Mechanical Tillage Diversely Affects Glomalin Content, Water Stable Aggregates and AM Fungal Community in the Soil Profiles of Two Differently Managed Olive Orchards
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Olive Grove
- Under the dripper, in the wet area during the irrigation season, (2 m from the trunk in the row) (SSr);
- Inter-rows (4 m from the trunk between the rows) where crop residues and pruning material were left on the ground as mulch (SSi).
- Conventional system:
- 2 m from the trunk of olive trees in the row (CSr);
- Inter-rows (4 m from the trunk between the rows) (CSi).
2.2. Soil Analysis
2.3. Glomalin and Water Stable Aggregates
2.4. Spore Isolation and Molecular Characterization of Autochthonous AMF
3. Results
3.1. Soil Analysis
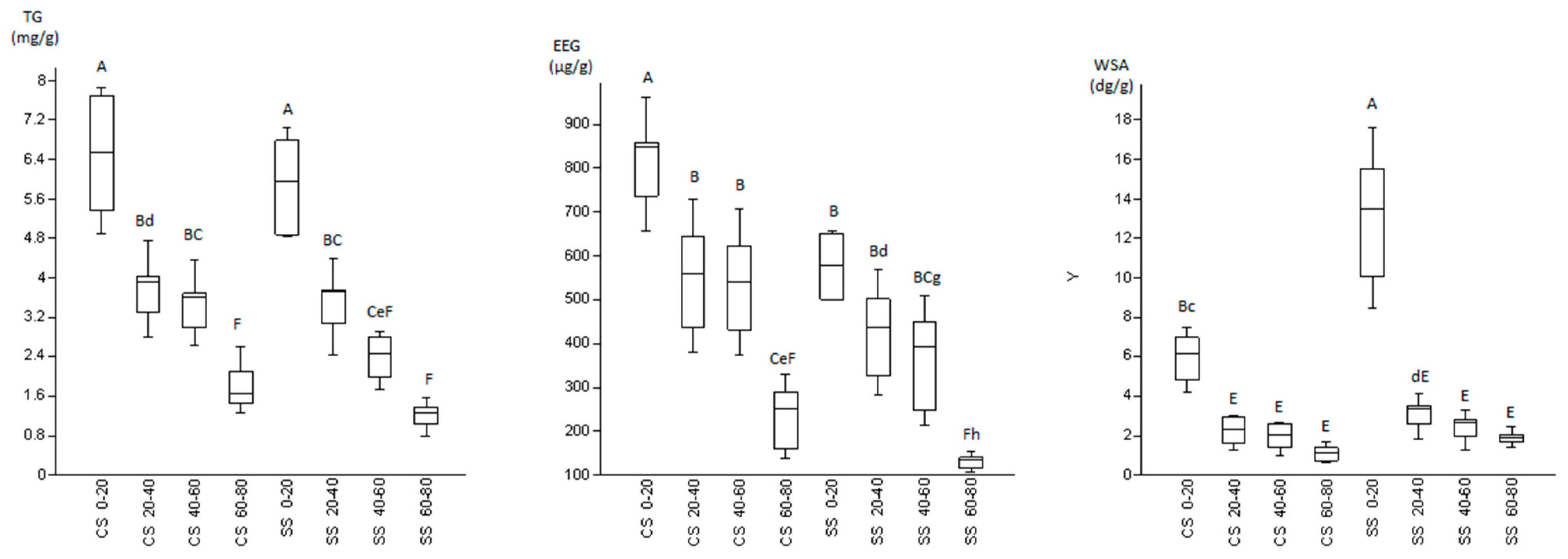
3.2. Glomalin and Water Stable Aggregates
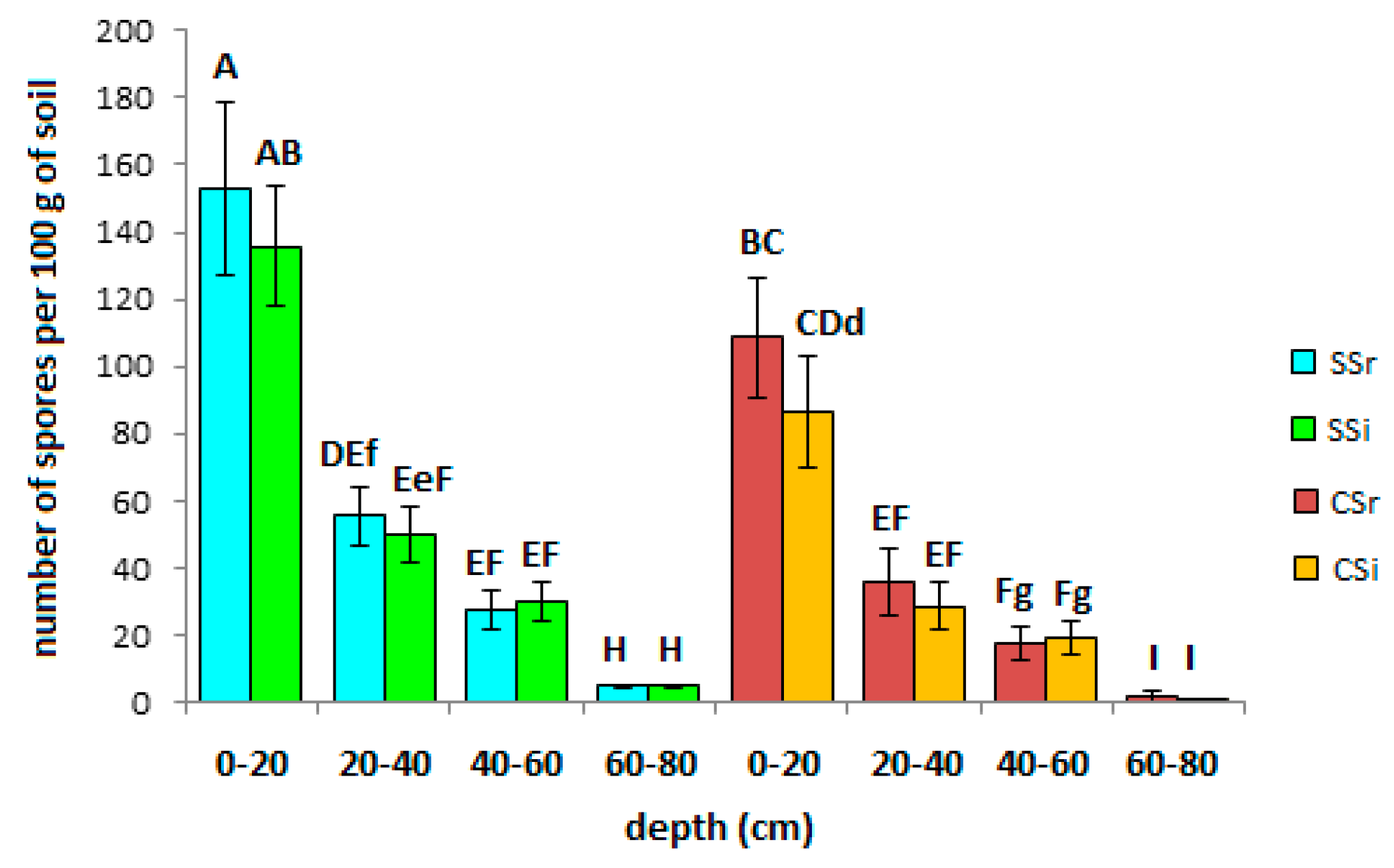
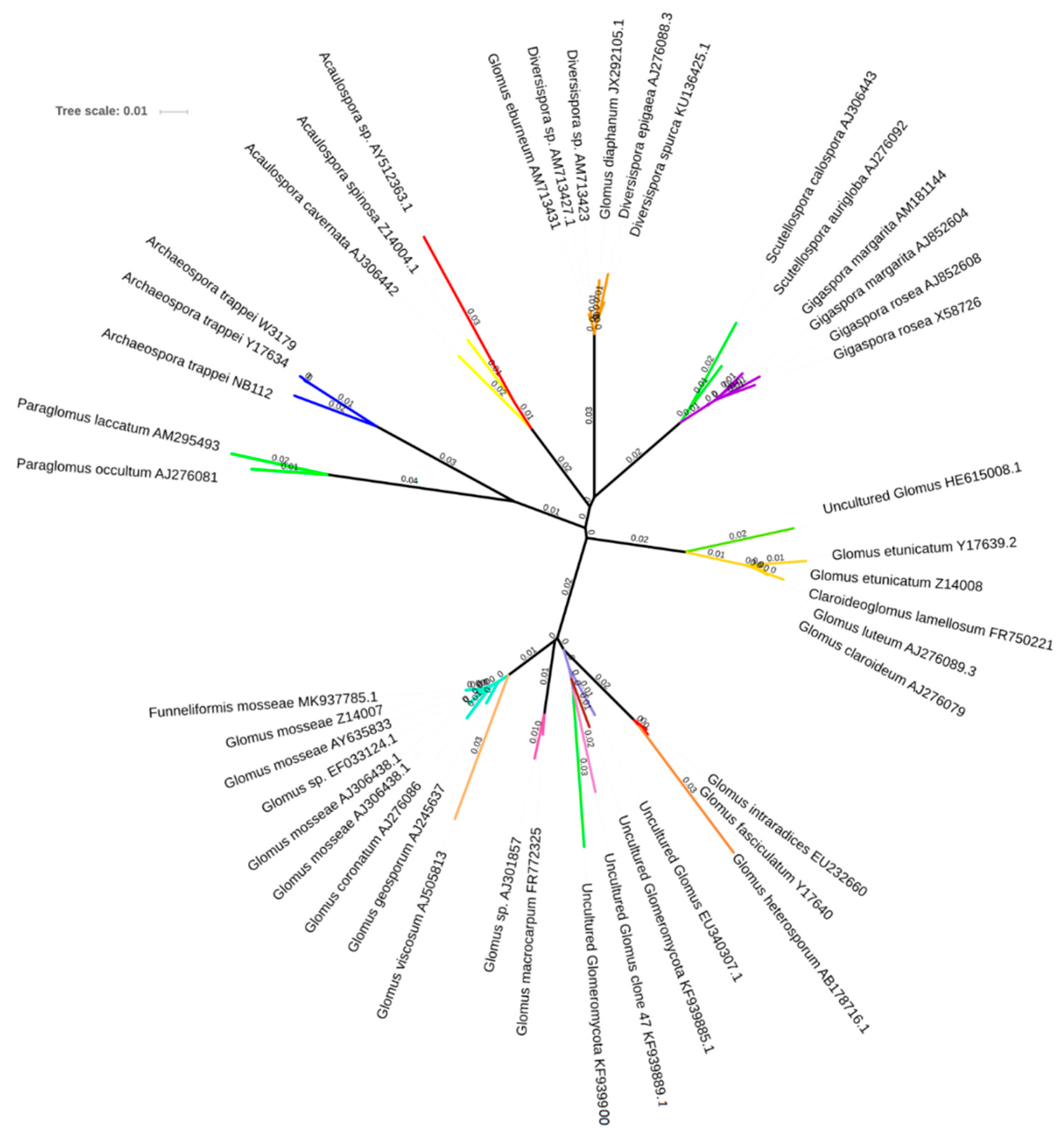
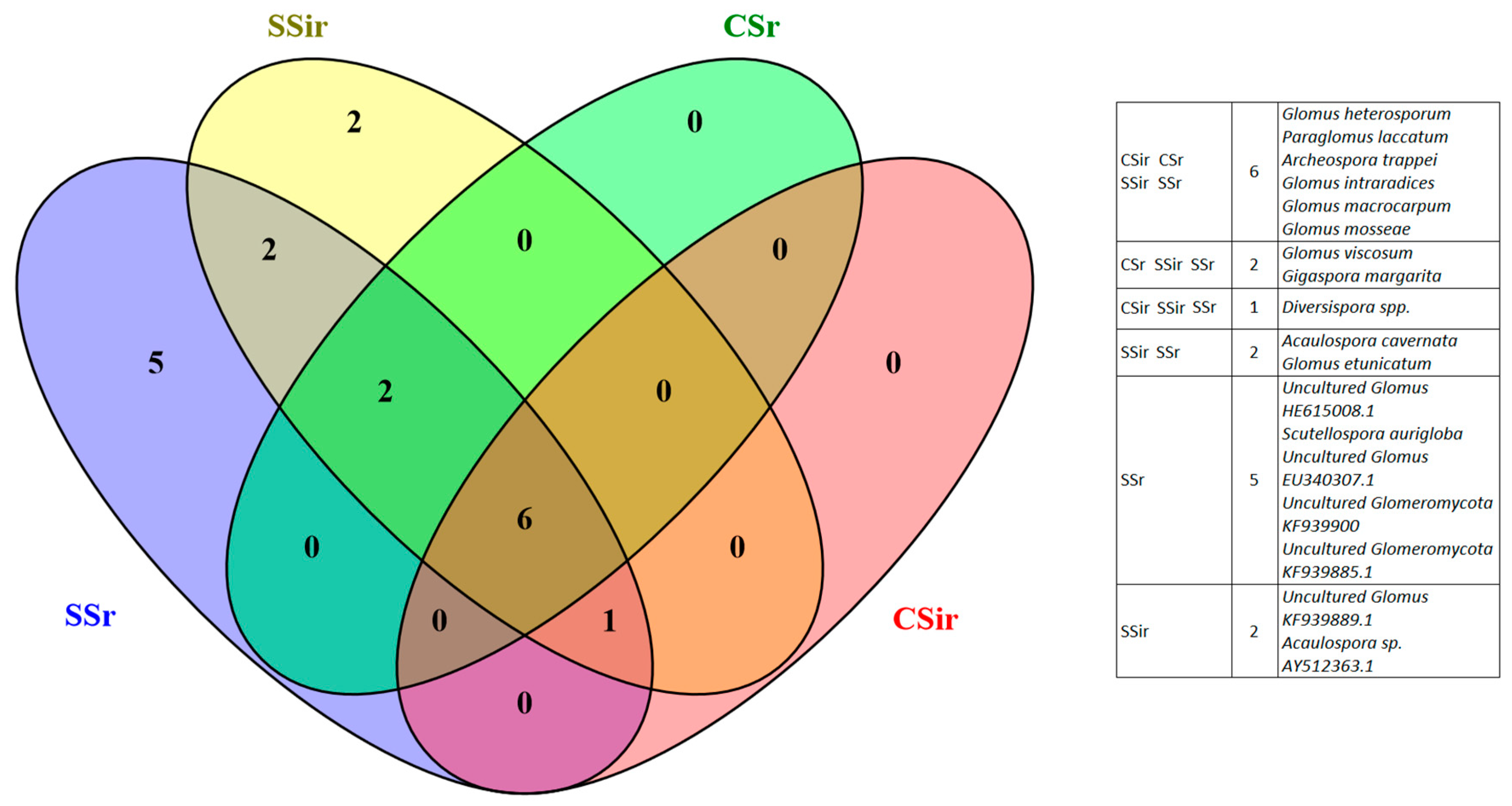
3.3. AMF Spore Abundance, Distribution and Identification
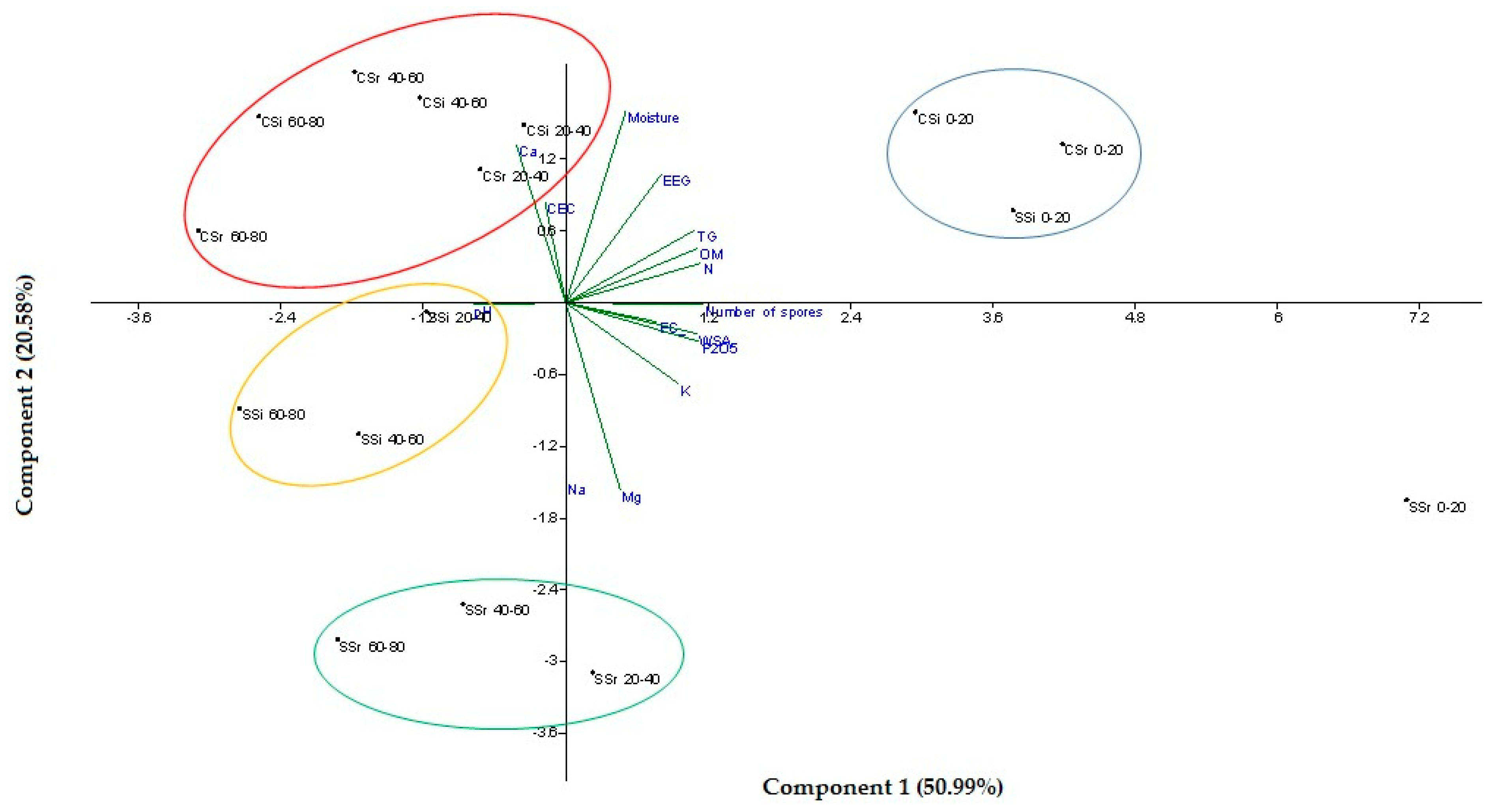
3.4. Principal Component Analysis
4. Discussions
4.1. Soil Analysis
4.2. Glomalin and Water Stable Aggregates
4.3. AMF Spore Abundance, Distribution, and Identification
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Driver, J.D.; Holben, W.E.; Rillig, M.C. Characterization of glomalin as a hyphal wall component of arbuscular mycorrhizal fungi. Soil Boil. Biochem. 2005, 37, 101–106. [Google Scholar] [CrossRef]
- Gispert, M.; Pardini, G.; Emran, M.; Doni, S.; Masciandaro, G. Seasonal evolution of soil organic matter, glomalin and enzymes and potential for C storage after land abandonment and renaturalization processes in soils of NE Spain. Catena 2018, 162, 402–413. [Google Scholar] [CrossRef]
- Rillig, M.C.; Wright, S.F.; Kimball, B.A.; Pinter, P.J.; Wall, G.W.; Ottman, M.J.; Leavitt, S.W. Elevated carbon dioxide and irrigation effects on water stable aggregates in a sorghum field: A possible role for arbuscular mycorrhizal fungi. Glob. Chang. Biol. 2001, 7, 333–337. [Google Scholar] [CrossRef]
- Rillig, M.C.; Steinberg, P.D. Glomalin production by an arbuscular mycorrhizal fungus: A mechanism of habitat modification? Soil Biol. Biochem. 2002, 34, 1371–1374. [Google Scholar] [CrossRef]
- Harner, M.J.; Ramsey, P.W.; Rillig, M.C. Protein accumulation and distribution in floodplain soils and river foam. Ecol. Lett. 2007, 7, 829–836. [Google Scholar] [CrossRef]
- Wright, S.; Upadhyaya, A. A survey of soils for aggregate stability and glomalin, a glycoprotein produced by hyphae of arbuscular mycorrhizal fungi. Plant Soil 1998, 198, 97–107. [Google Scholar] [CrossRef]
- Miller, R.M.; Jastrow, J.D. Mycorrhizal Fungi Influence Soil Structure. In Arbuscular Mycorrhizas: Physiology and Function; Kapulnik, Y., Douds, D.D., Jr., Eds.; Kluwer Academic Publishers: Amsterdam, The Netherlands, 2000; pp. 3–18. [Google Scholar]
