In Vitro and In Vivo Comparisons of Activated Charcoal and Biochar as Dietary Treatments for Controlling Boar Taint
Abstract
1. Introduction
2. Materials and Methods
2.1. In Vitro Binding Studies
2.1.1. Preparation of Adsorbent and Adsorbate Solutions
2.1.2. Adsorption Assays
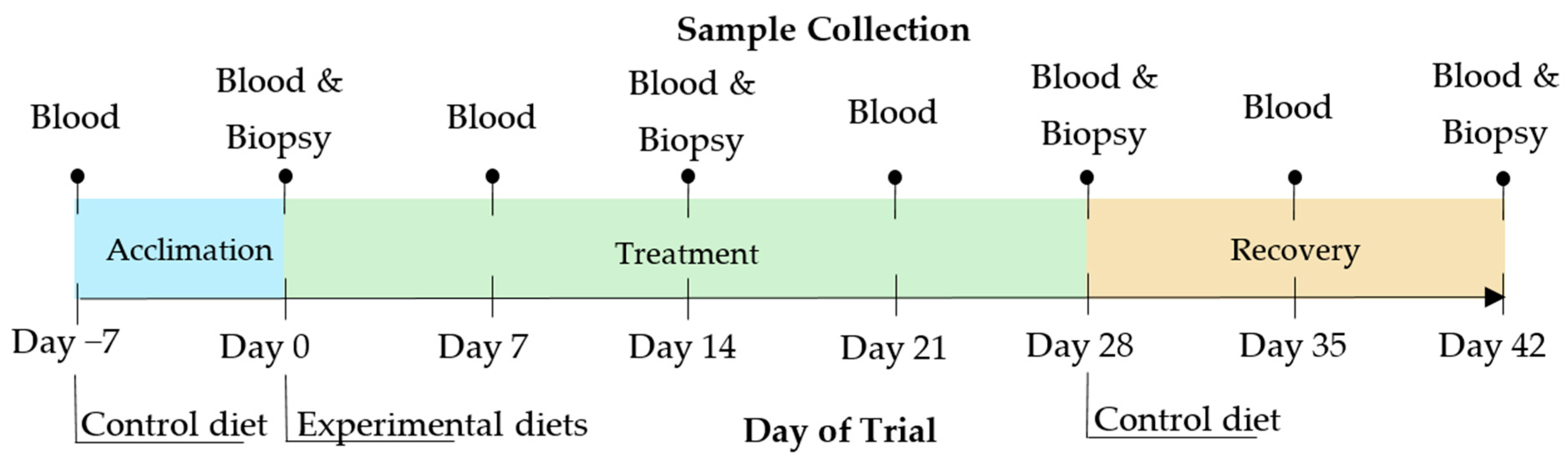
2.2. In Vivo Feeding Trial
2.2.1. Animals and Experimental Design
2.2.2. Diet Compositions
2.2.3. Immunoassays and Skatole Analysis
2.2.4. Growth and Performance Parameters
2.3. Statistical Analysis
3. Results
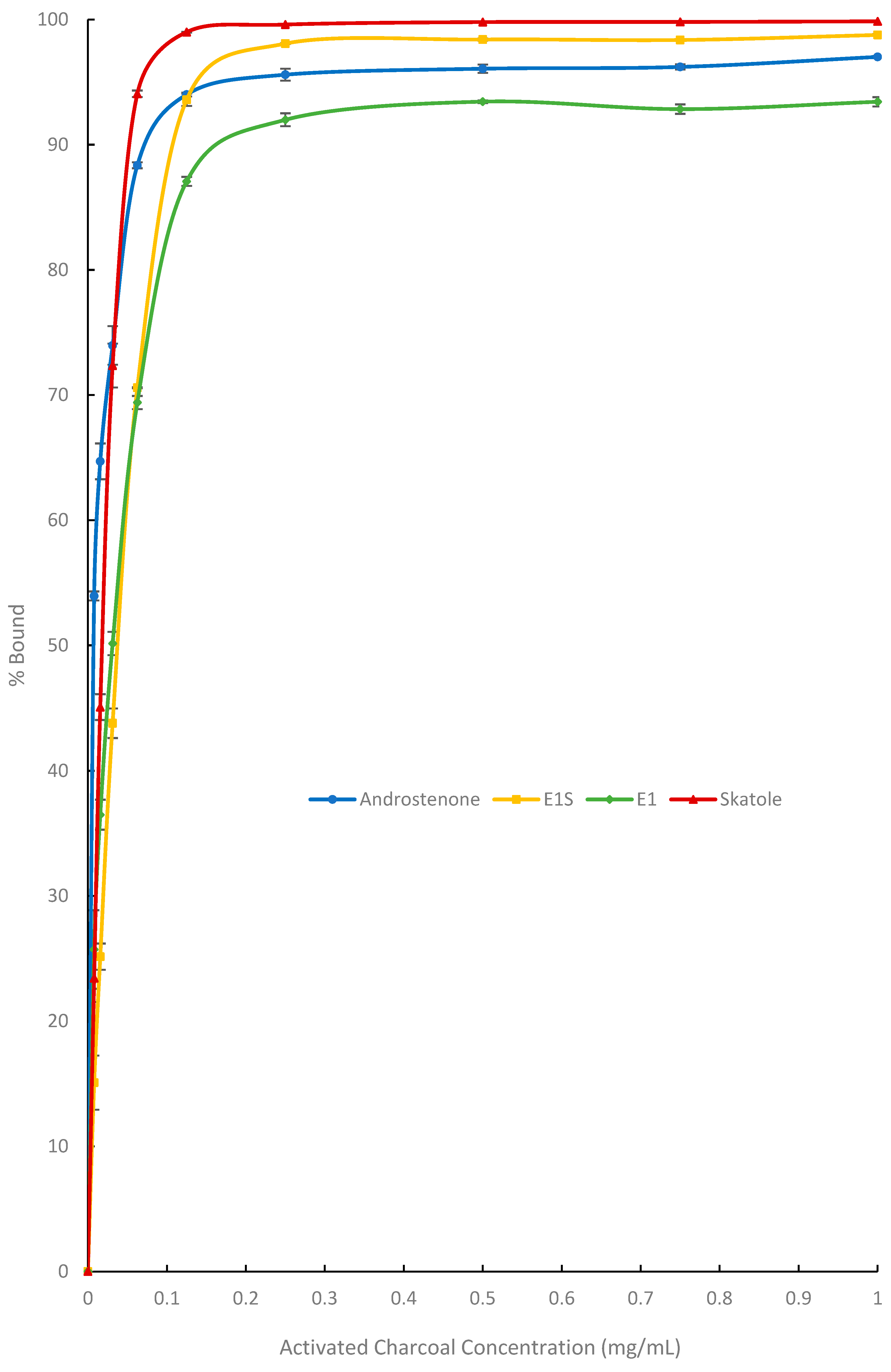
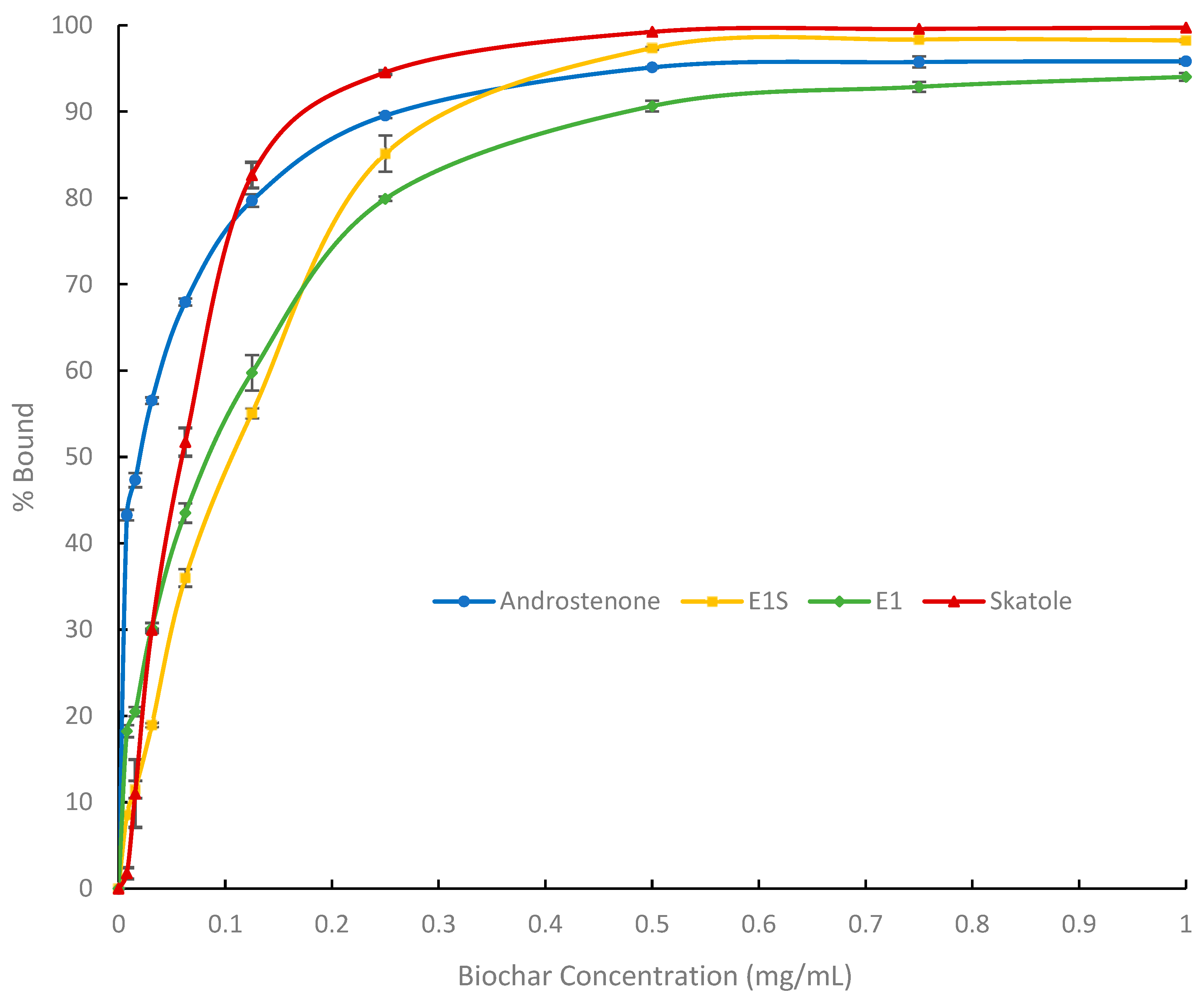
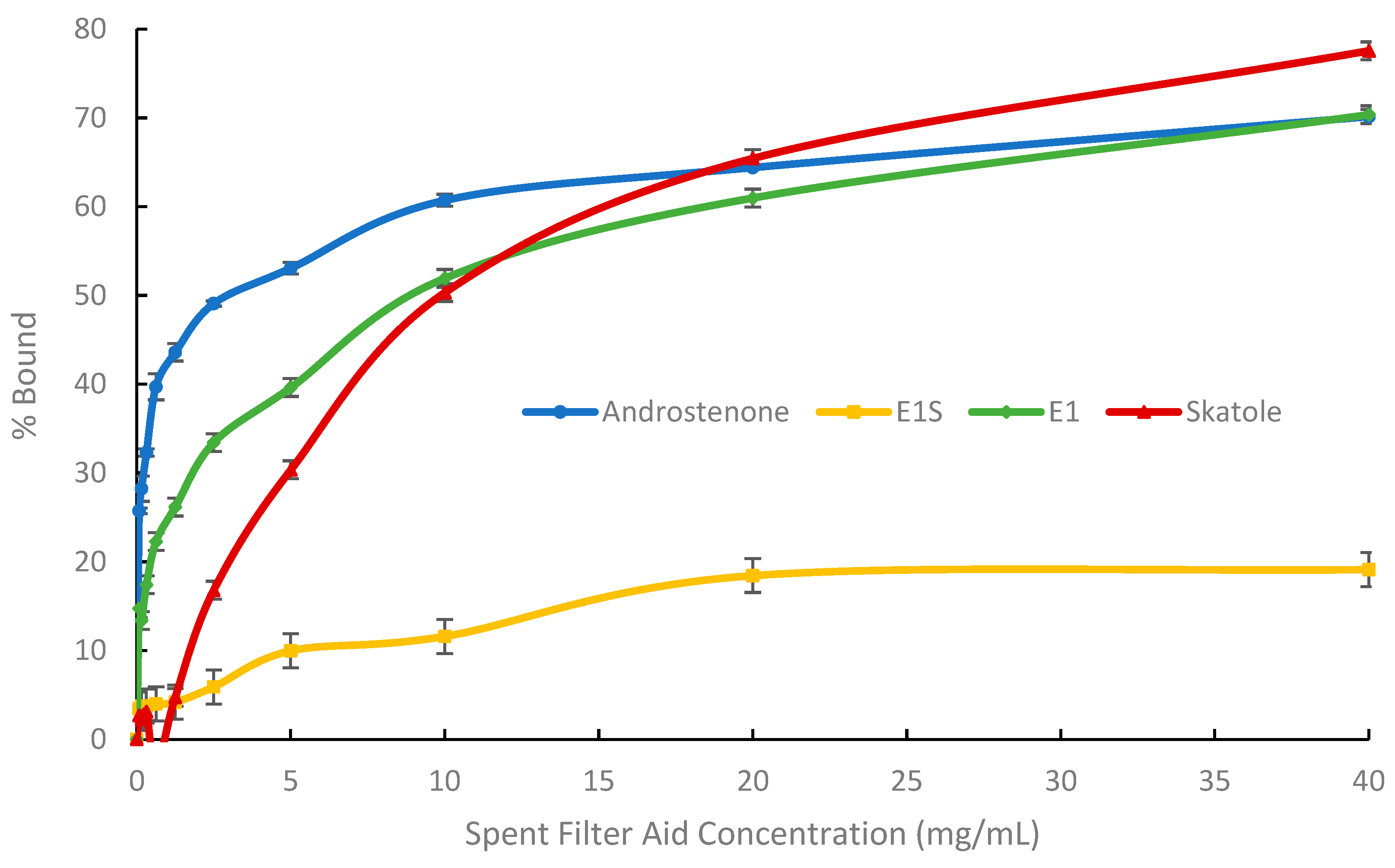
3.1. In Vitro Adsorption Assays and Michaelis-Menten Binding Kinetics
3.2. In Vivo Animal Feeding Trial
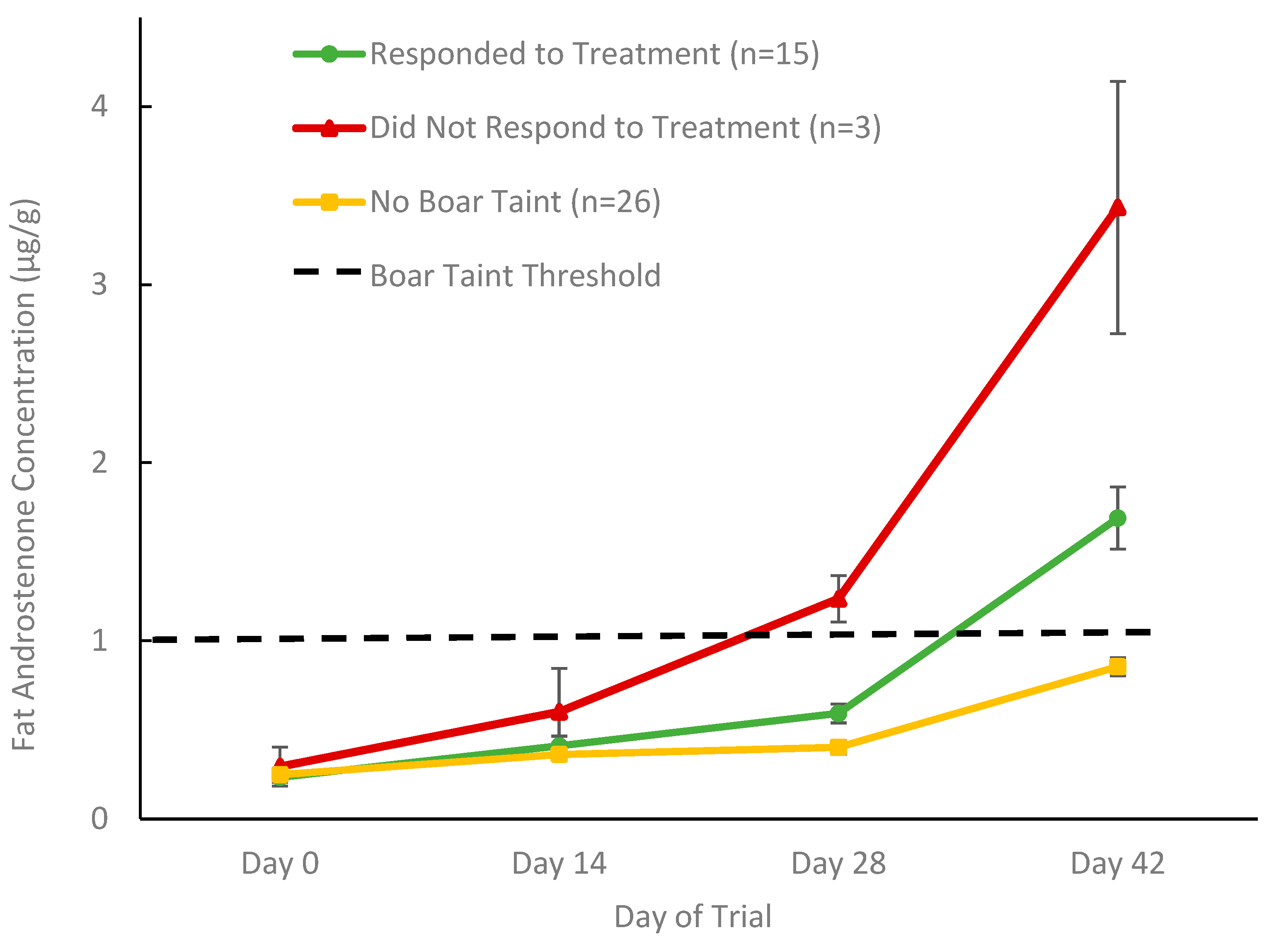
3.2.1. Classification of Boar Taint Status and Treatment Response
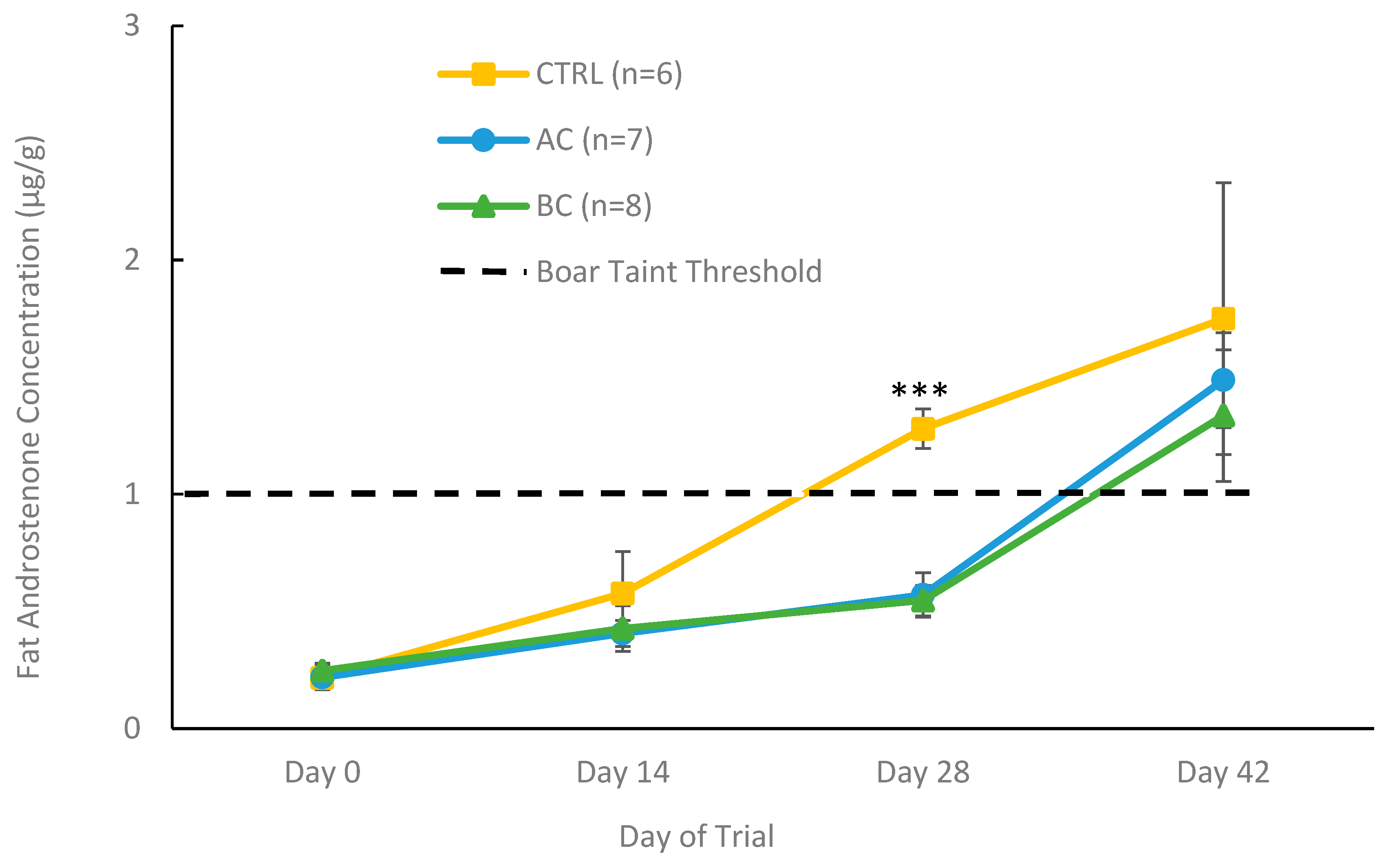
3.2.2. Fat Androstenone Concentrations in Treatment and Control Boars with Boar Taint
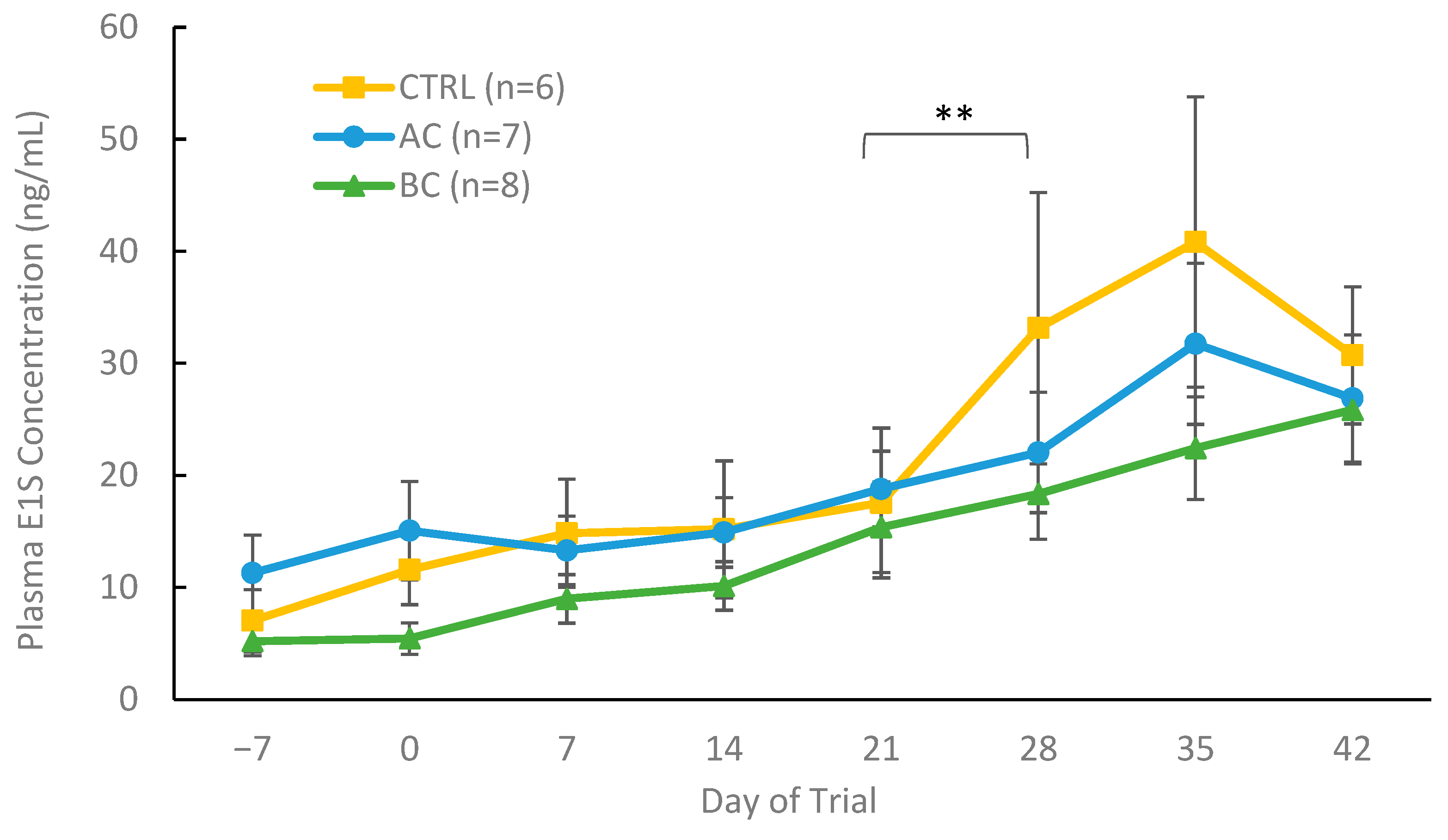
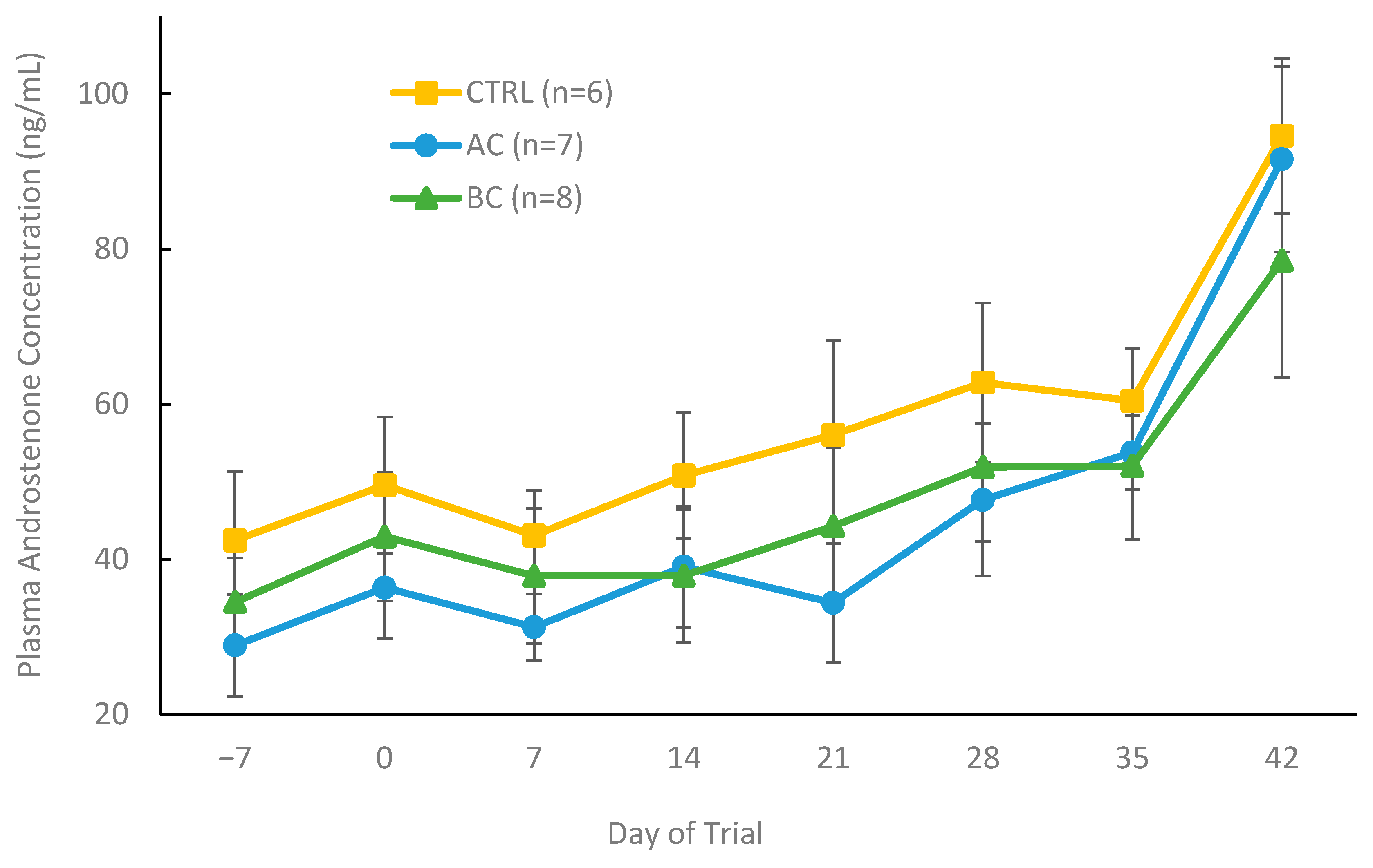
3.2.3. Plasma Hormone Concentrations in Treatment and Control Boars with Boar Taint
3.2.4. Animal Performance Parameters
4. Discussion
4.1. In Vitro Binding Affinity of Binding Agents for Estrogens and Boar Taint-Causing Compounds
4.2. Using AC and BC In Vivo as a Dietary Control for Boar Taint
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AC | Activated charcoal |
| ADG | Average daily gain |
| AND | Androstenone |
| BC | Biochar |
| CTRL | Control |
| EHC | Enterohepatic circulation |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| E1 | Estrone |
| E1S | Estrone-1-sulfate |
| FCR | Feed conversion ratio |
