Abstract
In recent decades, many efforts have been made to elucidate the genetic causes of non-syndromic cleft palate (nsCPO), a complex congenital disease caused by the interaction of several genetic and environmental factors. Since genome-wide association studies have evidenced a minor contribution of common polymorphisms in nsCPO inheritance, we used whole exome sequencing data to explore the role of ultra-rare variants in this study. In a cohort of 35 nsCPO cases and 38 controls, we performed a gene set enrichment analysis (GSEA) and a hypergeometric test for assessing significant overlap between genes implicated in nsCPO pathobiology and genes enriched in ultra-rare variants in our cohort. GSEA highlighted an enrichment of ultra-rare variants in genes principally belonging to cytoskeletal protein binding pathway (Probability Density Function corrected p-value = 1.57 × 10−4); protein-containing complex binding pathway (p-value = 1.06 × 10−2); cell adhesion molecule binding pathway (p-value = 1.24 × 10−2); ECM-receptor interaction pathway (p-value = 1.69 × 10−2); and in the Integrin signaling pathway (p-value = 1.28 × 10−2). Two genes implicated in nsCPO pathobiology, namely COL2A1 and GLI3, ranked among the genes (n = 34) with nominal enrichment in the ultra-rare variant collapsing analysis (Fisher’s exact test p-value < 0.05). These genes were also part of an independent list of genes highly relevant to nsCPO biology (n = 25). Significant overlap between the two sets of genes (hypergeometric test p-value = 5.86 × 10−3) indicated that enriched genes are likely to be implicated in physiological palate development and/or the pathological processes of oral clefting. In conclusion, ultra-rare variants collectively impinge on biological pathways crucial to nsCPO pathobiology and point to candidate genes that may contribute to the individual risk of disease. Sequencing can be an effective approach to identify candidate genes and pathways for nsCPO.
1. Introduction
The human secondary palate develops between the fifth and twelfth embryonic weeks in order to separate the nasal and oral cavities. It originates from two distinct and symmetric palatal shelves that initially grow next to the tongue in a vertical position, then rotate to acquire a horizontal position on the top of the tongue, and finally, shelves approach and fuse with each other in an antero-posterior direction [1,2]. The cleft of the secondary palate is a common birth defect that may arise in any one of these developmental steps, possibly caused by failure of growth, elevation, adhesion, or fusion of the palatal shelves.
Secondary cleft palate can occur as an independent defect, a disorder known as non-syndromic cleft palate (nsCPO). However, in about 50% of cases, cleft palate occurs in conjunction with other abnormalities that contribute to recognizable or non-recognizable syndrome. In addition, a cleft palate is often associated with other orofacial clefts, possibly involving the lip, the alveolar ridge, or the primary palate; this is a different condition known as cleft lip, with or without cleft palate, that can be non-syndromic (nsCL/P) or syndromic.
nsCPO, as well as nsCL/P, are considered complex diseases caused by the interaction of several genetic and environmental factors. Indeed, epidemiological data support the multifactorial threshold model of inheritance [3]. The genetic architecture underlying nsCPO is still largely unknown. Several candidate loci have been proposed but have rarely been replicated in subsequent studies, leaving many hypotheses open. Convincing evidence supporting a role in nsCPO etiology was collected for FOXE1, GRHL3, and PAX7 (reviewed by Martinelli et al. [4]).
In recent years, genome-wide association studies (GWAS) have identified numerous single nucleotide polymorphisms and several genes involved in nsCL/P etiology. In contrast, this approach has not been as successful for nsCPO [5,6], although nsCPO heritability suggests a strong contribution of genetic factors [3]. Although several explanations are conceivable, one possible reason could be related to inherent limitations of GWAS; this approach has high efficiency in detecting the role of common variants having moderate effect, but is less effective in the case of rare and ultra-rare variants [7]. Rare variants, which are numerous in every human genome [8] and are known to play a notable role in genetic diseases and in complex disorders [9], might have a greater effect in the etiology of nsCPO than in nsCL/P. Of note, in the GWAS study conducted by Leslie et al., the risk variant rs41268753 detected in the GRHL3 locus was relatively uncommon in the general population (minor allele frequency 3.2%), at a value close to the efficiency limits of GWAS. This evidence supports the idea that low-frequency variants may have relevance in the etiology of nsCPO [6]. In this scenario, sequencing might be a better approach to identify rare causative variants. To this purpose, two different research groups performed whole exome sequencing (WES), focusing on nsCPO familial cases [10,11]. Hoebel and colleagues analyzed WES data from 16 nsCPO first-degree relatives from eight multiplex families. They did not identify any genes with recurrent deleterious mutations; however, they suggested potential candidate genes [10]. Moreover, Liu et al. investigated five affected individuals from a single family and found evidence for a role of ARHGAP29 in the pathogenesis of nsCPO [11].
