A Multi-Ingredient Supplement Improves Body Re-Composition, Ovarian Aging Markers, and Reproductive Success in Young and Middle-Aged Female Mice
Abstract
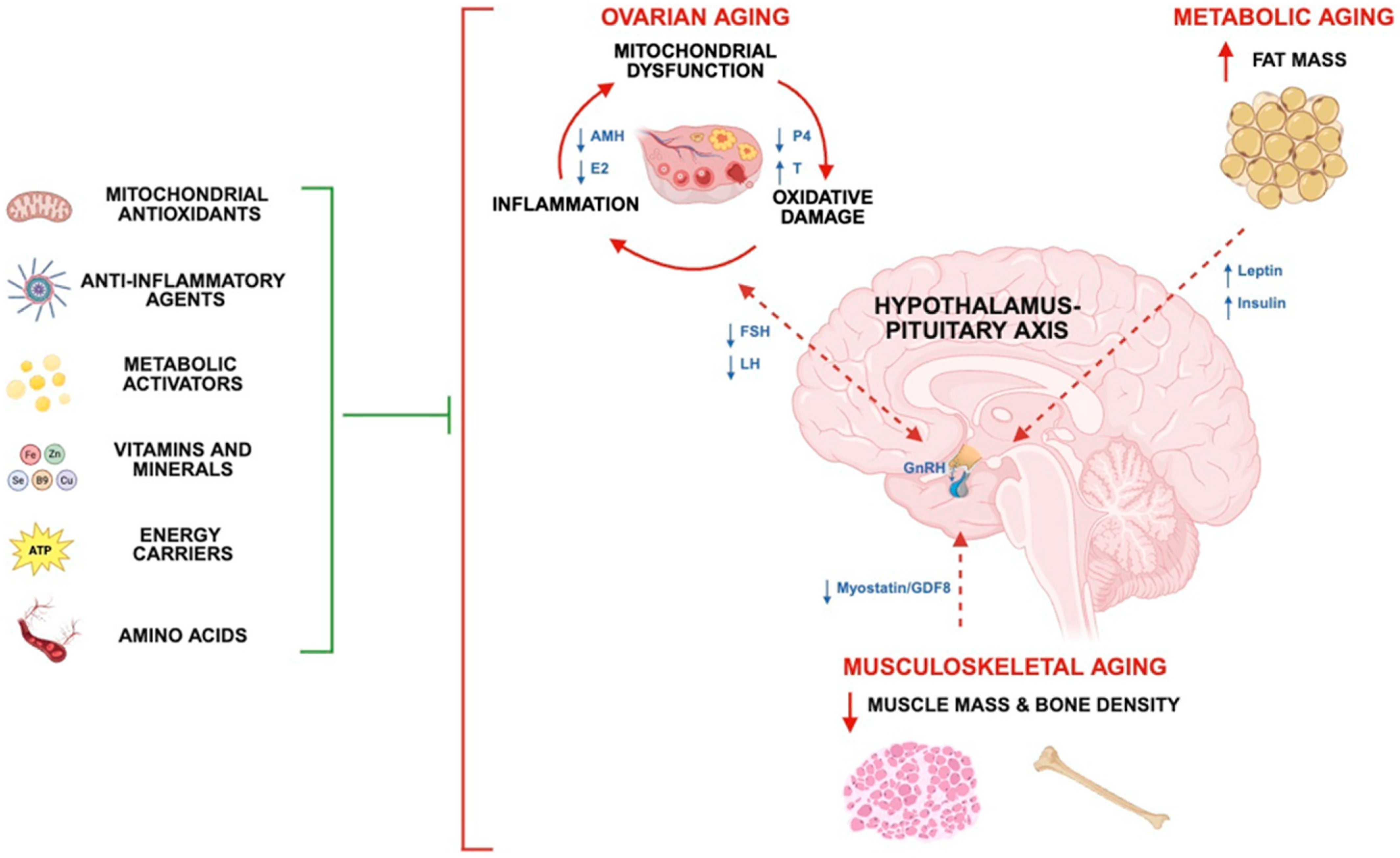
1. Introduction
2. Methods
2.1. Ethics Approval
2.2. Animals, Cohorts, and General Study Design
2.3. Diets and Nutraceuticals
2.4. Mating
2.5. Time-Domain NMR Whole-Body Composition
2.6. Metabolic Activity Analysis
2.7. Ovarian Immunoblotting
2.8. Ovarian Quality Index
2.9. Statistical Analyses
3. Results
3.1. Food Intakes
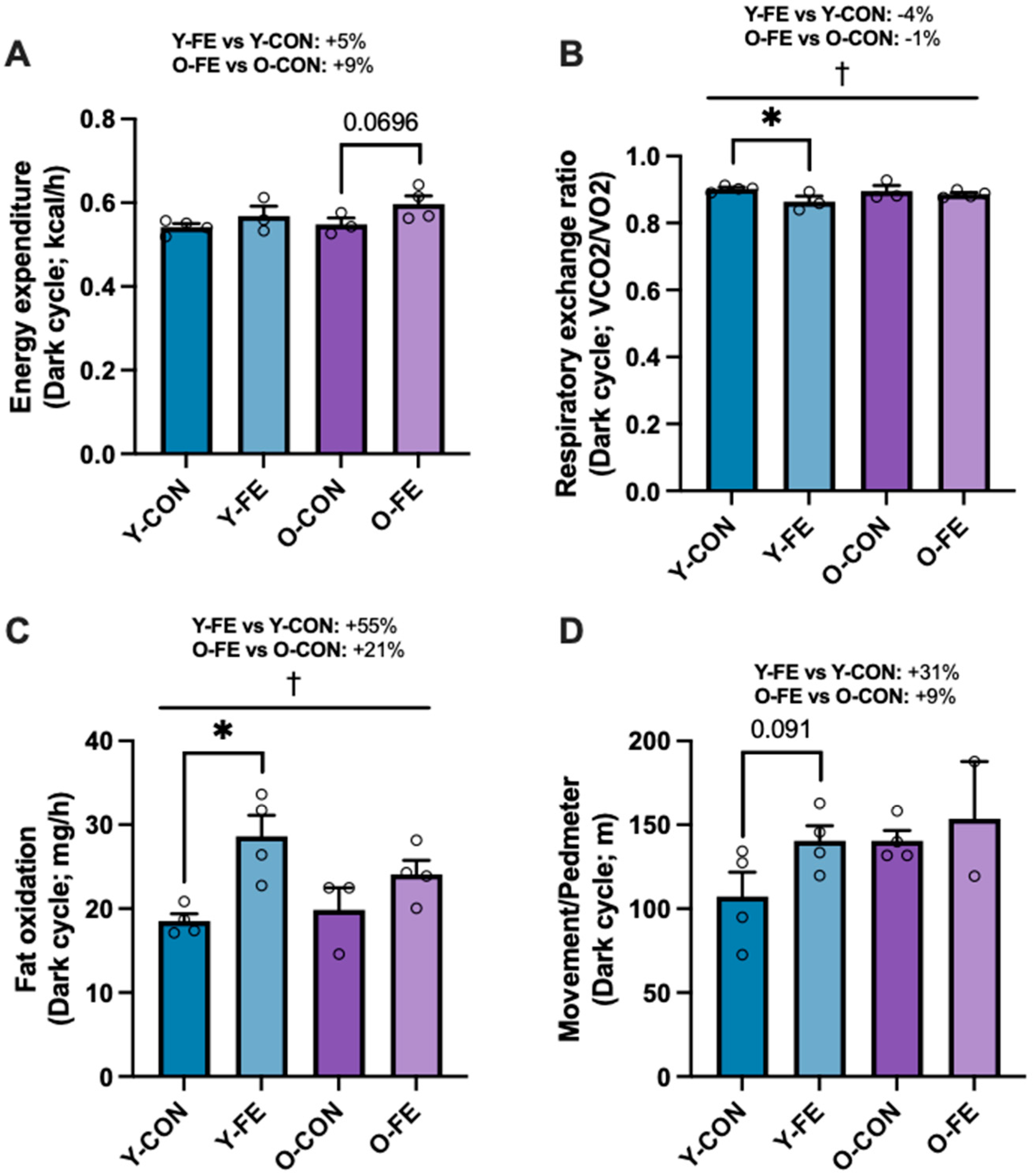
3.2. Metabolic Activity
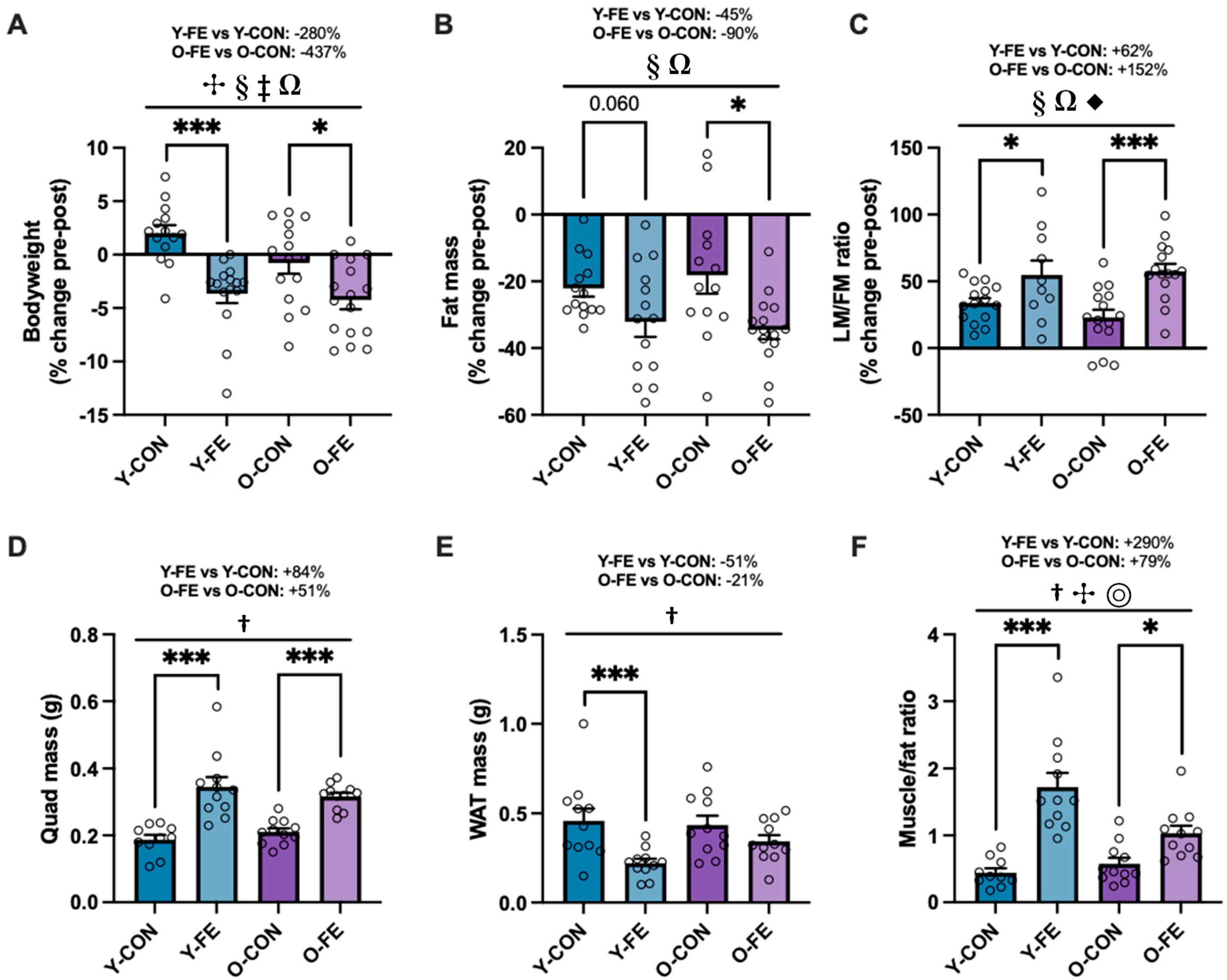
3.3. Anthropometry and Body Composition
3.4. Reproduction
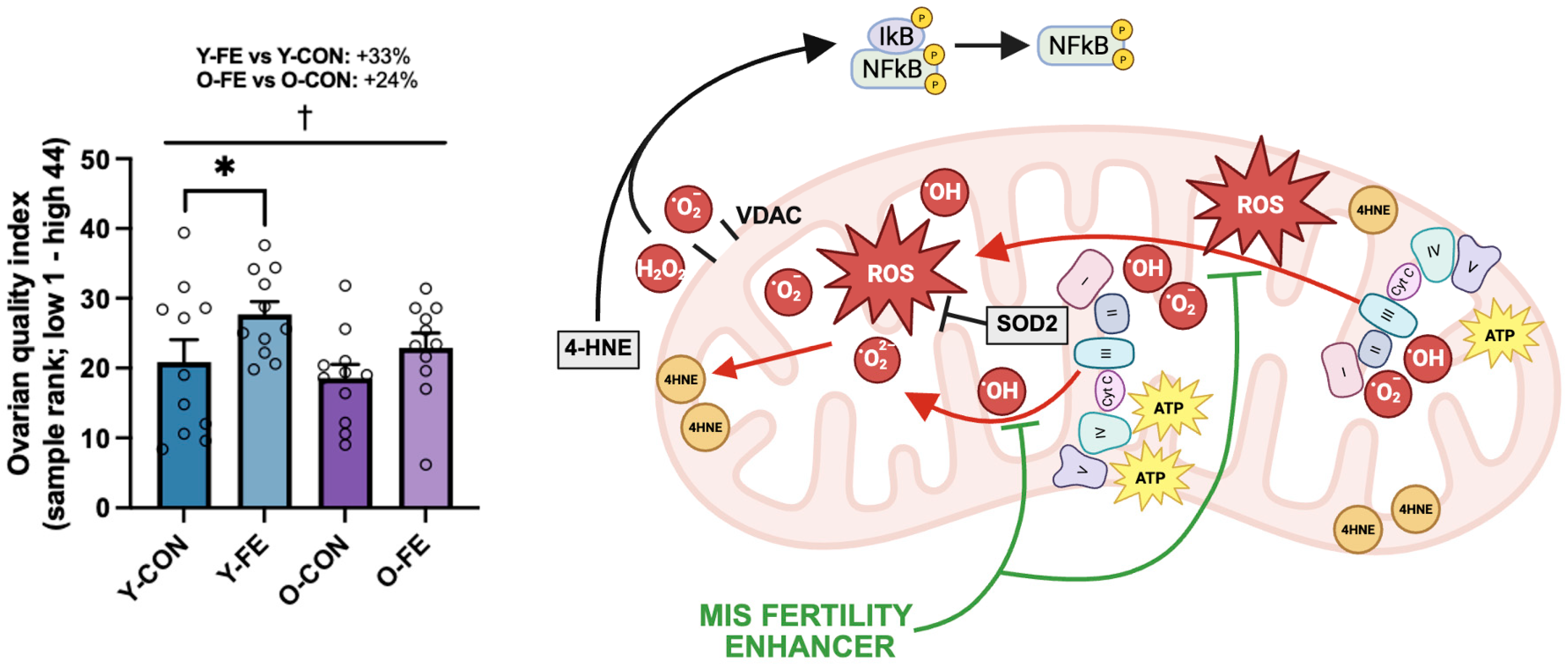
3.5. Ovarian Aging Markers
3.6. Ovarian Quality Index
3.7. Female Fertility Predictors
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nelson, S.M.; Telfer, E.E.; Anderson, R.A. The ageing ovary and uterus: New biological insights. Hum. Reprod. Update 2013, 19, 67–83. [Google Scholar] [CrossRef]
- Wu, J.; Liu, Y.; Song, Y.; Wang, L.; Ai, J.; Li, K. Aging conundrum: A perspective for ovarian aging. Front. Endocrinol. 2022, 13, 952471. [Google Scholar] [CrossRef]
- Wang, S.; Zheng, Y.; Li, J.; Yu, Y.; Zhang, W.; Song, M.; Liu, Z.; Min, Z.; Hu, H.; Jing, Y.; et al. Single-Cell Transcriptomic Atlas of Primate Ovarian Aging. Cell 2020, 180, 585–600.e19. [Google Scholar] [CrossRef]
- Jin, C.; Wang, X.; Yang, J.; Kim, S.; Hudgins, A.D.; Gamliel, A.; Pei, M.; Contreras, D.; Devos, M.; Guo, Q.; et al. Molecular and genetic insights into human ovarian aging from single-nuclei multi-omics analyses. Nat. Aging 2025, 5, 275–290. [Google Scholar] [CrossRef]
- Menken, J.; Trussell, J.; Larsen, U. Age and infertility. Science 1986, 233, 1389–1394. [Google Scholar] [CrossRef] [PubMed]
- Homan, G.F.; Davies, M.; Norman, R. The impact of lifestyle factors on reproductive performance in the general population and those undergoing infertility treatment: A review. Hum. Reprod. Update 2007, 13, 209–223. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Biedenharn, K.R.; Fedor, J.M.; Agarwal, A. Lifestyle factors and reproductive health: Taking control of your fertility. Reprod. Biol. Endocrinol. 2013, 11, 66. [Google Scholar] [CrossRef] [PubMed]
- Jura, M.; Kozak, L.P. Obesity and related consequences to ageing. Age 2016, 38, 23. [Google Scholar] [CrossRef]
- Eng, P.C.; Phylactou, M.; Qayum, A.; Woods, C.; Lee, H.; Aziz, S.; Moore, B.; Miras, A.D.; Comninos, A.N.; Tan, T.; et al. Obesity-Related Hypogonadism in Women. Endocr. Rev. 2024, 45, 171–189. [Google Scholar] [CrossRef]
- Ongaro, L.; Zhou, X.; Wang, Y.; Schultz, H.; Zhou, Z.; Buddle, E.R.S.; Brûlé, E.; Lin, Y.-F.; Schang, G.; Hagg, A.; et al. Muscle-derived myostatin is a major endocrine driver of follicle-stimulating hormone synthesis. Science 2025, 387, 329–336. [Google Scholar] [CrossRef]
- Steenwinkel, T.E.; Pangas, S.A. Myostatin’s flex on the reproductive hormone axis. Science 2025, 387, 249–250. [Google Scholar] [CrossRef]
- Nederveen, J.P.; Mastrolonardo, A.J.; Xhuti, D.; Di Carlo, A.; Manta, K.; Fuda, M.R.; Tarnopolsky, M.A. Novel Multi-Ingredient Supplement Facilitates Weight Loss and Improves Body Composition in Overweight and Obese Individuals: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Nutrients 2023, 15, 3693. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, M.I.; Xhuti, D.; de Maat, N.M.; Hettinga, B.P.; Tarnopolsky, M.A. Obesity and Metabolic Disease Impair the Anabolic Response to Protein Supplementation and Resistance Exercise: A Retrospective Analysis of a Randomized Clinical Trial with Implications for Aging, Sarcopenic Obesity, and Weight Management. Nutrients 2024, 16, 4407. [Google Scholar] [CrossRef]
- Chambers, G.M.; Sullivan, E.A.; Ishihara, O.; Chapman, M.G.; Adamson, G.D. The economic impact of assisted reproductive technology: A review of selected developed countries. Fertil. Steril. 2009, 91, 2281–2294. [Google Scholar] [CrossRef] [PubMed]
- Nachtigall, R.D. International disparities in access to infertility services. Fertil. Steril. 2006, 85, 871–875. [Google Scholar] [CrossRef] [PubMed]
- Njagi, P.; Groot, W.; Arsenijevic, J.; Dyer, S.; Mburu, G.; Kiarie, J. Financial costs of assisted reproductive technology for patients in low- and middle-income countries: A systematic review. Hum. Reprod. Open 2023, 2023, hoad007. [Google Scholar] [CrossRef]
