Knockout of RNA Binding Protein MSI2 Impairs Follicle Development in the Mouse Ovary: Characterization of MSI1 and MSI2 during Folliculogenesis
Abstract
:1. Introduction
2. Results and Discussion
2.1. MSI1 and MSI2 Time-Course Characterization
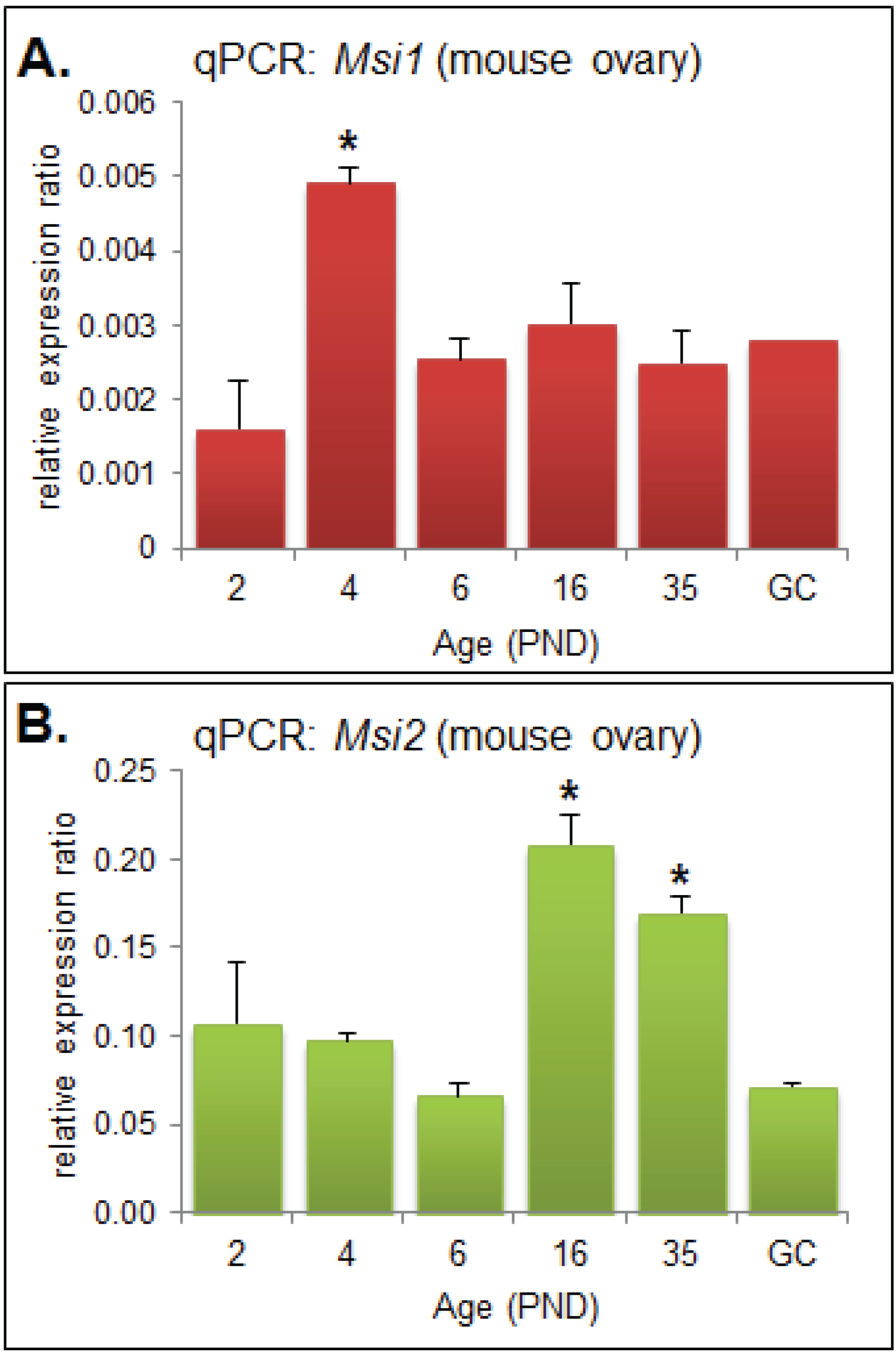
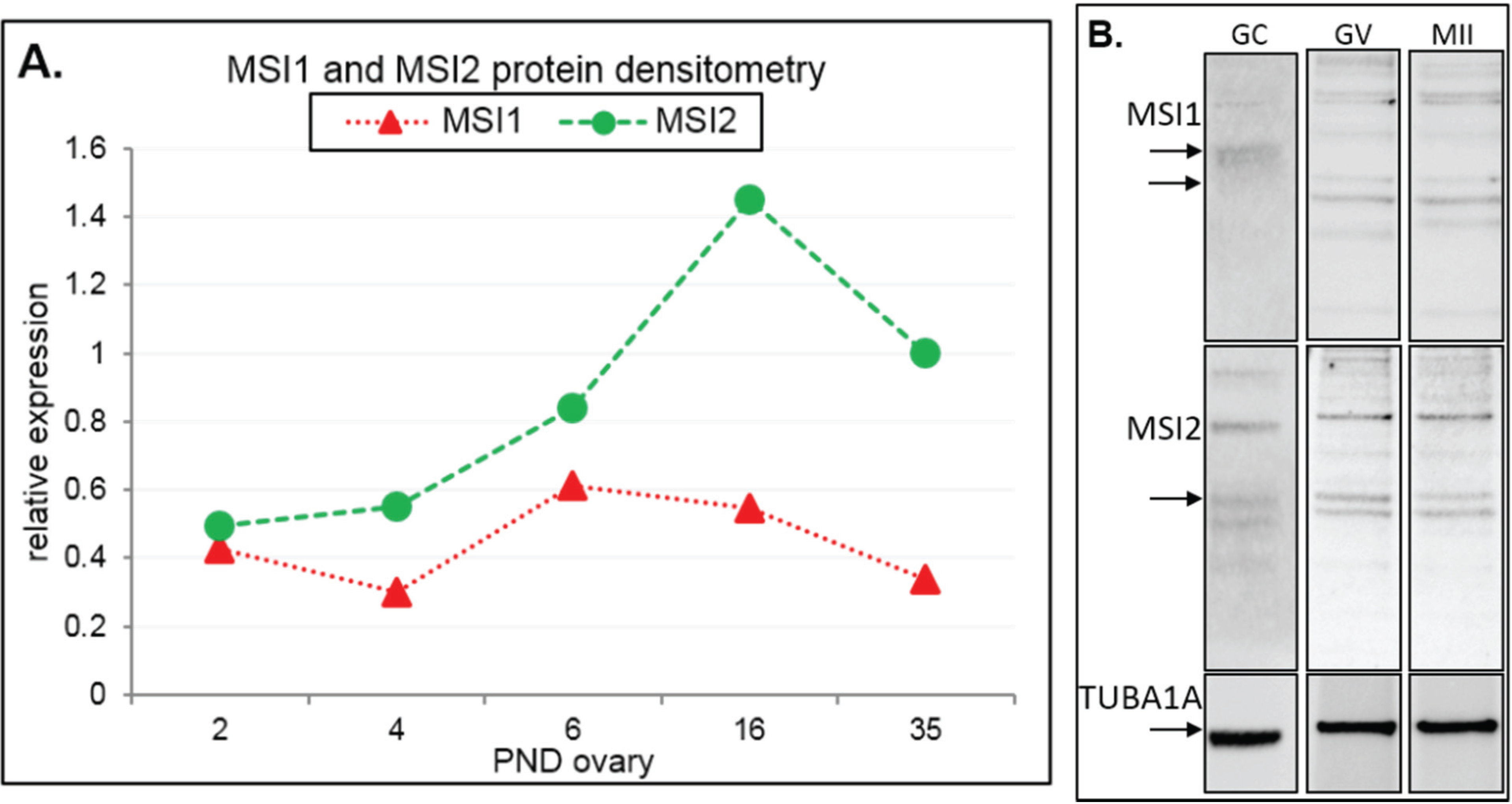
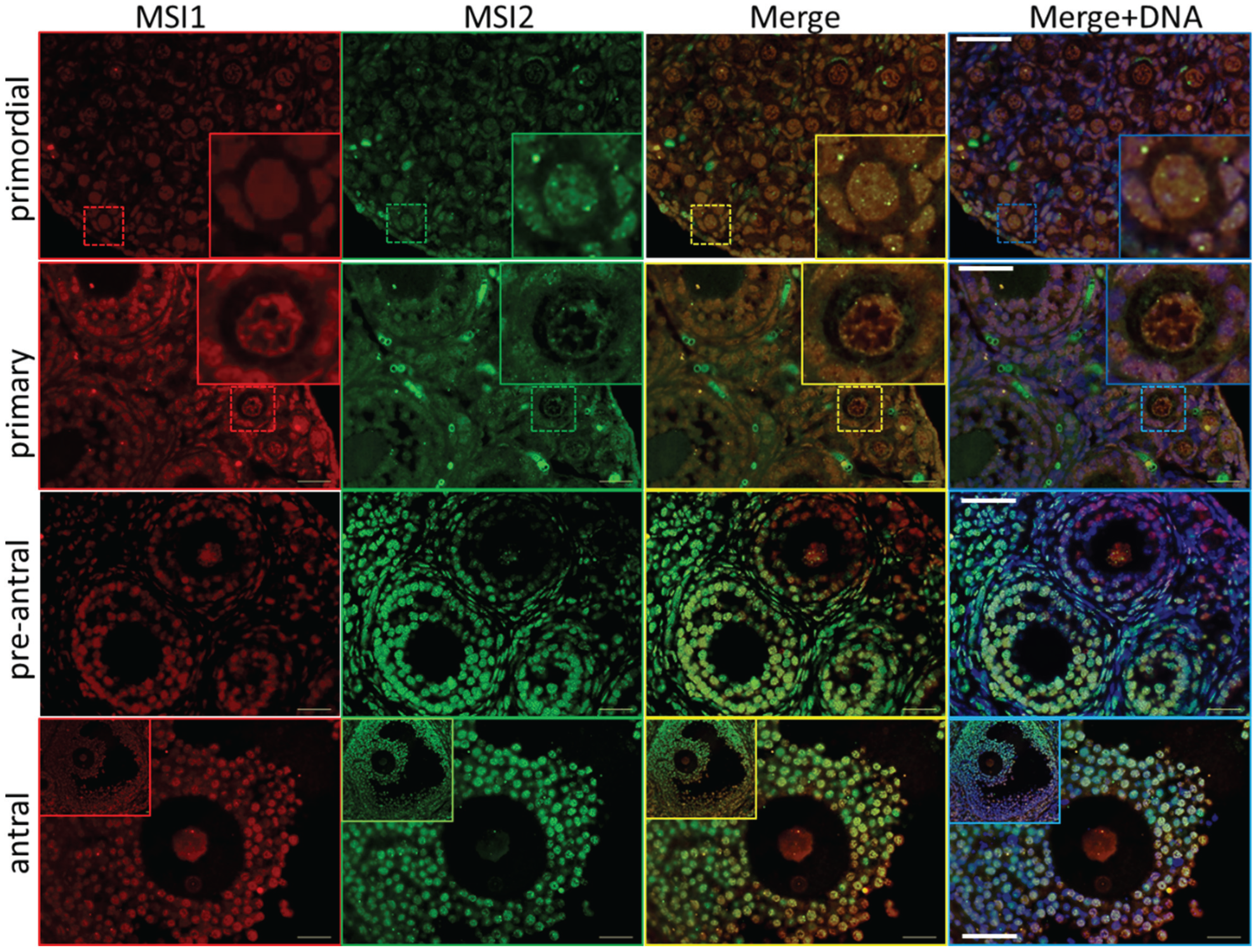
2.2. Analysis of Folliculogenesis in an Msi2-Deficient Mouse Model

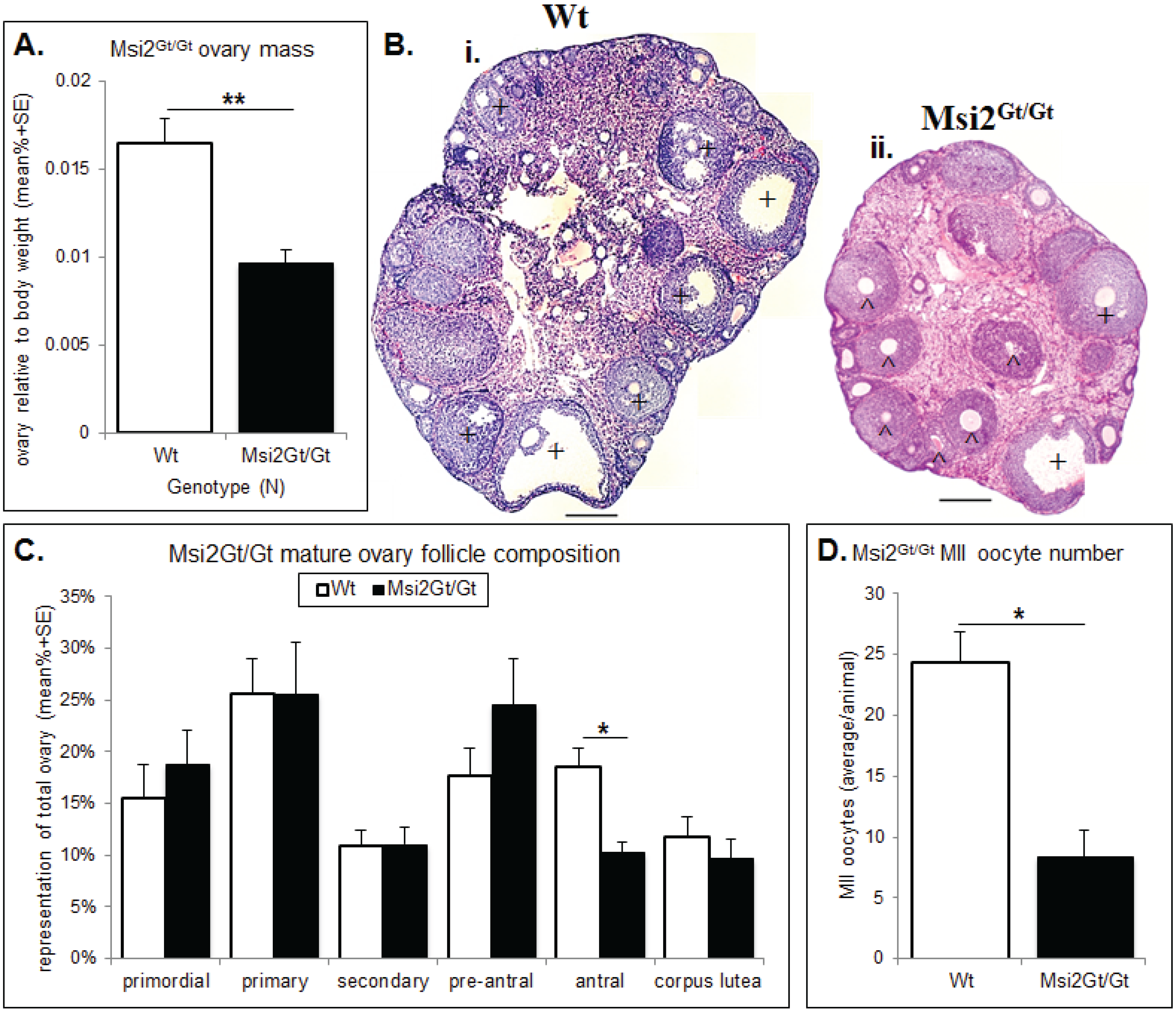
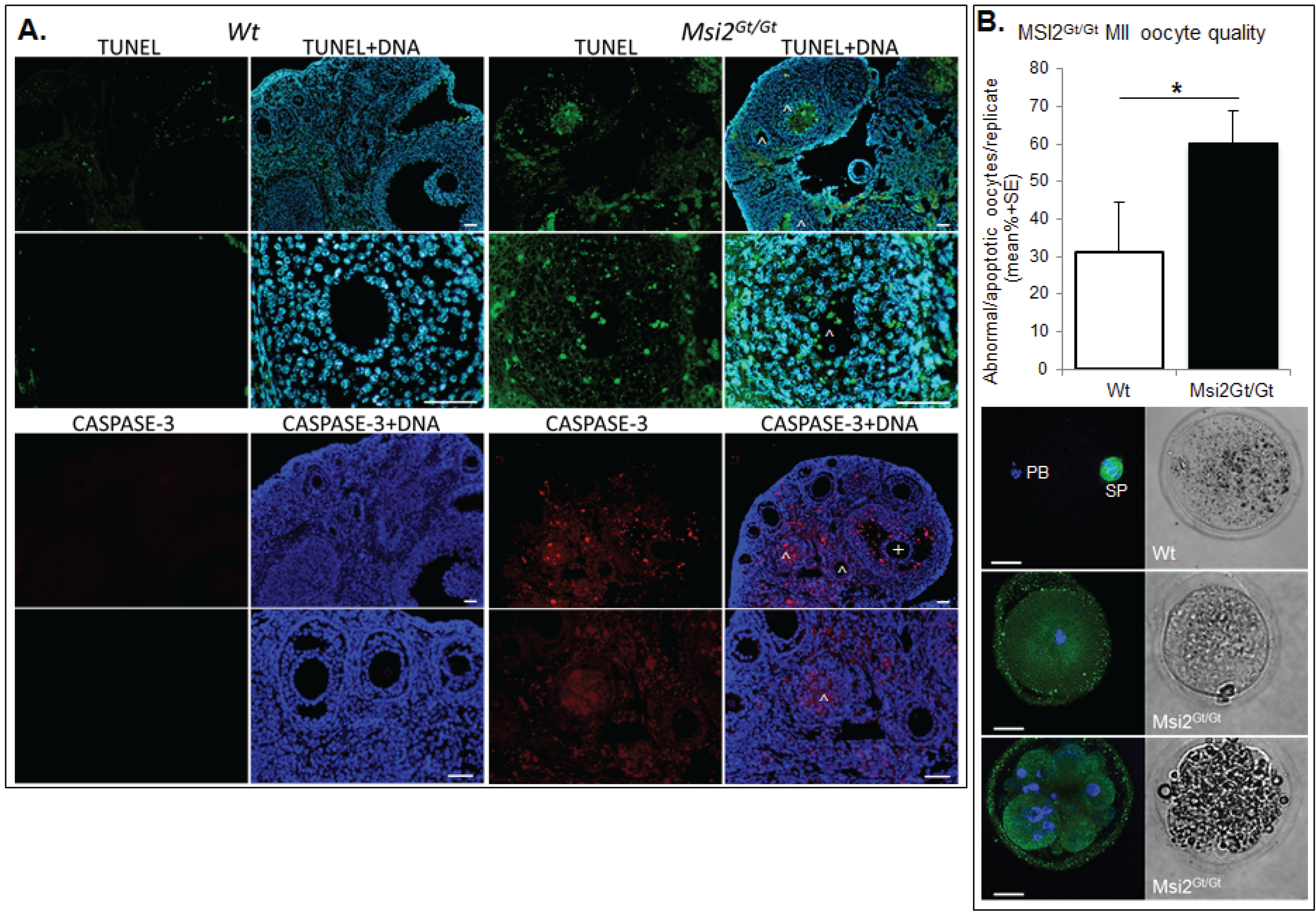
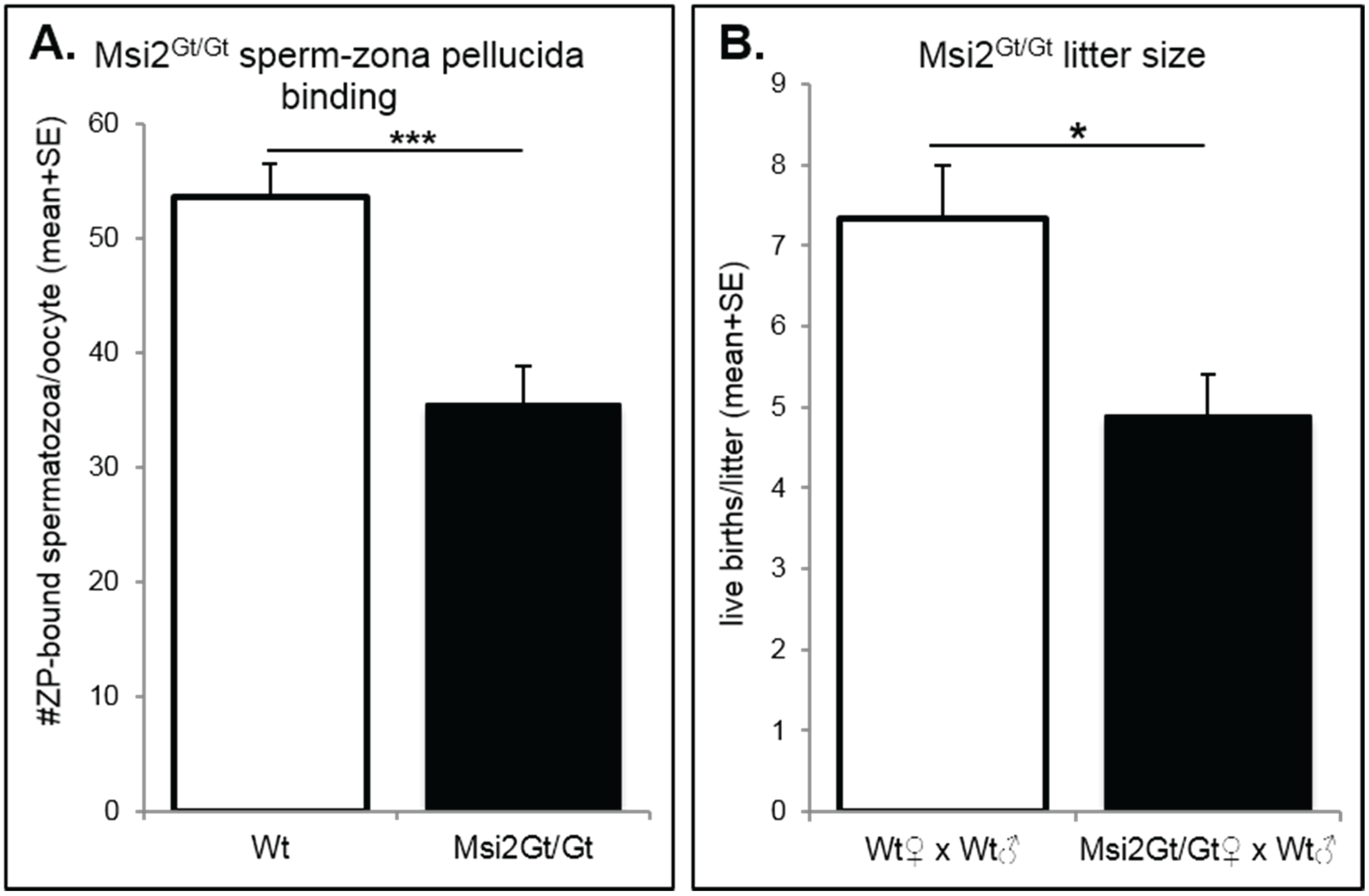
2.3. Discussion
3. Experimental Section
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Te Velde, E.R.; Pearson, P.L. The variability of female reproductive ageing. Hum. Reprod. Updat. 2002, 8, 141–154. [Google Scholar] [CrossRef]
- Dunson, D.B.; Colombo, B.; Baird, D.D. Changes with age in the level and duration of fertility in the menstrual cycle. Hum. Reprod. Oxf. Engl. 2002, 17, 1399–1403. [Google Scholar] [CrossRef]
- Faddy, M.J.; Gosden, R.G.; Gougeon, A.; Richardson, S.J.; Nelson, J.F. Accelerated disappearance of ovarian follicles in mid-life: Implications for forecasting menopause. Hum. Reprod. Oxf. Engl. 1992, 7, 1342–1346. [Google Scholar]
- Kezele, P.; Skinner, M.K. Regulation of ovarian primordial follicle assembly and development by estrogen and progesterone: Endocrine model of follicle assembly. Endocrinology 2003, 144, 3329–3337. [Google Scholar] [CrossRef] [PubMed]
