Short-Term Exposure to High Atmospheric Vapor Pressure Deficit (VPD) Severely Impacts Durum Wheat Carbon and Nitrogen Metabolism in the Absence of Edaphic Water Stress
Abstract
1. Introduction
2. Results
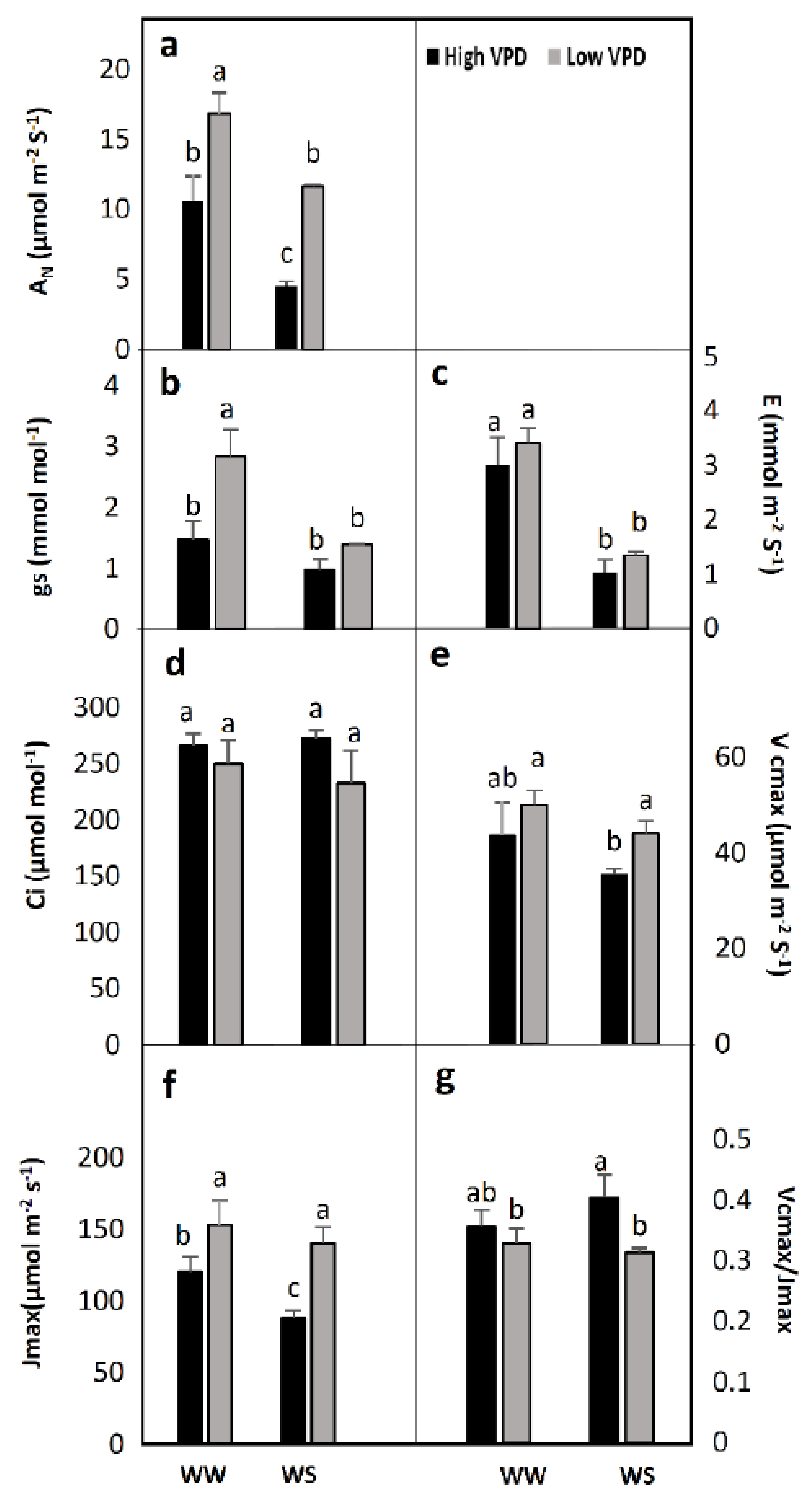
2.1. Gas Exchange Measurements
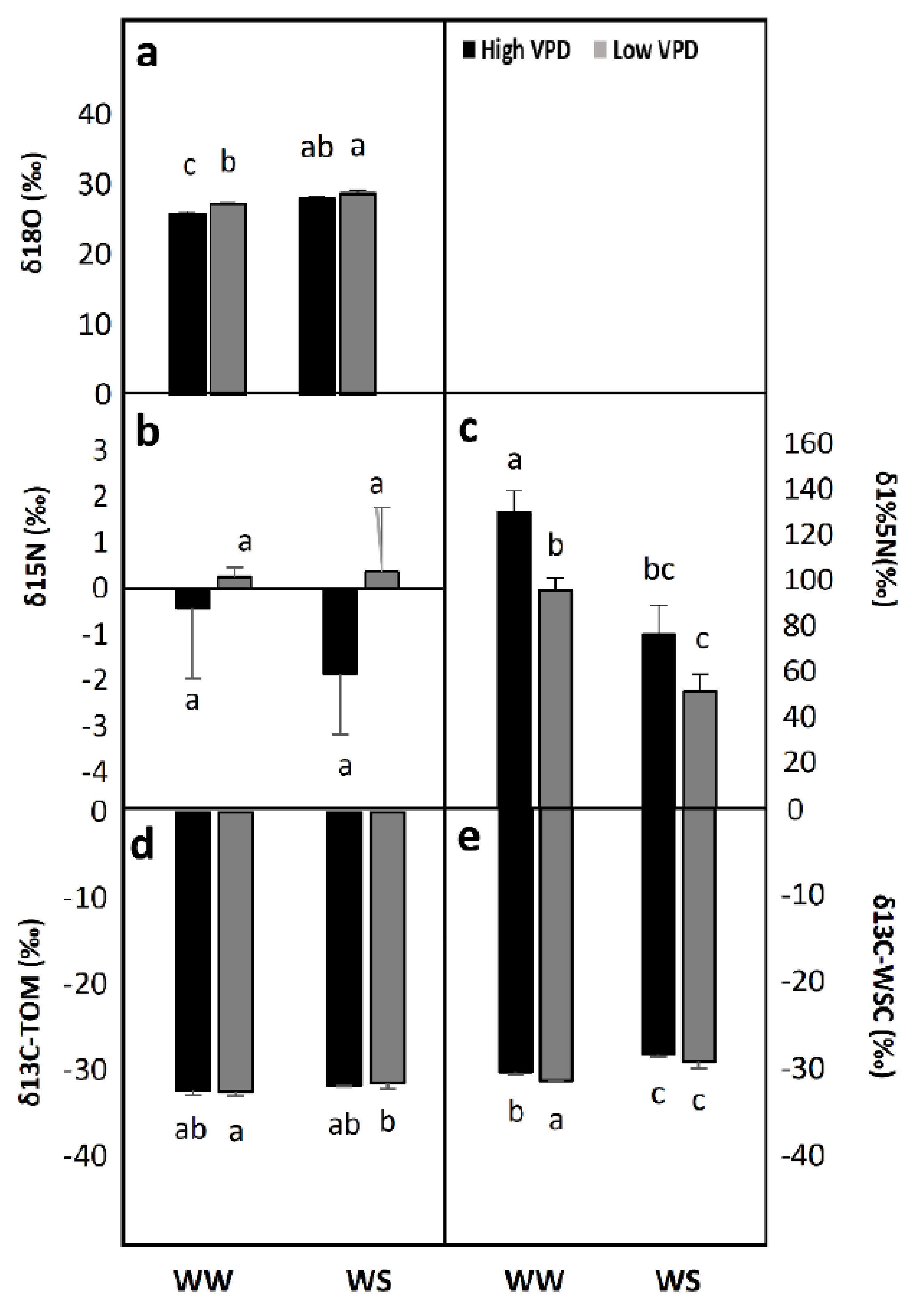
2.2. Stable Isotope Signatures
2.2.1. 18O Isotopic Composition, δ18O
2.2.2. 15N Isotopic Composition, δ15N
2.2.3. 13C Isotopic Composition, δ13C
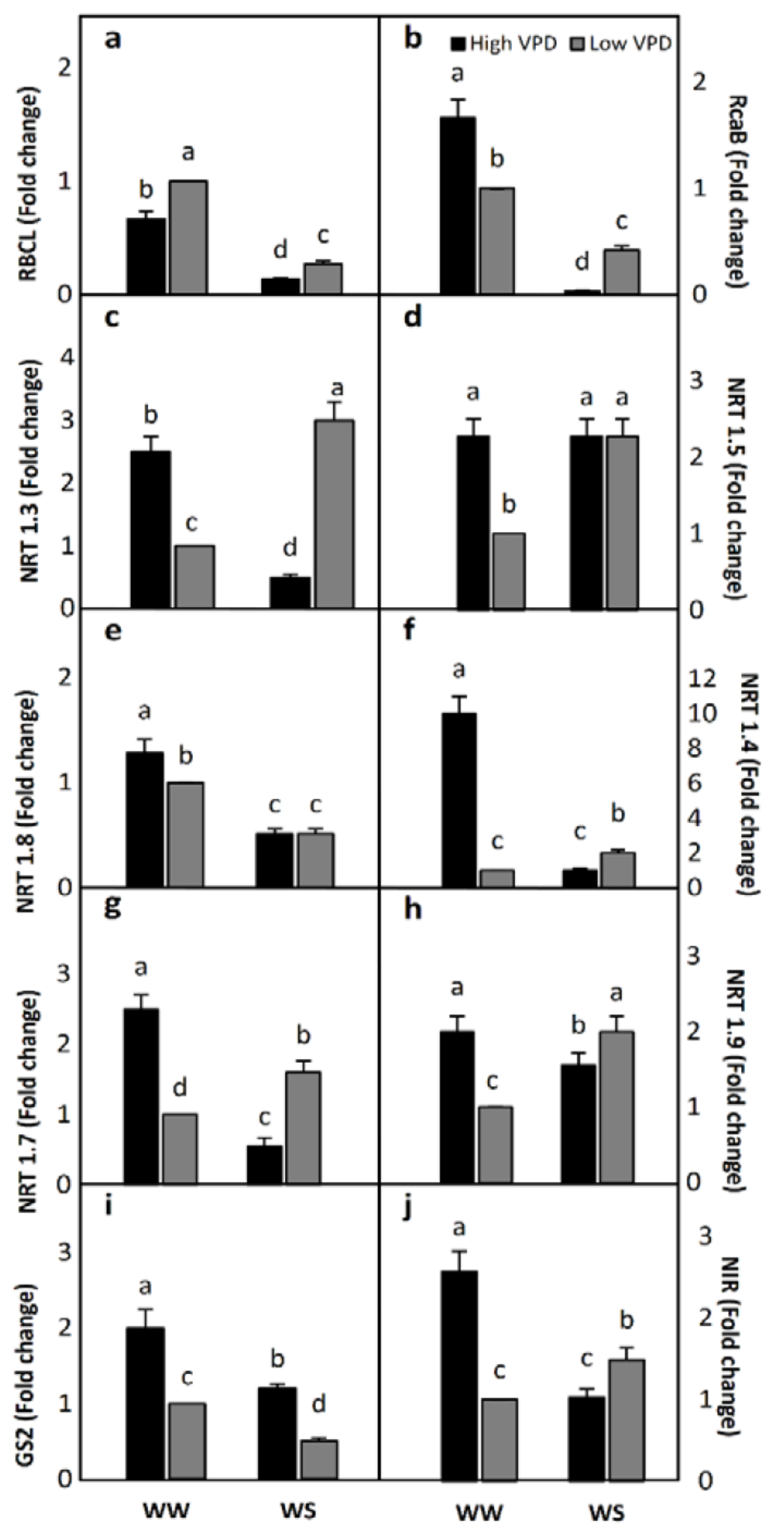
2.3. Gene Expression
2.3.1. RuBisCO Synthesis and RuBisCO Activase Genes
2.3.2. Nitrogen Assimilation and Transport Genes
Nitrate Transport Genes
2.3.3. Nitrogen Assimilation
2.4. Amino Acid Concentrations
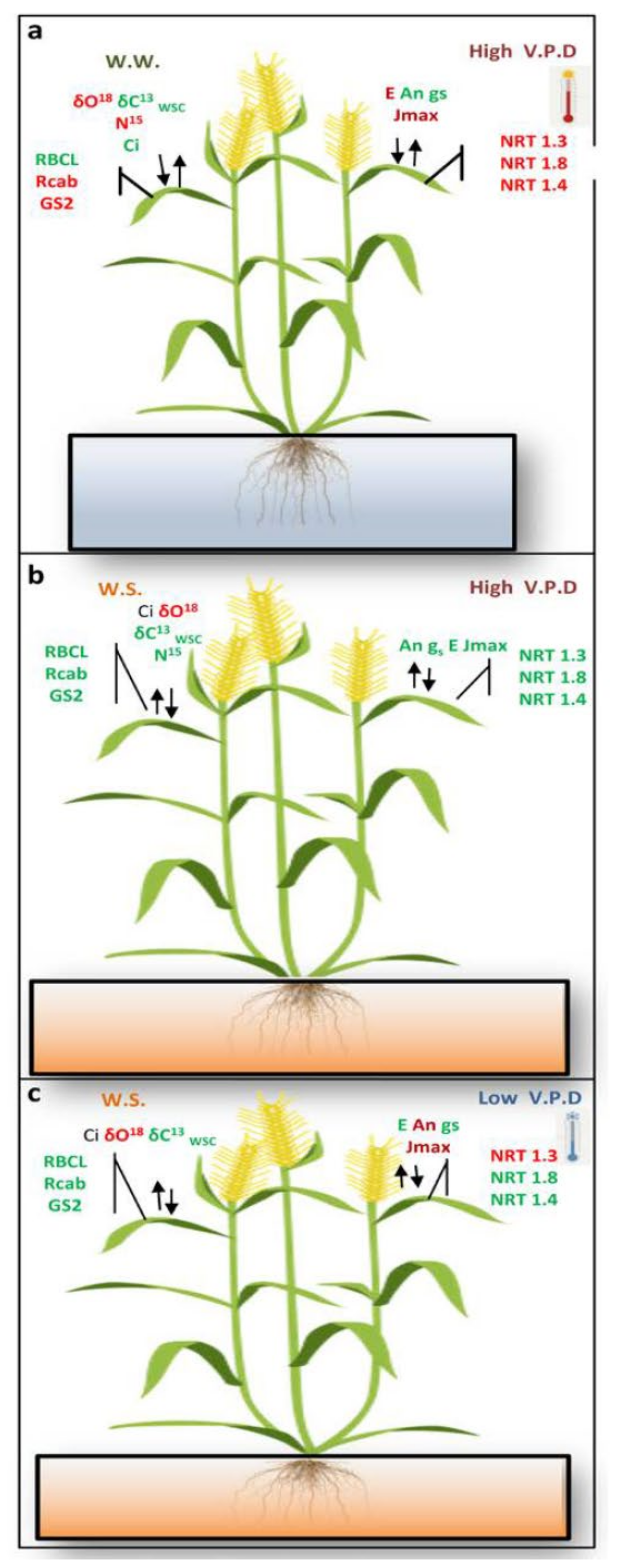
3. Discussion
4. Materials and Methods
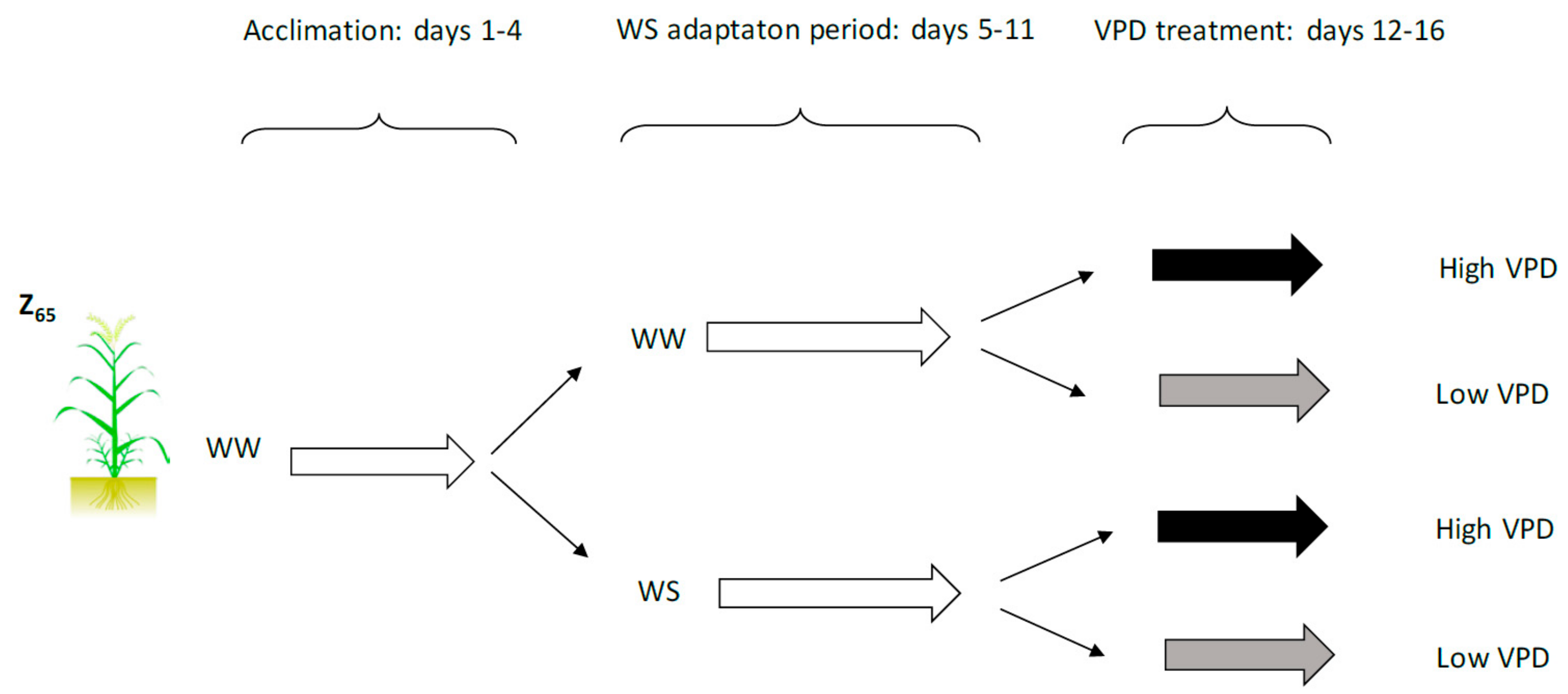
4.1. Experimental Setup
4.2. Gas Exchange
4.3. Nitrogen Labeling
4.4. Amino Acid Profile and Content
4.5. N, C, and O Isotope Composition Analyses (δ 15N, δ 13C and δ 18O)
4.6. Gene Expression
4.6.1. RNA Isolation
4.6.2. Quantitative Real-Time Reverse Transcription Polymerase Chain Reaction (RT-qPCR)
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- IPCC. 2018: Summary for Policymakers. In Global Warming of 1.5 °C. An IPCC Special Report on the Impacts of Global Warming of 1.5 °C above Pre-Industrial Levels and Related Global Greenhouse Gas Emission Pathways, in the Context of Strengthening the Global Response to the Threat of Climate Change, Sustainable Development, and Efforts to Eradicate Poverty; Masson-Delmotte, V., Zhai, P., Pörtner, H.-O., Roberts, D., Skea, J., Shukla, P.R., Pirani, A., Moufouma-Okia, W., Péan, C., Pidcock, R., et al., Eds.; IPCC: Geneva, Switzerland, 2018. [Google Scholar]
- Yuan, W.; Zheng, Y.; Piao, S.; Ciais, P.; Lombardozzi, D.; Wang, Y.; Ryu, Y.; Chen, G.; Dong, W.; Hu, Z.; et al. Increased atmospheric vapor pressure deficit reduces global vegetation growth. Sci. Adv. 2019, 5, 1–12. [Google Scholar] [CrossRef]
- Lobell, D.B.; Roberts, M.J.; Schlenker, W.; Braun, N.; Little, B.B.; Rejesus, R.M.; Hammer, G.L. Greater Sensitivity to Drought Accompanies Maize Yield Increase in the US Midwest. Science 2014, 344, 516–519. [Google Scholar] [CrossRef] [PubMed]
