Licorice (Glycyrrhiza glabra) Extracts-Suitable Pharmacological Interventions for COVID-19? A Review
Abstract
1. Introduction
2. Methodology
3. Phytochemical Actions on the Human Body
4. Licorice (Glycyrrhiza glabra)
4.1. Antimicrobial Activity
4.1.1. Antiviral Activity
In Vitro Studies
In Vivo Studies
Human Studies
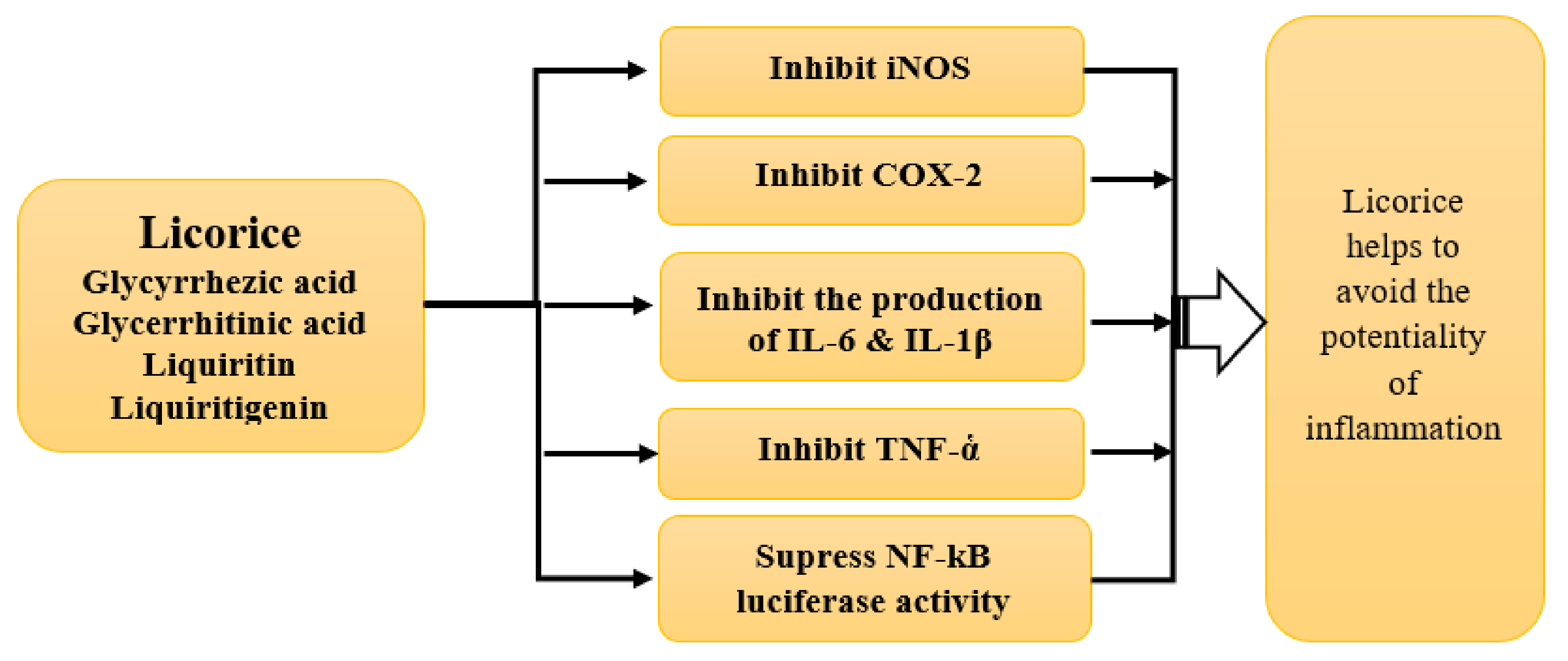
4.2. Anti-Inflammatory Property
4.2.1. In Vitro Studies
4.2.2. Animal Studies
| Compound | Tissue/Disease | Concentration | Method | Inhibition Rate | Reference(s) |
|---|---|---|---|---|---|
| In vitro studies | |||||
| 8β-GL | LPS (1 μg mL−1)-induced l Murine cell (RAW 264.7) | 75 μM | ELISA | 51%-NO, 51%-IL-1β, 49%-PGE2 & 42%-IL-6 | [144] |
| 18β-GL | LPS (1 μg mL−1)-induced murine Cell (RAW 264.7 macrophages) | 0.5 or 1 mg mL−1 | ELISA | Supress PGE2, PGI2,TXB2 & LTB4 | [155] |
| 18β-GL | Leishmania donovani infected Macrophages-BALB/c mice (age: 4–6 weeks) | 50 mg mL−1 | ELISA | 90.94%-parasite load | [156] |
| 18β-GA | LPS (1 μg mL−1)-induced murine cell (RAW 264.7 cell) | 75 μM | ELISA | 34%-NO | [144] |
| 18β-GA | 75 μM | ELISA | 58%-PEG2, 42%-1L-1β, 35%-IL-6, 34%-TNF-ἁ | [144] | |
| LID | LPS (0.1 μg mL−1)-induced U937 Cell line (human monoblastic leukaemia cell line) | 0.1, 0.5, 1 μg mL−1 | Decreased the secretion of IL-6, MMP-7, MMP-8, & MMP-9 | [157] | |
| DGC | Glutamte (5 nM)-induced HT22 cells | 2 μM | 2,7-DCF assay | Dose-dependent inhibition of ROS assay & WB production | [158] |
| LIA | LPS (0.1 μg mL−1)-induced U937 cell line (human monoblastic leukaemia cell line) | 0.1, 0.5, 1 μg mL−1 | Decreased the secretions of CCL5 @ (1 μg mL−1), MMP-7 @ (0.5, 1 μg mL−1) MMP-8 @ (0.5, 0.1, 1 μg mL−1) | [157] | |
| LCA | LPS (μg mL−1) induced murine cells (RAW 264.7) | 3 &10 μM | DCFH-DA | >80% PGE2 inhibition @ 10 μM fluorometric >50% NO inhibition at | [151] |
| 18β-GL | LPS (μg mL−1) induced | 75 μm | ELISA | 51% reduction in NO | [144] |
| Murine cells (RAW 264.7 cells) | 51% reduction in IL-1β 49% reduction in PGE2 46% reduction in TNF-ἁ 42% reduction in IL-6 | ||||
| 18β-GA | 75 μm | ELISA | 58% reduction in PEG2 | [159] | |
| Glabridin & isoliquiritigenin | 20-40 μg mL−1 | Cell culture & cell viability assay | anti-inflammatory activity is due to the individual or synergistic effects | ||
| In vivo studies | |||||
| Compounds | Inflammation Details | Models | Treatment | Result(s) | Reference |
| 18ἁ-GL | 20% paraquat poisoning solution @ 15 mg kg−1 | Sprague Dawley rats-male 30 Ns (180–200 g) | injection-IP 30 mg kg−1 | Significant decrease in intercellular adhesion molecules (ICAM-1) and matrix metalloproteinase-9 (MMP-9) | [147] |
| 18β-GL | LPS (1 mg kg−1)-Intratracheal installation | BALB/C mice (male 20–25 gm) | injection-IP 10, 25 & 50 mg kg−1 | Noted decrease in NO and MPO activity | [160] |
| LCA | Topical inflammation induced instantly at the posterior surface of the ear (using xylene 0.05 mL) | Kunming mice (20–25 gm) & Wistar rats (150–200 gm) | 50 mg kg−1 | Decrease in ear oedema rate by 30.3% | [161] |
| Human Studies | |||||
| GL | Hepatitis B virus induced inflammation | Humans | oral and IV (60 mL daily) for a week) | Effective in normalizing serum (for 7 days, later 3 days transaminases) | [115] |
| GL | Hepatitis C virus induced inflammation | Humans | 40 mL transaminases | Found effective in normalizing serum | [116] |
| GL | Hepatitis virus induced Inflammation | Humans | 40 mg of GL (IV) | Suppressed ALT | [80] |
4.2.3. Human Studies
4.2.4. Mechanism
4.3. Effect on Autophagy
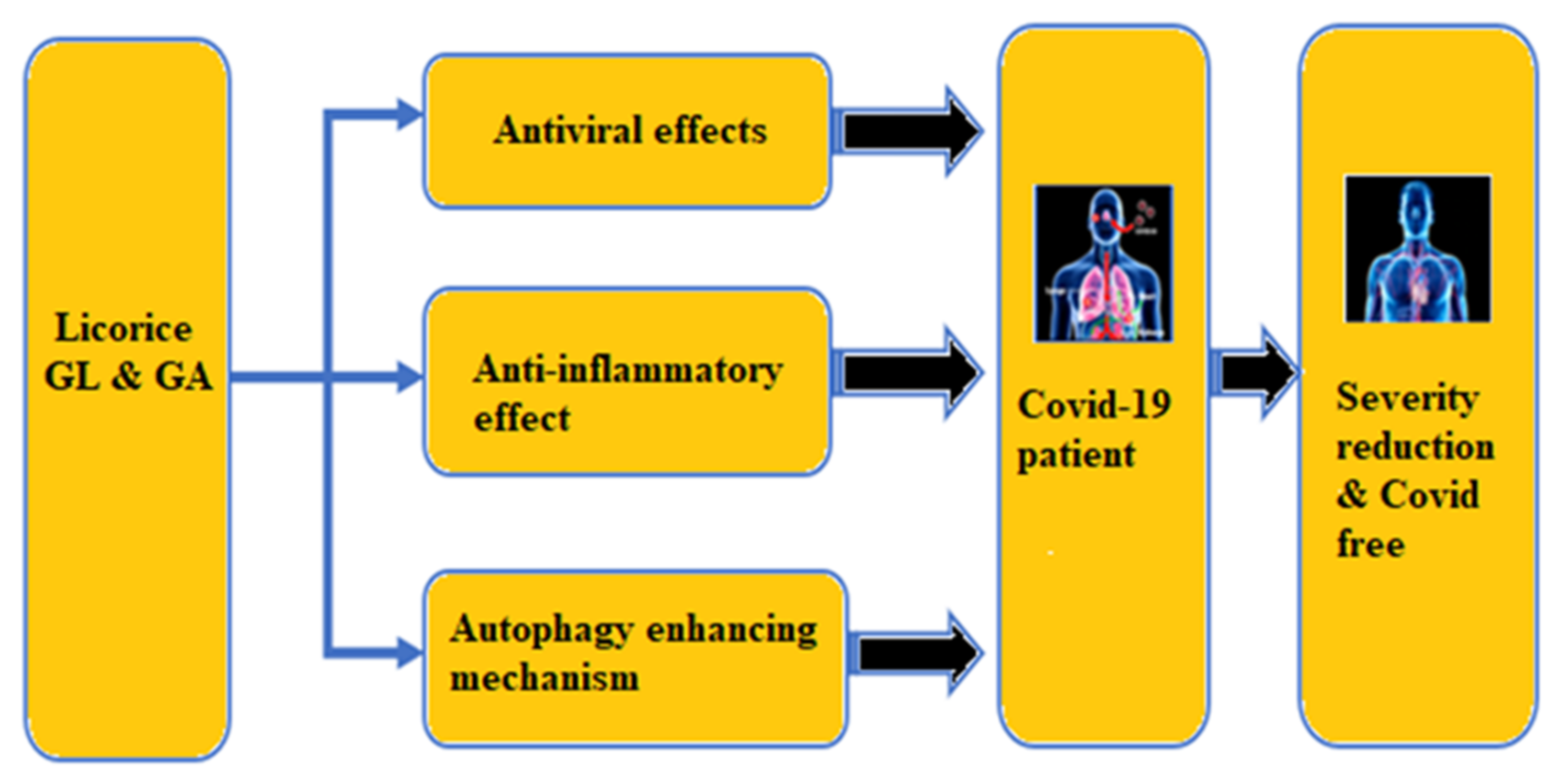
5. Discussion
- (i)
- If any system can block or inhibit the viral replication in the lung epithelial cells, it may be possible to prevent or reduce the body’s inflammatory response. Through several studies, it is found that the two triterpenes GL and GA, which are present in licorice, have significant antiviral characteristics against several viruses such as Hepatitis A, B, and C, HIV, Coxasackievirus, Influenza virus (H1N1 and H5N1), Duck Hepatitis virus, and SARS-CoV-1. In this respect, it is thus hypothesized that the GL and GA in licorice might prevent the spread of SARS-CoV-2, thereby significantly inhibiting any dangerous inflammatory response.