- Rillig, M.C. Arbuscular mycorrhizae, glomalin, and soil aggregation. Can. J. Soil Sci. 2004, 84, 355–363. [Google Scholar] [CrossRef]
- Fokom, R.; Adamou, S.; Teugwa, M.C.; Boyogueno, A.B.; Nana, W.L.; Ngonkeu, M.E.L.; Tchameni, N.S.; Nwaga, D.; Tsala Ndzomod, G.; Amvam Zollo, P.A. Glomalin related soil protein, 21carbon, nitrogen,and soil aggregate stability as affected by land use variation in the humid forest zone of 22south Cameron. Soil Tillage Res. 2012, 120, 69–75. [Google Scholar] [CrossRef]
- Kobierski, M.; Kondratowicz-Maciejewska, K.; Banach-Szott, M.; Wojewódzki, P.; Castejón, J.M.P. Humic substances and aggregate stability in rhizospheric and non-rhizospheric soil. J. Soils Sediments 2018, 18, 2777–2789. [Google Scholar] [CrossRef]
- Wang, Q.; Lu, H.; Chen, J.; Hong, H.; Liu, J.; Li, J.; Yan, C. Spatial distribution of glomalin-related soil protein and its relationship with sediment carbon sequestration across a mangrove forest. Sci. Total Environ. 2018, 613, 548–556. [Google Scholar] [CrossRef]
- Bedini, S.; Pellegrino, E.; Avio, L.; Pellegrini, S.; Bazzoffi, P.; Argese, E.; Giovannetti, M. Changes in soil aggregation and glomalin-related soil protein content as affected by the arbuscular mycorrhizal fungal species Glomus mosseae and Glomus intraradices. Soil Biol. Biochem. 2009, 41, 1491–1496. [Google Scholar] [CrossRef]
- Curaqueo, G.; Seguel, A.; Rubio, R.; Acevedo, E.; Cornejo, P.; Borie, F. Tillage effect on soil organic matter, mycorrhizal hyphae and aggregates in a mediterranean agroecosystem. Rev. Cienc. Suelo Nutr. Veg. 2010, 10, 12–21. [Google Scholar] [CrossRef]
- Sharifi, Z.; Azadi, N.; Rahimi, S.; Certini, G. The response of glomalin-related soil proteins to fire or tillage. Geoderma 2018, 329, 65–72. [Google Scholar] [CrossRef]
- Gómez, J.A.; Battany, M.; Renschler, C.S.; Fereres, E. Evaluating the impact of soil management on soil loss in olive orchards. Soil Use Manag. 2003, 19, 127–134. [Google Scholar] [CrossRef]
- Romero, P.; Castro, G.; Gomez, J.A.; Fereres, E. Curve Number Values for Olive Orchards under Different Soil Management. Soil Sci. Soc. Am. J. 2007, 71, 1758–1769. [Google Scholar] [CrossRef]
- FAO. FAO/Unesco Soil Map of the World: Revised Legend (FAO/Unesco/ISRIC); World Soil Resources Reports 60; FAO: Rome, Italy, 1988; p. 119. [Google Scholar]
- López, A.; Pollice, A.; Lonigro, A.; Masi, S.; Palese, A.; Cirelli, G.L.; Toscano, A.; Passino, R. Agricultural wastewater reuse in southern Italy. Desalination 2006, 187, 323–334. [Google Scholar] [CrossRef]
- Walkley, A.; Black, I.A. An examination of the Degtjareff method for determining organic carbon in soils: Effect of variations in digestion conditions and of inorganic soil constituents. Soil Sci. 1934, 63, 251–263. [Google Scholar] [CrossRef]