In this investigation, WES and pathway analysis were combined to find genes harboring deleterious ultra-rare variants and biological pathways associated with nsCPO by gene set enrichment analysis (GSEA) in a nsCPO cohort and in unaffected controls. Moreover, we assessed that genes implicated in nsCPO pathobiology ranked among the most enriched in ultra-rare variants in the nsCPO cohort.
The aim of this study was to examine the role of rare variants that are highly likely to be functionally damaging and/or pathogenic. We believe this may be a valuable resource for future investigations to define nsCPO etiology, a necessary step to develop prevention strategies and improve patient care.
2. Materials and Methods
2.1. Sample Collection
Following the approval by the Human Research Ethics Committee of Area Vasta Emilia Centro (Cod. CE: 14020/2017) and Ethics Committee of Isfahan University of Medical Sciences (IR.MUI.MED.REC.1399.178), we enrolled 30 Italian and 7 Iranian patients affected by cleft palate only as unique malformation (nsCPO). Exclusion criteria were the presence of other congenital anomalies or major diseases, exposure to known risk factors such as phenytoin and warfarin, and tobacco smoking and alcohol consumption during pregnancy. An additional inclusion criterion, only for the Iranian probands, was being offspring of a consanguineous mating, i.e., offspring of first- or second-degree cousins. Consanguinity may increase the risk factor of non-syndromic orofacial cleft, especially if rare autosomal recessive alleles are causally implicated. Nine out of 30 Italian and 2 out of 7 Iranian cases had one or more relatives (second, third, or fourth degree) reported as being affected by nsCPO. As control samples, we used 38 genomically unrelated parents from trios sequenced as part of the clinical routine at the Medical Genetics Unit, IRCCS AOU Bologna, Italy. WES data from these subjects were used as control, as their offspring were found to not be affected by orofacial cleft. Written informed consent was collected from all involved subjects at the time of recruitment at each clinical site, including patients, their parents, and/or additional informative on family members, when available.
2.2. Sequencing and Variant Detection
Genomic DNA of the 37 nsCPO cases were sequenced at Theragen Etex Bio (Kwanggyo Technovally, lui-dong Suwon, KOREA) (14 samples) or Novogene (Wan chai, Hong Kong) (23 samples), using the SureSelect XT Human All Exon V6 kit. Controls were sequenced at Macrogen (Seoul, Republic of Korea) using the SureSelect Human All Exon V6 (Agilent, Santa Clara, CA, USA).
Raw reads were processed as described elsewhere [12,13,14]. In particular, cases’ and controls’ variants were called and recalibrated according to GATK “Best Practices” [15] and annotated using the Ensembl tool Variant Effect Predictor v.76 [16] against GENCODE [17] canonical transcripts on reference genome GRCh37, using dbNSFP (dbNSFP v.3.5a) [18], CADD (GRCh37 v1.4) [19], and dbscSNV (dbscSNV v1.1) plugins. Runs of Homozygosity (ROHs) were detected from sequencing data in Iranian and Italian cases, and controls using AUDACITY [20]. Systematic ancestral differences between cases and controls were evaluated using Principal Component Analysis (PCA) through EIGENSOFT. PCA was conducted on 57,215 common HapMap SNPs extracted from sequencing data. Sequencing quality was measured per-sample using the GATK Depth of Coverage (GATK v.3.8, DoC) utility to obtain the mean sequencing coverage and percentage of bases covered <20× across all GENCODE protein-coding exons and across all exons for each gene. To address the confounding effect due to an imbalance of coverage, we excluded the genes with uneven coverage in cases and controls using the following criteria (coverage harmonization):
- Any gene should have <20% of bases covered <20× in each sample;
- Any gene should not differ by >10% in mean coverage between cases and controls.
