Effects of Winter Flounder Antifreeze Protein on the Growth of Ice Particles in an Ice Slurry Flow in Mini-Channels
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materals
2.2. Preheating
2.3. Ultrafiltration
2.4. Production of Ice Slurry
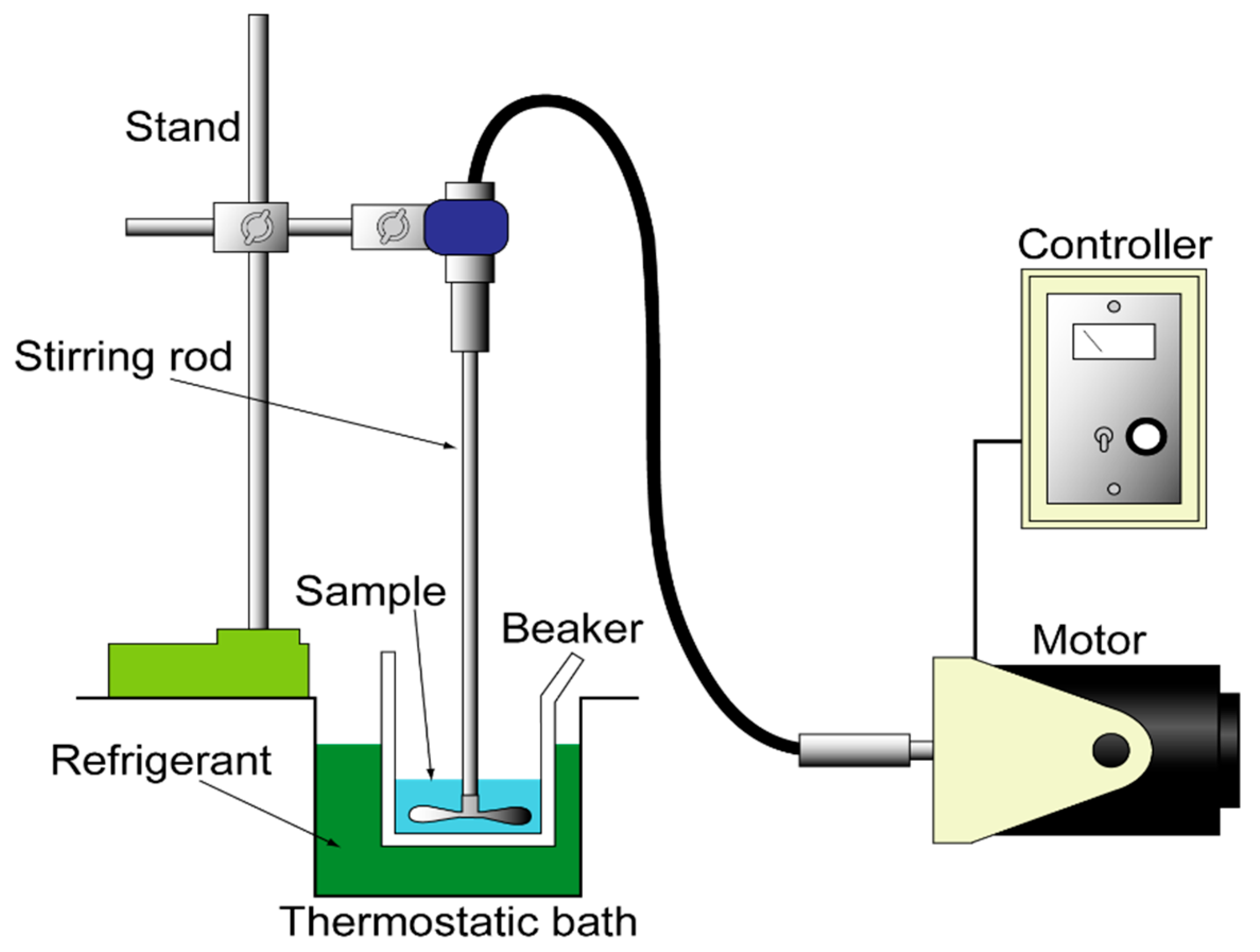
2.5. Apparatus
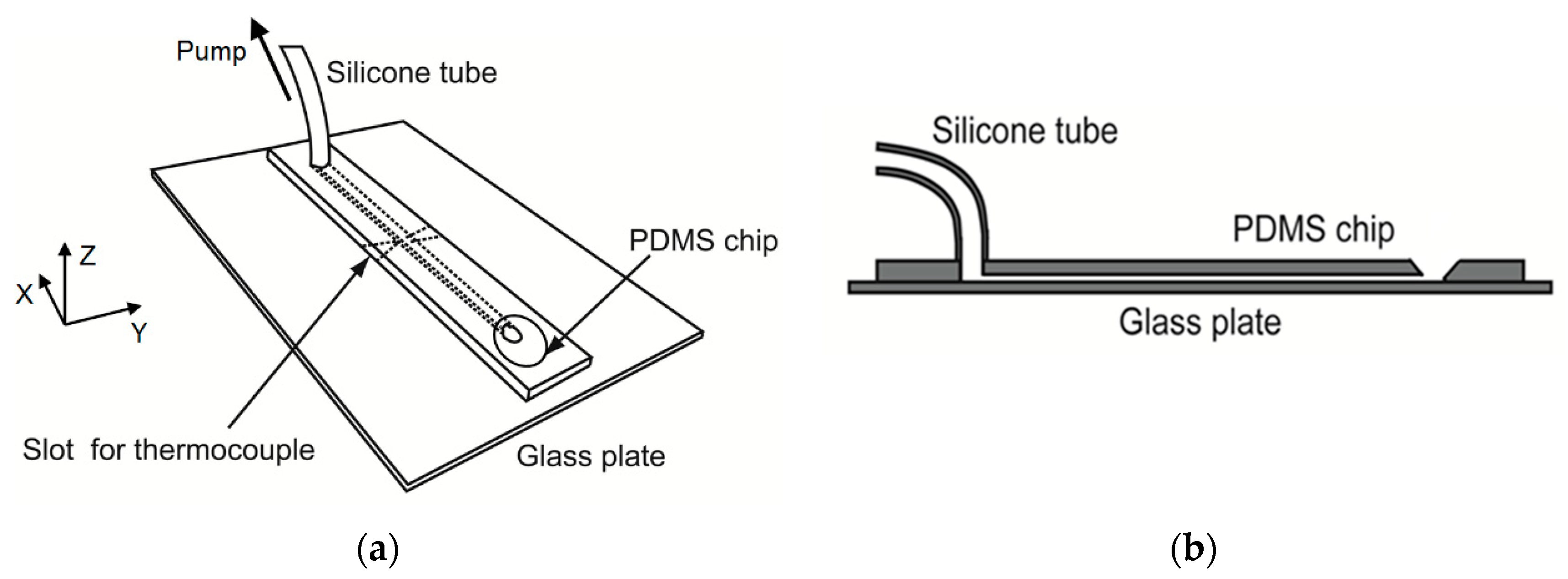
2.6. Mini-Channels
2.7. Cooling of Channels
2.8. Image-Capturing Condition
3. Results and Discussion
3.1. In the Case of Quiescent Ice Slurry
3.1.1. Ice Particle Shapes
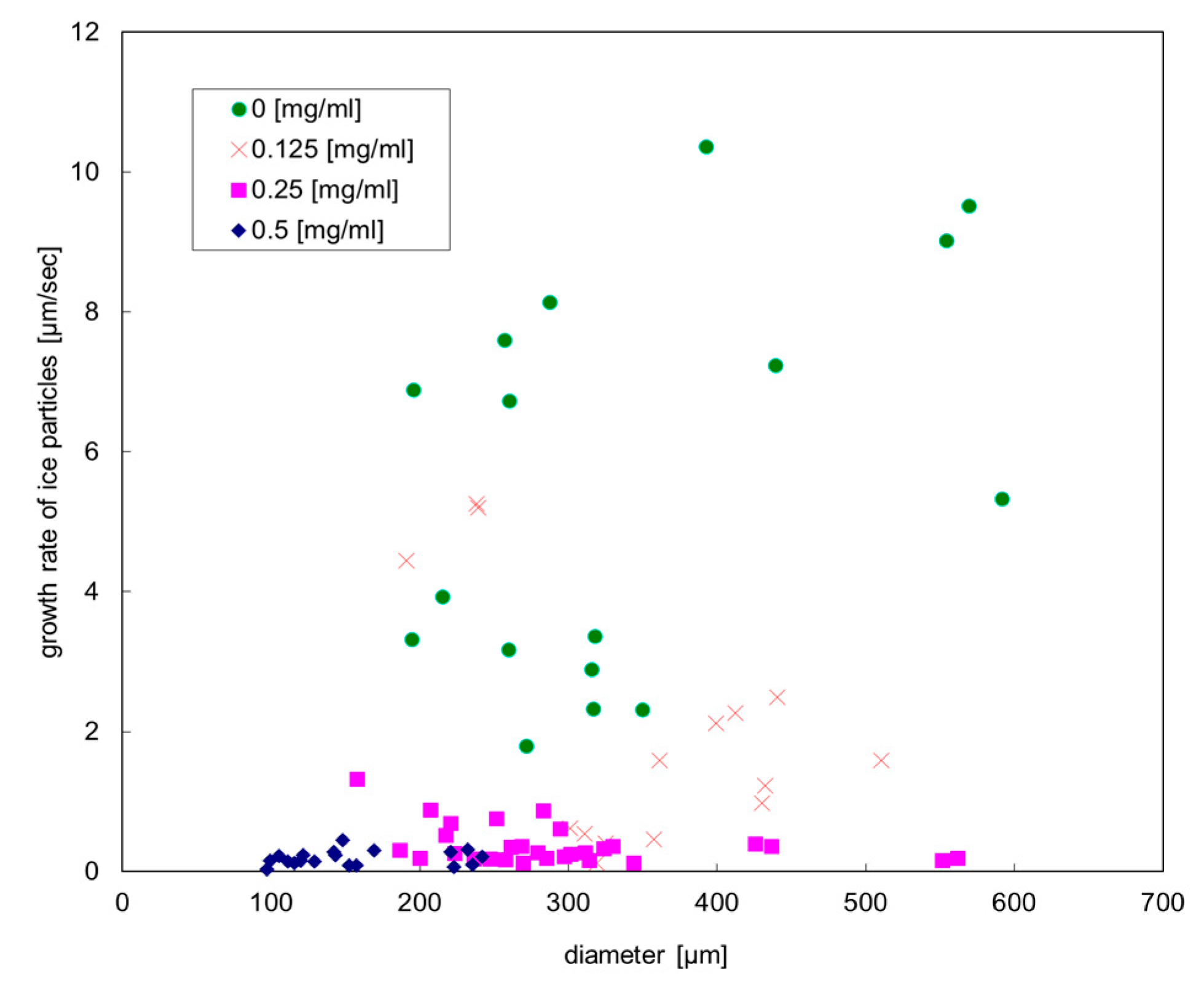
3.1.2. Ice Growth Rate
3.2. In the Case of Ice Slurry Flow
3.3. Effects of Preheating of HPLC6 Solution
3.4. Effects of Ultrafiltration of Preheated HPLC6 Solution
4. Conclusions
- (1)
- The ice growth inhibition by the antifreeze protein decreased in flowing solutions compared with that in quiescent solutions. This is because the slurry flow carried HPLC6 and the latent heat away from the surfaces of ice particles locally.
- (2)
- The preheating of the antifreeze protein solution before mixing with the ice slurry reduced the ice growth inhibition properties for the ice slurry flow. As the direction of flow, containing HPLC6 and its aggregates, to the ice particle surfaces can change as the ice particle grows, the probability of interaction between HPLC6 and ice surfaces does not increase with the ice particle growth.
- (3)
- The ultrafiltration after preheating the solution improved the ice growth inhibition. In a specific case, the inhibition was enhanced because many small aggregates and large aggregates remaining in the concentrate could interact with ice particle surfaces effectively.