- Wolff, E. Entwurf zur bodenanalyse. Z. Anal. Chem. 1864, 3, 83–115. [Google Scholar] [CrossRef]
- Kjeldahl, J. A new method for the determination of nitrogen in organic matter. Z. Anal. Chem. 1883, 22, 366–382. [Google Scholar] [CrossRef]
- Olsen, S.R.; Cole, V.; Watenabe, F.S.; Dean, L.A. Estimation of Available Phosphorus in Soils by Extraction with Sodium Bicarbonate; USDA: Washington, DC, USA, 1954. [Google Scholar]
- Wright, S.F.; Upadhyaya, A. Extraction of an abundant and unusual protein from soil and comparison with hyphal protein of arbuscular mycorrhizal fungi. Soil Sci. 1996, 161, 575–586. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Klute, A.; Kemper, W.D.; Rosenau, R.C. Aggregate Stability and Size Distribution. In Methods of Soil Analysis Part 1; Klute, A., Ed.; Soil Science Society of America: Madison, WI, USA, 1986. [Google Scholar]
- Oades, J.M.; Waters, A.G. Aggregate hierarchy in soils. Soil Res. 1991, 29, 815–828. [Google Scholar] [CrossRef]
- White, T.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal rna genes for phylogenetics. In PCR Protocols: A Guide to Methods and Application; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Lee, J.; Lee, S.; Young, J.P.W. Improved PCR primers for the detection and identification of arbuscular mycorrhizal fungi. FEMS Microbiol. Ecol. 2008, 65, 339–349. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [PubMed]
- Letunic, I.; Bork, P. Interactive Tree of Life (iTOL): An online tool for phylogenetic tree display and annotation. Bioinformatics 2006, 23, 127–128. [Google Scholar] [CrossRef] [PubMed]
- Shannon, C.E. A Mathematical Theory of Communication. Bell Syst. Tech. J. 1948, 27, 379–423. [Google Scholar] [CrossRef]
- Calvente, R.; Cano, C.; Ferrol, N.; Azcón-Aguilar, C.; Barea, J.M. Analysing natural diversity of arbuscular mycorrhizal fungi in olive tree (Olea europaea L.) plantations and assessment of the effective-ness of native fungal isolates as inoculants for commercial cultivarsof olive plantlets. Appl. Soil Ecol. 2004, 26, 11–19. [Google Scholar] [CrossRef]
- Briccoli Bati, C.; Santilli, E.; Lombardo, L. Effect of arbuscular mycorrhizal fungi on growthand on micronutrient and macronutrient uptake and allocationin olive plantlets growing under high total Mn levels. Mycorrhiza 2015, 25, 97–108. [Google Scholar] [CrossRef]
- Cogle, A.; Rao, K.; Yule, D.; George, P.; Srinivasan, S.; Smith, G.; Jangawad, L. Soil management options for Alfisols in the semi-arid tropics: Annual and perennial crop production. Soil Tillage Res. 1997, 44, 235–253. [Google Scholar] [CrossRef]
