Scutellaria baicalensis Georgi: A Promising Source of Bioactive Molecules for Kidney Disease Therapy
Abstract
1. Introduction
2. Main Components of S. baicalensis for Renoprotective Effects
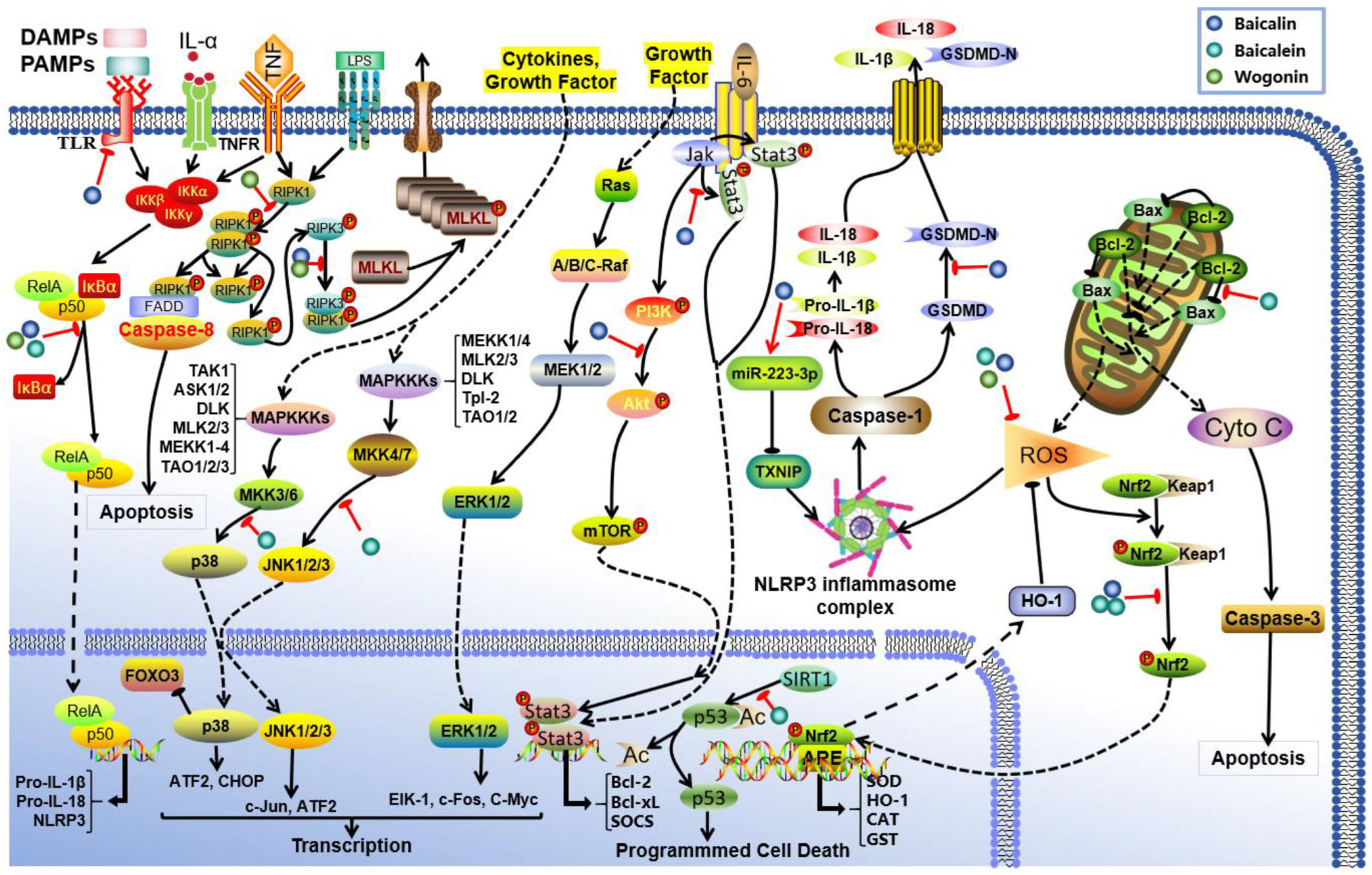
3. Effects and Mechanisms of S. baicalensis Against AKI
4. Effects and Mechanism of S. baicalensis Against CKD
4.1. Diabetic Kidney Disease (DKD)
4.2. Hyperuricemia
4.3. Renal Fibrosis (RF)
4.4. Renal Cell Carcinoma
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AKI | Acute kidney injury |
| ALOX12 | Arachidonate 12-lipoxygenase |
| CAT | Catalase |
| CKD | Chronic kidney disease |
| DKD | Diabetic kidney disease |
| DM | Diabetes mellitus |
| ECM | Extracellular matrix |
| ESRD | End-stage renal disease |
| HUA | Hyperuricemia |
| 12/15-LO | 12/15-Lipoxygenase |
| MDA | Malondialdehyde |
| RCC | Renal cell carcinoma |
| RF | Renal fibrosis |
| S. baicalensis | Scutellaria baicalensis |
| SOD | Superoxide dismutase |
| VEGF | Vascular endothelial growth factor |
| α-SMA | α-Smooth muscle actin |
References
- Menon, S.; Symons, J.M.; Selewski, D.T. Acute kidney injury. Pediatr. Rev. 2023, 44, 265–279. [Google Scholar] [CrossRef] [PubMed]
- Jager, K.J.; Kovesdy, C.; Langham, R.; Rosenberg, M.; Jha, V.; Zoccali, C. A single number for advocacy and communication-worldwide more than 850 million individuals have kidney diseases. Nephrol. Dial. Transplant. 2019, 34, 1803–1805. [Google Scholar] [CrossRef] [PubMed]
- Hall, R.K.; Kazancıoğlu, R.; Thanachayanont, T.; Wong, G.; Sabanayagam, D.; Battistella, M.; Ahmed, S.B.; Inker, L.A.; Barreto, E.F.; Fu, E.L.; et al. Drug stewardship in chronic kidney disease to achieve effective and safe medication use. Nat. Rev. Nephrol. 2024, 20, 386–401. [Google Scholar] [CrossRef]
- Hu, H.; Li, W.; Hao, Y.; Peng, Z.; Zou, Z.; Wei, J.; Zhou, Y.; Liang, W.; Cao, Y. The SGLT2 inhibitor dapagliflozin ameliorates renal fibrosis in hyperuricemic nephropathy. Cell Rep. Med. 2024, 5, 101690. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, N.; Dobre, M. Emerging preventive strategies in chronic kidney disease: Recent evidence and gaps in knowledge. Curr. Atheroscler. Rep. 2023, 25, 1047–1058. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, G.B.; Cederholm, T.; Avesani, C.M.; Bakker, S.J.L.; Bellizzi, V.; Cuerda, C.; Cupisti, A.; Sabatino, A.; Schneider, S.; Torreggiani, M.; et al. Nutritional status and the risk of malnutrition in older adults with chronic kidney disease-implications for low protein intake and nutritional care: A critical review endorsed by ERN-ERA and ESPEN. Clin. Nutr. 2023, 42, 443–457. [Google Scholar] [CrossRef]
- Ostermann, M.; Lumlertgul, N.; Jeong, R.; See, E.; Joannidis, M.; James, M. Acute kidney injury. Lancet 2025, 405, 241–256. [Google Scholar] [CrossRef]
- Baker, M.; Perazella, M.A. NSAIDs in CKD: Are they safe? Am. J. Kidney Dis. 2020, 76, 546–557. [Google Scholar] [CrossRef]
- Alicic, R.Z.; Neumiller, J.J.; Tuttle, K.R. Combination therapy: An upcoming paradigm to improve kidney and cardiovascular outcomes in chronic kidney disease. Nephrol. Dial. Transplant. 2025, 40, i3–i17. [Google Scholar] [CrossRef]
- Ahmadi, F.; Hwang, Y.J.; Muanda, F.T. Pros and cons of the use of fluoroquinolone antibiotics in patients with kidney disease. Nephrol. Dial. Transplant. 2024, 39, 1205–1209. [Google Scholar] [CrossRef]
- Chade, A.R.; Bidwell, G.L. Novel drug delivery technologies and targets for renal disease. Hypertension 2022, 79, 1937–1948. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, S.; Ives, N.; Brettell, E.A.; Valente, M.; Cockwell, P.; Topham, P.S.; Cleland, J.G.; Khwaja, A.; El Nahas, M. Multicentre randomized controlled trial of angiotensin-converting enzyme inhibitor/angiotensin receptor blocker withdrawal in advanced renal disease: The STOP-ACEi trial. Nephrol. Dial. Transpl. 2016, 31, 255–261. [Google Scholar] [CrossRef]
- Perazella, M.A. Pharmacology behind common drug nephrotoxicities. Clin. J. Am. Soc. Nephrol. 2018, 13, 1897–1908. [Google Scholar] [CrossRef] [PubMed]
- Jordan, M.A.; Wilson, L. Microtubules as a target for anticancer drugs. Nat. Rev. Cancer 2004, 4, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, H.; Colby, D.A.; Seto, S.; Va, P.; Tam, A.; Kakei, H.; Rayl, T.J.; Hwang, I.; Boger, D.L. Total synthesis of vinblastine, vincristine, related natural products, and key structural analogues. J. Am. Chem. Soc. 2009, 31, 4904–4916. [Google Scholar] [CrossRef]
- Olatunde, O.Z.; Yong, J.; Lu, C.; Ming, Y. A review on shikonin and its derivatives as potent anticancer agents targeted against topoisomerases. Curr. Med. Chem. 2023, 31, 920–937. [Google Scholar] [CrossRef]
- Tu, Y. The discovery of artemisinin (qinghaosu) and gifts from Chinese medicine. Nat. Med. 2011, 17, 1217–1220. [Google Scholar] [CrossRef]
- Chuk, M.K.; Balis, F.M.; Fox, E. Trabectedin. Oncologist 2009, 14, 794–799. [Google Scholar] [CrossRef]
- Zhao, Q.; Chen, X.Y.; Martin, C. Scutellaria baicalensis, the golden herb from the garden of chinese medicinal plants. Sci. Bull. 2016, 61, 1391–1398. [Google Scholar] [CrossRef]
