Anticoagulation Therapies and microRNAs in Heart Failure
Abstract
1. Introduction
2. Antiplatelet Drugs
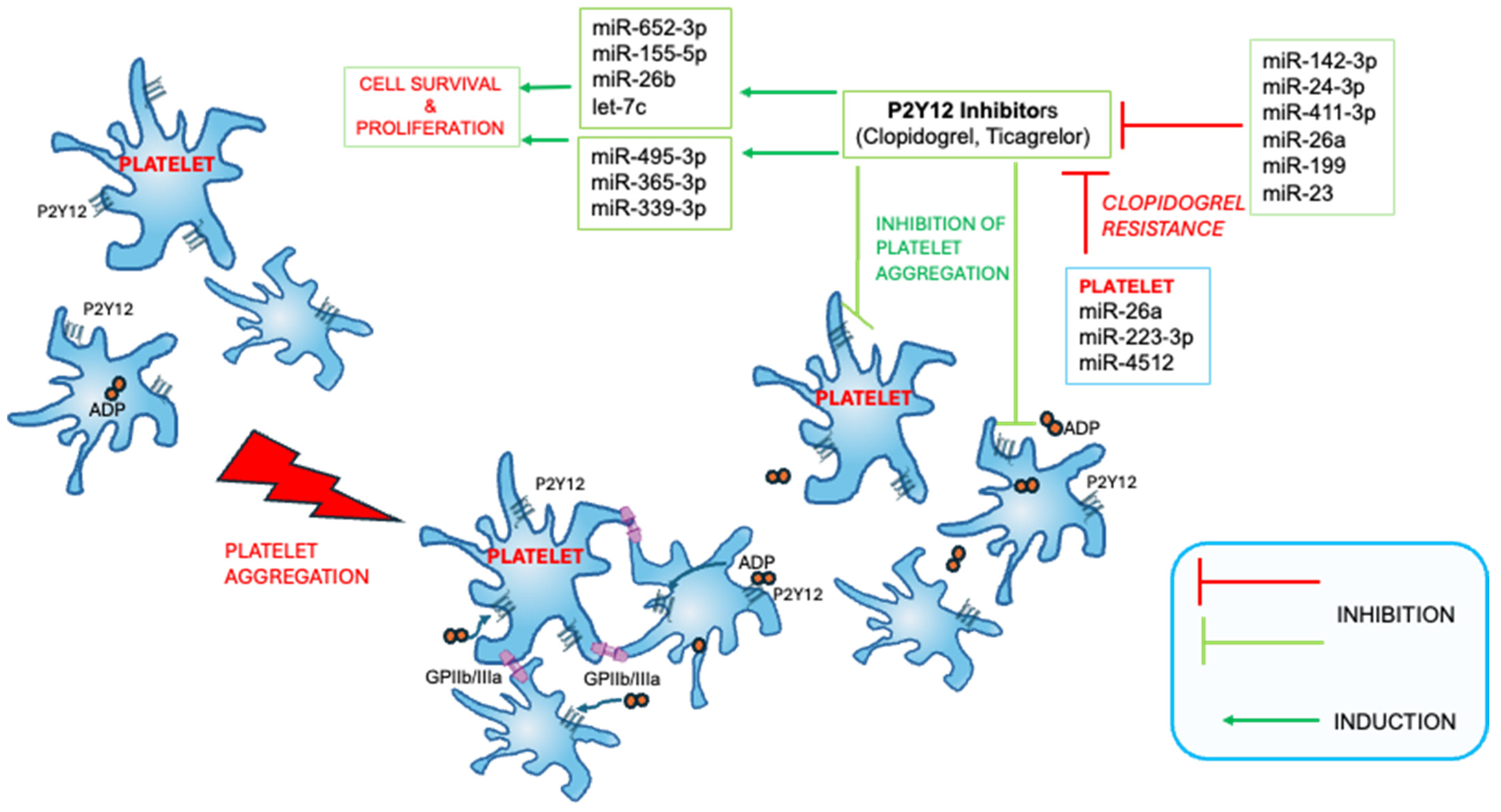
2.1. Clopidogrel and Ticagrelor
2.2. Aspirin
3. Anticoagulant Drugs
3.1. Vitamin K Inhibitor: Warfarin
3.2. Oral Anticoagulants
3.2.1. Factor X Inhibitors: Apixaban and Rivaroxaban
3.2.2. Thrombin Activation-Inhibitor: Edoxaban
3.2.3. Direct Thrombin Inhibitor: Dabigatran Etexilate
4. Digoxin
5. Sino-Atrial Node Current Inhibitor: Ivabradine
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bozkurt, B.; Ahmad, T.; Alexander, K.; Baker, W.L.; Bosak, K.; Breathett, K.; Carter, S.; Drazner, M.H.; Dunlay, S.M.; Fonarow, G.C.; et al. HF STATS 2024: Heart Failure Epidemiology and Outcomes Statistics An Updated 2024 Report from the Heart Failure Society of America. J. Card. Fail. 2024, 31, 66–116. [Google Scholar] [CrossRef]
- Rasmussen, M.; Prado, A.; Hominal, M.A.; Zaidman, C.J.; Cursack, G.; MacKinnon, I.; Zapata, G.; Rojas, D.G.; Duran, R.G.; Vilamajo, O.G.; et al. Global Variations in Heart Failure Etiology, Management, and Outcomes. JAMA 2023, 329, 1650–1661. [Google Scholar] [CrossRef]
- Leiner, J.; König, S.; Nitsche, A.; Hohenstein, S.; Nagel, J.; Seyfarth, M.; Baberg, H.; Lauten, A.; Neuser, H.; Staudt, A.; et al. A multicentre registry of hospitalized patients with acute and chronic heart failure: Study design of the H2-registry. ESC Heart Fail. 2025, 12, 3114–3133. [Google Scholar] [CrossRef]
- Bozkurt, B.; Coats, A.; Tsutsui, H. Universal definition and classification of heart failure: A report of the Heart Failure Society of America, Heart Failure Association of the European Society of Cardiology, Japanese Heart Failure Society and Writing Committee of the Universal Definition of Heart Failure. J. Card. Fail. 2021, 23, 352–380. [Google Scholar] [CrossRef]
- Heidenreich, P.A.; Bozkurt, B.; Aguilar, D.; Allen, L.A.; Byun, J.J.; Colvin, M.M.; Deswal, A.; Drazner, M.H.; Dunlay, S.M.; Evers, L.R.; et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2022, 145, e895–e1032. [Google Scholar] [CrossRef]
- Patolia, H.; Khan, M.S.; Fonarow, G.C.; Butler, J.; Greene, S.J. Implementing Guideline-Directed Medical Therapy for Heart Failure. JACC 2023, 82, 529–543. [Google Scholar] [CrossRef]
- Yancy, C.W.; Jessup, M.; Bozkurt, B.; Butler, J.; Casey, D.E., Jr.; Colvin, M.M.; Drazner, M.H.; Filippatos, G.S.; Fonarow, G.C.; Givertz, M.M.; et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. J. Am. Coll. Cardiol. 2017, 70, 776–803. [Google Scholar] [PubMed]
- Yancy, C.W.; Jessup, M.; Bozkurt, B.; Butler, J.; Casey, D.E., Jr.; Drazner, M.H.; Fonarow, G.C.; Geraci, S.A.; Horwich, T.; Januzzi, J.L.; et al. 2013 ACCF/AHA guideline for the management of heart failure: A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 2013, 62, e147–e239. [Google Scholar] [CrossRef] [PubMed]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.F.; Coats, A.J.S.; Falk, V.; González-Juanatey, J.R.; Harjola, V.P.; Jankowska, E.A.; et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC)Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2016, 37, 2129–2200. [Google Scholar] [CrossRef] [PubMed]
- Siniarski, A.; Gąsecka, A.; Borovac, J.A.; Papakonstantinou, P.E.; Bongiovanni, D.; Ehrlinder, H.; Giustozzi, M.; Guerreiro, R.A.; Parker, W.A. Blood Coagulation Disorders in Heart Failure: From Basic Science to Clinical Perspectives. J. Card. Fail. 2023, 29, 517–526. [Google Scholar] [CrossRef]
- Nohria, A. β-Adrenergic Blockade for Anthracycline- and Trastuzumab-Induced Cardiotoxicity. Circ. Heart Fail. 2013, 6, 358–361. [Google Scholar] [CrossRef][Green Version]
- van Rooij, E.; Sutherland, L.B.; Liu, N.; Williams, A.H.; McAnally, J.; Gerard, R.D.; Richardson, J.A.; Olson, E.N. A signature pattern of stress-responsive microRNAs that can evoke cardiac hypertrophy and heart failure. Proc. Natl. Acad. Sci. USA 2006, 103, 18255–18260. [Google Scholar] [CrossRef] [PubMed]
- Ambrosy, A.P.; Butler, J.; Ahmed, A.; Vaduganathan, M.; van Veldhuisen, D.J.; Colucci, W.S.; Gheorghiade, M. The Use of Digoxin in Patients With Worsening Chronic Heart Failure. JACC 2014, 63, 1823–1832. [Google Scholar] [CrossRef]
- Rahhal, A.; Habib, M.B.; Alani, H.; Chapra, A.; Mahfouz, A.; Alyafei, S.; Carr, C.; Alkhulaifi, A.; Omar, A. Impact of digoxin on gastrointestinal bleeding following left ventricular assist device implantation: A systematic review and meta-analysis. Eur. Heart J. 2024, 45, ehae666-1119. [Google Scholar] [CrossRef]
- Vukelic, S.; Vlismas, P.P.; Patel, S.R.; Xue, X.; Shitole, S.G.; Saeed, O.; Sims, D.B.; Chinnadurai, T.; Shin, J.J.; Forest, S.J.; et al. Digoxin Is Associated With a Decreased Incidence of Angiodysplasia-Related Gastrointestinal Bleeding in Patients With Continuous-Flow Left Ventricular Assist Devices. Circ. Heart Fail. 2018, 11, e004899. [Google Scholar] [CrossRef]
- Imamura, T. Clinical Implications of Ivabradine in the Contemporary Era. Medicina 2024, 60, 303. [Google Scholar] [CrossRef]
- Thum, T.; Galuppo, P.; Wolf, C.; Fiedler, J.; Kneitz, S.; van Laake, L.W.; Doevendans, P.A.; Mummery, C.L.; Borlak, J.; Haverich, A.; et al. MicroRNAs in the Human Heart. Circulation 2007, 116, 258–267. [Google Scholar] [CrossRef]
- Vegter, E.L.; van der Meer, P.; de Windt, L.J.; Pinto, Y.M.; Voors, A.A. MicroRNAs in heart failure: From biomarker to target for therapy. Eur. J. Heart Fail. 2016, 18, 457–468. [Google Scholar] [CrossRef] [PubMed]
- Shen, N.-N.; Wang, J.-L.; Fu, Y.-P. The microRNA Expression Profiling in Heart Failure: A Systematic Review and Meta-Analysis. Front. Cardiovasc. Med. 2022, 9, 856358. [Google Scholar] [CrossRef] [PubMed]
- Gargiulo, P.; Marzano, F.; Salvatore, M.; Basile, C.; Buonocore, D.; Parlati, A.L.M.; Nardi, E.; Asile, G.; Abbate, V.; Colella, A.; et al. MicroRNAs: Diagnostic, prognostic and therapeutic role in heart failure—A review. ESC Heart Fail. 2022, 10, 753–761. [Google Scholar] [CrossRef]