- Dwyer, J.T.; Coates, P.M.; Smith, M.J. Dietary Supplements: Regulatory Challenges and Research Resources. Nutrients 2018, 10, 41. [Google Scholar] [CrossRef]
- Vitagliano, A.; Petre, G.C.; Francini-Pesenti, F.; De Toni, L.; Di Nisio, A.; Grande, G.; Foresta, C.; Garolla, A. Dietary Supplements for Female Infertility: A Critical Review of Their Composition. Nutrients 2021, 13, 3552. [Google Scholar] [CrossRef]
- WHO Antenatal Care Recommendations for a Positive Pregnancy Experience. Nutritional Interventions Update: Multiple Micronutrient Supplements During Pregnancy; WHO: Geneva, Switzerland, 2020; pp. 1–68. [Google Scholar]
- De-Regil, L.M.; Pena-Rosas, J.P.; Fernandez-Gaxiola, A.C.; Rayco-Solon, P. Effects and safety of periconceptional oral folate supplementation for preventing birth defects. Cochrane Database Syst. Rev. 2015, 2015, CD007950. [Google Scholar] [CrossRef]
- Haider, B.A.; Olofin, I.; Wang, M.; Spiegelman, D.; Ezzati, M.; Fawzi, W.W. Anaemia, prenatal iron use, and risk of adverse pregnancy outcomes: Systematic review and meta-analysis. BMJ Br. Med. J. 2013, 346, f3443. [Google Scholar] [CrossRef]
- Pena-Rosas, J.P.; De-Regil, L.M.; Garcia-Casal, M.N.; Dowswell, T. Daily oral iron supplementation during pregnancy. Cochrane Database Syst. Rev. 2015, 12, CD004736. [Google Scholar] [CrossRef]
- Keats, E.C.; Haider, B.A.; Tam, E.; Bhutta, Z.A. Multiple-micronutrient supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2019, 3, CD004905. [Google Scholar] [CrossRef]
- Brough, L.; Rees, G.A.; Crawford, M.A.; Morton, R.H.; Dorman, E.K. Effect of multiple-micronutrient supplementation on maternal nutrient status, infant birth weight and gestational age at birth in a low-income, multi-ethnic population. Br. J. Nutr. 2010, 104, 437–445. [Google Scholar] [CrossRef]
- Hininger, I.; Favier, M.; Arnaud, J.; Faure, H.; Thoulon, J.M.; Hariveau, E.; Favier, A.; Roussel, A.M. Effects of a combined micronutrient supplementation on maternal biological status and newborn anthropometrics measurements: A randomized double-blind, placebo-controlled trial in apparently healthy pregnant women. Eur. J. Clin. Nutr. 2004, 58, 52–59. [Google Scholar] [CrossRef]
- Scholl, T.O.; Hediger, M.L.; Bendich, A.; Schall, J.I.; Smith, W.K.; Krueger, P.M. Use of Multivitamin/Mineral Prenatal Supplements: Influence on the Outcome of Pregnancy. Am. J. Epidemiol. 1997, 146, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Kapper, C.; Stelzl, P.; Oppelt, P.; Ganhör, C.; Gyunesh, A.A.; Arbeithuber, B.; Rezk-Füreder, M. The Impact of Minerals on Female Fertility: A Systematic Review. Nutrients 2024, 16, 4068. [Google Scholar] [CrossRef] [PubMed]
- Heshmati, J.; Morvaridzadeh, M.; Maroufizadeh, S.; Akbari, A.; Yavari, M.; Amirinejad, A.; Maleki-Hajiagha, A.; Sepidarkish, M. Omega-3 fatty acids supplementation and oxidative stress parameters: A systematic review and meta-analysis of clinical trials. Pharmacol. Res. 2019, 149, 104462. [Google Scholar] [CrossRef]
- Sley, E.G.; Rosen, E.M.; van ‘t Erve, T.J.; Sathyanarayana, S.; Barrett, E.S.; Nguyen, R.H.N.; Bush, N.R.; Milne, G.L.; Swan, S.H.; Ferguson, K.K. Omega-3 fatty acid supplement use and oxidative stress levels in pregnancy. PLoS ONE 2020, 15, e0240244. [Google Scholar] [CrossRef] [PubMed]
- Haghiac, M.; Yang, X.H.; Presley, L.; Smith, S.; Dettelback, S.; Minium, J.; Belury, M.A.; Catalano, P.M.; Hauguel-de Mouzon, S. Dietary Omega-3 Fatty Acid Supplementation Reduces Inflammation in Obese Pregnant Women: A Randomized Double-Blind Controlled Clinical Trial. PLoS ONE 2015, 10, e0137309. [Google Scholar] [CrossRef]
- Kavyani, Z.; Musazadeh, V.; Fathi, S.; Hossein Faghfouri, A.; Dehghan, P.; Sarmadi, B. Efficacy of the omega-3 fatty acids supplementation on inflammatory biomarkers: An umbrella meta-analysis. Int. Immunopharmacol. 2022, 111, 109104. [Google Scholar] [CrossRef]
- Li, K.; Huang, T.; Zheng, J.; Wu, K.; Li, D. Effect of marine-derived n-3 polyunsaturated fatty acids on C-reactive protein, interleukin 6 and tumor necrosis factor α: A meta-analysis. PLoS ONE 2014, 9, e88103. [Google Scholar] [CrossRef] [PubMed]
- Bernasconi, A.A.; Wiest, M.M.; Lavie, C.J.; Milani, R.V.; Laukkanen, J.A. Effect of Omega-3 Dosage on Cardiovascular Outcomes: An Updated Meta-Analysis and Meta-Regression of Interventional Trials. Mayo Clin. Proc. 2021, 96, 304–313. [Google Scholar] [CrossRef]
