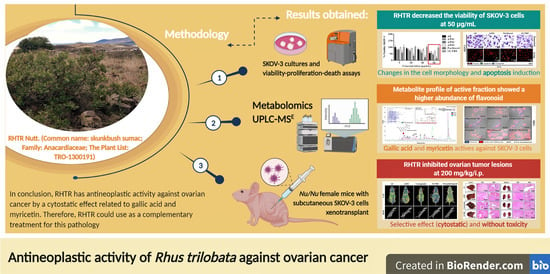
Antineoplastic Activity of Rhus trilobata Nutt. (Anacardiaceae) against Ovarian Cancer and Identification of Active Metabolites in This Pathology
Abstract
:1. Introduction
2. Results and Discussion
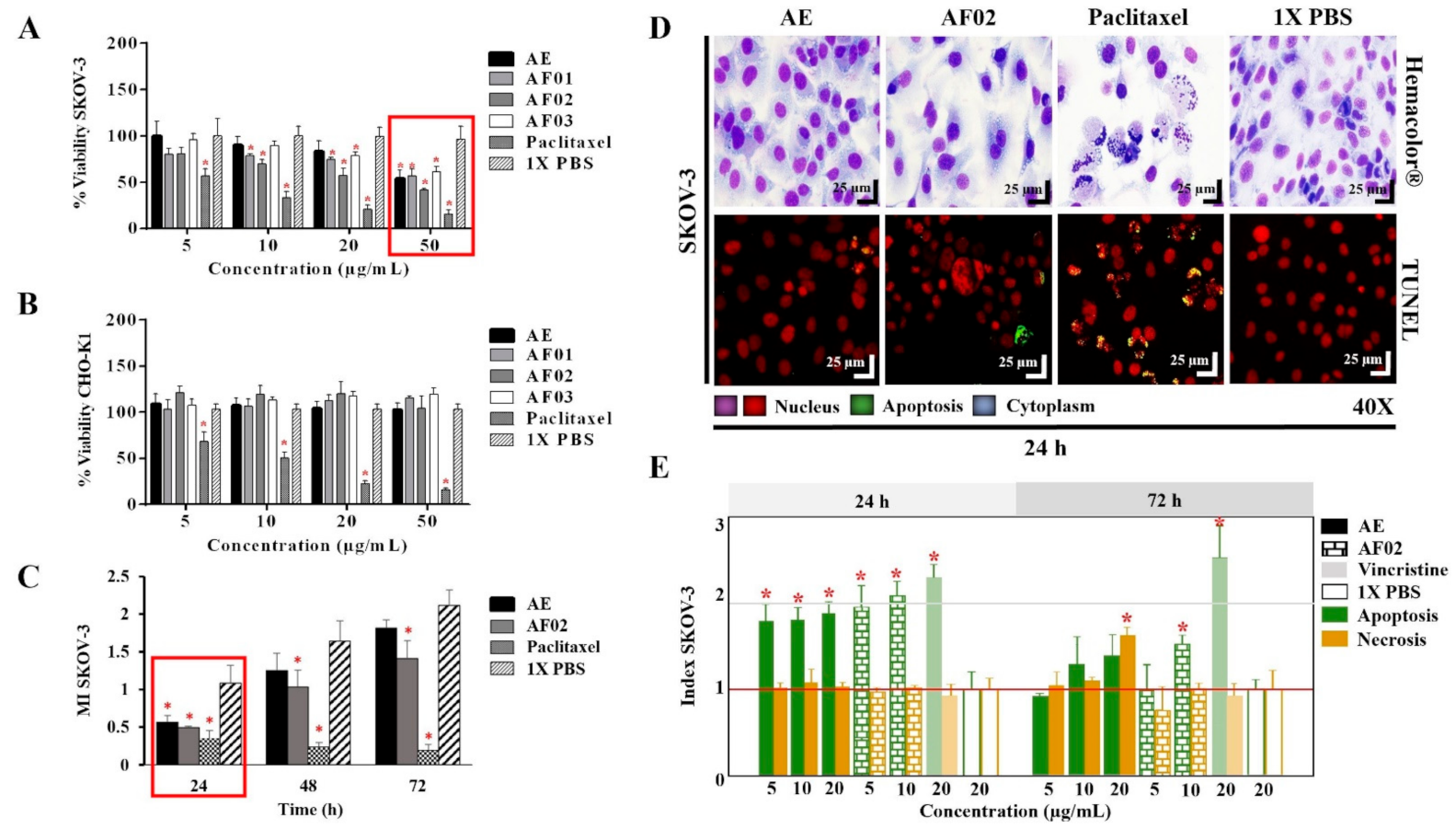
2.1. Biological Activity of AE-RHTR and Fractions in Cell Lines
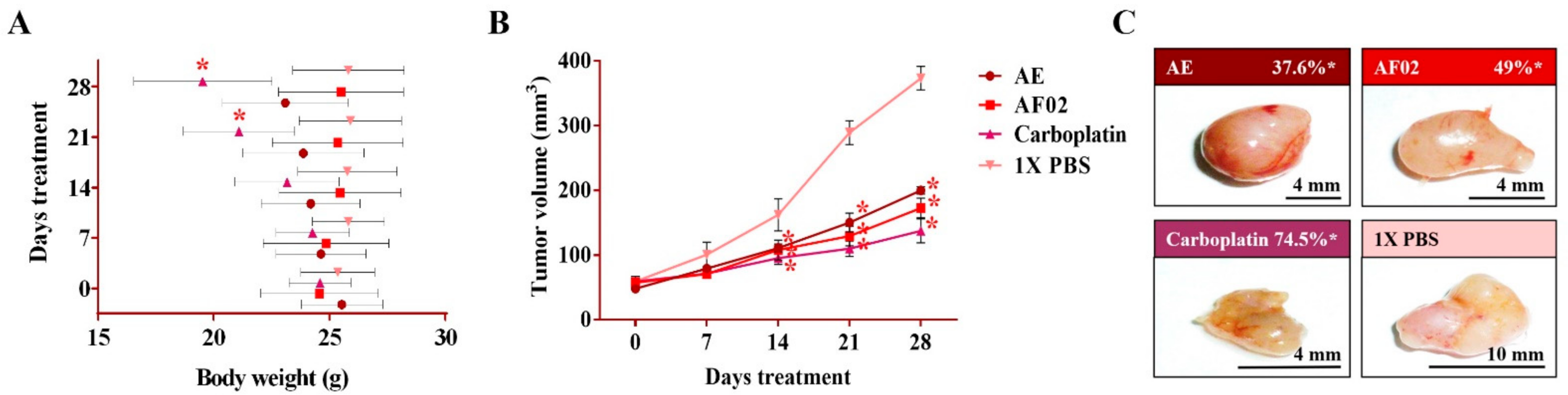
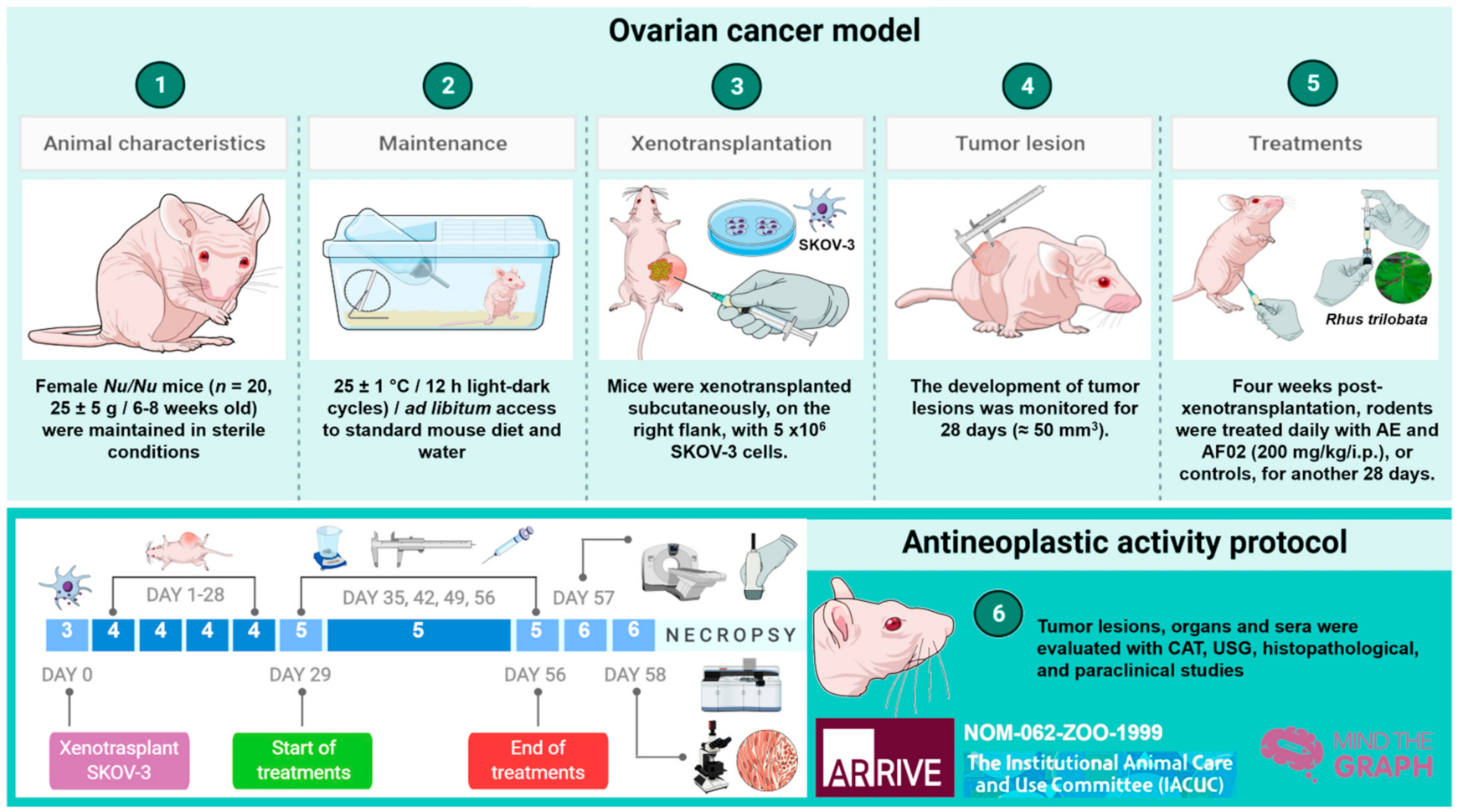
2.2. Antineoplastic Activity of RHTR in Mice Xenotransplanted with SKOV-3 Cells
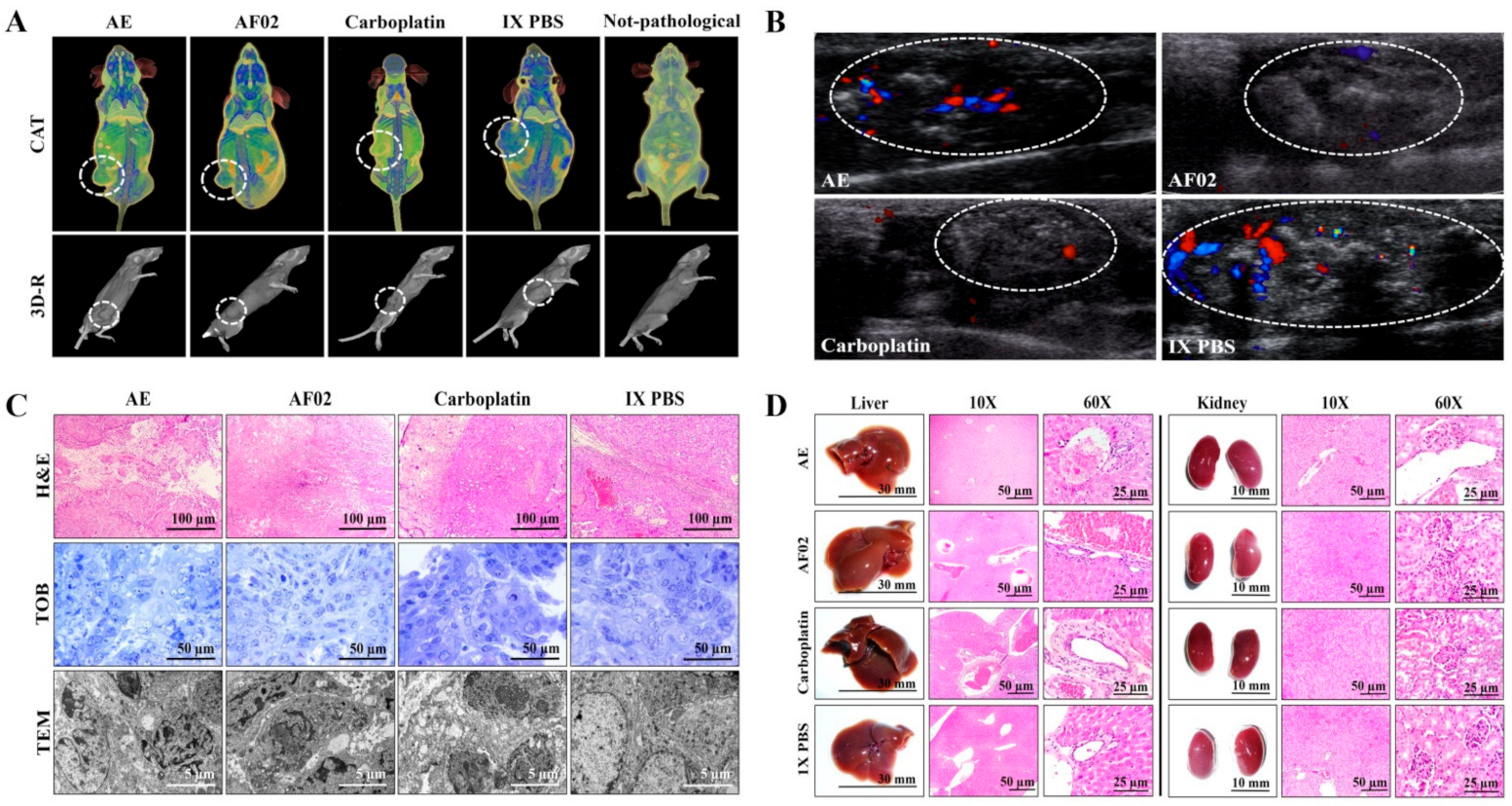
2.2.1. Imaging and Histopathological Studies of Tumor Lesions
2.2.2. Morphometric and Paraclinical Studies of Mice Treated with RHTR
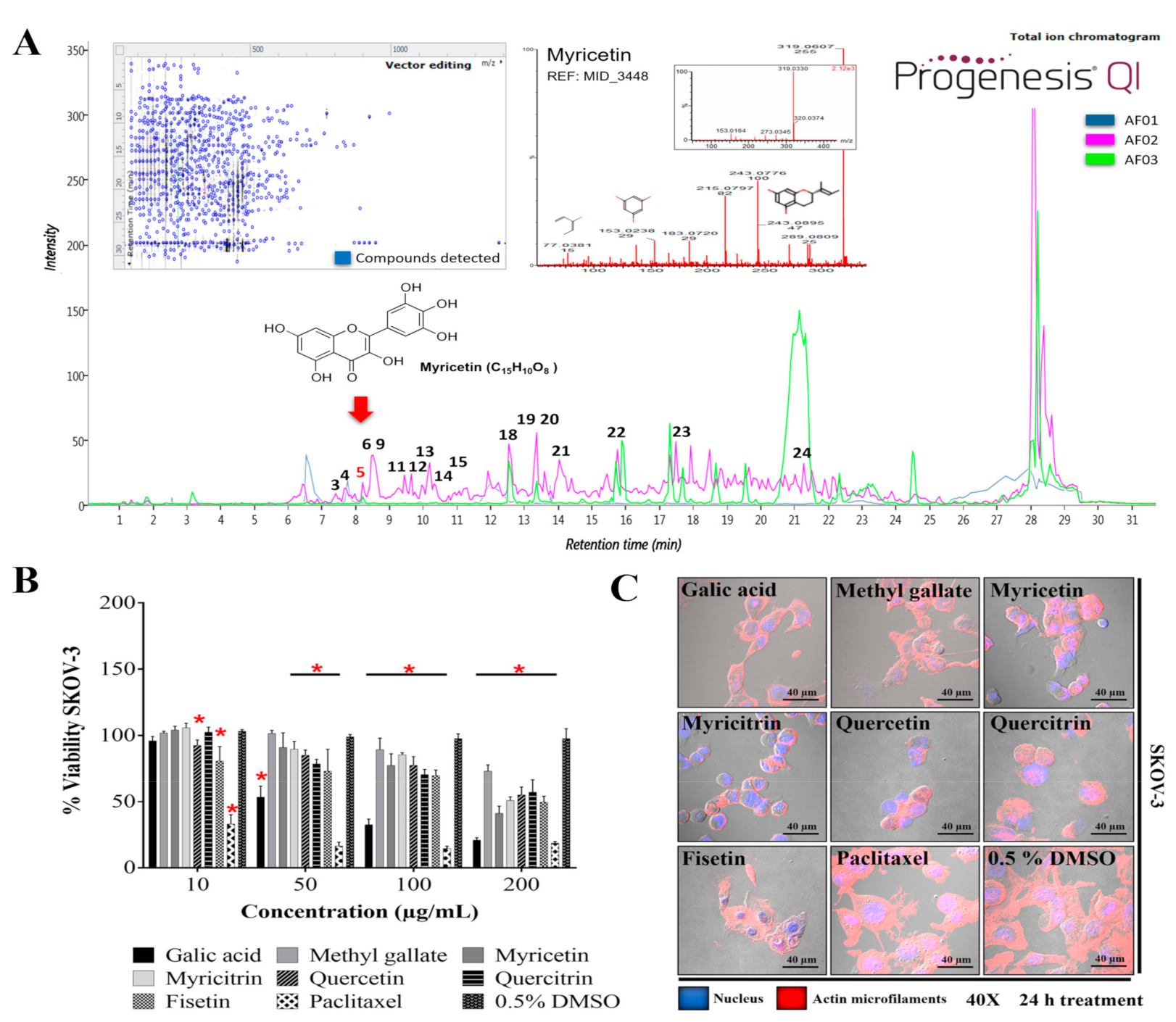
2.3. Purified Metabolites in RHTR
2.4. Phytochemical Composition and in Silico Analysis of AF02-RHTR
2.5. Biological Activity of Active Metabolites from RHTR in Cell Lines
