Alcoholic Liver Disease: Update on the Role of Dietary Fat
Abstract
:1. Introduction
2. Dietary Saturated and Unsaturated Fat: Role in ALD Pathogenesis
| Animal Models | Dietary Regimen ** | Significant Outcomes | References |
|---|---|---|---|
| Rats fed EtOH or control liquid diets with a Ritcher drinking tube for eight weeks. | CO + OO + SFO vs. L + SBO; 36%E | Hepatic steatosis and inflammation: CO + OO + SFO + EtOH > L + SBO + EtOH; Hepatic fibrosis: CO + OO + SFO + EtOH < L + SBO + EtOH | [20] |
| Rats fed intragastrically EtOH or control liquid diets for four weeks. | MCT vs. CO vs. FO; 25%E | Severity of liver pathology: FO + EtOH > CO + EtOH; MCT + EtOH (no pathological changes) | [15] |
| Rats fed intragastrically EtOH or control liquid diets for 10 weeks. | CO (USF diet) vs. BT + MCT (SF diet); 45%E | Liver steatosis, injury, and oxidative stress: USF + EtOH > SF + EtOH. SF protected rats from ALD in a dose-responsive fashion | [6] |
| Rats fed solid food and administered EtOH daily (IP) for six weeks | CO vs. CO + OO supplementation (5% wt/wt) | Liver injury, oxidative stress: CO + EtOH > CO + OO + EtOH | [19] |
| Rats fed at libitum EtOH or control liquid diets for eight weeks. | CO vs. CB vs. MCT; 30%E | Liver steatosis, macrophage activation, neutrophil infiltration, and hepatic endotoxin levels: CO + EtOH > CB + EtOH or MCT + EtOH; Serum endotoxin levels: CO + EtOH = CB + EtOH; CO + EtOH > MCT + EtOH | [7] |
| Mice fed intragastrically EtOH or control liquid diets for three weeks. | CO (USF diet), vs. Hydrogenated soya glyceride (12% palmitic and 85% stearic acids, SF diet); 35%E | Liver steatosis, injury, and oxidative stress: USF + EtOH > SF + EtOH | [8] |
| Mice fed at libitum EtOH or control liquid diets for four weeks. | CO vs. CB; 40%E | Liver injury and steatosis: CO + EtOH > CB + EtOH; Plasma adiponectin: CO + EtOH < CB + EtOH | [9] |
| Mice fed at libitum EtOH or control liquid diets for eight weeks. | CO (USF diet) vs. BT + MCT (SF diet); 40%E | Liver steatosis, inflammation, and injury: USF + EtOH > SF + EtOH. Intestinal inflammation, alterations in intestinal tight junctions, increased gut permeability and endotoxemia: USF + EtOH > SF + EtOH | [5,21] |
| FFAs | Lard | Beef Fat (Tallow) | Cocoa Butter | MCT Oil | Corn Oil | Palm Oil | Fish Oil a | Olive Oil | Safflower Oil | Soybean Oil |
|---|---|---|---|---|---|---|---|---|---|---|
| Saturated FFAs | ||||||||||
| C24:0 | - | - | - | - | - | 0.1 | - | 0.1 | 0.1 | 0.3 |
| C22:0 | - | - | 0.2 | - | - | 0.1 | - | 0.1 | 0.3 | 0.2 |
| C20:0 | - | 0.1 | 1.2 | - | 0.1 | 0.3 | - | 0.4 | 0.4 | 0.3 |
| C19:0 | - | 0.1 | - | - | - | - | - | - | - | |
| C18:0 | 14 | 21.6 | 36.4 | - | 2.2 | 4.4 | 2.1 | 2.6 | 2.3 | 3.9 |
| C17:0 | - | 1.5 | - | - | 0.1 | - | - | - | - | - |
| C16:0 | 26 | 25.5 | 25.1 | - | - | 43.8 | 13.0 | 12.1 | 6.1 | 10.8 |
| C15:0 | - | 1.3 | - | - | - | - | - | - | - | - |
| C14:0 | 2 | 3.3 | - | - | - | 1.1 | 11.6 | - | - | 0.1 |
| C12:0 | - | 0.1 | - | - | - | 0.4 | - | - | - | - |
