Aortic Oxidative Stress, Inflammation and DNA Damage Following Pulmonary Exposure to Cerium Oxide Nanoparticles in a Rat Model of Vascular Injury
Abstract
1. Introduction
2. Material and Methods
2.1. Particles
2.2. Animals and i.t. Instillation
2.3. Treatments
2.4. Sample Collection and Biochemical Analysis
2.5. Histology and Immunohistochemistry
2.6. Assessment of DNA Damage by COMET Assay
2.7. Statistics
3. Results
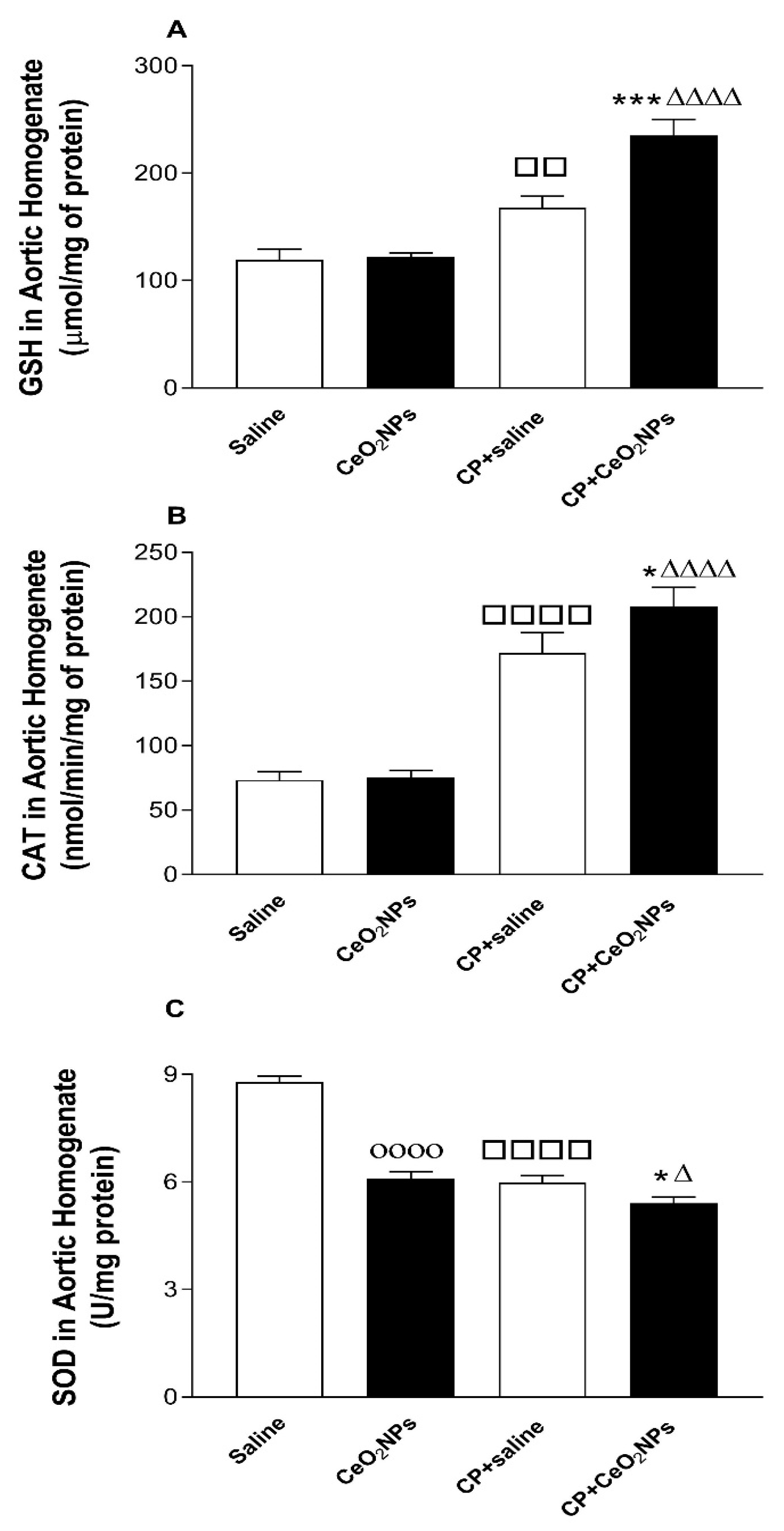
3.1. GSH Concentration and CAT and SOD Activities in Aortic Homogenates
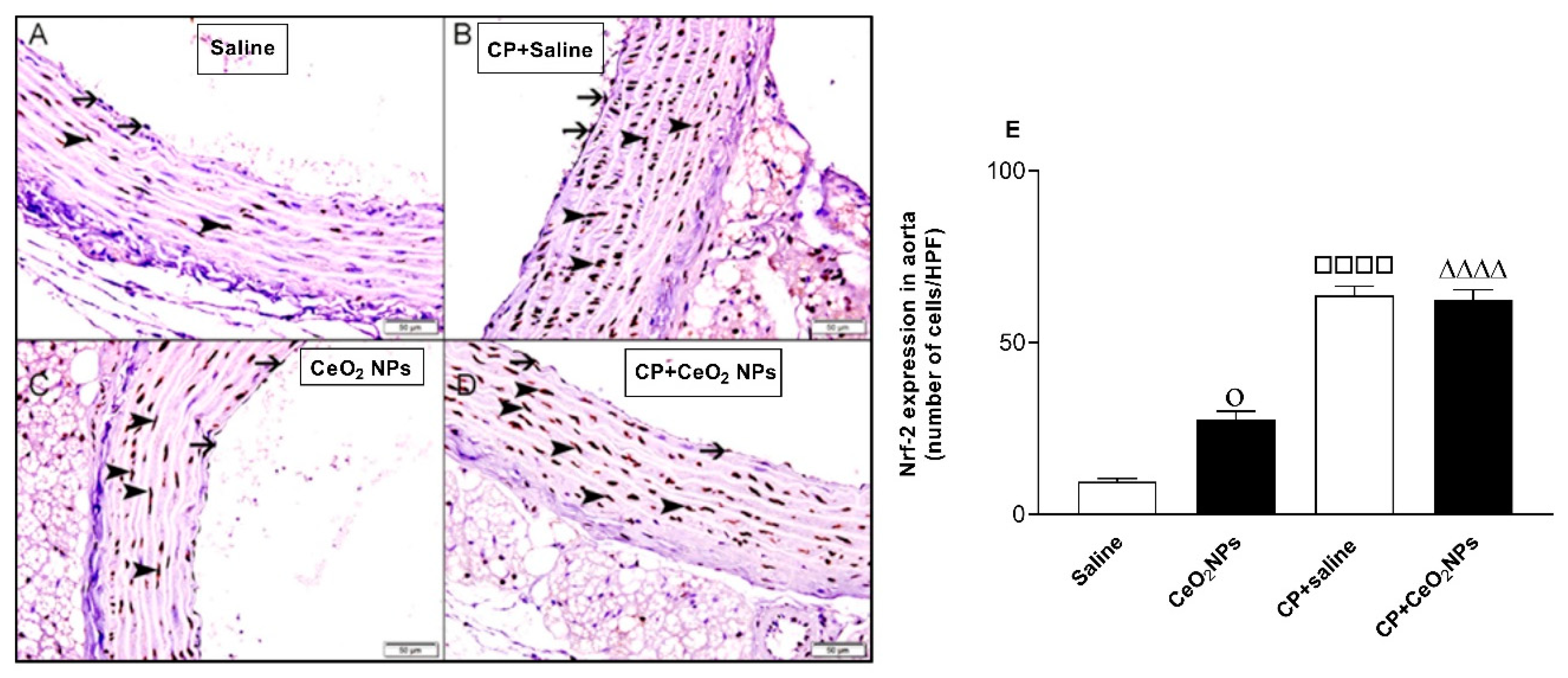
3.2. Histopathological Analysis of the Aorta and Expression of Nrf2
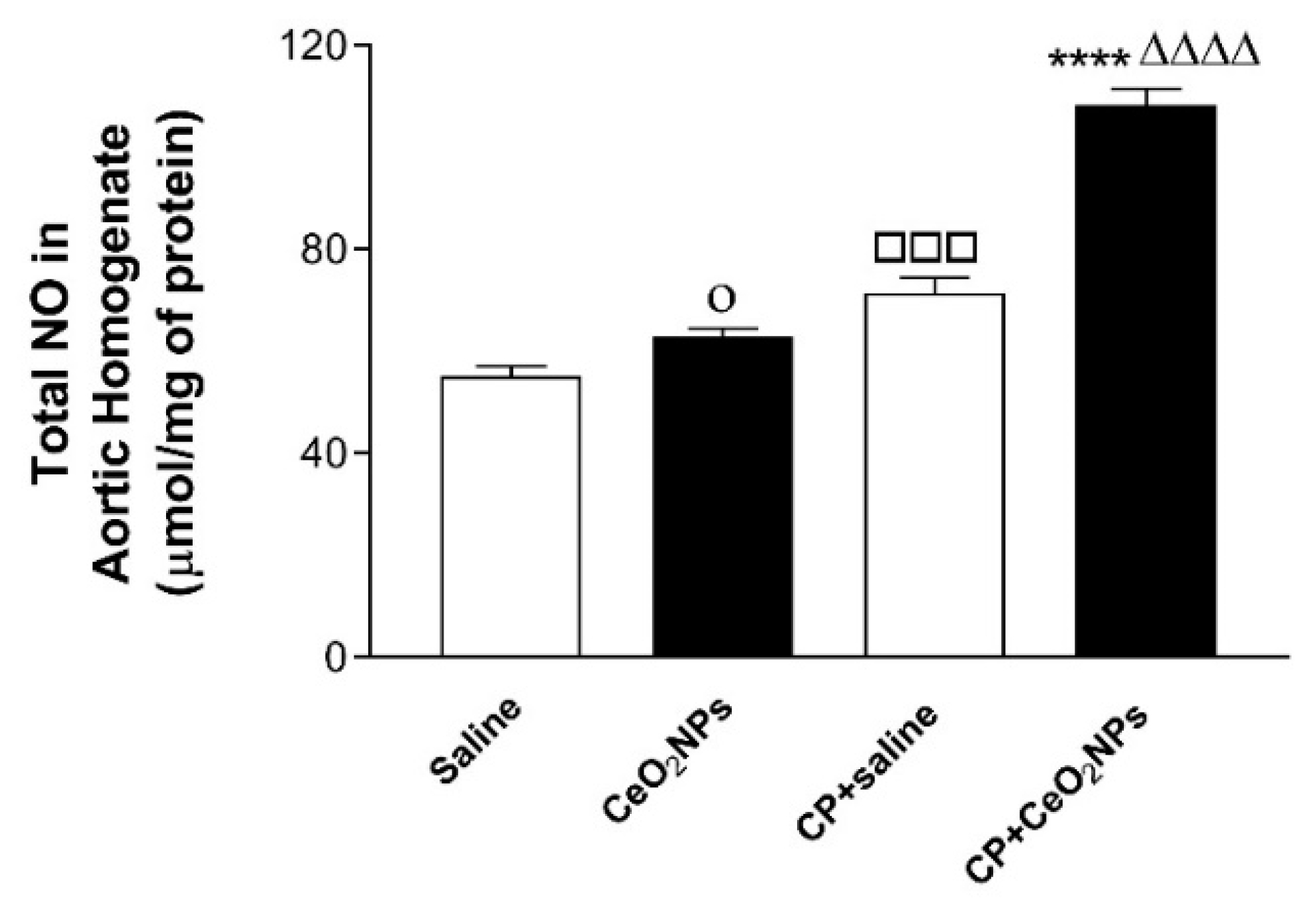
3.3. Total NO Concentration in Aortic Homogenates
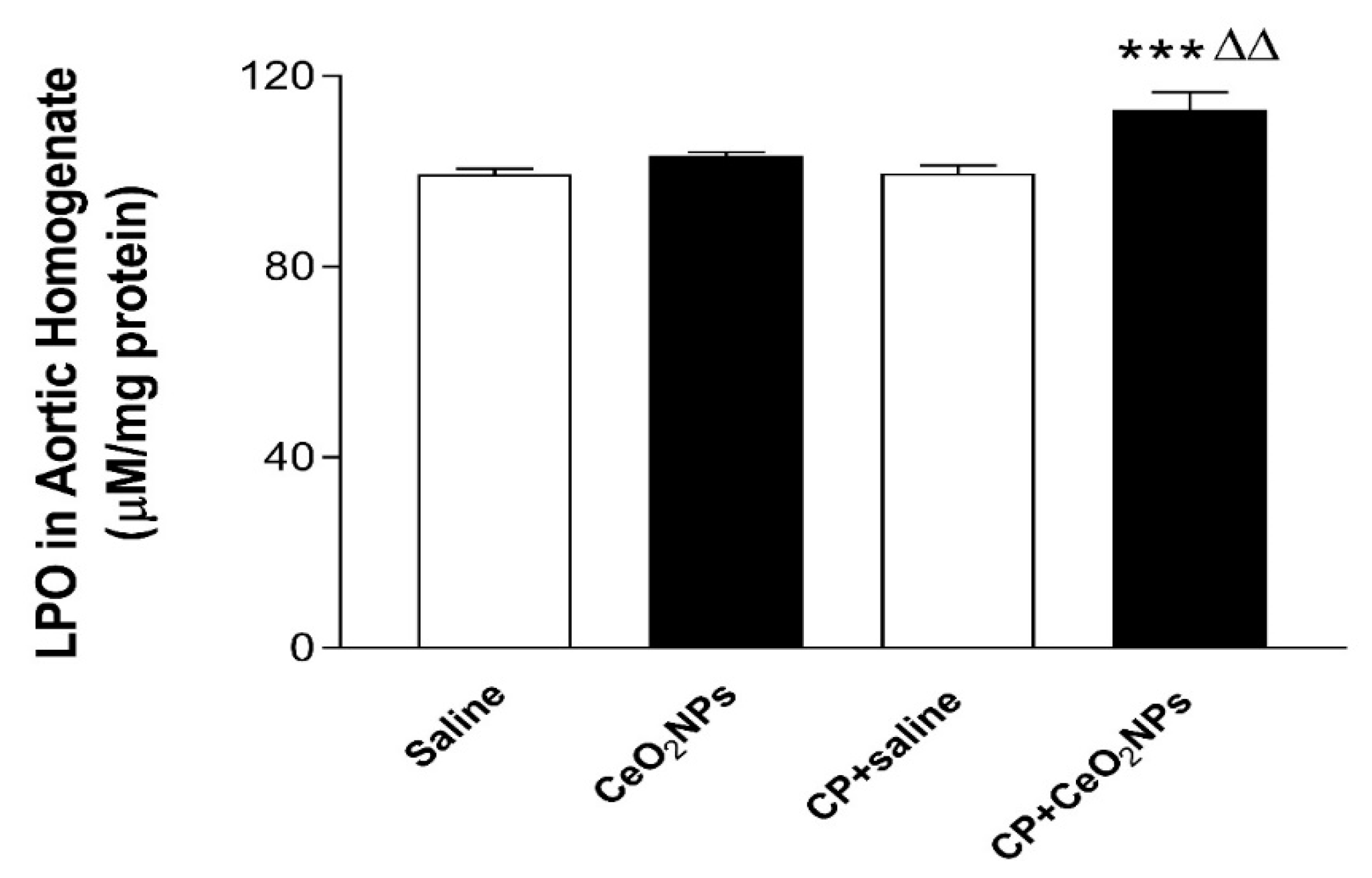
3.4. LPO Concentrations in Aortic Homogenates
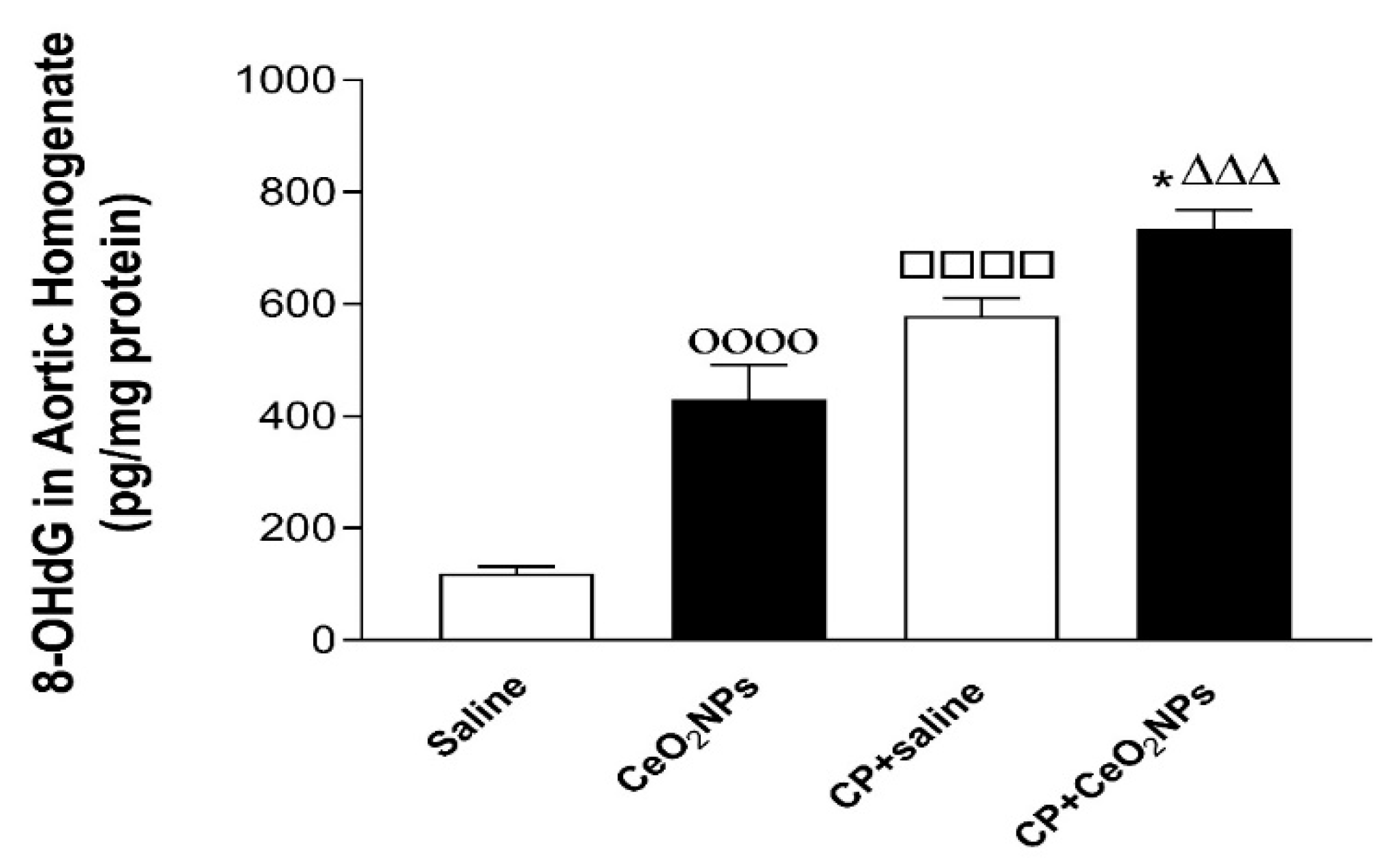
3.5. 8-OH-dG Concentrations in Aortic Homogenates
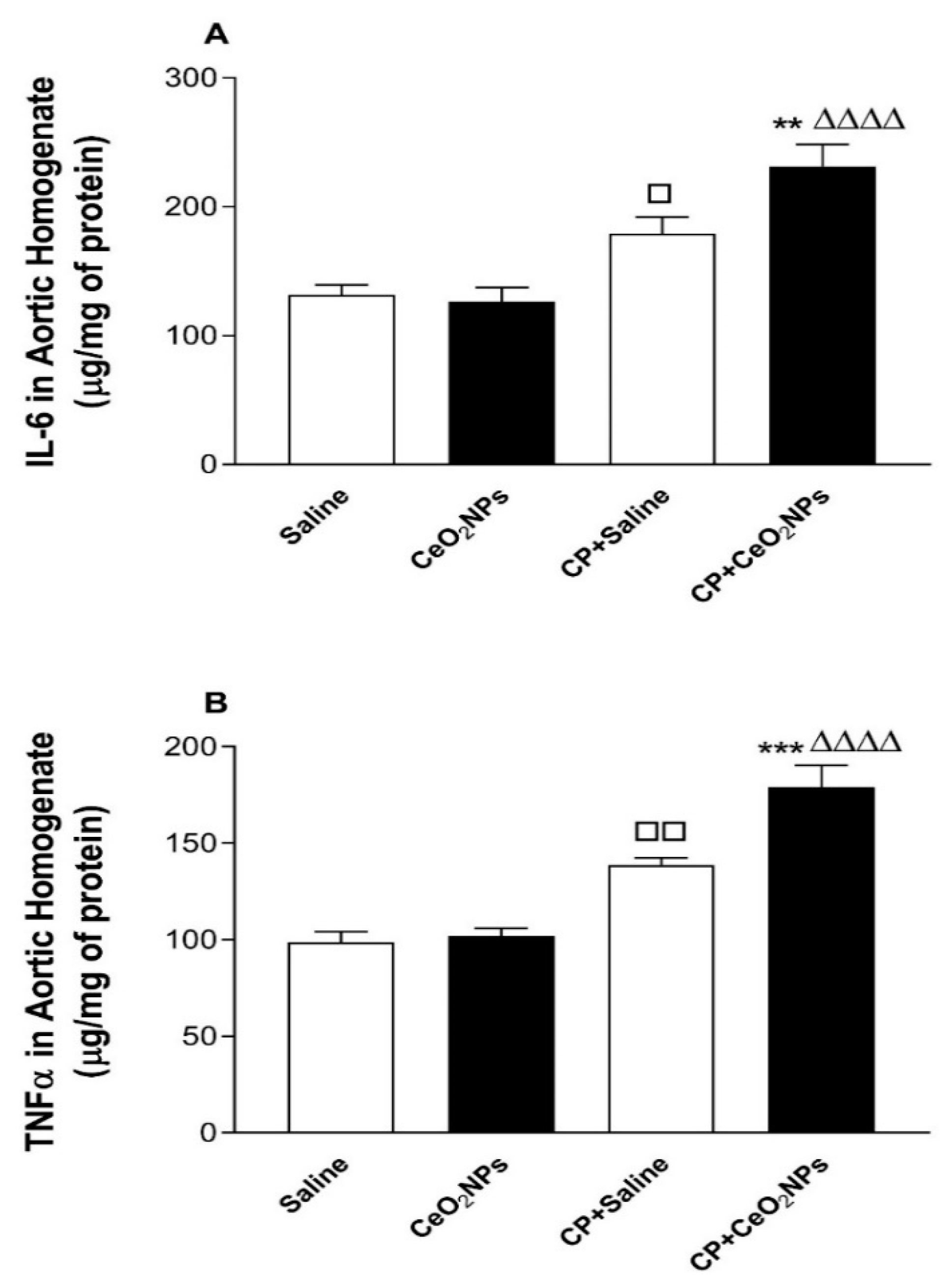
3.6. IL-6 and TNFα Concentrations in Aortic Homogenates
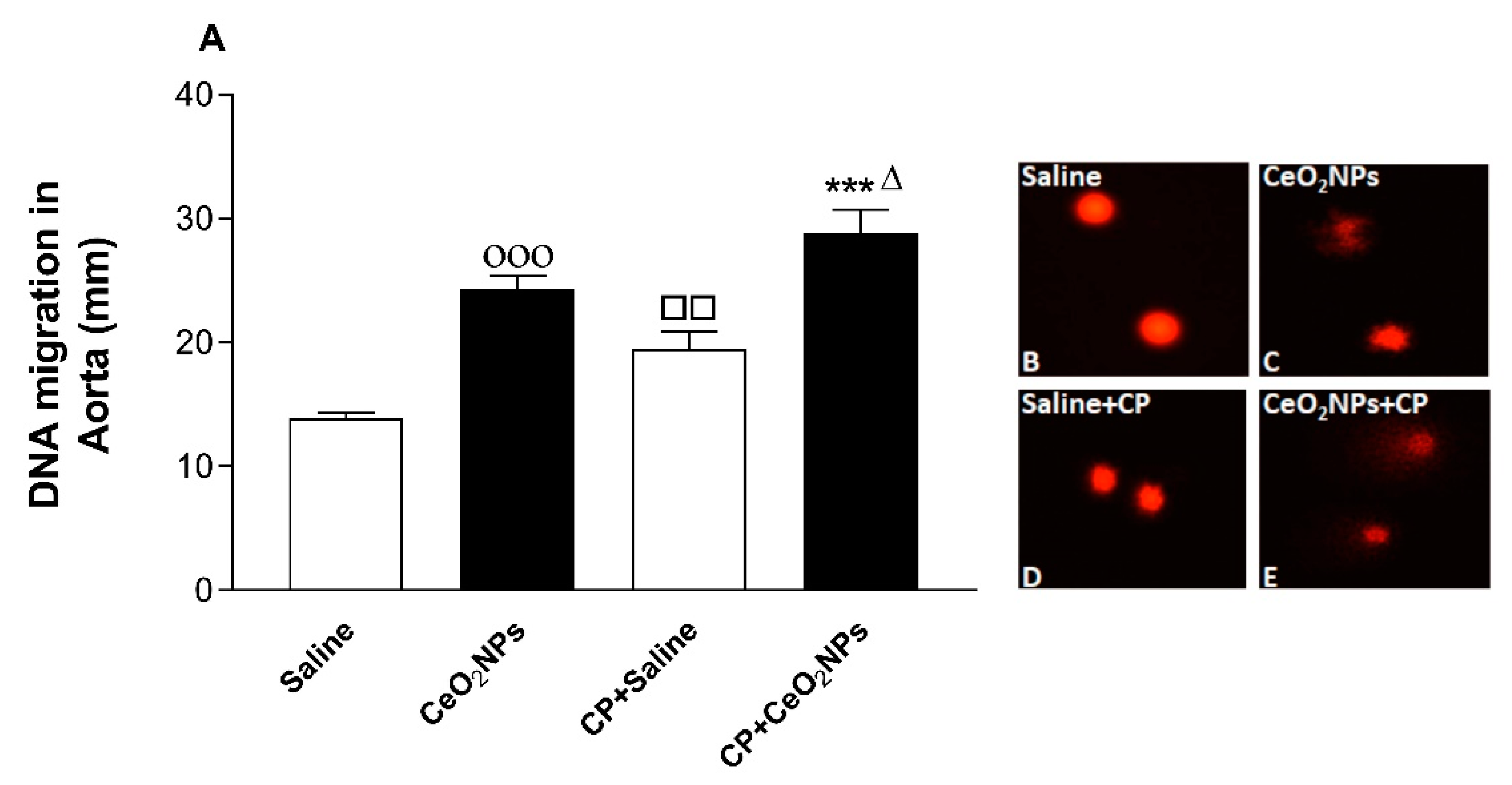
3.7. Aortic DNA Damage
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Radomska, A.; Leszczyszyn, J.; Radomski, M.W. The Nanopharmacology and Nanotoxicology of Nanomaterials: New Opportunities and Challenges. Adv. Clin. Exp. Med. 2016, 25, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Hoet, P.H.M.; Nemmar, A.; Nemery, B. Health impact of nanomaterials? Nat. Biotechnol. 2004, 22, 19. [Google Scholar] [CrossRef] [PubMed]