- (ii)
- Chen and Du [197] conducted a molecular docking test recently and highlighted the potential binding of GL to the ACE-2 molecule. We already knew that SARS-CoV-2 enters the cell through ACE-2. Thus, blocking the ACE-2 by binding with GL can reduce the SARS-CoV-2 infectivity and the resulting COVID-19.
- (iii)
- Autophagy is the suicidal mechanism that an infected cell adopts to protect adjacent cells from further infection., When SARS-CoV-2 infects a cell, the virus will forestall the autophagy mechanism by releasing SKP2 protein that can inhibit the cell’s Beclin-1 production. Whereas studies have shown that GL in licorice can increase the production of Beclin-1 by a factor of at least two times [107], which may help stimulate the autophagy mechanism and thus inhibit viral replication.
- (iv)
- In the worst case, if the above two mechanisms cannot stop viral replication, there is the potentiality of inflammation in the body. In such a situation, adaptive immunity can be severely compromised, and this is where the phytochemicals in licorice, specifically GL and GA, can significantly reduce inflammation in the body by inhibiting the pro-inflammatory cytokines. In this way, licorice can possibly mitigate the severity of COVID-19 symptoms.
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Worldometer. Coronavirus Cases. Available online: www.worldometers.info/coronoavirus (accessed on 29 August 2021).
- NDTV. Over 40,000 Vaccinated People Test Positive in Kerala, India: Sources. Media Report. Available online: https://www.ndtv.com/india-news/over-40-000-breakthrough-infections-or-covid-cases-in-vaccinated-people-in-kerala-source-2507884 (accessed on 29 August 2021).
- India Today. Kerala Reports over 40,000 Breakthrough Covid Cases, Raises Concerns of New Variant. Available online: https://www.indiatoday.in/coronavirus-outbreak/story/kerala-reports-40000-breakthrough-covid-cases-new-variant-1839476-2021-08-11 (accessed on 29 August 2021).
- Reuters. Israeli Doctors Find Severe COVID-19 Breakthrough Cases Moslt in Older, Sicker Patients. Available online: https://www.reuters.com/world/middle-east/israeli-doctors-find-severe-covid-19-breakthrough-cases-mostly-older-sicker-2021-08-20/ (accessed on 29 August 2021).
- Precision Vaccinations. Israel’s COVID-19 Vaccine Breakthrough Cases Exceeded 50%. Available online: https://www.precisionvaccinations.com/2021/08/11/israels-covid-19-vaccine-breakthrough-cases-exceed-50 (accessed on 29 August 2021).
- Becker’s Hospital Review. Nearly 60% of Hospitalised COVID-19 Patients in Israel Fully Vaccinated, Data Shows. Available online: https://www.beckershospitalreview.com/public-health/nearly-60-of-hospitalized-covid-19-patients-in-israel-fully-vaccinated-study-finds.html (accessed on 29 August 2021).
- Musser, J.M.; Christensen, P.A.; Olsen, R.J.; Long, S.W.; Subedi, S.; Davis, J.J.; Gollihar, J.D. Delta variants of SARS-CoV-2 cause significantly increased vaccine breakthrough COVID-19 cases in Houston, Texas. medRxiv 2021. [Google Scholar] [CrossRef]
- Birhane, M.; Bressler, S.; Chang, G.; Clark, T.; Dorough, L.; Fischer, M.; Watkins, L.F.; Goldstein, J.M.; Kugeler, K.; COVID-19 Vaccine Breakthrough Case Investigations Team.; et al. COVID-19 Vaccine Breakthrough Infections Reported to CDC—United States, January 1–April 30, 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 792–793. [Google Scholar] [CrossRef]
- Brown, C.M.; Vostok, J.; Johnson, H.; Burns, M.; Gharpure, R.; Sami, S.; Laney, A.S. Outbreak of SARS-CoV-2 infections, including COVID-19 vaccine breakthrough infections, associated with large public gatherings—Barnstable County, Massachusetts, July 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 1059. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.; Kaur, H.; Yadav, P.D.; Mukhopadhyay, L.; Sahay, R.R.; Kumar, A.; Nyayanit, D.A.; Shete, A.M.; Patil, S.; Majumdar, T.; et al. Clinical Characterization and Genomic Analysis of Samples from COVID-19 Breakthrough Infections during the Second Wave among the Various States of India. Viruses 2021, 13, 1782. [Google Scholar] [CrossRef]
- Wang, D.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; Cheng, Z.; Xiong, Y.; et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA 2020, 323, 1061–1069. [Google Scholar] [CrossRef] [PubMed]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Haberman, R.; Axelrad, J.; Chen, A.; Castillo, R.; Yan, D.; Izmirly, P.; Neimann, A.; Adhikari, S.; Hudesman, D.; Scher, J.U. Covid-19 in Immune-Mediated Inflammatory Diseases—Case Series from New York. N. Engl. J. Med. 2020, 383, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Bryant, A.; Lawrie, T.A.; Dowswell, T.; Fordham, E.J.; Scott, M.; Hill, S.R.; Tham, T.C. Ivermectin for prevention and treatment of COVID-19 infection: A systematic review, meta-analysis and trial sequential analysis to inform clinical guidelines. OSF Prepr. 2021. [Google Scholar] [CrossRef]
- Chaccour, C.; Casellas, A.; Matteo, A.B.-D.; Pineda, I.; Fernandez-Montero, A.; Ruiz-Castillo, P.; Richardson, M.-A.; Rodríguez-Mateos, M.; Jordán-Iborra, C.; Brew, J.; et al. The effect of early treatment with ivermectin on viral load, symptoms and humoral response in patients with non-severe COVID-19: A pilot, double-blind, placebo-controlled, randomized clinical trial. EClinicalMedicine 2021, 32, 100720. [Google Scholar] [CrossRef] [PubMed]
- Lucchino, B.; Di Franco, M.; Conti, F. COVID-19: An unexpected indication for anti-rheumatic therapies? Rheumatology 2020, 59, 1200–1203. [Google Scholar] [CrossRef]
- Kacprzak, K.M. Chemistry and Biology of Cinchona Alkaloids. In Natural Products; Springer: Berlin/Heidelberg, Germany, 2013; pp. 605–641. [Google Scholar] [CrossRef]
- Malkinson, F.D. Colchicine. New uses of an old, old drug. Arch. Dermatol. 1982, 118, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Luseba, D.; Elgorashi, E.; Ntloedibe, D.; Van Staden, J. Antibacterial, anti-inflammatory and mutagenic effects of some medicinal plants used in South Africa for the treatment of wounds and retained placenta in livestock. S. Afr. J. Bot. 2007, 73, 378–383. [Google Scholar] [CrossRef]
- Soltan, M.M.; Zaki, A.K. Antiviral screening of forty-two Egyptian medicinal plants. J. Ethnopharmacol. 2009, 126, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Ben-Shabat, S.; Yarmolinsky, L.; Porat, D.; Dahan, A. Antiviral effect of phytochemicals from medicinal plants: Applications and drug delivery strategies. Drug Deliv. Transl. Res. 2020, 10, 354–367. [Google Scholar] [CrossRef]
- Joshi, B.; Sah, G.P.; Basnet, B.B.; Bhatt, M.R.; Sharma, D.; Subedi, K.; Malla, R. Phytochemical extraction and antimicrobial properties of different medicinal plants: Ocimum sanctum (Tulsi), Eugenia caryophyllata (Clove), Achyranthes bidentata (Datiwan) and Azadirachta indica (Neem). J. Microbiol. Antimicrob. 2011, 3, 1–7. [Google Scholar]
