Modulating Effect of Carbohydrate Antigen 125 on the Prognostic Value of High-Sensitivity C-Reactive Protein in Heart Failure
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Group and Protocol
2.2. Biomarkers Measurement
2.3. Follow-Up and Endpoints
2.4. Statistical Analysis
3. Results
3.1. Baseline Characteristics Across hs-CRP/CA125 Categories
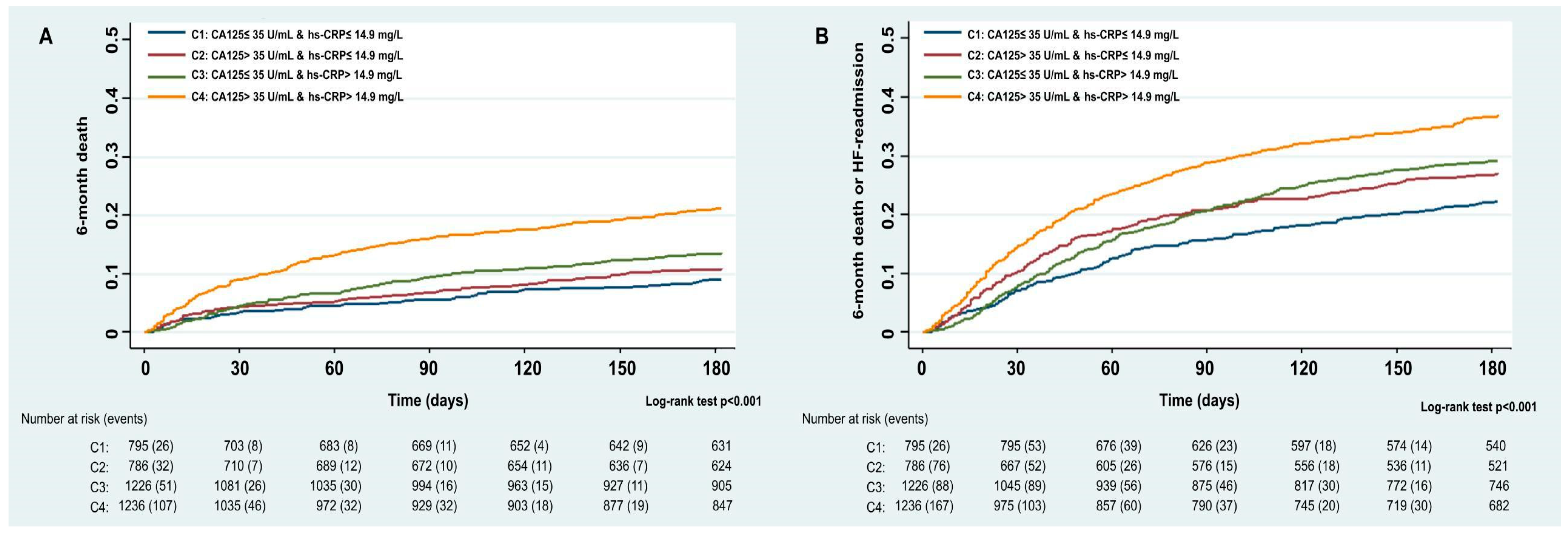
3.2. hs-CRP and 6-Month Adverse Outcomes
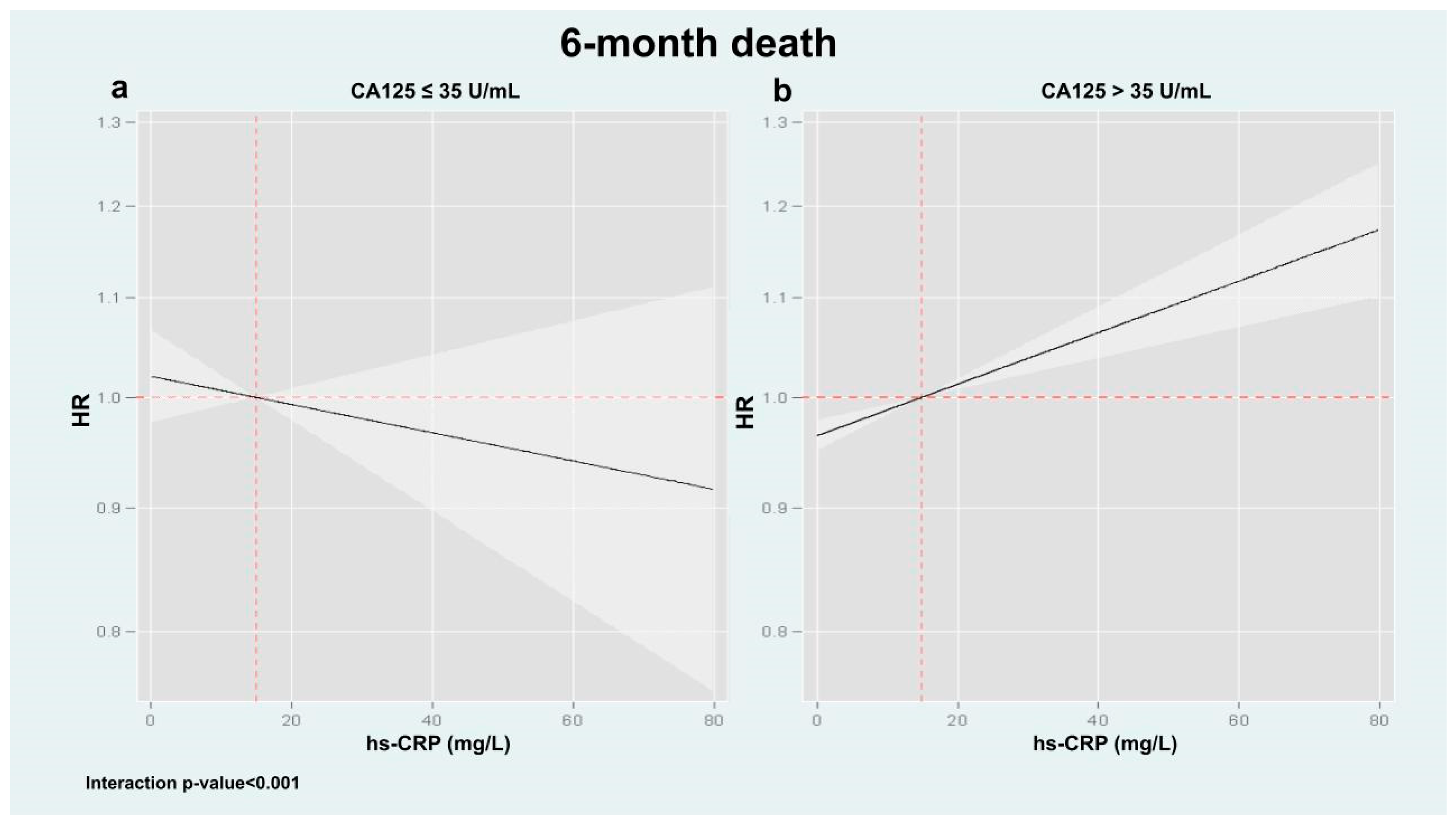
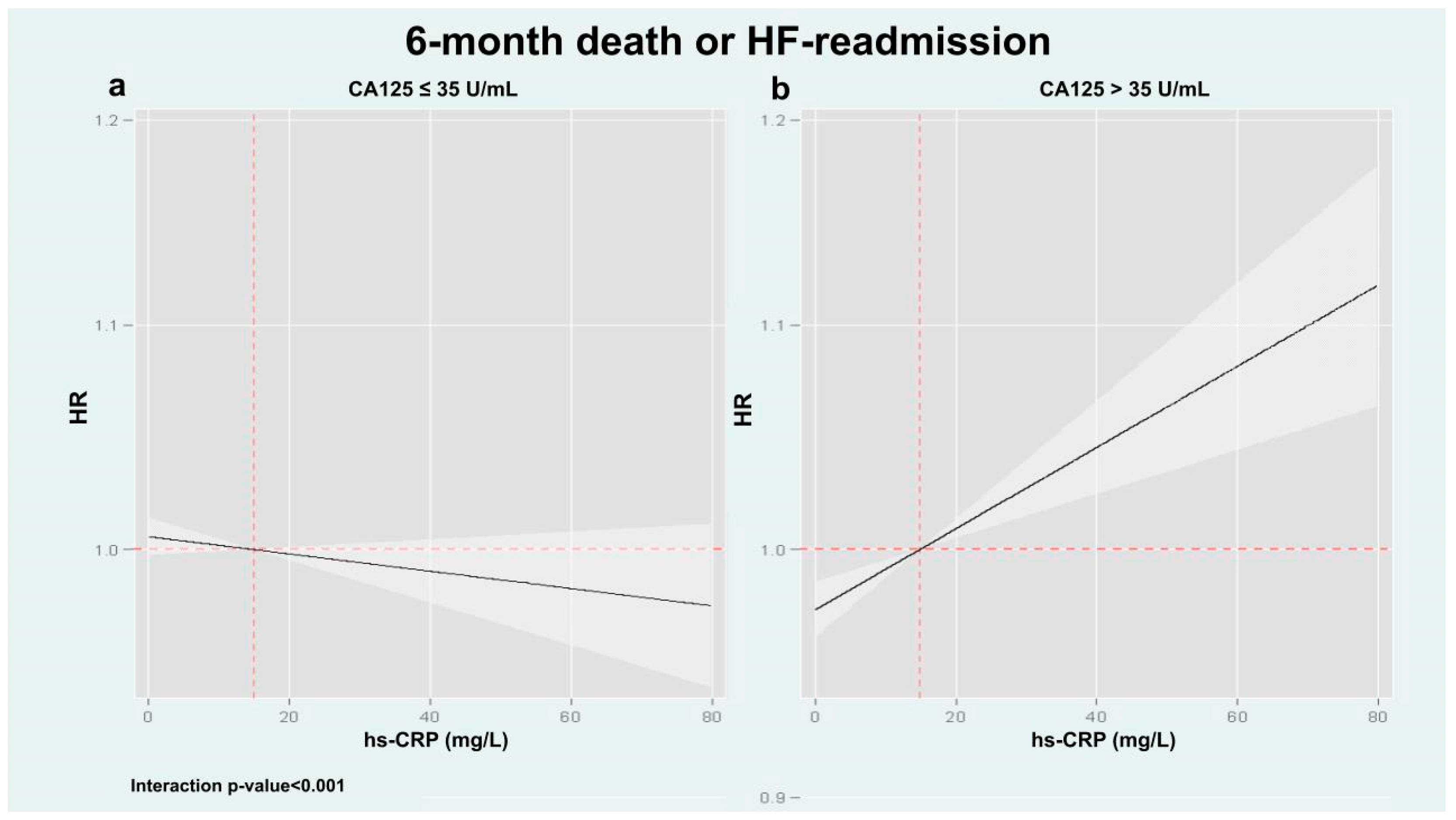
3.3. hs-CRP and Clinical Outcomes Across CA125
4. Discussion
4.1. Inflammation and Congestion: Two Converging Pathophysiological Axes in HF
4.2. CA125 as a Biomarker Beyond Congestion, a Modulator of Inflammatory Pathways?
4.3. Potential Clinical Implications
4.4. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| HF | Heart failure |
| IL-1 | Interleukin-1 |
| IL-6 | Interleukin-6 |
| hs-CRP | High-sensitivity C-reactive protein |
| CA125 | Carbohydrate antigen 125 |
| Gal-3 | Galectin-3 |
| sST2 | Soluble ST2 |
| IQR | Interquartile range |
| HR | Hazard ratios |
| CI | Confidence interval |
| NYHA | New York Heart Association |
| LVEF | Left ventricular ejection fraction |
| TAPSE | Tricuspid annular systolic plane excursion |
| RAAS | Renin-angiotensin-aldosterone system |
| MUC16 | Mucin 16 |
| CTD | C-terminal domain |
Appendix A
| Total Cohort (n = 4043) | |
|---|---|
| Medical history | |
| Valvular heart disease, n (%) | 1421 (35.1) |
| Chronic kidney disease, n (%) | 1037 (25.6) |
| COPD, n (%) | 819 (20.3) |
| PAD, n (%) | 413 (10.2) |
| Charlson index | 2.0 (1.0–3.0) |
| Peripheral edema, n (%) | 2528 (62.5) |
| Pleural effusion, n (%) | 1983 (49.0) |
| Stroke, n (%) | 450 (11.1) |
| Infection, n (%) | 492 (12.2) |
| NYHA I-II, n (%) | 3381 (83.6) |
| ICD carrier, n (%) | 113 (2.8) |
| Bundle Branch Block, n (%) | 1284 (31.8) |
| Risk factors | |
| Smoker, n (%) | 491 (12.1) |
| Ex-smoker, n (%) | 1123 (27.8) |
| Alcoholic, n (%) | 289 (7.1) |
| Laboratory | |
| Hematocrit (%) | 38.3 ± 5.8 |
| Iron, µg/dL | 49.5 ± 15.3 |
| Treatment | |
| Beta-blockers at admission, n (%) | 1637 (40.5) |
| ACE-I at admission, n (%) | 1512 (37.4) |
| Diuretics at admission, n (%) | 2430 (60.1) |
| Category 1 Low hs-CRP and Low CA125 (n = 795) | Category 2 High hs-CRP and Low CA125 (n = 786) | Category 3 Low hs-CRP and High CA125 (n = 1226) | Category 4 High hs-CRP and High CA125 (n = 1236) | p-Value | |
|---|---|---|---|---|---|
| Medical history | |||||
| Valvular heart disease, n (%) | 261 (32.8) | 254 (32.3) | 448 (36.5) | 458 (37.1) | 0.054 |
| Chronic kidney disease, n (%) | 188 (23.6) | 233 (29.6) | 268 (21.9) | 348 (28.2) | <0.001 |