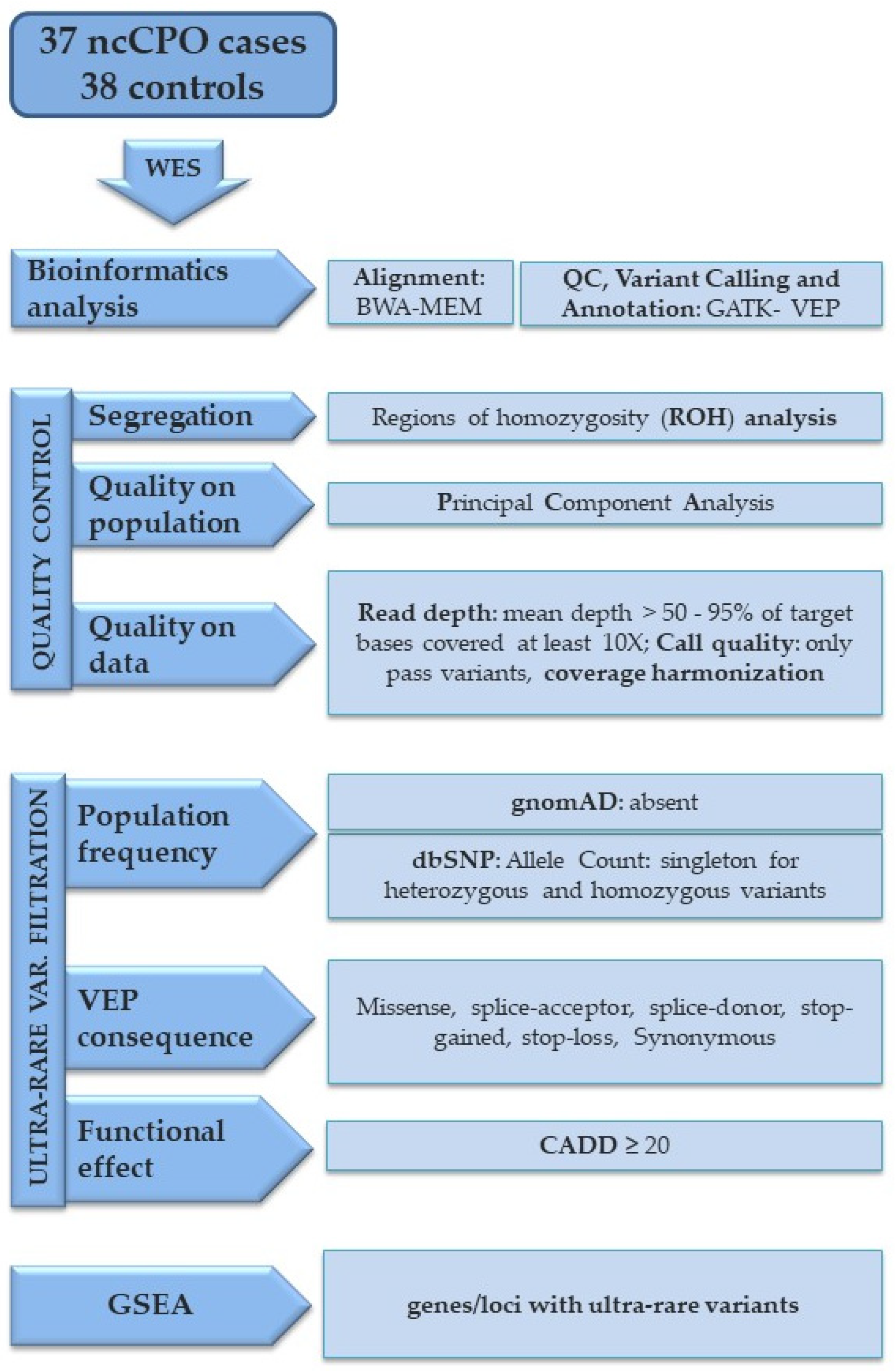
A schematic overview of the bioinformatics steps, the metrics used to decide whether to include samples and genes in the downstream analyses, and the filters used for selecting ultra-rare variants are summarized in Figure 1.
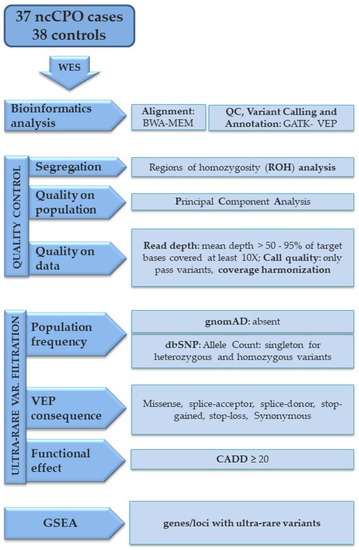
Figure 1.