- Hou, F.; Yu, Z.; Cheng, Y.; Liu, Y.; Liang, S.; Zhang, F. Deciphering the pharmacological mechanisms of Scutellaria baicalensis Georgi on oral leukoplakia by combining network pharmacology, molecular docking and experimental evaluations. Phytomedicine 2022, 103, 154195. [Google Scholar] [CrossRef]
- Pietta, P.G. Flavonoids as antioxidants. J. Nat. Prod. 2000, 63, 1035–1042. [Google Scholar] [CrossRef] [PubMed]
- Havsteen, B.H. The biochemistry and medical significance of the flavonoids. Pharmacol. Ther. 2002, 96, 67–202. [Google Scholar] [CrossRef] [PubMed]
- Heim, K.E.; Tagliaferro, A.R.; Bobilya, D.J. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Biochem. 2002, 13, 572–584. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.P.; Son, K.H.; Chang, H.W.; Kang, S.S. Anti-inflammatory plant flavonoids and cellular action mechanisms. J. Pharmacol. Sci. 2004, 96, 229–245. [Google Scholar] [CrossRef]
- Takasato, M.; Er, P.X.; Chiu, H.S.; Maier, B.; Baillie, G.J.; Ferguson, C.; Parton, R.G.; Wolvetang, E.J.; Roost, M.S.; Chuva de Sousa Lopes, S.M.; et al. Kidney organoids from human iPS cells contain multiple lineages and model human nephrogenesis. Nature 2015, 526, 564–568. [Google Scholar] [CrossRef]
- Khwaja, A. KDIGO clinical practice guidelines for acute kidney injury. Nephron. Clin. Pract. 2012, 120, c179–c184. [Google Scholar] [CrossRef]
- Jacob, J.; Dannenhoffer, J.; Rutter, A. Acute kidney injury. Prim. Care 2020, 47, 571–584. [Google Scholar] [CrossRef]
- Thadhani, R.; Pascual, M.; Bonventre, J.V. Acute renal failure. N. Engl. J. Med. 1996, 334, 1448–1460. [Google Scholar] [CrossRef]
- Adiyeke, E.; Ren, Y.; Guan, Z.; Ruppert, M.M.; Rashidi, P.; Bihorac, A.; Ozrazgat-Baslanti, T. Clinical courses of acute kidney injury in hospitalized patients: A multistate analysis. Sci. Rep. 2023, 13, 17781. [Google Scholar] [CrossRef]
- Molitoris, B.A. Low-flow acute kidney injury: The pathophysiology of prerenal azotemia, abdominal compartment syndrome, and obstructive uropathy. Clin. J. Am. Soc. Nephrol. 2022, 17, 1039–1049. [Google Scholar] [CrossRef]
- Hoste, E.A.; Bagshaw, S.M.; Bellomo, R.; Cely, C.M.; Colman, R.; Cruz, D.N.; Edipidis, K.; Forni, L.G.; Gomersall, C.D.; Govil, D.; et al. Epidemiology of acute kidney injury in critically ill patients: The multinational AKI-EPI study. Intensive Care Med. 2015, 41, 1411–1423. [Google Scholar] [CrossRef] [PubMed]
- Perazella, M.A. Drug-induced acute kidney injury: Diverse mechanisms of tubular injury. Curr. Opin. Crit. Care 2019, 25, 550–557. [Google Scholar] [CrossRef] [PubMed]
- Perazella, M.A.; Rosner, M.H. Drug-induced acute kidney injury. Clin. J. Am. Soc. Nephrol. 2022, 17, 1220–1233. [Google Scholar] [CrossRef] [PubMed]
- Kellum, J.A.; Romagnani, P.; Ashuntantang, G.; Ronco, C.; Zarbock, A.; Anders, H.J. Acute kidney injury. Nat. Rev. Dis. Primers 2021, 7, 52. [Google Scholar] [CrossRef]
- Webster, A.C.; Nagler, E.V.; Morton, R.L.; Masson, P. Chronic kidney disease. Lancet 2017, 389, 1238–1252. [Google Scholar] [CrossRef]
- Law, J.P.; Pickup, L.; Pavlovic, D.; Townend, J.N.; Ferro, C.J. Hypertension and cardiomyopathy associated with chronic kidney disease: Epidemiology, pathogenesis and treatment considerations. J. Hum. Hypertens. 2023, 37, 1–19. [Google Scholar] [CrossRef]
- Andrassy, K.M. Comments on ‘kdigo 2012 clinical practice guideline for the evaluation and management of chronic kidney disease’. Kidney Int. 2013, 84, 622–623. [Google Scholar] [CrossRef]
- Stevens, P.E.; Levin, A. Evaluation and management of chronic kidney disease: Synopsis of the kidney disease: Improving global outcomes 2012 clinical practice guideline. Ann. Intern. Med. 2013, 158, 825–830. [Google Scholar] [CrossRef]
- Levey, A.S.; Eckardt, K.U.; Dorman, N.M.; Christiansen, S.L.; Hoorn, E.J.; Ingelfinger, J.R.; Inker, L.A.; Levin, A.; Mehrotra, R.; Palevsky, P.M.; et al. Nomenclature for kidney function and disease: Report of a kidney disease: Improving global outcomes (KDIGO) consensus conference. Kidney Int. 2020, 97, 1117–1129. [Google Scholar] [CrossRef]
- Chawla, L.S.; Bellomo, R.; Bihorac, A.; Goldstein, S.L.; Siew, E.D.; Bagshaw, S.M.; Bittleman, D.; Cruz, D.; Endre, Z.; Fitzgerald, R.L.; et al. Acute kidney disease and renal recovery: Consensus report of the acute disease quality initiative (ADQI) 16 workgroup. Nat. Rev. Nephrol. 2017, 13, 241–257. [Google Scholar] [CrossRef]
- Neyra, J.A.; Chawla, L.S. Acute kidney disease to chronic kidney disease. Crit. Care Clin. 2021, 37, 453–474. [Google Scholar] [CrossRef] [PubMed]
- Chawla, L.S.; Eggers, P.W.; Star, R.A.; Kimmel, P.L. Acute kidney injury and chronic kidney disease as interconnected syndromes. N. Engl. J. Med. 2014, 371, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.L.; Wang, S.; Kuang, Y.; Hu, Z.M.; Qiao, X.; Ye, M. A comprehensive review on phytochemistry, pharmacology, and flavonoid biosynthesis of Scutellaria baicalensis. Pharm. Biol. 2018, 56, 465–484. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.Q.; Liang, H.; Lu, X.H.; Cai, S.Q.; Wang, B.; Zhao, Y.Y. Flavonoids from Scutellaria baicalensis and their bioactivities. J. Peking Univ. Health Sci. 2009, 41, 578–584. [Google Scholar]