| COPD, n (%) | 156 (19.6) | 166 (21.1) | 245 (20.0) | 252 (20.4) | 0.890 |
| PAD, n (%) | 78 (9.8) | 103 (13.1) | 98 (8.0) | 134 (10.8) | 0.002 |
| Charlson index | 2.0 (1.0–3.0) | 2.0 (1.0–4.0) | 2.0 (1.0–3.0) | 2.0 (1.0, 4.0) | <0.001 |
| Stroke, n (%) | 82 (10.3) | 90 (11.5) | 126 (10.3) | 152 (12.3) | 0.359 |
| Infection, n (%) | 61 (7.7) | 146 (18.6) | 51 (4.2) | 234 (18.9) | <0.001 |
| NYHA I-II, n (%) | 689 (86.7) | 649 (82.6) | 1039 (84.7) | 1004 (81.2) | 0.006 |
| ICD carrier, n (%) | 17 (2.1) | 19 (2.4) | 36 (2.9) | 41 (3.3) | 0.391 |
| Bundle Branch Block, n (%) | 250 (31.4) | 259 (33.0) | 387 (31.6) | 388 (31.4) | 0.885 |
| Risk factors | |||||
| Smoker, n (%) | 93 (11.7) | 74 (9.4) | 169 (13.8) | 155 (12.5) | 0.031 |
| Ex-smoker, n (%) | 211 (26.5) | 211 (26.9) | 327 (26.7) | 374 (30.3) | 0.141 |
| Alcoholic, n (%) | 39 (4.9) | 36 (4.6) | 106 (8.6) | 108 (8.7) | <0.001 |
| Laboratory | |||||
| Hematocrit (%) | 39.3 ± 5.7 | 37.8 ± 5.7 | 38.8 ± 5.6 | 37.4 ± 6.0 | <0.001 |
| Iron, µg/dL | 52.0 ± 14.3 | 53.8 ± 13.8 | 46.6 ± 15.9 | 48.1 ± 15.3 | <0.001 |
| Treatment | |||||
| Beta-blockers at admission, n (%) | 353 (44.4) | 296 (37.7) | 485 (39.6) | 503 (40.7) | 0.043 |
| ACEI at admission, n (%) | 334 (42) | 318 (40.5) | 440 (35.9) | 420 (34) | <0.001 |
| Diuretics at admission, n (%) | 458 (57.6) | 482 (61.3) | 700 (57.1) | 790 (63.9) | 0.002 |
| Variable | HR | 95% CI | p-Value |
|---|---|---|---|
| Age, years | 1.04 | 1.03–1.06 | <0.001 |
| Sex | 1.12 | 1.02–1.24 | 0.017 |
| NYHA III-IV | 1.40 | 1.21–1.63 | <0.001 |
| First hospital admission | 0.99 | 0.83–1.19 | 0.933 |
| Ischemic heart disease | 1.17 | 1.14–1.20 | <0.001 |
| Bundle Branch Block | 0.99 | 0.93–1.06 | 0.876 |
| Atrial fibrillation | 0.78 | 0.59–1.04 | 0.088 |
| Peripheral edema | 0.95 | 0.83–1.09 | 0.482 |
| Pleural effusion | 1.30 | 1.23–1.38 | <0.001 |
| Heart rate | 0.99 | 0.99–1.00 | 0.173 |
| SBP | 0.99 | 0.98–0.99 | 0.048 |
| DBP | 1.01 | 0.99–1.01 | 0.166 |
| Hemoglobin | 0.98 | 0.94–1.01 | 0.163 |
| eGFR (MDRD formula), mL/min/1.73 m2 | 0.99 | 0.99–0.993 | <0.001 |
| Sodium | 0.97 | 0.95–0.99 | 0.004 |
| TAPSE | 0.94 | 0.92–0.97 | <0.001 |
| Aortic insufficiency | 1.01 | 0.99–1.03 | 0.237 |
| Infection | 1.31 | 1.03–1.67 | 0.028 |
| Iron | 0.99 | 0.98–1.00 | 0.194 |
| Diuretics | 1.24 | 1.11–1.40 | <0.001 |
| ACE-I/ARB/ARNI | 0.85 | 0.23–3.14 | 0.802 |
| ACE-I | 0.88 | 0.32–2.41 | 0.798 |
| Beta-blockers | 0.47 | 0.36–0.61 | <0.001 |
| MRA | 0.64 | 0.33–1.24 | 0.188 |
| Statins | 0.68 | 0.45–1.04 | 0.074 |
| CA125 > 35 U/mL | 1.41 | 1.21–1.64 | <0.001 |
