Human Semen Samples with High Antioxidant Reservoir May Exhibit Lower Post-Cryopreservation Recovery of Sperm Motility
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects and Sample Collection
2.2. Ethical Considerations
2.3. Experimental Design
2.4. Assessment of Sperm Motility
2.5. Measurement of Total Antioxidant Capacity
2.6. Cryopreservation
2.7. Statistical Analysis
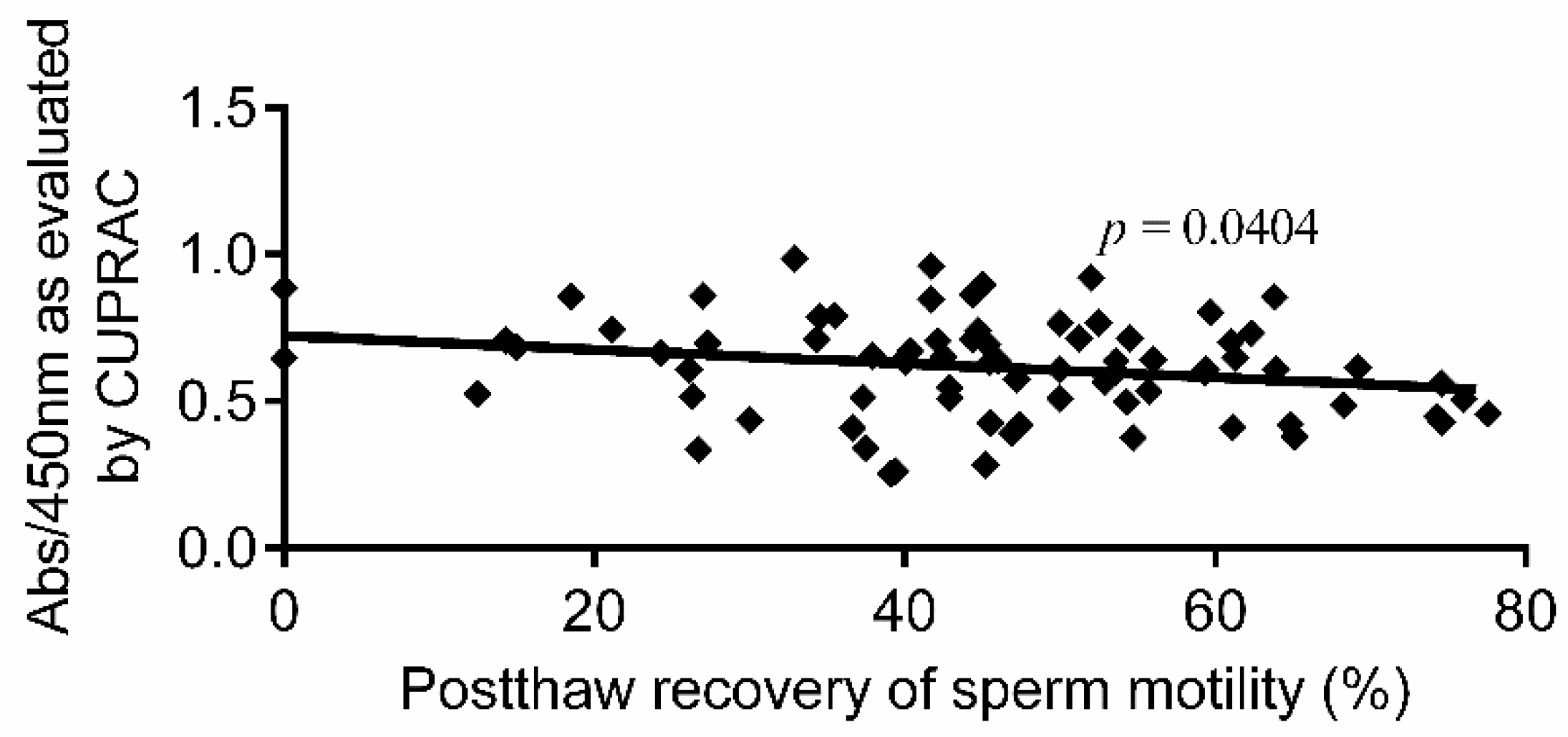
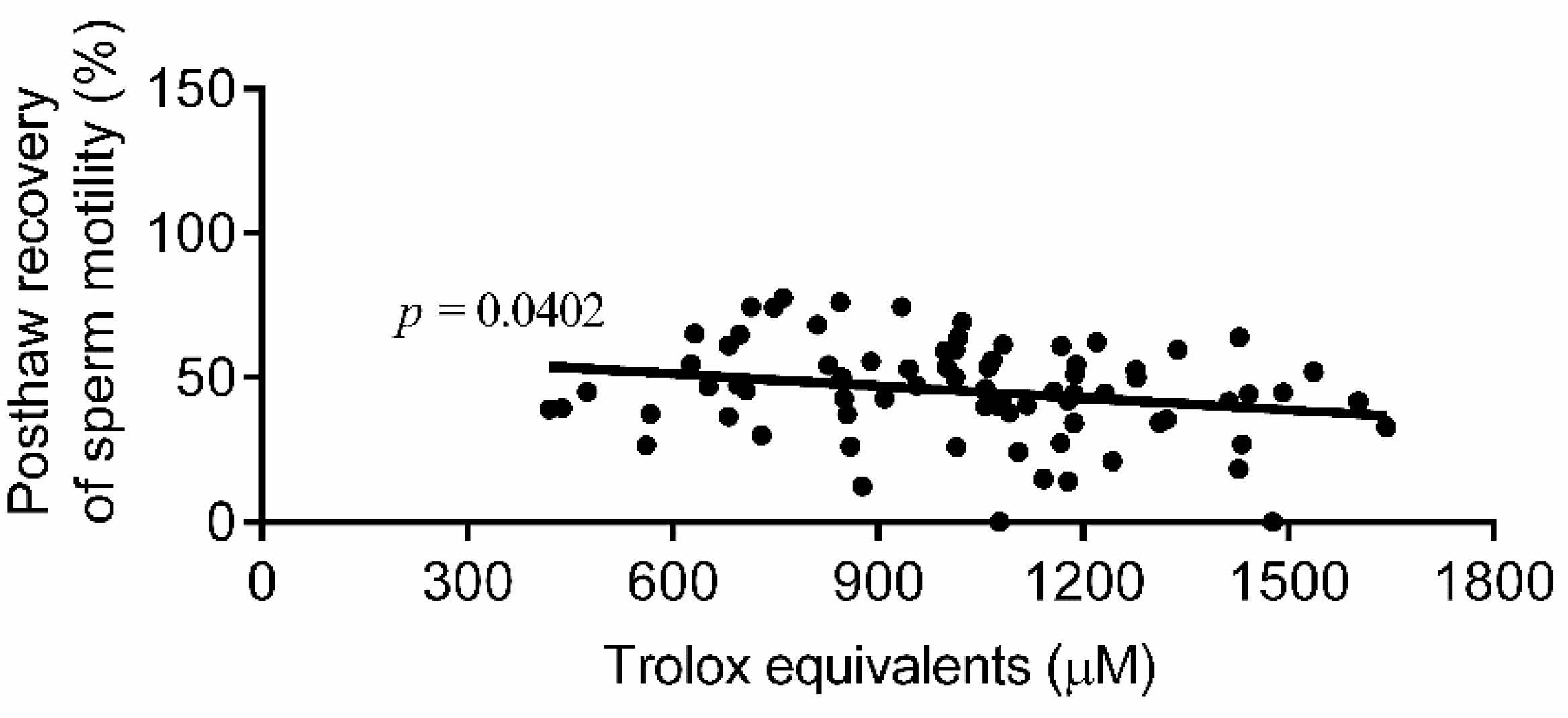
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Boitrelle, F.; Albert, M.; Theillac, C.; Ferfouri, F.; Bergere, M.; Vialard, F.; Wainer, R.; Bailly, M.; Selva, J. Cryopreservation of human spermatozoa decreases the number of motile normal spermatozoa, induces nuclear vacuolization and chromatin decondensation. J. Androl. 2012, 33, 1371–1378. [Google Scholar] [CrossRef] [PubMed]
- Azadi, L.; Tavalaee, M.; Deemeh, M.R.; Arbabian, M.; Nasr-Esfahani, M.H. Effects of tempol and quercetin on human sperm function after cryopreservation. Cryo Lett. 2017, 38, 29–36. [Google Scholar]
- Amidi, F.; Pazhohan, A.; Shabani Nashtaei, M.; Khodarahmian, M.; Nekoonam, S. The role of antioxidants in sperm freezing: A review. Cell Tissue Bank. 2016, 17, 745–756. [Google Scholar] [CrossRef]
- Nekoonam, S.; Nashtaei, M.S.; Naji, M.; Zangi, B.M.; Amidi, F. Effect of Trolox on sperm quality in normozospermia and oligozospermia during cryopreservation. Cryobiology 2016, 72, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Cooper, T.G.; Noonan, E.; von Eckardstein, S.; Auger, J.; Baker, H.W.; Behre, H.M.; Haugen, T.B.; Kruger, T.; Wang, C.; Mbizvo, M.T.; et al. World health organization reference values for human semen characteristics. Hum. Reprod. Update 2010, 16, 231–245. [Google Scholar] [CrossRef]
- El-Migdadi, F.; Banihani, I.; Banihani, S.A. Clinico-hormonal correlation of oligospermic patients in the below sea level environment (Jordan Valley). Neuro Endocrinol. Lett. 2005, 26, 13–18. [Google Scholar] [PubMed]