- Zhao, T.; Tang, H.; Xie, L.; Zheng, Y.; Ma, Z.; Sun, Q.; Li, X. Scutellaria baicalensis Georgi. (Lamiaceae): A review of its traditional uses, botany, phytochemistry, pharmacology and toxicology. J. Pharm. Pharmacol. 2019, 71, 1353–1369. [Google Scholar] [CrossRef]
- Song, J.W.; Long, J.Y.; Xie, L.; Zhang, L.L.; Xie, Q.X.; Chen, H.J.; Deng, M.; Li, X.F. Applications, phytochemistry, pharmacological effects, pharmacokinetics, toxicity of Scutellaria baicalensis Georgi. and its probably potential therapeutic effects on COVID-19: A review. Chin. Med. 2020, 15, 102. [Google Scholar] [CrossRef]
- Wen, H.Z.; Xiao, S.Y.; Wang, Y.M.; Luo, G.A. General situation of chemical constitutions anddrug-processing of Scutellaria baicalensis Georgi. Nat. Prod. Res. Dev. 2004, 16, 575–580. [Google Scholar]
- Ma, W.; Liu, T.; Ogaji, O.D.; Li, J.; Du, K.; Chang, Y. Recent advances in Scutellariae radix: A comprehensive review on ethnobotanical uses, processing, phytochemistry, pharmacological effects, quality control and influence factors of biosynthesis. Heliyon 2024, 10, e36146. [Google Scholar] [CrossRef]
- Yan, J.; Zhang, S.F.; Mao, X.X.; Xu, C.X.; Liu, B. Optimization of extraction process of flavonoids from Scutellaria baicalensis stem and leaf by response surface methodology and its content determination. Hubei Agric. Sci. 2024, 23, 131–137. [Google Scholar]
- Jiang, J.J.; Dong, H.R. Preparation and characterization of high-purity baicalein. J. Beijing Univ. Chem. Technol. 2008, 25, 31–34. [Google Scholar]
- Yu, H.; Chang, J.S.; Kim, S.Y.; Kim, Y.G.; Choi, H.K. Enhancement of solubility and dissolution rate of baicalein, wogonin and oroxylin A extracted from Radix scutellariae. Int. J. Pharm. 2017, 528, 602–610. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, J.; Zou, L.; Tang, J.; Zheng, J.; Luo, M.; Wang, G.; Liang, D.; Li, Y.; Chen, B.; et al. Preparation, characterization, and staphylococcus aureus biofilm elimination effect of baicalein-loaded β-cyclodextrin-grafted chitosan nanoparticles. Int. J. Nanomed. 2022, 17, 5287–5302. [Google Scholar] [CrossRef]
- Liu, Y.; Ma, Y.; Xu, J.; Chen, Y.; Xie, J.; Yue, P.; Zheng, Q.; Yang, M. Apolipoproteins adsorption and brain-targeting evaluation of baicalin nanocrystals modified by combination of Tween 80 and TPGS. Colloids Surf. B Biointerfaces 2017, 160, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Liang, J.; Zheng, X.; Pi, C.; Liu, H.; Yang, H.; Zou, Y.; Ye, Y.; Zhao, L. Lung-targeting drug delivery system of baicalin-loaded nanoliposomes: Development, biodistribution in rabbits, and pharmacodynamics in nude mice bearing orthotopic human lung cancer. Int. J. Nanomed. 2017, 12, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Bi, Y.; Wu, H. Formulation optimization and the absorption mechanisms of nanoemulsion in improving baicalin oral exposure. Drug Dev. Ind. Pharm. 2018, 44, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Wang, F.; Wang, X.; Zhang, D.; Bi, Y.; Gao, Y.; Zhao, X.; Zhang, Q. Development and optimization of baicalin-loaded solid lipid nanoparticles prepared by coacervation method using central composite design. Eur. J. Pharm. Sci. 2012, 47, 497–505. [Google Scholar] [CrossRef]
- Basile, D.P.; Anderson, M.D.; Sutton, T.A. Pathophysiology of acute kidney injury. Compr. Physiol. 2012, 2, 1303–1353. [Google Scholar] [CrossRef]
- Hoste, E.A.J.; Kellum, J.A.; Selby, N.M.; Zarbock, A.; Palevsky, P.M.; Bagshaw, S.M.; Goldstein, S.L.; Cerdá, J.; Chawla, L.S. Global epidemiology and outcomes of acute kidney injury. Nat. Rev. Nephrol. 2018, 14, 607–625. [Google Scholar] [CrossRef]
- Sun, Y.; Liu, M.W.; Zhao, Y.H.; Lu, Y.X.; Wang, Y.A.; Tong, C.W. Baicalin attenuates lipopolysaccharide-induced renal tubular epithelial cell injury by inhibiting the TXNIP/NLRP3 signalling pathway via increasing miR-223-3p expression. J. Biol. Regul. Homeost. Agents 2020, 34, 69–82. [Google Scholar] [CrossRef]
- Shi, J.; Wu, G.; Zou, X.; Jiang, K. Enteral baicalin, a flavone glycoside, reduces indicators of cardiac surgery-associated acute kidney injury in rats. Cardiorenal Med. 2019, 9, 31–40. [Google Scholar] [CrossRef]
- Li, Q.H.; Zhang, Y.L.; Xie, X.J.; Guo, H.X.; Wang, X.Y. Propofol’s protective effect enhancement by baicalin mediated activation of Nrf2/HO-1 pathway on renal ischemia-reperfusion injury rats. Acta Chin. Med. 2021, 36, 1704–1710. [Google Scholar]