| High-sensitivity C-reactive protein, mg/L | 0.87 | 0.65–1.17 | 0.372 |
| High hs-CRP and High CA125 | 1.46 | 1.02–2.08 | 0.036 |
| Variable | HR | 95% CI | p-Value |
|---|---|---|---|
| Age, years | 1.03 | 1.02–1.03 | <0.001 |
| Sex | 1.06 | 0.95–1.19 | 0.275 |
| NYHA III-IV | 1.32 | 1.23–1.43 | <0.001 |
| First hospital admission | 0.87 | 0.82–0.93 | <0.001 |
| Bundle Branch Block | 1.11 | 1.03–1.19 | 0.005 |
| Atrial fibrillation | 0.9 | 0.76–1.07 | 0.240 |
| Peripheral edema | 1.05 | 0.95–1.17 | 0.305 |
| Pleural effusion | 1.24 | 1.21–1.26 | <0.001 |
| Heart rate | 0.99 | 0.99–1.00 | 0.199 |
| SBP | 0.99 | 0.99–1.00 | <0.001 |
| DBP | 1.00 | 0.99–1.00 | 0.956 |
| eGFR (MDRD formula), mL/min/1.73 m2 | 0.99 | 0.99–1.00 | <0.001 |
| TAPSE | 0.97 | 0.95–0.99 | 0.010 |
| Aortic insufficiency | 1.01 | 1.00–1.01 | <0.001 |
| Iron | 0.99 | 0.99–1.01 | 0.750 |
| Diuretics | 1.42 | 1.34–1.51 | <0.001 |
| ACE-I/ARB/ARNI | 0.86 | 0.44–1.67 | 0.656 |
| ACE-I | 0.87 | 0.61–1.25 | 0.447 |
| Beta-blockers | 0.70 | 0.58–0.86 | 0.001 |
| MRA | 0.91 | 0.69–1.21 | 0.528 |
| CA125 > 35 U/mL | 1.23 | 1.20–1.26 | <0.001 |
| High-sensitivity C-reactive protein | 0.96 | 0.91–1.02 | 0.177 |
| High hs-CRP and High CA125 | 1.23 | 1.20–1.26 | <0.001 |
| Age, years | 1.03 | 1.02–1.03 | <0.001 |
| Sex | 1.06 | 0.95–1.19 | 0.275 |
| NYHA III-IV | 1.32 | 1.23–1.43 | <0.001 |
| First hospital admission | 0.87 | 0.82–0.93 | <0.001 |
| Bundle Branch Block | 1.11 | 1.03–1.19 | 0.005 |
References
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) With the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef] [PubMed]
- Murphy, S.P.; Kakkar, R.; McCarthy, C.P.; Januzzi, J.L., Jr. Inflammation in heart failure. J. Am. Coll. Cardiol. 2020, 75, 1324–1340. [Google Scholar] [CrossRef]
- Colombo, P.C.; Castagna, F.; Onat, D.; Wong, K.Y.; Harxhi, A.; Hayashi, Y.; Friedman, R.A.; Pinsino, A.; Ladanyi, A.; Mebazaa, A.; et al. Experimentally induced peripheral venous congestion exacerbates inflammation, oxidative stress, and neuhormonal and endothelial cell activation in patients with systolic heart failure. J. Card. Fail. 2023, 23, 580–591. [Google Scholar] [CrossRef]
- Kittipibul, V.; Fudim, M.; Sobotka, P.A. Congestion and inflammation in heart failure: Beyond the chicken or the egg. J. Card. Fail. 2023, 30, 592–595. [Google Scholar] [CrossRef] [PubMed]
- Adamo, L.; Rocha-Resende, C.; Prabhu, S.D.; Mann, D.L. Reappraising the role of inflammation in heart failure. Nat. Rev. Cardiol. 2020, 17, 269–286. [Google Scholar] [CrossRef]
- Dick, S.A.; Epelman, S. Chronic heart failure and inflammation: What do we really know? Circ. Res. 2016, 119, 159–171. [Google Scholar] [CrossRef] [PubMed]
