Red Light Enhances the Antioxidant Properties and Growth of Rubus hongnoensis
Abstract
:1. Introduction
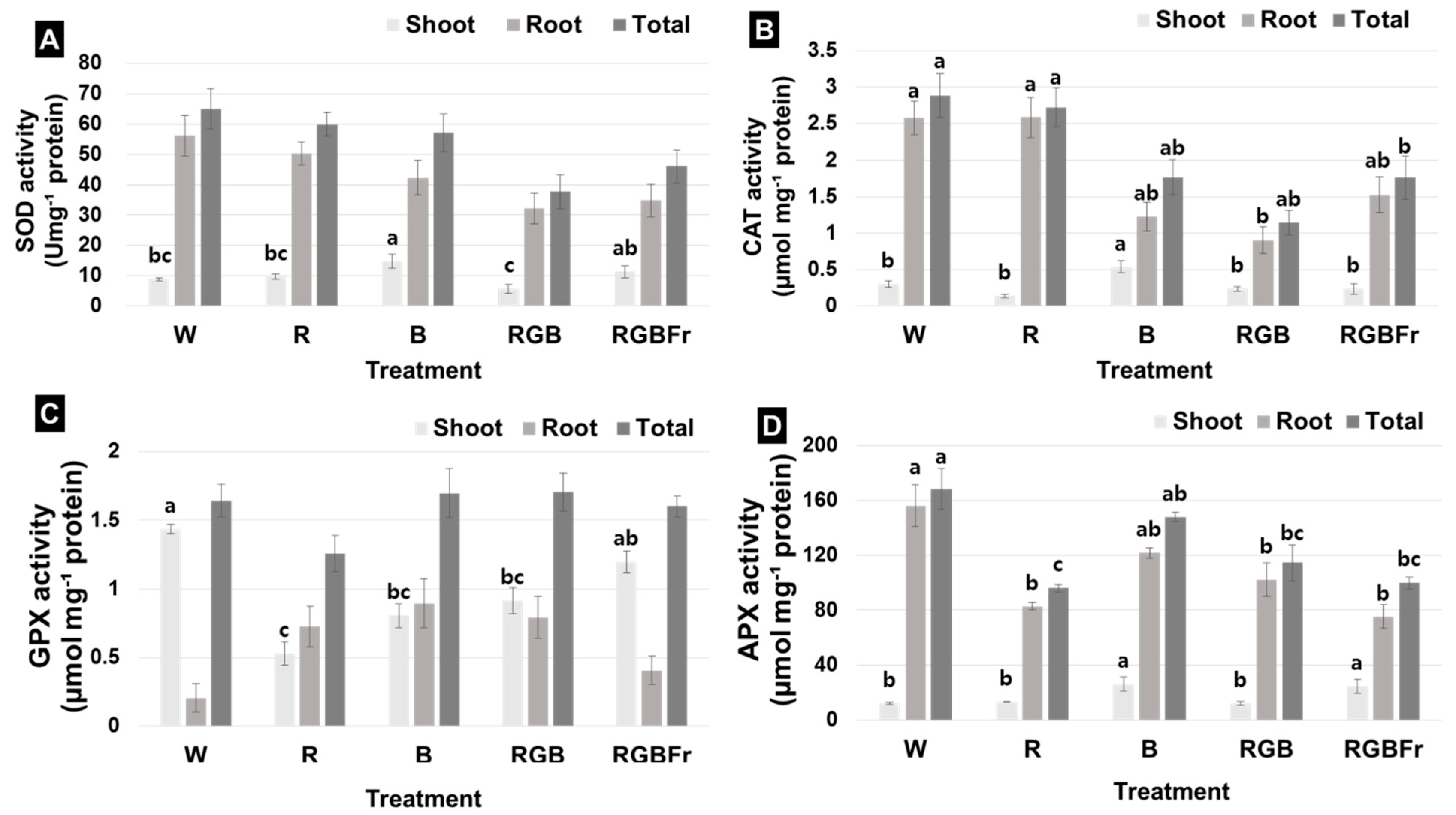
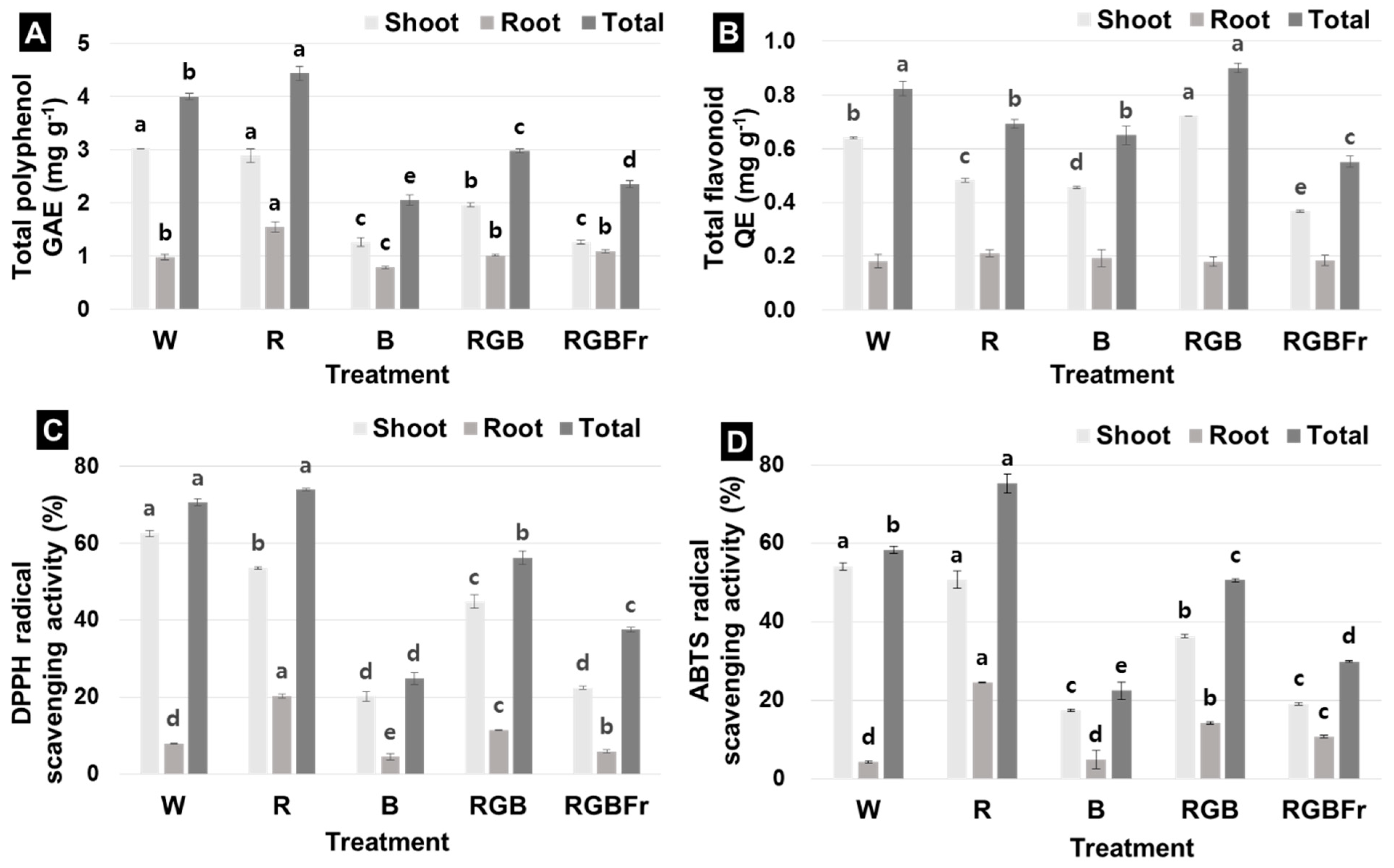
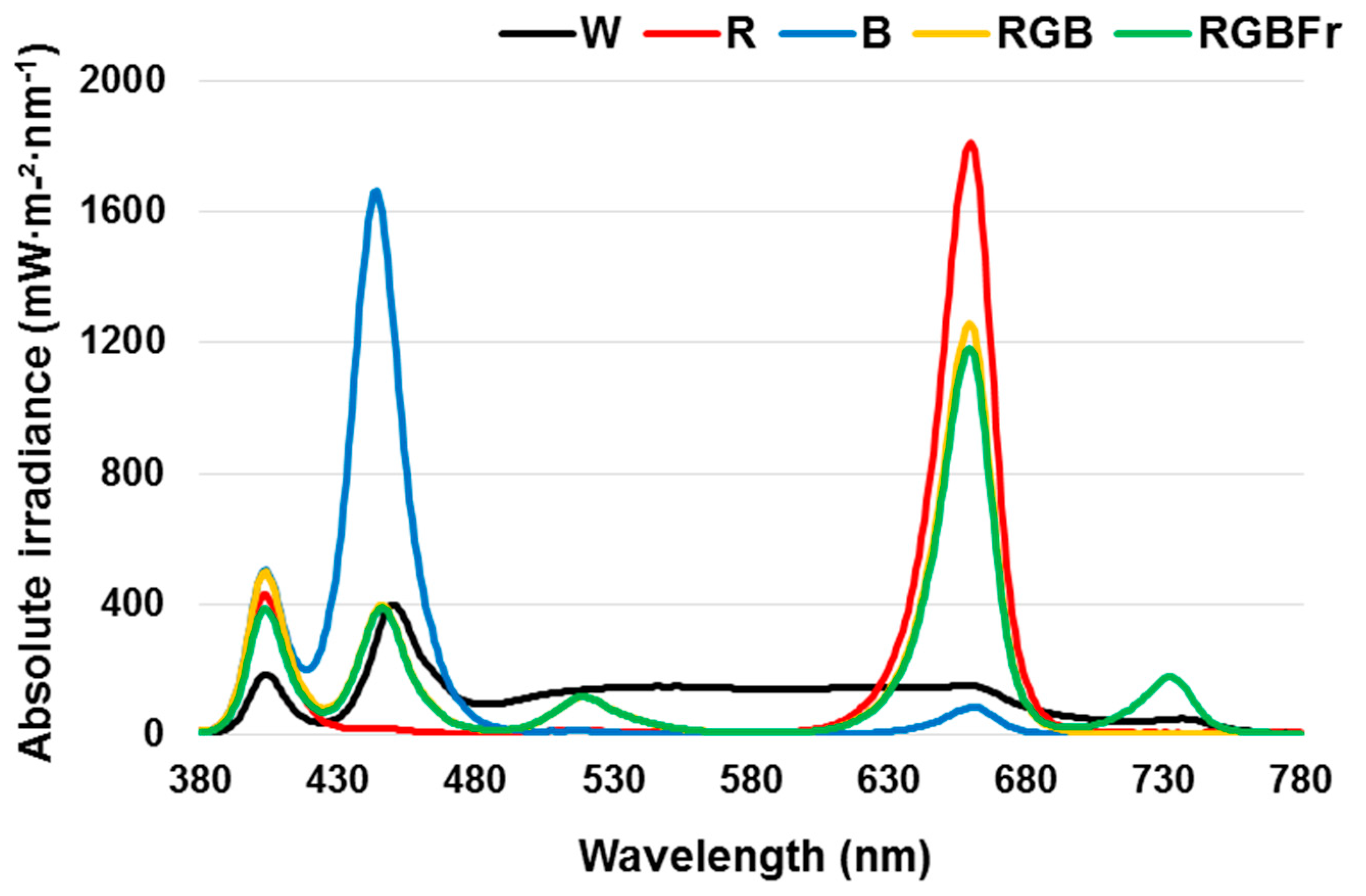
2. Results
3. Discussion
3.1. Morphogenesis
3.2. Photosynthesis
3.3. Antioxidant
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
4.2. Light Treatment
4.3. Growth Characteristics
4.4. Photosynthesis Measurements
4.5. Enzymatic Antioxidants
4.6. Extract Preparation
4.7. Total Phenol Content and Flavonoid Content
4.8. Total Flavonoid Content
4.9. DPPH Radical Scavenging Assay
4.10. ABTS Radical Scavenging Assay
4.11. Experimental Design and Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ma, J.; Park, C.-W. The Genera of Vascular Plants of Korea. TAXON 2008, 57, 681. [Google Scholar] [CrossRef]
- Nakai, T. Plantae novae Coreanae et Japonicae II. Repert. Spec. Nov. Regni Veg. 1914, 13, 277. [Google Scholar] [CrossRef]
- Patel, A.V.; Rojas-Vera, J.; Dacke, C.G. Therapeutic constituents and actions of Rubus species. Curr. Med. Chem. 2004, 11, 1501–1512. [Google Scholar] [CrossRef] [PubMed]
- Lechowicz, K.; Wrońska-Pilarek, D.; Bocianowski, J.; Maliński, T. Pollen morphology of Polish species from the genus Rubus L. (Rosaceae) and its systematic importance. PLoS ONE 2020, 15, e0221607. [Google Scholar] [CrossRef]
- Oszmiański, J.; Wojdyło, A.; Nowicka, P.; Teleszko, M.; Cebulak, T.; Wolanin, M. Determination of Phenolic Compounds and Antioxidant Activity in Leaves from Wild Rubus L. Species. Molecules 2015, 20, 4951–4966. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, M.; Masood, S.; Sultana, S.; Ben Hadda, T.; Bader, A.; Zafar, M. Report: Antioxidant and nutraceutical value of wild medicinal Rubus berries. Pak. J. Pharm. Sci. 2015, 28, 241–247. [Google Scholar]
