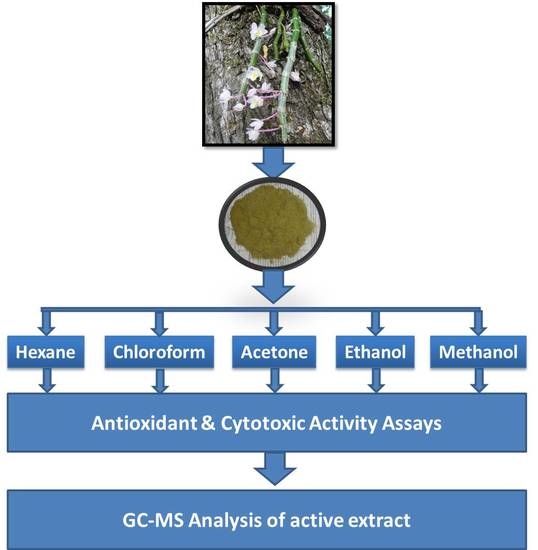
Assessment of Antioxidant and Cytotoxic Activities of Extracts of Dendrobium crepidatum
Abstract
:1. Introduction
2. Results and Discussion
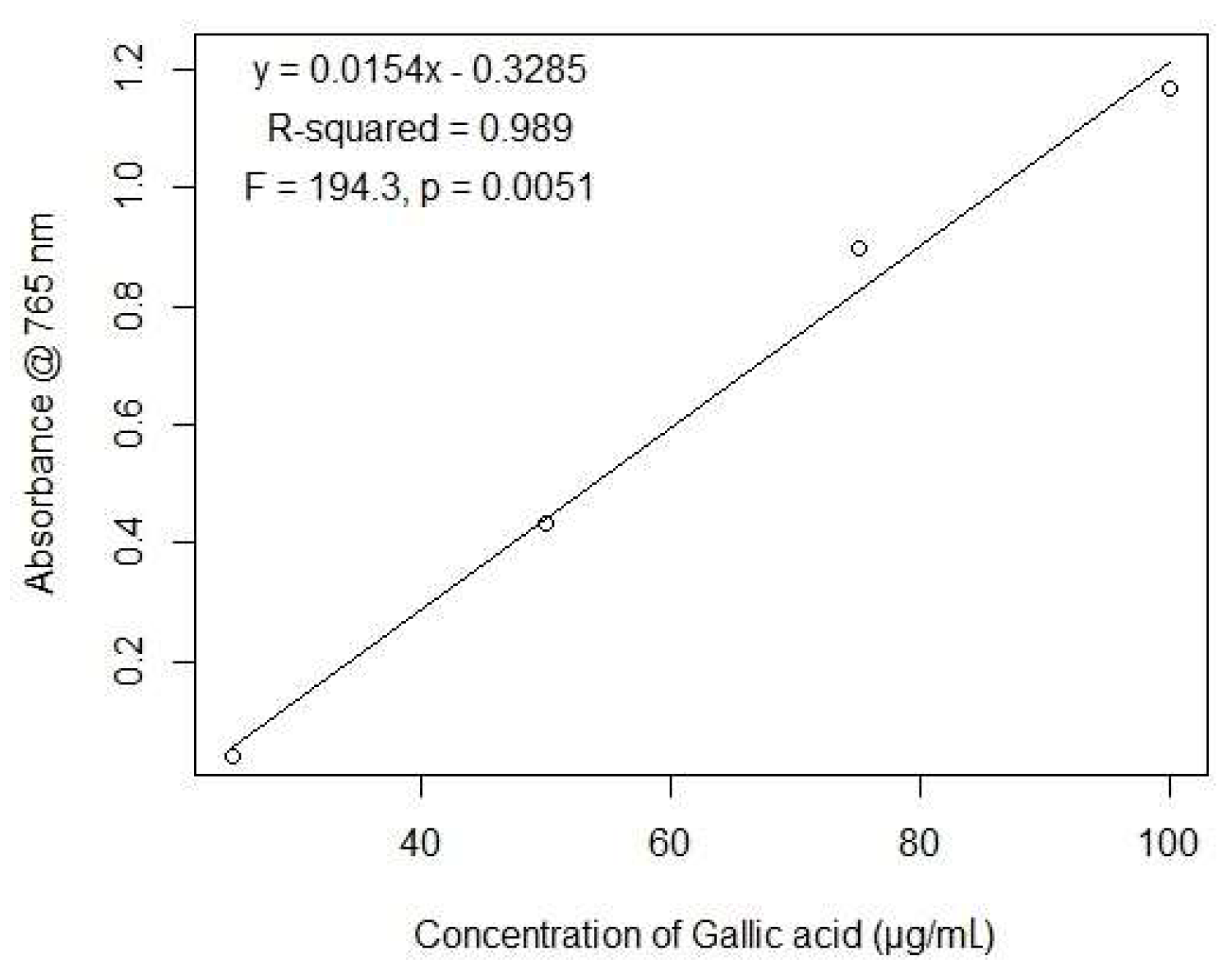
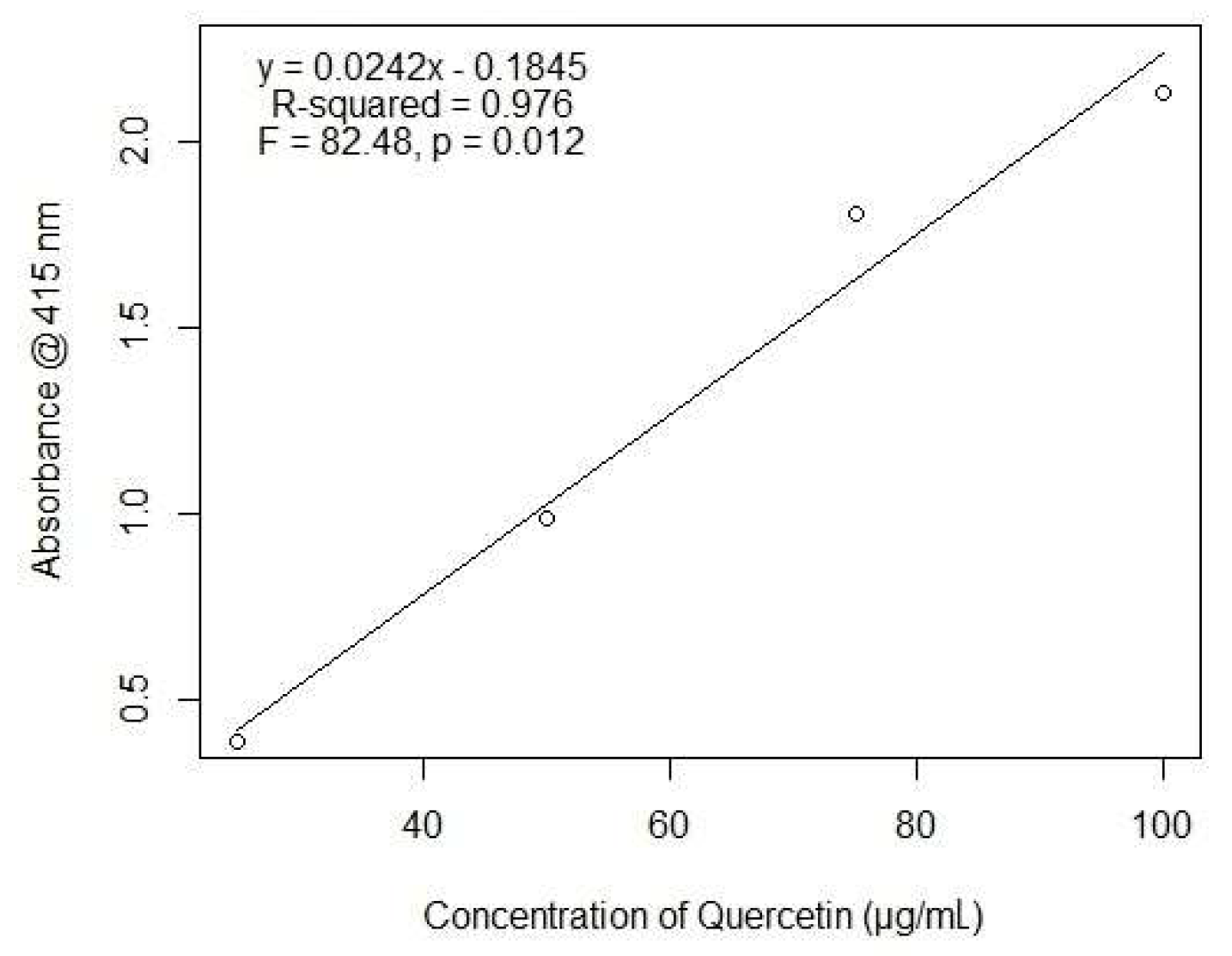
2.1. Total Polyphenols Content
2.2. DPPH Free-Radical Scavenging Potential and Antioxidant Capacity
2.3. Cytotoxic Activity toward HeLa and U251 Cell Lines
2.4. Detection and Identification of Compounds by GC–MS
3. Conclusions
4. Materials and Methods
4.1. Plant Materials
4.2. Preparation of Extracts
4.3. Quantification of Total Polyphenol Content
4.3.1. Quantification of Total Phenolic Content
4.3.2. Quantification of Total Flavonoid Content
4.4. Evaluation of Antioxidant Properties
4.5. Evaluation of Cytotoxic Properties
4.5.1. Human Cancer Cell Culture
4.5.2. MTT Assay
4.6. Identification of Compounds by GC–MS
4.7. Statistical Analysis
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Rajbhandari, K.R. Orchids of Nepal: Status, threat and conservation. In Proceedings of the National Workshop on NTFP/MAPs Sector Action Plant Development: Orchid, Kathmandu, Nepal, 9 July 2014; Department of Plant Resources, Ministry of Forest and Soil Conservation and Central Department of Botany, Tribhuvan University: Kathmandu, Nepal, 2014; pp. 1–40. [Google Scholar]
- Lam, Y.; Ng, T.B.; Yao, R.M.; Shi, J.; Xu, K.; Sze, S.C.W.; Zhang, K.Y. Evaluation of chemical constituents and important mechanism of pharmacological biology in Dendrobium plants. Evid.-Based Complement. Altern. Med. 2015, 2015, 841752. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Han, Q.B.; Li, S.L.; Chen, X.J.; Wang, X.N.; Zhao, Z.Z.; Chen, H.B. Chemistry, bioactivity and quality control of Dendrobium, a commonly used tonic herb in traditional Chinese medicine. Phytochem. Rev. 2013, 12, 341–367. [Google Scholar] [CrossRef]
- Elander, M.; Leander, K.; Rosenblom, J.; Ruusa, E. Crepidine, Crepidamine and Dendrocrepine, Three Alkaloids from Dendrobium crepidatum Lindl. Acta Chem. Scand. 1973, 27, 1907–1913. [Google Scholar] [CrossRef] [PubMed]
- Kierkegaard, P.; Pilotto, A.M.; Leander, K. The Constitution and Relative Configuration of Crepidine, an Alkaloid from Dendrobium crepidatum Lindl. Acta Chem. Scand. 1970, 24, 3757–3759. [Google Scholar] [CrossRef]
- Majumder, P.L.; Chatterjee, S. Crepidatin, a bibenzyl derivative from the orchid Dendrobium crepidatum. Phytochemistry 1989, 28, 1986–1988. [Google Scholar] [CrossRef]