- Banihani, S.; Agarwal, A.; Sharma, R.; Bayachou, M. Cryoprotective effect of l-carnitine on motility, vitality and DNA oxidation of human spermatozoa. Andrologia 2014, 46, 637–641. [Google Scholar] [CrossRef]
- Celik, S.E.; Ozyurek, M.; Guclu, K.; Apak, R. Differences in responsivity of original cupric reducing antioxidant capacity and cupric-bathocuproine sulfonate assays to antioxidant compounds. Anal. Biochem. 2012, 423, 36–38. [Google Scholar] [CrossRef]
- Gosmaro, F.; Bagnati, M.; Berto, S.; Bellomo, G.; Prenesti, E. Measurement of total antioxidant capacity of human plasma: Setting and validation of the CUPRAC-BCs method on routine apparatus ADIVA 2400. Talanta 2013, 115, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Banihani, S.A.; Alawneh, R.F.; Abu-Awad, A. Human semen cryopreservation reduces the seminal antioxidant reservoir. N. Z. J. Med. Lab. Sci. 2016, 70, 3–6. [Google Scholar]
- Donnelly, E.T.; McClure, N.; Lewis, S.E. Antioxidant supplementation in vitro does not improve human sperm motility. Fertil. Steril. 1999, 72, 484–495. [Google Scholar] [CrossRef]
- Banihani, S.; Sharma, R.; Bayachou, M.; Sabanegh, E.; Agarwal, A. Human sperm DNA oxidation, motility and viability in the presence of l-carnitine during in vitro incubation and centrifugation. Andrologia 2012, 44, 505–512. [Google Scholar] [CrossRef]
- Duru, N.K.; Morshedi, M.; Schuffner, A.; Oehninger, S. Semen treatment with progesterone and/or acetyl-l-carnitine does not improve sperm motility or membrane damage after cryopreservation-thawing. Fertil. Steril. 2000, 74, 715–720. [Google Scholar] [CrossRef]
- Sanchez-Partida, L.G.; Setchell, B.P.; Maxwell, W.M. Epididymal compounds and antioxidants in diluents for the frozen storage of ram spermatozoa. Reprod. Fertil. Dev. 1997, 9, 689–696. [Google Scholar] [CrossRef]
- Banihani, S.A. Role of uric acid in semen. Biomolecules 2018, 8, 65. [Google Scholar] [CrossRef]
- Banihani, S.A. Effect of captopril on semen quality. Andrologia 2017, 49, e12641. [Google Scholar] [CrossRef]
- Bisht, S.; Dada, R. Oxidative stress: Major executioner in disease pathology, role in sperm DNA damage and preventive strategies. Front. Biosci. 2017, 9, 420–447. [Google Scholar]