- Moore, F.C.; Lobell, D.B. The fingerprint of climate trends on European crop yields. Proc. Natl. Acad. Sci. USA 2015, 112, 2670–2675. [Google Scholar] [CrossRef] [PubMed]
- Drobinski, P.; Da Silva, N.; Bastin, S.; Mailler, S.; Muller, C.; Ahrens, B.; Christensen, O.B.; Lionello, P. How warmer and drier will the Mediterranean region be at the end of the twenty-first century? Reg. Environ. Chang. 2020, 20, ar78. [Google Scholar] [CrossRef]
- Savary, S.; Akter, S.; Almekinders, C.; Harris, J.; Korsten, L.; Roetter, R.; Waddington, S.; Watson, D. Mapping disruption and resilience mechanisms in food systems. Food Secur. 2020, 12, 695–717. [Google Scholar] [CrossRef]
- Lobell, D.B.; Schlenker, W.; Roberts, J.C. Climate Trends and Global Crop Production Since 1980. Science 2019, 333, 616–620. [Google Scholar] [CrossRef]
- Schlenker, W.; Lobell, D.B. Robust negative impacts of climate change on African agriculture. Environ. Res. Lett. 2010, 5, 1–8. [Google Scholar] [CrossRef]
- Sagardoy, R.; Vázquez-Reina, S.; Florez-Sarasa, I.D.; Albacete, A.; Ribas-Carbó, M.; Flexas, J.; Abadía, J.; Morales, F. Stomatal and mesophyll conductances to CO2 are the main limitations to photosynthesis in sugar beet (Beta vulgaris) plants grown with excess Zn. New Phytol. 2010, 187, 145–158. [Google Scholar] [CrossRef]
- Duursma, R.A.; Barton, C.V.M.; Lin, Y.S.; Medlyn, B.E.; Eamus, D.; Tissue, D.T.; Ellsworth, D.S.; McMurtire, R.E. The peaked response of transpiration rate to vapour pressure defict in field conditions can be explained by the temperature optimum of photosynthesis. Agric. Forest Meteorol. 2014, 189–190, 2–10. [Google Scholar] [CrossRef]
- Mendes, K.R.; Marenco, R.A. Stomatal opening in response to the simultaneous increase in vapor pressure deficit and temperature over a 24-h period under constant light in a tropical rainforest of the central Amazon. Theor. Exp. Plant Physiol. 2017, 2, 187–194. [Google Scholar] [CrossRef]
- Rashid, A.M.; Anderson, M.N.; Wollenweber, B.; Korupa, K.; Zhang, X.; Eivind, J. Acclimation to higher VPD and temperature minimized negative effects on assimilation and grain yield of wheat. Agric. For. Meteorol. 2018, 248, 119–129. [Google Scholar] [CrossRef]