- Song, R.; Zhang, L. MicroRNAs and therapeutic potentials in acute and chronic cardiac disease. Drug Discov. Today 2024, 29, 104179. [Google Scholar] [CrossRef]
- Zhou, S.-S.; Jin, J.-P.; Wang, J.-Q.; Zhang, Z.-G.; Freedman, J.H.; Zheng, Y.; Cai, L. miRNAS in cardiovascular diseases: Potential biomarkers, therapeutic targets and challenges. Acta Pharmacol. Sin. 2018, 39, 1073–1084. [Google Scholar] [CrossRef]
- Parvan, R.; Becker, V.; Hosseinpour, M.; Moradi, Y.; Louch, W.E.; Cataliotti, A.; Devaux, Y.; Frisk, M.; Silva, G.J.J.; CA21153, A.C.A. Prognostic and predictive microRNA panels for heart failure patients with reduced or preserved ejection fraction: A meta-analysis of Kaplan–Meier-based individual patient data. BMC Med. 2025, 23, 1–16. [Google Scholar] [CrossRef]
- Procyk, G.; Klimczak-Tomaniak, D.; Sygitowicz, G.; Tomaniak, M. Circulating and Platelet MicroRNAs in Cardiovascular Risk Assessment and Antiplatelet Therapy Monitoring. J. Clin. Med. 2022, 11, 1763. [Google Scholar] [CrossRef]
- Farid, N.A.; Kurihara, A.; Wrighton, S.A. Metabolism and disposition of the thienopyridine antiplatelet drugs ticlopidine, clopidogrel, and Prasugrel in humans. J. Clin. Pharmacol. 2010, 50, 126–142. [Google Scholar] [CrossRef]
- Herbert, J.-M.; Savi, P. P2Y12, A New Platelet ADP Receptor, Target of Clopidogrel. Semin. Vasc. Med. 2003, 03, 113–122. [Google Scholar] [CrossRef]
- Polasek, T.M.; Doogue, M.P.; Miners, J.O. Metabolic activation of clopidogrel: In Vitro data provide conflicting evidence for the contributions of CYP2C19 and PON1. Ther. Adv. Drug Saf. 2011, 2, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Lau, W.C.; Gurbel, P.A.; Watkins, P.B.; Neer, C.J.; Hopp, A.S.; Carville, D.G.; Guyer, K.E.; Tait, A.R.; Bates, E.R. Contribution of Hepatic Cytochrome P450 3A4 Metabolic Activity to the Phenomenon of Clopidogrel Resistance. Circulation 2004, 109, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Clarke, T.A.; Waskell, L.A. The Metabolism of Clopidogrel Is Catalyzed by Human Cytochrome P450 3A and Is Inhibited by Atorvastatin. Drug Metab. Dispos. 2003, 31, 53–59. [Google Scholar] [CrossRef]
- Biasucci, L.M.; Mandolini, C.; la Rosa, G.; Grimaldi, M.C.; Crea, F. Abstract 18464: Different Modulation of MicroRNA by Ticagrelor and Clopidogrel in Non STEMI Patients (TIGER M Study NCT02071966). Circulation 2017, 136, A18464. [Google Scholar]
- Maqbool, S.; Ali, M.S.; Rehman, A.; Rehman, M.E.U.; Iqbal, J.; Razzaq, A.; Kamal, A.; Shivamadhu, S.S.; Afzal, M.; Fazal, F.; et al. Efficacy and Safety Profile of Ticagrelor Versus Clopidogrel in Percutaneous Coronary Intervention (PCI) for Acute Coronary Syndrome (ACS): A Systematic Review and Meta-Analysis. Cureus 2023, 15, e46455. [Google Scholar] [CrossRef]
- Akyuz, S.; Calik, A.N.; Yaylak, B.; Onuk, T.; Eren, S.; Kolak, Z.; Mollaalioglu, F.; Durak, F.; Cetin, M.; Tanboga, I.H. Comparison of Ticagrelor and Clopidogrel in Patients With Acute Coronary Syndrome at High Bleeding or Ischemic Risk. Am. J. Cardiol. 2023, 210, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Wallentin, L.; Becker, R.C.; Budaj, A.; Cannon, C.P.; Emanuelsson, H.; Held, C.; Horrow, J.; Husted, S.; James, S.; Katus, H.; et al. Ticagrelor versus Clopidogrel in Patients with Acute Coronary Syndromes. N. Engl. J. Med. 2009, 361, 1045–1057. [Google Scholar] [CrossRef]
- Turgeon, R.D.; Koshman, S.L.; Youngson, E.; Har, B.; Wilton, S.B.; James, M.T.; Graham, M.M. Association of Ticagrelor vs Clopidogrel With Major Adverse Coronary Events in Patients With Acute Coronary Syndrome Undergoing Percutaneous Coronary Intervention. JAMA Intern. Med. 2020, 180, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-C.; Lin, F.-Y.; Lin, Y.-W.; Cheng, S.-M.; Chang, C.-C.; Lin, R.-H.; Chuang, C.-L.; Sheu, J.-S.; Chen, S.-M.; Tsai, C.-S. Platelet MicroRNA 365-3p Expression Correlates with High On-treatment Platelet Reactivity in Coronary Artery Disease Patients. Cardiovasc. Drugs Ther. 2019, 33, 129–137. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, H.; Li, X.; Wang, L.; Yu, H.; Huang, J.; Liu, Q.; Wang, C.; Jiang, A. Diagnostic and prognostic significance of aberrant miR-652-3p levels in patients with acute decompensated heart failure and acute kidney injury. J. Int. Med Res. 2020, 48, 1–12. [Google Scholar] [CrossRef]
- Stevens, M.T.; Saunders, B.M. Targets and regulation of microRNA-652-3p in homoeostasis and disease. J. Mol. Med. 2021, 99, 755–769. [Google Scholar] [CrossRef]
- Lin, S.; Xu, X.; Hu, H.; Cheng, J.; Chen, R.; Hu, Y.; Chen, X. The expression profile of platelet-derived miRNA in coronary artery disease patients with clopidogrel resistance. Pharmacol. Res. Perspect. 2021, 9, e00751. [Google Scholar] [CrossRef]
- Liu, Y.L.; Hu, X.L.; Song, P.Y.; Li, H.; Li, M.P.; Du, Y.X.; Li, M.Y.; Ma, Q.L.; Peng, L.M.; Song, M.Y.; et al. Influence of GAS5/MicroRNA-223-3p/P2Y12 Axis on Clopidogrel Response in Coronary Artery Disease. J. Am. Heart Assoc. 2021, 10, e021129. [Google Scholar] [CrossRef] [PubMed]
- Rytkin, E.; Mirzaev, K.; Bure, I.; Sychev, D. Selection of miRNAs for clopidogrel resistance prediction. Meta Gene 2020, 25, 100745. [Google Scholar] [CrossRef]
- Icli, B.; Wara, A.; Moslehi, J.; Sun, X.; Plovie, E.; Cahill, M.; Marchini, J.F.; Schissler, A.; Padera, R.F.; Shi, J.; et al. MicroRNA-26a Regulates Pathological and Physiological Angiogenesis by Targeting BMP/SMAD1 Signaling. Circ. Res. 2013, 113, 1231–1241. [Google Scholar] [CrossRef] [PubMed]
- Meloni, M.; Marchetti, M.; Garner, K.; Littlejohns, B.; Sala-Newby, G.; Xenophontos, N.; Floris, I.; Suleiman, M.-S.; Madeddu, P.; Caporali, A.; et al. Local Inhibition of MicroRNA-24 Improves Reparative Angiogenesis and Left Ventricle Remodeling and Function in Mice With Myocardial Infarction. Mol. Ther. 2013, 21, 1390–1402. [Google Scholar] [CrossRef]
- Wang, X.-Z.; Chen, L.; Sun, H.; Li, X.-Q.; Wang, H.; Zhang, X.-P.; Sun, J.-B.; Wang, H.-Y. MiR-199a-3p promotes repair of myocardial infarction by targeting NACC2. Int. J. Clin. Exp. Pathol. 2023, 16, 57–66. [Google Scholar]
