Characterization of the Ohmyungsamycin Biosynthetic Pathway and Generation of Derivatives with Improved Antituberculosis Activity
Abstract
1. Introduction
2. Results and Discussion
2.1. Identification of the OMS Biosynthetic Gene Cluster
2.2. Biosynthesis of the β-Hydroxy-l-Phe Moiety
2.3. Biosynthesis of the 4-Methoxy-l-Trp Moiety
2.4. Antituberculosis Activity
2.5. Antiproliferative Activity
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Bacterial Strains, Plasmids, and Culture Conditions
3.3. Draft Genome Sequencing and Bioinformatic Analysis
3.4. Construction of Gene Deletion Mutants and Complementation
3.5. UPLC-qTOF-HR-MS Analysis of OMS Derivatives
3.6. Large-Scale Culture and Extraction
3.7. Purification of OMS Derivatives
3.8. Characterization of OMS Derivatives
3.9. Minimum Inhibitory Concentration (MIC) Determination by REMA
3.10. Sulforhodamine B (SRB) Assay for the Determination of Proliferation Inhibition of Cancer Cells
4. Conclusions
5. Patents
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Um, S.; Choi, T.J.; Kim, H.; Kim, B.Y.; Kim, S.-H.; Lee, S.K.; Oh, K.-B.; Shin, J.; Oh, D.-C. Ohmyungsamycins A and B: Cytotoxic and antimicrobial cyclic peptides produced by Streptomyces sp. from a volcanic island. J. Org. Chem. 2013, 78, 12321–12329. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.S.; Shin, Y.-H.; Lee, H.-M.; Kim, J.K.; Choe, J.H.; Jang, J.-C.; Um, S.; Jin, H.S.; Komatsu, M.; Cha, G.-H.; et al. Ohmyungsamycins promote antimicrobial responses through autophagy activation via AMP-activated protein kinase pathway. Sci. Rep. 2017, 7, 3431. [Google Scholar] [CrossRef] [PubMed]
- Hur, J.; Jang, J.; Sim, J.; Son, W.S.; Ahn, H.-C.; Kim, T.S.; Shin, Y.-H.; Lim, C.; Lee, S.; An, H.; et al. Conformation-enabled total syntheses of ohmyungsamycins A and B and structural revision of ohmyungsamycin B. Angew. Chem. Int. Ed. 2018, 57, 3069–3073. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Suh, J.