- Khan, S.U.; Lone, A.N.; Khan, M.S.; Virani, S.S.; Blumenthal, R.S.; Nasir, K.; Miller, M.; Michos, E.D.; Ballantyne, C.M.; Boden, W.E.; et al. Effect of omega-3 fatty acids on cardiovascular outcomes: A systematic review and meta-analysis. eClinicalMedicine 2021, 38, 100997. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.X.; Yu, J.H.; Sun, J.H.; Ma, W.Q.; Wang, J.J.; Sun, G.J. Effects of Omega-3 Fatty Acids Supplementation on Serum Lipid Profile and Blood Pressure in Patients with Metabolic Syndrome: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Foods 2023, 12, 725. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y.; Shehzad, Q.; Su, Y.; Xu, L.; Yu, L.; Zeng, W.; Fang, Z.; Wu, G.; Wei, W.; et al. Does omega-3 PUFAs supplementation improve metabolic syndrome and related cardiovascular diseases? A systematic review and meta-analysis of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 2024, 64, 9455–9482. [Google Scholar] [CrossRef]
- Delpino, F.M.; Figueiredo, L.M.; da Silva, B.G.C.; da Silva, T.G.; Mintem, G.C.; Bielemann, R.M.; Gigante, D.P. Omega-3 supplementation and diabetes: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2022, 62, 4435–4448. [Google Scholar] [CrossRef]
- O’Mahoney, L.L.; Matu, J.; Price, O.J.; Birch, K.M.; Ajjan, R.A.; Farrar, D.; Tapp, R.; West, D.J.; Deighton, K.; Campbell, M.D. Omega-3 polyunsaturated fatty acids favourably modulate cardiometabolic biomarkers in type 2 diabetes: A meta-analysis and meta-regression of randomized controlled trials. Cardiovasc. Diabetol. 2018, 17, 98. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, X. Meta-analysis of the efficacy of ω-3 polyunsaturated fatty acids when treating patients with polycystic ovary syndrome. Medicine 2023, 102, e35403. [Google Scholar] [CrossRef]
- Yang, K.; Zeng, L.; Bao, T.; Ge, J. Effectiveness of Omega-3 fatty acid for polycystic ovary syndrome: A systematic review and meta-analysis. Reprod. Biol. Endocrinol. 2018, 16, 27. [Google Scholar] [CrossRef]
- Zhou, J.; Zuo, W.; Tan, Y.; Wang, X.; Zhu, M.; Zhang, H. Effects of n-3 polyunsaturated fatty acid on metabolic status in women with polycystic ovary syndrome: A meta-analysis of randomized controlled trials. J. Ovarian Res. 2023, 16, 54. [Google Scholar] [CrossRef] [PubMed]
- Cetin, I.; Carlson, S.E.; Burden, C.; da Fonseca, E.B.; di Renzo, G.C.; Hadjipanayis, A.; Harris, W.S.; Kumar, K.R.; Olsen, S.F.; Mader, S.; et al. Omega-3 fatty acid supply in pregnancy for risk reduction of preterm and early preterm birth. Am. J. Obstet. Gynecol. MFM 2024, 6, 101251. [Google Scholar] [CrossRef]
- Kar, S.; Wong, M.; Rogozinska, E.; Thangaratinam, S. Effects of omega-3 fatty acids in prevention of early preterm delivery: A systematic review and meta-analysis of randomized studies. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 198, 40–46. [Google Scholar] [CrossRef]
- Middleton, P.; Gomersall, J.C.; Gould, J.F.; Shepherd, E.; Olsen, S.F.; Makrides, M. Omega 3 fatty acid addition during pregnancy. Cochrane Database Syst. Rev. 2018, 11, CD003402. [Google Scholar] [CrossRef] [PubMed]
- Lawn, J.E.; Kinney, M. Preterm birth: Now the leading cause of child death worldwide. Sci. Transl. Med. 2014, 6, 263ed21. [Google Scholar] [CrossRef] [PubMed]
- Florou, P.; Anagnostis, P.; Theocharis, P.; Chourdakis, M.; Goulis, D.G. Does coenzyme Q(10) supplementation improve fertility outcomes in women undergoing assisted reproductive technology procedures? A systematic review and meta-analysis of randomized-controlled trials. J. Assist. Reprod. Genet. 2020, 37, 2377–2387. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Mariscal, F.M.; Yubero-Serrano, E.M.; Villalba, J.M.; Lopez-Miranda, J. Coenzyme Q(10): From bench to clinic in aging diseases, a translational review. Crit. Rev. Food Sci. Nutr. 2019, 59, 2240–2257. [Google Scholar] [CrossRef]
- Shang, Y.; Song, N.; He, R.; Wu, M. Antioxidants and Fertility in Women with Ovarian Aging: A Systematic Review and Meta-Analysis. Adv. Nutr. 2024, 15, 100273. [Google Scholar] [CrossRef]
- Zhang, T.; He, Q.; Xiu, H.; Zhang, Z.; Liu, Y.; Chen, Z.; Hu, H. Efficacy and Safety of Coenzyme Q10 Supplementation in the Treatment of Polycystic Ovary Syndrome: A Systematic Review and Meta-analysis. Reprod. Sci. 2023, 30, 1033–1048. [Google Scholar] [CrossRef]