- Michou, E.; Wussler, D.; Belkin, M.; Simmen, C.; Strebel, I.; Nowak, A.; Kozhuharov, N.; Shrestha, S.; Lopez-Ayala, P.; Sabti, Z.; et al. Quantifying inflammation using interleukin-6 for improved phenotyping and risk stratification in acute heart failure. Eur. J. Heart Fail. 2023, 25, 174–184. [Google Scholar] [CrossRef]
- Markousis-Mavrogenis, G.; Tromp, J.; Ouwerkerk, W.; Devalaraja, M.; Anker, S.D.; Cleland, J.G.; Dickstein, K.; Filippatos, G.S.; van der Harst, P.; Lang, C.C.; et al. The clinical significance of interleukin-6 in heart failure: Results from the BIOSTAT-CHF study. Eur. J. Heart Fail. 2019, 21, 965–973. [Google Scholar] [CrossRef]
- Ridker, P.M. C-reactive protein: Eighty years from discovery to emergence as a major risk marker for cardiovascular disease. Clin. Chem. 2009, 55, 209–215. [Google Scholar] [CrossRef]
- Ridker, P.M. From CRP to IL-6 and IL-1: Moving upstream to identify novel targets for atheroprotection. Circ. Res. 2016, 118, 145–156. [Google Scholar] [CrossRef]
- Zhang, L.; He, G.; Huo, X.; Tian, A.; Ji, R.; Pu, B.; Peng, Y. Long-term cumulative high-sensitivity C-reactive protein and mortality among patients with acute heart failure. J. Am. Heart Assoc. 2023, 12, e029386. [Google Scholar] [CrossRef]
- Santas, E.; Villar, S.; Palau, P.; Llàcer, P.; de la Espriella, R.; Miñana, G.; Lorenzo, M.; Núñez-Marín, G.; Górriz, J.L.; Carratalá, A.; et al. High sensitivity C reactive protein and risk of clinical outcomes in patients with acute heart failure. Sci. Rep. 2024, 14, 21672. [Google Scholar] [CrossRef] [PubMed]
- Núñez, J.; de la Espriella, R.; Miñana, G.; Santas, E.; Llácer, P.; Núñez, E.; Palau, P.; Bodí, V.; Chorro, F.J.; Sanchis, J.; et al. Antigen carbohydrate 125 as a biomarker in heart failure. A narrative review. Eur. J. Heart Fail. 2021, 9, 1445–1457. [Google Scholar] [CrossRef] [PubMed]
- Núñez, J.; Bayés-Genís, A.; Revuelta-López, E.; ter Maaten, J.M.; Miñana, G.; Barallat, J.; Cserkóová, A.; Bodi, V.; Fernández-Cisnal, A.; Núñez, E.; et al. Clinical role of CA125 in worsening heart failure. A BIOSTAT-CHF study subanalysis. J. Am. Coll. Cardiol. HF 2020, 8, 386–397. [Google Scholar] [CrossRef]
- Docherty, K.F.; McDowell, K.; Welsh, P.; Osmanska, J.; Anand, I.; de Boer, R.A.; Køber, L.; Kosiborod, M.N.; Martinez, F.A.; O’mEara, E.; et al. Association of Carbohydrate Antigen 125 on the Response to Dapagliflozin in Patients With Heart Failure. J. Am. Coll. Cardiol. 2023, 82, 142–157. [Google Scholar] [CrossRef]