- Nemmar, A.; Holme, J.A.; Rosas, I.; Schwarze, P.E.; Alfaro-Moreno, E. Recent advances in particulate matter and nanoparticle toxicology: A review of the in vivo and in vitro studies. BioMed Res. Int. 2013, 2013, 279371. [Google Scholar] [CrossRef] [PubMed]
- Oberdorster, G. Safety assessment for nanotechnology and nanomedicine: Concepts of nanotoxicology. J. Intern. Med. 2010, 267, 89–105. [Google Scholar] [CrossRef] [PubMed]
- Cassee, F.R.; van Balen, E.C.; Singh, C.; Green, D.; Muijser, H.; Weinstein, J.; Dreher, K. Exposure, health and ecological effects review of engineered nanoscale cerium and cerium oxide associated with its use as a fuel additive. Crit. Rev. Toxicol. 2011, 41, 213–229. [Google Scholar] [CrossRef] [PubMed]
- Selvan, A.M.; Anand, V.; Udayakumar, M.R.B. Effects of cerium oxide nanoparticle addition in diesel and diesel-biodiesel-ethanol blends on the performance and emission characteristics of a CI engine. J. Eng. Appl. Sci. 2009, 4, 1–6. [Google Scholar]
- Park, B.; Donaldson, K.; Duffin, R.; Tran, L.; Kelly, F.; Mudway, I.; Morin, J.P.; Guest, R.; Jenkinson, P.; Samaras, Z.; et al. Hazard and risk assessment of a nanoparticulate cerium oxide-based diesel fuel additive—A case study. Inhal. Toxicol. 2008, 20, 547–566. [Google Scholar] [CrossRef] [PubMed]
- Nemmar, A.; Yuvaraju, P.; Beegam, S.; Fahim, M.A.; Ali, B.H. Cerium Oxide Nanoparticles in Lung Acutely Induce Oxidative Stress, Inflammation, and DNA Damage in Various Organs of Mice. Oxid. Med. Cell. Longev. 2017, 2017, 9639035. [Google Scholar] [CrossRef]