| C10:0 | - | 0.1 | - | 23 (4 > 10:0) | - | 0.1 | - | - | - | - |
| C8:0 | - | - | - | 67 (6 < 8:0) | - | 0.1 | - | - | - | - |
| Unsaturated FFA (Monounsaturated FFAs) | ||||||||||
| C24:1 n9 | - | - | - | - | - | - | - | - | 0.2 | - |
| C20:1 n9 | 1 | - | - | - | - | 0.1 | - | 0.3 | 0.2 | 0.1 |
| C18:1 n9 | 44 | 38.7 | 34.1 | - | 27.5 | 39.1 | 6.7 | 72.5 | 13.4 | 23.9 |
| C18:1 n7 | - | - | - | - | - | - | 3.3 | - | - | - |
| C17:1 | - | 0.7 | - | - | - | - | - | 0.2 | - | - |
| C16:1 | 3 | 3.4 | - | - | 12.2 | 0.2 | 13.3 | 0.8 | 0.1 | 0.2 |
| C15:1 | - | 0.2 | - | - | - | - | - | - | - | |
| C14:1 | - | 0.2 | - | - | - | - | - | - | - | - |
| Unsaturated FFAs (Polyunsaturated FFAs) | ||||||||||
| C22:6 n3 | - | - | - | - | - | - | 8.2 | - | - | - |
| C22:5 n6 | - | - | - | - | - | - | 0.4 | - | - | - |
| C22:5 n3 | - | - | - | - | - | - | 2.0 | - | - | - |
| C20:5 n3 | - | - | - | - | - | - | 17.3 | - | 0.5 | - |
| C20:4 n3 | 1.9 | |||||||||
| C20:4 n6 | - | 0.4 | - | - | - | - | 0.7 | - | 0.5 | - |
| C18:3 n3 | - | 0.6 | 0.2 | - | 0.9 | 0.3 | - | 0.6 | 0.3 | 7.8 |
| C18:2 n6 | 10 | 2.2 | 2.8 | - | 57.0 | 10.2 | 1.1 | 9.4 | 76 | 52.1 |
| References | [22] | [6] | [23] | [6] | [6] | [23] | [15] | [23] | [23] | [23] |
3. Dietary PUFAs and ALD: The Bad, the Good, and the Controversy

| Fatty Acids | Food Sources | Dietary Intake |
|---|---|---|
| ω3 PUFAs | ||
| α-Linolenic Acid, 18:3 ω-3 | Vegetable oils (e.g., soybean and canola). Nuts, and seeds. | 1.4 g/d |
| Eicosapentanoic acid, 20:5 ω-3 | Fish (e.g., halibut, mackerel, herring, and salmon) and fish oils | 0.1–0.2 g/d |
| Docosahexaenoic acid, 22:6 ω-3 | ||
| ω6 PUFAs | ||
| Linoleic Acid, 18:2 ω-6 | Vegetable oils (e.g., sunflower, safflower, soybean, corn, and canola), nuts, seeds meats, and eggs | 12–17 g/d |
4. Oxidized Dietary Fat: Relevance to Liver Pathology
5. Oxidized Linoleic Acid Metabolites: Implication for ALD
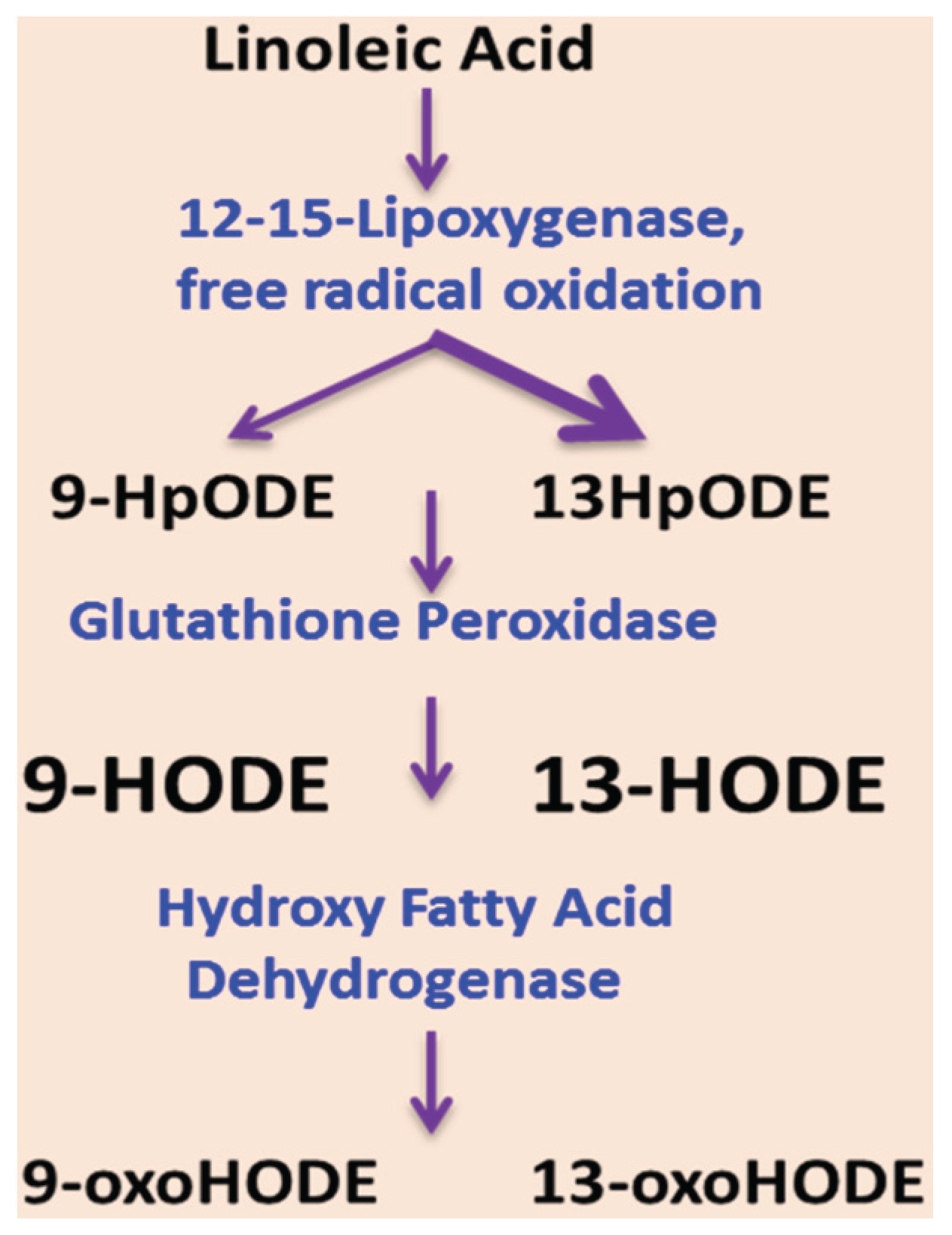
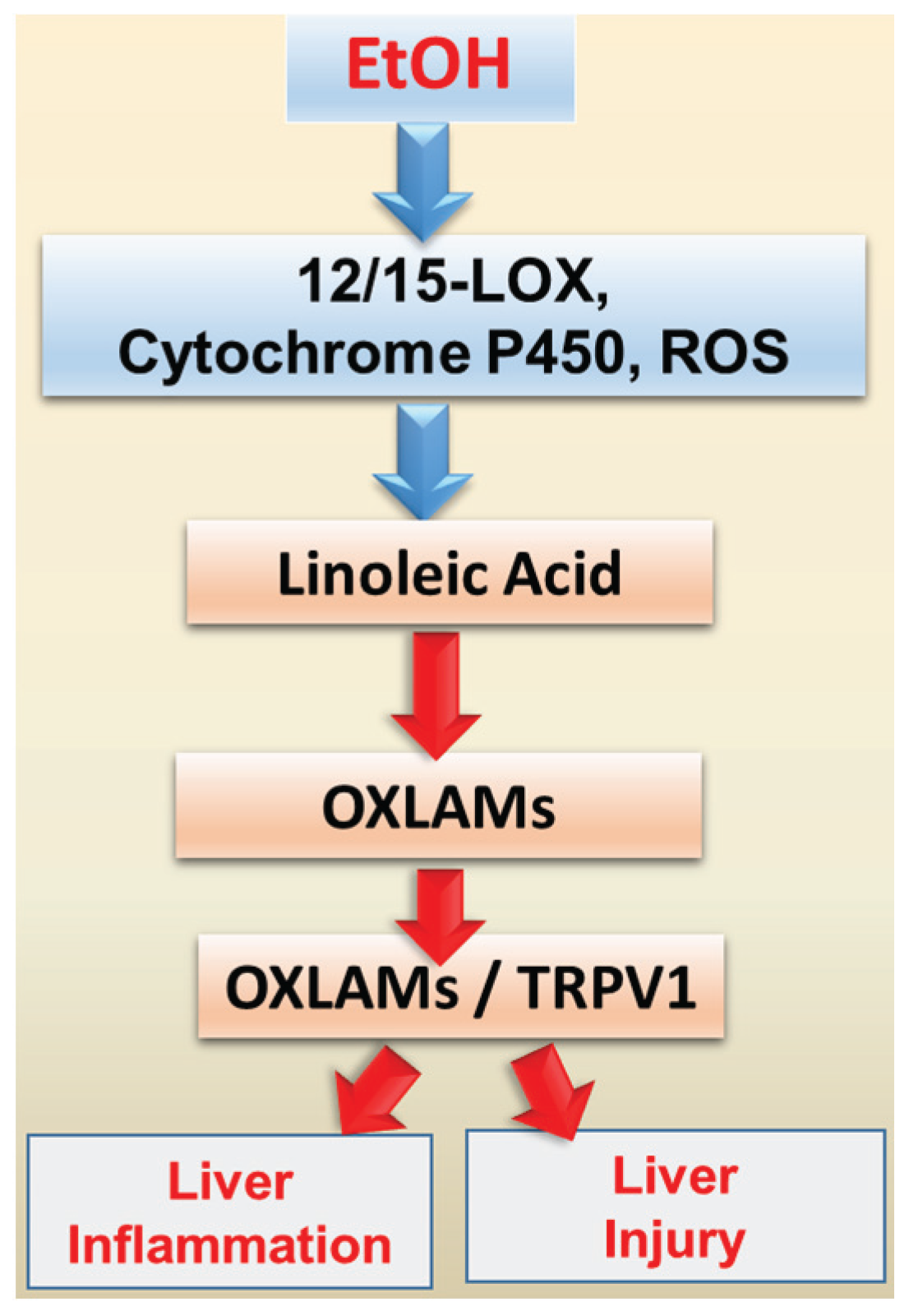
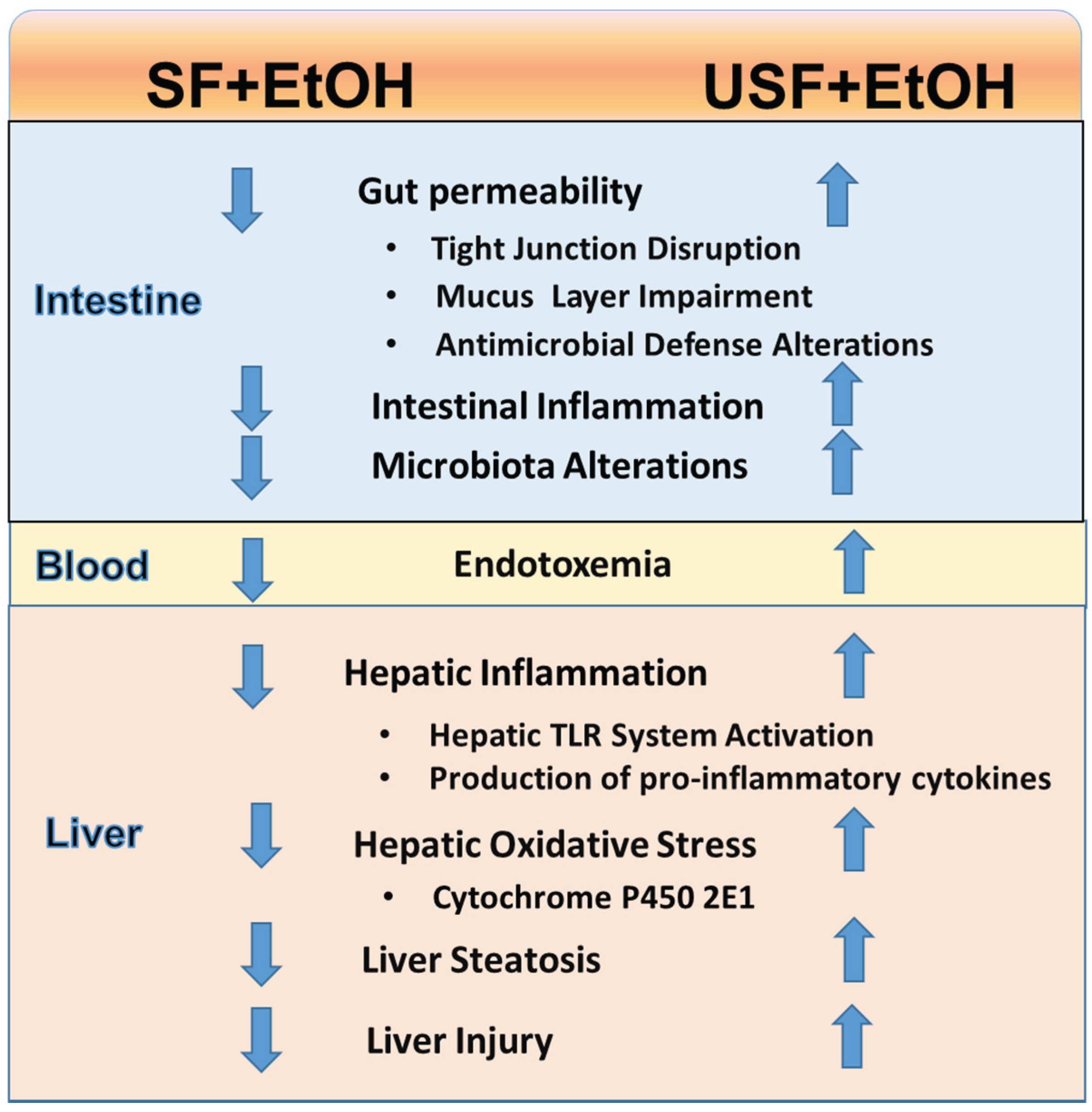
6. Dietary Fat and EtOH-mediated Changes in the Gut Microbiota
7. Therapeutic Implications of Dietary Lipids in ALD
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AA | arachidonic acid |
| AEA | N-arachidonoylethanolamine (anandamide) |
| 2-AG | 2-arachidonoylglycerol |
| ALA | alpha linolenic acid |
| ALD | alcoholic liver disease |
| ALT | alanine aminotransferase |
| AMPK | AMP-activated protein kinase |
| CYP2E1 | cytochrome P450 2E1 |
| DHA | docosahexaenoic acid |
| HNF4α | hepatocyte nuclear factor-4α |
| HO-1 | heme oxygenase-1 |
| HpODE | hydroperoxy-octadecadienoic acid |
| 9- and 13-HODEs | 9- and 13-hydroxy-octadecadienoic acids |
| EPA | eicosapentaenoic acid |
| EtOH | ethanol |
| IL-6 | interleukin 6 |
| LA | linoleic acid |
| LPS | lipopolysaccharide |
| 12/15-LOX | 12/15-lipoxygenase |
| LCFA | long chain fatty acids |
| LXA4 | lipoxin A4 |
| LXB4 | lipoxin B4 |
| MCT | medium chain triglyceride |
| NAFLD | nonalcoholic fatty liver disease |