- Shibuya, T.; Kano, K.; Endo, R.; Kitaya, Y. Effects of the interaction between vapor-pressure deficit and salinity on growth and photosynthesis of Cucumis sativus seedlings under different CO2 concentrations. Hort. J. 2018, 56, 893–900. [Google Scholar] [CrossRef]
- Jauregui, I.; Rothwel, S.A.; Taylor, S.A.; Parry, M.A.J.; Carmo-Silva, E.; Dodd, I.C. Whole plant chamber to examine sensitivity of cereal gas exchange to changes in evaporative demand. Plant Methods 2018, 14, ar97. [Google Scholar] [CrossRef] [PubMed]
- Lihavainen, J.; Ahonen, V.; Keski-saari, S.; Kontunen-soppela, S.; Oksanen, E.; Keinänen, M. Low vapour pressure deficit affects nitrogen nutrition and foliar metabolites in silver birch. J. Exp. Bot. 2016, 67, 4353–4365. [Google Scholar] [CrossRef]
- Duan, J.; Tian, H.; Gao, Y. Expression of nitrogen transporter genes in roots of winter wheat (Triticum aestivum L.) response to soil drought with contrasting nitrogen supplies. Crop Pasture Sci. 2016, 67, 128–136. [Google Scholar] [CrossRef]
- Thong, W.A.; Imai, R.; Tabata, S.; Shigenobu, K.; Yamada, Y.M.; Hasebe, M.; Sawa, S.; Motose, H.; Takahashi, T. Polyamine resistance is increased by mutations in a nitrate transporter gene NRT1.3 (AtNPF6.4) in Arabidopsis thaliana. Front. Plant Sci. 2016, 7, 1–10. [Google Scholar] [CrossRef]
- Wang, Y.-Y.; Tsay, Y.-F. Arabidopsis Nitrate Transporter NRT1.9 Is Important in Phloem Nitrate Transport. Plant Cell 2011, 23, 1945–1957. [Google Scholar] [CrossRef]
- Dechorgnat, J.; Nguyen, C.T.; Armengaud, P.; Jossier, M.; Diatloff, E.; Filleur, S.; Vedele, F.D. From the soil to the seeds: The long journey of nitrate in plants. J. Exp. Bot. 2011, 62, 1349–1359. [Google Scholar] [CrossRef]
- Chaves, M.M.; Maroco, J.P.; Pereira, J.S. Understanding plant responses to drought from genes to the whole plant. Funct. Plant Biol. 2003, 30, 239–264. [Google Scholar] [CrossRef]
- Yan, W.; Zhong, Y.; Shangguan, Z. Evaluation of physiological traits of summer maize under drought stress. Acta Agric. Scand. Sect. B Soil Plant Sci. 2016, 66, 133–140. [Google Scholar] [CrossRef]
