Long Chain Fatty Acid Acylated Derivatives of Quercetin-3-O-Glucoside as Antioxidants to Prevent Lipid Oxidation
Abstract
:1. Introduction
2. Material and Methods
2.1. Chemicals and Supplies
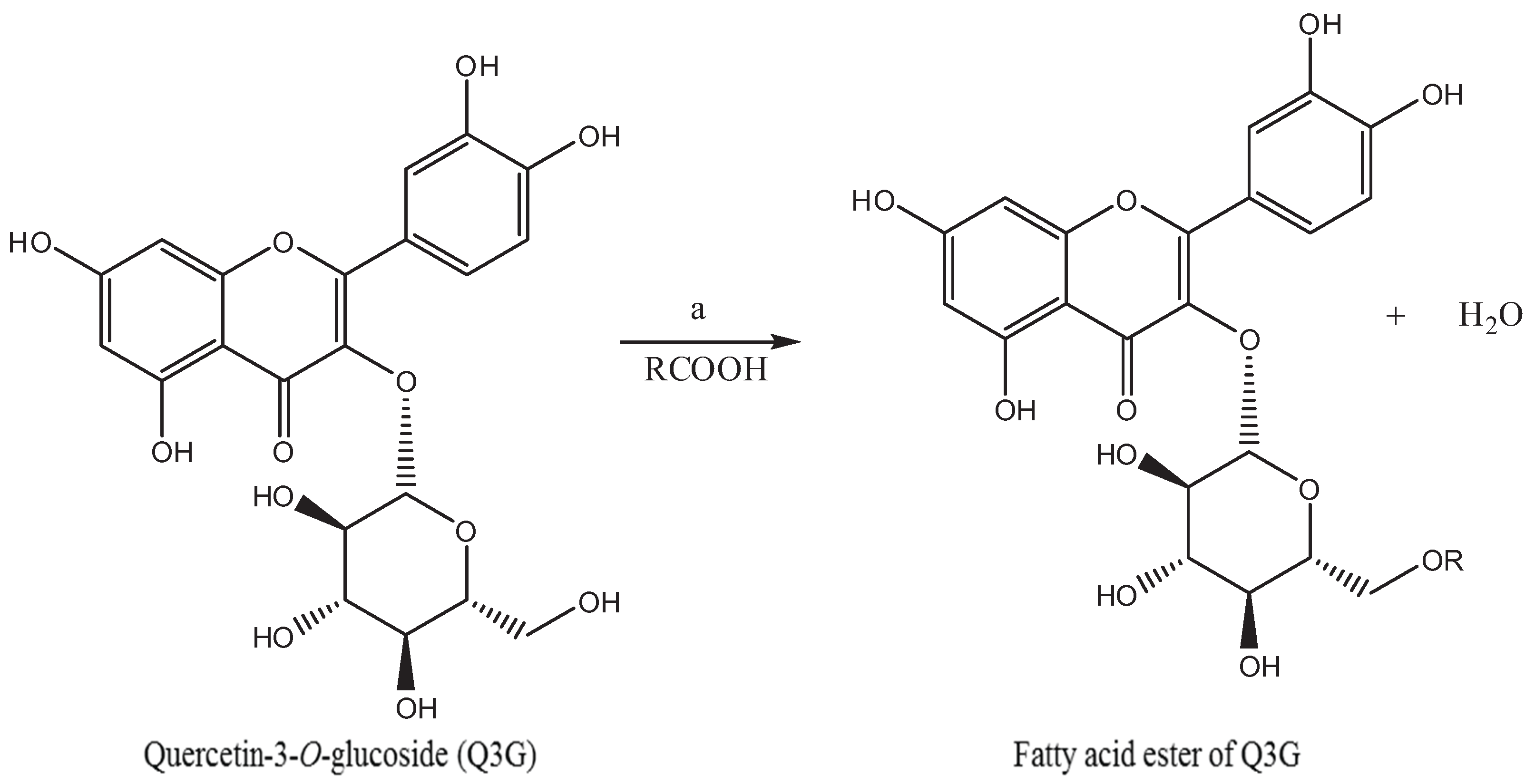
2.2. Synthesis of Fatty Acid Acylated Derivatives of Q3G
2.3. Determination of Primary Oxidation in Bulk Fish Oil Model System
2.4. Preparation of Aqueous Emulsion Model System
2.5. Determination of Primary Oxidation in Aqueous Emulsion Model System
2.6. Determining the Inhibition of Secondary Oxidation
2.7. LDL Oxidation
2.7.1. LDL Preparation
2.7.2. Determination of Protein Content of Dialysed LDL
2.7.3. Inhibition of LDL Oxidation
2.8. Statistical Analysis
3. Results
3.1. Inhibition of Oxidation in Bulk Fish Oil and Aqueous Emulsion
| Compound | % Inhibition (Relative to the Control) | |||||||
|---|---|---|---|---|---|---|---|---|
| Day 3 | Day 5 | |||||||
| Concentration (mmol·L−1) | Concentration (mmol·L−1) | |||||||
| 0.5 | 1 | 5 | 10 | 0.5 | 1 | 5 | 10 | |
| Q3G | 0 ± 0.0c | 0 ± 0.0c | 45 ± 7.1bc | 57 ± 8.7a | 8 ± 1.5D | 12 ± 1.4CD | 34 ± 4.4BC | 52 ± 6.3A |
| Q3G stearate | 0 ± 0.0c | 0 ± 0.0c | 24 ± 2.0bc | 26 ± 4.0bc | 13 ± 6.0CD | 22 ± 2.6CD | 34 ± 2.3BC | 29 ± 3.2BC |
| Q3G oleate | 7 ± 2.8c | 0 ± 0.0c | 14 ± 6.5c | 12 ± 9.3c | 0 ± 0.0D | 6 ± 3.8CD | 26 ± 6.7CD | 30 ± 15.2BC |
| Q3G linoleate | 18 ± 8.4bc | 16 ± 13.4bc | 14 ± 0.2c | 21 ± 2.7bc | 11 ± 1.0CD | 22 ± 3.4CD | 28 ± 3.0C | 26 ± 5.6C |
| Q3G α-linolenate | 14 ± 8.2c | 5 ± 3.5c | 12 ± 6.4c | 17 ± 3.4bc | 5 ± 3.5D | 11 ± 4.8CD | 22 ± 4.1CD | 40 ± 2.7AB |
| Q3G eicosapentaenoate | 18 ± 7.5bc | 21 ± 4.7bc | 34 ± 3.8b | 30 ± 5.0bc | 16 ± 2.6CD | 18 ± 0.5CD | 37 ± 2.1B | 34 ± 1.6BC |
| Q3G docosahexaenoate | 9 ± 7.0c | 17 ± 1.0bc | 26 ± 0.8bc | 21 ± 1.2bc | 19 ± 2.1CD | 21 ± 6.2CD | 30 ± 8.8BC | 38 ± 1.3AB |
| Compound | % Inhibition (Relative to the Control) | |||
|---|---|---|---|---|
| Concentration (mmol·L−1) | ||||
| 0.5 | 1 | 5 | 10 | |
| Q3G | 24 ± 5.8e | 35 ± 2.4d | 60 ± 2.5b | 69 ± 0.1a |
| Q3G stearate | 39 ± 4.4d | 35 ± 1.2d | 46 ± 1.2cd | 52 ± 2.1bc |
| Q3G oleate | 40 ± 1.4cd | 60 ± 0.7b | 64 ± 1.5ab | 67 ± 1.7ab |
| Q3G linoleate | 34 ± 3.0d | 49 ± 3.6c | 63 ± 1.6ab | 70 ± 0.6a |
| Q3G α-linolenate | 38 ± 1.0d | 33 ± 4.4de | 42 ± 0.6cd | 51 ± 0.9bc |
| Q3G eicosapentaenoate | 46 ± 2.6cd | 40 ± 1.9cd | 50 ± 4.1bc | 31 ± 1.1de |