- Hu, Y.; Zhang, C.; Zhao, X.; Wang, Y.; Feng, D.; Zhang, M.; Xie, H. (±)-Homocrepidine A, a Pair of Anti-inflammatory Enantiomeric Octahydroindolizine Alkaloid Dimers from Dendrobium crepidatum. J. Nat. Prod. 2016, 79, 252–256. [Google Scholar] [CrossRef]
- Li, C.B.; Wang, C.; Fan, W.W.; Dong, F.W.; Xu, F.Q.; Wan, Q.L.; Luo, H.R.; Liu, Y.Q.; Hu, J.M.; Zhou, J. Chemical components of Dendrobium crepidatum and their neurite outgrowth enhancing activities. Nat. Prod. Bioprospect. 2013, 3, 70–73. [Google Scholar] [CrossRef]
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Nita, M.; Grzybowski, A. The Role of the Reactive Oxygen Species and Oxidative Stress in the Pathomechanism of the Age-Related Ocular Diseases and Other Pathologies of the Anterior and Posterior Eye Segments in Adults. Oxid. Med. Cell. Longev. 2016, 2016, 3164734. [Google Scholar] [CrossRef]
- Tiwary, B.K.; Dutta, S.; Dey, P.; Hossain, M.; Kumar, A.; Bihani, S.; Nanda, A.K.; Chaudhuri, T.K.; Chakraborty, R. Radical Scavenging Activities of Lagerstroemia speciosa (L.) Pers. Petal Extracts and its hepato-protection in CCl4-intoxicated mice. BMC Complement. Altern. Med. 2017, 17, 55. [Google Scholar] [CrossRef] [PubMed]
- Sayin, V.I.; Ibrahim, M.X.; Larsson, E.; Nilsson, J.A.; Lindahl, P.; Bergo, M.O. Antioxidants accelerate lung cancer progression in mice. Sci. Transl. Med. 2014, 6, 221ra15. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Tang, X.L. Natural products against cancer: A comprehensive bibliometric study of the research projects, publications, patents and drugs. J. Cancer Res. Ther. 2014, 10, 27–37. [Google Scholar] [PubMed]
- Zhang, Y.; Luo, M.; Zu, Y.; Fu, Y.; Gu, C.; Wang, W.; Yao, L.; Efferth, T. Dryofragin, a phloroglucinol derivative, induces apoptosis in human breast cancer MCF-7 cells through ROS-mediated mitochondrial pathway. Chem. Biol. Interact. 2012, 199, 129–136. [Google Scholar] [CrossRef]
- Kangsamaksin, T.; Chaithongyot, S.; Wootthichairangsan, C.; Hanchaina, R.; Tangshewinsirikul, C.; Svasti, J. Lupeol and stigmasterol suppress tumor angiogenesis and inhibit cholangiocarcinoma growth in mice via downregulation of tumor necrosis factor-α. PLoS ONE 2017, 12, e0189628. [Google Scholar] [CrossRef]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.D.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef]
- Nimse, S.B.; Pal, D. Free radicals, natural antioxidants, and their reaction mechanisms. RSC Adv. 2015, 5, 27986–28006. [Google Scholar] [CrossRef] [Green Version]
- Phaniendra, A.; Jestadi, D.B.; Periyasamy, L. Free Radicals: Properties, Sources, Targets, and Their Implication in Various Diseases. Indian J. Clin. Biochem. 2015, 30, 11–26. [Google Scholar] [CrossRef]