- Kern, F.; Aparicio-Puerta, E.; Li, Y.; Fehlmann, T.; Kehl, T.; Wagner, V.; Ray, K.; Ludwig, N.; Lenhof, H.-P.; Meese, E.; et al. miRTargetLink 2.0—Interactive miRNA target gene and target pathway networks. Nucleic Acids Res. 2021, 49, W409–W416. [Google Scholar] [CrossRef] [PubMed]
- Szklarczyk, D.; Kirsch, R.; Koutrouli, M.; Nastou, K.; Mehryary, F.; Hachilif, R.; Gable, A.L.; Fang, T.; Doncheva, N.T.; Pyysalo, S.; et al. The STRING database in 2023: Protein–protein association networks and functional enrichment analyses for any sequenced genome of interest. Nucleic Acids Res. 2022, 51, D638–D646. [Google Scholar] [CrossRef]
- Shi, R.; Zhou, X.; Ji, W.-J.; Zhang, Y.-Y.; Ma, Y.-Q.; Zhang, J.-Q.; Li, Y.-M. The Emerging Role of miR-223 in Platelet Reactivity: Implications in Antiplatelet Therapy. BioMed Res. Int. 2015, 2015, 1–8. [Google Scholar] [CrossRef]
- Askari, S.; Goldfinger, L.E. Roles of miR-223 in Platelet Function and High On-Treatment Platelet Reactivity: A Brief Report and Review. Genes 2025, 16, 312. [Google Scholar] [CrossRef]
- Willeit, P.; Zampetaki, A.; Dudek, K.; Kaudewitz, D.; King, A.; Kirkby, N.S.; Crosby-Nwaobi, R.; Prokopi, M.; Drozdov, I.; Langley, S.R.; et al. Circulating MicroRNAs as Novel Biomarkers for Platelet Activation. Circ. Res. 2013, 112, 595–600. [Google Scholar] [CrossRef]
- Jha, S.; Loganathbabu, V.K.T.; Kumaran, K.; Krishnasamy, G.; Aruljothi, K.N. Long Non-Coding RNAs (lncRNAs) in Heart Failure: A Comprehensive Review. Non-Coding RNA 2023, 10, 3. [Google Scholar] [CrossRef]
- Landry, P.; Plante, I.; Ouellet, D.L.; Perron, M.P.; Rousseau, G.; Provost, P. Existence of a microRNA pathway in anucleate platelets. Nat. Struct. Mol. Biol. 2009, 16, 961–966. [Google Scholar] [CrossRef]
- Mo, Z.-Z.; Yuan, Z.; Peng, Y.-Y.; Zhou, W.-L.; Dai, W.; Wang, G.; Tang, J.; Zhang, W.; Chen, B.-L. miR-6076 rs1463411 polymorphisms are associated with bleeding during clopidogrel treatment in patients with acute coronary syndrome. Eur. J. Med Res. 2023, 28, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.-L.; Mo, Z.-Z.; Xiao, F.-Y.; Dai, W.; Wang, G.; Zhou, G.; Zhang, W.; Chen, B.-L. microRNA-605 rs2043556 polymorphisms affect clopidogrel therapy through modulation of CYP2B6 and P2RY12 in acute coronary syndrome patients. Platelets 2019, 31, 897–905. [Google Scholar] [CrossRef]
- Hu, X.; Li, M.; Li, H.; Song, P.; Zhang, Y.; Zhou, G.; Tang, J.; Peng, L.; Ma, Q.; Chen, X. Decreased platelet miR-199a-5p level might lead to high on-clopidogrel platelet reactivity in patients with coronary artery disease. Platelets 2023, 34, 2200860. [Google Scholar] [CrossRef]
- Syam, H.; Sukmawan, R.; Dharma, S.; Alaztha, G.; Giyantini, A.; Prakoso, R.; Rudiktyo, E.; Zamroni, D.; Rejeki, V.; Juzar, D.; et al. Epigenetic interaction of miRNA-26a and P2Y12 gene DNA methylation on platelet reactivity under clopidogrel and their impact to the coronary flow after primary PCI in STEMI. Eur. Heart J. 2020, 41, ehaa946-1547. [Google Scholar] [CrossRef]
- Xu, H.; Su, J.; Chen, X.; Li, J.; Li, Z.; Zheng, N.; Yu, R.; Li, X.; Song, Y.; Li, J.; et al. Identification of hsa_circ_0076957 and miR-4512-targeted COL19A1 as regulators in clopidogrel resistance among stable coronary heart disease patients through comprehensive circRNA and miRNA analysis. Eur. J. Pharmacol. 2024, 986, 177156. [Google Scholar] [CrossRef]
- Sneader, W. The discovery of aspirin: A reappraisal. BMJ 2000, 321, 1591–1594. [Google Scholar] [CrossRef] [PubMed]
- Tabari, M.A.K.; Mishan, M.A.; Moradi, M.; Khandan, M.; Khoshhal, H.; Mahrooz, A.; Bagheri, A. Noncoding RNA Roles in Pharmacogenomic Responses to Aspirin: New Molecular Mechanisms for an Old Drug. BioMed Res. Int. 2021, 2021, 6830560. [Google Scholar] [CrossRef] [PubMed]
- Vane, J.; Botting, R. The mechanism of action of aspirin. Thromb. Res. 2003, 110, 255–258. [Google Scholar] [CrossRef]
- Botting, R.M. Vane’s discovery of the mechanism of action of aspirin changed our understanding of its clinical pharmacology. Pharmacol. Rep. 2010, 62, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Bermingham, M.; Shanahan, M.K.; O’connell, E.; Dawkins, I.; Miwa, S.; O’hanlon, R.; Gilmer, J.; McDonald, K.; Ledwidge, M.; J, C.; et al. Aspirin Use in Heart Failure. Circ. Heart Fail. 2014, 7, 243–250. [Google Scholar] [CrossRef]
- Mujaj, B.; Zhang, Z.; Yang, W.; Thijs, L.; Wei, F.; Verhamme, P.; Delles, C.; Butler, J.; Sever, P.; Latini, R.; et al. Aspirin use is associated with increased risk for incident heart failure: A patient-level pooled analysis. ESC Heart Fail. 2021, 9, 685–694. [Google Scholar] [CrossRef]
- de Oliveira, A.B.M.; Luchiari, B.; Bonilha, I.; Barreto, J.; Nogueira, A.C.C.; Ceniccola, G.D.; Polanczyk, C.A.; Sposito, A.C.; de Carvalho, L.S.F. Aspirin in primary prevention and the risk of heart failure: A systematic review and meta-analysis of controlled trials. ESC Heart Fail. 2022, 10, 1488–1491. [Google Scholar] [CrossRef]
- La Rosa, G.; Biasucci, L.M.; Mandolini, C.; Massimi, I.; Copponi, G.; Pulcinelli, F.M.; Crea, F. Platelet miRNA-26b down-regulates multidrug resistance protein 4 in patients on chronic aspirin treatment. J. Cardiovasc. Med. 2018, 19, 611–613. [Google Scholar] [CrossRef] [PubMed]
- Massimi, I.; Guerriero, R.; Lotti, L.V.; Lulli, V.; Borgognone, A.; Romani, F.; Barillà, F.; Gaudio, C.; Gabbianelli, M.; Frati, L.; et al. Aspirin influences megakaryocytic gene expression leading to up-regulation of multidrug resistance protein-4 in human platelets. Br. J. Clin. Pharmacol. 2014, 78, 1343–1353. [Google Scholar] [CrossRef] [PubMed]
- Mattiello, T.; Guerriero, R.; Lotti, L.V.; Trifirò, E.; Felli, M.P.; Barbarulo, A.; Pucci, B.; Gazzaniga, P.; Gaudio, C.; Frati, L.; et al. Aspirin Extrusion From Human Platelets Through Multidrug Resistance Protein-4–Mediated Transport. J. Am. Coll. Cardiol. 2011, 58, 752–761. [Google Scholar] [CrossRef]