- Jamieson, P.D.; Martin, R.J.; Francis, G.S. Drought influences on grain yield of barley, wheat, and maize. N. Z. J. Crop Hortic. Sci. 1995, 23, 55–66. [Google Scholar] [CrossRef]
- Liu, X.; Wang, X.L.; Wang, X.Y.; Gao, J.; Luo, N.; Meng, Q.; Wang, P. Dissecting the critical stage in the response of maize kernel set to individual and combined drought and heat stress around flowering. Environ. Exp. Bot. 2020, 179, ar104213. [Google Scholar] [CrossRef]
- Cai, F.; Zhang, Y.; Mi, N.; Ming, H.; Zhang, S.; Zhang, H.; Zhao, X. Maize (Zea mays L.) physiological responses to drought and rewatering, and the associations with water stress degree. Agric. Water Manag. 2020, 241, ar106379. [Google Scholar] [CrossRef]
- Yang, Z.; Sinclairb, T.R.; Zhuc, M.; Messinac, C.D.; Cooperc, M.; Hammerd, G.L. Temperature effect on transpiration response of maize plants to vapour pressure deficit. Environ. Exp. Bot. 2012, 78, 157–162. [Google Scholar] [CrossRef]
- Verma, V.M.J.; Foulkes, A.J.; Bradley, W.R.S.; Caligari, P.D.S.; Snape, J.W. Mapping quantitative trait loci for flag leaf senescence as a yield determinant in winter wheat under optimal and drought-stressed environments. Euphytica 2004, 135, 255–263. [Google Scholar] [CrossRef]
- Rodriguez, D.; Aires, B. Effects of phosphorus and drought stresses on DW and Pi allocation in wheat. J. Plant Nutr. 1995, 18, 2501–2517. [Google Scholar] [CrossRef]
- Barber, S.A. A diffusion and mass-flow concept of soil nutrient availability. Soil Sci. 1962, 93, 39–49. [Google Scholar] [CrossRef]
- Pettigrew, W.T.L.; Hesketh, J.D.; Peters, D.B.; Woolley, J.T. A vapor pressure deficit on crop canopy photosynthesis. Photosynth. Res. 1990, 24, 27–34. [Google Scholar] [CrossRef]
- Shirke, P.A.; Pathre, U.V. Influence of leaf-to-air vapour pressure deficit (VPD) on the biochemistry and physiology of photosynthesis in Prosopis juliflora. J. Exp. Bot. 2004, 55, 2111–2120. [Google Scholar] [CrossRef]
- Ohsumi, A.; Hamasaki, A.; Nakagawa, H.; Homma, K.; Horie, T.; Shiraiwa, T. Response of Leaf Photosynthesis to Vapor Pressure Difference in Rice (Oryza sativa L.) Varieties in Relation to Stomatal and Leaf Internal Conductance. Plant Prod. Sci. 2015, 11, 184–191. [Google Scholar] [CrossRef]
