Antidiabetic Properties of Naringenin: A Citrus Fruit Polyphenol
Abstract
:1. Introduction
1.1. Glucose Homeostasis: Role of Insulin
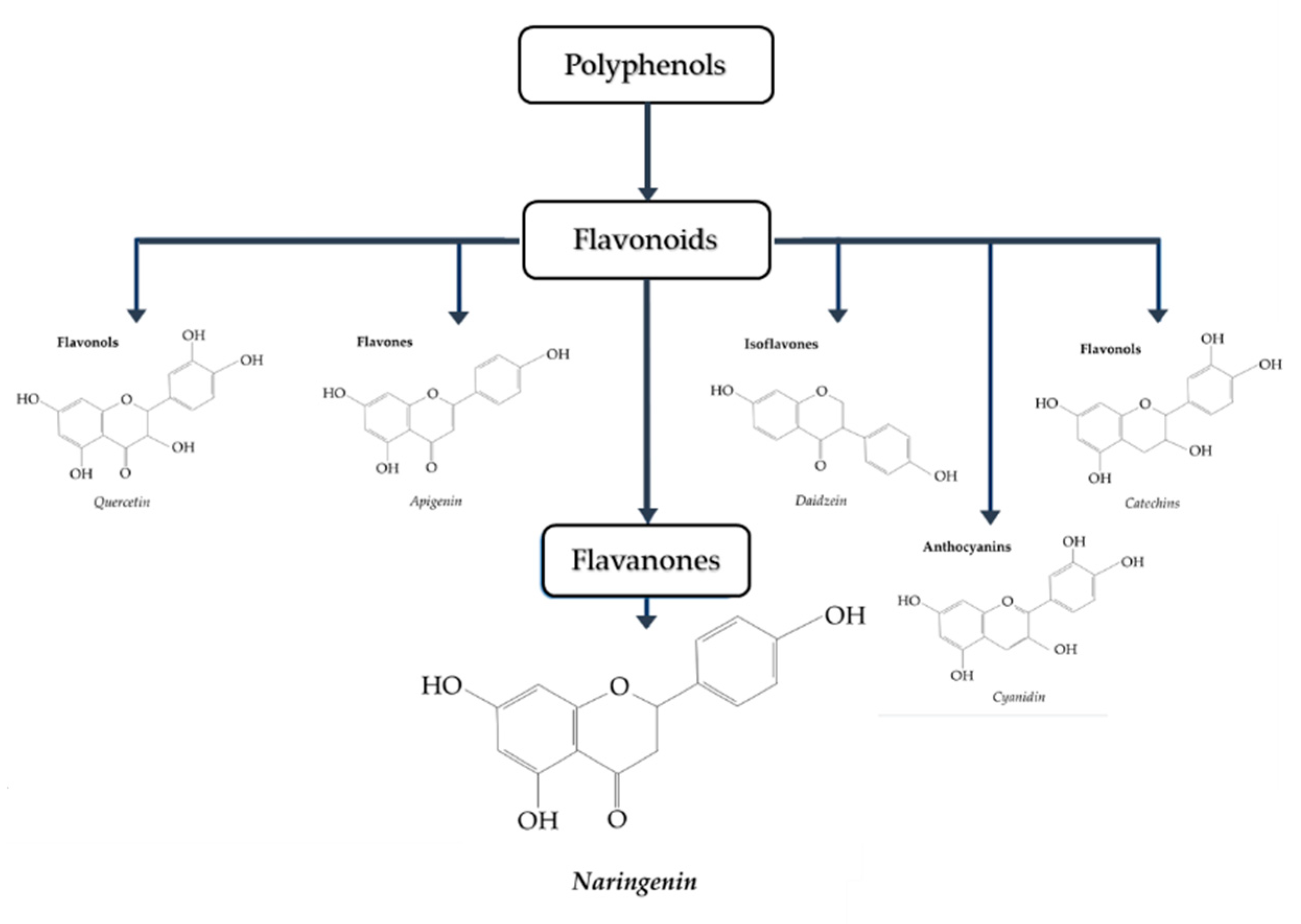
1.2. Naringenin
2. Anti-Diabetic Effects of Naringenin
2.1. Effects of Naringenin: In Vitro Skeletal Muscle Cell Studies
2.2. Effects of Naringenin: In Vitro Adipocyte Studies
2.3. Effects of Naringenin: In Vitro Hepatocyte Studies
2.4. Effects of Naringenin: In Vitro Beta Cell Studies
2.5. Evidence of Anti-Diabetic Effects of Naringenin: In Vivo Animal Studies
2.5.1. Streptozotocin (STZ)-Induced Diabetes Model
2.5.2. Alloxan-Induced Diabetes Model
2.5.3. Genetic Diabetes Model
2.5.4. Diet-Induced Diabetes Model
2.6. Biological Effects of Naringenin: Human Studies
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sweet, I.R.; Cook, D.L.; DeJulio, E.; Wallen, A.R.; Khalil, G.; Callis, J.; Reems, J. Regulation of ATP/ADP in Pancreatic Islets. Diabetes 2004, 53, 401–409. [Google Scholar] [CrossRef]
- Tripathy, D.; Chavez, A.O. Defects in insulin secretion and action in the pathogenesis of type 2 diabetes mellitus. Curr. Diab. Rep. 2010, 10, 184–191. [Google Scholar] [CrossRef]
- Petersen, M.C.; Shulman, G.I. Mechanisms of Insulin Action and Insulin Resistance. Physiol. Rev. 2018, 98, 2133–2223. [Google Scholar] [CrossRef]
- Saltiel, A.R. New perspectives into the molecular pathogenesis and treatment of type 2 diabetes. Cell 2001, 104, 517–529. [Google Scholar] [CrossRef]
- Alam, M.A.; Subhan, N.; Rahman, M.M.; Uddin, S.J.; Reza, H.M.; Sarker, S.D. Effect of citrus flavonoids, naringin and naringenin, on metabolic syndrome and their mechanisms of action. Adv. Nutr. 2014, 5, 404–417. [Google Scholar] [CrossRef] [PubMed]
- DeFronzo, R.A. Dysfunctional fat cells, lipotoxicity and type 2 diabetes. Int. J. Clin. Pract. Suppl. 2004, 58, 9–21. [Google Scholar] [CrossRef]
- Lee, C.-H.; Olson, P.; Hevener, A.; Mehl, I.; Chong, L.-W.; Olefsky, J.M.; Gonzalez, F.J.; Ham, J.; Kang, H.; Peters, J.M.; et al. PPAR regulates glucose metabolism and insulin sensitivity. Proc. Natl. Acad. Sci. USA 2006, 103, 3444–3449. [Google Scholar] [CrossRef]
- Defronzo, R.A. Banting Lecture. From the triumvirate to the ominous octet: A new paradigm for the treatment of type 2 diabetes mellitus. Diabetes 2009, 58, 773–795. [Google Scholar] [CrossRef]
- Frigolet, M.E.; Torres, N.; Tovar, A.R. The renin-angiotensin system in adipose tissue and its metabolic consequences during obesity. J. Nutr. Biochem. 2013, 24, 2003–2015. [Google Scholar] [CrossRef]
- Hotamisligil, G.S.; Spiegelman, B.M. Tumor necrosis factor alpha: A key component of the obesity-diabetes link. Diabetes 1994, 43, 1271–1278. [Google Scholar] [CrossRef]
- Cho, N.H.; Shaw, J.E.; Karuranga, S.; Huang, Y.; da Rocha Fernandes, J.D.; Ohlrogge, A.W.; Malanda, B. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 2018, 138, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, S.S.; Zhang, P.; Hora, I.; Geiss, L.S.; Luman, E.T.; Gregg, E.W. Factors Contributing to Increases in Diabetes-Related Preventable Hospitalization Costs Among U.S. Adults During 2001–2014. Diabetes Care 2019, 42, 77–84. [Google Scholar] [CrossRef]
- Baur, J.A.; Sinclair, D.A. Therapeutic potential of resveratrol: The in vivo evidence. Nat. Rev. Drug. Discov. 2006, 5, 493–506. [Google Scholar] [CrossRef] [PubMed]
- Park, E.-J.; Pezzuto, J.M. The pharmacology of resveratrol in animals and humans. Biochim. Biophys. Acta 2015, 1852, 1071–1113. [Google Scholar] [CrossRef] [PubMed]
- Anja, B.; Laura, R. The cost of diabetes in Canada over 10 years: Applying attributable health care costs to a diabetes incidence prediction model. Health Promot. Chronic Dis. Prev. Can. 2017, 37, 49–53. [Google Scholar]
- Vieira, A.R.; Abar, L.; Vingeliene, S.; Chan, D.S.M.; Aune, D.; Navarro-Rosenblatt, D.; Stevens, C.; Greenwood, D.; Norat, T. Fruits, vegetables and lung cancer risk: A systematic review and meta-analysis. Ann. Oncol. 2016, 27, 81–96. [Google Scholar] [CrossRef]
- Kuzma, J.N.; Schmidt, K.A.; Kratz, M. Prevention of metabolic diseases: Fruits (including fruit sugars) vs. vegetables. Curr. Opin. Clin. Nutr. Metab. Care 2017, 20, 286–293. [Google Scholar] [CrossRef]
- Stefan, N.; Häring, H.-U.; Schulze, M.B. Metabolically healthy obesity: The low-hanging fruit in obesity treatment? Lancet Diabetes Endocrinol. 2018, 6, 249–258. [Google Scholar] [CrossRef]