| Q3G docosahexaenoate | 50 ± 4.2cd | 50 ± 3.5bc | 47 ± 6.4cd | 33 ± 2.5de |
| Compound | % Inhibition (Relative to the Control) | |||||||
|---|---|---|---|---|---|---|---|---|
| Primary Oxidation | Secondary Oxidation | |||||||
| Concentration (mmol·L−1) | Concentration (mmol·L−1) | |||||||
| 0.5 | 1 | 5 | 10 | 0.5 | 1 | 5 | 10 | |
| Q3G | 29 ± 6.7c | 39 ± 9.4bc | 42 ± 9.1bc | 18 ± 15.3c | 0 ± 0.0F | 0 ± 0.0F | 9 ± 2.3EF | 25 ± 1.0DE |
| Q3G stearate | 55 ± 6.1bc | 74 ± 7.1ab | 100 ± 16.9a | 100 ± 3.6a | 16 ± 2.5E | 20 ± 5.2DE | 69 ± 2.3AB | 78 ± 3.1A |
| Q3G oleate | 53 ± 8.5bc | 56 ± 8.8bc | 55 ± 4.1bc | 63 ± 6.0bc | 11 ± 3.4EF | 30 ± 4.1D | 43 ± 9.7CD | 49 ± 14.4C |
| Q3G linoleate | 47 ± 2.2bc | 66 ± 0.6b | 60 ± 8.1bc | 73 ± 4.0ab | 0 ± 0.0F | 3 ± 1.0F | 45 ± 2.0CD | 67 ± 0.3AB |
| Q3G α-linolenate | 13 ± 6.7bc | 15 ± 0.1c | 34 ± 4.0c | 64 ± 3.0c | 12 ± 1.4EF | 24 ± 6.5DE | 51 ± 1.3BC | 63 ± 0.6B |
| Q3G eicosapentaenoate | 42 ± 3.2bc | 56 ± 5.4bc | 63 ± 17.3bc | 65 ± 12.2bc | 0 ± 0.0F | 0 ± 0.0F | 13 ± 1.9EF | 32 ± 1.8D |
| Q3G docosahexaenoate | 49 ± 5.0bc | 43 ± 4.0bc | 54 ± 19.3bc | 100 ± 8.4a | 27 ± 9.5DE | 26 ± 3.8DE | 53 ± 0.8BC | 58 ± 8.0BC |
| Compound | % Inhibition (Relative to the Control) | |||||||
|---|---|---|---|---|---|---|---|---|
| Cu2+-Induction | AAPH Derived Peroxyl Radical–Induction | |||||||
| Concentration (µmol·L−1) | Concentration (µmol·L−1) | |||||||
| 1 | 10 | 100 | 500 | 1 | 10 | 100 | 500 | |
| Q3G | 7 ± 4.5c | 20 ± 9.2bc | 20 ± 1.8bc | 33 ± 8.6ab | 10 ± 2.6C | 10 ± 2.7C | 16 ± 7.2C | 29 ± 2.0C |
| Q3G stearate | 4 ± 9.4c | 14 ± 7.5bc | 21 ± 8.6bc | 29 ± 4.2b | 4 ± 5.9C | 11 ± 5.0C | 14 ± 7.5C | 27 ± 8.5C |
| Q3G oleate | 8 ± 7.4c | 18 ± 5.8bc | 14 ± 5.3bc | 28 ± 2.8bc | 12 ± 7.2C | 9 ± 2.8C | 15 ± 3.9C | 17 ± 13.6C |
| Q3G linoleate | 14 ± 3.4bc | 27 ± 7.8bc | 29 ± 5.9b | 34 ± 6.3ab | 5 ± 4.7C | 15 ± 7.8C | 23 ± 5.6C | 25 ± 5.4C |
| Q3G α-linolenate | 40 ± 5.6ab | 34 ± 5.1ab | 31 ± 2.9ab | 31 ± 3.5ab | 28 ± 9.5AB | 23 ± 4.3AB | 23 ± 2.0AB | 34 ± 6.3AB |
| Q3G eicosapentaenoate | 20 ± 8.8bc | 43 ± 6.8ab | 45 ± 3.1ab | 51 ± 2.6a | 21 ± 3.8A | 36 ± 6.7A | 38 ± 1.7A | 41 ± 2.9A |
| Q3G docosahexaenoate | 7 ± 5.5c | 14 ± 7.9bc | 27 ± 6.7bc | 43 ± 1.4ab | 13 ± 3.7BC | 9 ± 8.1BC | 30 ± 10.0BC | 27 ± 8.1BC |
3.2. Inhibition of Oxidation in Human LDL
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Abbreviations