- Ferreira, J.P.; Packer, M.; Sattar, N.; Butler, J.; Pocock, S.J.; Anker, S.D.; Maldonado, S.G.; Panova-Noeva, M.; Sumin, M.; Masson, S.; et al. Carbohydrate antigen 125 concentrations across the ejection fraction spectrum in chronic heart failure: The EMPEROR programme. Eur. J. Heart Fail. 2024, 26, 788–802. [Google Scholar] [CrossRef]
- O’Brien, T.J.; Beard, J.B.; Underwood, L.J.; Dennis, R.A.; Santin, A.D.; York, L. The CA 125 gene: An extracellular superstructure dominated by repeat sequences. Tumour. Biol. 2001, 22, 348–366. [Google Scholar] [CrossRef] [PubMed]
- Giamougiannis, P.; Martin-Hirsch, P.L.; Martin, F.L. The evolving role of MUC16 (CA125) in the transformation of ovarian cells and the progression of neoplasia. Carcinogenesis 2021, 42, 327–343. [Google Scholar] [CrossRef]
- Eiras, S.; de la Espriella, R.; Fu, X.; Iglesias-Alvarez, D.; Basdas, R.; Nunez-Caamano, J.R.; Gonzalez-Juanatey, J.R. Carbohydrate antigen 125 on epicardial fat and its association with local inflammation and fibrosis-related markers. J. Transl. Med. 2024, 22, 619. [Google Scholar] [CrossRef]
- Núñez, J.; Rabinovich, G.A.; Sandino, J.; Mainar, L.; Palau, P.; Santas, E.; Villanueva, M.P.; Núñez, E.; Bodí, V.; Chorro, F.J.; et al. Prognostic value of the interaction between galectin-3 and antigen carbohydrate 125 in acute heart failure. PLoS ONE 2015, 10, e0122360. [Google Scholar] [CrossRef]
- Revuelta-López, E.; de la Espriella, R.; Miñana, G.; Santas, E.; Villar, S.; Sanchis, J.; Bayés-Genís, A.; Núñez, J. The modulating effect of circulating carbohydrate antigen 125 on ST2 and long-term recurrent morbidity burden. Sci. Rep. 2025, 15, 1905. [Google Scholar] [CrossRef]
- Pandhi, P.; ter Maaten, J.M.; Anker, S.D.; Ng, L.L.; Metra, M.; Samani, N.J.; Lang, C.C.; Dickstein, K.; de Boer, R.A.; van Veldhuisen, D.J.; et al. Pathophysiologic Processes and Novel Biomarkers Associated With Congestion in Heart Failure. JACC Heart Fail. 2022, 10, 623–632. [Google Scholar] [CrossRef]
- Colombo, P.C.; Onat, D.; Harxhi, A.; Demmer, R.T.; Hayashi, Y.; Jelic, S.; LeJemtel, T.H.; Bucciarelli, L.; Kebschull, M.; Papapanou, P.; et al. Peripheral venous congestion causes inflammation, neuhormonal, and endothelial cell activation. Eur. Heart J. 2014, 35, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Damman, K.; Voors, A.A.; Hillege, H.L.; Navis, G.; Lechat, P.; van Veldhuisen, D.J.; Dargie, H.J. Congestion in chronic systolic heart failure is related to renal dysfunction and increased mortality. Eur. J. Heart Fail. 2010, 12, 974–982. [Google Scholar] [CrossRef]
- Mullens, W.; Abrahams, Z.; Francis, G.S.; Sokos, G.; Taylor, D.O.; Starling, R.C.; Young, J.B.; Tang, W.W. Importance of venous congestion for worsening renal function in advanced decompensated heart failure. J. Am. Coll. Cardiol. 2009, 53, 589–596. [Google Scholar] [CrossRef]