- Kováč, K.; Macák, M.; Švančárková, M. The effect of soil conservation tillage on soil moisture dynamics under single cropping and crop rotation. Plant Soil Environ. 2005, 51, 124–130. [Google Scholar] [CrossRef]
- Palese, A.M.; Pasquale, V.; Celano, G.; Figliuolo, G.; Masi, S.; Xiloyannis, C. Irrigation of olive groves in Southern Italy with treated municipal wastewater: Effects on microbiological quality of soil and fruits. Agric. Ecosyst. Environ. 2009, 129, 43–51. [Google Scholar] [CrossRef]
- Palese, A.M.; Celano, G.; Lombardo, L.; Xiloyannis, C. Heavy metals in soil of an olive orchard drip-irrigated with reclaimed municipal wastewater. In Proceedings of the 4th International Conference “Oliviculture, Biotechnology and Quality of Olive Tree Products OLIVEBIOTEQ Chania, Crete, Greece, 31 October–4 November 2011. [Google Scholar]
- Nichols, K.A. Characterization of Glomalin-A Glycoprotein Produced by Arbuscular Mycorrhizal Fungi. Ph.D. Thesis, University of Maryland, College Park, MD, USA, 2003. [Google Scholar]
- Wright, S.F.; Anderson, R.L. Aggregate stability and glomalin in alternative crop rotations for the central Great Plains. Biol. Fertil. Soils 2000, 31, 249–253. [Google Scholar] [CrossRef]
- Rillig, C.M.; Ramsey, P.W.; Morris, S.; Paul, E.A. Glomalin and arbuscular-mycorrhizal fungal soil protein, responds to land-use change. Plant Soil 2003, 253, 293–299. [Google Scholar] [CrossRef]
- Bedini, S.; Avio, L.; Argese, E.; Giovannetti, M. Effect of long-term land use on arbuscular mycorrhizal fungi and glomalin-related soil protien. Agric. Ecosyst. Environ. 2007, 120, 463–466. [Google Scholar] [CrossRef]
- Preger, A.C.; Rillig, M.C.; Johns, A.R.; Du Preez, C.C.; Lobe, I.; Amelung, W. Losses of glomalin-related soil protein under prolonged arable cropping: A chronosequence study in sandy soils of the South African Highveld. Soil Biol. Biochem. 2007, 39, 445–453. [Google Scholar] [CrossRef]
- Lee, J.E.; Eom, A.H. Effect of Organic Farming on Spore Diversity of Arbuscular Mycorrhizal Fungi and Glomalin in Soil. Mycobiology 2009, 37, 272–276. [Google Scholar] [CrossRef]
- Carneiro, M.A.C.; Ferreira, D.A.; De Souza, E.D.; Paulino, H.B.; Júnior, O.J.S.; Siqueira, J.O. Arbuscular mycorrhizal fungi in soil aggregates from fields of “murundus” converted to agriculture. Pesqui. Agropecu. Bras. 2015, 50, 313–321. [Google Scholar] [CrossRef]
- Lovelock, C.E.; Wright, S.F.; Clark, D.A.; Ruess, R.W. Soil stocks of glomalin produced by arbuscular mycorrhizal fungi across a tropical rain forest landscape. J. Ecol. 2004, 92, 278–287. [Google Scholar] [CrossRef]
- Tisdall, J.M. Formation of soil aggregate and accumulation of soil organic matter. In Structure and Organic Matter Storage in Agricultural Soils; Carter, M.R., Stewart, B.A., Eds.; CRC Press: Boca Raton, FL, USA, 1996; pp. 57–96. [Google Scholar]