| NF-kappaB | nuclear factor kappaB |
| OXLAMs | oxidized linoleic acid metabolites |
| oxoODE | oxo-octadecadienoic acid |
| PPARα | peroxisome proliferator-activated receptor alpha |
| PGC-1α | peroxisome proliferator-activated receptor gamma, coactivator 1 alpha |
| PUFA | polyunsaturated fatty acid |
| ROS | reactive oxygen species |
| SF | saturated fat |
| SCD-1 | stearoyl-CoA desaturase-1 |
| SIRT1 | Sirtuin |
| SREBP-1 | sterol regulatory element binding protein-1c |
| TJs | tight junctions |
| TG | triacylglycerol |
| TLR | Toll-like receptors |
| TRPV1 | Transient Receptor Potential Vanilloid 1 |
| TNF-α | tumor necrosis factor α |
| USF | unsaturated fat |
References
- Rehm, J.; Samokhvalov, A.; Shield, K. Global burden of alcoholic liver disease. J. Hepatol. 2013, 59, 160–168. [Google Scholar] [CrossRef] [PubMed]
- D’Amico, G.; Garcia-Tsao, G.; Pagliaro, L. Natural history and prognostic indicators of survival in cirrhosis: A systematic review of 118 studies. J. Hepatol. 2006, 44, 217–231. [Google Scholar] [CrossRef] [PubMed]
- Wakim-Fleming, J.; Mullen, K.D. Long-term management of alcoholic liver disease. Clin. Liver Dis. 2005, 9, 135–149. [Google Scholar] [CrossRef] [PubMed]
- Nanji, A.A. Role of different dietary fatty acids in the pathogenesis of experimental alcoholic liver disease. Alcohol 2004, 34, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Kirpich, I.A.; Feng, W.; Wang, Y.; Liu, Y.; Barker, D.F.; Barve, S.S.; McClain, C.J. The type of dietary fat modulates intestinal tight junction integrity, gut permeability, and hepatic toll-like receptor expression in a mouse model of alcoholic liver disease. Alcohol. Clin. Exp. Res. 2012, 36, 835–846. [Google Scholar] [CrossRef] [PubMed]
- Ronis, M.J.; Korourian, S.; Zipperman, M.; Hakkak, R.; Badger, T.M. Dietary saturated fat reduces alcoholic hepatotoxicity in rats by altering fatty acid metabolism and membrane composition. J. Nutr. 2004, 134, 904–912. [Google Scholar] [PubMed]
- Zhong, W.; Li, Q.; Xie, G.; Sun, X.; Tan, X.; Sun, X.; Jia, W.; Zhou, Z. Dietary fat sources differentially modulate intestinal barrier and hepatic inflammation in alcohol-induced liver injury in rats. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 305, G919–G932. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Torralba, M.; Tan, J.; Embree, M.; Zengler, K.; Stärkel, P.; van Pijkeren, J.P.; DePew, J.; Loomba, R.; Ho, S.B.; et al. Supplementation of saturated long-chain fatty acids maintains intestinal eubiosis and reduces ethanol-induced liver injury in mice. Gastroenterology 2015, 148, 203–214. [Google Scholar] [CrossRef] [PubMed]
- You, M.; Considine, R.V.; Leone, T.C.; Kelly, D.P.; Crabb, D.W. Role of adiponectin in the protective action of dietary saturated fat against alcoholic fatty liver in mice. Hepatology 2005, 42, 568–577. [Google Scholar] [CrossRef] [PubMed]
- Mezey, E. Dietary fat and alcoholic liver disease. Hepatology 1998, 28, 901–905. [Google Scholar] [CrossRef] [PubMed]