- Jaiswal, Y.S.; Williams, L.L. A glimpse of Ayurveda—The forgotten history and principles of Indian traditional medicine. J. Tradit. Complement. Med. 2017, 7, 50–53. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.; Zhang, J.; Gao, X.; Xia, Y.; Ferrelli, R.M.; Fauci, A.; Guerra, R.; Hu, L. Clinical practice of traditional Chinese medicines for chronic heart failure. Hear Asia 2010, 2, 24–27. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.-Y.; Liu, A.-L.; Liu, S.-J.; Xu, X.-W.; Huang, L.-F. Screening for Neuraminidase Inhibitory Activity in Traditional Chinese Medicines Used to Treat Influenza. Molecules 2016, 21, 1138. [Google Scholar] [CrossRef] [PubMed]
- Van Wyk, B.E.; Wink, M. Medicinal Plants of the World; CABI: Pretoria, South Africa, 2018. [Google Scholar]
- Yarnell, E. Herbs for Upper Digestive Overgrowth of Flora. Altern. Complement. Ther. 2018, 24, 173–179. [Google Scholar] [CrossRef]
- Lelešius, R.; Karpovaitė, A.; Mickienė, R.; Drevinskas, T.; Tiso, N.; Ragažinskienė, O.; Kubilienė, L.; Maruška, A.; Šalomskas, A. In vitro antiviral activity of fifteen plant extracts against avian infectious bronchitis virus. BMC Vet. Res. 2019, 15, 178. [Google Scholar] [CrossRef]
- Fiore, C.; Eisenhut, M.; Ragazzi, E.; Zanchin, G.; Armanini, D. A history of the therapeutic use of liquorice in Europe. J. Ethnopharmacol. 2005, 99, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Halberstein, R.A. Medicinal Plants: Historical and Cross-Cultural Usage Patterns. Ann. Epidemiol. 2005, 15, 686–699. [Google Scholar] [CrossRef] [PubMed]
- Pan, S.-Y.; Litscher, G.; Gao, S.-H.; Zhou, S.-F.; Yu, Z.-L.; Chen, H.-Q.; Zhang, S.-F.; Tang, M.-K.; Sun, J.-N.; Ko, K.-M. Historical Perspective of Traditional Indigenous Medical Practices: The Current Renaissance and Conservation of Herbal Resources. Evid.-Based Complement. Altern. Med. 2014, 2014, 525340. [Google Scholar] [CrossRef]
- Mwitari, P.G.; Ayeka, P.A.; Ondicho, J.; Matu, E.N.; Bii, C.C. Antimicrobial Activity and Probable Mechanisms of Action of Medicinal Plants of Kenya: Withania somnifera, Warbugia ugandensis, Prunus africana and Plectrunthus barbatus. PLoS ONE 2013, 8, e65619. [Google Scholar] [CrossRef] [PubMed]
- Noor, A.; Bansal, V.S.; Vijayalakshmi, M.A. Current update on anti-diabetic biomolecules from key traditional Indian medicinal plants. Curr. Sci. 2013, 104, 721–727. [Google Scholar]
- Aggarwal, B.B.; Prasad, S.; Reuter, S.; Kannappan, R.; Yadav, V.R.; Park, B.; Kim, J.H.; Gupta, S.C.; Phromnoi, K.; Sundaram, C.; et al. Identification of Novel Anti-inflammatory Agents from Ayurvedic Medicine for Prevention of Chronic Diseases: “Reverse Pharmacology” and “Bedside to Bench” Approach. Curr. Drug Targets 2011, 12, 1595–1653. [Google Scholar] [CrossRef]
- Smith, J.V.; Burdick, A.J.; Golik, P.; Khan, I.; Wallace, U.; Luo, Y. Anti-apoptotic properties of Ginkgo biloba extract EGb 761 in differentiated PC12 cells. Cell. Mol. Boil. 2002, 48, 699–707. [Google Scholar]
- Li, W.; Mo, W.; Shen, D.; Sun, L.; Wang, J.; Lu, S.; Gitschier, J.M.; Zhou, B. Yeast Model Uncovers Dual Roles of Mitochondria in the Action of Artemisinin. PLoS Genet. 2005, 1, e36. [Google Scholar] [CrossRef]
- Wallace, D.C. Anecdotal, historical and critical commentaries on genetics: Mitochondria as chi. Genetics 2008, 179, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Dobos, G.J.; Rampp, T. The Significance of Ayurvedic Medicinal Plants. J. Evid.-Based Integr. Med. 2017, 22, 494–501. [Google Scholar] [CrossRef]
- Selitrennikoff, C.P. Antifungal Proteins. Appl. Environ. Microbiol. 2001, 67, 2883–2894. [Google Scholar] [CrossRef] [PubMed]
- Asres, K.; Bucar, F. Anti-HIV activity against immunodeficiency virus type 1 (HIV-I) and type II (HIV-II) of compounds isolated from the stem bark of Combretum molle. Ethiop. Med. J. 2005, 43, 15–20. [Google Scholar] [PubMed]
- Ghildiyal, R.; Prakash, V.; Chaudhary, V.; Gupta, V.; Gabrani, R. Phytochemicals as antiviral agents: Recent updates. In Plant-Derived Bioactives; Springer: Berlin/Heidelberg, Germany, 2020; pp. 279–295. [Google Scholar] [CrossRef]
- Ho, T.-Y.; Wu, S.-L.; Chen, J.-C.; Li, C.-C.; Hsiang, C.-Y. Emodin blocks the SARS coronavirus spike protein and angiotensin-converting enzyme 2 interaction. Antivir. Res. 2007, 74, 92–101. [Google Scholar] [CrossRef]
- McCutcheon, A.; Roberts, T.; Gibbons, E.; Ellis, S.; Babiuk, L.; Hancock, R.; Towers, G. Antiviral screening of British Columbian medicinal plants. J. Ethnopharmacol. 1995, 49, 101–110. [Google Scholar] [CrossRef]
- Lou, S.D. Research on Anti-HIV Activities of Chinese Herbal Medicines; Yunnan Science and Technology: Kunming, China, 1998. [Google Scholar]
- Li, S.-Y.; Chen, C.; Zhang, H.-Q.; Guo, H.-Y.; Wang, H.; Wang, L.; Zhang, X.; Hua, S.-N.; Yu, J.; Xiao, P.-G.; et al. Identification of natural compounds with antiviral activities against SARS-associated coronavirus. Antivir. Res. 2005, 67, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Selitrennikoff, C.P. Influenza virus variants with reduced susceptibility to inhibition by a polyphenol extract from Geranium sanguineum L. Die Pharm. Int. J. Pharm. Sci. 2003, 58, 53–57. [Google Scholar] [CrossRef]
- Tolo, F.M.; Rukunga, G.M.; Muli, F.W.; Njagi, E.N.; Njue, W.; Kumon, K.; Mungai, G.M.; Muthaura, C.N.; Muli, J.M.; Keter, L.K.; et al. Anti-viral activity of the extracts of a Kenyan medicinal plant Carissa edulis against herpes simplex virus. J. Ethnopharmacol. 2006, 104, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Lubbe, A.; Seibert, I.; Klimkait, T.; van der Kooy, F. Ethnopharmacology in overdrive: The remarkable anti-HIV activity of Artemisia annua. J. Ethnopharmacol. 2012, 141, 854–859. [Google Scholar] [CrossRef]
- Huang, X.; Ding, L.; Yang, J.; Bian, X.-J.; Zhang, L.-Y.; Zhang, Z.-X.; Liu, G.-Y. Study on the pharmacokinetics of diammonium glycyrrhizinate capsule in healthy volunteers by HPLC-MS. Chin. J. Clin. Pharmacol. 2006, 22, 365. [Google Scholar]
- Debiaggi, M.; Pagani, L.; Cereda, P.M.; Landini, P.; Romero, E. Antiviral activity of Chamaecyparis lawsoniana extract: Study with herpes simplex virus type 2. Microbiologica 1988, 11, 55–61. [Google Scholar]
- Vieira, R.F.; Bizzo, H.R.; Deschamps, C. Genetic resources of aromatic plants from Brazil. Isr. J. Plant Sci. 2010, 58, 263–271. [Google Scholar] [CrossRef]
- Akram, M.; Tahir, I.M.; Shah, S.M.A.; Mahmood, Z.; Altaf, A.; Ahmad, K.; Munir, N.; Daniyal, M.; Nasir, S.; Mehboob, H. Antiviral potential of medicinal plants against HIV, HSV, influenza, hepatitis, and coxsackievirus: A systematic review. Phytother. Res. 2018, 32, 811–822. [Google Scholar] [CrossRef]
- Dhama, K.; Karthik, K.; Khandia, R.; Munjal, A.; Tiwari, R.; Rana, R.; Khurana, S.K.; Ullah, S.; Khan, R.U.; Alagawany, M.; et al. Medicinal and therapeutic potential of herbs and plant metabolites/extracts countering viral pathogens-current knowledge and future prospects. Curr. Drug Metab. 2018, 19, 236–263. [Google Scholar] [CrossRef] [PubMed]
- Wink, M. Potential of DNA Intercalating Alkaloids and Other Plant Secondary Metabolites against SARS-CoV-2 Causing COVID-19. Diversity 2020, 12, 175. [Google Scholar] [CrossRef]
- Dioscorides. De Materia Medica. 1555. Available online: http://bdh.bne.es/bnesearch/detalle/bdh0000037225 (accessed on 25 September 2021).