| STA | Stearic acid (18:0) |
| OLA | Oleic acid (18:1n-9) |
| LNA | Linoleic acid (18:2n-6) |
| ALA | Alpha-linolenic acid (18:3n-3) |
| EPA | Eicosapentaenoic acid (20:5n-3) |
| DHA | Docosahexaenoic acid (22:6n-3) |
| PUFA | Polyunsaturated fatty acid(s) |
| AAPH | 2,2'-Azobis(2-amidinopropane) dihydrochloride |
| BHT | Butylated hydroxytoluene |
| LDL | Low density lipoprotein |
| Q3G | Quercetin-3-O-glucoside |
| TBA | 2-Thiobarbituric acid |
| TBARS | Thiobarbituric acid reactive substances |
| TCA | Trichloroacetic acid |
Conflicts of Interest
References
- Russo, G.L. Dietary n-6 and n-3 polyunsaturated fatty acids: From biochemistry to clinical implications in cardiovascular prevention. Biochem. Pharmacol. 2009, 77, 937–946. [Google Scholar]
- Sun, Y.; Wang, W.; Chen, H.; Li, C. Autoxidation of unsaturated lipids in food emulsion. Crit. Rev. Food Sci. Nutr. 2011, 5, 453–466. [Google Scholar] [CrossRef]
- Kathirvel, P.; Rupasinghe, H.P.V. Plant-derived antioxidants as potential omega-3 PUFA stabilizers. In Fish Oil: Production, Consumption and Health Benefits; Dijk, M.V., Vitel, J., Eds.; Nova Science Publishers USA: Hauppauge, NY, USA, 2011; pp. 143–154. [Google Scholar]
- Kanner, J. Dietary advance lipid oxidation end products are risk factors to human health. Mol. Nutr. Food Res. 2007, 51, 1094–1101. [Google Scholar] [CrossRef] [PubMed]
- Katsube, T.; Imawaka, N.; Kawano, Y.; Yamazaki, Y.; Shiwaku, L.; Yamane, Y. Antioxidant flavonol glycosides in mulberry (Morus alba L.) leaves isolated based on LDL antioxidant activity. Food Chem. 2006, 97, 25–31. [Google Scholar] [CrossRef]
- Chalas, J.; Claise, C.; Edeas, M.; Messaoudi, C.; Vergnes, L.; Abella, A.; Lindenbaum, A. Effect of ethyl esterification of phenolic acids on low-density lipoprotein oxidation. Biomed. Pharmacother. 2001, 55, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Salem, J.H.; Chevalot, I.; Harscoat-Schiavo, C.; Paris, C.; Fick, M.; Humeau, C. Biological activities of flavonoids from Nitraria retusa (Forssk.) Asch. and their acylated derivatives. Food Chem. 2011, 124, 486–494. [Google Scholar] [CrossRef]
- Lue, B.M.; Guo, Z.; Xu, X. Effect of room temperature ionic liquid structure on the enzymatic acylation of flavonoids. Process Biochem. 2010, 45, 1375–1382. [Google Scholar] [CrossRef]