- Serino, A.; Salazar, G. Protective Role of Polyphenols against Vascular Inflammation, Aging and Cardiovascular Disease. Nutrients 2018, 11, 53. [Google Scholar] [CrossRef]
- Moore, J.; Yousef, M.; Tsiani, E. Anticancer Effects of Rosemary (Rosmarinus officinalis L.) Extract and Rosemary Extract Polyphenols. Nutrients 2016, 8, 731. [Google Scholar] [CrossRef]
- Yousef, M.; Vlachogiannis, I.A.; Tsiani, E. Effects of Resveratrol against Lung Cancer: In Vitro and In Vivo Studies. Nutrients 2017, 9, 1231. [Google Scholar] [CrossRef]
- Naimi, M.; Vlavcheski, F.; Shamshoum, H.; Tsiani, E. Rosemary Extract as a Potential Anti-Hyperglycemic Agent: Current Evidence and Future Perspectives. Nutrients 2017, 9, 968. [Google Scholar] [CrossRef] [PubMed]
- Dreosti, I.E. Antioxidant polyphenols in tea, cocoa, and wine. Nutrition 2000, 16, 692–694. [Google Scholar] [CrossRef]
- Lagouge, M.; Argmann, C.; Gerhart-Hines, Z.; Meziane, H.; Lerin, C.; Daussin, F.; Messadeq, N.; Milne, J.; Lambert, P.; Elliott, P.; et al. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1alpha. Cell 2006, 127, 1109–1122. [Google Scholar] [CrossRef]
- Amor, S.; Châlons, P.; Aires, V.; Delmas, D. Polyphenol Extracts from Red Wine and Grapevine: Potential Effects on Cancers. Diseases 2018, 6, 106. [Google Scholar] [CrossRef]
- Yahfoufi, N.; Alsadi, N.; Jambi, M.; Matar, C. The Immunomodulatory and Anti-Inflammatory Role of Polyphenols. Nutrients 2018, 10, 1618. [Google Scholar] [CrossRef] [PubMed]
- D’Archivio, M.; Filesi, C.; Di Benedetto, R.; Gargiulo, R.; Giovannini, C.; Masella, R. Polyphenols, dietary sources and bioavailability. Ann. Ist. Super. Sanita 2007, 43, 348–361. [Google Scholar]
- Kumar, S.; Pandey, A.K. Chemistry and Biological Activities of Flavonoids: An Overview. Sci. World J. 2013, 2013, 162750. [Google Scholar] [CrossRef]
- Erlund, I. Review of the flavonoids quercetin, hesperetin, and naringenin. Dietary sources, bioactivities, bioavailability, and epidemiology. Nutr. Res. 2004, 24, 851–874. [Google Scholar] [CrossRef]
- Gattuso, G.; Barreca, D.; Gargiulli, C.; Leuzzi, U.; Caristi, C. Flavonoid Composition of Citrus Juices. Molecules 2007, 12, 1641–1673. [Google Scholar] [CrossRef]
- Aherne, S.A.; O’Brien, N.M. Dietary flavonols: Chemistry, food content, and metabolism. Nutrition 2002, 18, 75–81. [Google Scholar] [CrossRef]
- Zaidun, N.H.; Thent, Z.C.; Latiff, A.A. Combating oxidative stress disorders with citrus flavonoid: Naringenin. Life Sci. 2018, 208, 111–122. [Google Scholar] [CrossRef]
- Sharma, M.; Akhtar, N.; Sambhav, K.; Shete, G.; Bansal, A.K.; Sharma, S.S. Emerging potential of citrus flavanones as an antioxidant in diabetes and its complications. Curr. Top. Med. Chem. 2015, 15, 187–195. [Google Scholar] [CrossRef]
- Coelho, R.C.L.A.; Hermsdorff, H.H.M.; Bressan, J. Anti-inflammatory properties of orange juice: Possible favorable molecular and metabolic effects. Plant. Foods Hum. Nutr. 2013, 68, 1–10. [Google Scholar] [CrossRef]
- Rani, N.; Bharti, S.; Krishnamurthy, B.; Bhatia, J.; Sharma, C.; Kamal, M.A.; Ojha, S.; Arya, D.S. Pharmacological Properties and Therapeutic Potential of Naringenin: A Citrus Flavonoid of Pharmaceutical Promise. Curr. Pharm. Des. 2016, 22, 4341–4359. [Google Scholar] [CrossRef]
- Zeng, W.; Jin, L.; Zhang, F.; Zhang, C.; Liang, W. Naringenin as a potential immunomodulator in therapeutics. Pharmacol. Res. 2018, 135, 122–126. [Google Scholar] [CrossRef]
- Hernández-Aquino, E.; Muriel, P. Beneficial effects of naringenin in liver diseases: Molecular mechanisms. World J. Gastroenterol. 2018, 24, 1679–1707. [Google Scholar] [CrossRef]
- Mulvihill, E.E.; Assini, J.M.; Sutherland, B.G.; DiMattia, A.S.; Khami, M.; Koppes, J.B.; Sawyez, C.G.; Whitman, S.C.; Huff, M.W. Naringenin decreases progression of atherosclerosis by improving dyslipidemia in high-fat-fed low-density lipoprotein receptor-null mice. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 742–748. [Google Scholar] [CrossRef]
- Orhan, I.E.; Nabavi, S.F.; Daglia, M.; Tenore, G.C.; Mansouri, K.; Nabavi, S.M. Naringenin and atherosclerosis: A review of literature. Curr. Pharm. Biotechnol. 2015, 16, 245–251. [Google Scholar] [CrossRef]
- Mulvihill, E.E.; Burke, A.C.; Huff, M.W. Citrus Flavonoids as Regulators of Lipoprotein Metabolism and Atherosclerosis. Annu. Rev. Nutr. 2016, 36, 275–299. [Google Scholar] [CrossRef]
- Testai, L.; Calderone, V. Nutraceutical Value of Citrus Flavanones and Their Implications in Cardiovascular Disease. Nutrients 2017, 9, 502. [Google Scholar] [CrossRef]