- Le Bissionas, Y. Aggregate stability and assesment of soil crustability and erodibility I. Theory and methodology. Eur. J. Soil Sci. 1996, 47, 425–437. [Google Scholar] [CrossRef]
- Six, J.; Elliott, E.T.; Paustian, K. Soil Structure and Soil Organic Matter: II. A normalized stability index and the effect of mineralogy. Soil Sci. Soc. Am. J. 2000, 64, 1042–1049. [Google Scholar] [CrossRef]
- Garcia-Franco, N.; Martinez-Mena, M.; Goberna, M.; Albaladejo, J. Changes in soil aggregation and microbial community structure control carbon sequestration after afforestation of semiarid shrublands. Soil Biol. Biochem. 2015, 87, 110–121. [Google Scholar] [CrossRef]
- Kumari, M.; Chakraborty, D.; Gathala, M.K.; Pathak, H.; Dwivedi, B.S.; Tomar, R.K.; Garg, R.N. Soil aggregation and associated organic carbon fractions as affected by tillage in a rice-wheat rotation in North India. Soil Sci. Soc. Am. J. 2011, 75, 560–567. [Google Scholar] [CrossRef]
- Aziz, I.; Mahmood, T.; Islam, K.R. Effect of long term no-till and conventional tillage practices on soil quality. Soil Tillage Res. 2013, 131, 28–35. [Google Scholar] [CrossRef]
- Bai, Z.G.; Thomas, C.; Ruiperez, G.M.; Batjes, N.H.; Paul, M.; Bünemann, K. Effects of agricultural management practices on soil quality: A review of long-term experiments for Europe and China. Agric. Ecosyst. Environ. 2018, 265, 1–7. [Google Scholar] [CrossRef]
- Kumar, C.P.S.; Garampalli, R.H. Diversity of arbuscular mycorrhizal fungi in irrigated and non-irrigated fields of southern Karnataka, India. J. Environ. Biol. 2013, 34, 159–164. [Google Scholar] [PubMed]
- Mai, W.; Xue, X.; Feng, G.; Tian, C. Simultaneously maximizing root/mycorrhizal growth and phosphorus uptake by cotton plants by optimizing water and phosphorus management. BMC Plant Biol. 2018, 18, 334. [Google Scholar] [CrossRef]
- Oehl, F.; Sieverding, E.; Dubois, D.; Ineichen, K.; Boller, T.; Wiemken, A.; Mäder, P. Impact of long-term conventional and organic farming on the diversity of arbuscular mycorrhizal fungi. Oecologia 2004, 138, 574–583. [Google Scholar] [CrossRef]
- Lumini, E.; Vallino, M.; Alguacil, M.M.; Romani, M.; Bianciotto, V. Different farming and water regimes in Italian rice fields affect arbuscular mycorrhizal fungal soil communities. Ecol. Appl. 2011, 21, 1696–1707. [Google Scholar] [CrossRef]
- Sidney Luiz Stürmer, S.L.; Siqueira, J.O. Species richness and spore abundance of arbuscular mycorrhizal fungi across distinct land uses in Western Brazilian Amazon. Mycorrhiza 2011, 21, 255–267. [Google Scholar] [CrossRef]
- Sasvári, Z.; Magurno, F.; Galanics, D.; Hang, T.T.N.; Ha, T.T.H.; Luyen, N.D.; Huong, L.M.; Posta, K. Isolation and Identification of Arbuscular Mycorrhizal Fungi from Agricultural Fields of Vietnam. Am. J. Plant Sci. 2012, 3, 1796–1801. [Google Scholar] [CrossRef]