-W. Anti-tuberculosis lead molecules from natural products targeting Mycobacterium tuberculosis ClpC1. J. Ind. Microbiol. Biotechnol. 2016, 43, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Pogorevc, D.; Tang, Y.; Hoffmann, M.; Zipf, G.; Bernauer, H.S.; Popoff, A.; Steinmetz, H.; Wenzel, S.C. Biosynthesis and heterologous production of argyrins. ACS Synth. Biol. 2019, 8, 1121–1133. [Google Scholar] [CrossRef] [PubMed]
- Uhlmann, S.; Süssmuth, R.D.; Cryle, M.J. Cytochrome p450sky interacts directly with the nonribosomal peptide synthetase to generate three amino acid precursors in skyllamycin biosynthesis. ACS Chem. Biol. 2013, 8, 2586–2596. [Google Scholar] [CrossRef]
- Haslinger, K.; Brieke, C.; Uhlmann, S.; Sieverling, L.; Süssmuth, R.D.; Cryle, M.J. The structure of a transient complex of a nonribosomal peptide synthetase and a cytochrome P450 monooxygenase. Angew. Chem. Int. Ed. 2014, 53, 8518–8522. [Google Scholar] [CrossRef]
- Kokona, B.; Winesett, E.S.; von Krusenstiern, A.N.; Cryle, M.J.; Fairman, R.; Charkoudian, L.K. Probing the selectivity of β-hydroxylation reactions in non-ribosomal peptide synthesis using analytical ultracentrifugation. Anal. Biochem. 2016, 495, 42–51. [Google Scholar] [CrossRef]
- Cryle, M.J.; Meinhart, A.; Schlichting, I. Structural characterization of OxyD, a cytochrome P450 involved in β-hydroxytyrosine formation in vancomycin biosynthesis. J. Biol. Chem. 2010, 285, 24562–24574. [Google Scholar] [CrossRef]
- Zhang, C.; Kong, L.; Liu, Q.; Lei, X.; Zhu, T.; Yin, J.; Lin, B.; Deng, Z.; You, D. In vitro characterization of echinomycin biosynthesis: Formation and hydroxylation of l-tryptophanyl-S-enzyme and oxidation of (2S,3S) β-hydroxytryptophan. PLoS ONE 2013, 8, e56772. [Google Scholar] [CrossRef]
- Greule, A.; Charkoudian, L.K.; Cryle, M.J. Studying trans-acting enzymes that target carrier protein-bound amino acids during nonribosomal peptide synthesis. Methods Enzymol. 2019, 617, 113–154. [Google Scholar] [PubMed]
