Population Structure and Genetic Diversity of Cucurbita moschata Based on Genome-Wide High-Quality SNPs
Abstract
1. Introduction
2. Results
2.1. The Genome-Wide SNP Identification among 20 Chromosomes in C. moschata
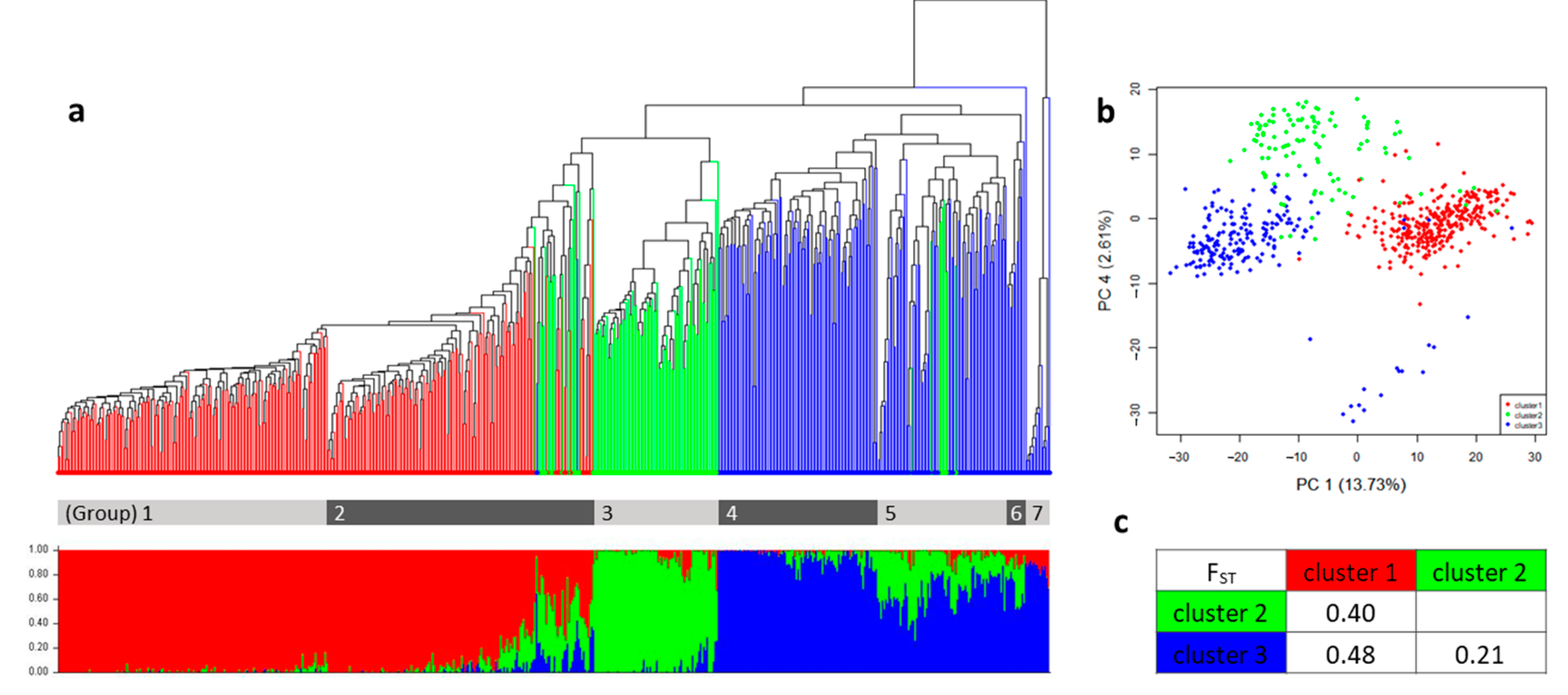
2.2. The Population Structure and the Genetic Relatedness of the Germplasm Collection
2.3. The Genetic Variations of the Germplasm Collection
2.4. The Construction of the C. moschata Core Collection and the Validation with the Phenotypic Variation
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Phenotypic Evaluation
4.2. The DNA Extraction and the SNP Set Construction Using the GBS Method
4.3. The Genetic Analyses
4.4. The Core Collection Selection and the Evaluation
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Eguiarte, L.E.; Hernández-Rosales, H.S.; Barrera-Redondo, J.; Castellanos-Morales, G.; Paredes-Torres, L.M.; Sánchez-de la Vega, G.; Ruiz-Mondragón, K.Y.; Vázquez-Lobo, A.; Montes-Hernández, S.; Aguirre-Planter, E.; et al. Domestication, diversity, genetic and genomic resources of Mexico: The case of pumpkins. TIP Rev. Espec. Cienc. Químico-Biológicas 2018, 21, 85. [Google Scholar] [CrossRef]
- Chomicki, G.; Schaefer, H.; Renner, S.S. Origin and domestication of Cucurbitaceae crops: Insights from phylogenies, genomics and archaeology. New Phytol. 2019, 226, 1240–1255. [Google Scholar] [CrossRef] [PubMed]
- Ferriol, M.; Pico, B. Pumpkin and winter squash. In Handbook of Plant Breeding, Vegetables I; Prohens, J., Nuez, F., Eds.; Springer: New York, NY, USA, 2008; pp. 317–349. [Google Scholar]
- Gong, L.; Paris, H.S.; Stift, G.; Pachner, M.; Vollmann, J.; Lelley, T. Genetic relationships and evolution in Cucurbita as viewed with simple sequence repeat polymorphisms: The centrality of C. Okeechobeensis. Genet. Resour. Crop. Evol. 2013, 60, 1531–1546. [Google Scholar] [CrossRef]
- Paris, H.S. Genetic resources of pumpkins and squash Cucurbita spp. In Genetics and Genomics of Cucurbitaceae; Grumet, R., Katzir, N., Garcia-Mas, J., Eds.; Springer: Cham, Switzerland, 2016; Volume 20, pp. 111–154. [Google Scholar]
- Hernández-Rosales, H.S.; Castellanos-Morales, G.; Sánchez-de la Vega, G.; Aguirre-Planter, E.; Montes-Hernández, S.; Lira-Saade, R.; Eguiarte, L.E. Phylogeographic and population genetic analyses of Cucurbita moschata reveal divergence of two mitochondrial lineages linked to an elevational gradient. Am. J. Bot. 2020, 107, 510–525. [Google Scholar] [CrossRef]
- Zizumbo-Villarreal, D.; Colunga-GarcíaMarín, P.; Flores-Silva, A. Pre-Columbian food system in West Mesoamerica. In Ethnobotany of Mexico: Interactions of People and Plants in Mesoamerica; Lira, R., Casas, A., Blancas, J., Eds.; Springer: New York, NY, USA, 2016; pp. 67–82. [Google Scholar]