- Liang, Y.; Zhao, D.; Ji, Q.; Liu, M.; Dai, S.; Hou, S.; Liu, Z.; Mao, Y.; Tian, Z.; Yang, Y. Effects of coenzyme Q10 supplementation on glycemic control: A GRADE-assessed systematic review and dose-response meta-analysis of randomized controlled trials. EClinicalMedicine 2022, 52, 101602. [Google Scholar] [CrossRef]
- Stojanović, M.; Radenković, M. A meta-analysis of randomized and placebo-controlled clinical trials suggests that coenzyme Q10 at low dose improves glucose and HbA1c levels. Nutr. Res. 2017, 38, 1–12. [Google Scholar] [CrossRef]
- Dludla, P.V.; Orlando, P.; Silvestri, S.; Marcheggiani, F.; Cirilli, I.; Nyambuya, T.M.; Mxinwa, V.; Mokgalaboni, K.; Nkambule, B.B.; Johnson, R.; et al. Coenzyme Q(10) Supplementation Improves Adipokine Levels and Alleviates Inflammation and Lipid Peroxidation in Conditions of Metabolic Syndrome: A Meta-Analysis of Randomized Controlled Trials. Int. J. Mol. Sci. 2020, 21, 3247. [Google Scholar] [CrossRef]
- Fan, L.; Feng, Y.; Chen, G.C.; Qin, L.Q.; Fu, C.L.; Chen, L.H. Effects of coenzyme Q10 supplementation on inflammatory markers: A systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 2017, 119, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Hou, S.; Tian, Z.; Zhao, D.; Liang, Y.; Dai, S.; Ji, Q.; Fan, Z.; Liu, Z.; Liu, M.; Yang, Y. Efficacy and Optimal Dose of Coenzyme Q10 Supplementation on Inflammation-Related Biomarkers: A GRADE-Assessed Systematic Review and Updated Meta-Analysis of Randomized Controlled Trials. Mol. Nutr. Food Res. 2023, 67, e2200800. [Google Scholar] [CrossRef]
- Dai, S.; Tian, Z.; Zhao, D.; Liang, Y.; Liu, M.; Liu, Z.; Hou, S.; Yang, Y. Effects of Coenzyme Q10 Supplementation on Biomarkers of Oxidative Stress in Adults: A GRADE-Assessed Systematic Review and Updated Meta-Analysis of Randomized Controlled Trials. Antioxidants 2022, 11, 1360. [Google Scholar] [CrossRef]
- Dludla, P.V.; Nyambuya, T.M.; Orlando, P.; Silvestri, S.; Mxinwa, V.; Mokgalaboni, K.; Nkambule, B.B.; Louw, J.; Muller, C.J.F.; Tiano, L. The impact of coenzyme Q(10) on metabolic and cardiovascular disease profiles in diabetic patients: A systematic review and meta-analysis of randomized controlled trials. Endocrinol. Diabetes Metab. 2020, 3, e00118. [Google Scholar] [CrossRef]
- Liu, Z.; Tian, Z.; Zhao, D.; Liang, Y.; Dai, S.; Liu, M.; Hou, S.; Dong, X.; Zhaxinima; Yang, Y. Effects of Coenzyme Q10 Supplementation on Lipid Profiles in Adults: A Meta-analysis of Randomized Controlled Trials. J. Clin. Endocrinol. Metab. 2022, 108, 232–249. [Google Scholar] [CrossRef]
- Jiang, Q. Natural forms of vitamin E: Metabolism, antioxidant, and anti-inflammatory activities and their role in disease prevention and therapy. Free. Radic. Biol. Med. 2014, 72, 76–90. [Google Scholar] [CrossRef] [PubMed]
- Kagan, V.E.; Fabisiak, J.P.; Quinn, P.J. Coenzyme Q and vitamin E need each other as antioxidants. Protoplasma 2000, 214, 11–18. [Google Scholar] [CrossRef]
- Kagan, V.E.; Serbinova, E.A.; Forte, T.; Scita, G.; Packer, L. Recycling of vitamin E in human low density lipoproteins. J. Lipid Res. 1992, 33, 385–397. [Google Scholar] [CrossRef]
- Lauridsen, C.; Jensen, S.K. α-Tocopherol incorporation in mitochondria and microsomes upon supranutritional vitamin E supplementation. Genes Nutr. 2012, 7, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Sohal, R.S. Coenzyme Q and vitamin E interactions. Methods Enzymol. 2004, 378, 146–151. [Google Scholar] [PubMed]
- Asbaghi, O.; Sadeghian, M.; Nazarian, B.; Sarreshtedari, M.; Mozaffari-Khosravi, H.; Maleki, V.; Alizadeh, M.; Shokri, A.; Sadeghi, O. The effect of vitamin E supplementation on selected inflammatory biomarkers in adults: A systematic review and meta-analysis of randomized clinical trials. Sci. Rep. 2020, 10, 17234. [Google Scholar] [CrossRef]
- Tefagh, G.; Payab, M.; Qorbani, M.; Sharifi, F.; Sharifi, Y.; Ebrahimnegad Shirvani, M.S.; Pourghazi, F.; Atlasi, R.; Shadman, Z.; Rezaei, N.; et al. Effect of vitamin E supplementation on cardiometabolic risk factors, inflammatory and oxidative markers and hormonal functions in PCOS (polycystic ovary syndrome): A systematic review and meta-analysis. Sci. Rep. 2022, 12, 5770. [Google Scholar] [CrossRef]
- Solmonson, A.; DeBerardinis, R.J. Lipoic acid metabolism and mitochondrial redox regulation. J. Biol. Chem. 2018, 293, 7522–7530. [Google Scholar] [CrossRef]
- Kucukgoncu, S.; Zhou, E.; Lucas, K.B.; Tek, C. Alpha-lipoic acid (ALA) as a supplementation for weight loss: Results from a meta-analysis of randomized controlled trials. Obes. Rev. 2017, 18, 594–601. [Google Scholar] [CrossRef]