- Lin, M.; Li, L.; Zhang, Y.; Zheng, L.; Xu, M.; Rong, R.; Zhu, T. Baicalin ameliorates H2O2 induced cytotoxicity in HK-2 cells through the inhibition of ER stress and the activation of Nrf2 signaling. Int. J. Mol. Sci. 2014, 15, 12507–12522. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, X.; Yan, C.; Xie, L.; Yang, Y. Baicalin exhibits a protective effect against cisplatin-induced cytotoxic damage in canine renal tubular epithelial cells. Metabolites 2023, 13, 1173. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Li, S.; Jiang, L.; Gao, X.; Liu, W.; Zhu, X.; Huang, W.; Zhao, H.; Wei, Z.; Wang, K.; et al. Baicalin protects against zearalenone-induced chicks liver and kidney injury by inhibiting expression of oxidative stress, inflammatory cytokines and caspase signaling pathway. Int. Immunopharmacol. 2021, 100, 108097. [Google Scholar] [CrossRef]
- Lin, M.; Li, L.; Li, L.; Pokhrel, G.; Qi, G.; Rong, R.; Zhu, T. The protective effect of baicalin against renal ischemia-reperfusion injury through inhibition of inflammation and apoptosis. BMC Complement. Altern. Med. 2014, 14, 19. [Google Scholar] [CrossRef]
- He, Q.; Sun, X.; Zhang, M.; Chu, L.; Zhao, Y.; Wu, Y.; Zhang, J.; Han, X.; Guan, S.; Ding, C. Protective effect of baicalin against arsenic trioxide-induced acute hepatic injury in mice through JAK2/STAT3 signaling pathway. Int. J. Immunopathol. Pharmacol. 2022, 36, 20587384211073397. [Google Scholar] [CrossRef]
- Xing, Z.G.; Wang, Y.Z.; Jia, D.Z.; Li, Q.J. Study on the mechanism of protective effect of baicalin on acute kidney injury in rats with crush syndrome. Mod. J. Integr. Tradit. Chin. West. Med. 2022, 31, 1204–1217. [Google Scholar]
- Ning, X.; Luo, D.; Chen, Y.; Shao, Y.; Xu, J. Baicalin reduces renal inflammation in mesangial proliferative glomerulonephritis through activation of Nrf2/ARE and PI3K/AKT pathways. Discov. Med. 2023, 35, 372–382. [Google Scholar] [CrossRef]
- Xi, B.S.; Huang, P.Z.; Tong, Z.Y.; Yao, C.L.; Hou, Y.Y.; Ji, Y. The role of NF-κB in the protective effects of baicalin for septic rats’ kidneys. Chin. J. Clin. Med. 2011, 18, 469–472. [Google Scholar]
- Li, Z.P.; Gu, J.; Zhang, Y.C.; Yang, Q.T.; Liu, M. Protective effect and mechanism of baicalin on acute kidney injury in duced by rhabdomyolysis syndrome in rats. Lab. Med. Clin. 2023, 20, 2465–2468. [Google Scholar]
- Jia, Y.C.; Yang, C.; Zheng, L.; Li, J.W.; Zeng, Y.G.; Xu, M.; Zhu, T.Y.; Rong, R.M. Protective effect of baicalin on acute kidney injury induced by aristolochic acid in mice. Chin. J. Clin. Med. 2019, 26, 432–436. [Google Scholar]
- Zhu, Y.; Fu, Y.; Lin, H. Baicalin inhibits renal cell apoptosis and protects against acute kidney injury in pediatric sepsis. Med. Sci. Monit. 2016, 22, 5109–5115. [Google Scholar] [CrossRef]
- Le, J.; Fan, H.; Sun, M.; Zhu, J. Protective effect and mechanism of baicalin on acute kidney injury in septic mice. Chin. Crit. Care Med. 2021, 33, 866–870. [Google Scholar]
- Wang, Y.; Jia, Y.; Yang, X.; Liang, B.; Gao, H.; Yang, T. A potential role of baicalin to inhibit apoptosis and protect against acute liver and kidney injury in rat preeclampsia model. Biomed. Pharmacother. 2018, 108, 1546–1552. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, J.; Huang, D.; Yu, C. Baicalin alleviates contrast-induced acute kidney injury through ROS/NLRP3/Caspase-1/GSDMD pathway-mediated proptosis in vitro. Drug Des. Dev. Ther. 2022, 16, 3353–3364. [Google Scholar] [CrossRef] [PubMed]
- Sahu, B.D.; Mahesh Kumar, J.; Sistla, R. Baicalein, a bioflavonoid, prevents cisplatin-induced acute kidney injury by up-regulating antioxidant defenses and down-regulating the MAPKs and NF-κB pathways. PLoS ONE 2015, 10, e0134139. [Google Scholar] [CrossRef]
- Chen, Y.; Zheng, Y.; Zhou, Z.; Wang, J. Baicalein alleviates tubular-interstitial nephritis in vivo and in vitro by down-regulating NF-κB and MAPK pathways. Braz. J. Med. Biol. Res. 2018, 51, e7476. [Google Scholar] [CrossRef]
- Lai, C.C.; Huang, P.H.; Yang, A.H.; Chiang, S.C.; Tang, C.Y.; Tseng, K.W.; Huang, C.H. Baicalein, a component of Scutellaria baicalensis, attenuates kidney injury induced by myocardial ischemia and reperfusion. Planta Med. 2016, 82, 181–189. [Google Scholar] [CrossRef]
- Hu, Y.; Lyu, C.Y.; Dai, X.; Wang, Y.H.; Zhao, R.Z.; Feng, J.X.; Lou, S.l.; Yan, H.; Sun, C. Baicalein intervenes in the Nrf-2/HO-1 signaling pathway reduces kidney injury in sepsis mice. Chin. J. Vet. Sci. 2025, 45, 121–128. [Google Scholar]
- Dai, C.; Tang, S.; Wang, Y.; Velkov, T.; Xiao, X. Baicalein acts as a nephroprotectant that ameliorates colistin-induced nephrotoxicity by activating the antioxidant defence mechanism of the kidneys and down-regulating the inflammatory response. J. Antimicrob. Chemother. 2017, 72, 2562–2569. [Google Scholar] [CrossRef]
- Wu, K.; Li, H.; Tian, J.; Lei, W. Protective effect of baicalein on renal ischemia/reperfusion injury in the rat. Ren. Fail. 2015, 37, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Li, H.; Wang, B.; Wu, Z.; Wu, S.; Jiang, G.; Wang, H.; Huang, Y. Baicalein ameliorates polymyxin B-induced acute renal injury by inhibiting ferroptosis via regulation of SIRT1/p53 acetylation. Chem. Biol. Interact. 2023, 382, 110607. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Zhou, L.; Liu, X.; Gao, L.; Li, Y.; Wu, Y. Baicalein alleviates cisplatin-induced acute kidney injury by inhibiting ALOX12-dependent ferroptosis. Phytomedicine 2024, 130, 155757. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.M.; Li, H.D.; Wu, W.F.; Ming-Kuen Tang, P.; Ren, G.L.; Gao, L.; Li, X.F.; Yang, Y.; Xu, T.; Ma, T.T.; et al. Wogonin protects against cisplatin-induced acute kidney injury by targeting RIPK1-mediated necroptosis. Lab. Invest 2018, 98, 79–94. [Google Scholar] [CrossRef]