- Chang, P.-Y.; Chen, Y.J.; Yang, T.-C.; Lu, S.-C.; Chen, C.-H. Low-Dose Aspirin Prevents Coronary Endothelial Injury by Oxidized-LDL through the Akt- FGF2 Prosurvival Pathway. J. Am. Coll. Cardiol. 2012, 59, E1726. [Google Scholar] [CrossRef]
- De Boer, H.C.; Van Solingen, C.; Prins, J.; Duijs, J.M.; Huisman, M.V.; Rabelink, T.J.; van Zonneveld, A.J. Aspirin treatment hampers the use of plasma microRNA-126 as a biomarker for the progression of vascular disease. Eur. Heart J. 2013, 34, 3451–3457. [Google Scholar] [CrossRef]
- Zapilko, V.; Fish, R.J.; Garcia, A.; Reny, J.-L.; Dunoyer-Geindre, S.; Lecompte, T.; Neerman-Arbez, M.; Fontana, P. MicroRNA-126 is a regulator of platelet-supported thrombin generation. Platelets 2020, 31, 746–755. [Google Scholar] [CrossRef]
- Ruan, D.T.; Gao, S.; Shelat, H.; King, B.; Geng, Y.-J. Differential expression of microRNA and arachidonic acid metabolism in aspirin-treated human cardiac and peri-cardiac fat-derived mesenchymal stem cells. Vasc. Pharmacol. 2020, 127, 106651. [Google Scholar] [CrossRef]
- Volodko, O.; Volinsky, N.; Yarkoni, M.; Margalit, N.; Kusniec, F.; Sudarsky, D.; Elbaz-Greener, G.; Carasso, S.; Amir, O. Characterization of Systemic and Culprit-Coronary Artery miR-483-5p Ex-pression in Chronic CAD and Acute Myocardial Infarction Male Patients. Int. J. Mol. Sci. 2023, 24, 8551. [Google Scholar] [CrossRef]
- Kok, M.; Mandolini, C.; Moerland, P.; de Ronde, M.; Sondermeijer, B.; Halliani, A.; Nieuwland, R.; Cipollone, F.; Creemers, E.; Meijers, J.; et al. Low miR-19b-1-5p expression in isolated platelets after aspirin use is related to aspirin insensitivity. Int. J. Cardiol. 2015, 203, 262–263. [Google Scholar] [CrossRef]
- Binderup, H.G.; Houlind, K.; Madsen, J.S.; Brasen, C.L. Aspirin resistance may be identified by miR-92a in plasma combined with platelet distribution width. Clin. Biochem. 2016, 49, 1167–1172. [Google Scholar] [CrossRef]
- Singh, S.; de Ronde, M.W.J.; Creemers, E.E.; Van der Made, I.; Meijering, R.; Chan, M.Y.; Tan, S.H.; Chin, C.T.; Richards, A.M.; Troughton, R.W.; et al. Low miR-19b-1-5p Expression Is Related to Aspirin Resistance and Major Adverse Cardio- Cerebrovascular Events in Patients With Acute Coronary Syndrome. J. Am. Heart Assoc. 2021, 10, e017120. [Google Scholar] [CrossRef]
- Kahner, B.N.; Shankar, H.; Murugappan, S.; Prasad, G.L.; Kunapuli, S.P. Nucleotide receptor signaling in platelets. J. Thromb. Haemost. 2006, 4, 2317–2326. [Google Scholar] [CrossRef] [PubMed]
- Mangin, P.; Ohlmann, P.; Eckly, A.; Cazenave, J.; Lanza, F.; Gachet, C. The P2Y1receptor plays an essential role in the platelet shape change induced by collagen when TxA2 formation is prevented. J. Thromb. Haemost. 2004, 2, 969–977. [Google Scholar] [CrossRef] [PubMed]
- Pan, D.; Ladds, G.; Rahman, K.M.; Pitchford, S.C. Exploring bias in platelet P2Y1 signalling: Host defence versus haemostasis. Br. J. Pharmacol. 2023, 181, 580–592. [Google Scholar] [CrossRef]
- Timur, A.A.; Murugesan, G.; Zhang, L.; Aung, P.P.; Barnard, J.; Wang, Q.K.; Gaussem, P.; Silverstein, R.L.; Bhatt, D.L.; Kottke-Marchant, K. P2RY1 and P2RY12 polymorphisms and on-aspirin platelet reactivity in patients with coronary artery disease. Int. J. Lab. Hematol. 2012, 34, 473–483. [Google Scholar] [CrossRef]
- Lordkipanidzé, M.; Diodati, J.G.; Palisaitis, D.A.; Schampaert, E.; Turgeon, J.; Pharand, C. Genetic determinants of response to aspirin: Appraisal of 4 candidate genes. Thromb. Res. 2011, 128, 47–53. [Google Scholar] [CrossRef]
- Lee, M.T.M.; Klein, T.E. Pharmacogenetics of warfarin: Challenges and opportunities. J. Hum. Genet. 2013, 58, 334–338. [Google Scholar] [CrossRef] [PubMed]
- Shahin, M.H.; Johnson, J.A. Clopidogrel and warfarin pharmacogenetic tests. Curr. Opin. Cardiol. 2013, 28, 305–314. [Google Scholar] [CrossRef]
- Kuruvilla, M.; Gurk-Turner, C. A Review of Warfarin Dosing and Monitoring. Bayl. Univ. Med. Cent. Proc. 2001, 14, 305–306. [Google Scholar] [CrossRef]
- Shoji, M.; Suzuki, S.; Otsuka, T.; Arita, T.; Yagi, N.; Semba, H.; Kano, H.; Matsuno, S.; Kato, Y.; Uejima, T.; et al. A Simple Formula for Predicting the Maintenance Dose of Warfarin with Reference to the Initial Response to Low Dosing at an Outpatient Clinic. Intern. Med. 2020, 59, 29–35. [Google Scholar] [CrossRef]
- A Tideman, P.; Tirimacco, R.; John, A.S.; Roberts, G.W. How to manage warfarin therapy. Aust. Prescr. 2022, 38, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.C.; Qian, M.; Lip, G.Y.; Di Tullio, M.R.; Graham, S.; Mann, D.L.; Nakanishi, K.; Teerlink, J.R.; Freudenberger, R.S.; Sacco, R.L.; et al. Heart Failure Severity and Quality of Warfarin Anticoagulation Control (From the WARCEF Trial). Am. J. Cardiol. 2018, 122, 821–827. [Google Scholar] [CrossRef]
- Klein, T.E.; Altman, R.B.; Eriksson, N.; Gage, B.F.; Kimmel, S.E.; Lee, M.-T.M.; Limdi, N.A.; Page, D.; Roden, D.M.; Wagner, M.J.; et al. Estimation of the Warfarin Dose with Clinical and Pharmacogenetic Data. N. Engl. J. Med. 2009, 360, 753–764. [Google Scholar] [CrossRef]
- Redman, A.R. Implications of Cytochrome P450 2C9 Polymorphism on Warfarin Metabolism and Dosing. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2001, 21, 235–242. [Google Scholar] [CrossRef]
- Higashi, M.K.; Veenstra, D.L.; Kondo, L.M.; Wittkowsky, A.K.; Srinouanprachanh, S.L.; Farin, F.M.; Rettie, A.E. Association Between CYP2C9 Genetic Variants and Anticoagulation-Related Outcomes During Warfarin Therapy. JAMA 2002, 287, 1690–1698. [Google Scholar] [CrossRef] [PubMed]
- Taube, J.; Halsall, D.; Baglin, T. Influence of cytochrome P-450 CYP2C9 polymorphisms on warfarin sensitivity and risk of over-anticoagulation in patients on long-term treatment. Blood 2000, 96, 1816–1819. [Google Scholar] [CrossRef]
- Aithal, G.P.; Day, C.P.; Kesteven, P.J.; Daly, A.K. Association of polymorphisms in the cytochrome P450 CYP2C9 with warfarin dose requirement and risk of bleeding complications. Lancet 1999, 353, 717–719. [Google Scholar] [CrossRef]