- Knight, P.G.; Glister, C. TGF-beta superfamily members and ovarian follicle development. Reproduction 2006, 132, 191–206. [Google Scholar] [CrossRef] [PubMed]
- Tilly, J.L.; Billig, H.; Kowalski, K.I.; Hsueh, A.J. Epidermal growth factor and basic fibroblast growth factor suppress the spontaneous onset of apoptosis in cultured rat ovarian granulosa cells and follicles by a tyrosine kinase-dependent mechanism. Mol. Endocrinol. Baltim. Md. 1992, 6, 1942–1950. [Google Scholar]
- Asai, R.; Okano, H.; Yasugi, S. Correlation between Musashi-1 and C-hairy-1 expression and cell proliferation activity in the developing intestine and stomach of both chicken and mouse. Dev. Growth Differ. 2005, 47, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Okano, H.; Imai, T.; Okabe, M. Musashi: A translational regulator of cell fate. J. Cell Sci. 2002, 115, 1355–1359. [Google Scholar] [PubMed]
- Okano, H.; Kawahara, H.; Toriya, M.; Nakao, K.; Shibata, S.; Imai, T. Function of RNA-binding protein Musashi-1 in stem cells. Exp. Cell Res. 2005, 306, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Sakakibara, S.; Nakamura, Y.; Yoshida, T.; Shibata, S.; Koike, M.; Takano, H.; Uchiyama, Y.; Ueda, S.; Noda, T.; Okano, H. RNA-binding protein Musashi family: Roles for CNS stem cells and a subpopulation of ependymal cells revealed by targeted disruption and antisense ablation. Proc. Natl. Acad. Sci. USA 2002, 99, 15194–15199. [Google Scholar] [CrossRef] [PubMed]