| PBS | Phosphate-buffered saline |
| RIA | Radioimmunoassay |
| SFA | Spent filter aid |
References
- Li, X.; Jensen, B.B.; Canibe, N. The Mode of Action of Chicory Roots on Skatole Production in Entire Male Pigs Is Neither via Reducing the Population of Skatole-Producing Bacteria nor via Increased Butyrate Production in the Hindgut. Appl. Environ. Microbiol. 2019, 85, e02327-18. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, P.A.; Squires, E.J.; Raeside, J.I.; Renaud, R. Synthesis of Free and Sulphoconjugated 16-Androstene Steroids by the Leydig Cells of the Mature Domestic Boar. J. Steroid Biochem. Mol. Biol. 2005, 96, 217–228. [Google Scholar] [CrossRef]
- Hofmann, A.F. The Enterohepatic Circulation of Bile Acids in Mammals: Form and Functions. Front. Biosci. 2009, 14, 2584. [Google Scholar] [CrossRef]
- Okour, M.; Brundage, R.C. Modeling Enterohepatic Circulation. Curr. Pharmacol. Rep. 2017, 3, 301–313. [Google Scholar] [CrossRef]
- Roberts, M.S.; Magnusson, B.M.; Burczynski, F.J.; Weiss, M. Enterohepatic Circulation: Physiological, Pharmacokinetic and Clinical Implications. Clin. Pharmacokinet. 2002, 41, 751–790. [Google Scholar] [CrossRef]
- Flores, R.; Shi, J.; Fuhrman, B.; Xu, X.; Veenstra, T.D.; Gail, M.H.; Gajer, P.; Ravel, J.; Goedert, J.J. Fecal Microbial Determinants of Fecal and Systemic Estrogens and Estrogen Metabolites: A Cross-Sectional Study. J. Transl. Med. 2012, 10, 253. [Google Scholar] [CrossRef]
- Gorbach, S.L.; Goldin, B.R. Diet and the Excretion and Enterohepatic Cycling of Estrogens. Prev. Med. 1987, 16, 525–531. [Google Scholar] [CrossRef]
- Ruoff, W.L.; Dziuk, P.J. Absorption and Metabolism of Estrogens from the Stomach and Duodenum of Pigs. Domest. Anim. Endocrinol. 1994, 11, 197–208. [Google Scholar] [CrossRef]