- Tian, Z.; Yang, Y.; Feng, Z.; Wu, D.; Yang, W.; Liu, D. Genetic variant in the promoter region of microRNA-137 reduces the warfarin maintenance dose in patients with atrial fibrillation. Mol. Med. Rep. 2019, 19, 5361–5367. [Google Scholar] [CrossRef] [PubMed]
- Elgebaly, S.A.; Christenson, R.H.; Kandil, H.; Ibrahim, M.; Rizk, H.; El-Khazragy, N.; Rashed, L.; Yacoub, B.; Eldeeb, H.; Ali, M.M.; et al. Nourin-Dependent miR-137 and miR-106b: Novel Biomarkers for Early Diagnosis of Myocardial Ischemia in Coronary Artery Disease Patients. Diagnostics 2021, 11, 703. [Google Scholar] [CrossRef]
- Lok, S.I.; van Mil, A.; Bovenschen, N.; van der Weide, P.; van Kuik, J.; van Wichen, D.; Peeters, T.; Siera, E.; Winkens, B.; Sluijter, J.P.; et al. Post-transcriptional Regulation of α-1-Antichymotrypsin by MicroRNA-137 in Chronic Heart Failure and Mechanical Support. Circ. Heart Fail. 2013, 6, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Ciccacci, C.; Rufini, S.; Politi, C.; Novelli, G.; Forte, V.; Borgiani, P. Could MicroRNA polymorphisms influence warfarin dosing? A pharmacogenetics study on mir133 genes. Thromb. Res. 2015, 136, 367–370. [Google Scholar] [CrossRef]
- Pérez-Andreu, V.; Teruel, R.; Corral, J.; Roldán, V.; García-Barberá, N.; Salloum-Asfar, S.; Gómez-Lechón, M.J.; Bourgeois, S.; Deloukas, P.; Wadelius, M.; et al. miR-133a Regulates Vitamin K 2,3-Epoxide Reductase Complex Subunit 1 (VKORC1), a Key Protein in the Vitamin K Cycle. Mol. Med. 2012, 18, 1466–1472. [Google Scholar] [CrossRef]
- Zhao, L.; Wang, J.; Shi, S.; Wu, Y.; Liu, J.; He, S.; Zou, Y.; Xie, H.; Ge, S.; Ye, H. Plasma miRNA profiles associated with stable warfarin dosage in Chinese patients. PeerJ 2020, 8, e9995. [Google Scholar] [CrossRef]
- Danowski, N.; Manthey, I.; Jakob, H.G.; Siffert, W.; Peters, J.; Frey, U.H. Decreased Expression of miR-133a but Not of miR-1 is Associated with Signs of Heart Failure in Patients Undergoing Coronary Bypass Surgery. Cardiology 2013, 125, 125–130. [Google Scholar] [CrossRef]
- Rad, H.K.; Mazaheri, M.; Firozabadi, A.D. Relative Expression of PBMC MicroRNA-133a Analysis in Patients Receiving Warfarin After Mechanical Heart Valve Replacement. Avicenna J. Med. Biotechnol. 2018, 10, 29–33. [Google Scholar]
- Canale, P.; Nicolini, G.; Pitto, L.; Kusmic, C.; Rizzo, M.; Balzan, S.; Iervasi, G.; Forini, F. Role of miR-133/Dio3 Axis in the T3-Dependent Modulation of Cardiac mitoK-ATP Expression. Int. J. Mol. Sci. 2022, 23, 6549. [Google Scholar] [CrossRef]
- Byon, W.; Garonzik, S.; Boyd, R.A.; Frost, C.E. Apixaban: A Clinical Pharmacokinetic and Pharmacodynamic Review. Clin. Pharmacokinet. 2019, 58, 1265–1279. [Google Scholar] [CrossRef]
- Raghavan, N.; Frost, C.E.; Yu, Z.; He, K.; Zhang, H.; Humphreys, W.G.; Pinto, D.; Chen, S.; Bonacorsi, S.; Wong, P.C.; et al. Apixaban Metabolism and Pharmacokinetics after Oral Administration to Humans. Drug Metab. Dispos. 2009, 37, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Skripka, A.I.; Krupenin, P.M.; Kozhanova, O.N.; Kudryavtseva, A.A.; Fedina, L.V.; Akmalova, K.A.; Bochkov, P.O.; Sokolova, A.A.; Napalkov, D.A.; Sychev, D.A. The impact of ABCB1, CYP3A4 and CYP3A5 gene polymorphisms on apixaban trough concentration and bleeding risk in patients with atrial fibrillation. Drug Metab. Pers. Ther. 2024, 39, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Ueshima, S.; Hira, D.; Fujii, R.; Kimura, Y.; Tomitsuka, C.; Yamane, T.; Tabuchi, Y.; Ozawa, T.; Itoh, H.; Horie, M.; et al. Impact of ABCB1, ABCG2, and CYP3A5 polymorphisms on plasma trough concentrations of apixaban in Japanese patients with atrial fibrillation. Pharm. Genom. 2017, 27, 329–336. [Google Scholar] [CrossRef]
- Gopinathannair, R.; Chen, L.Y.; Chung, M.K.; Cornwell, W.K.; Furie, K.L.; Lakkireddy, D.R.; Marrouche, N.F.; Natale, A.; Olshansky, B.; Joglar, J.A.; et al. Managing Atrial Fibrillation in Patients With Heart Failure and Reduced Ejection Fraction: A Scientific Statement From the American Heart Association. Circ. Arrhythmia Electrophysiol. 2021, 14, 688–705. [Google Scholar] [CrossRef]
- Monzo, L.; Baudry, G.; Hernandez, G.; Denquin, O.; Savarese, G.; Lip, G.Y.H.; Girerd, N. Apixaban in patients with nonvalvular atrial fibrillation, heart failure and low body weight: A report from a global federated research dataset. Eur. J. Clin. Investig. 2025, 55, e70012. [Google Scholar] [CrossRef]
- Li, G.; Lip, G.Y.H.; Holbrook, A.; Chang, Y.; Larsen, T.B.; Sun, X.; Tang, J.; Mbuagbaw, L.; Witt, D.M.; Crowther, M.; et al. Direct comparative effectiveness and safety between non-vitamin K antagonist oral anticoagulants for stroke prevention in nonvalvular atrial fibrillation: A systematic review and meta-analysis of observational studies. Eur. J. Epidemiol. 2018, 34, 173–190. [Google Scholar] [CrossRef]
- Archontakis-Barakakis, P.; Li, W.; Kalaitzoglou, D.; Tzelves, L.; Manolopoulos, A.; Giannopoulos, S.; Giamouzis, G.; Giannakoulas, G.; Batsidis, A.; Palaiodimos, L.; et al. Effectiveness and safety of intracranial events associated with the use of direct oral anticoagulants for atrial fibrillation: A systematic review and meta-analysis of 92 studies. Br. J. Clin. Pharmacol. 2022, 88, 4663–4675. [Google Scholar] [CrossRef]
- Kim, P.Y.; Yeh, C.H.; Dale, B.J.; Leslie, B.A.; Stafford, A.R.; Fredenburgh, J.C.; Hirsh, J.; Weitz, J.I. Mechanistic Basis for the Differential Effects of Rivaroxaban and Apixaban on Global Tests of Coagulation. TH Open 2018, 2, e190–e201. [Google Scholar] [CrossRef]
- Ray, W.A.; Chung, C.P.; Stein, C.M.; Smalley, W.; Zimmerman, E.; Dupont, W.D.; Hung, A.M.; Daugherty, J.R.; Dickson, A.; Murray, K.T. Association of Rivaroxaban vs Apixaban With Major Ischemic or Hemorrhagic Events in Patients With Atrial Fibrillation. JAMA 2021, 326, 2395–2404. [Google Scholar] [CrossRef] [PubMed]
- Anderer, S. FDA Approves Generic Form: Of Rivaroxaban for CAD and PAD. JAMA 2025, 333, 1570. [Google Scholar] [CrossRef] [PubMed]