- Jiménez, N.; Hormaza, L.C.; Pujol, A.; Álzate, F.; Osorio, E.; Guzman, O.L. Antioxidant capacity and phenolic content of commonly used anti-inflammatory medicinal plants in Colombia. Ind. Crops Prod. 2015, 70, 272–279. [Google Scholar] [CrossRef]
- Al-Muniri, R.M.S.; Hossain, M.A. Evaluation of antioxidant and cytotoxic activities of different extracts of folk medicinal plant Hapllophyllum tuberculatum. Egypt. J. Basic Appl. Sci. 2017, 4, 101–106. [Google Scholar] [CrossRef]
- Aksoy, L.; Kolay, E.; Aĝilönü, Y.; Aslan, Z.; Kargioĝlu, M. Free radical scavenging activity, total phenolic content, total antioxidant status, and total oxidant status of endemic Thermopsis turcica. Saudi J. Biol. Sci. 2013, 20, 235–239. [Google Scholar] [CrossRef]
- Paudel, M.R.; Chand, M.B.; Karki, N.; Pant, B. Antioxidant activity and total phenolic and flavonoid contents of Dendrobium amoenum Wall. ex Lindl. Bot. Orient. J. Plant Sci. 2015, 9, 20–26. [Google Scholar] [CrossRef]
- Saeed, N.; Khan, M.R.; Shabbir, M. Antioxidant activity, total phenolic and total flavonoid contents of whole plant extracts Torilis leptophylla L. BMC Complement. Altern. Med. 2012, 12, 221. [Google Scholar] [CrossRef]
- Dent, M.; Dragovic, V.; Penic, M.; Brncic, M.; Bosiljkov, T.; Levaj, B. The Effect of Extraction Solvents, Temperature and Time on the Composition and Mass Fraction of Polyphenols in Dalmatian Wild Sage (Salvia officinalis L.) Extracts. Food Technol. Biotechnol. 2013, 51, 84–91. [Google Scholar]
- Boeing, J.S.; Barizão, É.O.; e Silva, B.C.; Montanher, P.F.; de Cinque Almeida, V.; Visentainer, J.V. Evaluation of solvent effect on the extraction of phenolic compounds and antioxidant capacities from the berries: Application of principal component analysis. Chem. Cent. J. 2014, 8, 48. [Google Scholar] [CrossRef]
- Chand, M.B.; Paudel, M.R.; Pant, B. The antioxidant activity of selected wild orchids of Nepal. J. Coast. Life Med. 2016, 4, 731–736. [Google Scholar]
- Fu, L.; Xu, B.T.; Xu, X.R.; Gan, R.Y.; Zhang, Y.; Xia, E.Q.; Li, H.B. Antioxidant capacities and total phenolic contents of 62 fruits. Food Chem. 2011, 129, 345–350. [Google Scholar] [CrossRef]
- Habib, R.; Rana, S.; Hasan, R.; Imam, Z.; Hasan, R.; Saha, A. Antioxidant, Cytotoxicity and Antibacterial Potential of Different Extract of Geodorum densiflorum (Lam.) Schltr. Root. J. Herb. Med. Toxicol. 2011, 5, 63–70. [Google Scholar]