- Wang, J.N.; Wang, J.J.; Wang, M.X.; Wu, Y.G.; Qi, X.M. Protective effect of wogonin on lipopolysaccharide-induced acute kidney injury in mice. Acta Univ. Med. Anhui 2024, 59, 1411–1416. [Google Scholar]
- Qi, Q.; Li, Y.; Ding, M.; Huang, C.; Omar, S.M.; Shi, Y.; Liu, P.; Cai, G.; Zheng, Z.; Guo, X.; et al. Wogonin inhibits apoptosis and necroptosis induced by nephropathogenic infectious bronchitis virus in chicken renal tubular epithelial cells. Int. J. Mol. Sci. 2024, 25, 8194. [Google Scholar] [CrossRef]
- Alicic, R.Z.; Rooney, M.T.; Tuttle, K.R. Diabetic kidney disease: Challenges, progress, and possibilities. Clin. J. Am. Soc. Nephrol. 2017, 12, 2032–2045. [Google Scholar] [CrossRef]
- Gupta, S.; Dominguez, M.; Golestaneh, L. Diabetic kidney disease: An update. Med. Clin. N. Am. 2023, 107, 689–705. [Google Scholar] [CrossRef]
- Thomas, M.C.; Brownlee, M.; Susztak, K.; Sharma, K.; Jandeleit-Dahm, K.A.; Zoungas, S.; Rossing, P.; Groop, P.H.; Cooper, M.E. Diabetic kidney disease. Nat. Rev. Dis. Primers 2015, 1, 15018. [Google Scholar] [CrossRef]
- Ma, L.; Wu, F.; Shao, Q.; Chen, G.; Xu, L.; Lu, F. Baicalin alleviates oxidative stress and inflammation in diabetic nephropathy via Nrf2 and MAPK signaling pathway. Drug Des. Dev. Ther. 2021, 15, 3207–3221. [Google Scholar] [CrossRef]
- Ren, G.; Jiao, P.; Yan, Y.; Ma, X.; Qin, G. Baicalin exerts a protective effect in diabetic nephropathy by repressing inflammation and oxidative stress through the SphK1/S1P/NF-κB signaling pathway. Diabetes Metab. Syndr. Obes. 2023, 16, 1193–1205. [Google Scholar] [CrossRef] [PubMed]
- Yin, Q.Q.; Xia, Y.Y.; Chen, J.; Xiao, L.; Wang, Y.; Zhao, Y.Y.; Ye, G.; Wu, J. Baicalin improves diabetic nephropathy in mice by activating Sirt1/Nrf2 signal through miR-141 inhibition. Med. J. Wuhan Univ. 2019, 40, 186–191. [Google Scholar]
- Xi, Y.L.; Wang, M.; Zhou, J. Baicalin regulate oxidative stress to prevent and treat diabetic nephropathy. Chin. J. Bioprocess. Eng. 2024, 22, 419–425. [Google Scholar]
- Zhao, B.L.; Li, X.T.; Pan, S.M.; Zhang, X.N.; Huang, Z.J.; Wu, S.F.; Zhang, S.X.; Lin, R.S.; Su, N. Effect of baicalin capsules on kidney pathological morphology and protein kinase C in the rats of diabetic nephropath. World J. Integr. Tradit. West. Med. 2016, 11, 488–491. [Google Scholar]
- Zhang, Y.; Song, G.H. The research of kidney-protective effects of erigeron breviscapus on diabetic nephropathy in rats. Chin. J. Diabetes 2003, 11, 19–21. [Google Scholar]
- Hu, S.H.; Zhang, M.X. The protective effect of baicalin on the kidney of STZ induced diabeti crats and its relation to VEGF. Chin. J. Hosp. Pharm. 2007, 27, 885–888. [Google Scholar]
- Zhao, B.L. The effects of baicalin on phosphorylation sites on the Connexin43 gap junction protein in diabetic nephropathy rats kidney. Master’s Thesis, Guangzhou University, Guangzhou, China, 2015. [Google Scholar]
- Li, M.; Zhu, H.Y.; Zhao, S.Y.; Li, X.D.; Tong, S.M.; Ma, J.; Xu, A.J.; Zhang, J. Baicalin alleviates lipid metabolism disorders in diabetic kidney disease via targeting FKBP51. Phytomedicine 2025, 139, 156473. [Google Scholar] [CrossRef]
- Sun, P.; Lu, L.; Chen, J.; Liu, X.D.; Zhang, Q.; Wang, X. AMPKα, hs-CRP and FcγR in diabetic nephropathy and drug intervention. Exp. Ther. Med. 2018, 15, 4659–4664. [Google Scholar] [CrossRef]
- Faulkner, J.; Pye, C.; Al-Shabrawey, M.; Elmarakby, A.A. Inhibition of 12/15-lipoxygenase reduces renal inflammation and injury in streptozotocin-induced diabetic mice. J. Diabetes Metab. 2015, 6, 555. [Google Scholar]
- Ahad, A.; Mujeeb, M.; Ahsan, H.; Siddiqui, W.A. Prophylactic effect of baicalein against renal dysfunction in type 2 diabetic rats. Biochimie 2014, 106, 101–110. [Google Scholar] [CrossRef]
- Hou, Y.L.; Mei, W.; Guo, F.F.; He, M.J.; Wu, G.Q.; Lin, M. Effects of wogonin on blood glucose and renal tissue TLR4 and NF-κB p65 levels in rats with diabetic nephropathy. Guid. J. Tradit. Chin. Med. Pharm. 2021, 27, 5–9. [Google Scholar]
- Lei, L.; Zhao, J.; Liu, X.Q.; Chen, J.; Qi, X.M.; Xia, L.L.; Wu, Y.G. Wogonin alleviates kidney tubular epithelial injury in diabetic nephropathy by inhibiting PI3K/Akt/NF-κB signaling pathways. Drug Des. Dev. Ther. 2021, 15, 3131–3150. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.; Xu, X.X. Effect of wogonoside on KLF4/NF-κB signal pathway in RAW264.7 macrophages stimulated by high glucose. Act. Univ. Med. Anhui 2022, 57, 1523–1528. [Google Scholar]
- Xie, S. Wogonoside interferes with DNM3OS/KLF4 pathway and regulates macrophage activation in diabetic nephropathy. Master’s Thesis, Anhui Medical University, Hefei, China, 2022. [Google Scholar]