- Estrada, B.; Beltrán-Hermoso, M.; Palenzuela, J.; Iwase, K.; Lozano, J.M.R.; Barea, J.M.; Oehl, F. Diversity of arbuscular mycorrhizal fungi in the rhizosphere of Asteriscus maritimus (L.) Less., a representative plant species in arid and saline Mediterranean ecosystems. J. Arid Environ. 2013, 97, 170–175. [Google Scholar] [CrossRef]
- Casazza, G.; Lumini, E.; Ercole, E.; Dovana, F.; Guerrina, M.; Arnulfo, A.; Minuto, L.; Fusconi, A.; Mucciarelli, M. The abundance and diversity of arbuscular mycorrhizal fungi are linked to the soil chemistry of screes and to slope in the Alpic paleo-endemic Berardia subacaulis. PLoS ONE 2017, 12, e0171866. [Google Scholar] [CrossRef] [PubMed]
- Borriello, R.; Lumini, E.; Girlanda, M.; Bonfante, P.; Bianciotto, V. Effects of different management practices on arbuscular mycorrhizal fungal diversity in maize fields by a molecular approach. Biol. Fertil. Soils 2012, 48, 911–922. [Google Scholar] [CrossRef]
- Camenzind, T.; Hempel, S.; Homeier, J.; Horn, S.; Velescu, A.; Wilcke, W.; Rillig, M. Nitrogen and phosphorus additions impact arbuscular mycorrhizal abundance and molecular diversity in a tropical montane forest. Glob. Chang. Biol. 2014, 20, 3646–3659. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Yang, X.; Guo, R.; Guo, J. Response of AM fungi spore population to elevated temperature and nitrogen addition and their influence on the plant community composition and productivity. Sci. Rep. 2016, 6, 24749. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Che, R.; Liu, X.; Tan, Y.; Yang, R.; Zhang, W.; He, X.; Xu, Z.; Wang, K. Arbuscular mycorrhizal fungi abundance was sensitive to nitrogen addition but diversity was sensitive to phosphorus addition in karst ecosystems. Biol. Fertil. Soils 2019, 55, 457–469. [Google Scholar] [CrossRef]
- Sofo, A.; Ciarfaglia, A.; Scopa, A.; Camele, I.; Curci, M.; Crecchio, C.; Xiloyannis, C.; Palese, A.M. Soil microbial diversity and activity in a Mediterranean olive orchard using sustainable agricultural practices. Soil Use Manag. 2014, 30, 160–167. [Google Scholar] [CrossRef]

| Parameter | Unit of Measure | Value |
|---|---|---|
| pH | 7.6 | |
| Conductivity | µS cm−1 | 884 |
| Na | mg L−1 | 121.3 |
| Mg | mg L−1 | 13.8 |
| Ca | mg L−1 | 67.8 |
| N (NO3-) | mg L−1 | 18.3 |
| N (NH4+) | mg L−1 | 0.0 |
| B | mg L−1 | 1.0 |
| K | mg L−1 | 17.0 |
| P | mg L−1 | 1.0 |
| COD | mg L−1 | 180 |
| Treatment | Depth (cm) | pH | EC (µS/cm) | N (%) | P (ppm) | P2O5 (ppm) | CEC (meq/100g) | Base Cations (meq/100g) | Moisture (%) | OM (%) | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ca | Mg | K | Na | ||||||||||
| CSr | 0-20 | 8.19 ± 1.10 | 140.00 ± 17.98 | 0.13 ± 0.03 | 18.71 ± 2.08 | 42.85 ± 8.44 | 14.15 ± 1.62 | 12.24 ± 2.41 | 0.55 ± 0.08 | 1.09 ± 0.14 | 0.27 ± 0.04 | 13.23 ± 3.39 | 2.61 ± 0.61 |