- Snow, S.J.; Mcgee, J.; Miller, D.B.; Bass, V.; Schladweiler, M.C.; Thomas, R.F.; Krantz, T.; King, C.; Ledbetter, A.D.; Richards, J.; et al. Inhaled diesel emissions generated with cerium oxide nanoparticle fuel additive induce adverse pulmonary and systemic effects. Toxicol. Sci. 2014, 142, 403–417. [Google Scholar] [CrossRef]
- Wilson, S.J.; Miller, M.R.; Newby, D.E. Effects of Diesel Exhaust on Cardiovascular Function and Oxidative Stress. Antioxid. Redox Signal. 2018, 28, 819–836. [Google Scholar] [CrossRef]
- Newby, D.E.; Mannucci, P.M.; Tell, G.S.; Baccarelli, A.A.; Brook, R.D.; Donaldson, K.; Forastiere, F.; Franchini, M.; Franco, O.H.; Graham, I.; et al. Expert position paper on air pollution and cardiovascular disease. Eur. Heart J. 2015, 36, 83–93b. [Google Scholar] [CrossRef] [PubMed]
- Mathe, C.; Bohacs, A.; Duffek, L.; Lukacsovits, J.; Komlosi, Z.I.; Szondy, K.; Horvath, I.; Muller, V.; Losonczy, G. Cisplatin nephrotoxicity aggravated by cardiovascular disease and diabetes in lung cancer patients. Eur. Respir. J. 2011, 37, 888–894. [Google Scholar] [CrossRef] [PubMed]
- Ali, B.H.; Al-Salam, S.; Al Husseini, I.S.; Al-Lawati, I.; Waly, M.; Yasin, J.; Fahim, M.; Nemmar, A. Abrogation of cisplatin-induced nephrotoxicity by emodin in rats. Fundam. Clin. Pharmacol. 2013, 27, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Saleh, R.M.; Awadin, W.F.; El-Shafei, R.A.; Elseady, Y.Y.; Wehaish, F.E.; Elshal, M.F. Cardioprotective role of tadalafil against cisplatin-induced cardiovascular damage in rats. Eur. J. Pharmacol. 2015, 765, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.Y.; Zhao, H.; Mercer, R.R.; Barger, M.; Rao, M.; Meighan, T.; Schwegler-Berry, D.; Castranova, V.; Ma, J.K. Cerium oxide nanoparticle-induced pulmonary inflammation and alveolar macrophage functional change in rats. Nanotoxicology 2011, 5, 312–325. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.Y.; Young, S.H.; Mercer, R.R.; Barger, M.; Schwegler-Berry, D.; Ma, J.K.; Castranova, V. Interactive effects of cerium oxide and diesel exhaust nanoparticles on inducing pulmonary fibrosis. Toxicol. Appl. Pharmacol. 2014, 278, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Nemmar, A.; Al-Salam, S.; Al Ansari, Z.; Alkharas, Z.A.; Al Ahbabi, R.M.; Beegam, S.; Yuvaraju, P.; Yasin, J.; Ali, B.H. Impact of pulmonary exposure to cerium oxide nanoparticles on experimental acute kidney injury. Cell. Physiol. Biochem. 2019, 52, 439–454. [Google Scholar] [PubMed]
- Ali, B.H.; Al Moundhri, M.S.; Tag, E.M.; Nemmar, A.; Tanira, M.O. The ameliorative effect of cysteine prodrug L-2-oxothiazolidine-4-carboxylic acid on cisplatin-induced nephrotoxicity in rats. Fundam. Clin. Pharmacol. 2007, 21, 547–553. [Google Scholar] [CrossRef]
- Ali, B.H.; Al Moundhri, M.; Eldin, M.T.; Nemmar, A.; Al Siyabi, S.; Annamalai, K. Amelioration of cisplatin-induced nephrotoxicity in rats by tetramethylpyrazine, a major constituent of the Chinese herb Ligusticum wallichi. Exp. Biol. Med. (Maywood) 2008, 233, 891–896. [Google Scholar] [CrossRef]
- Jiang, Y.; Shan, S.; Gan, T.; Zhang, X.; Lu, X.; Hu, H.; Wu, Y.; Sheng, J.; Yang, J. Effects of cisplatin on the contractile function of thoracic aorta of Sprague-Dawley rats. BioMed Rep. 2014, 2, 893–897. [Google Scholar] [CrossRef][Green Version]