- Sandek, A.; Bauditz, J.; Swidsinski, A.; Buhner, S.; Weber-Eibel, J.; von Haehling, S.; Anker, S.D. The gut and heart failure: A target for therapeutic intervention? J. Am. Coll. Cardiol. 2007, 50, 1561–1569. [Google Scholar] [CrossRef]
- Llàcer, P.; Romero, G.; Trullàs, J.C.; de la Espriella, R.; Cobo, M.; Quiroga, B.; Casado, J.; Slon-Roblero, M.F.; Morales-Rull, J.L.; Morgado, J.I.; et al. Consensus on the approach to hydrosaline overload in acute heart failure. SEMI/SEC/S.E.N. recommendations. Rev. Esp. Cardiol. 2024, 77, 556–565. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Majhi, P.D.; Al-Mugotir, M.H.; Rachagani, S.; Sorgen, P.; Batra, S.K. Membrane proximal ectodomain cleavage of MUC16 occurs in the acidifying Golgi/post-Golgi compartments. Sci. Rep. 2015, 5, 9759. [Google Scholar] [CrossRef] [PubMed]
- Gubbels, J.A.A.; Felder, M.; Horibata, S.; Belisle, J.A.; Kapur, A.; Holden, H.; Petrie, S.; Migneault, M.; Rancourt, C.; Connor, J.P.; et al. MUC16 provides immune protection by inhibiting synapse formation between NK and ovarian tumor cells. Mol. Cancer 2010, 9, 11. [Google Scholar] [CrossRef]
- Das, S.; Rachagani, S.; Torres-Gonzalez, M.P.; Lakshmanan, I.; Majhi, P.D.; Smith, L.M.; Wagner, K.-U.; Batra, S.K. Carboxyl-terminal domain of MUC16 imparts tumorigenic and metastatic functions through nuclear translocation of JAK2 to pancreatic cancer cells. Oncotarget 2015, 6, 5772–5787. [Google Scholar] [CrossRef]
- Ballester, B.; Milara, J.; Montero, P.; Cortijo, J. MUC16 is overexpressed in idiopathic pulmonary fibrosis and induces fibrotic responses mediated by transforming growth Factor-β1 canonical pathway. Int. J. Mol. Sci 2021, 22, 6502. [Google Scholar] [CrossRef] [PubMed]
- Boulet, J.; Sridhar, V.S.; Bouabdallaoui, N.; Tardif, J.-C.; White, M. Inflammation in heart failure: Pathophysiology and therapeutic strategies. Inflamm. Res. 2024, 73, 709–723. [Google Scholar] [CrossRef] [PubMed]
| Total Cohort (n = 4043) | |
|---|---|
| Age, years a | 77 (68–83) |
| Female, n (%) | 1833 (45.3%) |
| Medical history | |
| Hypertension, n (%) | 3207 (79.3) |
| Diabetes mellitus, n (%) | 1769 (43.8) |
| Dyslipidemia, n (%) | 2175 (53.8) |
| HFpEF, n (%) | 748 (57.0) |
| eGFR (MDRD formula), mL/min/1.73 m2 | 62.5 ± 28.2 |
| Ischemic heart disease, n (%) | 1298 (32.1) |
| First hospital admission, n (%) | 2749 (68.0) |
| NYHA III-IV, n (%) | 662 (16.4) |
| Atrial fibrillation, n (%) | 1864 (46.1) |
| Vital signs | |
| Heart rate, bpm | 95.9 ± 27.5 |
| SBP, mmHg | 143.4 ± 31.0 |
| DBP, mmHg | 79.9 ±19.0 |
| Echocardiography | |