- Nanji, A.A.; French, S.W. Dietary factors and alcoholic cirrhosis. Alcohol. Clin. Exp. Res. 1986, 10, 271–273. [Google Scholar] [CrossRef] [PubMed]
- Lieber, C.S.; Lefèvre, A.; Spritz, N.; Feinman, L.; DeCarli, L.M. Difference in hepatic metabolism of long- and medium-chain fatty acids: The role of fatty acid chain length in the production of the alcoholic fatty liver. J. Clin. Investig. 1967, 46, 1451–1460. [Google Scholar] [CrossRef] [PubMed]
- Ronis, M.J.; Baumgardner, J.N.; Sharma, N.; Vantrease, J.; Ferguson, M.; Tong, Y.; Wu, X.; Cleves, M.A.; Badger, T.M. Medium chain triglycerides dose-dependently prevent liver pathology in a rat model of non-alcoholic fatty liver disease. Exp. Biol. Med. 2013, 238, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Nanji, A.A.; Mendenhall, C.L.; French, S.W. Beef fat prevents alcoholic liver disease in the rat. Alcohol. Clin. Exp. Res. 1989, 13, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Nanji, A.A.; Griniuviene, B.; Sadrzadeh, S.M.; Levitsky, S.; McCully, J.D. Effect of type of dietary fat and ethanol on antioxidant enzyme mRNA induction in rat liver. J. Lipid Res. 1995, 36, 736–744. [Google Scholar] [PubMed]
- Nanji, A.A.; French, S.W. Dietary linoleic acid is required for development of experimentally induced alcoholic liver injury. Life Sci. 1989, 44, 223–227. [Google Scholar] [CrossRef]
- Nanji, A.A.; Zhao, S.; Sadrzadeh, S.M.; Dannenberg, A.J.; Tahan, S.R.; Waxman, D.J. Markedly enhanced cytochrome P450 2E1 induction and lipid peroxidation is associated with severe liver injury in fish oil-ethanol-fed rats. Alcohol. Clin. Exp. Res. 1994, 18, 1280–1285. [Google Scholar] [CrossRef] [PubMed]
- Nanji, A.A.; Jokelainen, K.; Tipoe, G.L.; Rahemtulla, A.; Dannenberg, A.J. Dietary saturated fatty acids reverse inflammatory and fibrotic changes in rat liver despite continued ethanol administration. J. Pharmacol. Exp. Ther. 2001, 299, 638–644. [Google Scholar] [PubMed]
- Kasdallah-Grissa, A.; Nakbi, A.; Koubaa, N.; El-Fazaâ, S.; Gharbi, N.; Kamoun, A.; Hammami, M. Dietary virgin olive oil protects against lipid peroxidation and improves antioxidant status in the liver of rats chronically exposed to ethanol. Nutr. Res. 2008, 28, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.L.; Peng, H.C.; Wang, X.D.; Yang, S.C. Dietary saturated fatty acids reduce hepatic lipid accumulation but induce fibrotic change in alcohol-fed rats. Hepatobiliary Surg. Nutr. 2015, 4, 172–183. [Google Scholar] [PubMed]
- Kirpich, I.A.; Feng, W.; Wang, Y.; Liu, Y.; Beier, J.I.; Arteel, G.E.; Falkner, K.C.; Barve, S.S.; McClain, C.J. Ethanol and dietary unsaturated fat (corn oil/linoleic acid enriched) cause intestinal inflammation and impaired intestinal barrier defense in mice chronically fed alcohol. Alcohol 2013, 47, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Institute of Shortening and Edible Oils, Inc. Food Fats and Oils. Available online: http://www.iseo.org/httpdocs/Publications/FoodFatsOils2006.pdf (accessed on 5 May 2015).