- Aslam, W.; Noor, R.S.; Hussain, F.; Ameen, M.; Ullah, S.; Chen, H. Evaluating Morphological Growth, Yield, and Postharvest Fruit Quality of Cucumber (Cucumis Sativus L.) Grafted on Cucurbitaceous Rootstocks. Agriculture 2020, 10, 101. [Google Scholar] [CrossRef]
- Lee, J.M. Cultivation of Grafted Vegetables. I. Current Status, Grafting Methods, and Benefits. HortScience 1994, 29, 235–239. [Google Scholar] [CrossRef]
- Sakata, Y.; Sugiyama, M.; Ohara, T.; Morishita, M. Influence of Rootstocks on the Resistance of Grafted Cucumber (Cucumis sativus L.) Scions to Powdery Mildew (Podosphaera xanthii U. Braun & N. Shishkoff). J. Jpn. Soc. Hortic. Sci. 2006, 75, 135–140. [Google Scholar] [CrossRef][Green Version]
- Sánchez-de La Vega, G.; Castellanos-Morales, G.; Gámez, N.; Hernández-Rosales, H.S.; Vázquez-Lobo, A.; Aguirre-Planter, E.; Jaramillo-Correa, J.P.; Montes-Hernández, S.; Lira-Saade, R.; Eguiarte, L.E. Genetic Resources in the “Calabaza Pipiana” Squash (Cucurbita argyrosperma) in Mexico: Genetic Diversity, Genetic Differentiation and Distribution Models. Front. Plant. Sci. 2018, 9, 1–18. [Google Scholar] [CrossRef]
- Castellanos-Morales, G.; Ruiz-Mondragón, K.Y.; Hernández-Rosales, H.S.; Sánchez-De La Vega, G.; Gámez, N.; Aguirre-Planter, E.; Montes-Hernández, S.; Lira-Saade, R.; Eguiarte, L.E. Tracing back the origin of pumpkins (Cucurbita pepo ssp. pepo L.) in Mexico. Proc. R. Soc. B Biol. Sci. 2019, 286. [Google Scholar] [CrossRef]
- Yunli, W.; Yangyang, W.; Wenlong, X.; Chaojie, W.; Chongshi, C.; Shuping, Q. Genetic diversity of pumpkin based on morphological and SSR markers. Pak. J. Bot. 2020, 52, 477–487. [Google Scholar] [CrossRef]
- Barboza, N.; Albertazzi, F.J.; Sibaja-Cordero, J.A.; Mora-Umaña, F.; Astorga, C.; Ramírez, P. Analysis of genetic diversity of Cucurbita moschata (D.) germplasm accessions from Mesoamerica revealed by PCR SSCP and chloroplast sequence data. Sci. Hortic. 2012, 134, 60–71. [Google Scholar] [CrossRef]
- Gomes, R.S.; Machado, R.; De Almeida, C.F.; Chagas, R.R.; De Oliveira, R.L.; Delazari, F.T.; Da Silva, D.J.H. Brazilian germplasm of winter squash (Cucurbita moschata D.) displays vast genetic variability, allowing identification of promising genotypes for agro-morphological traits. PLoS ONE 2020, 15, e0230546. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-Y.; Ro, N.-Y.; Patil, A.; Lee, J.-H.; Kwon, J.-K.; Kang, B.-C. Uncovering Candidate Genes Controlling Major Fruit-Related Traits in Pepper via Genotype-by-Sequencing Based QTL Mapping and Genome-Wide Association Study. Front. Plant. Sci. 2020, 11, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-Y.; Ro, N.