- Schwarzenberger, F.; Toole, G.S.; Christie, H.L.; Raeside, J.I. Plasma Levels of Several Androgens and Estrogens from Birth to Puberty in Male Domestic Pigs. Acta Endocrinol. 1993, 128, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Ren, L.; Yang, S.M.; Qiu, J.; Kamal-Eldin, A.; Lundström, K. Determination of Androstenone Levels in Porcine Plasma by LC-MS/MS. Food Chem. 2010, 122, 1278–1282. [Google Scholar] [CrossRef]
- Jen, K.; Squires, E.J. Efficacy of Non-Nutritive Sorbent Materials as Intestinal-Binding Agents for the Control of Boar Taint. Animal 2011, 5, 1814–1820. [Google Scholar] [CrossRef]
- Wakabayashi, Y.; Maruyama, S.; Hachimura, K.; Ohwada, T. Activated Charcoal Interrupts Enteroenteric Circulation of Phenobarbital. J. Toxicol. Clin. Toxicol. 1994, 32, 419–424. [Google Scholar] [CrossRef]
- Chin, L.; Picchioni, A.L.; Duplisse, B.R. The Action of Activated Charcoal on Poisons in the Digestive Tract. Toxicol. Appl. Pharmacol. 1970, 16, 786–799. [Google Scholar] [CrossRef]
- Gęca, M.; Wiśniewska, M.; Nowicki, P. Biochars and Activated Carbons as Adsorbents of Inorganic and Organic Compounds from Multicomponent Systems—A Review. Adv. Colloid Interface Sci. 2022, 305, 102687. [Google Scholar] [CrossRef] [PubMed]
- Ayankoso, M.T.; Oluwagbamila, D.M.; Abe, O.S. Effects of Activated Charcoal on Livestock Production: A Review. Slovak J. Anim. Sci. 2023, 56, 46–60. [Google Scholar] [CrossRef]
- Park, S.; Squires, E.J. In Vitro Assessment of the Effectiveness of Mineral Adsorbents in Sequestering Boar Taint Compounds. Animals 2025, 15, 765. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, H.-P.; Hagemann, N.; Draper, K.; Kammann, C. The Use of Biochar in Animal Feeding. PeerJ 2019, 7, e7373. [Google Scholar] [CrossRef]