- Kharas, M.G.; Lengner, C.J.; Al-Shahrour, F.; Bullinger, L.; Ball, B.; Zaidi, S.; Morgan, K.; Tam, W.; Paktinat, M.; Okabe, R. Musashi-2 regulates normal hematopoiesis and promotes aggressive myeloid leukemia. Nat. Med. 2010, 16, 903–908. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, K.; MacNicol, M.C.; MacNicol, A.M. Autoregulation of Musashi1 mRNA translation during xenopus oocyte maturation. Mol. Reprod. Dev. 2012, 79, 553–563. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, K.; MacNicol, M.C.; Wang, Y.; Cragle, C.E.; Tackett, A.J.; Hardy, L.L.; MacNicol, A.M. Ringo/cyclin-dependent kinase and mitogen-activated protein kinase signaling pathways regulate the activity of the cell fate determinant Musashi to promote cell cycle re-entry in xenopus oocytes. J. Biol. Chem. 2012, 287, 10639–10649. [Google Scholar] [CrossRef] [PubMed]
- Cragle, C.; Macnicol, A.M. Musashi protein-directed translational activation of target mRNAs is mediated by the poly[A] polymerase, germline development defective-2. J. Biol. Chem. 2014, 18, 14239–14251. [Google Scholar] [CrossRef] [PubMed]
- Charlesworth, A.; Wilczynska, A.; Thampi, P.; Cox, L.L.; MacNicol, A.M. Musashi regulates the temporal order of mRNA translation during xenopus oocyte maturation. EMBO J. 2006, 25, 2792–2801. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, J.M.; Fraser, B.A.; Sobinoff, A.P.; Pye, V.J.; Davidson, T.L.; Siddall, N.A.; Koopman, P.; Hime, G.R.; McLaughlin, E.A. Developmental expression of Musashi-1 and Musashi-2 RNA-binding proteins during spermatogenesis: Analysis of the deleterious effects of dysregulated expression. Biol. Reprod. 2014. [Google Scholar] [CrossRef] [PubMed]