- Fiore, C.; Eisenhut, M.; Krausse, R.; Ragazzi, E.; Pellati, D.; Armanini, D.; Bielenberg, J. Antiviral effects ofGlycyrrhiza species. Phytother. Res. Int. J. Devoted Pharmacol. Toxicol. Eval. Nat. Prod. Deriv. 2008, 22, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yang, R.; Yuan, B.; Liu, Y.; Liu, C. The antiviral and antimicrobial activities of licorice, a widely-used Chinese herb. Acta Pharm. Sin. B 2015, 5, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Fatima, A.; Faridi, U.; Negi, A.S.; Shanker, K.; Kumar, J.; Rahuja, N.; Luqman, S.; Sisodia, B.S.; Saikia, D.; et al. Antimicrobial potential of Glycyrrhiza glabra roots. J. Ethnopharmacol. 2008, 116, 377–380. [Google Scholar] [CrossRef]
- Aly, A.M.; Al-Alousi, L.; Salem, H.A. Licorice: A possible anti-inflammatory and anti-ulcer drug. AAPS PharmSciTech 2005, 6, E74–E82. [Google Scholar] [CrossRef]
- Hanrahan, C. Gale Encyclopedia of Alternative Medicine, Licorice [CD-ROM]; Thomson Gale: Farmington Hills, MI, USA, 2001. [Google Scholar]
- Lust, J. The Herb Book; Bantam Books: New York, NY, USA, 1994. [Google Scholar]
- Yang, R.; Yuan, B.-C.; Ma, Y.-S.; Zhou, S.; Liu, Y. The anti-inflammatory activity of licorice, a widely used Chinese herb. Pharm. Biol. 2016, 55, 5–18. [Google Scholar] [CrossRef] [PubMed]
- Ayurvedic Pharmacopoeia of India. Vol. I: Part I, Department of Ayush, Ministry of Health and Family Welfare; Government of India: New Delhi, India, 2001.
- Obolentseva, G.V.; Litvinenko, I.V.I.; ammosov, A.S.; Popova, T.P.; Sampiev, A.M. Pharmacological and therapeutic properties of licorice preparations—A Review (Translated from Khimiko-Farmatsevticheskii Zhurnal). Med. Plants 1999, 33, 24–31. Available online: https://fdocuments.in/document/pharmacological-and-therapeutic-properties-of-licorice-preparations-a-review.html (accessed on 17 August 2021).
- RPRM. Resources of Plant Raw Materials in the USSR: Flowering Plants, Their Chemical Compositions, and Use; Nauka: Moscow, Russia, 1987. [Google Scholar]
- Somjen, D.; Tamir, S.; Vaya, J. Estrogen-Like Activity of Licorice Root Extract and Its Constituents. Oxidative Stress Dis. 2004, 14, 615–634. [Google Scholar] [CrossRef]
- Van Rossum, T.G.; Vulto, A.G.; Hop, W.C.; Brouwer, J.T.; Niesters, H.; Schalm, S.W. Intravenous glycyrrhizin for the treatment of chronic hepatitis C: A double-blind, randomized, placebo-controlled phase I/II trial. J. Gastroenterol. Hepatol. 1999, 14, 1093–1099. [Google Scholar] [CrossRef] [PubMed]
- Bahmani, M.; Rafieian-Kopaei, M.; Jeloudari, M.; Eftekhari, Z.; Delfan, B.; Zargaran, A.; Forouzan, S. A review of the health effects and uses of drugs of plant licorice (Glycyrrhiza glabra L.) in Iran. Asian Pac. J. Trop. Dis. 2014, 4, S847–S849. [Google Scholar] [CrossRef]
- Zang, Y. Pharmacological Activities of Coumarin Compounds in Licorice: A Review. Nat. Prod. Commun. 2020, 15. [Google Scholar] [CrossRef]
- Badkhane, Y.; Yadav, A.S.; Bajaj, A.; Sharma, A.K.; Raghuwanshi, D.K. Glycyrrhiza glabra L. a miracle medicinal herb. Indo Am. J. Pharm. Res. 2014, 4, 5808–5816. [Google Scholar]
- Dhingra, D.; Sharma, A. Evaluation of antidepressant-like activity of glycyrrhizin in mice. Indian J. Pharmacol. 2005, 37, 390. [Google Scholar] [CrossRef]
- Fuhrman, B.; Volkova, N.; Kaplan, M.; Presser, D.; Attias, J.; Hayek, T.; Aviram, M. Antiatherosclerotic effects of licorice extract supplementation on hypercholesterolemic patients: Increased resistance of LDL to atherogenic modifications, reduced plasma lipid levels, and decreased systolic blood pressure. Nutrition 2002, 18, 268–273. [Google Scholar] [CrossRef]
- Armanini, D.; De Palo, C.B.; Mattarello, M.J.; Spinella, P.; Zaccaria, M.; Ermolao, A.; Palermo, M.; Fiore, C.; Sartorato, P.; Francini-Pesenti, F.; et al. Effect of licorice on the reduction of body fat mass in healthy subjects. J. Endocrinol. Investig. 2003, 26, 646–650. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.-C.; Lee, Y.-H.; Kim, S.H.; Kim, K.-J.; Kim, K.-M.; Oh, S.; Jung, Y.-S. Hepatoprotective effect of licorice, the root of Glycyrrhiza uralensis Fischer, in alcohol-induced fatty liver disease. BMC Complement. Altern. Med. 2015, 16, 19. [Google Scholar] [CrossRef] [PubMed]
- Thakur, V.; Nargis, S.; Gonzalez, M.; Pradhan, S.; Terreros, D.; Chattopadhyay, M. Role of Glycyrrhizin in the Reduction of Inflammation in Diabetic Kidney Disease. Nephron 2017, 137, 137–147. [Google Scholar] [CrossRef]
- Li, X.; Sun, R.; Liu, R. Natural products in licorice for the therapy of liver diseases: Progress and future opportunities. Pharmacol. Res. 2019, 144, 210–226. [Google Scholar] [CrossRef]
- Armanini, D.; Fiore, C.; Mattarello, M.J.; Bielenberg, J.; Palermo, M. History of the Endocrine Effects of Licorice. Exp. Clin. Endocrinol. Diabetes 2002, 110, 257–261. [Google Scholar] [CrossRef]
- Li, Y.-J.; Chen, J.; Li, Y.; Li, Q.; Zheng, Y.-F.; Fu, Y.; Li, P. Screening and characterization of natural antioxidants in four Glycyrrhiza species by liquid chromatography coupled with electrospray ionization quadrupole time-of-flight tandem mass spectrometry. J. Chromatogr. A 2011, 1218, 8181–8191. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, J.; Zhou, W.; Wang, Y.; Yang, L. Systems approaches and polypharmacology for drug discovery from herbal medicines: An example using licorice. J. Ethnopharmacol. 2013, 146, 773–793. [Google Scholar] [CrossRef]
- Miyake, K.; Tango, T.; Ota, Y.; Mitamura, K.; Yoshiba, M.; Kako, M.; Hayashi, S.; Ikeda, Y.; Hayashida, N.; Iwabuchi, S.; et al. Efficacy of Stronger Neo-Minophagen C compared between two doses administered three times a week on patients with chronic viral hepatitis. J. Gastroenterol. Hepatol. 2002, 17, 1198–1204. [Google Scholar] [CrossRef] [PubMed]
- Takahara, T.; Watanabe, A.; Shiraki, K. Effects of glycyrrhizin on hepatitis B surface antigen: A biochemical and morphological study. J. Hepatol. 1994, 21, 601–609. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, B.; Coordinating Group of SNMC Trial’s Society of Hepatology in China. Randomized clinical trial with two doses (100 and 40 ml) of Stronger Neo-Minophagen C in Chinese patients with chronic hepatitis B. Hepatol. Res. 2002, 24, 220–227. [Google Scholar] [CrossRef]
- Celik, M.M.; Duran, N. An experimental in-vitro study to evaluate the anti-helicobacter activity of Glycyrrhetinic acid. Rev. Romana Med. Lab. 2019, 27, 63–71. [Google Scholar] [CrossRef]
- Long, D.R.; Mead, J.; Hendricks, J.M.; Hardy, M.E.; Voyich, J.M. 18bGlycyrrhetinic acid inhibits methicillin-resistant Staphylococcus aureus survival and attenuates virulence gene expression. Antimicrob. Agents Chemother. 2013, 57, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Krausse, R.; Bielenberg, J.; Blaschek, W.; Ullmann, U. In vitro anti-Helicobacter pylori activity of Extractum liquiritiae, glycyrrhizin and its metabolites. J. Antimicrob. Chemother. 2004, 54, 243–246. [Google Scholar] [CrossRef]
- Kolbe, L.; Immeyer, J.; Batzer, J.; Wensorra, U.; Dieck, K.T.; Mundt, C.; Wolber, R.; Stäb, F.; Schönrock, U.; Ceilley, R.I.; et al. Anti-inflammatory efficacy of Licochalcone A: Correlation of clinical potency and in vitro effects. Arch. Dermatol. Res. 2006, 298, 23–30. [Google Scholar] [CrossRef]
- Kwon, H.-J.; Kim, H.-H.; Ryu, Y.B.; Kim, J.H.; Jeong, H.J.; Lee, S.-W.; Chang, J.S.; Cho, K.-O.; Rho, M.-C.; Park, S.-J.; et al. In vitro anti-rotavirus activity of polyphenol compounds isolated from the roots of Glycyrrhizauralensis. Bioorg. Med. Chem. 2010, 18, 7668–7674. [Google Scholar] [CrossRef]
- Al-Turki, A.I.; El-Ziney, M.G.; Abdel-Salam, A.M. Chemical and antibacterial characterization of aqueous extracts of oregano, marjoram, sage and licorice and their application in milk and labneh. J. Food Agric. Environ. 2008, 6, 39–44. [Google Scholar]