- Domingues, T.F.; Martinelli, V.; Ehleringer, J.R. Plant Ecology & Diversity Seasonal patterns of leaf-level photosynthetic gas exchange in an eastern Amazonian rain forest. Plant Ecol. Divers. 2014, 7, 189–203. [Google Scholar] [CrossRef]
- Duursma, R.; Blackman, A.C.J.; Loppez, R.; StPaul, N.K.M.; Cochard, H.; Medlyn, B.E. On the minimum leaf conductance: Its role in models of plant water use, and ecological and environmental controls. New Phytol. 2018, 221, 693–705. [Google Scholar] [CrossRef]
- Onoda, Y.; Hikosaka, K.; Hirose, T. Seasonal change in the balance between capacities of RuBP carboxylation and RuBP regeneration affects CO2 response of photosynthesis in Polygonum cuspidatum. J. Exp. Bot. 2005, 56, 755–763. [Google Scholar] [CrossRef]
- Migge, A.; Bork, C.; Hell, R.; Becker, T.W. Negative regulation of nitrate reductase gene expression by glutamine or asparagine accumulating in leaves of sulfur-deprived tobacco. Planta 2000, 211, 587–595. [Google Scholar] [CrossRef]
- Faure, J.D.; Meyer, C.; Caboche, M. Nitrate assimilation: Nitrate and nitrite reductases. In Nitrogen Assimilation By Plants; MorotGaudry, J.F., Ed.; Science Publishers Inc.: Enfield, NH, USA, 2001; pp. 33–52. [Google Scholar]
- Farquhar, G.D. Feedforward Responses of Stomata to Humidity. Aust. J. Plant Physiol. 1978, 5, 787–800. [Google Scholar] [CrossRef]
- Monteith, J.L. A reinterpretation of stomatal responses to humidity. Plant Cell Environ. 1995, 18, 357–364. [Google Scholar] [CrossRef]
- Farquhar, G.D.; Sharkey, T.D. Stomatal conductance and photosynthesis. Annu. Rev. Plant. Physiol 1982, 33, 317–345. [Google Scholar] [CrossRef]
- Zargara, S.M.; Guptab, V.; Nazira, M.; Mahajanb, R.; Malikc, F.A.; Sofid, N.R.; Shikaria, A.B.; Salgotra, R.K. Impact of drought on photosynthesis: Molecular perspective. Plant Gene 2017, 11, 154–159. [Google Scholar] [CrossRef]
- El-Sharkaway, M.A.; Cock, J.H. Water Use Efficiency of Cassava. I. Effects of Air Humidity and Water Stress on Stomatal Conductance and Gas Exchange. Crop Sci. 1984, 24, 497–502. [Google Scholar] [CrossRef]