-Y.; Jeong, H.-J.; Kwon, J.-K.; Jo, J.; Ha, Y.; Jung, A.; Han, J.-W.; Venkatesh, J.; Kang, B.-C. Genetic diversity and population structure analysis to construct a core collection from a large Capsicum germplasm. BMC Genet. 2016, 17, 142. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.D. Eastern North America as an independent center of plant domestication. Proc. Natl. Acad. Sci. USA 2006, 103, 12223–12228. [Google Scholar] [CrossRef]
- Mapes, C.; Basurto, F. Biodiversity and edible plants of Mexico. In Ethnobotany of Mexico: Interactions of People and Plants in Mesoamerica; Lira, R., Casas, A., Blancas, J., Eds.; Springer: New York, NY, USA, 2016; pp. 83–131. [Google Scholar]
- Kates, H.R.; Soltis, P.S.; Soltis, D.E. Evolutionary and domestication history of Cucurbita (pumpkin and squash) species inferred from 44 nuclear loci. Mol. Phylogenet. Evol. 2017, 111, 98–109. [Google Scholar] [CrossRef]
- Han, K.; Lee, H.Y.; Ro, N.Y.; Hur, O.S.; Lee, J.H.; Kwon, J.K.; Kang, B.C. QTL mapping and GWAS reveal candidate genes controlling capsaicinoid content in Capsicum. Plant. Biotechnol. J. 2018, 16, 1546–1558. [Google Scholar] [CrossRef]
- Sun, H.; Wu, S.; Zhang, G.; Jiao, C.; Guo, S.; Ren, Y.; Zhang, J.; Zhang, H.; Gong, G.; Jia, Z.; et al. Karyotype Stability and Unbiased Fractionation in the Paleo-Allotetraploid Cucurbita Genomes. Mol. Plant. 2017, 10, 1293–1306. [Google Scholar] [CrossRef]
- Depristo, M.A.; Banks, E.; Poplin, R.; Garimella, K.V.; Maguire, J.R.; Hartl, C.; Philippakis, A.A.; Del Angel, G.; Rivas, M.A.; Hanna, M.; et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat. Genet. 2011, 43, 491–501. [Google Scholar] [CrossRef]
- Pritchard, J.K.; Stephens, M.; Donnelly, P. Inference of population structure using multilocus genotype data. Genetics 2000, 155, 945–959. [Google Scholar] [CrossRef]
- Evanno, G.; Regnaut, S.; Goudet, J. Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study. Mol. Ecol. 2005, 14, 2611–2620. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.W.; Chung, H.K.; Cho, G.T.; Ma, K.H.; Chandrabalan, D.; Gwag, J.G.; Kim, T.S.; Cho, E.G.; Park, Y.J. PowerCore: A program applying the advanced M strategy with a heuristic search for establishing core sets. Bioinformatics 2007, 23, 2155–2162. [Google Scholar] [CrossRef] [PubMed]

| Continent | Cluster_1 | Cluster_2 | Cluster_3 |
|---|---|---|---|
| Africa | 1 | 41 | 4 |
| America | 3 | 5 | 162 |
| Asia | 305 | 57 | 24 |
| Europe | 0 | 1 | 7 |
| Total | 309 | 104 | 197 |
| Cluster | Number of Accessions | Na | Ne | HO | HE | I | F |
|---|---|---|---|---|---|---|---|
| 1 | 309 | 1.985 | 1.311 | 0.045 | 0.202 | 0.328 | 0.756 |