- Montenegro, L.; Carbone, C.; Maniscalco, C.; Lambusta, D.; Nicolosi, G.; Ventura, C.A.; Puglisi, G. In vitro evaluation of quercetin-3-O-acyl esters as topical prodrugs. Int. J. Pharm. 2007, 336, 257–262. [Google Scholar] [CrossRef]
- Mellou, F.; Lazari, D.; Skattsa, H.; Tselepis, A.D.; Kolisis, F.N.; Stamatis, H. Biocatalytic preparation of acylated derivatives of flavonoid glycosides enhances their antioxidant and antimicrobial activity. J. Biotechnol. 2005, 116, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Laguerre, M.; Bayrasy, C.; Lecomte, J.; Chabi, B.; Decker, E.A.; Wrutniak-Cabello, C.; Cabello, G.; Villeneuve, P. How to boost antioxidants by lipophilization? Biochimie 2013, 95, 20–26. [Google Scholar] [CrossRef]
- Chebil, L.; Humeau, C.; Falcimaigne, A.; Engasser, J.M.; Ghoul, M. Review: Enzymatic acylation of flavonoids. Process Biochem. 2006, 41, 2237–2251. [Google Scholar] [CrossRef]
- Ardhaoui, M.; Falcimaigne, A.; Ognier, S.; Engasser, J.M.; Moussou, P.; Pauly, G.; Ghoul, M. Effect of acyl donor chain length and substitutions pattern on the enzymatic acylation of flavonoids. J. Biotechnol. 2004, 110, 256–271. [Google Scholar] [CrossRef]
- Ziaullah; Bhullar, K.S.; Warnakulasuriya, S.N.; Rupasinghe, H.P.V. Biocatalytic synthesis, structural elucidation, antioxidant capacity and tyrosinase inhibition activity of long chain fatty acid acylated derivatives of phloridzin and isoquercitrin. Bioorg. Med. Chem. 2013, 21, 684–692. [Google Scholar] [CrossRef]
- Official Methods and Recommended Practices of the American Oil Chemists’ Society Method Cd 8–53; Firestone, D. (Ed.) American Oil Chemists’ Society Champaign: Urbana, IL, USA, 1997.
- Rupasinghe, H.P.V.; Erkan, N.; Yasmin, A. Antioxidant protection of eicosapentaenoic acid and fish oil oxidation by polyphenolic-enriched apple skin extract. J. Agric. Food Chem. 2010, 58, 1233–1239. [Google Scholar] [CrossRef]
- Huber, G.M.; Rupasinghe, H.P.V.; Shahidi, F. Inhibition of oxidation of omega-3 polyunsaturated fatty acids and fish oil by quercetin glycosides. Food Chem. 2009, 117, 290–295. [Google Scholar] [CrossRef]
- Thilakarathna, S.H.; Rupasinghe, H.P.V. Anti-atherosclerotic effects of fruit bioactive compounds: A review of current scientific evidence. Can. J. Plant Sci. 2012, 92, 407–419. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 165–275. [Google Scholar]