- Dubois, V.; Breton, S.; Linder, M.; Fanni, J.; Parmentier, M. Fatty acid profiles of 80 vegetable oils with regard to their nutritional potential. Eur. J. Lipid Sci. Technol. 2007, 109, 710–732. [Google Scholar] [CrossRef]
- Nanji, A.A.; Zhao, S.; Lamb, R.G.; Dannenberg, A.J.; Sadrzadeh, S.M.; Waxman, D.J. Changes in cytochromes P-450, 2E1, 2B1, and 4A, and phospholipases A and C in the intragastric feeding rat model for alcoholic liver disease: Relationship to dietary fats and pathologic liver injury. Alcohol. Clin. Exp. Res. 1994, 18, 902–908. [Google Scholar] [CrossRef] [PubMed]
- Polavarapu, R.; Spitz, D.R.; Sim, J.E.; Follansbee, M.H.; Oberley, L.W.; Rahemtulla, A.; Nanji, A.A. Increased lipid peroxidation and impaired antioxidant enzyme function is associated with pathological liver injury in experimental alcoholic liver disease in rats fed diets high in corn oil and fish oil. Hepatology 1998, 27, 1317–1323. [Google Scholar] [CrossRef] [PubMed]
- Kono, H.; Enomoto, N.; Connor, H.D.; Wheeler, M.D.; Bradford, B.U.; Rivera, C.A.; Kadiiska, M.B.; Mason, R.P.; Thurman, R.G. Medium-chain triglycerides inhibit free radical formation and TNF-alpha production in rats given enteral ethanol. Am. J. Physiol. Gastrointest. Liver Physiol. 2000, 278, G467–G476. [Google Scholar] [PubMed]
- You, M.; Cao, Q.; Liang, X.; Ajmo, J.M.; Ness, G.C. Mammalian sirtuin 1 is involved in the protective action of dietary saturated fat against alcoholic fatty liver in mice. J. Nutr. 2008, 138, 497–501. [Google Scholar] [PubMed]
- You, M.; Rogers, C.Q. Adiponectin: A key adipokine in alcoholic fatty liver. Exp. Biol. Med. 2009, 234, 850–859. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zhong, W.; Qiu, Y.; Kang, X.; Sun, X.; Tan, X.; Zhao, Y.; Sun, X.; Jia, W.; Zhou, Z. Preservation of hepatocyte nuclear factor-4alpha contributes to the beneficial effect of dietary medium chain triglyceride on alcohol-induced hepatic lipid dyshomeostasis in rats. Alcohol. Clin. Exp. Res. 2013, 37, 587–598. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.; Zhao, Y.; McClain, C.J.; Kang, Y.J.; Zhou, Z. Inactivation of hepatocyte nuclear factor-4{alpha} mediates alcohol-induced downregulation of intestinal tight junction proteins. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 299, G643–G651. [Google Scholar] [CrossRef] [PubMed]
- Yashodhara, B.M.; Umakanth, S.; Pappachan, J.M.; Bhat, S.K.; Kamath, R.; Choo, B.H. Omega-3 fatty acids: A comprehensive review of their role in health and disease. Postgrad. Med. J. 2009, 85, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Lorente-Cebrian, S.; Costa, A.G.; Navas-Carretero, S.; Zabala, M.; Martínez, J.A.; Moreno-Aliaga, M.J. Role of omega-3 fatty acids in obesity, metabolic syndrome, and cardiovascular diseases: A review of the evidence. J. Physiol. Biochem. 2013, 69, 633–651. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Fernandez, L.; Laiglesia, L.M.; Huerta, A.E.; Martínez, J.A.; Moreno-Aliaga, M.J. Omega-3 fatty acids and adipose tissue function in obesity and metabolic syndrome. Prostaglandins Other Lipid Mediat. 2015. [Google Scholar] [CrossRef] [PubMed]
- Scorletti, E.; Byrne, C.D. Omega-3 fatty acids, hepatic lipid metabolism, and nonalcoholic fatty liver disease. Annu. Rev. Nutr. 2013, 33, 231–248. [Google Scholar] [CrossRef] [PubMed]
- Ellulu, M.S.; Khaza’ai, H.; Abed, Y.; Rahmat, A.; Ismail, P.; Ranneh, Y. Role of fish oil in human health and possible mechanism to reduce the inflammation. Inflammopharmacology 2015, 23, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Blasbalg, T.L.; Hibbeln, J.R.; Ramsden, C.E.; Majchrzak, S.F.; Rawlings, R.R. Changes in consumption of omega-3 and omega-6 fatty acids in the United States during the 20th century. Am. J. Clin. Nutr. 2011, 93, 950–962. [Google Scholar] [CrossRef] [PubMed]
- Zelber-Sagi, S.; Nitzan-Kaluski, D.; Goldsmith, R.; Webb, M.; Blendis, L.; Halpern, Z.; Oren, R. Long term nutritional intake and the risk for non-alcoholic fatty liver disease (NAFLD): A population based study. J. Hepatol. 2007, 47, 711–717. [Google Scholar] [CrossRef] [PubMed]
- Russo, G.L. Dietary n-6 and n-3 polyunsaturated fatty acids: From biochemistry to clinical implications in cardiovascular prevention. Biochem. Pharmacol. 2009, 77, 937–946. [Google Scholar] [CrossRef] [PubMed]