- Nemmar, A.; Al-Salam, S.; Yuvaraju, P.; Beegam, S.; Ali, B.H. Emodin mitigates diesel exhaust particles-induced increase in airway resistance, inflammation and oxidative stress in mice. Respir. Physiol. Neurobiol. 2015, 215, 51–57. [Google Scholar] [CrossRef]
- Zainalabidin, S.; Budin, S.B.; Ramalingam, A.; Lim, Y.C. Aortic remodelling in chronic nicotine-administered rat. Korean J. Physiol. Pharmacol. 2014, 18, 411–418. [Google Scholar] [CrossRef]
- Tsikas, D. Methods of quantitative analysis of the nitric oxide metabolites nitrite and nitrate in human biological fluids. Free Radic. Res. 2005, 39, 797–815. [Google Scholar] [CrossRef]
- Nemmar, A.; Al-Salam, S.; Beegam, S.; Yuvaraju, P.; Oulhaj, A.; Ali, B.H. Water-Pipe Smoke Exposure-Induced Circulatory Disturbances in Mice, and the Influence of Betaine Supplementation Thereon. Cell. Physiol. Biochem. 2017, 41, 1098–1112. [Google Scholar] [CrossRef]
- Nemmar, A.; Al-Salam, S.; Beegam, S.; Yuvaraju, P.; Ali, B.H. Thrombosis, systemic and cardiac oxidative stress and DNA damage induced by pulmonary exposure to diesel exhaust particles, and the effect of nootkatone thereon. Am. J. Physiol. Heart Circ. Physiol. 2018, 314, H917–H927. [Google Scholar] [CrossRef]
- Nemmar, A.; Al, H.A.; Al, H.N.; Yuvaraju, P.; Beegam, S.; Yasin, J.; Elwasila, M.; Ali, B.H.; Adeghate, E. Early pulmonary events of nose-only water pipe (shisha) smoking exposure in mice. Physiol. Rep. 2015, 3, e12258. [Google Scholar] [CrossRef]
- Al Suleimani, Y.M.; Al Mahruqi, A.S.; Al, Z.M.; Shalaby, A.; Ashique, M.; Nemmar, A.; Ali, B.H. Effect of diesel exhaust particles on renal vascular responses in rats with chronic kidney disease. Environ. Toxicol. 2017, 32, 541–549. [Google Scholar] [CrossRef]