- Namazi, N.; Larijani, B.; Azadbakht, L. Alpha-lipoic acid supplement in obesity treatment: A systematic review and meta-analysis of clinical trials. Clin. Nutr. 2017, 37, 419–428. [Google Scholar] [CrossRef]
- Vajdi, M.; Abbasalizad Farhangi, M. Alpha-lipoic acid supplementation significantly reduces the risk of obesity in an updated systematic review and dose response meta-analysis of randomised placebo-controlled clinical trials. Int. J. Clin. Pract. 2020, 74, e13493. [Google Scholar] [CrossRef] [PubMed]
- Akbari, M.; Ostadmohammadi, V.; Tabrizi, R.; Mobini, M.; Lankarani, K.B.; Moosazadeh, M.; Heydari, S.T.; Chamani, M.; Kolahdooz, F.; Asemi, Z. The effects of alpha-lipoic acid supplementation on inflammatory markers among patients with metabolic syndrome and related disorders: A systematic review and meta-analysis of randomized controlled trials. Nutr. Metab. 2018, 15, 39. [Google Scholar] [CrossRef]
- Akbari, M.; Ostadmohammadi, V.; Lankarani, K.B.; Tabrizi, R.; Kolahdooz, F.; Khatibi, S.R.; Asemi, Z. The effects of alpha-lipoic acid supplementation on glucose control and lipid profiles among patients with metabolic diseases: A systematic review and meta-analysis of randomized controlled trials. Metab. Clin. Exp. 2018, 87, 56–69. [Google Scholar] [CrossRef] [PubMed]
- Guarano, A.; Capozzi, A.; Cristodoro, M.; Di Simone, N.; Lello, S. Alpha Lipoic Acid Efficacy in PCOS Treatment: What Is the Truth? Nutrients 2023, 15, 3209. [Google Scholar] [CrossRef] [PubMed]
- de Guingand, D.L.; Palmer, K.R.; Bilardi, J.E.; Ellery, S.J. Acceptability of dietary or nutritional supplementation in pregnancy (ADONS)—Exploring the consumer’s perspective on introducing creatine monohydrate as a pregnancy supplement. Midwifery 2020, 82, 102599. [Google Scholar] [CrossRef]
- Dickinson, H.; Davies-Tuck, M.; Ellery, S.J.; Grieger, J.A.; Wallace, E.M.; Snow, R.J.; Walker, D.W.; Clifton, V.L. Maternal creatine in pregnancy: A retrospective cohort study. BJOG Int. J. Obstet. Gynaecol. 2016, 123, 1830–1838. [Google Scholar] [CrossRef]
- Dickinson, H.; Ellery, S.; Ireland, Z.; LaRosa, D.; Snow, R.; Walker, D.W. Creatine supplementation during pregnancy: Summary of experimental studies suggesting a treatment to improve fetal and neonatal morbidity and reduce mortality in high-risk human pregnancy. BMC Pregnancy Childbirth 2014, 14, 150. [Google Scholar] [CrossRef]
- Kreider, R.B.; Kalman, D.S.; Antonio, J.; Ziegenfuss, T.N.; Wildman, R.; Collins, R.; Candow, D.G.; Kleiner, S.M.; Almada, A.L.; Lopez, H.L. International Society of Sports Nutrition position stand: Safety and efficacy of creatine supplementation in exercise, sport, and medicine. J. Int. Soc. Sports Nutr. 2017, 14, 18. [Google Scholar] [CrossRef] [PubMed]
- Muccini, A.M.; Tran, N.T.; de Guingand, D.L.; Philip, M.; Della Gatta, P.A.; Galinsky, R.; Sherman, L.S.; Kelleher, M.A.; Palmer, K.R.; Berry, M.J.; et al. Creatine Metabolism in Female Reproduction, Pregnancy and Newborn Health. Nutrients 2021, 13, 490. [Google Scholar] [CrossRef] [PubMed]
- Asbaghi, O.; Rezaei Kelishadi, M.; Larky, D.A.; Bagheri, R.; Amirani, N.; Goudarzi, K.; Kargar, F.; Ghanavati, M.; Zamani, M. The effects of green tea extract supplementation on body composition, obesity-related hormones and oxidative stress markers: A grade-assessed systematic review and dose-response meta-analysis of randomised controlled trials. Br. J. Nutr. 2024, 131, 1125–1157. [Google Scholar] [CrossRef]
- Hursel, R.; Viechtbauer, W.; Westerterp-Plantenga, M.S. The effects of green tea on weight loss and weight maintenance: A meta-analysis. Int. J. Obes. 2009, 33, 956–961. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Luo, M.; Wei, S. The Bioprotective Effects of Polyphenols on Metabolic Syndrome against Oxidative Stress: Evidences and Perspectives. Oxidative Med. Cell. Longev. 2019, 2019, 6713194. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef]
- Rasaei, N.; Asbaghi, O.; Samadi, M.; Setayesh, L.; Bagheri, R.; Gholami, F.; Soveid, N.; Casazza, K.; Wong, A.; Suzuki, K.; et al. Effect of Green Tea Supplementation on Antioxidant Status in Adults: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Antioxidants 2021, 10, 1731. [Google Scholar] [CrossRef]
- Roychoudhury, S.; Agarwal, A.; Virk, G.; Cho, C.-L. Potential role of green tea catechins in the management of oxidative stress-associated infertility. Reprod. Biomed. Online 2017, 34, 487–498. [Google Scholar] [CrossRef]