- Cornic, G.; Massacci, A. Leaf Photosynthesis Under Drought Stress. In Photosynthesis and the Environment. Advances In Photosynthesis and Respiration; Baker, N.R., Sharkey, T.D., Eaton-Rye, J.J., Eds.; Springer: Dordrecht, The Netherlands, 1996; Volume 5, pp. 347–366. [Google Scholar] [CrossRef]
- Tezara, W.; Mitchell, V.J.; Driscoll, S.D.; Lawlor, D.W. Water stress inhibits plant photosynthesis by decreasing coupling factor and ATP. Nature 1999, 401, 914–917. [Google Scholar] [CrossRef]
- Sharkey, T.D.; Bernacchi, C.J.; Farquhar, G.D.; Singsaas, E.L. Fitting Photosynthetic carbon dioxide response curves for C3 leaves. Plant Cell Environ. 2007, 30, 1035–1040. [Google Scholar] [CrossRef]
- Patane, C. Leaf Area Index, Leaf Transpiration and Stomatal Conductance as Affected by Soil Water Deficit and VPD in Processing Tomato in Semi Arid Mediterranean Climate. J. Agron. Crop Sci. 2011, 197, 165–176. [Google Scholar] [CrossRef]
- Li, J.-Y.; Fu, Y.-L.; Pike, S.M.; Bao, J.; Tian, W.; Zhang, Y.; Chen, C.-Z.; Zhang, Y.; Li, H.-M.; Huang, J.; et al. The Arabidopsis Nitrate Transporter NRT1.8 Functions in Nitrate Removal from the Xylem Sap and Mediates Cadmium Tolerance. Plant Cell 2010, 22, 1633–1646. [Google Scholar] [CrossRef]
- Vendruscolo, E.C.G.; Schusterb, I.; Pileggic, M.; Scapimd, C.A.; Molinarie, H.B.C.; Marure, C.J.; Vieirae, L.G.E. Stress-induced synthesis of proline confers tolerance to water deficit in transgenic wheat. J. Plant Physiol. 2009, 164, 1367–1376. [Google Scholar] [CrossRef]
- Delauney, A.J.; Verma, D.P.S. Proline biosynthesis and osmoregulation in plants. Plant J. 1993, 4, 215–223. [Google Scholar] [CrossRef]
- Zulfiqar, F.; Akram, N.A.; Ashraf, M. Osmoprotection in plants under abiotic stresses: New insights into a classical phenomenon. Planta 2020, 251, ar3. [Google Scholar] [CrossRef]
- Cramer, G.R. Abiotic stress and plant responses from the whole vine to the genes. Aust. J. Grape Wine Res. 2010, 16, 86–93. [Google Scholar] [CrossRef]
- McDonald, K.C.; Zimmermann, R.; Kimball, J.S. Diurnal and Spatial Variation of Xylem Dielectric Constant in Norway Spruce (Picea abies [L.] Karst.) as Related to Microclimate, Xylem Sap Flow, and Xylem Chemistry. IEEE Trans. Geosci. Remote Sens. 2002, 40, 2063–2083. [Google Scholar] [CrossRef]