- Nemmar, A.; Yuvaraju, P.; Beegam, S.; Yasin, J.; Kazzam, E.E.; Ali, B.H. Oxidative stress, inflammation, and DNA damage in multiple organs of mice acutely exposed to amorphous silica nanoparticles. Int. J. Nanomed. 2016, 11, 919–928. [Google Scholar] [CrossRef]
- Nemmar, A.; Al-Salam, S.; Yuvaraju, P.; Beegam, S.; Yasin, J.; Ali, B.H. Chronic Exposure to Water-Pipe Smoke Induces Alveolar Enlargement, DNA Damage and Impairment of Lung Function. Cell. Physiol. Biochem. 2016, 38, 982–992. [Google Scholar] [CrossRef]
- Nemmar, A.; Beegam, S.; Yuvaraju, P.; Yasin, J.; Tariq, S.; Attoub, S.; Ali, B.H. Ultrasmall superparamagnetic iron oxide nanoparticles acutely promote thrombosis and cardiac oxidative stress and DNA damage in mice. Part. Fibre Toxicol. 2016, 13, 22. [Google Scholar] [CrossRef]
- Nemmar, A.; Al-Salam, S.; Beegam, S.; Yuvaraju, P.; Ali, B.H. The acute pulmonary and thrombotic effects of cerium oxide nanoparticles after intratracheal instillation in mice. Int. J. Nanomed. 2017, 12, 2913–2922. [Google Scholar] [CrossRef]
- Wingard, C.J.; Walters, D.M.; Cathey, B.L.; Hilderbrand, S.C.; Katwa, P.; Lin, S.; Ke, P.C.; Podila, R.; Rao, A.; Lust, R.M.; et al. Mast cells contribute to altered vascular reactivity and ischemia-reperfusion injury following cerium oxide nanoparticle instillation. Nanotoxicology 2011, 5, 531–545. [Google Scholar] [CrossRef]