Author Contributions
Funding
Conflicts of Interest
References
- Shitzer, A. Cryosurgery: Analysis and experimentation of cryoprobes in phase changing media. J. Heat Transf. 2011, 133, 011005. [Google Scholar] [CrossRef]
- Amir, G.; Rubinsky, B.; Basheer, S.Y.; Horowitz, L.; Jonathan, L.; Feinberg, M.S.; Smolinsky, A.K.; Lavee, L. Improved viability and reduced apoptosis in sub-zero 21-h preservation of transplanted rat hearts using anti-freeze proteins. J. Heart Lung Transpl. 2005, 24, 1915–1929. [Google Scholar] [CrossRef] [PubMed]
- Haymet, A.D.J.; Ward, L.G.; Harding, M.M. Winter flounder “antifreeze” proteins: Synthesis and ice growth inhibition of analogues that probe the relative importance of hydrophobic and hydrogen-bonding interactions. J. Am. Chem. Soc. 1999, 121, 941–948. [Google Scholar] [CrossRef]
- Loewen, M.C.; Chao, H.; Houston, M.E., Jr.; Baardsnes, J.; Hodges, R.S.; Kay, C.M.; Sykes, B.D.; Sönnichsen, F.D.; Davies, P.L. Alternative roles for putative ice-binding residues in type I antifreeze protein. Biochemistry 1999, 38, 4743–4749. [Google Scholar] [CrossRef] [PubMed]
- Chao, H.; Houston, M.E., Jr.; Hodges, R.S.; Kay, C.M.; Sykes, B.D.; Loewen, M.C.; Davies, P.L.; Sönnichsen, F.D. A diminished role for hydrogen bonds in antifreeze protein binding to ice. Biochemistry 1997, 36, 14652–14660. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.W.; Heneghan, A.F.; Haymet, A.D.J. Ice nucleation in nature: Supercooling point (SCP) measurements and the role of heterogeneous nucleation. Cryobiology 2003, 46, 88–98. [Google Scholar] [CrossRef]
- Coger, R.; Rubinsky, B.; Fletcher, G.L. Microscopic pattern of ice crystal growth in the presence of thermal hysteresis proteins. J. Offshore Mech. Arctic Eng. (Trans. ASME) 1994, 116, 173–179. [Google Scholar] [CrossRef]
- Furukawa, Y.; Inohara, N.; Yokoyama, E. Growth patterns and interfacial kinetic supercooling at ice/water interfaces at which anti-freeze glycoprotein molecules are adsorbed. J. Cryst. Growth 2005, 275, 167–174. [Google Scholar] [CrossRef]
- Butler, M.F. Freeze concentration of solutes at the ice/solution interface studied by optical interferometry. Cryst. Growth Des. 2002, 2, 541–548. [Google Scholar] [CrossRef]
- Hagiwara, Y.; Yamamoto, D. Temperature distribution and local heat flux in the unidirectional freezing of antifreeze-protein solution. Int. J. Heat Mass Transf. 2012, 55, 2384–2393. [Google Scholar] [CrossRef]
- Hagiwara, Y.; Aomatsu, H. Supercooling enhancement by adding antifreeze protein and ions to water in a narrow space. Int. J. Heat Mass Transf. 2015, 86, 55–64. [Google Scholar] [CrossRef]
- Miyamoto, T.; Nishi, N.; Waku, T.; Tanaka, N.; Hagiwara, Y. Effects of short-time preheating on ice growth in antifreeze polypeptides solutions in a narrow space. Heat Mass Transf. 2018, 54, 2415–2424. [Google Scholar] [CrossRef]
- Grandum, S.; Yabe, A.; Nakagomi, K.; Tanaka, M.; Takemura, F.; Kobayashi, Y.; Ikemoto, M.; Frivik, P. Investigation of the characteristics of ice slurry containing antifreeze protein for ice storage applications. Trans. Jpn. Soc. Mech. Ser. B 1997, 63, 1770–1776. (In Japanese) [Google Scholar] [CrossRef]