- Alkhalaf, L.M.; Ryan, K.S. Biosynthetic manipulation of tryptophan in bacteria: Pathways and mechanisms. Chem. Biol. 2015, 22, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Gunasekera, S.P.; Pomponi, S.A.; McCarthy, P.J. Discobahamins A and B, new peptides from the Bahamian deep water marine sponge Discodermia sp. J. Nat. Prod. 1994, 57, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M.; Wakimoto, T.; Egami, Y.; Tan, K.C.; Ise, Y.; Abe, I. Calyxamides A and B, cytotoxic cyclic peptides from the marine sponge Discodermia calyx. J. Nat. Prod. 2012, 75, 290–294. [Google Scholar] [CrossRef]
- Smith, D.R.M.; Uria, A.R.; Helfrich, E.J.N.; Milbredt, D.; van Pée, K.H.; Piel, J.; Goss, R.J.M. An unusual flavin-dependent halogenase from the metagenome of the marine sponge Theonella swinhoei WA. ACS Chem. Biol. 2017, 12, 1281–1287. [Google Scholar] [CrossRef]
- Fusetani, N.; Sugawara, T.; Matsunaga, S.; Hirota, H. Orbiculamide A: A novel cytotoxic cyclic peptide from a marine sponge Theonella sp. J. Am. Chem. Soc. 1991, 113, 7811–7812. [Google Scholar] [CrossRef]
- Cheng, H.-H.; Kuo, C.-C.; Yan, J.-L.; Chen, H.-L.; Lin, W.-C.; Wang, K.-H.; Tsai, K.K.-C.; Guvén, H.; Flaberg, E.; Szekely, L.; et al. Control of cyclooxygenase-2 expression and tumorigenesis by endogenous 5-methoxytryptophan. Proc. Natl. Acad. Sci. USA 2012, 109, 13231–13236. [Google Scholar] [CrossRef]
- Bülow, L.; Nickeleit, I.; Girbig, A.-K.; Brodmann, T.; Rentsch, A.; Eggert, U.; Sasse, F.; Steinmetz, H.; Frank, R.; Carlomagno, T.; et al. Synthesis and biological characterization of argyrin F. ChemMedChem 2010, 5, 832–836. [Google Scholar] [CrossRef]
- Chen, C.-H.; Genapathy, S.; Fischer, P.M.; Chan, W.C. A facile approach to tryptophan derivatives for the total synthesis of argyrin analogues. Org. Biomol. Chem. 2014, 12, 9764–9768. [Google Scholar] [CrossRef]
- Blin, K.; Shaw, S.; Steinke, K.; Villebro, R.; Ziemert, N.; Lee, S.Y.; Medema, M.H.; Weber, T. antiSMASH 5.0: Updates to the secondary metabolite genome mining pipeline. Nucleic Acids Res. 2019, 47, 81–87. [Google Scholar] [CrossRef]