- Liu, X.Q.; Jiang, L.; Li, Y.Y.; Huang, Y.B.; Hu, X.R.; Zhu, W.; Wang, X.; Wu, Y.G.; Meng, X.M.; Qi, X.M. Wogonin protects glomerular podocytes by targeting Bcl-2-mediated autophagy and apoptosis in diabetic kidney disease. Acta Pharmacol. Sin. 2022, 43, 96–110. [Google Scholar] [CrossRef]
- Wang, Y. Protective effect and mechanism of wogonin on diabetic kidney podocyte injury. Ph.D. Thesis, Anhui Medical University, Hefei, China, 2020. [Google Scholar]
- Li, X.; Zhao, S.; Li, M.; Xing, X.; Xie, J.; Wang, M.; Xu, A.; Zhao, Q.; Zhang, J. Wogonoside ameliorates oxidative damage in tubular epithelial cells of diabetic nephropathy by modulating the HNF4A-NRF2 axis. Int. Immunopharmacol. 2025, 152, 114481. [Google Scholar] [CrossRef]
- Major, T.J.; Dalbeth, N.; Stahl, E.A.; Merriman, T.R. An update on the genetics of hyperuricaemia and gout. Nat. Rev. Rheumatol. 2018, 14, 341–353. [Google Scholar] [CrossRef]
- El Ridi, R.; Tallima, H. Physiological functions and pathogenic potential of uric acid: A review. J. Adv. Res. 2017, 8, 487–493. [Google Scholar] [CrossRef]
- Casanova, A.G.; Morales, A.I.; Vicente-Vicente, L.; López-Hernández, F.J. Effect of uric acid reduction on chronic kidney disease. Systematic review and meta-analysis. Front. Pharmacol. 2024, 15, 1373258. [Google Scholar] [CrossRef]
- Su, H.Y.; Yang, C.; Liang, D.; Liu, H.F. Research advances in the mechanisms of hyperuricemia-induced renal injury. BioMed Res. Int. 2020, 2020, 5817348. [Google Scholar] [CrossRef]
- Fu, W.; Liu, Z.; Wang, Y.; Li, X.; Yu, X.; Li, Y.; Yu, Z.; Qiu, Y.; Mei, Z.; Xu, L. Baicalin inhibits monosodium urate crystal-induced pyroptosis in renal tubular epithelial cell line through Panx-1/P2X7 pathways: Molecular docking, molecular dynamics, and in vitro experiments. Chem. Biol. Drug Des. 2024, 103, e14522. [Google Scholar] [CrossRef]
- Liu, Y.J. Study on the effect and mechanism of baicalin against hyperuricemia nephropathy. Master’s Thesis, Wuhan Polytechnic University, Wuhan, China, 2020. [Google Scholar]
- Liu, Z.; Xiang, H.; Deng, Q.; Fu, W.; Li, Y.; Yu, Z.; Qiu, Y.; Mei, Z.; Xu, L. Baicalin and baicalein attenuate hyperuricemic nephropathy via inhibiting PI3K/AKT/NF-κB signalling pathway. Nephrology 2023, 28, 315–327. [Google Scholar] [CrossRef] [PubMed]
- Xiang, H.; Lei, H.; Liu, Z.; Liu, Y.; Li, Y.; Qiu, Y.; Xu, L. Network pharmacology and molecular docking analysis on molecular targets: Mechanisms of baicalin and baicalein against hyperuricemic nephropathy. Toxicol. Appl. Pharmacol. 2021, 424, 115594. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Mao, Z.; Li, X.; Zhong, D.; Li, M.; Jia, Y.; Wei, J.; Yang, B.; Zhou, H. Baicalein decreases uric acid and prevents hyperuricemic nephropathy in mice. Oncotarget 2017, 8, 40305–40317. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhao, Z.; Li, Y.; Yang, Y.; Li, L.; Jiang, Y.; Lin, C.; Cao, Y.; Zhou, P.; Tian, Y.; et al. Baicalein alleviates hyperuricemia by promoting uric acid excretion and inhibiting xanthine oxidase. Phytomedicine 2021, 80, 153374. [Google Scholar] [CrossRef]
- Wei, X.; Hou, Y.; Long, M.; Jiang, L.; Du, Y. Advances in energy metabolism in renal fibrosis. Life Sci. 2023, 312, 121033. [Google Scholar] [CrossRef]
- Zhao, D.; Zhu, X.; Jiang, L.; Huang, X.; Zhang, Y.; Wei, X.; Zhao, X.; Du, Y. Advances in understanding the role of adiponectin in renal fibrosis. Nephrology 2021, 26, 197–203. [Google Scholar] [CrossRef]
- Sears, S.M.; Sharp, C.N.; Krueger, A.; Oropilla, G.B.; Saforo, D.; Doll, M.A.; Megyesi, J.; Beverly, L.J.; Siskind, L.J. C57BL/6 mice require a higher dose of cisplatin to induce renal fibrosis and CCL2 correlates with cisplatin-induced kidney injury. Am. J. Physiol. Ren. Physiol. 2020, 319, F674–F685. [Google Scholar] [CrossRef]
- Zhou, S.; Yin, X.; Yuan, J.; Liang, Z.; Song, J.; Li, Y.; Peng, C.; Hylands, P.J.; Zhao, Z.; Xu, Q. Antifibrotic activities of Scutellariae Radix extracts and flavonoids: Comparative proteomics reveals distinct and shared mechanisms. Phytomedicine 2022, 100, 154049. [Google Scholar] [CrossRef]
- Fang, J.; Wang, W.; Sun, S.; Wang, Y.; Li, Q.; Lu, X.; Qiu, M.; Zhang, Y. Metabolomics study of renal fibrosis and intervention effects of total aglycone extracts of Scutellaria baicalensis in unilateral ureteral obstruction rats. J. Ethnopharmacol. 2016, 192, 20–29. [Google Scholar] [CrossRef]
- Hu, Q.; Gao, L.; Peng, B.; Liu, X. Baicalin and baicalein attenuate renal fibrosis in vitro via inhibition of the TGF-β1 signaling pathway. Exp. Ther. Med. 2017, 14, 3074–3080. [Google Scholar] [CrossRef]
- Wang, W.; Zhou, P.H.; Xu, C.G.; Zhou, X.J.; Hu, W.; Zhang, J. Baicalein ameliorates renal interstitial fibrosis by inducing myofibroblast apoptosis in vivo and in vitro. BJU Int. 2016, 118, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhou, P.H.; Xu, C.G.; Zhou, X.J.; Hu, W.; Zhang, J. Baicalein attenuates renal fibrosis by inhibiting inflammation via down-regulating NF-κB and MAPK signal pathways. J. Mol. Histol. 2015, 46, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Liang, G.Q.; Mu, W.; Jiang, C.B. Baicalein improves renal interstitial fibrosis by inhibiting the ferroptosis in vivo and in vitro. Heliyon 2024, 10, e28954. [Google Scholar] [CrossRef] [PubMed]