- Stankovic, M.S.; Jakovljevic, D.; Topuzovic, M.; Zlatkovic, B. Antioxidant activity and contents of phenolics and flavonoids in the whole plant and plant parts of Teucrium botrys L. Oxid. Commun. 2014, 37, 522–532. [Google Scholar]
- Paudel, M.R.; Chand, M.B.; Pant, B.; Pant, B. Antioxidant and cytotoxic activities of Dendrobium moniliforme extracts and the detection of related compounds by GC-MS. BMC Complement. Altern. Med. 2018, 18, 134. [Google Scholar] [CrossRef]
- Paudel, M.R.; Chand, M.B.; Pant, B.; Pant, B. Cytotoxic activity of antioxidant-riched Dendrobium longicornu. Pharmacogn. J. 2017, 9, 499–503. [Google Scholar] [CrossRef]
- Kedare, S.B.; Singh, R.P. Genesis and development of DPPH method of antioxidant assay. J. Food Sci. Technol. 2011, 48, 412–422. [Google Scholar] [CrossRef] [Green Version]
- Bondet, V.; Brand-Williams, W.; Berset, C. Kinetics and mechanisms of antioxidant activity using the DPPH•free radical method. LWT Food Sci. Technol. 1997, 30, 609–615. [Google Scholar] [CrossRef]
- Mensor, L.L.; Menezes, F.S.; Leitao, G.G.; Reis, A.S.; dos Santos, T.C.; Coube, C.S.; Leitao, S.G. Screening of Brazilian plant extracts for antioxidant activity by the use of DPPH free radical method. Phyther. Res. 2001, 15, 127–130. [Google Scholar] [CrossRef]
- Tiveron, A.P.; Melo, P.S.; Bergamaschi, K.B.; Vieira, T.M.F.S.; Regitano-d’Arce, M.A.B.; Alencar, S.M. Antioxidant activity of Brazilian vegetables and its relation with phenolic composition. Int. J. Mol. Sci. 2012, 13, 8943–8957. [Google Scholar] [CrossRef]
- Ozgen, M.; Reese, R.N.; Tulio, A.Z.; Scheerens, J.C.; Miller, A.R. Modified 2,2-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) method to measure antioxidant capacity of selected small fruits and comparison to ferric reducing antioxidant power (FRAP) and 2,2′-diphenyl-1- picrylhydrazyl (DPPH) methods. J. Agric. Food Chem. 2006, 54, 1151–1157. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Prasad, R.; Koch, B. Antitumor activity of ethanolic extract of Dendrobium formosum in T-cell lymphoma: An in vitro and in vivo study. Biomed Res. Int. 2014, 2014, 753451. [Google Scholar] [CrossRef]
- Peng, C.; Qi, X.M.; Miao, L.L.; Ren, J. 1,2:5,6-dianhydrogalactitol inhibits human glioma cell growth in vivo and in vitro by arresting the cell cycle at G2/M phase. Acta Pharmacol. Sin. 2017, 38, 1–10. [Google Scholar] [CrossRef]
- Chen, Y.; Li, Y.; Qing, C.; Zhang, Y.; Wang, L.; Liu, Y. 1,4,5-Trihydroxy-7-methoxy-9H-fluoren-9-one, a new cytotoxic compound from Dendrobium chrysotoxum. Food Chem. 2008, 108, 973–976. [Google Scholar] [CrossRef]