Flowchart of the analysis steps for generating gene lists with ultra-rare variants. The flowchart provides an overview of the approach used to select genes carrying ultra-rare variants from whole exome sequencing data for GSEA. Raw reads were aligned against hg19 reference genome using BWA MEM and, after duplicates were removed, SNVs were called using GATK. ROH analysis and PCA were performed as reported in the Methods section. Only the variants with a PASS filter were retained in the VCF file. Quality metrics were used to decide whether to include genes in downstream analysis; only genes with mean depth > 50× and 95% of target bases covered at least 10× were included. Moreover, genes with differences in coverage between cases and controls were excluded (see details for this in the Methods section). Variants predicted to be protein-altering (missense, splice-acceptor, splice-donor, stop-gained, stop-loss) were aggregated. Synonymous variants were only used in GSEA as a comparison group of variants, as they are not as likely to be implicated in nsCPO. Population frequency filter and functional effect predictor score were applied as described in the Methods section.
2.3. Gene-Set Enrichment Analysis (GSEA)
Because the analysis of ultra-rare variants in gene sets has more statistical power than a gene-per-gene analysis, we could use more stringent conditions for filtering variants in Gene-Set Enrichment Analysis (GSEA) than in Gene-based Collapsing Analysis (GCA). In particular, we limited GSEA analysis to ultra-rare variants absent in gnomAD database v2.1.1 with a CADD score ≥ 20 (Figure 1), while we included variants present in gnomAD at low minor allele frequency (MAF) (<0.01) and without CADD restrictions in GCA. GSEA was performed by up-loading lists of genes with ultra-rare variants separately, in cases and controls, on the ToppGene portal (https://toppgene.cchmc.org) accessed on 16 July 2022 [21]. p-values for each of the two groups resulted from Topp.Fun analysis corrected for multiple testing with Benjamini-Hochberg p-values of the terms for each GSEA category (Gene Ontology [GO], GO Molecular Function [GO:MF], and GO Cellular Function [GO:CF]). p-values of cases were compared to those of controls and of cases’ synonymous variants to confirm that the enrichment was observed only for protein-altering variants in nsCPO [22]. Gene pathway annotations were retrieved from the KEGG database.
2.4. Gene-Set Burden and Variants Analysis
A hypergeometric test was performed to assess whether genes relevant to nsCPO pathobiology (n = 25) preferentially achieved lower p-values (< 0.05 nominal p-values) in GCA compared to the rest of protein-coding genes. The list of genes relevant to nsCPO pathobiology was obtained by intersecting multiple lists derived from Online Mendelian Inheritance in Man (OMIM), Human Phenotype Ontology, Gene-Ontology, Genecard, and Malacard databases. OMIM and GenCards were searched by Boolean string: “cleft palate” OR “bifid uvula” OR “cleft uvula”; specific reference IDs were used in the other cases: HumanPhenotypeOntology HP:0000175, GeneOntology GO:0060021 and GO:1905748, MalaCards CLF027. The list of genes that are nominally enriched in ultra-rare variants in the nsCPO cohort was obtained by GCA to evaluate the per-gene rare variant burden in cases versus controls. In GCA, per-gene variant counts were performed in the whole cohort using an in-house Perl script. We assigned a binary variable to each subject based on absence/presence, respectively, of any number of variants per subject. The number of cases and controls with at least one variant was used to assess enrichment for ultra-rare variants in either group using Fisher’s exact test. The nominal significance level, to identify genes nominally enriched in ultra-rare variants in cases compared to controls, was 0.05.
Statistical tests were performed in R v 3.5.1. Clinical significance of ultra-rare variants was assessed by manual curation of their list according to the American College for Medical Genetics (ACMG) standards and guidelines [23], restricted to genes that had orofacial clefts in their OMIM clinical synopsis. Moreover, we investigated whether there was a significant difference in the number of ultra-rare homozygous variants due to reported consanguinity in families of Iranian nsCPO cases compared to Italian cases.
Segregation analysis in informative families was performed, when possible, by variant-site targeted PCR and Sanger sequencing, only for variants identified in genes with the nominally significant p-values from GCA.