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Recollection of the Plant Material
3.3. Preparation of the Plant Extract and Fractionation
3.4. Purification and Identification of Metabolites
3.5. Cell Culture
3.6. Determination of Cell Viability by Formazan Salts
3.7. Measurement of Proliferation, Cytotoxicity, and Apoptosis in Cells
3.8. Evaluation of Cell Morphology
3.9. Experimental Animals
3.10. Imaging, Paraclinical, and Histopathologic Studies
3.11. Phytochemical Composition of the RHTR Active Fraction
3.12. In Silico Studies and Statistical Analysis
4. Conclusions
5. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Gallardo-Rincón, D.; Espinosa-Romero, R.; Muñoz, W.R.; Mendoza-Martínez, R.; Villar-Álvarez, S.D.; Oñate-Ocaña, L.; Isla-Ortiz, D.; Márquez-Manríquez, J.P.; Apodaca-Cruz, Á.; Meneses-García, A. Epidemiological overview, advances in diagnosis, prevention, treatment and management of epithelial ovarian cancer in Mexico. Salud Publica Mex. 2016, 58, 302–308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DeFriend, D. Ovarios. In Ultrasonido Clinico; Allan, P., Baxter, G., Weston, M., Eds.; Amolca: Ciudad de México, Mexico, 2016; pp. 660–685. ISBN 9789588760827. [Google Scholar]
- Posada-Torres, J.A.; Del Real-Ordóñez, S.; Salcedo-Hernández, R.A. Capítulo 8, Tratamiento quirúrgico inicial: ¿rutina en ovario?, ¿existen variaciones en la cirugía para enfermedad temprana y avanzada. In COI: Cáncer de Ovario Epitelial; Gallardo-Rincón, D., Meneses-García, A., De la Garza-Salazar, J.G., Juárez-Sanchez, P., Aguilar-Ponce, J.L., Eds.; PyDESA: Ciudad de México, Mexico, 2016; pp. 87–93. [Google Scholar]