- Assini, J.M.; Mulvihill, E.E.; Huff, M.W. Citrus flavonoids and lipid metabolism. Curr. Opin. Lipidol. 2013, 24, 34–40. [Google Scholar] [CrossRef]
- Kannappan, S.; Anuradha, C.V. Naringenin enhances insulin-stimulated tyrosine phosphorylation and improves the cellular actions of insulin in a dietary model of metabolic syndrome. Eur. J. Nutr. 2010, 49, 101–109. [Google Scholar] [CrossRef]
- Kanaze, F.I.; Bounartzi, M.I.; Georgarakis, M.; Niopas, I. Pharmacokinetics of the citrus flavanone aglycones hesperetin and naringenin after single oral administration in human subjects. Eur. J. Clin. Nutr. 2007, 61, 472–477. [Google Scholar] [CrossRef]
- El Mohsen, M.A.; Marks, J.; Kuhnle, G.; Rice-Evans, C.; Moore, K.; Gibson, G.; Debnam, E.; Srai, S.K. The differential tissue distribution of the citrus flavanone naringenin following gastric instillation. Free Radic. Res. 2004, 38, 1329–1340. [Google Scholar] [CrossRef]
- Zygmunt, K.; Faubert, B.; MacNeil, J.; Tsiani, E. Naringenin, a citrus flavonoid, increases muscle cell glucose uptake via AMPK. Biochem. Biophys. Res. Commun. 2010, 398, 178–183. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Christensen, K.B.; Olsen, L.C.B.; Christensen, L.P.; Grevsen, K.; Færgeman, N.J.; Kristiansen, K.; Young, J.F.; Oksbjerg, N. Bioactive components from flowers of Sambucus nigra L. increase glucose uptake in primary porcine myotube cultures and reduce fat accumulation in Caenorhabditis elegans. J. Agric. Food Chem. 2013, 61, 11033–11040. [Google Scholar] [CrossRef]
- Mutlur Krishnamoorthy, R.; Carani Venkatraman, A. Polyphenols activate energy sensing network in insulin resistant models. Chem. Biol. Interact. 2017, 275, 95–107. [Google Scholar] [CrossRef]
- Di Meo, S.; Iossa, S.; Venditti, P. Skeletal muscle insulin resistance: Role of mitochondria and other ROS sources. J. Endocrinol. 2017, 233, R15–R42. [Google Scholar] [CrossRef]
- Haber, C.; Lam, T.; Yu, Z.; Gupta, N.; Goh, T.; Bogdanovic, E.; Giacca, A.; Fantus, I. N-acetylcysteine and taurine prevent hyperglycemia-induced insulin resistance in vivo: Possible role of oxidative stress. Am. J. Physiol. Endocrinol. Metab. 2003, 285, 744–753. [Google Scholar] [CrossRef]
- Harmon, A.W.; Harp, J.B. Differential effects of flavonoids on 3T3-L1 adipogenesis and lipolysis. Am. J. Physiol. Cell Physiol. 2001, 280, C807–C813. [Google Scholar] [CrossRef]
- Yoshida, H.; Takamura, N.; Shuto, T.; Ogata, K.; Tokunaga, J.; Kawai, K.; Kai, H. The citrus flavonoids hesperetin and naringenin block the lipolytic actions of TNF-alpha in mouse adipocytes. Biochem. Biophys. Res. Commun. 2010, 394, 728–732. [Google Scholar] [CrossRef]
- Claussnitzer, M.; Skurk, T.; Hauner, H.; Daniel, H.; Rist, M.J. Effect of flavonoids on basal and insulin-stimulated 2-deoxyglucose uptake in adipocytes. Mol. Nutr. Food Res. 2011, 55 (Suppl. 1), S26–S34. [Google Scholar] [CrossRef]
- Yoshida, H.; Watanabe, W.; Oomagari, H.; Tsuruta, E.; Shida, M.; Kurokawa, M. Citrus flavonoid naringenin inhibits TLR2 expression in adipocytes. J. Nutr. Biochem. 2013, 24, 1276–1284. [Google Scholar] [CrossRef]
- Richard, A.J.; Amini-Vaughan, Z.; Ribnicky, D.M.; Stephens, J.M. Naringenin inhibits adipogenesis and reduces insulin sensitivity and adiponectin expression in adipocytes. Evid. Based Complement. Altern. Med. 2013, 2013, 549750. [Google Scholar] [CrossRef]
- Rebello, C.J.; Greenway, F.L.; Lau, F.H.; Lin, Y.; Stephens, J.M.; Johnson, W.D.; Coulter, A.A. Naringenin Promotes Thermogenic Gene Expression in Human White Adipose Tissue. Obesity 2019, 27, 103–111. [Google Scholar] [CrossRef]
- Kaisanlahti, A.; Glumoff, T. Browning of white fat: Agents and implications for beige adipose tissue to type 2 diabetes. J. Physiol. Biochem. 2018, 75, 1–10. [Google Scholar] [CrossRef]
- Wilcox, L.J.; Borradaile, N.M.; de Dreu, L.E.; Huff, M.W. Secretion of hepatocyte apoB is inhibited by the flavonoids, naringenin and hesperetin, via reduced activity and expression of ACAT2 and MTP. J. Lipid Res. 2001, 42, 725–734. [Google Scholar]
- Borradaile, N.M.; de Dreu, L.E.; Huff, M.W. Inhibition of net HepG2 cell apolipoprotein B secretion by the citrus flavonoid naringenin involves activation of phosphatidylinositol 3-kinase, independent of insulin receptor substrate-1 phosphorylation. Diabetes 2003, 52, 2554–2561. [Google Scholar] [CrossRef]