- Stachelhaus, T.; Mootz, H.D.; Marahiel, M.A. The specificity-conferring code of adenylation domains in nonribosomal peptide synthetases. Chem. Biol. 1999, 6, 493–505. [Google Scholar] [CrossRef]
- Challis, G.L.; Ravel, J.; Townsend, C.A. Predictive, structure-based model of amino acid recognition by nonribosomal peptide synthetase adenylation domains. Chem. Biol. 2000, 7, 211–224. [Google Scholar] [CrossRef]
- Miao, V.; Coëffet-Legal, M.-F.; Brian, P.; Brost, R.; Penn, J.; Whiting, A.; Martin, S.; Ford, R.; Parr, I.; Bouchard, M.; et al. Daptomycin biosynthesis in Streptomyces roseosporus: Cloning and analysis of the gene cluster and revision of peptide stereochemistry. Microbiology 2005, 151, 1507–1523. [Google Scholar] [CrossRef] [PubMed]
- Seipke, R.F.; Hutchings, M.I. The regulation and biosynthesis of antimycins. Beilstein J. Org. Chem. 2013, 9, 2556–2563. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Khullar, A.; Chou, S.; Sacramo, A.; Gerratana, B. Biosynthesis of sibiromycin, a potent antitumor antibiotic. Appl. Environ. Microbiol. 2009, 75, 2869–2878. [Google Scholar] [CrossRef] [PubMed]
- Kieser, T.; Bibb, M.J.; Buttner, M.J.; Chater, K.F.; Hoopwood, D.A. Practical Streptomyces Genetics; The John Innes Foundation: Norwich, UK, 2000. [Google Scholar]
- Bierman, M.; Logan, R.; O’Brien, K.; Seno, E.T.; Rao, R.N.; Schoner, B.E. Plasmid cloning vectors for the conjugal transfer of DNA from Escherichia coli to Streptomyces spp. Gene 1992, 116, 43–49. [Google Scholar] [CrossRef]
- Bachmann, B.O.; Ravel, J. Chapter 8. Methods for in silico prediction of microbial polyketide and nonribosomal peptide biosynthetic pathways from DNA sequence data. Methods Enzymol. 2009, 458, 181–217. [Google Scholar]
- Jang, J.; Kim, R.; Woo, M.; Jeong, J.; Park, D.E.; Kim, G.; Delorme, V. Efflux attenuates the antibacterial activity of Q203 in Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2017, 61, e02637-16. [Google Scholar] [CrossRef]