- Banihani, S.A. Vitamin B12 and semen quality. Biomolecules 2017, 7, 42. [Google Scholar] [CrossRef] [PubMed]
- Banihani, S.A. Effect of paracetamol on semen quality. Andrologia 2018, 50, e12874. [Google Scholar] [CrossRef] [PubMed]
- Bisht, S.; Faiq, M.; Tolahunase, M.; Dada, R. Oxidative stress and male infertility. Nat. Rev. Urol. 2017, 14, 470–485. [Google Scholar] [CrossRef] [PubMed]
- Mostofa, A.G.; Punganuru, S.R.; Madala, H.R.; Al-Obaide, M.; Srivenugopal, K.S. The process and regulatory components of inflammation in brain oncogenesis. Biomolecules 2017, 7, 34. [Google Scholar] [CrossRef]
- Banihani, S.A. Ginger and testosterone. Biomolecules 2018, 8, 119. [Google Scholar] [CrossRef] [PubMed]
- Gutteridge, J.M. Antioxidants, nutritional supplements and life-threatening diseases. Br. J. Biomed. Sci. 1994, 51, 288–295. [Google Scholar] [PubMed]
- Menezo, Y.J.; Hazout, A.; Panteix, G.; Robert, F.; Rollet, J.; Cohen-Bacrie, P.; Chapuis, F.; Clement, P.; Benkhalifa, M. Antioxidants to reduce sperm DNA fragmentation: An unexpected adverse effect. Reprod. Biomed. Online 2007, 14, 418–421. [Google Scholar] [CrossRef]
- Zareba, P.; Colaci, D.S.; Afeiche, M.; Gaskins, A.J.; Jorgensen, N.; Mendiola, J.; Swan, S.H.; Chavarro, J.E. Semen quality in relation to antioxidant intake in a healthy male population. Fertil. Steril. 2013, 100, 1572–1579. [Google Scholar] [CrossRef] [PubMed]
- Sardi, B. High-dose vitamin C and iron overload. Ann. Intern. Med. 2004, 140, 846–847. [Google Scholar] [CrossRef] [PubMed]
- Hininger, I.; Waters, R.; Osman, M.; Garrel, C.; Fernholz, K.; Roussel, A.M.; Anderson, R.A. Acute prooxidant effects of vitamin c in EDTA chelation therapy and long-term antioxidant benefits of therapy. Free Radic. Biol. Med. 2005, 38, 1565–1570. [Google Scholar] [CrossRef] [PubMed]
- Podmore, I.D.; Griffiths, H.R.; Herbert, K.E.; Mistry, N.; Mistry, P.; Lunec, J. Vitamin C exhibits pro-oxidant properties. Nature 1998, 392, 559. [Google Scholar] [CrossRef]