- Spahis, S.; Alvarez, F.; Dubois, J.; Ahmed, N.; Peretti, N.; Levy, E. Plasma fatty acid composition in French-Canadian children with non-alcoholic fatty liver disease: Effect of n-3 PUFA supplementation. Prostaglandins Leukot. Essent. Fatty Acids 2015, 99, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Capanni, M.; Calella, F.; Biagini, M.R.; Genise, S.; Raimondi, L.; Bedogni, G.; Svegliati-Baroni, G.; Sofi, F.; Milani, S.; Abbate, R.; et al. Prolonged n-3 polyunsaturated fatty acid supplementation ameliorates hepatic steatosis in patients with non-alcoholic fatty liver disease: A pilot study. Aliment. Pharmacol. Ther. 2006, 23, 1143–1151. [Google Scholar] [CrossRef] [PubMed]
- Nobili, V.; Alisi, A.; Della Corte, C.; Risé, P.; Galli, C.; Agostoni, C.; Bedogni, G. Docosahexaenoic acid for the treatment of fatty liver: Randomised controlled trial in children. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 1066–1070. [Google Scholar] [CrossRef] [PubMed]
- Pachikian, B.D.; Essaghir, A.; Demoulin, J.B.; Neyrinck, A.M.; Catry, E.; de Backer, F.C.; Dejeans, N.; Dewulf, E.M.; Sohet, F.M.; Portois, L.; et al. Hepatic n-3 polyunsaturated fatty acid depletion promotes steatosis and insulin resistance in mice: Genomic analysis of cellular targets. PLoS ONE 2011, 6, e23365. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, R.; Espinosa, A.; González-Mañán, D.; D’Espessailles, A.; Fernández, V.; Videla, L.A.; Tapia, G. N-3 long-chain polyunsaturated fatty acid supplementation significantly reduces liver oxidative stress in high fat induced steatosis. PLoS ONE 2012, 7, e46400. [Google Scholar] [CrossRef] [PubMed]
- Kajikawa, S.; Harada, T.; Kawashima, A.; Imada, K.; Mizuguchi, K. Highly purified eicosapentaenoic acid ethyl ester prevents development of steatosis and hepatic fibrosis in rats. Dig. Dis. Sci. 2010, 55, 631–641. [Google Scholar] [CrossRef] [PubMed]
- Kajikawa, S.; Harada, T.; Kawashima, A.; Imada, K.; Mizuguchi, K. Highly purified eicosapentaenoic acid prevents the progression of hepatic steatosis by repressing monounsaturated fatty acid synthesis in high-fat/high-sucrose diet-fed mice. Prostaglandins Leukot. Essent. Fatty Acids 2009, 80, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Tapia, G.; Valenzuela, R.; Espinosa, A.; Romanque, P.; Dossi, C.; Gonzalez-Mañán, D.; Videla, L.A.; D’Espessailles, A. N-3 long-chain PUFA supplementation prevents high fat diet induced mouse liver steatosis and inflammation in relation to PPAR-alpha upregulation and NF-kappaB DNA binding abrogation. Mol. Nutr. Food Res. 2014, 58, 1333–1341. [Google Scholar] [CrossRef] [PubMed]
- Dossi, C.G.; Tapia, G.S.; Espinosa, A.; Videla, L.A.; D’Espessailles, A. Reversal of high-fat diet-induced hepatic steatosis by n-3 LCPUFA: Role of PPAR-alpha and SREBP-1c. J. Nutr. Biochem. 2014, 25, 977–984. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, M.; Zern, M.A.; Hagbjörk, A.L.; Ingelman-Sundberg, M.; French, S.W. Fish oil, alcohol, and liver pathology: Role of cytochrome P450 2E1. Proc. Soc. Exp. Biol. Med. 1994, 207, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Wada, S.; Yamazaki, T.; Kawano, Y.; Miura, S.; Ezaki, O. Fish oil fed prior to ethanol administration prevents acute ethanol-induced fatty liver in mice. J. Hepatol. 2008, 49, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Song, B.J.; Moon, K.H.; Olsson, N.U.; Salem, N., Jr. Prevention of alcoholic fatty liver and mitochondrial dysfunction in the rat by long-chain polyunsaturated fatty acids. J. Hepatol. 2008, 49, 262–273. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.L.; Wan, J.B.; Wang, B.; He, C.W.; Ma, H.; Li, T.W.; Kang, J.X. Suppression of acute ethanol-induced hepatic steatosis by docosahexaenoic acid is associated with downregulation of stearoyl-CoA desaturase 1 and inflammatory cytokines. Prostaglandins Leukot. Essent. Fatty Acids 2013, 88, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Pawlosky, R.J.; Flynn, B.M.; Salem, N., Jr. The effects of low dietary levels of polyunsaturates on alcohol-induced liver disease in rhesus monkeys. Hepatology 1997, 26, 1386–1392. [Google Scholar] [CrossRef] [PubMed]
- Pawlosky, R.J.; Salem, N., Jr. Development of alcoholic fatty liver and fibrosis in rhesus monkeys fed a low n-3 fatty acid diet. Alcohol. Clin. Exp. Res. 2004, 28, 1569–1576. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.; McLean, C.H.; Silvers, K.M. Are the health benefits of fish oils limited by products of oxidation? Nutr. Res. Rev. 2006, 19, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids; The National Academy Press: Washington, DC, USA, 2005.