- Nilsson, M.I.; May, L.; Roik, L.J.; Fuda, M.R.; Luo, A.; Hettinga, B.P.; Bujak, A.L.; Tarnopolsky, M.A. A Multi-Ingredient Supplement Protects against Obesity and Infertility in Western Diet-Fed Mice. Nutrients 2023, 15, 611. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Townsend, L.K.; DesOrmeaux, G.J.; Frangos, S.M.; Batchuluun, B.; Dumont, L.; Kuhre, R.E.; Ahmadi, E.; Hu, S.; Rebalka, I.A.; et al. GDF15 promotes weight loss by enhancing energy expenditure in muscle. Nature 2023, 619, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Lexell, J.; Taylor, C.C.; Sjöström, M. What is the cause of the ageing atrophy?: Total number, size and proportion of different fiber types studied in whole vastus lateralis muscle from 15- to 83-year-old men. J. Neurol. Sci. 1988, 84, 275–294. [Google Scholar] [CrossRef] [PubMed]
- Brzezinski, A.; Saada, A.; Miller, H.; Brzezinski-Sinai, N.A.; Ben-Meir, A. Is the aging human ovary still ticking? Expression of clock-genes in luteinized granulosa cells of young and older women. J. Ovarian Res. 2018, 11, 95. [Google Scholar] [CrossRef]
- Palmer, A.K.; Jensen, M.D. Metabolic changes in aging humans: Current evidence and therapeutic strategies. J. Clin. Investig. 2022, 132, e158451. [Google Scholar] [CrossRef]
- Spitler, K.M.; Davies, B.S.J. Aging and plasma triglyceride metabolism. J. Lipid Res. 2020, 61, 1161–1167. [Google Scholar] [CrossRef]
- Bell, K.E.; Snijders, T.; Zulyniak, M.; Kumbhare, D.; Parise, G.; Chabowski, A.; Phillips, S.M. A whey protein-based multi-ingredient nutritional supplement stimulates gains in lean body mass and strength in healthy older men: A randomized controlled trial. PLoS ONE 2017, 12, e0181387. [Google Scholar] [CrossRef]
- Nilsson, M.I.; Mikhail, A.; Lan, L.; Di Carlo, A.; Hamilton, B.; Barnard, K.; Hettinga, B.P.; Hatcher, E.; Tarnopolsky, M.G.; Nederveen, J.P.; et al. A Five-Ingredient Nutritional Supplement and Home-Based Resistance Exercise Improve Lean Mass and Strength in Free-Living Elderly. Nutrients 2020, 12, 2391. [Google Scholar] [CrossRef]
- Melov, S.; Tarnopolsky, M.A.; Beckman, K.; Felkey, K.; Hubbard, A. Resistance exercise reverses aging in human skeletal muscle. PLoS ONE 2007, 2, e465. [Google Scholar] [CrossRef]
- Nederveen, J.P.; Manta, K.; Bujak, A.L.; Simone, A.C.; Fuda, M.R.; Nilsson, M.I.; Hettinga, B.P.; Hughes, M.C.; Perry, C.G.R.; Tarnopolsky, M.A. A Novel Multi-Ingredient Supplement Activates a Browning Program in White Adipose Tissue and Mitigates Weight Gain in High-Fat Diet-Fed Mice. Nutrients 2021, 13, 3726. [Google Scholar] [CrossRef]
- Platko, K.; Lebeau, P.F.; Nederveen, J.P.; Byun, J.H.; MacDonald, M.E.; Bourgeois, J.M.; Tarnopolsky, M.A.; Austin, R.C. A Metabolic Enhancer Protects against Diet-Induced Obesity and Liver Steatosis and Corrects a Pro-Atherogenic Serum Profile in Mice. Nutrients 2023, 15, 2410. [Google Scholar] [CrossRef]
- Xhuti, D.; Rebalka, I.A.; Minhas, M.; May, L.; Murphy, K.; Nederveen, J.P.; Tarnopolsky, M.A. The Acute Effect of Multi-Ingredient Antioxidant Supplementation following Ionizing Radiation. Nutrients 2023, 15, 207. [Google Scholar] [CrossRef]
- Tarnopolsky, M.A.; Mahoney, D.J.; Vajsar, J.; Rodriguez, C.; Doherty, T.J.; Roy, B.D.; Biggar, D. Creatine monohydrate enhances strength and body composition in Duchenne muscular dystrophy. Neurology 2004, 62, 1771–1777. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, M.C.; MacDonald, J.R.; Mahoney, D.J.; Parise, G.; Beal, M.F.; Tarnopolsky, M.A. Beneficial effects of creatine, CoQ10, and lipoic acid in mitochondrial disorders. Muscle Nerve 2007, 35, 235–242. [Google Scholar] [CrossRef]
- Tarnopolsky, M.A. The mitochondrial cocktail: Rationale for combined nutraceutical therapy in mitochondrial cytopathies. Adv. Drug Deliv. Rev. 2008, 60, 1561–1567. [Google Scholar] [CrossRef] [PubMed]
- Zong, Y.; Li, H.; Liao, P.; Chen, L.; Pan, Y.; Zheng, Y.; Zhang, C.; Liu, D.; Zheng, M.; Gao, J. Mitochondrial dysfunction: Mechanisms and advances in therapy. Signal Transduct. Target. Ther. 2024, 9, 124. [Google Scholar] [CrossRef]
- Centonze, M.; Aloisio Caruso, E.; De Nunzio, V.; Cofano, M.; Saponara, I.; Pinto, G.; Notarnicola, M. The Antiaging Potential of Dietary Plant-Based Polyphenols: A Review on Their Role in Cellular Senescence Modulation. Nutrients 2025, 17, 1716. [Google Scholar] [CrossRef]