- Siddall, N.A.; McLaughlin, E.A.; Marriner, N.L.; Hime, G.R. The RNA-binding protein Musashi is required intrinsically to maintain stem cell identity. Proc. Natl. Acad. Sci. USA 2006, 103, 8402–8407. [Google Scholar] [CrossRef] [PubMed]
- Siddall, N.A.; Kalcina, M.; Johanson, T.M.; Monk, A.C.; Casagranda, F.; Been, R.P.; Hime, G.R.; McLaughlin, E.A. Drosophila Rbp6 is an orthologue of vertebrate Msi-1 and Msi-2, but does not function redundantly with dMsi to regulate germline stem cell behaviour. PLoS ONE 2012, 7, e49810. [Google Scholar] [CrossRef] [PubMed]
- Peters, H. The development of the mouse ovary from birth to maturity. Acta Endocrinol 1969, 62, 98–116. [Google Scholar] [CrossRef] [PubMed]
- De Andrés-Aguayo, L.; Varas, F.; Kallin, E.M.; Infante, J.F.; Wurst, W.; Floss, T.; Graf, T. Musashi 2 is a regulator of the hsc compartment identified by a retroviral insertion screen and knockout mice. Blood 2011, 118, 554–564. [Google Scholar] [CrossRef] [PubMed]
- Albertini, D.F.; Combelles, C.M.; Benecchi, E.; Carabatsos, M.J. Cellular basis for paracrine regulation of ovarian follicle development. Reproduction 2001, 121, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Yong, E.L.; Baird, D.T.; Yates, R.; Reichert, L.E., Jr.; Hillier, S.G. Hormonal regulation of the growth and steroidogenic function of human granulosa cells. J. Clin. Endocrinol. Metab. 1992, 74, 842–849. [Google Scholar] [CrossRef] [PubMed]