- Gali-Muhtasib, H.; Hmadi, R.; Kareh, M.; Tohme, R.; Darwiche, N. Cell death mechanisms of plant-derived anticancer drugs: Beyond apoptosis. Apoptosis 2015, 20, 1531–1562. [Google Scholar] [CrossRef]
- Paudel, M.R.; Pant, B. Cytotoxic activity of crude extracts of Dendrobium amoenum and detection of bioactive compounds by GC-MS. Bot. Orient. J. Plant Sci. 2017, 11, 38–42. [Google Scholar] [CrossRef]
- Milutinović, M.G.; Stanković, M.S.; Cvetković, D.M.; Topuzović, M.D.; Mihailović, V.B.; Marković, S.D. Antioxidant and anticancer properties of leaves and seed cones from European yew (Taxus baccata L.). Arch. Biol. Sci. 2015, 67, 525–534. [Google Scholar] [CrossRef]
- Uddin, S.J.; Grice, D.; Tiralongo, E. Evaluation of cytotoxic activity of patriscabratine, tetracosane and various flavonoids isolated from the Bangladeshi medicinal plant Acrostichum aureum. Pharm. Biol. 2012, 50, 1276–1280. [Google Scholar] [CrossRef]
- Abubakar, M.N.; Majinda, R.T. GC-MS Analysis and Preliminary Antimicrobial Activity of Albizia adianthifolia (Schumach) and Pterocarpus angolensis (DC). Medicines 2016, 3, 3. [Google Scholar] [CrossRef]
- Stankovic, M.S. Total phenolic content, flavonoid concentration and antioxidant activity of Marrubium peregrinum L. extracts. Kragujev. J. Sci. 2011, 33, 63–72. [Google Scholar]
- Zhao, G.R.; Xiang, Z.J.; Ye, T.X.; Yuan, Y.J.; Guo, Z.X. Antioxidant activities of Salvia miltiorrhiza and Panax notoginseng. Food Chem. 2006, 99, 767–774. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Sargent, J.M.; Taylor, C.G. Appraisal of the MTT assay as a rapid test of chemosensitivity in acute myeloid leukaemia. Br. J. Cancer 1989, 60, 206–210. [Google Scholar] [CrossRef]
| Extract | TPC (µg GAE/mg Extract) | TFC (µg QE/mg Extract) |
|---|---|---|
| DCH | 45.34 ± 5.60c | 82.62 ± 1.13a |
| DCC | 37.18 ± 0.70d | 53.36 ± 0.83d |
| DCA | 61.27 ± 3.55b | 71.93 ± 1.59b |
| DCE | 78.11 ± 0.72a | 61.57 ± 3.46c |
| DCM | 28.89 ± 2.11e | 40.14 ± 0.25e |
| Plant Extract | Concentration of Extract (µg/mL) | ||||
|---|---|---|---|---|---|
| 50 | 100 | 200 | 400 | 800 | |
| DCH | 35.14 ± 2.89b | 41.43 ± 5.08b | 50.31 ± 6.20b | 60.01 ± 13.55b | 88.17 ± 5.60a |
| DCC | 20.13 ± 4.65c | 23.43 ± 6.22c | 42.71 ± 3.98c | 59.36 ± 3.53b | 74.11 ± 11.34b |
| DCA | 34.54 ± 4.62b | 46.31 ± 1.74b | 78.53 ± 0.59a | 92.76 ± 1.86a | 93.41 ± 0.86a |
| DCE | 44.02 ± 7.22a | 64.31 ± 0.62a | 84.36 ± 2.47a | 94.10 ± 0.26a | 94.69 ± 0.10a |
| DCM | 40.03 ± 1.24ab | 43.90 ± 2.46b | 55.42 ± 2.43b | 69.92 ± 0.22b | 85.23 ± 5.57ab |
| Plant Extracts and Ascorbic Acid (AA) | ||||||
|---|---|---|---|---|---|---|