- Allister, E.M.; Mulvihill, E.E.; Barrett, P.H.R.; Edwards, J.Y.; Carter, L.P.; Huff, M.W. Inhibition of apoB secretion from HepG2 cells by insulin is amplified by naringenin, independent of the insulin receptor. J. Lipid Res. 2008, 49, 2218–2229. [Google Scholar] [CrossRef]
- Purushotham, A.; Tian, M.; Belury, M.A. The citrus fruit flavonoid naringenin suppresses hepatic glucose production from Fao hepatoma cells. Mol. Nutr. Food Res. 2009, 53, 300–307. [Google Scholar] [CrossRef]
- Goldwasser, J.; Cohen, P.Y.; Yang, E.; Balaguer, P.; Yarmush, M.L.; Nahmias, Y. Transcriptional regulation of human and rat hepatic lipid metabolism by the grapefruit flavonoid naringenin: Role of PPARalpha, PPARgamma and LXRalpha. PLoS ONE 2010, 5, e12399. [Google Scholar] [CrossRef]
- Constantin, R.P.; Constantin, R.P.; Bracht, A.; Yamamoto, N.S.; Ishii-Iwamoto, E.L.; Constantin, J. Molecular mechanisms of citrus flavanones on hepatic gluconeogenesis. Fitoterapia 2014, 92, 148–162. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Oksbjerg, N.; Young, J.F.; Jeppesen, P.B. Caffeic acid, naringenin and quercetin enhance glucose-stimulated insulin secretion and glucose sensitivity in INS-1E cells. Diabetes Obes. Metab. 2014, 16, 602–612. [Google Scholar] [CrossRef]
- Li, J.M.; Che, C.T.; Lau, C.B.S.; Leung, P.S.; Cheng, C.H.K. Inhibition of intestinal and renal Na+-glucose cotransporter by naringenin. Int. J. Biochem. Cell Biol. 2006, 38, 985–995. [Google Scholar] [CrossRef]
- Ortiz-Andrade, R.R.; Sánchez-Salgado, J.C.; Navarrete-Vázquez, G.; Webster, S.P.; Binnie, M.; García-Jiménez, S.; León-Rivera, I.; Cigarroa-Vázquez, P.; Villalobos-Molina, R.; Estrada-Soto, S. Antidiabetic and toxicological evaluations of naringenin in normoglycaemic and NIDDM rat models and its implications on extra-pancreatic glucose regulation. Diabetes Obes. Metab. 2008, 10, 1097–1104. [Google Scholar] [CrossRef]
- Punithavathi, V.R.; Anuthama, R.; Prince, P.S.M. Combined treatment with naringin and vitamin C ameliorates streptozotocin-induced diabetes in male Wistar rats. J. Appl. Toxicol. 2008, 28, 806–813. [Google Scholar] [CrossRef]
- Tsai, S.-J.; Huang, C.-S.; Mong, M.-C.; Kam, W.-Y.; Huang, H.-Y.; Yin, M.-C. Anti-inflammatory and antifibrotic effects of naringenin in diabetic mice. J. Agric. Food Chem. 2012, 60, 514–521. [Google Scholar] [CrossRef]
- Sharma, A.K.; Bharti, S.; Ojha, S.; Bhatia, J.; Kumar, N.; Ray, R.; Kumari, S.; Arya, D.S. Up-regulation of PPARγ, heat shock protein-27 and -72 by naringin attenuates insulin resistance, β-cell dysfunction, hepatic steatosis and kidney damage in a rat model of type 2 diabetes. Br. J. Nutr. 2011, 106, 1713–1723. [Google Scholar] [CrossRef]
- Annadurai, T.; Muralidharan, A.R.; Joseph, T.; Hsu, M.J.; Thomas, P.A.; Geraldine, P. Antihyperglycemic and antioxidant effects of a flavanone, naringenin, in streptozotocin-nicotinamide-induced experimental diabetic rats. J. Physiol. Biochem. 2012, 68, 307–318. [Google Scholar] [CrossRef]
- Mahmoud, A.M.; Ashour, M.B.; Abdel-Moneim, A.; Ahmed, O.M. Hesperidin and naringin attenuate hyperglycemia-mediated oxidative stress and proinflammatory cytokine production in high fat fed/streptozotocin-induced type 2 diabetic rats. J. Diabetes Complicat. 2012, 26, 483–490. [Google Scholar] [CrossRef]
- Priscilla, D.H.; Roy, D.; Suresh, A.; Kumar, V.; Thirumurugan, K. Naringenin inhibits α-glucosidase activity: A promising strategy for the regulation of postprandial hyperglycemia in high fat diet fed streptozotocin induced diabetic rats. Chem. Biol. Interact. 2014, 210, 77–85. [Google Scholar] [CrossRef]
- Hasanein, P.; Fazeli, F. Role of naringenin in protection against diabetic hyperalgesia and tactile allodynia in male Wistar rats. J. Physiol. Biochem. 2014, 70, 997–1006. [Google Scholar] [CrossRef]
- Kapoor, R.; Kakkar, P. Naringenin accords hepatoprotection from streptozotocin induced diabetes in vivo by modulating mitochondrial dysfunction and apoptotic signaling cascade. Toxicol. Rep. 2014, 1, 569–581. [Google Scholar] [CrossRef]
- Al-Rejaie, S.S.; Aleisa, A.M.; Abuohashish, H.M.; Parmar, M.Y.; Ola, M.S.; Al-Hosaini, A.A.; Ahmed, M.M. Naringenin neutralises oxidative stress and nerve growth factor discrepancy in experimental diabetic neuropathy. Neurol. Res. 2015, 37, 924–933. [Google Scholar] [CrossRef]
- Ren, B.; Qin, W.; Wu, F.; Wang, S.; Pan, C.; Wang, L.; Zeng, B.; Ma, S.; Liang, J. Apigenin and naringenin regulate glucose and lipid metabolism, and ameliorate vascular dysfunction in type 2 diabetic rats. Eur. J. Pharmacol. 2016, 773, 13–23. [Google Scholar] [CrossRef]