| TAPSE | 18.6 ± 3.7 |
| Septum, mm | 11.9 ± 2.7 |
| DTDVI, mm | 54.4 ± 9.3 |
| LVPW, mm | 11.3 ± 2.0 |
| Laboratory | |
| Hemoglobin, g/dL a | 12.5 (11.1–13.9) |
| Creatinine, mg/dL a | 1.1 (0.9–1.5) |
| Sodium, mEq/L | 138.3 ± 4.5 |
| NT-proBNP, pg/mL a | 3723.0 (1977.8–7694.0) |
| hs-CRP, mg/L a | 14.9 (6.0–36.2) |
| CA125, U/mL a | 50.2 (22.2–114.5) |
| Treatment | |
| ACE-I/ARB/ARNI, n (%) | 2423 (59.9) |
| MRA, n (%) | 1303 (32.2) |
| Diuretics prescribed at discharge, n (%) | 3754 (93.6) |
| Beta-blockers prescribed at discharge, n (%) | 2816 (69.7) |
| Statins prescribed at discharge, n (%) | 2030 (50.2) |
| Category 1 Low hs-CRP and Low CA125 (n = 795) | Category 2 High hs-CRP and Low CA125 (n = 786) | Category 3 Low hs-CRP and High CA125 (n = 1226) | Category 4 High hs-CRP and High CA125 (n = 1236) | p-Value | |
|---|---|---|---|---|---|
| Age, years | 75.8 ± 10.4 | 75.9 ± 10.3 | 73.5 ± 11.8 | 73.9 ± 11.2 | <0.001 |
| Male, n (%) | 386 (48.6) | 414 (52.7) | 697 (56.9) | 713 (57.7) | <0.001 |
| Medical history | |||||
| Hypertension, n (%) | 665 (83.6) | 682 (86.8) | 905 (73.8) | 955 (77.3) | <0.001 |
| Diabetes mellitus, n (%) | 341 (42.9) | 362 (46.1) | 519 (42.3) | 547 (44.3) | 0.382 |
| Dyslipidemia, n (%) | 470 (59.1) | 424 (53.9) | 628 (51.2) | 653 (52.8) | 0.005 |
| HFpEF, n (%) | 215 (65.5) | 217 (70.5) | 134 (41.6) | 182 (51.3) | <0.001 |
| eGFR (MDRD formula), mL/min/1,73 m2 | 65.0 ± 26.7 | 60.4 ± 26.1 | 64.9 ± 30.7 | 59.8 ± 27.4 | <0.001 |
| Ischemic heart disease, n (%) | 266 (33.5) | 258 (32.8) | 373 (30.4) | 401 (32.4) | 0.474 |
| Peripheral edema, n (%) | 406 (51.1) | 424 (53.9) | 832 (67.9) | 866 (70.1) | <0.001 |
| Pleural effusion, n (%) | 253 (31.8) | 259 (33) | 730 (59.5) | 741 (60) | <0.001 |
| First hospital admission, n (%) | 556 (69.9) | 488 (62.1) | 883 (72) | 822 (66.5) | <0.001 |
| NYHA III-IV, n (%) | 106 (13.3) | 137 (17.4) | 187 (15.3) | 232 (18.8) | 0.006 |
| Atrial fibrillation, n (%) | 324 (40.8) | 335 (42.6) | 588 (48.0) | 617 (49.9) | <0.001 |
| Vital signs | |||||
| Heart rate, bpm | 92.2 ± 26.9 | 94.9 ± 27.1 | 96.4 ± 27.6 | 98.5 ± 27.8 | <0.001 |
| SBP, mmHg | 146.8 ± 31.4 | 146.1 ± 31.7 | 141.7 ± 29.9 | 141.1 ± 30.9 | <0.001 |
| DBP, mmHg | 81.3 ± 20.3 | 79.3 ± 19.2 | 80.2 ± 17.9 | 79.0 ± 18.9 | 0.049 |
| Echocardiography | |||||
| TAPSE | 19.7 ± 3.6 | 19.5 ± 3.4 | 17.9 ± 3.6 | 18.0 ± 3.8 | <0.001 |
| Septum, mm | 12.4 ± 3.3 | 12.1 ± 2.4 | 11.6 ± 2.6 | 11.8 ± 2.6 | <0.001 |
| DTDVI, mm | 54.0 ± 9.2 | 53.1 ± 8.8 | 55.2 ± 9.5 | 54.7 ± 9.3 | <0.001 |
| LVPW, mm | 11.6 ± 1.9 | 11.5 ± 1.9 | 11.1 ± 2.1 | 11.3 ± 2.0 | <0.001 |
| Laboratory | |||||