- Xu, B.J.; Yuan, S.H.; Chang, S.K.C. Comparative studies on the antioxidant activities of nine common food legumes against copper induced LDL oxidation in vitro. J. Food Sci. 2007, 72, 522–527. [Google Scholar] [CrossRef]
- Danihelova, M.; Viskupičova, J.; Šturdika, E. Lipophilization of flavonoids for their food, therapeutic and cosmetic applications. Acta Chim. Slovaca 2012, 5, 59–69. [Google Scholar]
- Passicos, E.; Santarelli, X.; Coulon, D. Regioselective acylation of flavonoids catalyzed by immobilized Candida antarctica lipase under reduced pressure. Biotechnol. Lett. 2004, 26, 1073–1076. [Google Scholar] [CrossRef]
- Lorentz, C.; Dulac, A.; Pencreach, G.; Ergan, F.; Richomme, P.; Soultani-Vigneron, S. Lipase-catalyzed synthesis of two new antioxidants: 4-O- and 3-O-palmitoyl chlorogenic acids. Biotechnol. Lett. 2010, 32, 1955–1960. [Google Scholar] [CrossRef] [PubMed]
- Chebil, L.; Anthoni, J.; Humeau, C.; Gerardin, C.; Engasser, J.M.; Goul, M. Enzymatic acylation of flavonoids: Effect of the nature of the substrate, origin of lipase and operating conditions on conversion yield and regioselectivity. J. Agric. Food Chem. 2007, 55, 9496–9502. [Google Scholar] [CrossRef] [PubMed]
- Silva, F.A.; Borges, F.; Guimarães, C.; Lima, J.L.; Matos, C.; Reis, S. Phenolic acids and derivatives: Studies on the relationship among structure, radical scavenging activity, and physicochemical parameters. J. Agric. Food Chem. 2000, 48, 2122–2126. [Google Scholar] [CrossRef] [PubMed]
- Viskupicova, J.; Danihelova, M.; Ondrejovic, M.; Liptaj, T.; Sturdik, E. Lipophilic rutin derivatives for antioxidant protection of oil-based foods. Food Chem. 2010, 123, 45–55. [Google Scholar] [CrossRef]
- Sharma, O.P.; Bhat, T.K. DPPH antioxidant assay revisited. Food Chem. 2009, 113, 1202–1205. [Google Scholar] [CrossRef]
- Porter, W.L. Recent trends in food applications of antioxidants. In Autoxidation in Food and Biological Systems; Simic, M.G., Karel, M., Eds.; Plenum Press: New York, NY, USA, 1980; pp. 295–365. [Google Scholar]
- Frankel, E.N. Antioxidants in lipid foods and their impact on food quality. Food Chem. 1996, 57, 51–55. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Revisiting the polar paradox theory: A critical overview. J. Agric. Food Chem. 2011, 59, 3499–3504. [Google Scholar] [CrossRef] [PubMed]
- Chaiyasit, W.; Elias, R.J.; McClements, D.J.; Decker, E.A. Role of physical structures in bulk oils on lipid oxidation. Crit. Rev. Food Sci. Nutr. 2007, 47, 299–317. [Google Scholar] [CrossRef]