- Weber, K.; Quicker, P. Properties of Biochar. Fuel 2018, 217, 240–261. [Google Scholar] [CrossRef]
- Lanthier, F.; Lou, Y.; Squires, E.J. Skatole Metabolism in the Intact Pre-Pubescent Male Pig: The Relationship between Hepatic Enzyme Activity and Skatole Concentrations in Plasma and Fat. Livest. Sci. 2007, 106, 145–153. [Google Scholar] [CrossRef]
- Jen, K.; Squires, E.J. In Vitro Assessment of the Effectiveness of Non-Nutritive Sorbent Materials as Binding Agents for Boar Taint Compounds. Animal 2011, 5, 1821–1828. [Google Scholar] [CrossRef]
- Raeside, J.I.; Renaud, R.L.; Friendship, R.M. Differences in Aromatase Activity between Leydig Cells from the Scrotal and Abdominal Testis in the Naturally Unilateral-Cryptorchid Boar1. Biol. Reprod. 1988, 39, 566–572. [Google Scholar] [CrossRef]
- Claus, R.; Mahler, G.; Munster, E. Determination of boar taint steroid 5a-androst-16-en-3-one in adipose tissue of pigs with a rapid microtiter plate enzyme-immunoassay (MTE). Arch. Fuer Leb. 1988, 39, 87–90. [Google Scholar]
- Squires, E.J.; Lundström, K. Relationship between Cytochrome P450IIE1 in Liver and Levels of Skatole and Its Metabolites in Intact Male Pigs. J. Anim. Sci. 1997, 75, 2506. [Google Scholar] [CrossRef]
- Dehnhard, M.; Bernal-Barragan, H.; Claus, R. Rapid and Accurate High-Performance Liquid Chromatographic Method for the Determination of 3-Methylindole (Skatole) in Faeces of Various Species. J. Chromatogr. B Biomed. Sci. Appl. 1991, 566, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Prusa, K.; Nederveld, H.; Runnels, P.L.; Li, R.; King, V.L.; Crane, J.P. Prevalence