- Van Ieperen, W. Plant Morphological and Developmental Responses to Light Quality in a Horticultural Context. Acta Hortic. 2012, 131–139. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.; Liang, Y.; Yang, M. Effects of Composite LED Light on Root Growth and Antioxidant Capacity of Cunninghamia lanceolata Tissue Culture Seedlings. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef]
- Wang, H.; Gu, M.; Cui, J.; Shi, K.; Zhou, Y.; Yu, J. Effects of light quality on CO2 assimilation, chlorophyll-fluorescence quenching, expression of Calvin cycle genes and carbohydrate accumulation in Cucumis sativus. J. Photochem. Photobiol. B Biol. 2009, 96, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Cheng, Z. Hoagland nutrient solution promotes the growth of cucumber seedlings under light-emitting diode light. Acta Agric. Scand. Sect. B Plant. Soil Sci. 2014, 65, 74–82. [Google Scholar] [CrossRef]
- Jishi, T. LED Lighting Technique to Control Plant Growth and Morphology. In Smart Plant Factory; Springer: Singapore, 2018; pp. 211–222. [Google Scholar] [CrossRef]
- Kim, H.-H.; Goins, G.D.; Wheeler, R.M.; Sager, J.C. Stomatal Conductance of Lettuce Grown under or Exposed to Different Light Qualities. Ann. Bot. 2004, 94, 691–697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, T.; Maruhnich, S.A.; Folta, K.M. Green Light Induces Shade Avoidance Symptoms. Plant. Physiol. 2011, 157, 1528–1536. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shibuya, T.; Endo, R.; Kitaya, Y.; Hayashi, S. Growth Analysis and Photosynthesis Measurements of Cucumber Seedlings Grown under Light with Different Red to Far-red Ratios. HortScience 2016, 51, 843–846. [Google Scholar] [CrossRef] [Green Version]
- Ohashi-Kaneko, K.; Takase, M.; Kon, N.; Fujiwara, K.; Kurata, K. Effect of Light Quality on Growth and Vegetable Quality in Leaf Lettuce, Spinach and Komatsuna. Environ. Control. Biol. 2007, 45, 189–198. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.-C.; Lin, C.-C. Red Light-emitting Diode Light Irradiation Improves Root and Leaf Formation in Difficult-to-propagate Protea cynaroides L. Plantlets In Vitro. HortScience 2012, 47, 1490–1494. [Google Scholar] [CrossRef] [Green Version]