- Park, I.-K.; Kim, J.; Lee, Y.-S.; Shin, S.-C. In vivofungicidal activity of medicinal plant extracts against six phytopathogenic fungi. Int. J. Pest Manag. 2008, 54, 63–68. [Google Scholar] [CrossRef]
- Messier, C.; Grenier, D. Effect of licorice compounds licochalcone A, glabridin and glycyrrhizic acid on growth and virulence properties of Candida albicans. Mycoses 2011, 54, e801–e806. [Google Scholar] [CrossRef]
- Maas, H.; Cock, I.E. The Interactive Antimicrobial Activity of Glycyrrhiza glabra L. Root Extracts and Conventional Antibiotics Against some Bacterial Triggers of Autoimmune Inflammatory Diseases. Pharmacogn. Commun. 2018, 8, 66–74. [Google Scholar] [CrossRef]
- Wu, S.-C.; Yang, Z.-Q.; Liu, F.; Peng, W.-J.; Qu, S.-Q.; Li, Q.; Song, X.-B.; Zhu, K.; Shen, J.-Z. Antibacterial Effect and Mode of Action of Flavonoids from Licorice against Methicillin-Resistant Staphylococcus aureus. Front. Microbiol. 2019, 10, 2489. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.G. Inhibitory Actions of Glycyrrhizic Acid on Arylamine N-Acetyltransferase Activity in Strains of Helicobacter Pylori from Peptic Ulcer Patients. Drug Chem. Toxicol. 1998, 21, 355–370. [Google Scholar] [CrossRef] [PubMed]
- Pastorino, G.; Cornara, L.; Soares, S.; Rodrigues, F.; Oliveira, M.B.P.P. Liquorice (Glycyrrhiza glabra): A phytochemical and pharmacological review. Phytother. Res. 2018, 32, 2323–2339. [Google Scholar] [CrossRef] [PubMed]
- Fukai, T.; Marumo, A.; Kaitou, K.; Kanda, T.; Terada, S.; Nomura, T. Antimicrobial activity of licorice flavonoids against methicillin-resistant Staphylococcus aureus. Fitoter 2002, 73, 536–539. [Google Scholar] [CrossRef]
- Demizu, S.; Kajiyama, K.; Takahashi, K.; Hiraga, Y.; Yamamoto, S.; Tamura, Y.; Okada, K.; Kinoshita, T. Antioxidant and antimicrobial constituents of licorice: Isolation and structure elucidation of a new benzofuran derivative. Chem. Pharm. Bull. 1988, 36, 3474–3479. [Google Scholar] [CrossRef] [PubMed]
- Haraguchi, H.; Tanimoto, K.; Tamura, Y.; Mizutani, K.; Kinoshita, T. Mode of antibacterial action of retrochalcones from Glycyrrhiza inflata. Phytochemistry 1998, 48, 125–129. [Google Scholar] [CrossRef]
- Zhou, T. Antimicrobial Activity of Licochalcone E against Staphylococcus aureus and Its Impact on the Production of Staphylococcal Alpha-Toxin. J. Microbiol. Biotechnol. 2012, 22, 800–805. [Google Scholar] [CrossRef] [PubMed]
- Fukai, T.; Marumo, A.; Kaitou, K.; Kanda, T.; Terada, S.; Nomura, T. Anti-Helicobacter pylori flavonoids from licorice extract. Life Sci. 2002, 71, 1449–1463. [Google Scholar] [CrossRef]
- Pompei, R.; Flore, O.; Marccialis, M.A.; Pani, A.; Loddo, B. Glycyrrhizic acid inhibits virus growth and inactivates virus particles. Nat. Cell Biol. 1979, 281, 689–690. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, Y.; Matsuura, T.; Aoyagi, H.; Matsuda, M.; Hmwe, S.S.; Date, T.; Watanabe, N.; Watashi, K.; Suzuki, R.; Ichinose, S.; et al. Antiviral Activity of Glycyrrhizin against Hepatitis C Virus In Vitro. PLoS ONE 2013, 8, e68992. [Google Scholar] [CrossRef]
- Ashfaq, U.A.; Masoud, M.S.; Nawaz, Z.; Riazuddin, S. Glycyrrhizin as antiviral agent against Hepatitis C Virus. J. Transl. Med. 2011, 9, 112. [Google Scholar] [CrossRef]
- Huang, W.; Chen, X.; Li, Q.; Li, P.; Zhao, G.; Xu, M.; Xie, P. Inhibition of Intercellular Adhesion in Herpex Simplex Virus Infection by Glycyrrhizin. Cell Biophys. 2011, 62, 137–140. [Google Scholar] [CrossRef] [PubMed]
- Michaelis, M.; Geiler, J.; Naczk, P.; Sithisarn, P.; Ogbomo, H.; Altenbrandt, B.; Leutz, A.; Doerr, H.W.; Cinatl, J., Jr. Glycyrrhizin inhibits highly pathogenic H5N1 influenza A virus-induced pro-inflammatory cytokine and chemokine expression in human macrophages. Med. Microbiol. Immunol. 2010, 199, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Moisy, D.; Avilov, S.V.; Jacob, Y.; Laoide, B.M.; Ge, X.; Baudin, F.; Naffakh, N.; Jestin, J.-L. HMGB1 Protein Binds to Influenza Virus Nucleoprotein and Promotes Viral Replication. J. Virol. 2012, 86, 9122–9133. [Google Scholar] [CrossRef]
- Smirnov, V.S.; Zarubaev, V.V.; Anfimov, P.M.; Shtro, A.A. Effect of a combination of glutamyl-tryptophan and glycyrrhizic acid on the course of acute infection caused by influenza (H3H2) virus in mice. Vopr. Virusol. 2012, 57, 23–27. [Google Scholar] [PubMed]
- Laconi, S.; Madeddu, M.A.; Pompei, R. Autophagy Activation and Antiviral Activity by a Licorice Triterpene. Phytother. Res. 2014, 28, 1890–1892. [Google Scholar] [CrossRef]
- Soufy, H.; Yassein, S.; Ahmed, A.R.; Khodier, M.H.; Kutkat, M.; Nasr, S.M.; Okda, F. Antiviral and immune stimulant activities of glycyrrhizin against duck hepatitis virus. Afr. J. Tradit. Complement. Altern. Med. 2012, 9, 389–395. [Google Scholar] [CrossRef]
- Fukuchi, K.; Okudaira, N.; Adachi, K.; Odai-Ide, R.; Watanabe, S.; Ohno, H.; Yamamoto, M.; Kanamoto, T.; Terakubo, S.; Nakashima, H.; et al. Antiviral and Antitumor Activity of Licorice Root Extracts. In Vivo 2016, 30, 777–786. [Google Scholar] [CrossRef] [PubMed]
- Hoever, G.; Baltina, L.; Michaelis, M.; Kondratenko, R.; Baltina, L.; Tolstikov, G.A.; Doerr, H.W.; Cinatl, J. Antiviral Activity of Glycyrrhizic Acid Derivatives against SARS−Coronavirus. J. Med. Chem. 2005, 48, 1256–1259. [Google Scholar] [CrossRef] [PubMed]
- Cinatl, J.; Morgenstern, B.; Bauer, G.; Chandra, P.; Rabenau, H.; Doerr, H. Glycyrrhizin, an active component of liquorice roots, and replication of SARS-associated coronavirus. Lancet 2003, 361, 2045–2046. [Google Scholar] [CrossRef]
- Murray, M.T. Glycyrrhiza glabra (Licorice). In Textbook of Natural Medicine; Elsevier: Amsterdam, The Netherlands, 2020; pp. 641–647.e3. [Google Scholar]
- Crance, J.M.; Biziagos, E.; Passagot, J.; van Cuyck-Gandré, H.; Deloince, R. Inhibition of hepatitis A virus replication in vitro by antiviral compounds. J. Med. Virol. 1990, 31, 155–160. [Google Scholar] [CrossRef]
- Sato, H.; Goto, W.; Yamamura, J.-I.; Kurokawa, M.; Kageyama, S.; Takahara, T.; Watanabe, A.; Shiraki, K. Therapeutic basis of glycyrrhizin on chronic hepatitis B. Antivir. Res. 1996, 30, 171–177. [Google Scholar] [CrossRef]
- Tandon, A. Treatment of subacute hepatitis with Lamivudine and intravenous Glycyrrhizin: A pilot study. Hepatol. Res. 2001, 20, 1–8. [Google Scholar] [CrossRef]
- Orlent, H.; Hansen, B.; Willems, M.; Brouwer, J.T.; Huber, R.; Kullak-Ublick, G.A.; Gerken, G.; Zeuzem, S.; Nevens, F.; Tielemans, W.C.; et al. Biochemical and histological effects of 26 weeks of glycyrrhizin treatment in chronic hepatitis C: A randomized phase II trial. J. Hepatol. 2006, 45, 539–546. [Google Scholar] [CrossRef]
- Ikeda, K. Glycyrrhizin injection therapy prevents hepatocellular carcinogenesis in patients with interferon-resistant active chronic hepatitis C. Hepatol. Res. 2007, 37, S287–S293. [Google Scholar] [CrossRef] [PubMed]
- De Clercq, E. Current lead natural products for the chemotherapy of human immunodeficiency virus (HIV) infection. Med. Res. Rev. 2000, 20, 323–349. [Google Scholar] [CrossRef]
- Sasaki, H.; Takei, M.; Kobayashi, M.; Pollard, R.B.; Suzuki, F. Effect of Glycyrrhizin, an Active Component of Licorice Roots, on HIV Replication in Cultures of Peripheral Blood Mononuclear Cells from HIV-Seropositive Patients. Pathobiology 2002, 70, 229–236. [Google Scholar] [CrossRef]
- Harada, S. The broad anti-viral agent glycyrrhizin directly modulates the fluidity of plasma membrane and HIV-1 envelope. Biochem. J. 2005, 392, 191–199. [Google Scholar] [CrossRef]
- Baba, M.; Shigeta, S. Antiviral activity of glycyrrhizin against varicella-zoster virus in vitro. Antivir. Res. 1987, 7, 99–107. [Google Scholar] [CrossRef]
- Lin, J.-C. Mechanism of action of glycyrrhizic acid in inhibition of Epstein-Barr virus replication in vitro. Antivir. Res. 2003, 59, 41–47. [Google Scholar] [CrossRef]