- Helin, T.A.; Lemponen, M.; Immonen, K.; Lakkisto, P.; Joutsi-Korhonen, L. Circulating microRNAs targeting coagulation and fibrinolysis in patients with severe COVID-19. Thromb. J. 2024, 22, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bonde, A.N.; Martinussen, T.; Lee, C.J.-Y.; Lip, G.Y.; Staerk, L.; Bang, C.N.; Bhattacharya, J.; Gislason, G.; Torp-Pedersen, C.; Olesen, J.B.; et al. Rivaroxaban Versus Apixaban for Stroke Prevention in Atrial Fibrillation. Circ. Cardiovasc. Qual. Outcomes 2020, 13, e006058. [Google Scholar] [CrossRef]
- Xiang, Q.; Wang, Z.; Mu, G.; Xie, Q.; Liu, Z.; Zhou, S.; Zhang, H.; Wang, Z.; Jiang, J.; Hu, K.; et al. Genetic variants influenced the risk of bleeding and pharmacodynamics of rivaroxaban in patients with nonvalvular atrial fibrillation: A multicentre prospective cohort study. Clin. Transl. Med. 2023, 13, e1263. [Google Scholar] [CrossRef]
- Lauriol, J.; Keith, K.; Jaffré, F.; Couvillon, A.; Saci, A.; Goonasekera, S.A.; McCarthy, J.R.; Kessinger, C.W.; Wang, J.; Ke, Q.; et al. RhoA signaling in cardiomyocytes protects against stress-induced heart failure but facilitates cardiac fibrosis. Sci. Signal. 2014, 7, ra100. [Google Scholar] [CrossRef] [PubMed]
- Rose, B.A.; Force, T.; Wang, Y. Mitogen-Activated Protein Kinase Signaling in the Heart: Angels Versus Demons in a Heart-Breaking Tale. Physiol. Rev. 2010, 90, 1507–1546. [Google Scholar] [CrossRef] [PubMed]
- Kilian, L.S.; Voran, J.; Frank, D.; Rangrez, A.Y. RhoA: A dubious molecule in cardiac pathophysiology. J. Biomed. Sci. 2021, 28, 1–21. [Google Scholar] [CrossRef]
- Liu, R.; Molkentin, J.D. Regulation of cardiac hypertrophy and remodeling through the dual-specificity MAPK phosphatases (DUSPs). J. Mol. Cell. Cardiol. 2016, 101, 44–49. [Google Scholar] [CrossRef]
- Wang, X.; Nie, X.; Wang, H.; Ren, Z. Roles of small GTPases in cardiac hypertrophy (Review). Mol. Med. Rep. 2024, 30, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Poulakos, M.; Walker, J.N.; Baig, U.; David, T. Edoxaban: A direct oral anticoagulant. Am. J. Heal. Pharm. 2017, 74, 117–129. [Google Scholar] [CrossRef]
- Parasrampuria, D.A.; Truitt, K.E. Pharmacokinetics and Pharmacodynamics of Edoxaban, a Non-Vitamin K Antagonist Oral Anticoagulant that Inhibits Clotting Factor Xa. Clin. Pharmacokinet. 2016, 55, 641–655. [Google Scholar] [CrossRef]
- Ogata, K.; Mendell-Harary, J.; Tachibana, M.; Masumoto, H.; Oguma, T.; Kojima, M.; Kunitada, S. Clinical Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of the Novel Factor Xa Inhibitor Edoxaban in Healthy Volunteers. J. Clin. Pharmacol. 2010, 50, 743–753. [Google Scholar] [CrossRef]
- Kanuri, S.H.; Kreutz, R.P. Pharmacogenomics of Novel Direct Oral Anticoagulants: Newly Identified Genes and Genetic Variants. J. Pers. Med. 2019, 9, 7. [Google Scholar] [CrossRef]
- Guerrero-Orriach, J.L.; Carmona-Luque, M.D.; Rodriguez-Capitan, M.J.; Quesada-Muñoz, G. MicroRNA-197-3p Transfection: Variations in Cardiomyocyte Gene Expression with Anaesthetics Drugs in a Model of Hypoxia/Reperfusion. Pharmaceuticals 2025, 18, 146. [Google Scholar] [CrossRef]
- Ekström, L.; Skilving, I.; Ovesjö, M.; Aklillu, E.; Nylén, H.; Rane, A.; Diczfalusy, U.; Björkhem-Bergman, L. miRNA-27b levels are associated with CYP3A activity in vitro and in vivo. Pharmacol. Res. Perspect. 2015, 3, e00192. [Google Scholar] [CrossRef]
- Pan, Y.-Z.; Gao, W.; Yu, A.-M. MicroRNAs Regulate CYP3A4 Expression via Direct and Indirect Targeting. Drug Metab. Dispos. 2009, 37, 2112–2117. [Google Scholar] [CrossRef]
- Li, G.; Shao, Y.; Guo, H.C.; Zhi, Y.; Qiao, B.; Ma, K.; Du, J.; Lai, Y.Q.; Li, Y. MicroRNA-27b-3p down-regulates FGF1 and aggravates pathological cardiac remodelling. Cardiovasc. Res. 2021, 118, 2139–2151. [Google Scholar] [CrossRef]
- Wang, J.; Song, Y.; Zhang, Y.; Xiao, H.; Sun, Q.; Hou, N.; Guo, S.; Wang, Y.; Fan, K.; Zhan, D.; et al. Cardiomyocyte overexpression of miR-27b induces cardiac hypertrophy and dysfunction in mice. Cell Res. 2011, 22, 516–527. [Google Scholar] [CrossRef]
- Veliceasa, D.; Biyashev, D.; Qin, G.; Misener, S.; Mackie, A.R.; Kishore, R.; Volpert, O.V. Therapeutic manipulation of angiogenesis with miR-27b. Vasc. Cell 2015, 7, 6. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, S.; Gao, Y.; Zhang, S. Serum MicroRNA-27b as a Screening Biomarker for Left Ventricular Hypertrophy. Tex. Heart Inst. J. 2017, 44, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Rincón, L.M.; Rodríguez-Serrano, M.; Conde, E.; Lanza, V.F.; Sanmartín, M.; González-Portilla, P.; Paz-García, M.; Del Rey, J.M.; Menacho, M.; Bermejo, M.G.; et al. Serum microRNAs are key predictors of long-term heart failure and cardiovascular death after myocardial infarction. ESC Heart Fail. 2022, 9, 3367–3379. [Google Scholar] [CrossRef]
- Trabold, K.; Makhoul, S.; Gambaryan, S.; van Ryn, J.; Walter, U.; Jurk, K. The Direct Thrombin Inhibitors Dabigatran and Lepirudin Inhibit GPIbα-Mediated Platelet Aggregation. Thromb. Haemost. 2019, 119, 916–929. [Google Scholar] [CrossRef]
- Stangier, J.; Clemens, A. Pharmacology, Pharmacokinetics, and Pharmacodynamics of Dabigatran Etexilate, an Oral Direct Thrombin Inhibitor. Clin. Appl. Thromb. 2009, 15, 9S–16S. [Google Scholar] [CrossRef] [PubMed]
- Redondo, S.; Martínez, M.-P.; Ramajo, M.; Navarro-Dorado, J.; Barez, A.; Tejerina, T. Pharmacological basis and clinical evidence of dabigatran therapy. J. Hematol. Oncol. 2011, 4, 53. [Google Scholar] [CrossRef]
- Dólleman, S.C.; Agten, S.M.; Spronk, H.M.; Hackeng, T.M.; Bos, M.H.; Versteeg, H.H.; van Zonneveld, A.J.; de Boer, H.C. Thrombin in complex with dabigatran can still interact with PAR-1 via exosite-I and instigate loss of vascular integrity. J. Thromb. Haemost. 2022, 20, 996–1007. [Google Scholar] [CrossRef]
- Woźniak, E.; Broncel, M.; Bukowska, B.; Gorzelak-Pabiś, P. The Protective Effect of Dabigatran and Rivaroxaban on DNA Oxidative Changes in a Model of Vascular Endothelial Damage with Oxidized Cholesterol. Int. J. Mol. Sci. 2020, 21, 1953. [Google Scholar] [CrossRef]