- Varela-Rodríguez, L.; Sánchez-Ramírez, B.; Rodríguez-Reyna, I.S.; Ordaz-Ortiz, J.J.; Chávez-Flores, D.; Salas-Muñoz, E.; Osorio-Trujillo, J.C.; Ramos-Martínez, E.; Talamás-Rohana, P. Biological and toxicological evaluation of Rhus trilobata Nutt. (Anacardiaceae) used traditionally in Mexico against cancer. BMC Complementary Med. Ther. 2019, 19, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Abbott, B.J.; Leiter, J.; Hartwell, J.L.; Caldwell, M.E.; Beal, J.L.; Perdue, R.E.; Schepartz, S.A. Screening data from the cancer chemotherapy national service center screening laboratories. XXXIV. Plant extracts. Cancer Res. 1966, 26, 1131–1271. [Google Scholar]
- Pettit, G.R.; Saldana, E.I.; Lehto, E. Antineoplastic agents 35. Rhus trilobata. Lloydia 1974, 37, 539–540. [Google Scholar] [PubMed]
- National Comprehensive Cancer Network (NCCN). Guidelines V. 1.2016: Ovarian Cancer. 2016. Available online: https://jnccn.org/view/journals/jnccn/14/9/articlep1134.xml#container-4698-item-4697 (accessed on 7 July 2021).