- Kris-Etherton, P.M.; Taylor, D.S.; Yu-Poth, S.; Huth, P.; Moriarty, K.; Fishell, V.; Hargrove, R.L.; Zhao, G.; Etherton, T.D. Polyunsaturated fatty acids in the food chain in the United States. Am. J. Clin. Nutr. 2000, 71, 179S–188S. [Google Scholar] [PubMed]
- Choe, E.; Min, D.B. Chemistry of deep-fat frying oils. J. Food Sci. 2007, 72, R77–R86. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.C. Chemical and biological properties related to toxicity of heated fats. J. Toxicol. Environ. Health 1981, 7, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Halvorsen, B.L.; Blomhoff, R. Determination of lipid oxidation products in vegetable oils and marine omega-3 supplements. Food Nutr. Res. 2011. [Google Scholar] [CrossRef] [PubMed]
- Perez-Herrera, A.; Rangel-Zuñiga, O.A.; Delgado-Lista, J.; Marin, C.; Perez-Martinez, P.; Tasset, I.; Tunez, I.; Quintana-Navarro, G.M.; Lopez-Segura, F.; Luque de Castro, M.D.; et al. The antioxidants in oils heated at frying temperature, whether natural or added, could protect against postprandial oxidative stress in obese people. Food Chem. 2013, 138, 2250–2259. [Google Scholar] [CrossRef] [PubMed]
- Kanner, J. Dietary advanced lipid oxidation endproducts are risk factors to human health. Mol. Nutr. Food Res. 2007, 51, 1094–1101. [Google Scholar] [CrossRef] [PubMed]
- Ng, C.Y.; Leong, X.F.; Masbah, N.; Adam, S.K.; Kamisah, Y.; Jaarin, K. Heated vegetable oils and cardiovascular disease risk factors. Vasc. Pharmacol. 2014, 61, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Perez-Herrera, A.; Delgado-Lista, J.; Torres-Sanchez, L.A.; Rangel-Zuñiga, O.A.; Camargo, A.; Moreno-Navarrete, J.M.; Garcia-Olid, B.; Quintana-Navarro, G.M.; Alcala-Diaz, J.F.; Muñoz-Lopez, C.; et al. The postprandial inflammatory response after ingestion of heated oils in obese persons is reduced by the presence of phenol compounds. Mol. Nutr. Food Res. 2012, 56, 510–514. [Google Scholar] [CrossRef] [PubMed]
- Owu, D.U.; Osim, E.E.; Ebong, P.E. Serum liver enzymes profile of Wistar rats following chronic consumption of fresh or oxidized palm oil diets. Acta Trop. 1998, 69, 65–73. [Google Scholar] [CrossRef]
- Nwanguma, B.C.; Achebe, A.C.; Ezeanyika, L.U.; Eze, L.C. Toxicity of oxidized fats II: Tissue levels of lipid peroxides in rats fed a thermally oxidized corn oil diet. Food Chem. Toxicol. 1999, 37, 413–416. [Google Scholar] [CrossRef]
- Shibayama, Y. Hepatotoxicity of heated and oxygenated corn oil. Exp. Toxicol. Pathol. 1992, 44, 255–258. [Google Scholar] [CrossRef]
- Varma, P.S.; Aruna, K.; Rukkumani, R.; Menon, V.P. Alcohol and thermally oxidized pufa induced oxidative stress: Role of N-acetyl cysteine. Ital. J. Biochem. 2004, 53, 10–15. [Google Scholar] [PubMed]
- Aruna, K.; Rukkumani, R.; Varma, P.S.; Menon, V.P. Therapeutic role of Cuminum cyminum on ethanol and thermally oxidized sunflower oil induced toxicity. Phytother. Res. 2005, 19, 416–421. [Google Scholar] [CrossRef] [PubMed]
- Rukkumani, R.; Aruna, K.; Varma, P.S.; Viswanathan, P.; Rajasekaran, K.N.; Menon, V.P. Protective role of a novel curcuminoid on alcohol and PUFA-induced hyperlipidemia. Toxicol. Mech. Methods 2005, 15, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Rukkumani, R.; Aruna, K.; Varma, P.S.; Menon, V.P. Influence of ferulic acid on circulatory prooxidant-antioxidant status during alcohol and PUFA induced toxicity. J. Physiol. Pharmacol. 2004, 55, 551–561. [Google Scholar] [PubMed]
- Latha, P.; Chaitanya, D.; Rukkumani, R. Protective effect of Phyllanthus niruri on alcohol and heated sunflower oil induced hyperlipidemia in Wistar rats. Toxicol. Mech. Methods 2010, 20, 498–503. [Google Scholar] [CrossRef] [PubMed]
- Ringseis, R.; Muschick, A.; Eder, K. Dietary oxidized fat prevents ethanol-induced triacylglycerol accumulation and increases expression of PPARalpha target genes in rat liver. J. Nutr. 2007, 137, 77–83. [Google Scholar] [PubMed]
- Feldstein, A.E.; Lopez, R.; Tamimi, T.A.; Yerian, L.; Chung, Y.M.; Berk, M.; Zhang, R.; McIntyre, T.M.; Hazen, S.L. Mass spectrometric profiling of oxidized lipid products in human nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. J. Lipid Res. 2010, 51, 3046–3054. [Google Scholar] [CrossRef] [PubMed]
- Zein, C.O.; Yerian, L.M.; Gogate, P.; Lopez, R.; Kirwan, J.P.; Feldstein, A.E.; McCullough, A.J. Pentoxifylline improves nonalcoholic steatohepatitis: A randomized placebo-controlled trial. Hepatology 2011, 54, 1610–1619. [Google Scholar] [CrossRef] [PubMed]
- Raszeja-Wyszomirska, J.; Safranow, K.; Milkiewicz, M.; Milkiewicz, P.; Szynkowska, A.; Stachowska, E. Lipidic last breath of life in patients with alcoholic liver disease. Prostaglandins Other Lipid Mediat. 2012, 99, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Latchoumycandane, C.; McMullen, M.R.; Pratt, B.T.; Zhang, R.; Papouchado, B.G.; Nagy, L.E.; Feldstein, A.E.; McIntyre, T.M. Chronic alcohol exposure increases circulating bioactive oxidized phospholipids. J. Biol. Chem. 2010, 285, 22211–22220. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Beier, J.I.; Arteel, G.E.; Ramsden, C.E.; Feldstein, A.E.; McClain, C.J.; Kirpich, I.A. Transient receptor potential vanilloid 1 gene deficiency ameliorates hepatic injury in a mouse model of chronic binge alcohol-induced alcoholic liver disease. Am. J. Pathol. 2015, 185, 43–54. [Google Scholar] [CrossRef] [PubMed]
- Patwardhan, A.M.; Akopian, A.N.; Ruparel, N.B.; Diogenes, A.; Weintraub, S.T.; Uhlson, C.; Murphy, R.C.; Hargreaves, K.M. Heat generates oxidized linoleic acid metabolites that activate TRPV1 and produce pain in rodents. J. Clin. Investig. 2010, 120, 1617–1626. [Google Scholar] [CrossRef] [PubMed]
- Patwardhan, A.M.; Scotland, P.E.; Akopian, A.N.; Hargreaves, K.M. Activation of TRPV1 in the spinal cord by oxidized linoleic acid metabolites contributes to inflammatory hyperalgesia. Proc. Natl. Acad. Sci. USA 2009, 106, 18820–18824. [Google Scholar] [CrossRef] [PubMed]