- Siti, H.N.; Kamisah, Y.; Kamsiah, J. The role of oxidative stress, antioxidants and vascular inflammation in cardiovascular disease (a review). Vascul. Pharmacol. 2015, 71, 40–56. [Google Scholar] [CrossRef]
- Xue, M.; Qian, Q.; Adaikalakoteswari, A.; Rabbani, N.; Babaei-Jadidi, R.; Thornalley, P.J. Activation of NF-E2-related factor-2 reverses biochemical dysfunction of endothelial cells induced by hyperglycemia linked to vascular disease. Diabetes 2008, 57, 2809–2817. [Google Scholar] [CrossRef]
- Teasdale, J.E.; Newby, A.C.; Timpson, N.J.; Munafo, M.R.; White, S.J. Cigarette smoke but not electronic cigarette aerosol activates a stress response in human coronary artery endothelial cells in culture. Drug Alcohol Depend. 2016, 163, 256–260. [Google Scholar] [CrossRef]
- Manke, A.; Wang, L.; Rojanasakul, Y. Mechanisms of nanoparticle-induced oxidative stress and toxicity. BioMed Res. Int. 2013, 2013, 942916. [Google Scholar] [CrossRef]
- Ali, D.; Alarifi, S.; Alkahtani, S.; AlKahtane, A.A.; Almalik, A. Cerium Oxide Nanoparticles Induce Oxidative Stress and Genotoxicity in Human Skin Melanoma Cells. Cell Biochem. Biophys. 2015, 71, 1643–1651. [Google Scholar] [CrossRef]
- Benameur, L.; Auffan, M.; Cassien, M.; Liu, W.; Culcasi, M.; Rahmouni, H.; Stocker, P.; Tassistro, V.; Bottero, J.Y.; Rose, J.; et al. DNA damage and oxidative stress induced by CeO2 nanoparticles in human dermal fibroblasts: Evidence of a clastogenic effect as a mechanism of genotoxicity. Nanotoxicology 2015, 9, 696–705. [Google Scholar] [CrossRef]
- Preaubert, L.; Courbiere, B.; Achard, V.; Tassistro, V.; Greco, F.; Orsiere, T.; Bottero, J.Y.; Rose, J.; Auffan, M.; Perrin, J. Cerium dioxide nanoparticles affect in vitro fertilization in mice. Nanotoxicology 2016, 10, 111–117. [Google Scholar]