| DCH | DCC | DCA | DCE | DCM | AA | |
| IC50 | 306.77 ± 51.14* | 277.08 ± 27.80* | 99.35 ± 2.40 | 74.00 ± 5.75 | 186.39 ± 72.79* | 38.21 ± 2.00 |
| Plant Extract | Concentration of Extract (µg/mL) | |||
|---|---|---|---|---|
| 100 | 200 | 400 | 800 | |
| DCH | 19.84 ± 4.31c | 31.96 ± 3.57c | 58.16 ± 0.71c | 62.10 ± 5.04c |
| DCC | 20.96 ± 1.67c | 24.56 ± 1.17d | 55.25 ± 3.95c | 81.49 ± 0.43a |
| DCA | 25.97 ± 1.90bc | 42.06 ± 2.28b | 74.35 ± 0.59a | 78.21 ± 2.53ab |
| DCE | 30.05 ± 1.87ab | 48.66 ± 0.10a | 52.89 ± 1.48c | 59.55 ± 0.63c |
| DCM | 35.61 ± 3.92a | 47.40 ± 1.24a | 67.01 ± 1.97b | 73.05 ± 1.39b |
| Plant Extract | Concentration of Extract (µg/mL) | |||
|---|---|---|---|---|
| 100 | 200 | 400 | 800 | |
| DCH | 16.35 ± 4.26b | 18.26 ± 0.83b | 70.79 ± 9.72a | 76.45 ± 4.26a |
| DCC | 9.76 ± 2.71c | 23.02 ± 11.33b | 39.96 ± 8.25c | 71.24 ± 10.98a |
| DCA | 20.82 ± 4.14a | 16.94 ± 1.65b | 41.06 ± 14.03c | 52.53 ± 6.60c |
| DCE | 15.31 ± 5.73b | 26.89 ± 6.48b | 49.62 ± 23.55b | 61.28 ± 11.96b |
| DCM | 21.69 ± 1.22a | 44.43 ± 2.99a | 54.36 ± 0.21b | 64.41 ± 6.49b |
| Plant Extracts and Cisplatin (CDDP) | ||||||
|---|---|---|---|---|---|---|
| DCH | DCC | DCA | DCE | DCM | CDDP | |
| IC50 HeLa | 325.38 ± 5.03* | 369.16 ± 3.03* | 229.27 ± 3.80* | 223.07 ± 2.86* | 194.14 ± 4.94* | 25.00 ± 0.5 |
| IC50 U251 | 314.70 ± 4.20* | 539.58 ± 1.04* | 687.34 ± 6.83* | 558.63 ± 3.61* | 301.99 ± 2.00* | 25.00 ± 0.5 |
| RT (min) | Compound Name | Peak Area Percentage in the Extract | ||||
|---|---|---|---|---|---|---|
| DCH | DCC | DCA | DCE | DCM | ||
| 4.060 | Dimethylsulfoxonium formylmethylide | 7.15 | 14.94 | |||
| 4.253 | 2-Phenylethanol | 0.94 | ||||
| 4.261 | 1-Butoxy-2-propanol acetate | 4.14 | ||||
| 4.340 | 2,3-dihydro-3,5-dihydroxy-6-methyl-4H-Pyran-4-one | 5.21 | 13.60 | |||
| 4.375 | 2,5-Dimethyl-4-hydroxy-3(2H)-furanone | 4.56 | ||||
| 4.585 | (l)-alpha-Terpineol | 1.12 | ||||
| 4.652 | (−)-alpha-Terpineol | 0.86 | ||||
| 4.806 | 2-Furaldehyde | 5.78 | ||||
| 5.217 | 5-(Hydroxymethyl)- 2-Furancarboxaldehyde | 21.90 | 1.39 | |||
| 5.282 | 2-Methoxy-4-vinylphenol | 0.84 | 2.22 | |||
| 5.430 | 4-methyl-1-naphthalenol | 0.40 | 0.99 | |||
| 5.458 | 4-Methyl-1-naphthol | 0.97 | 1.20 | |||
| 5.528 | Eugenol | 1.05 | ||||
| 5.672 | 2-Methoxy-5-(1-propenyl)- phenol | 0.79 | ||||
| 6.286 | N,N’-Dibutylidene-hydrazine | 0.46 | ||||
| 6.768 | p-Mesyloxyphenol | 0.49 | ||||
| 7.285 | , 2,6-Dimethoxy-4-(2-propenyl)-phenol | 1.08 | ||||
| 7.362 | 1,7-Trimethylene-2,3,5-trimethylindole | 3.93 | ||||