- Lindner, H.R.; Amsterdam, A.; Salomon, Y.; Tsafriri, A.; Nimrod, A.; Lamprecht, S.A.; Zor, U.; Koch, Y. Intraovarian factors in ovulation: Determinants of follicular response to gonadotrophins. J. Reprod. Fertil. 1977, 51, 215–235. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, R.Q.; Ou, S.B.; Zhang, N.F.; Ren, L.; Wei, L.N.; Zhang, Q.X.; Yang, D.Z. Increased GDF9 and BMP15 mRNA levels in cumulus granulosa cells correlate with oocyte maturation, fertilization, and embryo quality in humans. Reprod. Biol. Endocrinol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.A.; Sciorio, R.; Kinnell, H.; Bayne, R.A.; Thong, K.J.; de Sousa, P.A.; Pickering, S. Cumulus gene expression as a predictor of human oocyte fertilisation, embryo development and competence to establish a pregnancy. Reproduction 2009, 138, 629–637. [Google Scholar] [CrossRef] [PubMed]
- Robker, R.L.; Watson, L.N.; Robertson, S.A.; Dunning, K.R.; McLaughlin, E.A.; Russell, D.L. Identification of sites of STAT3 action in the female reproductive tract through conditional gene deletion. PLoS ONE 2014, 9, e101182. [Google Scholar] [CrossRef] [PubMed]
- Holt, J.E.; Tran, S.M.; Stewart, J.L.; Minahan, K.; Garcia-Higuera, I.; Moreno, S.; Jones, K.T. The APC/C activator FZR1 coordinates the timing of meiotic resumption during prophase i arrest in mammalian oocytes. Development 2011, 138, 905–913. [Google Scholar] [CrossRef] [PubMed]
- Gunter, K.; McLaughlin, E. Translational control in germ cell development: A role for the RNA-binding proteins Musashi-1 and Musashi-2. IUBMB Life 2011, 63, 678–685. [Google Scholar] [CrossRef] [PubMed]