- Numazaki, K.; Nagata, N.; Sato, T.; Chiba, S. Effect of glycyrrhizin, cyclosporin A, and tumor necrosis factor α on infection of U-937 and MRC-5 cells by human cytomegalovirus. J. Leukoc. Biol. 1994, 55, 24–28. [Google Scholar] [CrossRef]
- Zhang, H.; Song, Y.; Zhang, Z. Glycyrrhizin Administration Ameliorates Coxsackievirus B3-Induced Myocarditis in Mice. Am. J. Med. Sci. 2012, 344, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Lampis, G.; Deidda, D.; Pinza, M.; Pompei, R. Enhancement of Anti-Herpetic Activity of Glycyrrhizic Acid by Physiological Proteins. Antivir. Chem. Chemother. 2001, 12, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Hardy, R.S.; Raza, K.; Cooper, M.S. Endogenous glucocorticoids in inflammation: Contributions of systemic and local responses. Swiss Med. Wkl. 2012, 142, w13650. [Google Scholar] [CrossRef]
- Utsunomiya, T.; Kobayashi, M.; Pollard, R.B.; Suzuki, F. Glycyrrhizin, an active component of licorice roots, reduces morbidity and mortality of mice infected with lethal doses of influenza virus. Antimicrob. Agents Chemother. 1997, 41, 551–556. [Google Scholar] [CrossRef] [PubMed]
- Sekizawa, T.; Yanagi, K.; Itoyama, Y. Glycyrrhizin increases survival of mice with herpes simplex encephalitis. Acta Virol. 2001, 45, 51–54. [Google Scholar] [PubMed]
- Lu, H.; Huo, N.; Wang, G.; Li, H.; Nie, L.; Xu, X. Clinical observation of therapeutic effect of compound glycyrrhizin on SARS. China Pharm. 2003, 10, 34–36. [Google Scholar]
- Wu, H.B.; Li, H.M.; Lu, C.A.; Gao, Y.J.; Li, X.Q.; Zhou, A.X. Experimental study on Shuanghuanglian dispersible tablets against viruses. Chin. J. Exp. Tradit. Med. Formul. 2004, 10, 48–50. [Google Scholar]
- Yang, H.; Ko, H.-J.; Yang, J.-Y.; Kim, J.-J.; Seo, S.-U.; Park, S.G.; Choi, S.S.; Seong, J.K.; Kweon, M.-N. Interleukin-1 Promotes Coagulation, Which Is Necessary for Protective Immunity in the Lung against Streptococcus pneumoniae Infection. J. Infect. Dis. 2012, 207, 50–60. [Google Scholar] [CrossRef]
- Krishnaswamy, K. Traditional Indian spices and their health significance. Asia Pac. J. Clin. Nutr. 2008, 17, 265–268. [Google Scholar] [PubMed]
- Tapsell, L.C.; Hemphill, I.; Cobiac, L.; Sullivan, D.R.; Fenech, M.; Patch, C.S.; Roodenrys, S.; Keogh, J.; Clifton, P.; Williams, P.; et al. Health benefits of herbs and spices: The past, the present, the future. Med. J. Aust. 2006, 185 (Suppl. 5), S1–S24. [Google Scholar] [CrossRef] [PubMed]
- Mueller, M.; Hobiger, S.; Jungbauer, A. Anti-inflammatory activity of extracts from fruits, herbs and spices. Food Chem. 2010, 122, 987–996. [Google Scholar] [CrossRef]
- Hanada, T.; Yoshimura, A. Regulation of cytokine signaling and inflammation. Cytokine Growth Factor Rev. 2002, 13, 413–421. [Google Scholar] [CrossRef]
- Guo, J.; Shang, E.; Zhao, J.; Fan, X.; Duan, J.; Qian, D.; Tao, W.; Tang, Y. Data mining and frequency analysis for licorice as a “on Chinese Formulae Database” herb in Chinese Formulae based on Chinese Formulae. Phytomedicine 2014, 21, 1281–1286. [Google Scholar] [CrossRef]
- Murray, M.T. The Healing Power of Herbs, 2nd ed.; Three Rivers Press: New York, NY, USA, 1995. [Google Scholar]
- Szapary, P.O. Tyler’s Herbs of Choice: The Therapeutic Use of Phytomedicinals; Robberts, J.E., Tyler, V.E., Eds.; Haworth Herbal: Binghamton, NY, USA, 1999; ISBN 0789001608. [Google Scholar] [CrossRef]
- Wu, T.-Y.; Khor, T.O.; Saw, C.; Loh, S.C.; Chen, A.I.; Lim, S.S.; Park, J.H.Y.; Cai, L.; Kong, A.-N.T. Anti-inflammatory/Anti-oxidative Stress Activities and Differential Regulation of Nrf2-Mediated Genes by Non-Polar Fractions of Tea Chrysanthemum zawadskii and Licorice Glycyrrhiza uralensis. AAPS J. 2010, 13, 1–13. [Google Scholar] [CrossRef]
- Chandrasekaran, C.V.; Deepak, H.B.; Thiyagarajan, P.; Kathiresan, S.; Sangli, G.K.; Deepak, M.; Agarwal, A. Dual inhibitory effect of Glycyrrhiza glabra (GutGardTM) on COX and LOX products. Phytomedicine 2011, 18, 278–284. [Google Scholar] [CrossRef]
- Kao, T.-C.; Wu, C.-H.; Yen, G.-C. Bioactivity and Potential Health Benefits of Licorice. J. Agric. Food Chem. 2014, 62, 542–553. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.-Y.; Ha, J.Y.; Kim, K.-M.; Jung, Y.-S.; Jung, J.-C.; Oh, S. Anti-Inflammatory Activities of Licorice Extract and Its Active Compounds, Glycyrrhizic Acid, Liquiritin and Liquiritigenin, in BV2 Cells and Mice Liver. Molecules 2015, 20, 13041–13054. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Li, X.; Johnson, T.; Seidel, C.; Wallace, D.P.; Li, R. Polycystin-dependent fluid flow sensing targets histone deacetylase 5 to prevent the development of renal cysts. Development 2010, 137, 1075–1084. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-Y.; Kao, T.-C.; Lo, W.-H.; Yen, G.-C. Glycyrrhizic Acid and 18β-Glycyrrhetinic Acid Modulate Lipopolysaccharide-Induced Inflammatory Response by Suppression of NF-κB through PI3K p110δ and p110γ Inhibitions. J. Agric. Food Chem. 2011, 59, 7726–7733. [Google Scholar] [CrossRef] [PubMed]
- Bai, M.; Yao, G.-D.; Ren, Q.; Li, Q.; Liu, Q.-B.; Zhang, Y.; Wang, X.-B.; Huang, X.-X.; Song, S.-J. Triterpenoid saponins and flavonoids from licorice residues with anti-inflammatory activity. Ind. Crops Prod. 2018, 125, 50–58. [Google Scholar] [CrossRef]
- Frattaruolo, L.; Carullo, G.; Brindisi, M.; Mazzotta, S.; Bellissimo, L.; Rago, V.; Curcio, R.; Dolce, V.; Aiello, F.; Cappello, A.R. Antioxidant and Anti-Inflammatory Activities of Flavanones from Glycyrrhiza glabra L. (licorice) Leaf Phytocomplexes: Identification of Licoflavanone as a Modulator of NF-kB/MAPK Pathway. Antioxidants 2019, 8, 186. [Google Scholar] [CrossRef]
- Xiao, Y.; Xu, J.; Mao, C.; Jin, M.; Wu, Q.; Zou, J.; Gu, Q.; Zhang, Y.; Zhang, Y. 18β-Glycyrrhetinic Acid Ameliorates Acute Propionibacterium acnes-induced Liver Injury through Inhibition of Macrophage Inflammatory Protein-1α. J. Biol. Chem. 2010, 285, 1128–1137. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-R.; Hao, H.-G.; Chu, L. Glycyrrhizin inhibits LPS-induced inflammatory mediator production in endometrial epithelial cells. Microb. Pathog. 2017, 109, 110–113. [Google Scholar] [CrossRef] [PubMed]
- Ishida, T.; Miki, I.; Tanahashi, T.; Yagi, S.; Kondo, Y.; Inoue, J.; Kawauchi, S.; Nishiumi, S.; Yoshida, M.; Maeda, H.; et al. Effect of 18β-glycyrrhetinic acid and hydroxypropyl γcyclodextrin complex on indomethacin-induced small intestinal injury in mice. Eur. J. Pharmacol. 2013, 714, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.W.; Ki, S.H.; Lee, J.R.; Lee, S.J.; Kim, C.W.; Kim, S.C.; Kim, S.G. Liquiritigenin, an aglycone of liquiritin in Glycyrrhizae radix, prevents acute liver injuries in rats induced by acetaminophen with or without buthionine sulfoximine. Chem. Biol. Interact. 2006, 161, 125–138. [Google Scholar] [CrossRef]
- Fu, Y.; Chen, J.; Li, Y.-J.; Zheng, Y.-F.; Li, P. Antioxidant and anti-inflammatory activities of six flavonoids separated from licorice. Food Chem. 2013, 141, 1063–1071. [Google Scholar] [CrossRef] [PubMed]
- Yao, L.; Sun, T. Glycyrrhizin administration ameliorates Streptococcus aureus-induced acute lung injury. Int. Immunopharmacol. 2019, 70, 504–511. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Ohtaki, Y.; Kai, K.; Sasano, T.; Shimauchi, H.; Yokochi, T.; Takada, H.; Sugawara, S.; Kumagai, K.; Endo, Y. Critical roles of platelets in lipopolysaccharide-induced lethality: Effects of glycyrrhizin and possible strategy for acute respiratory distress syndrome. Int. Immunopharmacol. 2005, 5, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Menegazzi, M.; di Paola, R.; Mazzon, E.; Genovese, T.; Crisafulli, C.; Dalbosco, M.; Zou, Z.; Suzuki, H.; Cuzzocrea, S. Glycyrrhizin attenuates the development of carrageenan-induced lung injury in mice. Pharmacol. Res. 2008, 58, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.; Li, X.; Wu, J.; Liang, Z.; Deng, F.; Xie, W.; Zhu, M.; Zhu, J.; Zhu, W.; Geng, S.; et al. Anti-inflammatory activity of magnesium isoglycyrrhizinate through inhibition of phospholipase A2/Arachidonic acid pathway. Inflammation 2015, 38, 1639–1648. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, S.; Bhattacharjee, A.; Majumder, S.; Majumdar, S.B. Glycyrrhizic acid suppresses Cox-2-mediated anti-inflammatory responses during Leishmania donovani infection. J. Antimicrob. Chemother. 2012, 67, 1905–1914. [Google Scholar] [CrossRef] [PubMed]