- Erice, G.; Sanz-Sáez, A.; González-Torralba, J.; Méndez-Espinoza, A.M.; Urretavizcaya, I.; Nieto, M.T.; Serret, M.D.; Araus, J.L.; Irigoyen, J.J.; Aranjuelo, I. Impact of elevated CO2 and drought on yield and quality traits of a historical (Blanqueta) and a modern (Sula) durum wheat. J. Cereal Sci. 2019, 87, 194–201. [Google Scholar] [CrossRef]
- Morales, F.; Pascual, I.; Sánchez-Díaz, M.; Aguirreolea, J.; Irigoyen, J.J.; Goicoechea, N.; Antolín, M.C.; Oyarzun, M.; Urdiain, A. Methodological advances: Using greenhouses to simulate climate change scenarios. Plant Sci. 2014, 226, 30–40. [Google Scholar] [CrossRef]
- Caemmerer, S.V.; Farquhar, G.D. Some relationships between the biochemistry of photosynthesis and the gas exchange of leaves. Planta 1981, 153, 376–387. [Google Scholar] [CrossRef] [PubMed]
- Chomczynski, P.; Sacchi, N. Single-Step Method of RNA Isolation by Acid Guanidinium Thiocyanate-Phenol-Chloroform Extraction. Anal. Biochem. 1987, 162, 156–159. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001, 4, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res. 2001, 29, 2003–2007. [Google Scholar] [CrossRef] [PubMed]
| Parameter | WW High VPD | WW Low VPD | WS High VPD | WS Low VPD |
|---|---|---|---|---|
| N% | 2.85 ± 0.31 a | 2.9 ± 0.37 a | 2.91 ± 0.31 a | 2.73 ± 0.25 a |
| ARG | 1134.33 ± 122.4 b | 581.3 ± 23.7 c | 2346.7 ± 265.1 a | 2041.7 ± 192.7 a |
| LYS | 386 ± 91.6 b | 106.3 ± 14.3 c | 854.7 ± 86.3 a | 621.7 ± 94.3 ab |
| LEU | 186.6 ± 33.3 b | 55 ± 12.4 c | 269 ± 32.8 ab | 356.7 ± 45.6 a |
| ILE | 199.66 ± 47.2 b | 57.3 ± 9.8 c | 365.7 ± 34 a | 410.7 ± 56.8 a |
| MET | 46 ± 3.5 bc | 7.67 ± 1.3 c | 164.3 ± 62.7 b | 310.7 ± 50.4 a |
| PHE | 578.3 ± 59.4 b | 186.3 ± 45.7 c | 1456.7 ± 347.2 a | 1453 ± 113.2 a |
| TRP | 619 ± 38.9 b | 104.6 ± 43.7 c | 1375 ± 320.4 a | 1101 ± 173.5 a |
| HIS | 155.6 ± 31.3 bc | 73.3 ± 12.4 c | 275 ± 14.7 a | 194 ± 31.2 ab |
| TYR | 202 ± 1.7 b | 74.3 ± 9.8 c | 520.3 ± 49.8 a | 479.3 ± 77.8 a |
| VAL | 530 ± 162.5 b | 124 ± 25.7 c | 784 ± 101.8 ab | 1099.3 ± 153.5 a |
| GLN | 4982.6 ± 885.7 b | 1406.3 ± 225.8 c | 6460.7 ± 476.9 b | 10829.5 ± 967.5 a |
| PRO | 1413.6 ± 148.8 b | 158.3 ± 24.9 c | 5515 ± 1801.7 a | 5507 ± 1334.3 a |