- Venables, J.P.; Eperon, I. The roles of RNA-binding proteins in spermatogenesis and male infertility. Curr. Opin. Genet. Dev. 1999, 9, 346–354. [Google Scholar] [CrossRef]
- Brook, M.; Smith, J.W.; Gray, N.K. The DAZL and PABP families: RNA-binding proteins with interrelated roles in translational control in oocytes. Reproduction 2009, 137, 595–617. [Google Scholar] [CrossRef] [PubMed]
- Stitzel, M.L.; Seydoux, G. Regulation of the oocyte-to-zygote transition. Science 2007, 316, 407–408. [Google Scholar] [CrossRef] [PubMed]
- Detwiler, M.R.; Reuben, M.; Li, X.; Rogers, E.; Lin, R. Two zinc finger proteins, OMA-1 and OMA-2, are redundantly required for oocyte maturation in C. elegans. Dev. Cell 2001, 1, 187–199. [Google Scholar] [CrossRef]
- Spike, C.A.; Coetzee, D.; Nishi, Y.; Guven-Ozkan, T.; Oldenbroek, M.; Yamamoto, I.; Greenstein, D.; Lin, R. Translational control of the oogenic program by components of OMA ribonucleoprotein particles in Caenorhabditis elegans. Genetics 2014, 198, 1513–1533. [Google Scholar] [CrossRef] [PubMed]
- Collier, B.; Gorgoni, B.; Loveridge, C.; Cooke, H.J.; Gray, N.K. The DAZL family proteins are PABP-binding proteins that regulate translation in germ cells. EMBO J. 2005, 24, 2656–2666. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, E.; Barbagallo, F.; Valeri, C.; Geremia, R.; Salustri, A.; de Felici, M.; Sette, C. Ablation of the SAM68 gene impairs female fertility and gonadotropin-dependent follicle development. Hum. Mol. Genet. 2010, 19, 4886–4894. [Google Scholar] [CrossRef] [PubMed]