- La, V.D.; Tanabe, S.-I.; Bergeron, C.; Gafner, S.; Grenier, D. Modulation of Matrix Metalloproteinase and Cytokine Production by Licorice Isolates Licoricidin and Licorisoflavan A: Potential Therapeutic Approach for Periodontitis. J. Periodontol. 2011, 82, 122–128. [Google Scholar] [CrossRef]
- Kim, H.J.; Seo, J.-Y.; Suh, H.-J.; Lim, S.S.; Kim, J.-S. Antioxidant activities of licorice-derived prenylflavonoids. Nutr. Res. Pract. 2012, 6, 491–498. [Google Scholar] [CrossRef]
- Thiyagarajan, P.; Chandrasekaran, C.V.; Deepak, H.B.; Agarwal, A. Modulation of lipopolysaccharide-induced pro-inflammatory mediators by an extract of Glycyrrhiza glabra and its phytoconstituents. Inflammopharmacology 2011, 19, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.-F.; Kuai, J.-K.; Lu, Z.-F.; Yang, G.-D.; Fu, H.-Y.; Wang, J.; Tian, F.; Yan, X.-L.; Zhao, Y.-C.; Wang, Y.-J.; et al. Glycyrrhizin Treatment Is Associated with Attenuation of Lipopolysaccharide-Induced Acute Lung Injury by Inhibiting Cyclooxygenase-2 and Inducible Nitric Oxide Synthase Expression. J. Surg. Res. 2011, 165, e29–e35. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Ao, M.; Hu, J.; Yu, L. Anti-Inflammatory Activity of Licochalcone A Isolated from Glycyrrhiza inflata. Zeitschrift für Naturforschung C 2008, 63, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Acharya, S.K.; Dasarathy, S.; Tandon, A.; Joshi, Y.K.; Tandon, B.N. A preliminary open trial on interferon stimulator (SNMC) derived from Glycyrrhiza glabra in the treatment of subacute hepatic failure. Indian J. Med. Res. 1993, 98, 69–74. [Google Scholar] [CrossRef]
- Seo, E.-H.; Song, G.-Y.; Kwak, B.O.; Oh, C.-S.; Lee, S.H.; Kim, S.-H. Effects of Glycyrrhizin on the Differentiation of Myeloid Cells of the Heart and Lungs in Lipopolysaccharide-Induced Septic Mice. Shock 2017, 48, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Edwards, C.R.; Benediktsson, R.; Lindsay, R.; Seckl, J.R. 11β-Hydroxysteroid dehydrogenases: Key enzymes in determining tissue-specific glucocorticoid effects. Steroids 1996, 61, 263–269. [Google Scholar] [CrossRef]
- Kao, T.-C.; Shyu, M.-H.; Yen, G.-C. Glycyrrhizic Acid and 18β-Glycyrrhetinic Acid Inhibit Inflammation via PI3K/Akt/GSK3β Signaling and Glucocorticoid Receptor Activation. J. Agric. Food Chem. 2010, 58, 8623–8629. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Lian, Q.-Q.; Dong, Q.; Ge, R.-S. Environmental inhibitors of 11β-hydroxysteroid dehydrogenase type 2. Toxicology 2011, 285, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Kao, T.-C.; Wu, C.-H.; Yen, G.-C. Glycyrrhizic acid and 18β-glycyrrhetinic acid recover glucocorticoid resistance via PI3K-induced AP1, CRE and NFAT activation. Phytomedicine 2013, 20, 295–302. [Google Scholar] [CrossRef]
- Mollica, L.; De Marchis, F.; Spitaleri, A.; Dallacosta, C.; Pennacchini, D.; Zamai, M.; Agresti, A.; Trisciuoglio, L.; Musco, G.; Bianchi, M.E. Glycyrrhizin Binds to High-Mobility Group Box 1 Protein and Inhibits Its Cytokine Activities. Chem. Biol. 2007, 14, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Bodet, C.; La, V.D.; Gafner, S.; Bergeron, C.; Grenier, D. A Licorice Extract Reduces Lipopolysaccharide-Induced Proinflammatory Cytokine Secretion by Macrophages and Whole Blood. J. Periodontol. 2008, 79, 1752–1761. [Google Scholar] [CrossRef] [PubMed]
- Michaelis, M.; Geiler, J.; Naczk, P.; Sithisarn, P.; Leutz, A.; Doerr, H.W.; Cinatl, J. Glycyrrhizin Exerts Antioxidative Effects in H5N1 Influenza A Virus-Infected Cells and Inhibits Virus Replication and Pro-Inflammatory Gene Expression. PLoS ONE 2011, 6, e19705. [Google Scholar] [CrossRef]
- Suzuki, M.; Tetsuka, T.; Yoshida, S.; Watanabe, N.; Kobayashi, M.; Matsui, N.; Okamoto, T. The role of p38 mitogen-activated protein kinase in IL-6 and IL-8 production from the TNF-α- or IL-1β-stimulated rheumatoid synovial fibroblasts. FEBS Lett. 2000, 465, 23–27. [Google Scholar] [CrossRef]
- Haworth, C.; Brennan, F.M.; Chantry, D.; Turner, M.; Maini, R.N.; Feldmann, M. Expression of granulocyte-macrophage colony-stimulating factor in rheumatoid arthritis: Regulation by tumor necrosis factor-α. Eur. J. Immunol. 1991, 21, 2575–2579. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.-J.; Lim, H.-S.; Seo, C.-S.; Kim, J.-H.; Jin, S.-E.; Yoo, S.-R.; Shin, H.-K. Traditional herbal formula Jakyakgamcho-tang (Paeonia lactiflora and Glycyrrhiza uralensis) impairs inflammatory chemokine production by inhibiting activation of STAT1 and NF-κB in HaCaT cells. Phytomedicine 2015, 22, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Nagy, G.; Clark, J.M.; Buzas, E.; Gorman, C.; Pasztoi, M.; Koncz, A.; Falus, A.; Cope, A. Nitric oxide production of T lymphocytes is increased in rheumatoid arthritis. Immunol. Lett. 2008, 118, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Gumpricht, E.; Dahl, R.; Devereaux, M.W.; Sokol, R.J. Licorice compounds glycyrrhizin and 18beta-glycyrrhetinic acid are potent modulators of bile acid-induced cytotoxicity in rat hepatocytes. J. Biol. Chem. 2005, 280, 10556–10563. [Google Scholar] [CrossRef]
- Manns, M.P.; Wedemeyer, H.; Singer, A.; Khomutjanskaja, N.; Dienes, H.P.; Roskams, T.; Goldin, R.; Hehnke, U.; Inoue, H.; The European SNMC Study Group. Glycyrrhizin in patients who failed previous interferon alpha-based therapies: Biochemical and histological effects after 52 weeks. J. Viral Hepat. 2012, 19, 537–546. [Google Scholar] [CrossRef]
- Li, J.-Y.; Cao, H.-Y.; Liu, P.; Cheng, G.-H.; Sun, M.-Y. Glycyrrhizic Acid in the Treatment of Liver Diseases: Literature Review. BioMed Res. Int. 2014, 2014, 872139. [Google Scholar] [CrossRef]
- Ye, S.; Zhu, Y.; Ming, Y.; She, X.; Liu, H.; Ye, Q. Glycyrrhizin protects mice against renal ischemia-reperfusion injury through inhibition of apoptosis and inflammation by downregulating p38 mitogen-activated protein kinase signaling. Exp. Ther. Med. 2014, 7, 1247–1252. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Xiao, Z.; He, F.; Zou, J.; Wu, S.; Liu, Z. MicroRNAs Regulate the Pathogenesis of CVB3-Induced Viral Myocarditis. Intervirology 2013, 56, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Fujisawa, Y.; Sakamoto, M.; Matsushita, M.; Fujita, T.; Nishioka, K. Glycyrrhizin Inhibits the Lytic Pathway of Complement—Possible Mechanism of Its Anti-Inflammatory Effect on Liver Cells in Viral Hepatitis. Microbiol. Immunol. 2000, 44, 799–804. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.R.; Jeong, C.-K.; Park, K.-K.; Choi, J.; Park, J.H.Y.; Lim, S.S.; Chung, W.-Y. Anti-Inflammatory Effects of Licorice and Roasted Licorice Extracts on TPA-Induced Acute Inflammation and Collagen-Induced Arthritis in Mice. J. Biomed. Biotechnol. 2010, 2010, 709378. [Google Scholar] [CrossRef]
- Mizgerd, J.P.; Spieker, M.R.; Doerschuk, C.M. Early response cytokines and innate immunity: Essential roles for TNF receptor 1 and type I IL-1 receptor during Escherichia coli pneumonia in mice. J. Immunol. 2001, 166, 4042–4048. [Google Scholar] [CrossRef] [PubMed]
- Lauzurica, P.; Martínez-Martínez, S.; Marazuela, M.; del Arco, P.G.; Martínez-A, C.; Sánchez-Madrid, F.; Redondo, J.M. Pyrrolidine dithiocarbamate protects mice from lethal shock induced by LPS or TNF-α. Eur. J. Immunol. 1999, 29, 1890–1900. [Google Scholar] [CrossRef]