and Relationships of Sensory Taint, 5α-Androstenone and Skatole in Fat and Lean Tissue from the Loin (Longissimus Dorsi) of Barrows, Gilts, Sows, and Boars from Selected Abattoirs in the United States. Meat Sci. 2011, 88, 96–101. [Google Scholar] [CrossRef]
- Zamaratskaia, G.; Rydhmer, L.; Chen, G.; Madej, A.; Andersson, H.; Lundström, K. Boar Taint Is Related to Endocrine and Anatomical Changes at Puberty but Not to Aggressive Behaviour in Entire Male Pigs. Reprod. Domest. Anim. 2005, 40, 500–506. [Google Scholar] [CrossRef]
- Kojima, M. Sex and Breed Differences in the Constitutive Gene Expression of Hepatic Drug Metabolizing Enzymes in Meishan and Landrace Pigs: Testosterone-Mediated Differences. Jpn. Agric. Res. Q. 2020, 54, 7–12. [Google Scholar] [CrossRef]
- Appell, M.; Wegener, E.C.; Sharma, B.K.; Eller, F.J.; Evans, K.O.; Compton, D.L. In Vitro Evaluation of the Adsorption Efficacy of Biochar Materials on Aflatoxin B1, Ochratoxin A and Zearalenone. Animals 2023, 13, 3311. [Google Scholar] [CrossRef]
- Zamaratskaia, G.; Squires, E.J. Biochemical, Nutritional and Genetic Effects on Boar Taint in Entire Male Pigs. Animal 2009, 3, 1508–1521. [Google Scholar] [CrossRef] [PubMed]
- Hansen, L.L.; Mejer, H.; Thamsborg, S.M.; Byrne, D.V.; Roepstorff, A.; Karlsson, A.H.; Hansen-Møller, J.; Jensen, M.T.; Tuomola, M. Influence of Chicory Roots (Cichorium intybus L.) on Boar Taint in Entire Male and Female Pigs. Anim. Sci. 2006, 82, 359–368. [Google Scholar] [CrossRef]
- Zammerini, D.; Wood, J.D.; Whittington, F.M.; Nute, G.R.; Hughes, S.I.; Hazzledine, M.; Matthews, K. Effect of Dietary Chicory on Boar Taint. Meat Sci. 2012, 91, 396–401. [Google Scholar] [CrossRef]