- Whitman, W.B.; Coleman, D.C.; Wiebe, W.J. Prokaryotes: The unseen majority. Proc. Natl. Acad. Sci. USA 1998, 95, 6578–6583. [Google Scholar] [CrossRef] [PubMed]
- Tremaroli, V.; Backhed, F. Functional interactions between the gut microbiota and host metabolism. Nature 2012, 489, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, J.K.; Holmes, E.; Kinross, J.; Burcelin, R.; Gibson, G.; Jia, W.; Pettersson, S. Host-gut microbiota metabolic interactions. Science 2012, 336, 1262–1267. [Google Scholar] [CrossRef] [PubMed]
- Mutlu, E.; Keshavarzian, A.; Engen, P.; Forsyth, C.B.; Sikaroodi, M.; Gillevet, P. Intestinal dysbiosis: A possible mechanism of alcohol-induced endotoxemia and alcoholic steatohepatitis in rats. Alcohol. Clin. Exp. Res. 2009, 33, 1836–1846. [Google Scholar] [CrossRef] [PubMed]
- Mutlu, E.A.; Gillevet, P.M.; Rangwala, H.; Sikaroodi, M.; Naqvi, A.; Engen, P.A.; Kwasny, M.; Lau, C.K.; Keshavarzian, A. Colonic microbiome is altered in alcoholism. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 302, G966–G978. [Google Scholar] [CrossRef] [PubMed]
- Yan, A.W.; Fouts, D.E.; Brandl, J.; Stärkel, P.; Torralba, M.; Schott, E.; Tsukamoto, H.; Nelson, K.E.; Brenner, D.A.; Schnabl, B. Enteric dysbiosis associated with a mouse model of alcoholic liver disease. Hepatology 2011, 53, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Bull-Otterson, L.; Feng, W.; Kirpich, I.; Wang, Y.; Qin, X.; Liu, Y.; Gobejishvili, L.; Joshi-Barve, S.; Ayvaz, T.; Petrosino, J.; et al. Metagenomic analyses of alcohol induced pathogenic alterations in the intestinal microbiome and the effect of Lactobacillus rhamnosus GG treatment. PLoS ONE 2013, 8, e53028. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Stärkel, P.; Turner, J.R.; Ho, S.B.; Schnabl, B. Dysbiosis-induced intestinal inflammation activates tumor necrosis factor receptor I and mediates alcoholic liver disease in mice. Hepatology 2015, 61, 883–894. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Wei, X.; Yin, X.; Wang, Y.; Zhang, M.; Zhao, C.; Zhao, H.; McClain, C.J.; Feng, W.; Zhang, X. Hepatic and fecal metabolomic analysis of the effects of Lactobacillus rhamnosus GG on alcoholic fatty liver disease in mice. J. Proteome Res. 2015, 14, 1174–1182. [Google Scholar] [CrossRef] [PubMed]
- Kirpich, I.A.; Solovieva, N.V.; Leikhter, S.N.; Shidakova, N.A.; Lebedeva, O.V.; Sidorov, P.I.; Bazhukova, T.A.; Soloviev, A.G.; Barve, S.S.; McClain, C.J.; et al. Probiotics restore bowel flora and improve liver enzymes in human alcohol-induced liver injury: A pilot study. Alcohol 2008, 42, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, M.A.; Hoffmann, C.; Sherrill-Mix, S.A.; Keilbaugh, S.A.; Hamady, M.; Chen, Y.Y.; Knight, R.; Ahima, R.S.; Bushman, F.; Wu, G.D. High-fat diet determines the composition of the murine gut microbiome independently of obesity. Gastroenterology 2009. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Bäckhed, F.; Fulton, L.; Gordon, J.I. Diet-induced obesity is linked to marked but reversible alterations in the mouse distal gut microbiome. Cell Host Microbe 2008, 3, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ridaura, V.K.; Faith, J.J.; Rey, F.E.; Knight, R.; Gordon, J.I. The effect of diet on the human gut microbiome: A metagenomic analysis in humanized gnotobiotic mice. Sci. Transl. Med. 2009. [Google Scholar] [CrossRef] [PubMed]
- De Wit, N.; Derrien, M.; Bosch-Vermeulen, H.; Oosterink, E.; Keshtkar, S.; Duval, C.; de Vogel-van den Bosch, J.; Kleerebezem, M.; Müller, M.; van der Meer, R. Saturated fat stimulates obesity and hepatic steatosis and affects gut microbiota composition by an enhanced overflow of dietary fat to the distal intestine. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 303, G589–G599. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.Y.; Keilbaugh, S.A.; Bewtra, M.; Knights, D.; Walters, W.A.; Knight, R.; et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 2011, 334, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Duncan, S.H.; Lobley, G.E.; Holtrop, G.; Ince, J.; Johnstone, A.M.; Louis, P.; Flint, H.J. Human colonic microbiota associated with diet, obesity and weight loss. Int. J. Obes. 2008, 32, 1720–1724. [Google Scholar] [CrossRef] [PubMed]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Kirpich, I.; Petrosino, J.; Ajami, N.; Feng, W.; Wang, Y.; Liu, Y.; Beier, J.; Barve, S.; Yin, X.; Wei, X.; et al. Saturated and unsaturated dietary fats differentially modulate ethanol-induced changes in gut microbiome and metabolome in a mouse model of alcoholic liver disease. Am. J. Pathol. 2015. accepted. [Google Scholar]
- Vanek, V.W.; Seidner, D.L.; Allen, P.; Bistrian, B.; Collier, S.; Gura, K.; Miles, J.M.; Valentine, C.J.; Kochevar, M.; Novel Nutrient Task Force, Intravenous Fat Emulsions Workgroup; et al. A.S.P.E.N. position paper: Clinical role for alternative intravenous fat emulsions. Nutr. Clin. Pract. 2012, 27, 150–192. [Google Scholar] [CrossRef] [PubMed]
- Cresci, G.A.; Bush, K.; Nagy, L.E. Tributyrin supplementation protects mice from acute ethanol-induced gut injury. Alcohol. Clin. Exp. Res. 2014, 38, 1489–1501. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kirpich, I.A.; Miller, M.E.; Cave, M.C.; Joshi-Barve, S.; McClain, C.J. Alcoholic Liver Disease: Update on the Role of Dietary Fat. Biomolecules 2016, 6, 1. https://doi.org/10.3390/biom6010001
Kirpich IA, Miller ME, Cave MC, Joshi-Barve S, McClain CJ. Alcoholic Liver Disease: Update on the Role of Dietary Fat. Biomolecules. 2016; 6(1):1. https://doi.org/10.3390/biom6010001
Chicago/Turabian StyleKirpich, Irina A., Matthew E. Miller, Matthew C. Cave, Swati Joshi-Barve, and Craig J. McClain. 2016. "Alcoholic Liver Disease: Update on the Role of Dietary Fat" Biomolecules 6, no. 1: 1. https://doi.org/10.3390/biom6010001