- Miao, S.H. Effects of baicalein on endoplasmic reticulum stresspathway in rats with renal interstitial fibrosis. Master’s Thesis, Lanzhou University, Lanzhou, China, 2017. [Google Scholar]
- Wang, H.; Jiang, Q.; Zhang, L. Baicalin protects against renal interstitial fibrosis in mice by inhibiting the TGF-β/Smad signalling pathway. Pharm. Biol. 2022, 60, 1407–1416. [Google Scholar] [CrossRef]
- Zheng, L.; Zhang, C.; Li, L.; Hu, C.; Hu, M.; Sidikejiang, N.; Wang, X.; Lin, M.; Rong, R. Baicalin ameliorates renal fibrosis via inhibition of transforming growth factor β1 production and downstream signal transduction. Mol. Med. Rep. 2017, 15, 1702–1712. [Google Scholar] [CrossRef]
- Liu, H.X.; Guo, M.H.; Liu, Y.; Zhu, Y.J.; Liu, X.D.; Xu, Q.Y.; Tan, J. Effect of baicalin on human renal tubular epithelial cell transdifferentiation induced by TGF-β1 in vitro. Chin. J. Integr. Tradit. West. Nephrol. 2011, 12, 864–866. [Google Scholar]
- Tan, Y.J.; Zhu, C.L.; Mao, H.X. Therapeutic effect of baicalin in treatment of renal interstitial fibrosis in rats with unliateral ureteral obstruction and related mechanisms. Chin. J. Contemp. Pediatr. 2016, 18, 365–371. [Google Scholar]
- Su, N.; Li, F.; Chen, J.Y.; Chen, Z.X.; Zhou, L.Q.; Luo, R.J. Effect of baicalin on plasma and renal AngⅡ in diabetic nephropathy rats. Tradit. Chin. Drug Res. Clin. Pharmacol. 2009, 20, 201–203. [Google Scholar]
- Zhang, S.; Xu, L.; Liang, R.; Yang, C.; Wang, P. Baicalin suppresses renal fibrosis through microRNA-124/TLR4/NF-κB axis in streptozotocin-induced diabetic nephropathy mice and high glucose-treated human proximal tubule epithelial cells. J. Physiol. Biochem. 2020, 76, 407–416. [Google Scholar] [CrossRef]
- Hu, H.; Li, W.; Hao, Y.; Peng, Z.; Zou, Z.; Liang, W. Baicalin ameliorates renal fibrosis by upregulating CPT1α-mediated fatty acid oxidation in diabetic kidney disease. Phytomedicine 2024, 122, 155162. [Google Scholar] [CrossRef]
- Cai, Y.; Ma, W.; Xiao, Y.; Wu, B.; Li, X.; Liu, F.; Qiu, J.; Zhang, G. High doses of baicalin induces kidney injury and fibrosis through regulating TGF-β/Smad signaling pathway. Toxicol. Appl. Pharmacol. 2017, 333, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Li, F. The role of baicalin bai against glomerulosclerosis by regulating CTGF. Master’s Thesis, Guangzhou University of Chinese Medicine, Guangzhou, China, 2011. [Google Scholar]
- Zheng, X.P.; Feng, J.; Song, C.Y.; Wang, F.F.; Zhang, H.M.; Sui, H.Y.; Xin, H.; Chen, G. Effect of baicalin on renal fibrosis in rats with diabetic nephropathy by TGF-β1. J. Med. Res. Combat. Trauma. Care 2020, 33, 471–475. [Google Scholar]
- Liu, Q.P.; Shao, X.; Li, Y.T.; Liu, M.; Tan, P.; Wang, Y. Baicalin improves renal fibrosis in rats with chronic kidney disease by regulating the galectin-3/Akt/GSK-3B/Snail signaling pathway. Hebei Med. J. 2024, 46, 2261–2265. [Google Scholar]
- Zhang, X.T.; Wang, G.; Ye, L.F.; Pu, Y.; Li, R.T.; Liang, J.; Wang, L.; Lee, K.K.H.; Yang, X. Baicalin reversal of DNA hypermethylation-associated Klotho suppression ameliorates renal injury in type 1 diabetic mouse model. Cell Cycle 2020, 19, 3329–3347. [Google Scholar] [CrossRef]
- Nam, J.E.; Jo, S.Y.; Ahn, C.W.; Kim, Y.S. Baicalin attenuates fibrogenic process in human renal proximal tubular cells (HK-2) exposed to diabetic milieu. Life Sci. 2020, 254, 117742. [Google Scholar] [CrossRef]
- Ke, Y.Q.; Hao, J.P.; Lu, L.; Chen, X.L.; Fu, Y.Q. Study on the mechanism of wogonoside protecting against renal injury and fibrosis in diabetic rats. J. Guangzhou Univ. Tradit. Chin. Med. 2022, 39, 2109–2115. [Google Scholar]
- Meng, X.M.; Ren, G.L.; Gao, L.; Li, H.D.; Wu, W.F.; Li, X.F.; Xu, T.; Wang, X.F.; Ma, T.T.; Li, Z.; et al. Anti-fibrotic effect of wogonin in renal tubular epithelial cells via Smad3-dependent mechanisms. Eur. J. Pharmacol. 2016, 789, 134–143. [Google Scholar] [CrossRef]
- Zhang, X.D.; Ren, G.L.; Chen, H.H.; Chen, Y.; Zhu, J. Effect of baicalin modulation of TLR4/MAPK/NF-KB signaling pathway on renal fibrosis in rats with diabetic nephropathy. Chin. Pharmacol. Bull. 2023, 39, 1840–1846. [Google Scholar]
- Kuo, H.L.; Chuang, H.L.; Chen, C.M.; Chen, Y.Y.; Chen, Y.S.; Lin, S.C.; Weng, P.Y.; Liu, T.C.; Wang, P.Y.; Huang, C.F.; et al. Wogonin ameliorates ER stress-associated inflammatory response, apoptotic death and renal fibrosis in a unilateral ureteral obstruction mouse model. Eur. J. Pharmacol. 2024, 977, 176676. [Google Scholar] [CrossRef]
- Guo, J.; Mei, Z.W.; Wang, X.J.; Li, Q.; Qin, J. Molecular docking and network pharmacological analysis of Scutellaria baicalensis against renal cell carcinoma. Eur. Rev. Med. Pharmacol. Sci. 2023, 27, 11574–11586. [Google Scholar]
- Wang, Y. The role of wogonin in the malignant behavior and resistance to targeted drug of renal cell carcinoma. Ph.D. Thesis, Shandong University, Jinan, China, 2021. [Google Scholar]