- Vichai, V.; Kirtikara, K. Sulforhodamine B colorimetric assay for cytotoxicity screening. Nat. Protoc. 2006, 1, 1112–1116. [Google Scholar] [CrossRef]
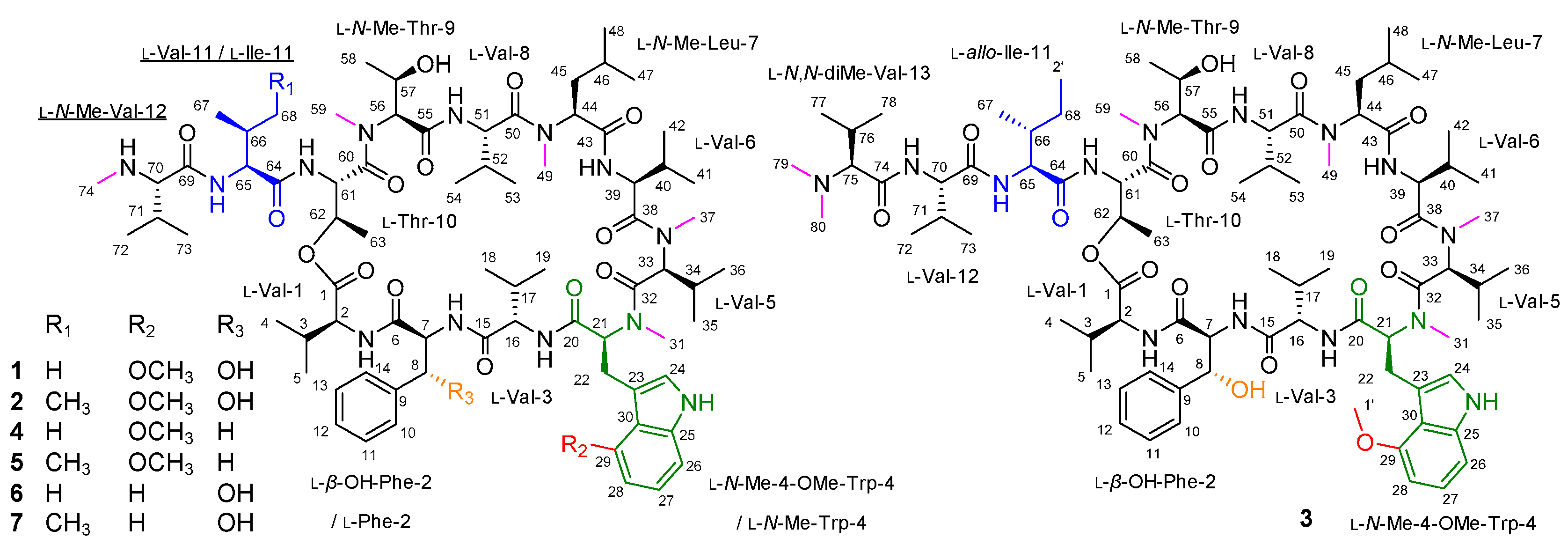
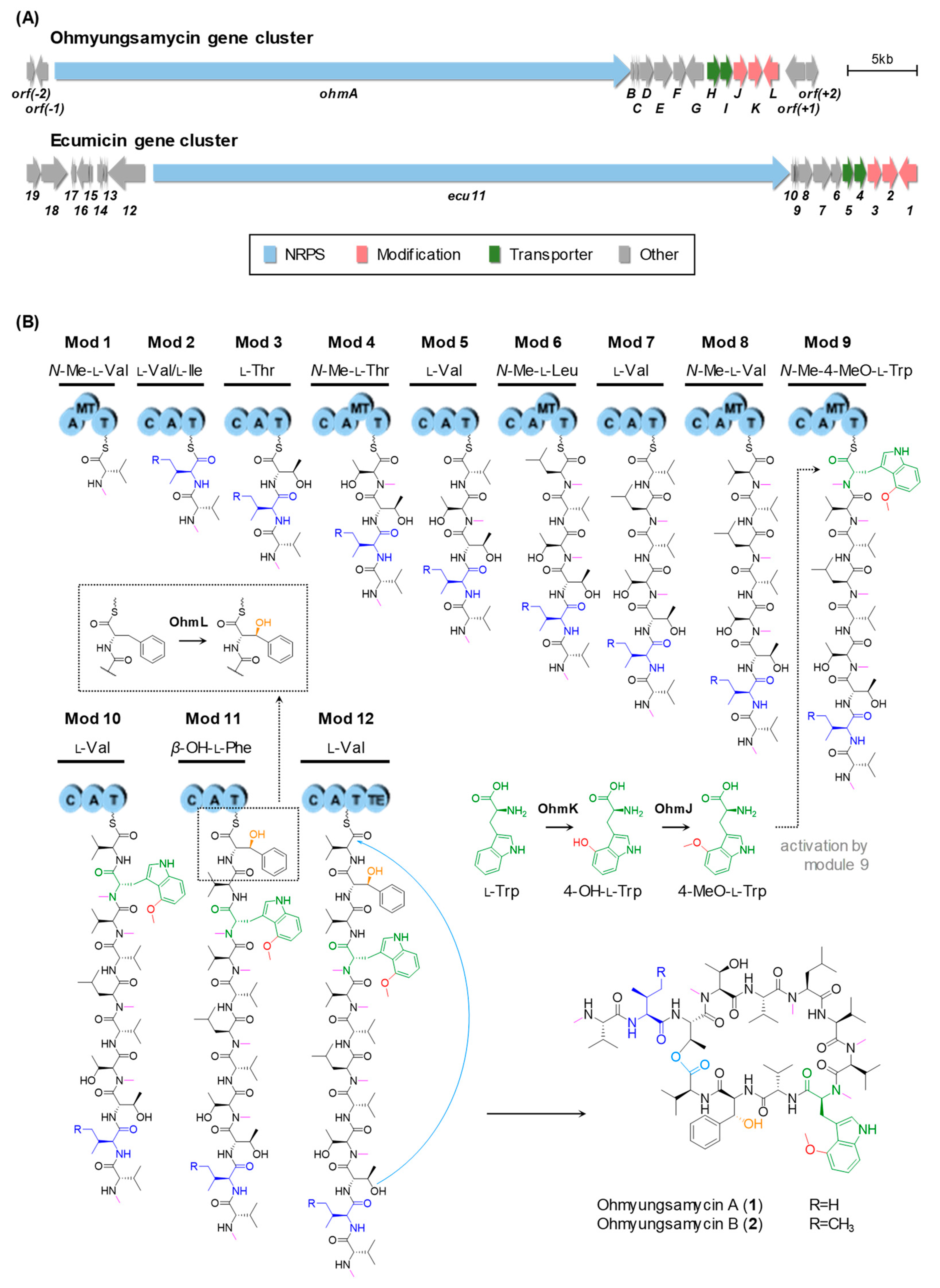
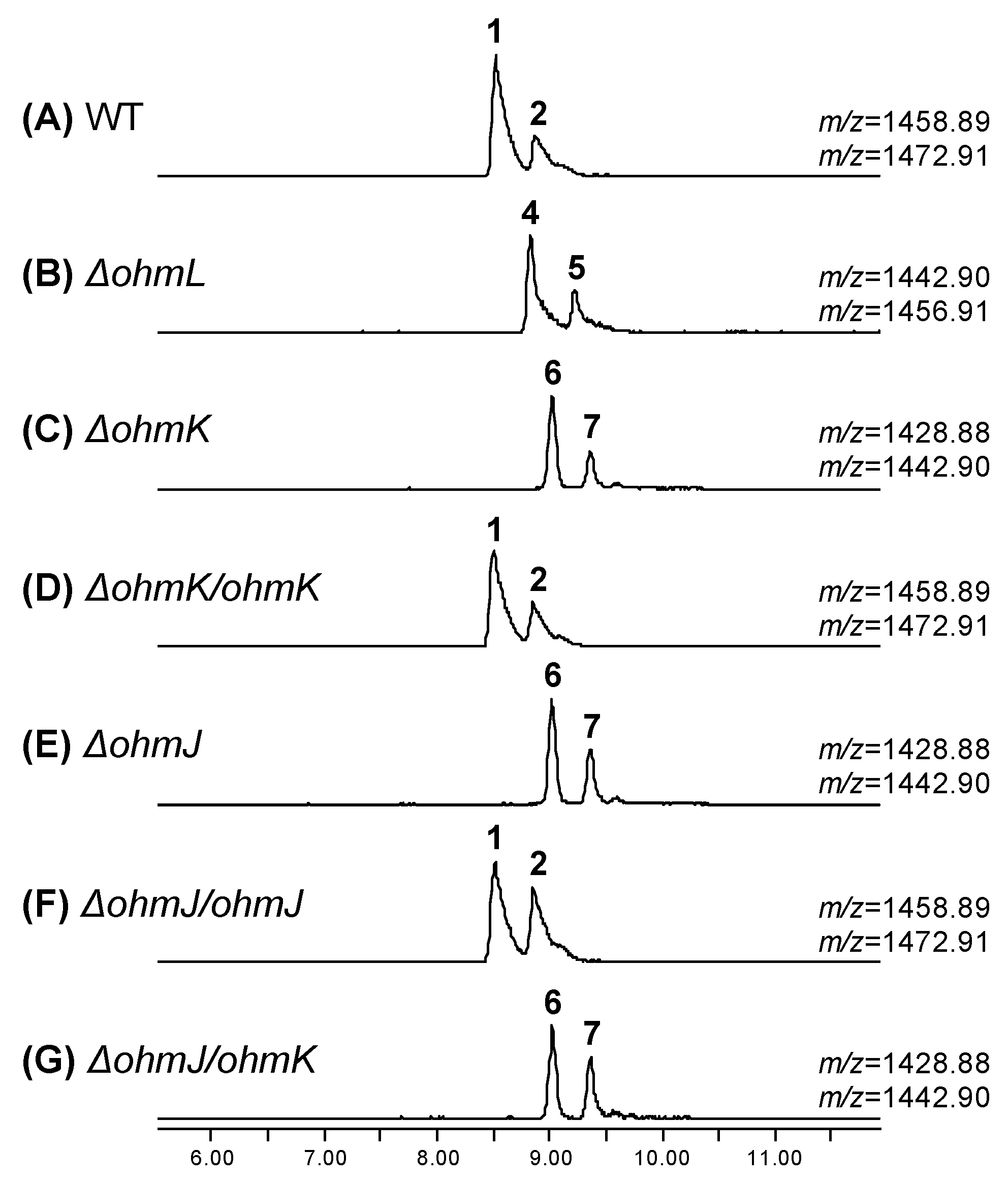
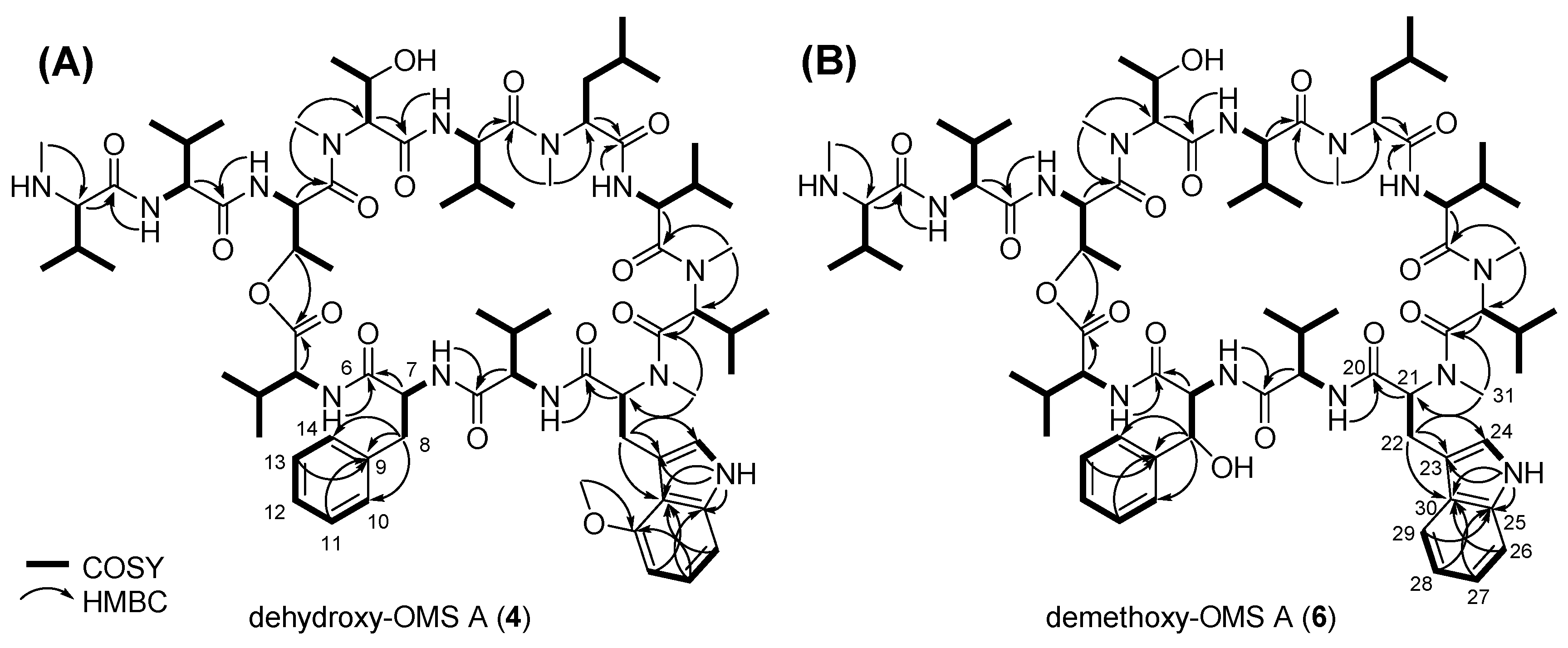
| Position | δCa, Type | δHb, Mult (J in Hz) | Position | δCa, Type | δHb, Mult (J in Hz) | ||
|---|---|---|---|---|---|---|---|
| Val-1 | 1 | 174.6, C | Val-6 | 38 | 173.0, C | ||
| 2 | 58.1, CH | 4.72, dd (9.0, 7.5) | 39 | 54.4, CH | 4.96, dd (9.5, 9.5) | ||
| 2-NH | 9.17, d (9.0) | 39-NH | 9.46, d (9.5) | ||||
| 3 | 33.0, CH | 2.22, m | 40 | 31.8, CH | 2.42, m | ||
| 4 | 18.9, CH3 | 1.13, mc | 41 | 19.7, CH3 | 1.14, d (7.0) | ||
| 5 | 18.8, CH3 | 0.99, d (7.0) | 42 | 18.9, CH3 | 1.03, d (7.0) | ||
| Phe-2 | 6 | 172.8, C | N-Me-Leu-7 | 43 | 171.7, C | ||
| 7 | 56.8, CH | 5.49, ddd (9.5, 8.0, 4.5) | 44 | 54.7, CH | 5.68, dd (7.5, 7.5) | ||
| 7-NH | 10.40, d (8.0) | 45a | 39.0, CH2 | 1.75, m | |||
| 8a | 38.2, CH2 | 3.80, dd (14.0,4.0) | 45b | 1.58, m | |||
| 8b | 3.28, dd (14.0, 9.5) | 46 | 25.1, CH | 1.38, m | |||
| 9 | 139.3, C | 47 | 23.1, CH3 | 0.69, d (6.5) | |||
| 10 | 129.8, CH | 7.34, d (7.5) | 48 | 21.9, CH3 | 0.66, d (6.5) | ||
| 11 | 128.9, CH | 7.40, dd (7.5, 7.0) | 49 | 31.3, CH3 | 3.53, s | ||
| 12 | 126.7, CH | 7.24, dd (7.0, 7.0) | Val-8 | 50 | 173.6, C | ||
| 13 | 128.9, CH | 7.40, dd (7.5, 7.0) | 51 | 55.7, CH | 5.28, dd (8.5, 8.5) | ||
| 14 | 129.8, CH | 7.34, d (7.5) | 51-NH | 8.08, d (8.5) | |||
| Val-3 | 15 | 174.2, C | 52 | 31.1, CH | 2.58, m | ||
| 16 | 58.6, CH | 5.15, dd (9.5, 9.5) | 53 | 19.9, CH3 | 1.16, d (7.0) | ||
| 16-NH | 8.04, d (9.5) | 54 | 19.2, CH3 | 1.20, d (6.5) | |||
| 17 | 32.8, CH | 2.66, m | N-Me-Thr-9 | 55 | 170.6, C | ||
| 18 | 20.2, CH3 | 1.11, d (7.0) | 56 | 62.4, CH | 5.57, d (3.5) | ||
| 19 | 19.9, CH3 | 1.31, d (7.0) | 57 | 66.6, CH | 5.07, qd (6.5, 3.5) | ||