- Yan, N.; Wen, L.; Peng, R.; Li, H.; Liu, H.; Peng, H.; Sun, Y.; Wu, T.; Chen, L.; Duan, Q.; et al. Naringenin Ameliorated Kidney Injury through Let-7a/TGFBR1 Signaling in Diabetic Nephropathy. J. Diabetes Res. 2016, 2016, 8738760. [Google Scholar] [CrossRef]
- Roy, S.; Ahmed, F.; Banerjee, S.; Saha, U. Naringenin ameliorates streptozotocin-induced diabetic rat renal impairment by downregulation of TGF-β1 and IL-1 via modulation of oxidative stress correlates with decreased apoptotic events. Pharm. Biol. 2016, 54, 1616–1627. [Google Scholar] [CrossRef]
- Al-Dosari, D.I.; Ahmed, M.M.; Al-Rejaie, S.S.; Alhomida, A.S.; Ola, M.S. Flavonoid Naringenin Attenuates Oxidative Stress, Apoptosis and Improves Neurotrophic Effects in the Diabetic Rat Retina. Nutrients 2017, 9, 1161. [Google Scholar] [CrossRef]
- Ahmed, O.M.; Hassan, M.A.; Abdel-Twab, S.M.; Abdel Azeem, M.N. Navel orange peel hydroethanolic extract, naringin and naringenin have anti-diabetic potentials in type 2 diabetic rats. Biomed. Pharmacother. 2017, 94, 197–205. [Google Scholar] [CrossRef]
- Singh, A.K.; Raj, V.; Keshari, A.K.; Rai, A.; Kumar, P.; Rawat, A.; Maity, B.; Kumar, D.; Prakash, A.; De, A.; et al. Isolated mangiferin and naringenin exert antidiabetic effect via PPARγ/GLUT4 dual agonistic action with strong metabolic regulation. Chem. Biol. Interact. 2018, 280, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Oršolić, N.; Gajski, G.; Garaj-Vrhovac, V.; Dikić, D.; Prskalo, Z.Š.; Sirovina, D. DNA-protective effects of quercetin or naringenin in alloxan-induced diabetic mice. Eur. J. Pharmacol. 2011, 656, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Sirovina, D.; Oršolić, N.; Gregorović, G.; Končić, M.Z. Naringenin ameliorates pathological changes in liver and kidney of diabetic mice: A preliminary study. Arh. Hig. Rada Toksikol. 2016, 67, 19–24. [Google Scholar] [CrossRef]
- Jung, U.J.; Lee, M.-K.; Park, Y.B.; Kang, M.A.; Choi, M.-S. Effect of citrus flavonoids on lipid metabolism and glucose-regulating enzyme mRNA levels in type-2 diabetic mice. Int. J. Biochem. Cell Biol. 2006, 38, 1134–1145. [Google Scholar] [CrossRef] [PubMed]
- Burke, A.C.; Telford, D.E.; Edwards, J.Y.; Sutherland, B.G.; Sawyez, C.G.; Huff, M.W. Naringenin Supplementation to a Chow Diet Enhances Energy Expenditure and Fatty Acid Oxidation, and Reduces Adiposity in Lean, Pair-Fed Ldlr−/− Mice. Mol. Nutr. Food Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.-M.; Park, Y.B.; Choi, M.-S. Antihypercholesterolemic property of naringin alters plasma and tissue lipids, cholesterol-regulating enzymes, fecal sterol and tissue morphology in rabbits. Clin. Nutr. 2004, 23, 1025–1034. [Google Scholar] [CrossRef] [PubMed]
- Mulvihill, E.E.; Allister, E.M.; Sutherland, B.G.; Telford, D.E.; Sawyez, C.G.; Edwards, J.Y.; Markle, J.M.; Hegele, R.A.; Huff, M.W. Naringenin prevents dyslipidemia, apolipoprotein B overproduction, and hyperinsulinemia in LDL receptor-null mice with diet-induced insulin resistance. Diabetes 2009, 58, 2198–2210. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.W.; Kim, Y.O.; Andrade, J.E.; Burgess, J.R.; Kim, Y.-C. Dietary naringenin increases hepatic peroxisome proliferators-activated receptor α protein expression and decreases plasma triglyceride and adiposity in rats. Eur. J. Nutr. 2011, 50, 81–88. [Google Scholar] [CrossRef]
- Pu, P.; Gao, D.-M.; Mohamed, S.; Chen, J.; Zhang, J.; Zhou, X.-Y.; Zhou, N.-J.; Xie, J.; Jiang, H. Naringin ameliorates metabolic syndrome by activating AMP-activated protein kinase in mice fed a high-fat diet. Arch. Biochem. Biophys. 2012, 518, 61–70. [Google Scholar] [CrossRef]
- Alam, M.A.; Kauter, K.; Brown, L. Naringin improves diet-induced cardiovascular dysfunction and obesity in high carbohydrate, high fat diet-fed rats. Nutrients 2013, 5, 637–650. [Google Scholar] [CrossRef]
- Assini, J.M.; Mulvihill, E.E.; Sutherland, B.G.; Telford, D.E.; Sawyez, C.G.; Felder, S.L.; Chhoker, S.; Edwards, J.Y.; Gros, R.; Huff, M.W. Naringenin prevents cholesterol-induced systemic inflammation, metabolic dysregulation, and atherosclerosis in Ldlr−/− mice. J. Lipid Res. 2013, 54, 711–724. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, H.; Watanabe, H.; Ishida, A.; Watanabe, W.; Narumi, K.; Atsumi, T.; Sugita, C.; Kurokawa, M. Naringenin suppresses macrophage infiltration into adipose tissue in an early phase of high-fat diet-induced obesity. Biochem. Biophys. Res. Commun. 2014, 454, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Assini, J.M.; Mulvihill, E.E.; Burke, A.C.; Sutherland, B.G.; Telford, D.E.; Chhoker, S.S.; Sawyez, C.G.; Drangova, M.; Adams, A.C.; Kharitonenkov, A.; et al. Naringenin prevents obesity, hepatic steatosis, and glucose intolerance in male mice independent of fibroblast growth factor 21. Endocrinology 2015, 156, 2087–2102. [Google Scholar] [CrossRef]