- Pauly, C.; Spring, P.; O’Doherty, J.V.; Kragten, S.A.; Bee, G. Performances, Meat Quality and Boar Taint of Castrates and Entire Male Pigs Fed a Standard and a Raw Potato Starch-Enriched Diet. Animal 2008, 2, 1707–1715. [Google Scholar] [CrossRef] [PubMed]
- Pinto, R.P.; Mata, F.; Pires, P.; Barros, M.; Araújo, J.P.; Vaz-Velho, M. The Use of Sugar Beet Pulp in Pig Diet to Control Skatole Analysed by HPLC Quantification Method. Sci. Agric. 2023, 80, e20220093. [Google Scholar] [CrossRef]
- Claus, R.; Weiler, U. Endocrine Regulation of Growth and Metabolism in the Pig: A Review. Livest. Prod. Sci. 1994, 37, 245–260. [Google Scholar] [CrossRef]
- Robic, A.; Faraut, T.; Prunier, A. Pathways and Genes Involved in Steroid Hormone Metabolism in Male Pigs: A Review and Update. J. Steroid Biochem. Mol. Biol. 2014, 140, 44–55. [Google Scholar] [CrossRef]
- Sui, Y.; Wu, J.; Chen, J. The Role of Gut Microbial β-Glucuronidase in Estrogen Reactivation and Breast Cancer. Front. Cell Dev. Biol. 2021, 9, 631552. [Google Scholar] [CrossRef]
- Fuhrman, B.J.; Feigelson, H.S.; Flores, R.; Gail, M.H.; Xu, X.; Ravel, J.; Goedert, J.J. Associations of the Fecal Microbiome With Urinary Estrogens and Estrogen Metabolites in Postmenopausal Women. J. Clin. Endocrinol. Metab. 2014, 99, 4632–4640. [Google Scholar] [CrossRef]
- Schubert, D.C.; Chuppava, B.; Witte, F.; Terjung, N.; Visscher, C. Evaluation of Coated Biochar as an Intestinal Binding Agent for Skatole and Indole in Male Intact Finishing Pigs. Animals 2021, 11, 760. [Google Scholar] [CrossRef]
- Huwig, A.; Freimund, S.; Käppeli, O.; Dutler, H. Mycotoxin Detoxication of Animal Feed by Different Adsorbents. Toxicol. Lett. 2001, 122, 179–188. [Google Scholar] [CrossRef] [PubMed]