- Ozturk, S.; Sozen, B.; Demir, N. EPAB and PABPC1 are differentially expressed in the postnatal mouse ovaries. J. Assist. Reprod. Genet. 2015, 32, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Pique, M.; Lopez, J.M.; Foissac, S.; Guigo, R.; Mendez, R. A combinatorial code for CPE-mediated translational control. Cell 2008, 132, 434–448. [Google Scholar] [CrossRef] [PubMed]
- Sobinoff, A.P.; Pye, V.; Nixon, B.; Roman, S.D.; McLaughlin, E.A. Adding insult to injury: Effects of xenobiotic-induced preantral ovotoxicity on ovarian development and oocyte fusibility. Toxicol. Sci. 2010, 118, 653–666. [Google Scholar] [CrossRef] [PubMed]
- Sobinoff, A.; Beckett, E.; Jarnicki, A.; Sutherland, J.; McCluskey, A.; Hansbro, P.; McLaughlin, E. Scrambled and fried: Cigarette smoke exposure causes antral follicle destruction and oocyte dysfunction through oxidative stress. Toxicol. Appl. Pharmacol. 2013, 271, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Nixon, B.; Bielanowicz, A.; Mclaughlin, E.A.; Tanphaichitr, N.; Ensslin, M.A.; Aitken, R.J. Composition and significance of detergent resistant membranes in mouse spermatozoa. J. Cell. Physiol. 2009, 218, 122–134. [Google Scholar] [CrossRef] [PubMed]