| 2 | 104 | 1.994 | 1.549 | 0.094 | 0.327 | 0.494 | 0.701 |
| 3 | 182 | 1.998 | 1.526 | 0.068 | 0.315 | 0.48 | 0.777 |
| Total | 595 | 1.992 | 1.462 | 0.069 | 0.281 | 0.434 | 0.745 |
| Source of Variation | Degrees of Freedom (df) | Sum of Squares | Mean Sum of Squares | Estimated Variance | Percentage of Variation (%) |
|---|---|---|---|---|---|
| Among Pops | 2 | 80,683 | 40,342 | 212 | 9.50% |
| Within Pops | 592 | 1,202,784 | 2032 | 2032 | 90.50% |
| Total | 594 | 1,283,468 | 2244 | 100% |
| Traits | Type | Unit | 2018 | 2019 | 2020 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Min | Max | Mean | SE | Min | Max | Mean | SE | Min | Max | Mean | SE | |||
| Cotyledon length | W | mm | 16.1 | 78.7 | 55.3 | 0.3 | 13.7 | 65.0 | 46.4 | 0.8 | 28.3 | 95.6 | 69.3 | 0.7 |
| CC | mm | 27.8 | 64.5 | 52.3 | 0.9 | 13.7 | 53.7 | 42.3 | 3.0 | 48.0 | 85.0 | 62.7 | 2.1 | |
| Cotyledon width | W | mm | 8.8 | 53.1 | 36.3 | 0.2 | 10.8 | 50.5 | 31.3 | 0.5 | 23.7 | 58.5 | 42.5 | 0.4 |
| CC | mm | 23.4 | 45.5 | 34.3 | 0.6 | 15.6 | 35.8 | 30.1 | 1.6 | 31.3 | 49.0 | 40.0 | 1.0 | |
| Hypocotyl length | W | mm | - | - | - | - | 15.5 | 61.7 | 35.2 | 0.7 | 22.0 | 94.4 | 54.1 | 0.9 |
| CC | mm | - | - | - | - | 23.6 | 48.0 | 36.1 | 1.8 | 22.0 | 74.2 | 45.9 | 2.3 | |
| Internode length | W | cm | 2.4 | 24.8 | 13.5 | 0.1 | 6.8 | 26.8 | 15.9 | 0.2 | 4.0 | 30.3 | 17.4 | 0.2 |
| CC | cm | 2.4 | 24.4 | 14.2 | 0.4 | 7.0 | 26.8 | 15.9 | 0.6 | 11.2 | 30.3 | 18.7 | 0.8 | |
| No. of female flower among 10 nodes | W | ea | 1.0 | 5.0 | 1.8 | 0.1 | 0.2 | 5.5 | 1.1 | 0.1 | 0.2 | 3.8 | 0.7 | 0.0 |
| CC | ea | 1.0 | 4.0 | 1.8 | 0.2 | 0.4 | 5.5 | 1.7 | 0.5 | 0.2 | 1.0 | 0.6 | 0.1 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.-Y.; Jang, S.; Yu, C.-R.; Kang, B.-C.; Chin, J.-H.; Song, K. Population Structure and Genetic Diversity of Cucurbita moschata Based on Genome-Wide High-Quality SNPs. Plants 2021, 10, 56. https://doi.org/10.3390/plants10010056
Lee H-Y, Jang S, Yu C-R, Kang B-C, Chin J-H, Song K. Population Structure and Genetic Diversity of Cucurbita moschata Based on Genome-Wide High-Quality SNPs. Plants. 2021; 10(1):56. https://doi.org/10.3390/plants10010056
Chicago/Turabian StyleLee, Hea-Young, Siyoung Jang, Chea-Rin Yu, Byoung-Cheorl Kang, Joong-Hyoun Chin, and Kihwan Song. 2021. "Population Structure and Genetic Diversity of Cucurbita moschata Based on Genome-Wide High-Quality SNPs" Plants 10, no. 1: 56. https://doi.org/10.3390/plants10010056
APA StyleLee, H.-Y., Jang, S., Yu, C.-R., Kang, B.-C., Chin, J.-H., & Song, K. (2021). Population Structure and Genetic Diversity of Cucurbita moschata Based on Genome-Wide High-Quality SNPs. Plants, 10(1), 56. https://doi.org/10.3390/plants10010056