- Heo, J.W.; Shin, K.S.; Kim, S.K.; Paek, K.Y. Light quality affectsin Vitro growth of grape ‘Teleki 5BB’. J. Plant. Biol. 2006, 49, 276–280. [Google Scholar] [CrossRef]
- Lee, J.G.; Oh, S.S.; Cha, S.H.; Jang, Y.A.; Kim, S.Y.; Um, Y.C.; Cheong, S.R. Effects of red/blue light ratio and short-term light quality conversion on growth and anthocyanin contents of baby leaf lettuce. J. Bio-Environ. 2010, 19, 351–359. [Google Scholar]
- Pecháčková, S. Root response to above-ground light quality–Differences between rhizomatous and non-rhizomatous clones of Festuca rubra. Plant Ecol. 1999, 141, 67–77. [Google Scholar] [CrossRef]
- Li, H.; Xu, Z.; Tang, C. Effect of light-emitting diodes on growth and morphogenesis of upland cotton (Gossypium hirsutum L.) plantlets in vitro. Plant Cell Tissue Organ Cult. 2010, 103, 155–163. [Google Scholar] [CrossRef]
- Simlat, M.; Ślęzak, P.; Moś, M.; Warchoł, M.; Skrzypek, E.; Ptak, A. The effect of light quality on seed germination, seedling growth and selected biochemical properties of Stevia rebaudiana Bertoni. Sci. Hortic. 2016, 211, 295–304. [Google Scholar] [CrossRef]
- Wu, Q.; Su, N.; Shen, W.; Cui, J. Analyzing photosynthetic activity and growth of Solanum lycopersicum seedlings exposed to different light qualities. Acta Physiol. Plant. 2014, 36, 1411–1420. [Google Scholar] [CrossRef]
- Park, Y.G.; Park, J.E.; Hwang, S.J.; Jeong, B.R. Light source and CO2 concentration affect growth and anthocyanin content of lettuce under controlled environment. Hortic. Environ. Biotechnol. 2012, 53, 460–466. [Google Scholar] [CrossRef]
- Emerson, R.; Rabinowitch, E. Red Drop and Role of Auxiliary Pigments in Photosynthesis. Plant Physiol. 1960, 35, 477–485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- YYorio, N.C.; Goins, G.D.; Kagie, H.R.; Wheeler, R.M.; Sager, J.C. Improving Spinach, Radish, and Lettuce Growth under Red Light-emitting Diodes (LEDs) with Blue Light Supplementation. HortScience 2001, 36, 380–383. [Google Scholar] [CrossRef] [Green Version]