- Paar, V.; Jirak, P.; Gruber, S.; Prodinger, C.; Cadamuro, J.; Wernly, B.; Motloch, L.J.; Haschke-Becher, E.; Hoppe, U.C.; Lichtenauer, M. Influence of dabigatran on pro-inflammatory cytokines, growth factors and chemokines—Slowing the vicious circle of coagulation and inflammation. Life Sci. 2020, 262, 118474. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, X. miRDB: An online database for prediction of functional microRNA targets. Nucleic Acids Res. 2019, 48, D127–D131. [Google Scholar] [CrossRef] [PubMed]
- Ragia, G.; Pallikarou, M.; Michou, C.; Thomopoulos, T.; Chalikias, G.; Trikas, A.; Tziakas, D.N.; Manolopoulos, V.G. Direct oral anticoagulants do not affect miR-27a-3p expression, a regulator of coagulation cascade, in atrial fibrillation patients. J. Thromb. Thrombolysis 2025, 58, 636–645. [Google Scholar] [CrossRef]
- Gheorghiade, M.; Adams, K.F.; Colucci, W.S. Digoxin in the Management of Cardiovascular Disorders. Circulation 2004, 109, 2959–2964. [Google Scholar] [CrossRef]
- Whayne, T.F., Jr. Clinical Use of Digitalis: A State of the Art Review. Am. J. Cardiovasc. Drugs 2018, 18, 427–440. [Google Scholar] [CrossRef]
- Erdmann, E.; Brown, L. The cardiac glycoside-receptor system in the human heart. Eur. Heart J. 1983, 4, 61–65. [Google Scholar] [CrossRef]
- Watanabe, A.M. Digitalis and the autonomic nervous system. JACC 1985, 5, 35A–42A. [Google Scholar] [CrossRef] [PubMed]
- Quan, K.J.; Van Hare, G.F.; Biblo, L.A.; Mackall, J.A.; Carlson, M.D. Endocardial Stimulation of Efferent Parasympathetic Nerves to the Atrioventricular Node in Humans: Optimal Stimulation Sites and the Effects of Digoxin. J. Interv. Card. Electrophysiol. 2001, 5, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Andersson, T.L.; Zygmunt, P.; Vinge, E. Some substances with proposed digitalis-like effects evaluated on platelet functions sensitive for cardiac glycosides. Gen. Pharmacol. Vasc. Syst. 1991, 22, 749–753. [Google Scholar] [CrossRef]
- Chirinos, J.A.; Castrellon, A.; Zambrano, J.P.; Jimenez, J.J.; Jy, W.; Horstman, L.L.; Willens, H.J.; Castellanos, A.; Myerburg, R.J.; Ahn, Y.S. Digoxin use is associated with increased platelet and endothelial cell activation in patients with nonvalvular atrial fibrillation. Heart Rhythm 2005, 2, 525–529. [Google Scholar] [CrossRef] [PubMed]
- Pastori, D.; Carnevale, R.; Nocella, C.; Bartimoccia, S.; Novo, M.; Cammisotto, V.; Piconese, S.; Santulli, M.; Vasaturo, F.; Violi, F.; et al. Digoxin and Platelet Activation in Patients With Atrial Fibrillation: In Vivo and In Vitro Study. J. Am. Heart Assoc. 2018, 7, e009509. [Google Scholar] [CrossRef] [PubMed]
- Eisen, A.; Ruff, C.T.; Braunwald, E.; Hamershock, R.A.; Lewis, B.S.; Hassager, C.; Chao, T.; Le Heuzey, J.Y.; Mercuri, M.; Rutman, H.; et al. Digoxin Use and Subsequent Clinical Outcomes in Patients With Atrial Fibrillation With or Without Heart Failure in the ENGAGE AF-TIMI 48 Trial. J. Am. Heart Assoc. 2017, 6, e006035. [Google Scholar] [CrossRef]
- Pastori, D.; Farcomeni, A.; Bucci, T.; Cangemi, R.; Ciacci, P.; Vicario, T.; Violi, F.; Pignatelli, P. Digoxin treatment is associated with increased total and cardiovascular mortality in anticoagulated patients with atrial fibrillation. Int. J. Cardiol. 2015, 180, 1–5. [Google Scholar] [CrossRef]
- Washam, J.B.; Stevens, S.R.; Lokhnygina, Y.; Halperin, J.L.; Breithardt, G.; E Singer, D.; Mahaffey, K.W.; Hankey, G.J.; Berkowitz, S.D.; Nessel, C.C.; et al. Digoxin use in patients with atrial fibrillation and adverse cardiovascular outcomes: A retrospective analysis of the Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET AF). Lancet 2015, 385, 2363–2370. [Google Scholar] [CrossRef]
- Gona, S.R.; Rosenberg, J.; Fyffe-Freil, R.C.; Kozakiewicz, J.M.; Money, M.E. Review: Failure of current digoxin monitoring for toxicity: New monitoring recommendations to maintain therapeutic levels for efficacy. Front. Cardiovasc. Med. 2023, 10, 1179892. [Google Scholar] [CrossRef]
- Johnson, B.F.; Bye, C.E.; Jones, G.E.; Sabey, G.A. The pharmacokinetics of beta-methyl digoxin compared with digoxin tablets and capsules. Eur. J. Clin. Pharmacol. 1976, 10, 231–236. [Google Scholar] [CrossRef]
- Iisalo, E. Clinical Pharmacokinetics of Digoxin. Clin. Pharmacokinet. 1977, 2, 1–16. [Google Scholar] [CrossRef]
- Matheny, C.J.; Lamb, M.W.; Brouwer, K.L.R.; Pollack, G.M. Pharmacokinetic and Pharmacodynamic Implications of P-glycoprotein Modulation. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2001, 21, 778–796. [Google Scholar] [CrossRef] [PubMed]
- Aronson, J.K. Clinical Pharmacokinetics of Digoxin 1980. Clin. Pharmacokinet. 1980, 5, 137–149. [Google Scholar] [CrossRef]
- Nguyen, L.D.; Wei, Z.; Silva, M.C.; Barberán-Soler, S.; Zhang, J.; Rabinovsky, R.; Muratore, C.R.; Stricker, J.M.S.; Hortman, C.; Young-Pearse, T.L.; et al. Small molecule regulators of microRNAs identified by high-throughput screen coupled with high-throughput sequencing. Nat. Commun. 2023, 14, 1–18. [Google Scholar] [CrossRef]
- Hinkel, R.; Batkai, S.; Bähr, A.; Bozoglu, T.; Straub, S.; Borchert, T.; Viereck, J.; Howe, A.; Hornaschewitz, N.; Oberberger, L.; et al. AntimiR-132 Attenuates Myocardial Hypertrophy in an Animal Model of Percutaneous Aortic Constriction. JACC 2021, 77, 2923–2935. [Google Scholar] [CrossRef]
- Xu, K.; Chen, C.; Wu, Y.; Wu, M.; Lin, L. Advances in miR-132-Based Biomarker and Therapeutic Potential in the Cardiovascular System. Front. Pharmacol. 2021, 12, 751487. [Google Scholar] [CrossRef]
- Ucar, A.; Gupta, S.K.; Fiedler, J.; Erikci, E.; Kardasinski, M.; Batkai, S.; Dangwal, S.; Kumarswamy, R.; Bang, C.; Holzmann, A.; et al. The miRNA-212/132 family regulates both cardiac hypertrophy and cardiomyocyte autophagy. Nat. Commun. 2012, 3, 1078. [Google Scholar] [CrossRef]
- Foinquinos, A.; Batkai, S.; Genschel, C.; Viereck, J.; Rump, S.; Gyöngyösi, M.; Traxler, D.; Riesenhuber, M.; Spannbauer, A.; Lukovic, D.; et al. Preclinical development of a miR-132 inhibitor for heart failure treatment. Nat. Commun. 2020, 11, 1–10. [Google Scholar] [CrossRef]