- Baleato, R.M.; Aitken, R.J.; Roman, S.D. Vitamin a regulation of BMP4 expression in the male germ line. Dev. Biol. 2005, 286, 78–90. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sutherland, J.M.; Sobinoff, A.P.; Gunter, K.M.; Fraser, B.A.; Pye, V.; Bernstein, I.R.; Boon, E.; Siddall, N.A.; De Andres, L.I.; Hime, G.R.; et al. Knockout of RNA Binding Protein MSI2 Impairs Follicle Development in the Mouse Ovary: Characterization of MSI1 and MSI2 during Folliculogenesis. Biomolecules 2015, 5, 1228-1244. https://doi.org/10.3390/biom5031228
Sutherland JM, Sobinoff AP, Gunter KM, Fraser BA, Pye V, Bernstein IR, Boon E, Siddall NA, De Andres LI, Hime GR, et al. Knockout of RNA Binding Protein MSI2 Impairs Follicle Development in the Mouse Ovary: Characterization of MSI1 and MSI2 during Folliculogenesis. Biomolecules. 2015; 5(3):1228-1244. https://doi.org/10.3390/biom5031228
Chicago/Turabian StyleSutherland, Jessie M., Alexander P. Sobinoff, Kara M. Gunter, Barbara A. Fraser, Victoria Pye, Ilana R. Bernstein, Evan Boon, Nicole A. Siddall, Luisa I. De Andres, Gary R. Hime, and et al. 2015. "Knockout of RNA Binding Protein MSI2 Impairs Follicle Development in the Mouse Ovary: Characterization of MSI1 and MSI2 during Folliculogenesis" Biomolecules 5, no. 3: 1228-1244. https://doi.org/10.3390/biom5031228
APA StyleSutherland, J. M., Sobinoff, A. P., Gunter, K. M., Fraser, B. A., Pye, V., Bernstein, I. R., Boon, E., Siddall, N. A., De Andres, L. I., Hime, G. R., Holt, J. E., Graf, T., & McLaughlin, E. A. (2015). Knockout of RNA Binding Protein MSI2 Impairs Follicle Development in the Mouse Ovary: Characterization of MSI1 and MSI2 during Folliculogenesis. Biomolecules, 5(3), 1228-1244. https://doi.org/10.3390/biom5031228