- Micetick, K.C.; Barnes, D.; Erickson, L.C. A comparative study of the cytotoxicity and DNA-damaging effects of cis-(dimamino) (1,1-cyclobutanedicarboxylato)-platinum (II) and cis-diamminedichloroplatinum (II) on L1210 cells. Cancer Res. 1985, 45, 4043–4047. [Google Scholar]
- Zhu, L.; Chen, L. Progress in research on paclitaxel and tumor immunotherapy. Cell. Mol. Biol. Lett. 2019, 24, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adeneye, A.A.; Ajagbonna, O.P.; Adeleke, T.I.; Bello, S.O. Preliminary toxicity and phytochemical studies of the stem bark aqueous extract of Musangacecropioides in rats. J. Ethnopharmacol. 2006, 105, 374–379. [Google Scholar] [CrossRef]
- Fernandes, D.P.; Pimentel, M.M.; Dos Santos, F.A.; Praxedes, E.A.; De Brito, P.D.; Lima, M.A.; Lelis, I.C.N.; De Macedo, M.F.; Bezerra, M.B. Hematological and biochemical profile of BALB/c nude and C57BL/6 SCID female mice after ovarian xenograft. An. Da Acad. Bras. De Ciências 2018, 90, 3941–3948. [Google Scholar] [CrossRef]
- Otto, G.P.; Rathkolb, B.; Oestereicher, M.A.; Lengger, C.J.; Moerth, C.; Micklich, K.; Fuchs, H.; Gailus-Durner, V.; Wolf, E.; Hrabě de Angelis, M. Clinical chemistry reference intervals for C57BL/6J, C57BL/6N, and C3HeB/FeJ mice (Mus musculus). J. Am. Assoc. Lab. Anim. Sci. 2016, 55, 375–386. [Google Scholar]
- Kamatham, S.; Kumar, N.; Gudipalli, P. Isolation and characterization of gallic acid and methyl gallate from the seed coats of Givotia rottleriformis Griff. and their anti-proliferative effect on human epidermoid carcinoma A431 cells. Toxicol. Rep. 2015, 14, 520–529. [Google Scholar] [CrossRef]
- Mohd Nazrul Hisham, D.; Mohd Lip, J.; Mohd Noh, J.; Normah, A.; Nurul Nabilah, M.F. Identification and isolation of methyl gallate as a polar chemical marker for Labisia pumila Benth. J. Trop. Agric. Food Sci. 2011, 39, 279–284. [Google Scholar]
- Rayne, S.; Mazza, G. Biological activities of extracts from Sumac (Rhus spp.): A review. Plant Foods Hum. Nutr. 2007, 62, 165–175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.; Lee, G.; Sohn, S.-H.; Lee, C.; Kwak, J.W.; Bae, H. Immunotherapy with methyl gallate, an inhibitor of Treg cell migration, enhances the anti-cancer effect of cisplatin therapy. Korean J. Physiol. Pharmacol. 2016, 20, 261–268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khurana, S.; Hollingsworth, A.; Piche, M.; Venkataraman, K.; Kumar, A.; Ross, G.M.; Tai, T.C. Antiapoptotic Actions of Methyl Gallate on Neonatal Rat Cardiac Myocytes Exposed to H2O2. Oxidative Med. Cell. Longev. 2014, 2014, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Lee, H.; Kwon, Y.; Lee, J.-H.; Kim, J.; Shin, M.-K.; Kim, S.-H.; Bae, H. Methyl gallate exhibits potent antitumor activities by inhibiting tumor infiltration of CD4+ CD25+ regulatory T cells. J. Immunol. 2010, 185, 6698–6705. [Google Scholar] [CrossRef] [Green Version]
- Devi, K.P.; Rajavel, T.; Habtemariam, S.; Nabavi, S.F.; Nabavi, S.M. Molecular mechanisms underlying anticancer effects of myricetin. Life Sci. 2015, 142, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, C.A.; Swartz, C.; Maronpot, R.; Davis, J.; Recio, L.; Koyanagi, M.; Hayashi, S.-M. Genotoxicity evaluation of the flavonoid, myricitrin, and its aglycone, myricetin. Food Chem. Toxicol. 2015, 83, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Marcarini, J.C.; Ferreira-Tsuboy, M.S.; Cabral-Luiz, R.; Ribeiro, L.R.; Hoffmann-Campo, C.B.; Ségio-Mantovani, M. Investigation of cytotoxic, apoptosis-inducing, genotoxic and protective effects of the flavonoid rutin in HTC hepatic cells. Exp. Toxicol. Pathol. 2011, 63, 459–465. [Google Scholar] [CrossRef]