- Davinelli, S.; Medoro, A.; Hu, F.B.; Scapagnini, G. Dietary polyphenols as geroprotective compounds: From Blue Zones to hallmarks of ageing. Ageing Res. Rev. 2025, 108, 102733. [Google Scholar] [CrossRef] [PubMed]
- Pazoki-Toroudi, H.; Amani, H.; Ajami, M.; Nabavi, S.F.; Braidy, N.; Kasi, P.D.; Nabavi, S.M. Targeting mTOR signaling by polyphenols: A new therapeutic target for ageing. Ageing Res. Rev. 2016, 31, 55–66. [Google Scholar] [CrossRef]
- Xu, W.; Luo, Y.; Yin, J.; Huang, M.; Luo, F. Targeting AMPK signaling by polyphenols: A novel strategy for tackling aging. Food Funct. 2023, 14, 56–73. [Google Scholar] [CrossRef]
- Stohs, S.J.; Badmaev, V. A Review of Natural Stimulant and Non-stimulant Thermogenic Agents. Phytother. Res. PTR 2016, 30, 732–740. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Risi, R.; Masi, D.; Caputi, A.; Balena, A.; Rossini, G.; Tuccinardi, D.; Mariani, S.; Basciani, S.; Manfrini, S.; et al. Current Evidence to Propose Different Food Supplements for Weight Loss: A Comprehensive Review. Nutrients 2020, 12, 2873. [Google Scholar] [CrossRef] [PubMed]
- De Guingand, D.L.; Ellery, S.J.; Davies-Tuck, M.L.; Dickinson, H. Creatine and pregnancy outcomes, a prospective cohort study in low-risk pregnant women: Study protocol. BMJ Open 2019, 9, e026756. [Google Scholar] [CrossRef]
- Cieri-Hutcherson, N.E.; Jaenecke, A.; Bahia, A.; Lucas, D.; Oluloro, A.; Stimmel, L.; Hutcherson, T.C. Systematic Review of l-Arginine for the Treatment of Hypoactive Sexual Desire Disorder and Related Conditions in Women. Pharmacy 2021, 9, 71. [Google Scholar] [CrossRef] [PubMed]
- Goto, E. Effects of prenatal oral l-arginine on birth outcomes: A meta-analysis. Sci. Rep. 2021, 11, 22748. [Google Scholar] [CrossRef]
- Menichini, D.; Feliciello, L.; Neri, I.; Facchinetti, F. L-Arginine supplementation in pregnancy: A systematic review of maternal and fetal outcomes. J. Matern. Neonatal Med. 2023, 36, 2217465. [Google Scholar] [CrossRef]
- Astrup, A.; Bügel, S. Overfed but undernourished: Recognizing nutritional inadequacies/deficiencies in patients with overweight or obesity. Int. J. Obes. 2019, 43, 219–232. [Google Scholar] [CrossRef]
- Dutta, S.; Sengupta, P. Men and mice: Relating their ages. Life Sci. 2016, 152, 244–248. [Google Scholar] [CrossRef] [PubMed]

| Age | n-Size | Acclimation Diet | Intervention Diet | ||
|---|---|---|---|---|---|
| Start (Months) | End (Months) | ||||
| Y-CON | 6.2 | 9.2 | 15 | LabDiet 5K52 (JAX) | 8604 (Inotiv) |
| Y-FE | 6.2 | 9.2 | 15 | LabDiet 5K52 (JAX) | TD.230256 (Inotiv) |
| O-CON | 11.2 | 14.2 | 16 | LabDiet 5K52 (JAX) | 8604 (Inotiv) |
| O-FE | 11.2 | 14.2 | 16 | LabDiet 5K52 (JAX) | TD.230256 (Inotiv) |
| Control (8604) | Fertility Enhancer (TD.230256) | Manufacturer (Purity) | |
|---|---|---|---|
| Macronutrients and energy | |||
| kcal/g | 3.00 | 3.00 | Inotiv |
| Carbohydrate (%kcal) | 54.0 | 49.4 | Inotiv |
| Protein (%kcal) | 32.0 | 30.2 | Inotiv |
| Fat (%kcal) | 14.0 | 20.5 | Inotiv |
| Mitochondrial antioxidants | |||
| CoQ10 (ubiquinone; g/kg) | - | 2.50 | MTC (98–101%) |
| Vitamin E (α-tocopherol; IU/kg) | 120 | 1155 | Inotiv |
| α-Lipoic acid (g/kg) | - | 1.00 | Alzchem (98%) |
| Anti-inflammatory agents | |||
| n-3 oil (g/kg) | - | 28.0 | NutraSea (EPA and DHA) |
| Metabolic activators | |||
| Green coffee extract (g/kg) | - | 2.50 | AFS (CGA 45%, caffeine 40%) |
| Green tea extract (g/kg) | - | 0.75 | ENI (EGCG 50%, caffeine < 3%) |
| Forskolin extract (g/kg) | - | 0.13 | Sabinsa (40%) |
| Beet root extract (g/kg) | - | 10.0 | Enovate (100%) |
| Vitamins and minerals | |||
| Iron/ferric citrate (mg/kg) | 300 | 700 | Inotiv |
| Folic acid (mg/kg) | 3.00 | 8.00 | Inotiv |
| Zinc (mg/kg) | 80.0 | 80.0 | Inotiv |
| Selenium (mg/kg) | 0.34 | 0.34 | Inotiv |
| Copper (mg/kg) | 25.0 | 25.0 | Inotiv |
| Vitamin B12 (mg/kg) | 0.05 | 0.05 | Inotiv |
| Vitamin D3 (IU/g) | 2.40 | 2.40 | Inotiv |
| Energy carriers | |||
| Creatine monohydrate (g/kg) | - | 30.0 | Alzchem (99–100%) |
| Amino acids | |||
| L-arginine (g/kg) | 15.0 | 20.0 | Inotiv |
| Food and Energy Intakes | ||
|---|---|---|
| g/d | kcal/d | |
| Y-CON | 3.85 ± 0.07 a | 11.53 ± 0.22 a |
| Y-FE | 4.17 ± 0.15 b | 12.52 ± 0.45 b |
| O-CON | 3.77 ± 0.06 a | 11.31 ± 0.17 a |
| O-FE | 4.26 ± 0.11 b | 12.78 ± 0.32 b |
| Main Effects | 2 × 2 ANOVA | 2 × 2 ANOVA |
| Age (A) | 0.96398 | 0.95148 |
| Diet (D) | 0.00002 | 0.00001 |