| Ingredient Inclusion | Experimental Diets | ||
|---|---|---|---|
| (g/kg) | Control | Activated Charcoal | Biochar |
| Corn | 485 | 485 | 485 |
| Soybean meal 1 | 200 | 200 | 200 |
| Corn distillers dried grains and solubles | 200 | 200 | 200 |
| Corn oil | 37.5 | 37.5 | 37.5 |
| Vitamin/mineral premix 2 | 6 | 6 | 6 |
| Mono-calcium phosphate | 6.5 | 6.5 | 6.5 |
| Limestone | 9 | 9 | 9 |
| Sodium chloride | 3 | 3 | 3 |
| L-Lysine-HCl | 2.7 | 2.7 | 2.7 |
| L-Threonine | 0.3 | 0.3 | 0.3 |
| Cellulose | 50 | - | - |
| Activated charcoal | - | 50 | - |
| Biochar | - | - | 50 |
| Total | 1000 | 1000 | 1000 |
| Parameters | Activated Charcoal | Biochar | Spent Filter Aid | |
|---|---|---|---|---|
| Bmax (%) | 98.3 ± 0.5 a | 99.7 ± 1.2 a | 39.8 ± 1.4 b | |
| E1 | K (µg/mL) | 25.4 ± 1.1 a | 72.0 ± 3.1 a | 423.3 ± 71.1 b |
| Bmax/K (% mL/mg) | 3880.9 ± 163.1 a | 1390.1 ± 52.6 b | 99.6 ± 16.0 c | |
| Bmax (%) | 107.7 ± 0.4 a | 116.9 ± 0.3 b | 7.5 ± 0.8 c | |
| E1S | K (µg/mL) | 38.1 ± 1.1 a | 128.5 ± 3.5 ab | 225.0 ± 58.3 b |
| Bmax/K (% mL/mg) | 2891.8 ± 69.7 a | 910.8 ± 23.6 b | 34.1 ± 23.3 c | |
| Bmax (%) | 97.2 ± 0.4 a | 84.5 ± 0.8 b | 50.5 ± 0.2 c | |
| AND | K (µg/mL) | 7.3 ± 0.2 a | 11.2 ± 0.5 a | 123.4 ± 7.9 b |
| Bmax/K (% mL/mg) | 13,404.8 ± 334.3 a | 7553.1 ± 252.5 b | 412.5 ± 24.9 c | |
| Bmax (%) | 106.1 ± 0.2 a | 113.2 ± 0.7 a | 97.1 ± 5.3 b | |
| Skatole | K (µg/mL) | 16.7 ± 0.9 a | 72.6 ± 4.6 a | 11,320.2 ± 1187.6 b |
| Bmax/K (% mL/mg) | 6387.0 ± 325.2 a | 1383.3 ± 88.5 b | 8.6 ± 0.4 c | |
| Performance Parameter | Control | Activated Charcoal | Biochar | p-Value |
|---|---|---|---|---|