- Mateos, R.; Trujillo, M.; Pereira-Caro, G.; Madrona, A.; Cert, A.; Espartero, J.L. New lipophilic tyrosyl esters: Comparative antioxidant evaluation with hydroxytyrosyl esters. J. Agric. Food Chem. 2008, 56, 10960–10966. [Google Scholar] [CrossRef] [PubMed]
- Frankel, E.N.; Kanner, J.; German, J.B.; Parks, E.; Kinsella, J.E. Inhibition of oxidation of human low-density lipoprotein by phenolic substances in red wine. Lancet 1993, 341, 454–457. [Google Scholar] [CrossRef]
- Safari, M.; Sheikh, N. Effects of flavonoids on the susceptibility of low-density lipoprotein to oxidative modification. Prostaglandins Leukot. Essent. Fatty Acids 2003, 69, 73–77. [Google Scholar] [CrossRef]
- Thilakarathna, S.H.; Rupasinghe, H.P.V.; Needs, P.W. Apple peel bioactive rich extracts effectively inhibit in vitro human LDL oxidation. Food Chem. 2013, 138, 463–470. [Google Scholar] [CrossRef]
- Miyagi, Y.; Miwa, K.; Inoue, H. Inhibition of human low-density lipoprotein oxidation by flavonoids in red wine and grape juice. Am. J. Cardiol. 1997, 80, 1627–1631. [Google Scholar] [CrossRef]
- Kerry, N.; Abbey, M. Red wine and fractionated phenolic compounds prepared from red wine inhibit low density lipoprotein oxidation in vitro. Atherosclerosis 1997, 135, 93–102. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Warnakulasuriya, S.N.; Ziaullah; Rupasinghe, H.P.V. Long Chain Fatty Acid Acylated Derivatives of Quercetin-3-O-Glucoside as Antioxidants to Prevent Lipid Oxidation. Biomolecules 2014, 4, 980-993. https://doi.org/10.3390/biom4040980
Warnakulasuriya SN, Ziaullah, Rupasinghe HPV. Long Chain Fatty Acid Acylated Derivatives of Quercetin-3-O-Glucoside as Antioxidants to Prevent Lipid Oxidation. Biomolecules. 2014; 4(4):980-993. https://doi.org/10.3390/biom4040980
Chicago/Turabian StyleWarnakulasuriya, Sumudu N., Ziaullah, and H. P. Vasantha Rupasinghe. 2014. "Long Chain Fatty Acid Acylated Derivatives of Quercetin-3-O-Glucoside as Antioxidants to Prevent Lipid Oxidation" Biomolecules 4, no. 4: 980-993. https://doi.org/10.3390/biom4040980
APA StyleWarnakulasuriya, S. N., Ziaullah, & Rupasinghe, H. P. V. (2014). Long Chain Fatty Acid Acylated Derivatives of Quercetin-3-O-Glucoside as Antioxidants to Prevent Lipid Oxidation. Biomolecules, 4(4), 980-993. https://doi.org/10.3390/biom4040980