| ASN | 160.5 ± 6.5 b | 234.6 ± 49.5 b | 781.3 ± 91.3 a | 388.3 ± 62.4 b |
| GABA | 204.3 ± 67.4 a | 219.6 ± 59 a | 255 ± 70 a | 284.3 ± 59.6 a |
| THR | 250 ± 19.9 b | 176 ± 23 b | 286 ± 28.3 a | 319 ± 48.4 a |
| SER | 1446.5 ± 12.5 b | 789 ± 154 c | 1939.33 ± 63.8 a | 1371.3 ± 178.3 b |
| GLY | 127 ± 11 b | 100.6 ± 19.5 b | 142 ± 20.1 ab | 206.7 ± 37.3 a |
| ALA | 1366.3 ± 214.9 a | 1604.6 ± 140.8 a | 1424.3 ± 198.3 a | 713.5 ± 5.5 b |
| GLU | 2999.6 ± 65.8 a | 2476 ± 96 b | 2636.5 ± 1.5 ab | 2599.7 ± 178.5 b |
| ASP | 1546 ± 160.6 a | 776 ± 7.3 b | 1267 ± 138.6 ab | 1106.3 ± 172.8 ab |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fakhet, D.; Morales, F.; Jauregui, I.; Erice, G.; Aparicio-Tejo, P.M.; González-Murua, C.; Aroca, R.; Irigoyen, J.J.; Aranjuelo, I. Short-Term Exposure to High Atmospheric Vapor Pressure Deficit (VPD) Severely Impacts Durum Wheat Carbon and Nitrogen Metabolism in the Absence of Edaphic Water Stress. Plants 2021, 10, 120. https://doi.org/10.3390/plants10010120
Fakhet D, Morales F, Jauregui I, Erice G, Aparicio-Tejo PM, González-Murua C, Aroca R, Irigoyen JJ, Aranjuelo I. Short-Term Exposure to High Atmospheric Vapor Pressure Deficit (VPD) Severely Impacts Durum Wheat Carbon and Nitrogen Metabolism in the Absence of Edaphic Water Stress. Plants. 2021; 10(1):120. https://doi.org/10.3390/plants10010120
Chicago/Turabian StyleFakhet, Dorra, Fermín Morales, Iván Jauregui, Gorka Erice, Pedro M. Aparicio-Tejo, Carmen González-Murua, Ricardo Aroca, Juan J. Irigoyen, and Iker Aranjuelo. 2021. "Short-Term Exposure to High Atmospheric Vapor Pressure Deficit (VPD) Severely Impacts Durum Wheat Carbon and Nitrogen Metabolism in the Absence of Edaphic Water Stress" Plants 10, no. 1: 120. https://doi.org/10.3390/plants10010120
APA StyleFakhet, D., Morales, F., Jauregui, I., Erice, G., Aparicio-Tejo, P. M., González-Murua, C., Aroca, R., Irigoyen, J. J., & Aranjuelo, I. (2021). Short-Term Exposure to High Atmospheric Vapor Pressure Deficit (VPD) Severely Impacts Durum Wheat Carbon and Nitrogen Metabolism in the Absence of Edaphic Water Stress. Plants, 10(1), 120. https://doi.org/10.3390/plants10010120