| ADG over trial (kg/day) | 1.37 ± 0.042 | 1.41 ± 0.045 | 1.39 ± 0.035 | 0.8 |
| ADG during treatment (kg/day) | 1.33 ± 0.050 | 1.28 ± 0.054 | 1.31 ± 0.050 | 0.6 |
| FCR over trial | 2.60 ± 0.052 | 2.56 ± 0.056 | 2.65 ± 0.088 | 0.06 |
| FCR during treatment | 2.60 ± 0.062 | 2.62 ± 0.076 | 2.74 ± 0.11 | 0.3 |
| Weight at start of acclimation (kg) | 70.98 ± 1.92 | 71.21 ± 2.59 | 71.71 ± 2.09 | 0.8 |
| Weight at start of treatment (kg) | 80.85 ± 2.57 | 82.60 ± 3.09 | 82.71 ± 3.07 | 0.8 |
| Weight at end of treatment (kg) | 125.06 ± 4.33 | 125.47 ± 4.20 | 125.74 ± 4.14 | 0.9 |
| Weight at end of recovery (kg) | 143.31 ± 4.92 | 145.68 ± 4.88 | 144.58 ± 4.72 | 0.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parent, M.; Bone, C.; Huber, L.-A.; Squires, E.J. In Vitro and In Vivo Comparisons of Activated Charcoal and Biochar as Dietary Treatments for Controlling Boar Taint. Biomolecules 2025, 15, 1257. https://doi.org/10.3390/biom15091257
Parent M, Bone C, Huber L-A, Squires EJ. In Vitro and In Vivo Comparisons of Activated Charcoal and Biochar as Dietary Treatments for Controlling Boar Taint. Biomolecules. 2025; 15(9):1257. https://doi.org/10.3390/biom15091257
Chicago/Turabian StyleParent, Melissa, Christine Bone, Lee-Anne Huber, and E. James Squires. 2025. "In Vitro and In Vivo Comparisons of Activated Charcoal and Biochar as Dietary Treatments for Controlling Boar Taint" Biomolecules 15, no. 9: 1257. https://doi.org/10.3390/biom15091257
APA StyleParent, M., Bone, C., Huber, L.-A., & Squires, E. J. (2025). In Vitro and In Vivo Comparisons of Activated Charcoal and Biochar as Dietary Treatments for Controlling Boar Taint. Biomolecules, 15(9), 1257. https://doi.org/10.3390/biom15091257