- Gajc-Wolska, J.; Kowalczyk, K.; Przybysz, A.; Mirgos, M.; Orliński, P. Photosynthetic Efficiency and Yield of Cucumber (Cucumis sativus L.) Grown under HPS and LED Lighting in Autumn–Winter Cultivation. Plants 2021, 10, 2042. [Google Scholar] [CrossRef]
- Yang, X.; Xu, H.; Shao, L.; Li, T.; Wang, Y.; Wang, R. Response of photosynthetic capacity of tomato leaves to different LED light wavelength. Environ. Exp. Bot. 2018, 150, 161–171. [Google Scholar] [CrossRef]
- Kobayashi, K.; Amore, T.; Lazaro, M. Light-Emitting Diodes (LEDs) for Miniature Hydroponic Lettuce. Opt. Photon. J. 2013, 3, 74–77. [Google Scholar] [CrossRef] [Green Version]
- Hwang, C.H.; Park, Y.G.; Jeong, B.R. Changes in content of total polyphenol and activities of antioxidizing enzymes in Perilla frutescens var. acuta Kudo and Salvia plebeia R. Br. as affected by light intensity. Hortic. Environ. Biotechnol. 2014, 55, 489–497. [Google Scholar] [CrossRef]
- Dewir, Y.; Chakrabarty, D.; Ali, M.; Hahn, E.; Paek, K.Y. Lipid peroxidation and antioxidant enzyme activities of Euphorbia millii hyperhydric shoots. Environ. Exp. Bot. 2006, 58, 93–99. [Google Scholar] [CrossRef]
- Yu, W.; Liu, Y.; Song, L.; Jacobs, D.; Du, X.; Ying, Y.; Shao, Q.; Wu, J. Effect of Differential Light Quality on Morphology, Photosynthesis, and Antioxidant Enzyme Activity in Camptotheca acuminata Seedlings. J. Plant. Growth Regul. 2016, 36, 148–160. [Google Scholar] [CrossRef]
- Bantis, F. Light Spectrum Differentially Affects the Yield and Phytochemical Content of Microgreen Vegetables in a Plant Factory. Plants 2021, 10, 2182. [Google Scholar] [CrossRef] [PubMed]
- Ye, F.; Liang, Q.; Li, H.; Zhao, G. Solvent effects on phenolic content, composition, and antioxidant activity of extracts from florets of sunflower (Helianthus annuus L.). Ind. Crop. Prod. 2015, 76, 574–581. [Google Scholar] [CrossRef]