- Chtourou, Y.; Kamoun, Z.; Zarrouk, W.; Kebieche, M.; Kallel, C.; Gdoura, R.; Fetoui, H. Naringenin ameliorates renal and platelet purinergic signalling alterations in high-cholesterol fed rats through the suppression of ROS and NF-κB signaling pathways. Food Funct. 2016, 7, 183–193. [Google Scholar] [CrossRef]
- Burke, A.C.; Sutherland, B.G.; Telford, D.E.; Morrow, M.R.; Sawyez, C.G.; Edwards, J.Y.; Drangova, M.; Huff, M.W. Intervention with citrus flavonoids reverses obesity and improves metabolic syndrome and atherosclerosis in obese Ldlr−/− mice. J. Lipid Res. 2018, 59, 1714–1728. [Google Scholar] [CrossRef]
- Knekt, P.; Kumpulainen, J.; Järvinen, R.; Rissanen, H.; Heliövaara, M.; Reunanen, A.; Hakulinen, T.; Aromaa, A. Flavonoid intake and risk of chronic diseases. Am. J. Clin. Nutr. 2002, 76, 560–568. [Google Scholar] [CrossRef]
- Farook, V.S.; Reddivari, L.; Chittoor, G.; Puppala, S.; Arya, R.; Fowler, S.P.; Hunt, K.J.; Curran, J.E.; Comuzzie, A.G.; Lehman, D.M.; et al. Metabolites as Novel Biomarkers for Childhood Obesity-Related Traits in Mexican American Children. Pediatr. Obes. 2015, 10, 320–327. [Google Scholar] [CrossRef]
- Jung, U.J.; Kim, H.J.; Lee, J.S.; Lee, M.K.; Kim, H.O.; Park, E.J.; Kim, H.K.; Jeong, T.S.; Choi, M.S. Naringin supplementation lowers plasma lipids and enhances erythrocyte antioxidant enzyme activities in hypercholesterolemic subjects. Clin. Nutr. 2003, 22, 561–568. [Google Scholar] [CrossRef]
- Dallas, C.; Gerbi, A.; Elbez, Y.; Caillard, P.; Zamaria, N.; Cloarec, M. Clinical Study to Assess the Efficacy and Safety of a Citrus Polyphenolic Extract of Red Orange, Grapefruit, and Orange (Sinetrol-XPur) on Weight Management and Metabolic Parameters in Healthy Overweight Individuals. Phytother. Res. 2014, 28, 212–218. [Google Scholar] [CrossRef]
| Cell | Naringenin Concentration/Duration | Effect | Reference |
|---|---|---|---|
| L6 muscle cells | 10–75 µM, 2 h | ↑ Glucose uptake ↑ Phospho-AMPK | [46] |
| Primary porcine myotubes | Sambucus nigra flower (elderflower); Naringenin 0.1–10 µM, 1 h | ↑ Glucose uptake ↓ ROS levels | [47] |
| L6 myotubes insulin resistance induced by palmitate (750 µM) | 50 and 75 µM, 16 h | ↑ Glucose uptake ↑ GLUT4 translocation ↑ Phospho-AMPK ↑ SIRT1 ↑ PGC-1α | [48] |
| Cell | Naringenin Concentration/Duration | Effect | Reference |
|---|---|---|---|
| 3T3-L1 preadipocytes | 5–100 µM, 48 h | ↓ Adipocyte proliferation ↑ LDH release | [51] |
| 3T3-L1 adipocytes | 100 µM, 30 min | ↓ TNF-α FFA secretion ↓ IκB-α degradation ↓ Phospho-ERK protein expression ↑ Perilipin mRNA ↑ PDE3B mRNA | [52] |
| 3T3-L1 adipocytes and mature human adipocytes | 20 µM, 2 min | ↓ Insulin-stimulated glucose uptake ↓ GLUT4 recruitment | [53] |
| 3T3-L1 adipocytes | 10, 50 and 100 µM, 0.5–3 h | ↓ Inflammation ↓ TLR2 expression ↓ TNF-α ↓ MCP-1 | [54] |
| 3T3-L1 differentiating and mature adipocytes | 0–50 µM, 0–120 h (Pre-adipocytes) and 0–24 h (Mature) | ↓ Adipogenesis ↓ Lipid accumulation ↓ aP2, PPARγ, STAT5A and adiponectin protein ↓ IRS-1 (Y896) ↓ Adiponectin | [55] |
| Human white adipocytes | 8 µM, 7 to 14 days | ↑ GLUT4 mRNA ↑ Adiponectin mRNA ↑ UCP1, ATGL, CPT1β, PGC-1α and PGC-1β mRNA ↑ Oxygen consumption rate | [56] |
| Cell | Naringenin Concentration/Duration | Effect | Reference |
|---|---|---|---|
| HepG2 human hepatoma cells | 10–200 µM, 24 h | ↓ Apo B secretion ↓ ACAT2 mRNA ↓ MTP protein and mRNA ↑ LDL receptor mRNA ↑ LDL uptake ↑ LDL degradation | [58] |
| HepG2 human hepatoma cells | 200 µM, 6 h | ↓ Apolipoprotein B secretion ↑ SREBP-1 and LDLr expression ↑ PI3K activity | [59] |
| HepG2 human hepatoma cells | 0–200 µM, 60 min | ↓ Apo B secretion ↑ ERK activity ↓ Microsomal triglyceride transfer protein | [60] |
| Hepatoma (Fao) cells | 6–100 µM, 6 h | ↓ Glucose production ↓ Cellular ATP levels | [61] |
| Huh7 hepatocytes and Lewis rat primary hepatocytes | 0–380 µM, 16–24 h | ↓ Triglyceride production ↑ Fatty acid oxidation ↓Trap220/Drip-2 and LBD ↓ LXRα response element ↑ mRNA of CYP4A11, ACOX, UCP1 and ApoAI | [62] |