- Badhani, B.; Sharma, N.; Kakkar, R. Gallic acid: A versatile antioxidant with promising therapeutic and industrial applications. RSC Adv. 2015, 5, 27540–27557. [Google Scholar] [CrossRef]
- Varela-Rodríguez, L.; Sánchez-Ramírez, B.; Hernández-Ramírez, V.I.; Varela-Rodríguez, H.; Castellanos-Mijangos, R.D.; González-Horta, C.; Chávez-Munguía, B.; Talamás-Rohana, P. Effect of Gallic acid and Myricetin on ovarian cancer models: A possible alternative antitumoral treatment. BMC Complementary Med. Ther. 2020, 20, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Li, S.; Xu, B.; Luo, H. Cancer evolution: A means by which tumors evade treatment. Biomed. Pharmacother. 2021, 133, 1–13. [Google Scholar] [CrossRef]
- Shin, H.; Park, Y.; Choi, J.H.; Jeon, Y.H.; Byun, Y.; Sung, S.H.; Lee, K.Y. Structure elucidation of a new triterpene from Rhus trichocarpa roots. Magn. Reson. Chem. 2017, 55, 763–766. [Google Scholar] [CrossRef] [PubMed]
- Gross, G.G. Enzymes in the Biosynthesis of Hydrolyzable Tannins. In Plant Polyphenols: Synthesis, Properties, Significance; Hemingway, R.W., Laks, P.E., Branham, S.J., Eds.; Springer: New York, NY, USA, 1992; pp. 43–60. [Google Scholar]
- Chen, W.J.; Chang, C.Y.; Lin, J.K. Induction of G1 phase arrest in MCF human breast cancer cells by pentagalloylglucose through the down-regulation of CDK4 and CDK2 activities and up-regulation of the CDK inhibitors p27(Kip) and p21(Cip). Biochem. Pharmacol. 2003, 65, 1777–1785. [Google Scholar] [CrossRef]
- Hu, H.; Lee, H.J.; Jiang, C.; Zhang, J.; Wang, L.; Zhao, Y.; Xiang, Q.; Lee, E.O.; Kim, S.H.; Lü, J. Penta-1,2,3,4,6-O-galloyl-β-D-glucose induces p53 and inhibits STAT3 in prostate cancer cells in vitro and suppresses prostate xenograft tumor growth in vivo. Mol. Cancer Ther. 2008, 7, 2681–2691. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Velázquez-Domínguez, J.A.; Hernández-Ramírez, V.I.; Calzada, F.; Varela-Rodríguez, L.; Pichardo-Hernández, D.L.; Bautista, E.; Herrera-Martínez, M.; Castellanos-Mijangos, R.D.; Matus-Meza, A.S.; Chávez-Munguía, B.; et al. Linearolactone and kaempferol disrupt the actin cytoskeleton in Entamoeba histolytica: Inhibition of amoebic liver abscess development. J. Nat. Prod. 2020, 83, 3671–3680. [Google Scholar] [CrossRef] [PubMed]
- Varela-Rodríguez, H.; Abella-Quintana, D.G.; Espinal-Centeno, A.; Varela-Rodríguez, L.; Gomez-Zepeda, D.; Caballero-Pérez, J.; García-Medel, P.L.; Brieba, L.G.; Ordaz-Ortiz, J.J.; Cruz-Ramirez, A. Functional characterization of the Lin28/let-7 circuit during forelimb regeneration in Ambystoma mexicanum and its influence on metabolic reprogramming. Front. Cell Dev. Biol. 2020, 8, 562940. [Google Scholar] [CrossRef]
- Gobierno de México. Norma Oficial Mexicana: NOM-062-ZOO-1999. Especificaciones Técnicas Para la Producción, Cuidado y Uso de los Animales de Laboratorio. 2001. Available online: https://www.gob.mx/cms/uploads/attachment/file/203498/NOM-062-ZOO-1999_220801.pdf (accessed on 7 July 2021).
- Kilkenny, C.; Browne, W.; Cuthill, I.C.; Emerson, M.; Altman, D.G. Animal research: Reporting in vivo experiments: The ARRIVE guidelines. BJP Br. J. Pharmacol. 2010, 160, 1577–1579. [Google Scholar] [CrossRef]
- Molsoft LLC. Drug-Likeness and Molecular Property Prediction. 2021. Available online: http://molsoft.com/mprop/ (accessed on 21 September 2021).