- Zheng, L.; Van Labeke, M.-C. Chrysanthemum morphology, photosynthetic efficiency and antioxidant capacity are differentially modified by light quality. J. Plant Physiol. 2017, 213, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Giménez, A.; Martínez-Ballesta, M.; Egea-Gilabert, C.; Gómez, P.; Artés-Hernández, F.; Pennisi, G.; Orsini, F.; Crepaldi, A.; Fernández, J. Combined Effect of Salinity and LED Lights on the Yield and Quality of Purslane (Portulaca oleracea L.) Microgreens. Horticulturae 2021, 7, 180. [Google Scholar] [CrossRef]
- Hashim, M.; Ahmad, B.; Drouet, S.; Hano, C.; Abbasi, B.; Anjum, S. Comparative Effects of Different Light Sources on the Production of Key Secondary Metabolites in Plants In Vitro Cultures. Plants 2021, 10, 1521. [Google Scholar] [CrossRef]
- Hogewoning, S.; Trouwborst, G.; Maljaars, H.; Poorter, H.; Van Ieperen, W.; Harbinson, J. Blue light dose-responses of leaf photosynthesis, morphology, and chemical composition of Cucumis sativus grown under different combinations of red and blue light. J. Exp. Bot. 2010, 61, 3107–3117. [Google Scholar] [CrossRef] [PubMed]
- Shohael, A.; Ali, M.; Yu, K.; Hahn, E.; Islam, R.; Paek, K. Effect of light on oxidative stress, secondary metabolites and induction of antioxidant enzymes in Eleutherococcus senticosus somatic embryos in bioreactor. Process. Biochem. 2006, 41, 1179–1185. [Google Scholar] [CrossRef]
- Taulavuori, K.; Hyöky, V.; Oksanen, J.; Taulavuori, E.; Julkunen-Tiitto, R. Species-specific differences in synthesis of flavonoids and phenolic acids under increasing periods of enhanced blue light. Environ. Exp. Bot. 2016, 121, 145–150. [Google Scholar] [CrossRef]
- Liu, Y.; Fang, S.; Yang, W.; Shang, X.; Fu, X. Light quality affects flavonoid production and related gene expression in Cyclocarya paliurus. J. Photochem. Photobiol. B Biol. 2018, 179, 66–73. [Google Scholar] [CrossRef]
- Ouzounis, T.; Heuvelink, E.; Ji, Y.; Schouten, H.; Visser, R.; Marcelis, L. Blue and red LED lighting effects on plant biomass, stomatal conductance, and metabolite content in nine tomato genotypes. Acta Hortic. 2016, 251–258. [Google Scholar] [CrossRef]