| 7.370 | 1-(3,4-Dihydro-1-naphthalenyl)- Pyrrolidine | 4.22 | 1.04 | |||
| 7.495 | 3-(2-Phenylethylamino)-5-(4-methylphenyl)-2-cyclohexen-1-one | 1.25 | ||||
| 7.740 | 2-Benzylhexahydropyrrolizin-3-one | 1.03 | 1.07 | |||
| 8.830 | 5-Acetyl-4-amino-3-(2-N-pyrrolidinylethylthio) thieno [3,2-d]isothiazole | 2.58 | ||||
| 8.841 | 2-t-Butyl-4-quinolinealdehyde | 6.54 | ||||
| 8.851 | 2,3-Dihydro-2,4,8-trimethyl-furo [2,3-b] quinoline | 5.82 | 0.99 | |||
| 8.861 | 1,3,4,10-Tetrahydro-2-methyl-9(2H)-Acridinone | 5.96 | ||||
| 9.097 | 1,2-Benzenedicarboxylic acid, bis (2-methylpropyl) ester | 2.05 | 1.38 | 1.19 | 16.08 | |
| 9.099 | Diisobutyl phthalate | 3.73 | ||||
| 9.547 | 4-Acetyl-3-amino-5-butyl-2,4-cyclopentadiene-1,1,2-tricarbonitrile | 2.09 | 3.40 | |||
| 9.551 | N-(N’-Ethoxycarbonylisonipecotinoyl)-isonipecotic acid ethyl ester | 3.78 | ||||
| 9.611 | 2,3,9-Trimethyl-5-trimethylsilyloxy-2-azabicyclodecane | 1.44 | ||||
| 9.680 | 2-Butoxy-5-(4-methylphenyl)- benzaldehyde | 2.26 | ||||
| 9.687 | 5,7-Dimethyl-9-hydroxy-2,3-dihydro-1H-cyclopenta [b] quinoline | 3.45 | 4.30 | |||
| 9.724 | 7-Phenyl-1,5-dihydro-imidazo [4,5-d] pyridazin-4-one | 1.64 | ||||
| 9.733 | Dibutyl phthalate | 2.79 | ||||
| 9.801 | 1-Heneicosanol | 0.69 | ||||
| 10.837 | 2,4-Dimethyl-3a-phenyldecahydrofuro[3,2-E] indolizin2-ol | 5.23 | ||||
| 11.191 | 1-Nonadecanol | 1.10 | ||||
| 11.415 | 9,12-Octadecadienoic acid, methyl ester | 17.27 | ||||
| 11.574 | 6-Isopentyl-2-tert-pentyl-tryptophan methyl ester | 1.14 | ||||
| 11.592 | 2-(1,1-Dimethylpropyl)-6-(3-methylbutyl)-tryptophan methyl ester | 2.21 | 0.32 | |||
| 11.899 | 2-Dodecyl-5-methylpyrrolidine | 1.60 | ||||
| 11.997 | Octahydro-2H-Inden-2-one, cis-oxime | 1.44 | ||||
| 12.175 | 5-sec-butylpyrogallol | 5.54 | ||||
| 12.187 | 1,2-Dihydro-2-(2-oxocyclohexylidene) quinoline | 5.02 | ||||
| 12.269 | Isocrepidamine | 2.80 | 2.28 | |||
| 12.326 | 1-(6-Hydroxy-7-methyl-6-phenyloctahydro-5-indolizinyl) acetone | 3.73 | ||||
| 12.438 | 1-Eicosanol, trifluoroacetate | 0.70 | ||||
| 12.728 | 2-(Diethylamino)-7-methyl-7phenyl-2,4-cycloheptadien-1-one | 2.26 | ||||
| 12.802 | 2,4-Cycloheptadien-1-one, 2-(diethylamino)-7-methyl-7-phenyl | 2.37 | 0.36 | |||
| 12.915 | 3-Bornanone, oxime | 1.89 | ||||
| 12.931 | 3,12-Didehydro-9,10-dimethoxy-galanthan-1,2-diol | 4.66 | ||||
| 13.064 | iso-Propyl 9-cis,11-trans-octadecadienoate | 5.48 | ||||
| 13.092 | 4-Methoxy-6-(1-pyrrolidinylmethyl)-1,3,5-triazin-2-amine | 6.48 | ||||
| 13.093 | 2-Hydrazino-6-methylpyrimidin-4-ol | 7.01 | ||||
| 13.125 | Tetracosane | 3.74 | ||||
