [21] | |
| 21 | 9.85 | quillaic acid or gypsogenic acid-triterpene glycoside derivative | 972 | 972, 648, 485, 323 | TS | R | [25] | ||
| 22 | 10.09 | ecdysone | 465 | 465, 447, 429 | E | R | [24,26,27] | ||
| 23 | 10.6 | apigenin-5-O-β-D-glucopyranosyl, 8-C-(6″acetyl)-β-D-glucopyranoside derivative | 219, 271, 330 | 621 | 621, 475, 379, 313 | F | H | [6,21] | |
| 24 | 10.76 | viticosterone E | 222 | 521 | 567, 521 | E | R | [27,28] | |
| 25 | 10.83 | unknown C-glycosyl derivative | 216, 270, 338 | 661 | 661, 619, 509 | F | H | [21] | |
| 26 | 11.34 | unknown C-glycosyl derivative | 217, 270, 338 | 703 | 747, 703, 661 | F | H | [6,21] | |
| 27 | 11.81 | quillaic acid or gypsogenic acid-triterpene glycoside derivative | 649 | 649, 487, 469, 325 | TS | R | [25] | ||
| 28 | 12.13 | quillaic acid or gypsogenic acid-triterpene glycoside derivative | 730 | 730, 649, 487, 325 | TS | R | [25] | ||
| 29 | 12.53 | quillaic acid or gypsogenic acid-triterpene glycoside derivative | 1150 | 1150, 663, 485, 351 | TS | H | [25] | ||
| 30 | 12.68 | quillaic acid or gypsogenic acid-triterpene glycoside derivative | 735 | 735, 648, 485, 323 | TS | R | [25] | ||
| 31 | 12.95 | quillaic acid or gypsogenic acid-triterpene glycoside derivative, isomer I | 1134 | 1134, 647, 485, 310 | TS | H | [25] | ||
| 32 | 13.88 | quillaic acid or gypsogenic acid-triterpene glycoside derivative | 1134 | 1134, 711, 647, 485, 323 | TS | R | [25] | ||
| 33 | 13.89 | quillaic acid or gypsogenic acid-triterpene glycoside derivative, isomer II | 1134 | 1134, 647, 485, 310 | TS | H | [25] | ||
| 34 | 14.4 | unidentified | not detected | 659 | 211, 329, 659 | TS | C | not found | |
| 35 | 14.61 | gypsogenin-triterpene glycoside derivative | 1231 | 639, 469, 350 | TS | H | [25] | ||
| 36 | 14.62 | gypsogenin-triterpene glycoside derivative | 1231 | 638, 469, 307 | TS | R | [25] | ||
| 37 | 15.3 | quillaic acid or gypsogenic acid-triterpene glycoside derivative | not detected | 880 | 191, 405, 485, 761, 880 | TS | C | not found | |
| 38 | 15.48 | quillaic acid or gypsogenic acid-triterpene glycoside derivative, isomer III | 972 | 972, 485, 312 | TS | H | [25] | ||
| 39 | 15.8 | quillaic acid or gypsogenic acid-triterpene glycoside derivative | not detected | 799 | 191, 330, 405, 485, 661, 799 | TS | C | not found | |
| 40 | 16.14 | unidentified | 1116 | 1116, 771, 329 | TS | H | not found | ||
| 41 | 16.49 | unidentified | 1684 | 842, 792, 624, 329 | TS | H | not found | ||
| 42 | 16.54 | quillaic acid or gypsogenic acid-triterpene glycoside derivative | not detected | 901 | 191, 405, 485, 761, 901 | TS | C | [25] | |
| 43 | 16.61 | quillaic acid or gypsogenic acid-triterpene glycoside derivative | not detected | 769 | 405, 411, 485, 761, 769 | TS | C | not found | |
| 44 | 16.94 | oleanolic acid-triterpene glycoside derivative | 1452 | 726, 467 | TS | R | [25] | ||
| 45 | 16.97 | quillaic acid or gypsogenic acid-triterpene glycoside derivative | not detected | 820 | 191, 330, 405, 485, 661, 820 | TS | C | not found | |
| 46 | 17.41 | quillaic acid or gypsogenic acid-triterpene glycoside derivative | 1138 | 726, 487, 469, 189 | TS | R | [25] | ||