- Alma, M.H.; Mavi, A.; Yildirim, A.; Digrak, M.; Hirata, T. Screening Chemical Composition and in Vitro Antioxidant and Antimicrobial Activities of the Essential Oils from Origanum syriacum L. Growing in Turkey. Biol. Pharm. Bull. 2003, 26, 1725–1729. [Google Scholar] [CrossRef] [Green Version]
- Shiga, T.; Shoji, K.; Shimada, H.; Hashida, S.-N.; Goto, F.; Yoshihara, T. Effect of light quality on rosmarinic acid content and antioxidant activity of sweet basil, Ocimum basilicum L. Plant Biotechnol. 2009, 26, 255–259. [Google Scholar] [CrossRef] [Green Version]
- Denoya, G.I.; Pataro, G.; Ferrari, G. Effects of postharvest pulsed light treatments on the quality and antioxidant properties of persimmons during storage. Postharvest Biol. Technol. 2020, 160. [Google Scholar] [CrossRef]
- Chen, X.N.; Fan, J.F.; Yue, X.; Wu, X.R.; Li, L.T. Radical Scavenging Activity and Phenolic Compounds in Persimmon (Diospyros kaki L. cv. Mopan). J. Food Sci. 2007, 73, C24–C28. [Google Scholar] [CrossRef] [PubMed]
- Alici, E.H.; Arabaci, G. Determination of SOD, POD, PPO and CAT Enzyme Activities in Rumex obtusifolius L. Annu. Res. Rev. Biol. 2016, 11, 1–7. [Google Scholar] [CrossRef]
- Aebi, H. Catalase. In Methods of Enzymatic Analysis; Academic Press: New York, NY, USA, 1974; pp. 673–684. [Google Scholar]
- Sadasivam, S.; Manickam, A. Biochemical Methods; New Age International (P) Limited: New Delhi, India, 1996; Volume 2, pp. 124–126. [Google Scholar]
- Chen, G.-X.; Asada, K. Ascorbate Peroxidase in Tea Leaves: Occurrence of Two Isozymes and the Differences in Their Enzymatic and Molecular Properties. Plant. Cell Physiol. 1989, 30, 987–998. [Google Scholar] [CrossRef]
- Zhou, Y.; Singh, B.R. Red light stimulates flowering and anthocyanin biosynthesis in American cranberry. Plant Growth Regul. 2002, 38, 165–171. [Google Scholar] [CrossRef]
- Kumaran, A.; Karunakaran, R.J. In vitro antioxidant activities of methanol extracts of five Phyllanthus species from India. LWT Food Sci. Technol. 2007, 40, 344–352. [Google Scholar] [CrossRef]