| Hemoglobin, g/dL a | 12.9 (11.6–14.1) | 12.3 (11.0–13.7) | 12.6 (11.3–13.9) | 12.0 (10.8–13.6) | <0.001 |
| Creatinine, mg/dL a | 1.1 (0.8–1.4) | 1.1 (0.9–1.5) | 1.1 (0.9–1.4) | 1.2 (0.9–1.6) | <0.001 |
| Sodium, mEq/L | 138.8 ± 3.9 | 138.4 ± 4.4 | 138.8 ± 4.6 | 137.5 ± 4.8 | <0.001 |
| NT-proBNP, pg/mL a | 2541.4 (1292–4583) | 3254.6 (1791–6000) | 3889.8 (2093–7843) | 5283.8 (2765.8–10,566) | <0.001 |
| hs-CRP, mg/L | 6.0 ± 3.8 | 58.7 ± 52.9 | 6.8 ± 4 | 53.9 ± 52 | <0.001 |
| CA125, U/mL a | 18.1 (11.6–25.9) | 18.7 (12.7–26) | 96.5 (58.8–174) | 95.1 (56.8–159) | <0.001 |
| Treatment | |||||
| ACE-I/ARB/ARNI, n (%) | 505 (63.5) | 458 (58.3) | 756 (61.7) | 704 (57) | 0.010 |
| MRA, n (%) | 259 (32.6) | 265 (33.7) | 417 (34.0) | 362 (29.3) | 0.057 |
| Diuretics prescribed at discharge, n (%) | 719 (90.9) | 724 (93.3) | 1162 (95.3) | 1149 (93.8) | 0.001 |
| Beta-blockers prescribed at discharge, n (%) | 568 (71.4) | 513 (65.3) | 886 (72.3) | 849 (68.7) | 0.005 |
| Statins prescribed at discharge, n (%) | 465 (58.5) | 389 (49.5) | 606 (49.4) | 570 (46.1) | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santas, E.; Martí-Martínez, A.; Revuelta-López, E.; Villar, S.; de la Espriella, R.; Palau, P.; Llàcer, P.; Miñana, G.; Rodriguez-Borja, E.; Carratalá, A.; et al. Modulating Effect of Carbohydrate Antigen 125 on the Prognostic Value of High-Sensitivity C-Reactive Protein in Heart Failure. Biomolecules 2025, 15, 1260. https://doi.org/10.3390/biom15091260
Santas E, Martí-Martínez A, Revuelta-López E, Villar S, de la Espriella R, Palau P, Llàcer P, Miñana G, Rodriguez-Borja E, Carratalá A, et al. Modulating Effect of Carbohydrate Antigen 125 on the Prognostic Value of High-Sensitivity C-Reactive Protein in Heart Failure. Biomolecules. 2025; 15(9):1260. https://doi.org/10.3390/biom15091260
Chicago/Turabian StyleSantas, Enrique, Arancha Martí-Martínez, Elena Revuelta-López, Sandra Villar, Rafael de la Espriella, Patricia Palau, Pau Llàcer, Gema Miñana, Enrique Rodriguez-Borja, Arturo Carratalá, and et al. 2025. "Modulating Effect of Carbohydrate Antigen 125 on the Prognostic Value of High-Sensitivity C-Reactive Protein in Heart Failure" Biomolecules 15, no. 9: 1260. https://doi.org/10.3390/biom15091260
APA StyleSantas, E., Martí-Martínez, A., Revuelta-López, E., Villar, S., de la Espriella, R., Palau, P., Llàcer, P., Miñana, G., Rodriguez-Borja, E., Carratalá, A., Gonzalez, A., Bayés-Genís, A., Sanchis, J., & Núñez, J. (2025). Modulating Effect of Carbohydrate Antigen 125 on the Prognostic Value of High-Sensitivity C-Reactive Protein in Heart Failure. Biomolecules, 15(9), 1260. https://doi.org/10.3390/biom15091260