| 47 | 17.63 | unidentified | 929 | 929, 883 | TS | H | not found | ||
| 48 | 18.39 | unidentified | 1364 | 682 | TS | H | not found | ||
| 49 | 18.79 | quillaic acid or gypsogenic acid-triterpene glycoside derivative | 1494 | 749, 487, 393, 189 | TS | R | [25] | ||
| 50 | 19.06 | gypsogenin-triterpene glycoside derivative | 717 | 717, 469, 453, 189 | TS | R | [25] | ||
| 51 | 19.2 | quillaic acid or gypsogenic acid-triterpene glycoside derivative | not detected | 1275 | 177, 405, 485, 599, 660, 1275 | TS | C | not found | |
| 52 | 19.5 | unidentified | 1406 | 703 | TS | H | not found | ||
| 53 | 19.75 | quillaic acid or gypsogenic acid-triterpene glycoside derivative, isomer I | 768 | 768, 485, 435, 323 | TS | R | [25] | ||
| 54 | 20.02 | quillaic acid or gypsogenic acid-triterpene glycoside derivative, isomer II | 768 | 768, 485, 435, 323 | TS | R | [25] | ||
| 55 | 20.37 | gypsogenin-triterpene glycoside derivative | 739 | 739, 469, 453, 189 | TS | R | [25] |
| Plant Material | Total Phenolics (mg GAE g−1 d.w.) | Total Phenolic Acids (mg CAE g−1 d.w.) | Total Flavonoids (µg QE g−1 d.w.) | FRAP (mg AAE g−1 d.w.) | DPPH, IC50 (mg d.w. mL−1) |
|---|---|---|---|---|---|
| Ns-F | 195.40 ± 4.68 a | 3.88 ± 0.22 a | 1469 ± 76 a | 20.14 ± 0.62 a | 4.33 ± 1.51 a |
| Ns-H | 112.10 ± 6.77 b | 2.62 ± 0.14 b | 1232 ± 55 b | 11.46 ± 0.16 b | 10.97 ± 0.33 b |
| Ns-R | 41.86 ± 1.31 d | 0.52 ± 0.03 f | 14 ± 3 f | 5.72 ± 0.37 c | 52.78 ± 3.03 e |
| InV-S | 106.38 ± 5.91 b | 1.60 ± 0.05 d | 701 ± 50 d | 5.96 ± 0.23 c | 43.47 ± 1.77 d |
| InV-R | 63.60 ± 6.31 c | 1.08 ± 0.27 e | 470 ± 23 e | 5.13 ± 0.13 d | 44.22 ± 2.24 d |
| InV-C | 22.07 ± 0.68 e | 2.13 ± 0.17 c | 0 ± 0 g | 2.02 ± 0.13 e | >100 1 |
| ExV-H | 113.47 ± 4.95 b | 2.25 ± 0.09 c | 886 ± 49 c | 11.42 ± 0.63 b | 19.58 ± 0.75 c |
| ExV-R | 43.54 ± 5.30 d | 0.44 ± 0.05 g | 7 ± 3 f | 5.18 ± 0.41 d | 93.30 ± 3.99 f |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maliński, M.P.; Kikowska, M.A.; Soluch, A.; Kowalczyk, M.; Stochmal, A.; Thiem, B. Phytochemical Screening, Phenolic Compounds and Antioxidant Activity of Biomass from Lychnis flos-cuculi L. In Vitro Cultures and Intact Plants. Plants 2021, 10, 206. https://doi.org/10.3390/plants10020206
Maliński MP, Kikowska MA, Soluch A, Kowalczyk M, Stochmal A, Thiem B. Phytochemical Screening, Phenolic Compounds and Antioxidant Activity of Biomass from Lychnis flos-cuculi L. In Vitro Cultures and Intact Plants. Plants. 2021; 10(2):206. https://doi.org/10.3390/plants10020206
Chicago/Turabian StyleMaliński, Michał P., Małgorzata Anna Kikowska, Agata Soluch, Mariusz Kowalczyk, Anna Stochmal, and Barbara Thiem. 2021. "Phytochemical Screening, Phenolic Compounds and Antioxidant Activity of Biomass from Lychnis flos-cuculi L. In Vitro Cultures and Intact Plants" Plants 10, no. 2: 206. https://doi.org/10.3390/plants10020206
APA StyleMaliński, M. P., Kikowska, M. A., Soluch, A., Kowalczyk, M., Stochmal, A., & Thiem, B. (2021). Phytochemical Screening, Phenolic Compounds and Antioxidant Activity of Biomass from Lychnis flos-cuculi L. In Vitro Cultures and Intact Plants. Plants, 10(2), 206. https://doi.org/10.3390/plants10020206