- Castro, M.; Carson, G.; McConnell, M.; Herst, P. High-dose ascorbate causes both genotoxic and metabolic stress in glioma cells. Antioxidants 2017, 6, 58. [Google Scholar] [CrossRef]
- Buettner, G.R.; Jurkiewicz, B.A. Catalytic metals, ascorbate and free radicals: Combinations to avoid. Radiat. Res. 1996, 145, 532–541. [Google Scholar] [CrossRef]
- Hani, S.B.; Bayachou, M. Salvia fruticosa reduces intrinsic cellular and H2O2-induced DNA oxidation in HEK 293 cells; assessment using flow cytometry. Asian Pac. J. Trop. Biomed. 2014, 4, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Otto, C.C.; Koehl, J.L.; Solanky, D.; Haydel, S.E. Metal ions, not metal-catalyzed oxidative stress, cause clay leachate antibacterial activity. PLoS ONE 2014, 9, e115172. [Google Scholar] [CrossRef] [PubMed]
| Semen Parameter | Mean ± SEM |
|---|---|
| Total semen volume (mL) | 3.44 ± 0.183 |
| Sperm count (mL−1) | 56.76 ± 5.08 |
| Sperm motility before cryopreservation (%) | 52.95 ± 2.07 |
| Sperm motility after cryopreservation (%) | 22.66 ± 1.51 |
| Sperm recovery (%) | 42.79 ± 1.82 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Banihani, S.A.; Alawneh, R.F. Human Semen Samples with High Antioxidant Reservoir May Exhibit Lower Post-Cryopreservation Recovery of Sperm Motility. Biomolecules 2019, 9, 111. https://doi.org/10.3390/biom9030111
Banihani SA, Alawneh RF. Human Semen Samples with High Antioxidant Reservoir May Exhibit Lower Post-Cryopreservation Recovery of Sperm Motility. Biomolecules. 2019; 9(3):111. https://doi.org/10.3390/biom9030111
Chicago/Turabian StyleBanihani, Saleem A., and Razan F. Alawneh. 2019. "Human Semen Samples with High Antioxidant Reservoir May Exhibit Lower Post-Cryopreservation Recovery of Sperm Motility" Biomolecules 9, no. 3: 111. https://doi.org/10.3390/biom9030111
APA StyleBanihani, S. A., & Alawneh, R. F. (2019). Human Semen Samples with High Antioxidant Reservoir May Exhibit Lower Post-Cryopreservation Recovery of Sperm Motility. Biomolecules, 9(3), 111. https://doi.org/10.3390/biom9030111