| N-Me-4-OMe-Trp-4 | 20 | 169.8, C | 58 | 20.5, CH3 | 1.35, d (6.5) | ||
| 21 | 70.7, CH | 4.60, dd (11.0, 4.5) | 59 | 34.8, CH3 | 3.71, s | ||
| 22a | 27.2, CH2 | 4.45, dd (13.5, 4.5) | Thr-10 | 60 | 171.3, C | ||
| 22b | 4.33, dd (13.5, 11.0) | 61 | 52.6, CH | 5.94, dd (9.0, 2.5) | |||
| 23 | 112.9, C | 61-NH | 10.31, d (9.0) | ||||
| 24 | 124.3, CH | 7.19, d (1.5) | 62 | 69.4, CH | 6.10, qd (6.5, 2.5) | ||
| 24-NH | 11.80, d (1.5) | 63 | 16.7, CH3 | 1.56, d (6.5) | |||
| 25 | 139.6, C | Val-11 | 64 | 172.6, C | |||
| 26 | 105.9, CH | 7.30, d (7.5) | 65 | 59.1, CH | 5.29, dd (8.5, 8.5) | ||
| 27 | 122.8, CH | 7.27, dd (7.5, 7.5) | 65-NH | 9.92, d (8.5) | |||
| 28 | 99.6, CH | 6.68, d (7.5) | 66 | 31.5, CH | 2.27, m | ||
| 29 | 154.9, C | 67 | 19.5, CH3 | 0.94, d (7.0) | |||
| 29-OMe | 55.5, CH3 | 3.85, s | 68 | 18.8, CH3 | 0.93, d (6.5) | ||
| 30 | 118.5, C | N-Me-Val-12 | 69 | 168.1, C | |||
| 31 | 40.9, CH3 | 2.51, s | 70 | 67.9, CH | 4.33, d (6.0) | ||
| N-Me-Val-5 | 32 | 169.5, C | 71 | 30.9, CH | 2.64, m | ||
| 33 | 71.0, CH | 3.21, d (8.5) | 72 | 19.3, CH3 | 1.25, d (7.0) | ||
| 34 | 29.0, CH | 3.03, m | 73 | 18.5, CH3 | 1.19, d (7.0) | ||
| 35 | 21.9, CH3 | 1.22, d (6.5) | 74 | 32.4, CH3 | 2.99, s | ||
| 36 | 19.7, CH3 | 1.02, d (6.5) | |||||
| 37 | 40.0, CH3 | 3.20, s | |||||
| Position | δCa, Type | δHb, Mult (J in Hz) | Position | δCa, Type | δHb, Mult (J in Hz) | ||
|---|---|---|---|---|---|---|---|
| Val-1 | 1 | 174.3, C | Val-6 | 38 | 173.1, C | ||
| 2 | 58.4, CH | 4.65, dd (8.5, 8.0) | 39 | 54.4, CH | 4.96, dd (9.0, 8.5) | ||
| 2-NH | 9.26, d (8.5) | 39-NH | 9.36, d (8.5) | ||||
| 3 | 33.0, CH | 2.22, m | 40 | 32.0, CH | 2.39, mc | ||
| 4 | 19.0, CH3 | 0.92, mc | 41 | 19.3, CH3 | 1.12, mc | ||
| 5 | 18.9, CH3 | 1.12, mc | 42 | 19.3, CH3 | 1.02, mc | ||
| β-OH-Phe-2 | 6 | 172.7, C | N-Me-Leu-7 | 43 | 171.6, C | ||
| 7 | 60.1, CH | 5.50, mc | 44 | 54.8, CH | 5.61, mc | ||
| 7-NH | 9.73, br s | 45a | 38.9, CH2 | 1.73, mc | |||