- Batkai, S.; Genschel, C.; Viereck, J.; Rump, S.; Bär, C.; Borchert, T.; Traxler, D.; Riesenhuber, M.; Spannbauer, A.; Lukovic, D.; et al. CDR132L improves systolic and diastolic function in a large animal model of chronic heart failure. Eur. Heart J. 2020, 42, 192–201. [Google Scholar] [CrossRef]
- Täubel, J.; Hauke, W.; Rump, S.; Viereck, J.; Batkai, S.; Poetzsch, J.; Rode, L.; Weigt, H.; Genschel, C.; Lorch, U.; et al. Novel antisense therapy targeting microRNA-132 in patients with heart failure: Results of a first-in-human Phase 1b randomized, double-blind, placebo-controlled study. Eur. Heart J. 2020, 42, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Bauersachs, J.; Solomon, S.D.; Anker, S.D.; Antorrena-Miranda, I.; Batkai, S.; Viereck, J.; Rump, S.; Filippatos, G.; Granzer, U.; Ponikowski, P.; et al. Efficacy and safety of CDR132L in patients with reduced left ventricular ejection fraction after myocardial infarction: Rationale and design of the HF-REVERT trial. Eur. J. Heart Fail. 2024, 26, 674–682. [Google Scholar] [CrossRef] [PubMed]
- Sjögren, B.; Parra, S.; Atkins, K.B.; Karaj, B.; Neubig, R.R. Digoxin-Mediated Upregulation of RGS2 Protein Protects against Cardiac Injury. J. Pharmacol. Exp. Ther. 2016, 357, 311–319. [Google Scholar] [CrossRef]
- Lin, Y.; He, S.; Feng, R.; Xu, Z.; Chen, W.; Huang, Z.; Liu, Y.; Zhang, Q.; Zhang, B.; Wang, K.; et al. Digoxin-induced anemia among patients with atrial fibrillation and heart failure: Clinical data analysis and drug-gene interaction network. Oncotarget 2017, 8, 57003–57011. [Google Scholar] [CrossRef] [PubMed]
- Koch, V.; Nieger, M.; Bräse, S. Towards the synthesis of calotropin and related cardenolides from 3-epiandrosterone: A-ring related modifications. Org. Chem. Front. 2020, 7, 2670–2681. [Google Scholar] [CrossRef]
- Chen, Z.; Yao, H.; Yao, X.; Zheng, R.; Yang, Y.; Liu, Z.; Zhang, R.; Cheng, Y. Calotropin attenuates ischemic heart failure after myocardial infarction by modulating SIRT1/FOXD3/SERCA2a pathway. Biomed. Pharmacother. 2024, 179, 117384. [Google Scholar] [CrossRef]
- Fala, L. Corlanor (Ivabradine), First HCN Channel Blocker, FDA Approved for the Treatment of Patients with Heart Failure. Am Health Drug Benefits 2016, 9, 56–59. [Google Scholar]
- Yang, J.; Zhao, C.; Yang, L.; Yang, Y.; Wang, N.; Gao, A.; Wang, X. Disproportionality Analysis of Ivabradine in the US FDA Adverse Event Reporting System: A Real-World Study Across Overall and Indication-Specific Populations. Am. J. Cardiovasc. Drugs 2025, 25, 703–713. [Google Scholar] [CrossRef] [PubMed]
- Koruth, J.S.; Lala, A.; Pinney, S.; Reddy, V.Y.; Dukkipati, S.R. The Clinical Use of Ivabradine. JACC 2017, 70, 1777–1784. [Google Scholar] [CrossRef]
- Müller-Werdan, U.; Stöckl, G.; Werdan, K. Advances in the management of heart failure: The role of ivabradine. Vasc. Heal. Risk Manag. 2016, ume 12, 453–470. [Google Scholar] [CrossRef]
- Lauzier, B.; Vaillant, F.; Gélinas, R.; Bouchard, B.; Brownsey, R.; Thorin, E.; Tardif, J.-C.; Rosiers, C.D. Ivabradine reduces heart rate while preserving metabolic fluxes and energy status of healthy normoxic working hearts. Am. J. Physiol. Circ. Physiol. 2011, 300, H845–H852. [Google Scholar] [CrossRef]
- Lin, Y.; Jan, J.; Chang, J.; Lin, M.; Yang, T.; Wang, P.; Chen, M. Ivabradine in heart failure patients with reduced ejection fraction and history of paroxysmal atrial fibrillation. ESC Heart Fail. 2022, 9, 2548–2557. [Google Scholar] [CrossRef]
- Wang, J.; Yang, Y.; Li, Y.; Zhu, J.; Lian, H.; Shao, X.; Zhang, H.; Fu, Y.; Zhang, L. Long-term treatment with ivabradine in transgenic atrial fibrillation mice counteracts hyperpolarization-activated cyclic nucleotide gated channel overexpression. J. Cardiovasc. Electrophysiol. 2018, 30, 242–252. [Google Scholar] [CrossRef]
- Gomes, F.A.; Noronha, S.I.; Silva, S.C.; Machado-Júnior, P.A.; Ostolin, T.L.; Chírico, M.T.; Ribeiro, M.C.; Reis, A.B.; Cangussú, S.D.; Montano, N.; et al. Ivabradine treatment lowers blood pressure and promotes cardiac and renal protection in spontaneously hypertensive rats. Life Sci. 2022, 308, 120919. [Google Scholar] [CrossRef]
- Shao, S.; Zhang, Y.; Gong, M.; Yang, Q.; Yuan, M.; Yuan, M.; Suo, Y.; Wang, X.; Li, Y.; Bao, Q.; et al. Ivabradine Ameliorates Cardiac Function in Heart Failure with Preserved and Reduced Ejection Fraction via Upregulation of miR-133a. Oxidative Med. Cell. Longev. 2021, 2021, 1257283. [Google Scholar] [CrossRef]
- Yanni, J.; D’sOuza, A.; Wang, Y.; Li, N.; Hansen, B.J.; Zakharkin, S.O.; Smith, M.; Hayward, C.; Whitson, B.A.; Mohler, P.J.; et al. Silencing miR-370-3p rescues funny current and sinus node function in heart failure. Sci. Rep. 2020, 10, 1–23. [Google Scholar] [CrossRef]
- Miziuk, V.M. Effect of ivabradin and clopidogrel on platelet hemostasis indices in patients with acute coronary syndrome without ST-segment elevation and with concurrent cardiac insufficiency. Likars’ Ka Sprav. 2008, 3–4, 71–75. [Google Scholar]
- Huang, X.; Liu, C.; Sharma, S.N.; You, X.; Chen, S.; Li, Y.; Liu, H.-J.; Liu, B.; Saiding, Q.; Chen, W.; et al. Oral delivery of liquid mRNA therapeutics by an engineered capsule for treatment of preclinical intestinal disease. Sci. Transl. Med. 2025, 17, eadu1493. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spartano, L.; Lombardi, M.; Foglieni, C. Anticoagulation Therapies and microRNAs in Heart Failure. Biomolecules 2025, 15, 1411. https://doi.org/10.3390/biom15101411
Spartano L, Lombardi M, Foglieni C. Anticoagulation Therapies and microRNAs in Heart Failure. Biomolecules. 2025; 15(10):1411. https://doi.org/10.3390/biom15101411
Chicago/Turabian StyleSpartano, Lucia, Maria Lombardi, and Chiara Foglieni. 2025. "Anticoagulation Therapies and microRNAs in Heart Failure" Biomolecules 15, no. 10: 1411. https://doi.org/10.3390/biom15101411
APA StyleSpartano, L., Lombardi, M., & Foglieni, C. (2025). Anticoagulation Therapies and microRNAs in Heart Failure. Biomolecules, 15(10), 1411. https://doi.org/10.3390/biom15101411