| Wistar rat hepatocytes | 300 µM, 30–50 min | ↓ Glucose production ↓ Gluconeogenesis ↓ Pyruvate transport | [63] |
| Cell | Naringenin Concentration/Duration | Effect | Reference |
|---|---|---|---|
| INS-1E cells | 100–1000 µM, 1 and 72 h | ↑ Glucose-stimulated insulin secretion ↑ Glut2, Gck, Ins1/2, Beta2 and Pdx1 mRNA ↑ Akt1, Akt2, Irs1, Bcl2 and Hsp70/90 mRNA ↓ Bax, Casp3, and Acc1 mRNA | [64] |
| Streptozotocin (STZ)-Induced Diabetes Animal Models | ||||
|---|---|---|---|---|
| Animal | Naringenin Concentration/Duration | Blood Measures | Other Measures | Reference |
| Neonatal Wistar rats and New Zealand White rabbits | 0.1–1000 mM, 1–2 min | No effect | ↓ Intestinal BBMV glucose uptake ↓ renal BBMV glucose uptake | [65] |
| Wistar rats | 5 and 50 mg/kg b.w., 5 days | ↓ Glucose levels ↓ Total Cholesterol ↓ Triglycerides ↑ HDL levels | No effect | [66] |
| Wistar rats | 30 mg/kg b.w., 21 days | ↓ Glucose levels ↑ Insulin levels ↑ Vitamin E levels | ↑ Kidney and liver hexokinase activity ↓ Liver and kidney G6Pase and F16BPase ↓ Glycoprotein levels | [67] |
| Male BALB/cA mice | 0.5–2% dietary intake, 10 weeks | ↓ Glucose levels ↑ Insulin levels | ↓ Blood urea nitrogen levels ↓ Kidney NF-κB p65 ↓ Renal TNFα ↓ Renal IL-1β ↓ Renal IL-6 ↓ Renal CCP-1 | [68] |
| Wistar albino rats | 25, 50 and 100 mg/kg. b.w/day, 28 days | ↓ Glucose levels ↑ Insulin levels ↓ Triglyceride, TAG, LDL and NEFA levels | ↑ Liver and kidney PPARγ ↑ HSP-27 and HSP-72 protein ↓ Pancreatic, liver and kidney NF-κB protein ↓ Liver TNFα ↓ Renal IL-6 | [69] |
| Albino Wistar rats | 50 mg/kg b.w./day, 21 days | ↓ Fasting-glucose levels ↓ HbA1c ↑ Insulin levels | ↑ SOD ↑ Catalase ↑ GSH peroxidase ↑ Glutathione-S-transferase activity | [70] |
| Male albino rats | 50 mg/kg b.w., 4 weeks | ↓ Glucose levels ↑ Insulin levels ↓ HbA1c levels | ↓ Lipid peroxide and NO levels ↑ Vitamin C, vitamin E and GSH levels ↓ TNF-α and IL-6 levels | [71] |
| Albino Wistar rats | 25 mg/kg p.o., 2 h | ↓ Glucose in response to maltose and sucrose load | ↓ α-glucosidase activity | [72] |
| Male Wistar rats | 20, 50, and 100 mg/kg p.o, 8 weeks | ↓ Glucose levels | ↓ Hyperalgesia ↑ SOD ↑ Body weight | [73] |
| Male Wistar rats | 50 mg/kg b.w., 30 days | ↓ Glucose levels | ↓ Oxidative stress ↓ Liver ROS ↓ Liver lipid peroxidation ↓ Bax and Bcl-2 mRNA and protein | [74] |
| Male Wistar rats | 25 and 50 mg/kg b.w./day, 5 weeks | ↓ Glucose levels ↑ Insulin levels ↓ TNF-α, IL-1β and IL-6 levels | ↑ Sciatic IGF expression ↑ Sciatic NGF expression ↓ TBARS, GSH levels ↑ SOD and catalase activity ↓ Sciatic histology | [75] |
| Sprague-Dawley rats | 50 and 100 mg/kg/day, 6 weeks | ↑ Glucose tolerance ↓ Glucose levels ↓ Lipid levels | ↓ ICAM-1 ↓ Malonaldehyde levels | [76] |
| Sprague–Dawley rats | 50 mg/kg b.w./day, 6 weeks | ↓ Glucose levels | ↓ Urinary protein levels ↓ Kidney index ↑ Creatinine clearance ratio ↓ ECM deposition ↑ let-7a signaling ↓ TGF-β1 and TGFBR1 mRNA and protein ↓ Smad2 and smad7 mRNA and protein | [77] |
| Male Wistar rats | 50 and 100 mg/kg b.w., 10 weeks | ↓ Glucose levels ↓ Total cholesterol levels | ↑ Kidney SOD ↑ Kidney Catalase ↑ Kidney GSH ↓ Renal IL-1 | [78] |
| Male Wistar rats | 50 mg/kg b.w./day, 5 weeks | ↓ Glucose levels ↑ Insulin levels | ↓ Retinal TBAR levels ↓ GSH levels ↑ BDNF and TrkB levels ↑ Synaptophysin levels ↑ Bcl-2 protein ↓ Bax and Caspase-3 protein | [79] |
| Male albino rats | 100 mg/kg b.w./day, 4 weeks | ↑ Insulin levels ↑ Lipid profile | ↑ GLUT4 mRNA ↓ Liver G6Pase activity | [80] |
| Male albino rats | 100 mg/kg/day, 15 days | ↓ Glucose levels ↑ Lipid levels | ↑ GLUT4 activity ↑ PPARγ in the pancreas | [81] |
| Alloxan-Induced Diabetes Animal Model | ||||
|---|---|---|---|---|
| Animals | Naringenin Concentration/Duration | Blood Measures | Other Measures | Reference |
| Swiss albino mice | 50 mg/kg b.w., 7 days | ↑ Immunological parameters ↓ Cholesterol levels | ↑ Survival | [82] |
| Swiss albino mice | 50 mg/kg b.w., 7 days | ↓ Lipid peroxidation levels | ↑ Tissue repair ↓ Liver vacuolisation | [83] |
| Genetically-Induced Diabetes Animal Model | ||||
|---|---|---|---|---|
| Animals | Naringenin Concentration/Duration | Blood Measures | Other Measures | Reference |
| C57BL/KsJ-db/db mice | 0.2 g/kg diet, 5 weeks | ↓ Glucose levels ↓ Lipid levels | ↓ Hepatic FAS, G6Pase and PAP activity ↓ Hepatic fatty acid β-oxidation ↑ Hepatic GK mRNA ↑ GLUT4 levels ↓ GLUT2 levels | [84] |