| Interactions | ||
| A*D | 0.36969 | 0.36378 |
| 5-Day Mating Period | 10-Day Mating Period | Cumulative Results | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Plugs | Litters | Reproductive Success (%) | Plugs | Litters | Reproductive Success (%) | Plugs | Litters | Reproductive Success (%) | |
| Y-CON | 3.0 ab | 2.0 ab | 66.7 | 6.0 | 2.0 | 33.3 | 9.0 | 4.0 ab | 44.4 |
| Y-FE | 6.0 ab | 5.0 a | 83.3 | 3.0 | 3.0 | 100.0 | 9.0 | 8.0 a | 88.9 |
| O-CON | 2.0 a | 0.0 b | 0.0 | 5.0 | 0.0 | 0.0 | 7.0 | 0.0 b | 0.0 |
| O-FE | 8.0 b | 0.0 b | 0.0 | 3.0 | 2.0 | 66.7 | 11.0 | 2.0 b | 18.2 |
| Main Effects | 2 × 2 ANOVA | 2 × 2 ANOVA | 2 × 2 ANOVA | ||||||
| Age (A) | 0.91335 | 0.00292 | NA | 0.93797 | 0.28014 | NA | 0.82145 | 0.00571 | NA |
| Diet (D) | 0.01475 | 0.18828 | NA | 0.14439 | 0.22359 | NA | 0.45292 | 0.10121 | NA |
| Interactions | |||||||||
| A*D | 0.44739 | 0.18828 | NA | 0.81547 | 0.64873 | NA | 0.452921 | 0.54925 | NA |
| Mitochondrial | Antioxidant | Oxidative Damage | Inflammation | ||||
|---|---|---|---|---|---|---|---|
| Complex II | Complex III | SOD2 | 4HNE | pNFkb (Ser536) | tNFkb | p/tNFkb | |
| Y-CON | 0.020 ± 0.002 a | 0.021 ± 0.002 | 0.028 ± 0.004 | 0.041 ± 0.003 | 0.025 ± 0.010 | 0.023 ± 0.008 | 1.351 ± 0.650 |
| Y-FE | 0.026 ± 0.002 a | 0.026 ± 0.002 | 0.035 ± 0.004 | 0.035 ± 0.004 | 0.022 ± 0.007 | 0.30 ± 0.008 | 0.859 ± 0.367 |
| O-CON | 0.021 ± 0.002 a | 0.021 ± 0.002 | 0.026 ± 0.004 | 0.046 ± 0.003 | 0.025 ± 0.011 | 0.025 ± 0.008 | 1.133 ± 0.566 |
| O-FE | 0.024 ± 0.002 a | 0.023 ± 0.001 | 0.031 ± 0.004 | 0.043 ± 0.004 | 0.021 ± 0.014 | 0.025 ± 0.008 | 1.055 ± 0.818 |
| Main Effects | 2 × 2 ANOVA | 2 × 2 ANOVA | 2 × 2 ANOVA | 2 × 2 ANOVA | 2 × 2 ANOVA | 2 × 2 ANOVA | 2 × 2 ANOVA |
| Age (A) | 0.94530 | 0.43540 | 0.42328 | 0.07822 | 0.84741 | 0.78773 | 0.73208 |
| Diet (D) | 0.03715 | 0.05595 | 0.11030 | 0.20410 | 0.21210 | 0.22683 | 0.06406 |
| Interactions | |||||||
| A*D | 0.49087 | 0.36999 | 0.90435 | 0.80935 | 0.59630 | 0.37531 | 0.79310 |
| Fertility Predictors | Linear Regression Results | ||
|---|---|---|---|
| Anthropometrics | |||
| Bodyweight | r = −0.170 | r2 = 0.029 | p = 0.270 |
| Ex vivo body composition | |||
| Muscle (quad) | r = 0.2414 | r2 = 0.058 | p = 0.119 |
| Fat (gWAT) | r = −0.3390 | r2 = 0.115 | p = 0.026 |
| Muscle/fat ratio | r = 0.3702 | r2 = 0.137 | p = 0.015 |
| Ovarian aging markers | |||
| Complex II | r = 0.1830 | r2 = 0.033 | p = 0.235 |
| Complex III | r = 0.2816 | r2 = 0.079 | p = 0.064 |
| SOD2 | r = 0.4866 | r2 = 0.237 | p = 0.001 |
| 4HNE | r = −0.3013 | r2 = 0.091 | p = 0.047 |
| p/tNFkb | r = −0.0917 | r2 = 0.008 | p = 0.554 |
| Ovarian quality index | r = 0.3296 | r2 = 0.109 | p = 0.029 |
| Best multiple regression model | R = 0.637 | R2 = 0.406 | p = 0.00013 |
| Muscle/fat ratio | p = 0.00633 | ||
| SOD2 | p = 0.00109 | ||
| 4HNE | p = 0.11814 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiarot, A.; Minhas, M.; de Maat, N.M.; Doan, J.; Nilsson, M.I.; Hettinga, B.P.; Faghih, M.; Neal, M.S.; Nederveen, J.P.; Tarnopolsky, M.A. A Multi-Ingredient Supplement Improves Body Re-Composition, Ovarian Aging Markers, and Reproductive Success in Young and Middle-Aged Female Mice. Biomolecules 2025, 15, 1258. https://doi.org/10.3390/biom15091258
Chiarot A, Minhas M, de Maat NM, Doan J, Nilsson MI, Hettinga BP, Faghih M, Neal MS, Nederveen JP, Tarnopolsky MA. A Multi-Ingredient Supplement Improves Body Re-Composition, Ovarian Aging Markers, and Reproductive Success in Young and Middle-Aged Female Mice. Biomolecules. 2025; 15(9):1258. https://doi.org/10.3390/biom15091258
Chicago/Turabian StyleChiarot, Alessandra, Mahek Minhas, Nicoletta M. de Maat, Jenny Doan, Mats I. Nilsson, Bart P. Hettinga, Mehrnoosh Faghih, Michael S. Neal, Joshua P. Nederveen, and Mark A. Tarnopolsky. 2025. "A Multi-Ingredient Supplement Improves Body Re-Composition, Ovarian Aging Markers, and Reproductive Success in Young and Middle-Aged Female Mice" Biomolecules 15, no. 9: 1258. https://doi.org/10.3390/biom15091258
APA StyleChiarot, A., Minhas, M., de Maat, N. M., Doan, J., Nilsson, M. I., Hettinga, B. P., Faghih, M., Neal, M. S., Nederveen, J. P., & Tarnopolsky, M. A. (2025). A Multi-Ingredient Supplement Improves Body Re-Composition, Ovarian Aging Markers, and Reproductive Success in Young and Middle-Aged Female Mice. Biomolecules, 15(9), 1258. https://doi.org/10.3390/biom15091258