- Onishi, Y.; Nakagawa, Y.; Kitagawa, A.; Hagiwara, Y. Temperature, flow velocity and protein concentration near ice surfaces in mini-channels. In Proceedings of the 10th International Conference on Phase-change Materials and Slurries for Refrigeration and Air Conditioning (ISBN 978-2-913149-91-5), Kobe, Japan, 29 July–1 August 2012; pp. 203–210. [Google Scholar]
- Baardsnes, J.; Kondejewski, L.H.; Hodges, R.S.; Chao, H.; Kay, C.; Davies, P.L. New ice-binding face for type I antifreeze protein. FEBS Lett. 1999, 463, 87–91. [Google Scholar] [CrossRef]
- Davies, P.L.; Baardsnes, J.; Kuiper, M.J.; Walker, V.K. Structure and function of antifreeze proteins. Philos. Trans. R. Soc. Lond. B 2002, 357, 927–935. [Google Scholar] [CrossRef] [PubMed]
- Jorov, A.; Zhorov, B.S.; Yang, D.S.C. Theoretical study of interaction of winter flounder antifreeze protein with ice. Protein Sci. 2004, 13, 1524–1537. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, P.V. Ice Physics; Clarendon Press: Oxford, UK, 1974. [Google Scholar]





| Quiescent, Flowing | Preheated, Ultra-Filtrated | |
|---|---|---|
| Mini-channel | 1 | 2 |
| Cooling type | A | B |
| Magnification | ×10 | ×5 |
| Pixel number | 1344 × 1024 | |
| Area size (μm2) | 867 × 660 | 1234 × 940 |
| Pixel resolution (μm2) | 0.645 × 0.645 | 0.918 × 0.918 |
| Binning | 1 × 1 | |
| Depth | 12 bit | |
| Exposure time (s) | 0.03 | |
| c = 0 mg/ml | c = 0.125 mg/mL | c = 0.25 mg/mL | c = 0.50 mg/mL |
|---|---|---|---|
| 5.0 (2.7) μm/s | 2.0 (1.7) μm/s | 0.30 (0.28) μm/s | 0.18 (0.10) μm/s |
| Q = 40 μL/min | Q = 0 μL/min | ||
|---|---|---|---|
| c = 0 mg/mL | c = 0.25 mg/mL | c = 0 mg/mL | c = 0.25 mg/mL |
| 13 (8.6) μm/s | 4.1 (1.8) μm/s | 5.0 (2.5) μm/s | 0.30 (0.30) μm/s |
| c = 0.25 mg/mL | c = 0.50 mg/mL | ||
|---|---|---|---|
| Unheated | Preheated | Unheated | Preheated |
| 2.5 (1.2) μm/s | 3.8 (2.4) μm/s | 0.35 (0.40) μm/s | 0.71 (0.85) μm/s |
| Normal Vol. | Normal Vol. | Reduced Vol. | Reduced Vol. | Normal Vol. | Normal Vol. |
|---|---|---|---|---|---|
| Mw = 105 | Mw = 104 | Mw = 105 | Mw = 104 | Mw = 105 | Mw = 104 |
| Filtrate | Filtrate | Concentrate | Concentrate | Concentrate | Concentrate |
| 0.94 (0.60) μm/s | 1.25 (0.54) μm/s | 1.25 (0.59) μm/s | 1.11 (0.63) μm/s | 0.35 (0.20) μm/s | 0.25 (0.22) μm/s |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takeshita, Y.; Waku, T.; Wilson, P.W.; Hagiwara, Y. Effects of Winter Flounder Antifreeze Protein on the Growth of Ice Particles in an Ice Slurry Flow in Mini-Channels. Biomolecules 2019, 9, 70. https://doi.org/10.3390/biom9020070
Takeshita Y, Waku T, Wilson PW, Hagiwara Y. Effects of Winter Flounder Antifreeze Protein on the Growth of Ice Particles in an Ice Slurry Flow in Mini-Channels. Biomolecules. 2019; 9(2):70. https://doi.org/10.3390/biom9020070
Chicago/Turabian StyleTakeshita, Yuki, Tomonori Waku, Peter W. Wilson, and Yoshimichi Hagiwara. 2019. "Effects of Winter Flounder Antifreeze Protein on the Growth of Ice Particles in an Ice Slurry Flow in Mini-Channels" Biomolecules 9, no. 2: 70. https://doi.org/10.3390/biom9020070
APA StyleTakeshita, Y., Waku, T., Wilson, P. W., & Hagiwara, Y. (2019). Effects of Winter Flounder Antifreeze Protein on the Growth of Ice Particles in an Ice Slurry Flow in Mini-Channels. Biomolecules, 9(2), 70. https://doi.org/10.3390/biom9020070