| Ldlr−/− mice | 3% wt/wt, 8 weeks | ↓ Glucose levels ↓ Lipid levels ↓ Insulin levels ↑ β-hydroxybutyrate | ↑ Energy expenditure ↑ Pgc1a mRNA ↑ Cpt1a mRNA ↑ Pnpla2 mRNA | [85] |
| Diet-Induced Diabetes Model | ||||
|---|---|---|---|---|
| Animal Model | Naringenin Concentration/Duration | Blood Measures | Other Measures | Reference |
| Male rabbits; high-cholesterol diet | 0.05% naringin, 8 weeks | ↓ LDL-cholesterol ↑ HDLD cholesterol | ↑ Hepatic CoA reductase ↓ Acyl-CoA cholesterol acyltransferase activity | [86] |
| Ldlr−/− mice; high fat diet | 1–3% wt/wt, 4 weeks | ↑ Hepatic glucose tolerance | ↓ Hepatic cholesterol ↓ Muscle VLDL fatty acids ↑ Hepatic fatty acid oxidation | [87] |
| Ldlr−/− mice; high fat diet | 3% wt/wt, 6 months | ↓ Triglyceride levels ↓ Cholesterol levels | ↓ Aortic plaque deposits ↓ Atherosclerosis ↓ Liver triglyceride ↓ Liver cholesteryl ester mass | [38] |
| Wistar rats; high fructose diet | 50 mg/kg b.w, 45 days | ↑ Glycogen content (liver and skeletal muscle) | ↑ phospho-PTK | [43] |
| Male Long-Evans hooded rats; high-sucrose diet | 0.003, 0.006 and 0.012% dietary consumption, 6 weeks | ↓ Triglyceride levels ↓ Cholesterol levels | ↓ Hepatic cholesterol ↓ Adipose triglyceride levels ↑ PPARα protein ↑ CPT-1 and UCP2 protein | [88] |
| C57BL/6 mice; high-fat diet | 0.2 g/kg diet, 20 weeks | No effect | ↓ MAPK signaling ↑ IRS1 ↑ Fatty acid oxidation ↑ AMPK activation | [89] |
| BALB/cA mice; high fat-fed diet | 0.5–2% dietary intake, 4 months | ↓ Glucose levels ↑ Insulin levels | ↓ TNF-α ↓ MCP-1 ↓ TLR2 in adipose tissues | [54] |
| Wistar rats; high-fat-high-cholesterol diet | 100 mg/kg/day, 16 weeks | ↓ Glucose levels ↓ Insulin levels ↓ Cholesterol levels ↓ Triglyceride levels ↓ NEFA levels ↓ AST and ALT activity | ↓ Inflammatory cell infiltration ↓ Collagen deposition ↑ Mitochondrial respiration state 3 rates | [90] |
| Ldlr−/− mice | 3% naringenin, 12 weeks | ↓ Glucose levels ↓ Insulin levels ↓ Lipid levels | ↓ ApoB100 secretion ↓ Srebf1c and Acox1 mRNA ↑ Fgf21, Pgc1a, and Cpt1a mRNA ↓ Tnfa, Il1b, Ccl2, and Ccl3 mRNA | [91] |
| C57BL/6J; high fat diet | 100 mg/kg/day, 14 days | No effect | ↓ MCP-1 ↓ Macrophage infiltration in adipose tissues | [92] |
| Albino Wistar rats; high fat diet | 25 mg/kg p.o., 2 h | ↓ Glucose levels | ↓ α-glucosidase activity | [72] |
| C57BL6/J and FGF21−/− mice; high-fat diet | 3% wt/wt, 4 weeks | ↓ Glucose levels ↑ Insulin levels ↓ Leptin levels ↓ TNFα levels | ↓ Obesity ↓ Adipose tissue volume ↓ Pgc1a and Cpt1a mRNA ↑ Pnpla2 and Lipe mRNA | [93] |
| Male Wistar rats; high cholesterol diet | 50 mg/kg b.w./day, 90 days | ↓ Lipid levels | ↓ Renal urea levels ↑ Creatinine clearance rate ↓ Renal WBCs and platelets levels ↓ NTPDases, CD73, iNOS, TNF-α, IL-6 and NF-κB mRNA | [94] |
| Albino Wistar rats; high fructose diet | 50 mg/kg b.w./day, 6 weeks | No effect | ↑ Skeletal muscle GLUT4 translocation ↑ Phospho-AMPK ↑ SIRT1 ↑ PGC-1α | [48] |
| Ldlr−/− mice; high-fat/high-cholesterol diet | 3% wt/wt, 12 weeks | ↓ Fasting glucose levels ↓ Fasting insulin levels | ↓ Hepatic lipid levels ↓ Skeletal muscle lipid levels ↑ Pgc1a and Cpt1a mRNA | [95] |
| Naringenin Concentration | Blood Measures | Other Measures | Reference |
|---|---|---|---|
| 3.7–5.0 mg/kg/day | ↓ Cholesterol levels | ↓ T2DM risk ↓ Asthma ↓ Ischemic heart disease ↓ Mortality | [96] |
| 273.08 mg/mL | ↓ Triglyceride levels ↑ HDL levels | ↓ Obesity ↓ Systolic and diastolic blood pressure | [97] |
| 400 mg/capsule/day | ↓ Cholesterol levels ↓ LDL levels ↓ ApoB levels ↓ TBARS levels | ↑ SOD activity ↑ CAT activity ↓ GSH-Px activity | [98] |
| 900 mg/kg | ↓ Glucose levels | ↓ Body weight | [99] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Den Hartogh, D.J.; Tsiani, E. Antidiabetic Properties of Naringenin: A Citrus Fruit Polyphenol. Biomolecules 2019, 9, 99. https://doi.org/10.3390/biom9030099
Den Hartogh DJ, Tsiani E. Antidiabetic Properties of Naringenin: A Citrus Fruit Polyphenol. Biomolecules. 2019; 9(3):99. https://doi.org/10.3390/biom9030099
Chicago/Turabian StyleDen Hartogh, Danja J., and Evangelia Tsiani. 2019. "Antidiabetic Properties of Naringenin: A Citrus Fruit Polyphenol" Biomolecules 9, no. 3: 99. https://doi.org/10.3390/biom9030099
APA StyleDen Hartogh, D. J., & Tsiani, E. (2019). Antidiabetic Properties of Naringenin: A Citrus Fruit Polyphenol. Biomolecules, 9(3), 99. https://doi.org/10.3390/biom9030099