3. Results
3.1. Quality Checks
After quality checks of sequencing data, two cases did not achieve per-sample coverage thresholds (Table S1). As expected, PCA showed ample demarcation between Italian and Iranian samples, but suggested substantial clustering between cases and controls of the same geographic origin (Figure S1). As stated in the Methods section, GSEA has more power than GCA because it is applied to groups of genes instead of single genes. For this reason, although population stratification is not considered to be a crucial confounder for ultra-rare variants [24,25], we decided to apply PCA indications in GSEA, limiting this analysis to the 28 Italian cases and 38 controls. As GCA in this study is not used to carry out an association analysis, but rather to obtain a list of top-ranking genes for the Gene-Set burden test, we instead included all the samples passing quality checks and all variants with MAF ≤ 0.01 and no CADD restriction.
3.2. GSEA Results
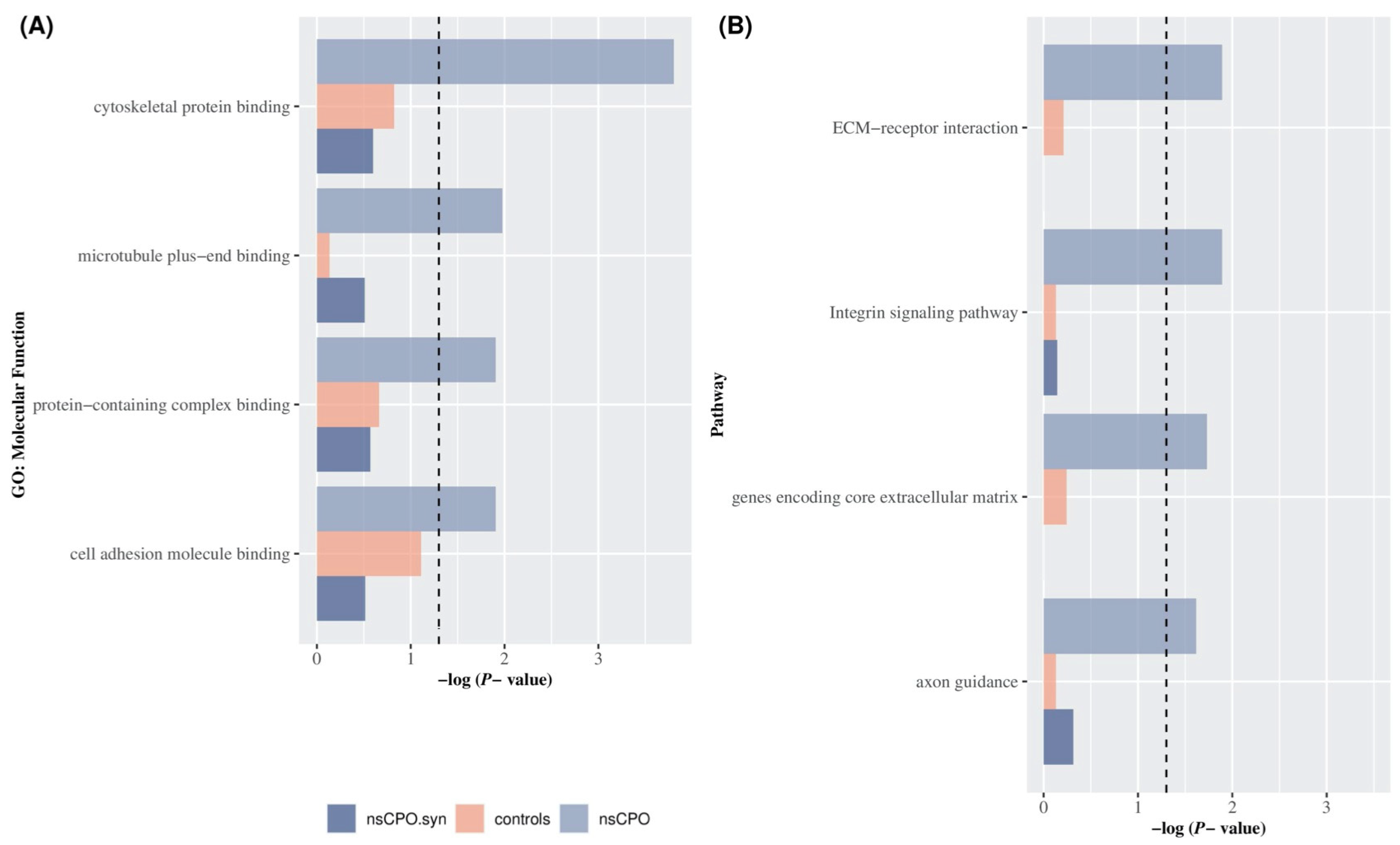
Using the most stringent conditions defined in the Methods section, ultra-rare variants identified 586 genes in cases and 708 in controls, in which most variants were missense across both groups. Molecular functions [GO:MF], KEGG pathways, and Cellular Function [GO:CF] terms were significantly enriched in gene sets identified by mutations in nsCPO cases. However, only a few terms showed exclusive enrichment in cases, i.e., there was no evidence of enrichment in controls or for gene sets identified by cases’ synonymous variants (Figure 2A). The most significantly enriched MF process was the cytoskeletal protein binding (GO:0008092) (BH corrected p-value = 1.57 × 10−4 for cases; p-value = 0.15 for controls; p-value = 0.25 for synonymous variants). Other significantly enriched MF included microtubule plus-end binding (GO:0051010), the protein-containing complex binding (GO:0044877), and the cell adhesion molecule binding (GO:0050839) (BH p-value = 1.06 × 10−2; 1.24 × 10−2; 1.24 × 10−2, respectively). Moreover, GSEA detected different pathways showing enrichment for genes with ultra-rare variants in cases (Figure 2B); among them were the ECM-receptor interaction pathway (BH p-value = 1.69 × 10−2) and the Integrin signaling pathway (p-value = 1.28 × 10−2) (Table 1).