- Wang, Y.; Chen, S.; Sun, S.; Liu, G.; Chen, L.; Xia, Y.; Cui, J.; Wang, W.; Jiang, X.; Zhang, L.; et al. Wogonin induces apoptosis and reverses sunitinib resistance of renal cell carcinoma cells via inhibiting CDK4-RB pathway. Front. Pharmacol. 2020, 11, 1152. [Google Scholar] [CrossRef]


| Compound | Molecular Structure | CAS | Molecular Formula | Molecular Weight | Plant Part |
|---|---|---|---|---|---|
| Baicalein |  | 491-67-8 | C15H10O5 | 270.237 | Root; Hairy Root |
| Wogonin |  | 632-85-9 | C16H12O5 | 284.263 | Root; Aerial Part Hairy Root |
| Baicalin |  | 21967-41-9 | C21H18O11 | 446.361 | Root; Aerial Part Hairy Root |
| Wogonoside |  | 51059-44-0 | C22H20O11 | 460.388 | Root; Hairy Root |
| Compound | In Vivo or In Vitro Models | Mechanism of Action | Outcome | Ref. |
|---|---|---|---|---|
| Baicalin | db/db and db/m mouse model; palmitic acid-induced MPC-5 cells; high-glucose-induced HK-2 cells | Activation of Nrf2 signaling pathway; inhibition of MAPK and SphK1/S1P/NF-κB signaling pathway, as well as inhibition of the expression of miR-141 to activate the Sirt1/Nrf2 signaling axis | Oxidative stress ↓ Inflammation ↓ Apoptosis ↓ | [90,91] |
| STZ-induced DKD mouse model; high-glucose-induced SV40-Mes-13 cells | Down-regulation of the expression of miR-141 to activate the Sirt1/Nrf2 signaling axis | Oxidative stress ↓ Apoptosis ↓ | [92] | |
| STZ combined with high fat and high-glucose-induced DKD mouse model | Down-regulation the expression of MDA and 8-hydroxydeoxyguanosine; up-regulation of the expression of the genes encoding CAT and Mn-SOD, as well as forkhead box O3a protein | Oxidative stress ↓ | [93] | |
| STZ-induced and STZ combined with high-fat-induced DKD rat model | Inhibition of the activity of PKC and the expression of phosphorylated connexin 43 to decrease the content of ECM and TGF-β1; inhibition of the production of advanced glycation end products and the expression of VEGF | Inflammation ↓ ECM production ↓ | [94,95,96,97] | |
| High-fat-diet/STZ-induced DKD mouse model | Targeting FK506-binding protein 51 | Lipid metabolism ↑ | [98] | |
| Baicalein | STZ combined with high-fat and high-glucose-induced DKD rat model | Down-regulation the expression of AMPKα, hs-CRP and FcγR | Inflammation ↓ | [99] |
| STZ-induced DKD model in C57BL/6J (WT) and 12/15-LO-deficient mice | Down-regulation the activity of 12/15-LO | Inflammation ↓ | [100] | |
| STZ combined with high-fat -induced DKD rat model | Inhibition of NF-κB signaling pathway to down-regulation the expression of iNOS and TGF-β1 | Lipid and glucose metabolism ↑ Oxidative stress ↓ Inflammation ↓ | [101] | |
| Wogonin | STZ combined with high-fat-induced DKD rat model | Inhibition of TLR/NF-κB signaling pathway | Inflammation ↓ | [102] |
| STZ-induced DKD mouse model; high-glucose-induced HK-2 cells | Inhibition of PI3K/Akt/NF-κB signaling pathway | Autophagy ↓ Inflammation ↓ | [103] | |
| High-glucose-induced RAW264.7 cells | Regulation of KLF4/NF-κB signaling pathway in macrophages | Inflammation ↓ | [104] | |
| db/db and db/m mouse model; high-glucose-induced RAW264.7 cells | Inhibition DNM3OS/KLF4 signaling pathway in macrophages | Oxidative stress ↓ Inflammation ↓ | [105] | |
| STZ-induced DKD mouse model; high-glucose-induced podocyte cells | Regulation of the balance between apoptosis and autophagy mediated by Bcl-2 | Apoptosis ↓ Autophagy ↓ | [106,107] | |
| db/db mouse model; high-glucose-induced HK-2 cells | Inhibition of TLR4-mediated JAK/STAT/AIM2 signaling pathway | Apoptosis ↓ Autophagy ↓ | [106] | |
| Wogonoside | HFD/STZ-induced DKD mouse model; high-glucose-induced MPC-5 cells | Targeting the NF-κB p65-MMP28 axis | Apoptosis ↓ Autophagy ↓ | [106] |
| STZ combined with high-fat-induced DKD mouse model; high-glucose-induced TCMK-1 cells | Targeting the HNF4A-NRF2 axis | Oxidative stress ↓ | [108] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yang, X.; Dou, F.; Hai, L.; Xiao, Y.; Cui, J.; Cai, Y.; Wang, R.; Ji, K.; Feng, Y.; Chen, H. Scutellaria baicalensis Georgi: A Promising Source of Bioactive Molecules for Kidney Disease Therapy. Biomolecules 2026, 16, 64. https://doi.org/10.3390/biom16010064
Yang X, Dou F, Hai L, Xiao Y, Cui J, Cai Y, Wang R, Ji K, Feng Y, Chen H. Scutellaria baicalensis Georgi: A Promising Source of Bioactive Molecules for Kidney Disease Therapy. Biomolecules. 2026; 16(1):64. https://doi.org/10.3390/biom16010064
Chicago/Turabian StyleYang, Xia, Fang Dou, Lang Hai, Yating Xiao, Jie Cui, Yangyang Cai, Rui Wang, Kai Ji, Yalong Feng, and Hua Chen. 2026. "Scutellaria baicalensis Georgi: A Promising Source of Bioactive Molecules for Kidney Disease Therapy" Biomolecules 16, no. 1: 64. https://doi.org/10.3390/biom16010064
APA StyleYang, X., Dou, F., Hai, L., Xiao, Y., Cui, J., Cai, Y., Wang, R., Ji, K., Feng, Y., & Chen, H. (2026). Scutellaria baicalensis Georgi: A Promising Source of Bioactive Molecules for Kidney Disease Therapy. Biomolecules, 16(1), 64. https://doi.org/10.3390/biom16010064