| 13.135 | Crepidamine | 3.79 | 10.17 | 4.50 | 1.43 | |
| 13.139 | 9-Methoxy-9-Borabicyclo [3.3.1] nonane | 1.32 | ||||
| 13.190 | Gephyrotoxin 207a | 3.51 | 0.64 | |||
| 13.253 | 2-Methoxy-3-(1-methylpropyl)- pyrazine | 3.14 | ||||
| 13.399 | 1-(2-Hydroxy-3,4-dimethoxyphenyl)-2-(4-methoxyphenyl) ethanone | 5.20 | ||||
| 13.410 | 1-[2-(5-Hydroxy-1,1-dimethylhexyl)-3-methyl-2-cyclopropen-1-yl]-ethanone | 2.93 | ||||
| 13.464 | N-(9H-Xanthen-9-yl)- 4-pyridinecarbothioamide | 3.25 | ||||
| 13.519 | 2-Hydroxy-3,4-dimethoxy-alpha-(p-methoxyphenyl) acetophenone | 4.15 | 0.54 | |||
| 13.537 | (−)-Nortrachelogenin | 2.60 | ||||
| 13.793 | Heneicosane | 1.25 | ||||
| 13.942 | Crepidine | 3.19 | 4.87 | 7.08 | 0.37 | 0.56 |
| 14.041 | Dimethoxymethylphenylsilane | 3.30 | 1.75 | |||
| 14.544 | n-Tetratriacontane | 3.42 | ||||
| 14.599 | Tetratriacontane | 8.10 | ||||
| 14.767 | Monolinolein | 0.67 | ||||
| 14.791 | n-Propyl 9,12-octadecadienoate | 3.01 | ||||
| 14.913 | cis-3,3,5-Trimethyl-cyclohexanol acetate | 0.87 | ||||
| 15.019 | 6-(6-Acetonyl-5-hydroxy-1,4-dimethyl-5-phenyl-2-piperidyl)-3-hexen-2-one | 1.34 | ||||
| 15.240 | Hexatriacontane | 0.90 | ||||
| 15.796 | Oxalic acid, di-(1-menthyl) ester | 2.07 | ||||
| 15.945 | n-Hexatriacontane | 1.22 | ||||
| 15.960 | Triacontane | 2.65 | ||||
| 16.050 | 1-Heptacosanol | 1.44 | 0.75 | |||
| 17.611 | gamma-Tocopherol | 1.37 | ||||
| 18.477 | alpha-Tocopherol-beta-d-mannoside | 2.19 | ||||
| 19.908 | (3beta)-ergost-5-en-3-ol | 0.85 | ||||
| 20.355 | Stigmasterol | 2.03 | ||||
| 21.115 | 2-(1-methylimidazol-4-yl)-1,2,3,4-tetrahydroquinoline | 7.44 | ||||
| 21.159 | 1,2,3,4-Tetrahydro-2-(1-methylimidazol-4-yl)-quinoline | 2.53 | 2.70 | |||
| 21.424 | Estrone-3-acetate-17-methyloxime | 3.98 | 1.05 | |||
| 23.109 | dl-alpha-Tocopherol | 2.66 | ||||
| 24.466 | 3,4,5-Trimethoxy-nitrobenzene | 0.60 | 1.69 | 2.62 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paudel, M.R.; Chand, M.B.; Pant, B.; Pant, B. Assessment of Antioxidant and Cytotoxic Activities of Extracts of Dendrobium crepidatum. Biomolecules 2019, 9, 478. https://doi.org/10.3390/biom9090478
Paudel MR, Chand MB, Pant B, Pant B. Assessment of Antioxidant and Cytotoxic Activities of Extracts of Dendrobium crepidatum. Biomolecules. 2019; 9(9):478. https://doi.org/10.3390/biom9090478
Chicago/Turabian StylePaudel, Mukti Ram, Mukesh Babu Chand, Basant Pant, and Bijaya Pant. 2019. "Assessment of Antioxidant and Cytotoxic Activities of Extracts of Dendrobium crepidatum" Biomolecules 9, no. 9: 478. https://doi.org/10.3390/biom9090478