- Blois, M.S. Antioxidant Determinations by the Use of a Stable Free Radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]


| Light Quality z | Plant Height (cm) | Leaf | No. of Five-Leaflets | Chlorophyll (SPAD) | Stem Diameter (mm) | ||
|---|---|---|---|---|---|---|---|
| Length (cm) | Width (cm) | Area (cm2) | |||||
| W | 6.8 b y | 13.4 a | 8.4 a | 44.7 ab | 5.7 ab | 38.0 | 5.3 a |
| R | 11.2 a | 16.7 a | 10.4 a | 66.2 a | 6.6 a | 40.7 | 6.6 a |
| B | 3.7 c | 8.8 b | 5.5 b | 24.1 c | 5.1 b | 34.3 | 2.8 b |
| RGBFr | 7.9 b | 15.0 a | 8.5 a | 51.6 b | 6.8 a | 39.6 | 5.2 a |
| RGB | 8.2 b | 15.3 a | 9.0 a | 55.2 ab | 6.4 a | 39.8 | 4.9 a |
| F-test | *** | *** | *** | *** | ** | NS | ** |
| Light Quality z | Root Length (cm) | Fresh Weight (g) | Dry Weight (g) | ||
|---|---|---|---|---|---|
| Shoot | Root | Shoot | Root | ||
| W | 27.1 bc y | 5.13 a | 3.68 bc | 0.95 ab | 0.28 bc |
| R | 33.1 ab | 11.65 a | 8.34 a | 2.05 a | 0.64 ab |
| B | 25.2 c | 2.76 b | 2.06 c | 0.35 b | 0.12 c |
| RGBFr | 34.4 ab | 8.29 ab | 7.98 ab | 1.59 a | 0.52 ab |
| RGB | 38.4 a | 11.90 a | 8.59 a | 1.94 a | 0.66 a |
| F-test | ** | ** | ** | ** | ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oh, H.E.; Yoon, A.; Park, Y.G. Red Light Enhances the Antioxidant Properties and Growth of Rubus hongnoensis. Plants 2021, 10, 2589. https://doi.org/10.3390/plants10122589
Oh HE, Yoon A, Park YG. Red Light Enhances the Antioxidant Properties and Growth of Rubus hongnoensis. Plants. 2021; 10(12):2589. https://doi.org/10.3390/plants10122589
Chicago/Turabian StyleOh, Hyeon Eui, Ara Yoon, and Yoo Gyeong Park. 2021. "Red Light Enhances the Antioxidant Properties and Growth of Rubus hongnoensis" Plants 10, no. 12: 2589. https://doi.org/10.3390/plants10122589
APA StyleOh, H. E., Yoon, A., & Park, Y. G. (2021). Red Light Enhances the Antioxidant Properties and Growth of Rubus hongnoensis. Plants, 10(12), 2589. https://doi.org/10.3390/plants10122589