- Hussein, W.; Sağlık, B.N.; Levent, S.; Korkut, B.; Ilgın, S.; Özkay, Y.; Kaplancıklı, Z.A. Synthesis and biological evaluation of new cholinesterase inhibitors for Alzheimer’s disease. Molecules 2018, 23, 2033. [Google Scholar] [CrossRef] [Green Version]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2001, 46, 3–26. [Google Scholar] [CrossRef]
- Macalalad, M.; Gonzales, A.A., 3rd. In-silico screening and identification of phytochemicals from Centella asiatica as potential inhibitors of sodium-glucose co-transporter 2 for treating diabetes. J. Biomol. Struct. Dyn. 2021, 2021, 1–18. [Google Scholar] [CrossRef]
| Treatments | AE | AF02 | Carboplatin | 1× PBS |
|---|---|---|---|---|
| Weight (g) | 0.24 ± 0.09 * | 0.19 ± 0.10 * | 0.14 ± 0.06 * | 0.34 ± 0.09 |
| Larger diameter (mm) | 9.07 ± 0.25 * | 7.40 ± 0.95 * | 3.70 ± 0.26 * | 14.53 ± 2.4 |
| Tumor volume (mm3) | 201.4 ± 11.6 * | 172.7 ± 15.3 * | 137.3 ± 18.6 * | 373.3 ± 18.1 |
| Attenuation coefficient (HU) | 47 ± 2.16 | 48.8 ± 0.83 | 48.7 ± 0.95 | 47 ± 2.1 |
| Vascularity | + | + | + | +++ |
| Fibrosis | + | + | + | + |
| Morphology | Cystic | Cystic | Solid | Cystic |
| Treatments | AE | AF02 | Carboplatin | 1× PBS | Reference Range (mean) |
|---|---|---|---|---|---|
| Glucose (mg/dL) | 72 ± 23.9 | 114 ± 21.2 * | 107 ± 14.8 * | 78.3 ± 16.6 | 63–176 (89) |
| Triglycerides (mg/dL) | 71.5 ± 26.2 | 80 ± 11.35 | 103.7 ± 12.7 * | 87.6 ± 12.01 | 55–115 (85) |
| Cholesterol (mg/dL) | 66 ± 25.4 * | 62 ± 2.52 * | 108.6 ± 19.4 * | 85.5 ± 13.4 | 26–82 (64) |
| Protein (g/dL) | 4.48 ± 0.6 | 4.42 ± 0.62 | 5.1 ± 1.46 | 4.7 ± 0.56 | 4–8.6 (6.2) |
| Albumin (g/dL) | 2.1 ± 0.8 * | 2.03 ± 0.35 * | 1.33 ± 0.29 | 1.36 ± 0.21 | 2.5–4 (3.2) |
| AST (GOT) (U/L) | 132.6 ± 45.7 | 145.7 ± 7.69 | 162.9 ± 8.2 | 149.8 ± 27.29 | 55–251 (139) |
| ALT (GPT) (U/L) | 41.2 ± 9.8 | 32.43 ± 19.2 | 64.4 ± 6.87 * | 40.75 ± 14.5 | 17–77 (47) |
| Total bilirubin (mg/dL) | 0.47 ± 0.07 * | 0.39 ± 0.08 * | 0.58 ± 0.2 | 0.67 ± 0.16 | 0.20–1.0 (0.6) |
| Alkaline phosphatase (U/L) | 46.5 ± 23.2 * | 83.6 ± 24.04 | 66.6 ± 15.6 | 71 ± 10.36 | 9–88 (48.5) |
| Creatinine (mg/dL) | 0.74 ± 0.4 | 0.7 ± 0.25 | 0.37 ± 0.26 * | 0.65 ± 0.2 | 0.2–0.9 (0.5) |
| Urea (mg/dL) | 40.8 ± 8.2 | 57.6 ± 10.7 | 59.4 ± 9.99 | 47.9 ± 7.16 | 46.9–73 (60.1) |
| BUN (g/dL) | 19 ± 3.8 | 26.8 ± 4.7 | 27.8 ± 4.63 | 22.36 ± 3.32 | 11–27 (19) |
| Hemoglobin (g/dL) | 15.2 ± 0.98 | 12.35 ± 2.2 | 13.1 ± 1.61 | 14.4 ± 2.13 | 10–17 (13.1) |
| Hematocrit (%) | 48.3 ± 7.92 | 43.6 ± 17.8 | 40.57 ± 4.16 | 44.52 ± 2.17 | 39–49 (40.4) |
| Erythrocytes (×106/mm3) | 7.24 ± 1.06 | 7.67 ± 2.26 | 7.81 ± 0.88 | 7.92 ± 1.93 | 8.3 |
| Leukocytes (×100/mm3) | 3600 ± 903 * | 4242.6 ± 980 | 2750 ± 777 * | 5720 ± 684 | 5–12 (6.33) |
| Platelets (×106/μL) | 736 ± 1.414 | 689 ± 5.425 | 759 ± 19.143 | 710 ± 6.716 | 116 |
| PCT | 0.53 ± 0.07 | 0.49 ± 0.10 | 0.55 ± 0.29 | 0.55 ± 0.30 | -- |
| No. | RT (min) | PI | Compound Name | Biological Activity | Presence in Plants | DLMS |
|---|---|---|---|---|---|---|
| 1 | 6.18 | HMDB41635 | myricetin 3-arabinoside | Antioxidant, anticarcinogenic | Rhus spp. | 0.90 |
| 2 | 6.44 | CSID70398 | methyl gallate * | Antioxidant | Rhus spp. | −0.49 |
| 3 | 7.05 | HMDB38831 | epigallocatechin 3-cinnamate | Antioxidant, antibacterial | Ocotea porosa | 0.24 |
| 4 | 7.36 | CSID3525015 | 3,5-digalloylepicatechin | Antioxidant | -- | 1.52 |
| 5 | 8.27 | CHEBI:18152 | myricetin * | Antioxidant, antineoplastic | Rhus spp. | −0.04 |
| 6 | 8.47 | LMPK12112436 | myricitrin * | Antioxidant, antineoplastic | Rhus spp. | 0.78 |
| 7 | 8.52 | LMPK12110568 | quercetin 3-(2’’’-galloylglucosyl)-(1→2)-alpha-L-arabinofuranoside | Antioxidant | Euphorbia pachyrhiza | 1.24 |
| 8 | 8.52 | CHEBI18082 | 1,2,3,4,6-pentakis-O-galloyl-β-D-glucose | Antineoplastic | Rhus spp. | 0.35 |
| 9 | 8.60 | CHEBI:16243 | quercetin * | Antioxidant, anti-inflammatory, antineoplastic | Rhus spp. | 0.93 |
| 10 | 9.13 | CSID24784962 | 4-O-digalloyl-1,2,3,6-tetra-O-β-D-galloylglucose | Antibacterial, antineoplastic, antithrombotic | Rhus typhina | 0.35 |