| 20-40 | 8.33 ± 1.08 | 117.67 ± 15.73 | 0.09 ± 0.01 | 6.77 ± 0.82 | 15.51 ± 3.19 | 12.39 ± 1.38 | 11.45 ± 2.11 | 0.38 ± 0.06 | 0.35 ± 0.06 | 0.21 ± 0.03 | 12.94 ± 2.76 | 1.75 ± 0.47 | |
| 40-60 | 8.43 ± 1.07 | 116.33 ± 15.58 | 0.08 ± 0.01 | 4.93 ± 0.62 | 11.29 ± 2.37 | 16.35 ± 1.76 | 15.18 ± 2.47 | 0.44 ± 0.08 | 0.41 ± 0.07 | 0.32 ± 0.04 | 13.09 ± 3.06 | 1.51 ± 0.48 | |
| 60-80 | 8.47 ± 1.06 | 121.33 ± 16.08 | 0.05 ± 0.01 | 3.80 ± 0.46 | 8.71 ± 1.79 | 16.10 ± 1.69 | 14.88 ± 2.43 | 0.48 ± 0.08 | 0.29 ± 0.06 | 0.35 ± 0.04 | 12.24 ± 2.89 | 1.47 ± 0.46 | |
| CSi | 0-20 | 8.55 ± 1.05 | 109.33 ± 14.87 | 0.12 ± 0.02 | 19.07 ± 2.10 | 43.67 ± 8.57 | 12.36 ± 1.43 | 11.00 ± 2.04 | 0.36 ± 0.06 | 0.78 ± 0.10 | 0.22 ± 0.03 | 13.52 ± 3.31 | 2.55 ± 0.63 |
| 20-40 | 8.29 ± 1.09 | 120.67 ± 16.03 | 0.09 ± 0.01 | 7.63 ± 0.91 | 17.48 ± 3.56 | 13.09 ± 1.45 | 12.15 ± 2.18 | 0.32 ± 0.06 | 0.37 ± 0.06 | 0.25 ± 0.03 | 12.48 ± 2.82 | 1.80 ± 0.48 | |
| 40-60 | 8.21 ± 1.10 | 126.33 ± 16.61 | 0.06 ± 0.01 | 4.43 ± 0.54 | 10.13 ± 2.09 | 14.60 ± 1.56 | 13.58 ± 2.33 | 0.39 ± 0.06 | 0.39 ± 0.06 | 0.24 ± 0.03 | 11.98 ± 2.77 | 1.63 ± 0.45 | |
| 60-80 | 8.32 ± 1.08 | 112.33 ± 15.19 | 0.07 ± 0.01 | 3.12 ± 0.42 | 7.14 ± 1.56 | 15.76 ± 1.69 | 14.68 ± 2.43 | 0.46 ± 0.07 | 0.35 ± 0.06 | 0.28 ± 0.04 | 12.77 ± 2.90 | 1.39 ± 0.45 | |
| SSr | 0-20 | 7.91 ± 1.14 | 173.00 ± 21.31 | 0.16 ± 0.04 | 24.68 ± 2.72 | 56.52 ± 11.10 | 13.15 ± 1.57 | 10.11 ± 2.02 | 1.58 ± 0.21 | 0.99 ± 0.15 | 0.47 ± 0.06 | 13.03 ± 3.42 | 2.71 ± 0.61 |
| 20-40 | 8.34 ± 1.08 | 135.00 ± 17.46 | 0.07 ± 0.03 | 10.57 ± 1.17 | 24.21 ± 4.76 | 12.60 ± 1.37 | 9.88 ± 1.95 | 1.28 ± 0.18 | 0.93 ± 0.14 | 0.51 ± 0.06 | 8.42 ± 2.31 | 1.53 ± 0.40 | |
| 40-60 | 8.46 ± 1.06 | 118.50 ± 15.80 | 0.07 ± 0.03 | 8.94 ± 1.01 | 20.47 ± 4.06 | 13.38 ± 1.45 | 10.79 ± 2.02 | 1.21 ± 0.17 | 0.85 ± 0.14 | 0.52 ± 0.06 | 9.66 ± 2.45 | 1.59 ± 0.43 | |
| 60-80 | 8.47 ± 1.06 | 115.67 ± 15.51 | 0.06 ± 0.03 | 8.49 ± 0.95 | 19.45 ± 3.84 | 13.40 ± 1.44 | 11.01 ± 2.05 | 1.15 ± 0.17 | 0.69 ± 0.12 | 0.55 ± 0.06 | 9.27 ± 2.41 | 1.41 ± 0.40 | |
| SSi | 0-20 | 8.18 ± 1.10 | 110.67 ± 15.04 | 0.12 ± 0.02 | 12.98 ± 1.49 | 29.72 ± 5.95 | 12.97 ± 1.49 | 11.02 ± 2.08 | 0.48 ± 0.07 | 1.23 ± 0.15 | 0.24 ± 0.04 | 12.78 ± 2.98 | 2.93 ± 0.60 |
| 20-40 | 8.40 ± 1.07 | 89.67 ± 12.92 | 0.06 ± 0.01 | 8.61 ± 0.96 | 19.72 ± 3.89 | 11.81 ± 1.28 | 10.65 ± 2.02 | 0.37 ± 0.06 | 0.55 ± 0.08 | 0.24 ± 0.03 | 10.42 ± 2.50 | 1.50 ± 0.42 | |
| 40-60 | 8.29 ± 1.09 | 85.33 ± 12.50 | 0.06 ± 0.01 | 8.32 ± 0.93 | 19.04 ± 3.76 | 10.46 ± 1.14 | 9.43 ± 1.91 | 0.35 ± 0.06 | 0.45 ± 0.07 | 0.23 ± 0.03 | 8.30 ± 2.15 | 1.28 ± 0.36 | |
| 60-80 | 8.39 ± 1.07 | 78.00 ± 11.75 | 0.06 ± 0.01 | 6.06 ± 0.70 | 13.88 ± 2.79 | 11.20 ± 1.22 | 10.12 ± 1.97 | 0.40 ± 0.07 | 0.43 ± 0.07 | 0.25 ± 0.03 | 10.38 ± 2.33 | 1.40 ± 0.39 | |