- Courbiere, B.; Auffan, M.; Rollais, R.; Tassistro, V.; Bonnefoy, A.; Botta, A.; Rose, J.; Orsiere, T.; Perrin, J. Ultrastructural interactions and genotoxicity assay of cerium dioxide nanoparticles on mouse oocytes. Int. J. Mol. Sci. 2013, 14, 21613–21628. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nemmar, A.; Al-Salam, S.; Beegam, S.; Yuvaraju, P.; Ali, B.H. Aortic Oxidative Stress, Inflammation and DNA Damage Following Pulmonary Exposure to Cerium Oxide Nanoparticles in a Rat Model of Vascular Injury. Biomolecules 2019, 9, 376. https://doi.org/10.3390/biom9080376
Nemmar A, Al-Salam S, Beegam S, Yuvaraju P, Ali BH. Aortic Oxidative Stress, Inflammation and DNA Damage Following Pulmonary Exposure to Cerium Oxide Nanoparticles in a Rat Model of Vascular Injury. Biomolecules. 2019; 9(8):376. https://doi.org/10.3390/biom9080376
Chicago/Turabian StyleNemmar, Abderrahim, Suhail Al-Salam, Sumaya Beegam, Priya Yuvaraju, and Badreldin H. Ali. 2019. "Aortic Oxidative Stress, Inflammation and DNA Damage Following Pulmonary Exposure to Cerium Oxide Nanoparticles in a Rat Model of Vascular Injury" Biomolecules 9, no. 8: 376. https://doi.org/10.3390/biom9080376
APA StyleNemmar, A., Al-Salam, S., Beegam, S., Yuvaraju, P., & Ali, B. H. (2019). Aortic Oxidative Stress, Inflammation and DNA Damage Following Pulmonary Exposure to Cerium Oxide Nanoparticles in a Rat Model of Vascular Injury. Biomolecules, 9(8), 376. https://doi.org/10.3390/biom9080376