| 11 | 9.45 | CHEBI:5715 | hibiscoquinone a | Antioxidant | Hibiscus spp. | −0.52 |
| 12 | 9.45 | LMPK12112171 | quercitrin * | Antioxidant, antineoplastic | Rhus spp. | 1.04 |
| 13 | 9.96 | LMPK12112447 | myricetin 3-(4’’-galloylrhamnoside) | Antioxidant | -- | 1.12 |
| 14 | 10.20 | LMPK12100067 | obtusaquinol | Antiparasitic, nitric oxide inhibitor | Dalbergia spp. | −0.03 |
| 15 | 10.94 | CSID35013429 | epifisetinidol-(4β→8)-catechin | Antibacterial, α-amylase and lipase inhibitor | Cotinus coggyria | 0.61 |
| 16 | 11.92 | LMPK12111566 | fisetin * | Antineoplastic, antioxidant, antiangiogenic | Rhus spp. | 0.76 |
| 17 | 12.54 | LMFA01010048 | margaric acid | Antioxidant, antifungal | Rhus typhina | −0.33 |
| 18 | 12.54 | LMFA01050146 | 12S-hydroxy-16-heptadecynoic acid | Anti-inflammatory | -- | −0.38 |
| 19 | 13.37 | CSID8034429 | (-)-pinellic acid | Metabolism, adjuvant activity | Pinelliae tuber | −1.08 |
| 20 | 13.37 | LMFA01030130 | 11,14-eicosadienoic acid | Antioxidant, anti-inflammatory | Rhus typhina | −0.08 |
| 21 | 13.9 | LMPK12040009 | amentoflavone | Antioxidant, anti-inflammatory | Rhus spp. | 0.51 |
| 22 | 15.94 | CSID10297786 | 3-(1,1-dimethylallyl)-8-(3,3-dimethylallyl) xanthyletin | -- | Ruta graveolens | −1.31 |
| 23 | 17.7 | CHEBI:31014 | lignocerate | Anti-inflammatory | Oleandra neriiformis | −0.33 |
| 24 | 21.28 | LMFA02000057 | 2R-hydroxy-9Z,12Z-octadecadienoic acid | Anti-allergic | Brassica campestris | −0.96 |
| Sample/Cell Line | SKOV-3 | OVCAR-3 | CACO-2 | BEAS-2B | CHO-K1 |
|---|---|---|---|---|---|
| Gallic acid | 50 (294) | 43 (253) | 25 (147) | 25 (147) | 100 (588) |
| Methyl gallate | >200 (1,086) | >200 (1,086) | 100 (543) | 200 (1,086) | 130 (706) |
| Myricetin | 166 (522) | 94 (295) | 62 (195) | 64 (201) | 33 (104) |
| Myricitrin | 197 (424) | 200 (431) | >200 (431) | 160 (344) | 94 (202) |
| Quercetin | >200 (662) | 200 (662) | 150 (496) | 189 (625) | 127 (420) |
| Quercitrin | >200 (446) | >200 (446) | 179 (399) | >200 (446) | >200 (446) |
| Fisetin | 200 (699) | 200 (699) | 100 (349) | 50 (175) | 46 (161) |
| Paclitaxel | 7 (8.20) | 8 (9.37) | 20 (23.42) | 8 (9.37) | 10 (11.71) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varela-Rodríguez, L.; Sánchez-Ramírez, B.; Saenz-Pardo-Reyes, E.; Ordaz-Ortiz, J.J.; Castellanos-Mijangos, R.D.; Hernández-Ramírez, V.I.; Cerda-García-Rojas, C.M.; González-Horta, C.; Talamás-Rohana, P. Antineoplastic Activity of Rhus trilobata Nutt. (Anacardiaceae) against Ovarian Cancer and Identification of Active Metabolites in This Pathology. Plants 2021, 10, 2074. https://doi.org/10.3390/plants10102074
Varela-Rodríguez L, Sánchez-Ramírez B, Saenz-Pardo-Reyes E, Ordaz-Ortiz JJ, Castellanos-Mijangos RD, Hernández-Ramírez VI, Cerda-García-Rojas CM, González-Horta C, Talamás-Rohana P. Antineoplastic Activity of Rhus trilobata Nutt. (Anacardiaceae) against Ovarian Cancer and Identification of Active Metabolites in This Pathology. Plants. 2021; 10(10):2074. https://doi.org/10.3390/plants10102074
Chicago/Turabian StyleVarela-Rodríguez, Luis, Blanca Sánchez-Ramírez, Erika Saenz-Pardo-Reyes, José Juan Ordaz-Ortiz, Rodrigo Daniel Castellanos-Mijangos, Verónica Ivonne Hernández-Ramírez, Carlos Martín Cerda-García-Rojas, Carmen González-Horta, and Patricia Talamás-Rohana. 2021. "Antineoplastic Activity of Rhus trilobata Nutt. (Anacardiaceae) against Ovarian Cancer and Identification of Active Metabolites in This Pathology" Plants 10, no. 10: 2074. https://doi.org/10.3390/plants10102074
APA StyleVarela-Rodríguez, L., Sánchez-Ramírez, B., Saenz-Pardo-Reyes, E., Ordaz-Ortiz, J. J., Castellanos-Mijangos, R. D., Hernández-Ramírez, V. I., Cerda-García-Rojas, C. M., González-Horta, C., & Talamás-Rohana, P. (2021). Antineoplastic Activity of Rhus trilobata Nutt. (Anacardiaceae) against Ovarian Cancer and Identification of Active Metabolites in This Pathology. Plants, 10(10), 2074. https://doi.org/10.3390/plants10102074