- Chu, X.; Ci, X.; Wei, M.; Yang, X.; Cao, Q.; Guan, M.; Li, H.; Deng, Y.; Feng, H.; Deng, X. Licochalcone A Inhibits Lipopolysaccharide-Induced Inflammatory Response in Vitro and in Vivo. J. Agric. Food Chem. 2012, 60, 3947–3954. [Google Scholar] [CrossRef]
- Levine, B.; Deretic, V. Unveiling the roles of autophagy in innate and adaptive immunity. Nat. Rev. Immunol. 2007, 7, 767–777. [Google Scholar] [CrossRef]
- Leib, D.A.; Alexander, D.E.; Cox, D.; Yin, J.; Ferguson, T.A. Interaction of ICP34.5 with Beclin 1 Modulates Herpes Simplex Virus Type 1 Pathogenesis through Control of CD4 + T-Cell Responses. J. Virol. 2009, 83, 12164–12171. [Google Scholar] [CrossRef] [PubMed]
- Gassen, N.C.; Niemeyer, D.; Muth, D.; Corman, V.M.; Martinelli, S.; Gassen, A.; Hafner, K.; Papies, J.; Mösbauer, K.; Zellner, A.; et al. SKP2 attenuates autophagy through Beclin1-ubiquitination and its inhibition reduces MERS-Coronavirus infection. Nat. Commun. 2019, 10, 5770. [Google Scholar] [CrossRef] [PubMed]
- Shojaei, S.; Suresh, M.; Klionsky, D.J.; Labouta, H.I.; Ghavami, S. Autophagy and SARS-CoV-2 infection: A possible smart targeting of the autophagy pathway. Virulence 2020, 11, 805–810. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.-H.; Li, T.; Tong, Y.-G.; Chen, X.-J.; Chen, X.-P.; Wang, Y.-T.; Lu, J. A Systematic Review of the Anticancer Properties of Compounds Isolated from Licorice (Gancao). Planta Medica 2015, 81, 1670–1687. [Google Scholar] [CrossRef] [PubMed]
- Vlaisavljevic, S.; Šibul, F.; Sinka, I.; Zupko, I.; Ocsovszki, I.; Jovanović-Šanta, S. Chemical composition, antioxidant and anticancer activity of licorice from Fruska Gora locality. Ind. Crops Prod. 2018, 112, 217–224. [Google Scholar] [CrossRef]
- Glavač, N.K.; Kreft, S. Excretion profile of glycyrrhizin metabolite in human urine. Food Chem. 2012, 131, 305–308. [Google Scholar] [CrossRef]
- Egashira, T.; Takayama, F.; Yufu, F.; Shoyama, Y. Pharmacokinetics of glycyrrhizin and glycyrrhetic acid following glycyrrhizin administration to rats with single and multiple doses via different routes. Jpn. Pharmacol. Ther. 2003, 31, 75–83. [Google Scholar]
- Bai, H.; Bao, F.; Fan, X.; Han, S.; Zheng, W.; Sun, L.; Yan, N.; Du, H.; Zhao, H.; Yang, Z. Metabolomics study of different parts of licorice from different geographical origins and their anti-inflammatory activities. J. Sep. Sci. 2020, 43, 1593–1602. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Sakata, Y.; Morimoto, K.; Tambe, Y.; Watanabe, Y.; Honda, G.; Tabata, M.; Oshima, T.; Masuda, T.; Umezawa, T.; et al. Influence of Natural and Synthetic Compounds on Cell Surface Expression of Cell Adhesion Molecules, ICAM-1 and VCAM-1. Planta Medica 2001, 67, 108–113. [Google Scholar] [CrossRef]
- Shi, C.-C.; Chen, T.-R.; Zhang, Q.-H.; Wei, L.-H.; Huang, C.; Zhu, Y.-D.; Liu, H.-B.; Bai, Y.-K.; Wang, F.-J.; Guo, W.-Z.; et al. Inhibition of human thrombin by the constituents of licorice: Inhibition kinetics and mechanistic insights through in vitro and in silico studies. RSC Adv. 2020, 10, 3626–3635. [Google Scholar] [CrossRef]
- Guan, W.-J.; Ni, Z.-Y.; Hu, Y.; Liang, W.-H.; Ou, C.-Q.; He, J.-X.; Liu, L.; Shan, H.; Lei, C.-L.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Du, Q. Potential natural compounds for preventing SARS-CoV-2 (2019-nCoV) infection. Preprints 2020, 2020010358. [Google Scholar] [CrossRef]
- Cosmetic Ingredient Review Expert Panel. Final Report on the Safety Assessment of Glycyrrhetinic Acid, Potassium Glycyrrhetinate, Disodium Succinoyl Glycyrrhetinate, Glyceryl Glycyrrhetinate, Glycyrrhetinyl Stearate, Stearyl Glycyrrhetinate, Glycyrrhizic Acid, Ammonium Glycyrrhizate, Dipotassium Glycyrrhizate, Disodium Glycyrrhizate, Trisodium Glycyrrhizate, Methyl Glycyrrhizate, and Potassium Glycyrrhizinate1. Int. J. Toxicol. 2007, 26 (Suppl. 2), 79–112. [Google Scholar] [CrossRef]
- Chen, F.; Chan, K.H.; Jiang, Y.; Kao, R.Y.T.; Lu, H.T.; Fan, K.W.; Cheng, V.C.C.; Tsui, W.H.W.; Hung, I.F.N.; Lee, T.S.W. In vitro susceptibility of 10 clinical isolates of SARS coronavirus to selected antiviral compounds. J. Clin. Virol. 2004, 31, 69–75. [Google Scholar] [CrossRef]
- Ruiz-Granados, E.S.; Shouls, G.; Sainsbury, C.; Antonios, T. A salty cause of severe hypertension. BMJ Case Rep. 2012, 2012, bcr1220115336. [Google Scholar] [CrossRef] [PubMed]
- Mu, Y.; Zhang, J.; Zhang, S.; Zhou, H.-H.; Toma, D.; Ren, S.; Huang, L.; Yaramus, M.; Baum, A.; Venkataramanan, R.; et al. Traditional Chinese Medicines Wu Wei Zi (Schisandra chinensis Baill) and Gan Cao (Glycyrrhiza uralensis Fisch) Activate Pregnane X Receptor and Increase Warfarin Clearance in Rats. J. Pharmacol. Exp. Ther. 2005, 316, 1369–1377. [Google Scholar] [CrossRef] [PubMed]
- Cavallari, L.H.; Limdi, N.A. Warfarin pharmacogenomics. Curr. Opin. Mol. Ther. 2009, 11, 243–251. [Google Scholar] [PubMed]


| Bioactive Compounds | Properties | References |
|---|---|---|
| Glycerrhizin (GL) | Antimicrobial | [80,81,82] |
| 18β-Glycerrhitinic acid | Antimicrobial, anti-inflammatory | |
| (GA) | Against Helicobacter pylori, | [83,84,85] |
| MRSA, | ||
| Clarithromycin-resistant H. pylori | ||
| 18α-GC, 18β-GC | Anti-inflammatory | |
| Flavonoids (13 Ns below) | Anti-inflammatory | |
| Licochalcone A/B/C/D/E, isoliquiritigenin (ISL) echinatin (EC), glabridin (GLD), soangustone A (ISOA), licoricidin (LID), licorisoflavan A (LIA), dehydroglyasperin C (DGC), & dehydroglyasperin D (DGD) Glabridin | Antimicrobial, anti-inflammatory | [86] |
| Antimicrobial | [87] | |
| Aqueous extract | B. Subtilis and E. Coli | [88] |
| Methanol extract | Phytopathogenic fungi | [89] |
| Glycyrhetinic acid | MRSA | [84] |
| Licochalcone and Anti-fungal | [90] | |
| Acetate root extract | K. pneumonia and A. baylyi | [91] |
| Glabron | ||
| Licochalcone A/C/E | Staphylococcus aureus | [92] |
| Glycyrrhizin | Helicobacter pylori | [93] |
| 18β-Glycerrhetinic acid | Clarithromycin-resistant H. pylori | [85] |
| Compounds in Licorice | Antiviral Property against | Reference |
| Glycyrrhizic acid | SARS-CoV | [111] |
| Glycyrrhizic acid derivatives | SARS-CoV | [110] |
| Glycyrrhizic acid | Hepatitis A (HAV) | [113] |
| Glycyrrhizic acid | Hepatitis B (HBV) | [81,114,115] |
| Glycyrrhizic acid | Hepatitic C virus | [101,102,116,117] |
| Glycyrrhizic acid | Human immune deficiency (HIV) Virus | [103,118,119,120] |
| Alkali root extract | HIV | [109] |
| Glycyrrhizic acid | Herpes viridae (varicella) | [121] |
| Zoster virus (VZV) | [122] | |
| Epstien-Barr virus (EBV) | [122] | |
| Cytomegalovirus (CMV) | [123] | |
| Coxasackievirus B3 (CVB3) | [124] | |
| Coxasackievirus A16 (CVA16) | [57] | |
| Glycyrrhizic acid | H5N1 influenza virus | [104,105,106] |
| Duck Hepatitis virus | [108] | |
| Herpes simplex virus–1 | [107] | |
| [125] | ||
| Water extract | HSV | [109] |
| 18β-glycyrrhetinic acid | Rotavirus | [126] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abraham, J.; Florentine, S. Licorice (Glycyrrhiza glabra) Extracts-Suitable Pharmacological Interventions for COVID-19? A Review. Plants 2021, 10, 2600. https://doi.org/10.3390/plants10122600
Abraham J, Florentine S. Licorice (Glycyrrhiza glabra) Extracts-Suitable Pharmacological Interventions for COVID-19? A Review. Plants. 2021; 10(12):2600. https://doi.org/10.3390/plants10122600
Chicago/Turabian StyleAbraham, Joji, and Singarayer Florentine. 2021. "Licorice (Glycyrrhiza glabra) Extracts-Suitable Pharmacological Interventions for COVID-19? A Review" Plants 10, no. 12: 2600. https://doi.org/10.3390/plants10122600
APA StyleAbraham, J., & Florentine, S. (2021). Licorice (Glycyrrhiza glabra) Extracts-Suitable Pharmacological Interventions for COVID-19? A Review. Plants, 10(12), 2600. https://doi.org/10.3390/plants10122600