| 8 | 73.3, CH | 5.92, mc | 45b | 1.50, mc | |||
| 9 | 143.0, C | 46 | 25.1, CH | 1.36, mc | |||
| 10 | 127.0, CH | 7.65, mc | 47 | 23.3, CH3 | 0.63, d (6.0) | ||
| 11 | 128.6, CH | 7.38, mc | 48 | 22.0, CH3 | 0.59, d (6.0) | ||
| 12 | 127.6, CH | 7.24, mc | 49 | 31.4, CH3 | 3.49, s | ||
| 13 | 128.6, CH | 7.38, mc | Val-8 | 50 | 173.5, C | ||
| 14 | 127.0, CH | 7.65, mc | 51 | 55.7, CH | 5.27, dd (8.5, 8.0) | ||
| Val-3 | 15 | 174.4, C | 51-NH | 8.08, d (8.5) | |||
| 16 | 58.5, CH | 5.43, dd (9.5, 9.0) | 52 | 31.2, CH | 2.55, mc | ||
| 16-NH | 8.15, d (9.5) | 53 | 19.3, CH3 | 1.19, mc | |||
| 17 | 33.1, CH | 2.68, m | 54 | 19.3, CH3 | 1.14, mc | ||
| 18 | 20.2, CH3 | 1.22, mc | N-Me-Thr-9 | 55 | 170.6, C | ||
| 19 | 19.8, CH3 | 1.35, d (6.0) | 56 | 62.5, CH | 5.60, d (3.5) | ||
| N-Me-Trp-4 | 20 | 169.4, C | 57 | 66.5, CH | 5.06, m | ||
| 21 | 70.7, CH | 4.21, d (10.5, 3.0) | 58 | 20.5, CH3 | 1.32, d (6.0) | ||
| 22a | 25.1, CH2 | 4.40, mc | 59 | 34.7, CH3 | 3.71, s | ||
| 22b | 4.30, dd (15.0, 3.0) | Thr-10 | 60 | 171.4, C | |||
| 23 | 113.1, C | 61 | 52.6, CH | 5.92, m | |||
| 24 | 124.5, CH | 7.35, mc | 61-NH | 10.29, d (8.5) | |||
| 24-NH | 11.87, br s | 62 | 69.8, CH | 6.09, m | |||
| 25 | 137.8, C | 63 | 16.8, CH3 | 1.56, d (6.5) | |||
| 26 | 127.0, CH | 7.65, d (7.5) | Val-11 | 64 | 172.6, C | ||
| 27 | 128.7, CH | 7.38, mc | 65 | 59.2, CH | 5.23, m | ||
| 28 | 119.0, CH | 7.24, mc | 65-NH | 9.96, d (8.5) | |||
| 29 | 119.4, CH | 7.86, d (7.5) | 66 | 31.4, CH | 2.25, m | ||
| 30 | 128.5, C | 67 | 19.0, CH3 | 0.92, mc | |||
| 31 | 41.0, CH3 | 2.46, s | 68 | 19.0, CH3 | 0.92, mc | ||
| N-Me-Val-5 | 32 | 169.5, C | N-Me-Val-12 | 69 | 168.0, C | ||
| 33 | 71.1, CH | 3.28, d (7.0) | 70 | 67.8, CH | 4.37, mc | ||
| 34 | 29.3, CH | 3.06, m | 71 | 30.9, CH | 2.65, mc | ||
| 35 | 22.0, CH3 | 1.25, mc | 72 | 19.3, CH3 | |||
| 36 | 19.7, CH3 | 1.05, mc | 73 | 18.5, CH3 | 1.20, mc | ||
| 37 | 40.0, CH3 | 3.19, s | 74 | 32.3, CH3 | 2.99, s |