| Treatment | Depth (cm) | TG (mg/g) | EEG (μg/g) | WSA (dg/g) | Macro-Aggregates (%) |
|---|---|---|---|---|---|
| CSr | 0–20 | 6.53 ± 1.16 a | 757.75 ± 101.25 a,b | 6.16 ± 1.33 c | 76.33 |
| 20–40 | 3.36 ± 0.57 d,e | 438.26 ± 58.14 defg | 2.31 ± 0.66 d,e | 72.84 | |
| 40–60 | 3.11 ± 0.49 d,e,f | 432.14 ± 57.13 d,e,f,g | 2.03 ± 0.64 d,e | 71.23 | |
| 60–80 | 1.46 ± 0.19 e,f | 159.23 ± 21.36 h,i | 1.40 ± 0.29 d,e | 70.97 | |
| CSi | 0–20 | 6.37 ± 1.48 a | 849.19 ± 112.52 a | 5.57 ± 1.40 c,d | 77.38 |
| 20–40 | 4.03 ± 0.72 b,c,d | 643.50 ± 85.27 b,c | 2.13 ± 0.85 d,e | 71.66 | |
| 40–60 | 3.68 ± 0.68 cd | 623.63 ± 82.63 b,c,d | 1.82 ± 0.80 d,e | 73.08 | |
| 60–80 | 2.11 ± 0.50 d,e,f | 290.54 ± 38.43 f,g,h,i | 0.72 ± 0.06 e | 69.13 | |
| SSr | 0–20 | 5.96 ± 1.09 a,b | 579.53 ± 78.40 b,c,d | 15.55 ± 2.05 a | 78.16 |
| 20–40 | 3.75 ± 0.63 b,c,d | 325.96 ± 44.50 e,f,g,h | 3.49 ± 0.61 c,d,e | 75.51 | |
| 40–60 | 2.46 ± 0.46 d,e,f | 248.60 ± 33.54 g,h,i | 2.83 ± 0.50 d,e | 72.42 | |
| 60–80 | 1.37 ± 0.21 e,f | 123.39 ± 16.79 i | 1.88 ± 0.18 d,e | 70.88 | |
| SSi | 0–20 | 5.81 ± 0.98 a,b,c | 575.37 ± 75.73 c,d | 10.07 ± 1.60 b | 72.01 |
| 20–40 | 3.08 ± 0.64 d,e,f | 501.58 ± 65.78 c,d,e | 2.60 ± 0.79 d,e | 75.56 | |
| 40–60 | 2.26 ± 0.53 d,e,f | 450.06 ± 58.52 c,d,e,f | 1.95 ± 0.70 d,e | 70.54 | |
| 60–80 | 1.03 ± 0.24 f | 134.66 ± 17.86 h,i | 1.93 ± 0.53 d,e | 73.26 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lombardo, L.; Palese, A.M.; Grasso, F.; Duffy, D.H., III; Briccoli Bati, C.; Xiloyannis, C. Mechanical Tillage Diversely Affects Glomalin Content, Water Stable Aggregates and AM Fungal Community in the Soil Profiles of Two Differently Managed Olive Orchards. Biomolecules 2019, 9, 639. https://doi.org/10.3390/biom9100639
Lombardo L, Palese AM, Grasso F, Duffy DH III, Briccoli Bati C, Xiloyannis C. Mechanical Tillage Diversely Affects Glomalin Content, Water Stable Aggregates and AM Fungal Community in the Soil Profiles of Two Differently Managed Olive Orchards. Biomolecules. 2019; 9(10):639. https://doi.org/10.3390/biom9100639
Chicago/Turabian StyleLombardo, Luca, Assunta Maria Palese, Filomena Grasso, Donald H. Duffy, III, Caterina Briccoli Bati, and Cristos Xiloyannis. 2019. "Mechanical Tillage Diversely Affects Glomalin Content, Water Stable Aggregates and AM Fungal Community in the Soil Profiles of Two Differently Managed Olive Orchards" Biomolecules 9, no. 10: 639. https://doi.org/10.3390/biom9100639
APA StyleLombardo, L., Palese, A. M., Grasso, F., Duffy, D. H., III, Briccoli Bati, C., & Xiloyannis, C. (2019). Mechanical Tillage Diversely Affects Glomalin Content, Water Stable Aggregates and AM Fungal Community in the Soil Profiles of Two Differently Managed Olive Orchards. Biomolecules, 9(10), 639. https://doi.org/10.3390/biom9100639