| MIC50 (nM) | IC50 (μM) | ||||||
|---|---|---|---|---|---|---|---|
| Cell Lines/Strain | Mycobacterium Tuberculosis mc2 6230 | A549 | HCT 116 | MDA-MB-231 | SK-HEP-1 | SNU 638 | MRC-5 |
| OMS A (1) | 29.3 | 11.26 | 10.91 | 14.90 | 14.41 | 13.27 | >50 |
| Dehydroxy-OMS A (4) | 4.9 | 29.87 | 25.10 | 27.26 | 33.26 | 29.38 | >50 |
| Demethoxy-OMS A (6) | 40.2 | 9.56 | 7.68 | 9.52 | 10.11 | 11.58 | >50 |
| Etoposide | - | 0.42 | 1.23 | 7.79 | 1.11 | 1.24 | 14.68 |
| Isoniazid (INH) | 142.9 | - | - | - | - | - | - |
| Ethambutol (EMB) | 2700 | - | - | - | - | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, E.; Shin, Y.-H.; Kim, T.H.; Byun, W.S.; Cui, J.; Du, Y.E.; Lim, H.-J.; Song, M.C.; Kwon, A.S.; Kang, S.H.; et al. Characterization of the Ohmyungsamycin Biosynthetic Pathway and Generation of Derivatives with Improved Antituberculosis Activity. Biomolecules 2019, 9, 672. https://doi.org/10.3390/biom9110672
Kim E, Shin Y-H, Kim TH, Byun WS, Cui J, Du YE, Lim H-J, Song MC, Kwon AS, Kang SH, et al. Characterization of the Ohmyungsamycin Biosynthetic Pathway and Generation of Derivatives with Improved Antituberculosis Activity. Biomolecules. 2019; 9(11):672. https://doi.org/10.3390/biom9110672
Chicago/Turabian StyleKim, Eunji, Yern-Hyerk Shin, Tae Ho Kim, Woong Sub Byun, Jinsheng Cui, Young Eun Du, Hyung-Ju Lim, Myoung Chong Song, An Sung Kwon, Sang Hyeon Kang, and et al. 2019. "Characterization of the Ohmyungsamycin Biosynthetic Pathway and Generation of Derivatives with Improved Antituberculosis Activity" Biomolecules 9, no. 11: 672. https://doi.org/10.3390/biom9110672
APA StyleKim, E., Shin, Y.-H., Kim, T. H., Byun, W. S., Cui, J., Du, Y. E., Lim, H.-J., Song, M. C., Kwon, A. S., Kang, S. H., Shin, J., Lee, S. K., Jang, J., Oh, D.-C., & Yoon, Y. J. (2019). Characterization of the Ohmyungsamycin Biosynthetic Pathway and Generation of Derivatives with Improved Antituberculosis Activity. Biomolecules, 9(11), 672. https://doi.org/10.3390/biom9110672