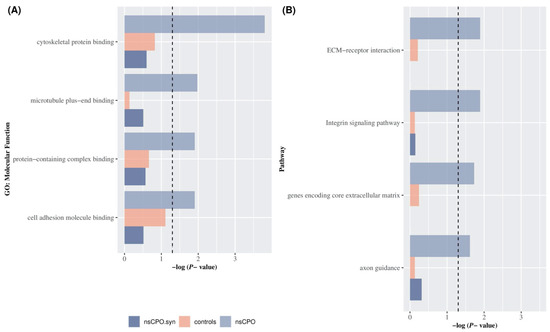
Figure 2.
Gene Set Enrichment Analysis for ultra-rare variants in nsCPO and unaffected controls. GSEA for genes carrying protein-altering (lilac), synonymous (purple) ultra-rare variants in nsCPO cases, and protein-altering ultra-rare variants in unaffected controls (pink). The X-axis shows a negative log-transformed BH p-value, and the horizontal dashed line indicates the nominally significance threshold (p-value = 0.05). (A) BH p-value for genes with protein-altering and synonymous ultra-rare variants in nsCPO and protein-altering ultra-rare variants in unaffected controls for the top 5 significant Molecular Function (GO:MF) terms. (B) BH p-value for genes with protein-altering and synonymous ultra-rare variants in nsCPO cases and protein-altering ultra-rare variants in unaffected controls for the top 5 significant pathway terms.

Table 1.
Genes carrying ultra-rare variants in nsCPO patients belonging to significant Gene Ontology categories identified by gene set enrichment analysis.
3.3. Gene-Set Burden Test Results
While GSEA pointed to interesting pathways, which turned out to be significantly enriched ultra-rare variants in nsCPO cases, GCA ranked genes according to their enrichment of ultra-rare variants in cases compared to controls, and was instrumental in demonstrating that GCA top-ranking genes (that is, genes having the lowest p-values) were significantly over-represented in the list of genes relevant to nsCPO pathobiology [24,25]. All samples considered, there were 16,749 ultra-rare variants (16,230 missense, 348 stop-gained or -loss, 171 splice-acceptor or -donor) in 7496 genes, according to the quality criteria defined in the Methods section. Although no gene reached the genome-wide significance level of association (Table 2), two genes in the nsCPO pathobiology list, namely COL2A1 and GLI3, achieved a nominal level of significance in the association test (Fisher’s Exact Test, p-value < 0.05) for the presence of an excess of ultra-rare damaging variants in cases compared to controls (Table 2 and Table 3). Genes relevant to nsCPO pathobiology achieved lower p-values compared to the other protein-coding genes included in GCA analysis (hypergeometric test, p-value = 5.86 × 10−3).

Table 2.
Genes obtaining nominal p-value < 0.05 in GCA.

Table 3.
Variants identified by GCA in COL2A1 and GLI3 are detailed.
3.4. Clinical Variants
An analysis guided by ACMG criteria revealed that four nsCPO cases (10.8% of the sample study) carried pathogenic (P) or likely pathogenic (LP) variants in genes causing autosomal dominant syndromes, with cleft palate in their clinical synopsis (Table 4). Four of the homozygous ultra-rare variants identified in Iranian cases were inside large ROHs, and one of those involves an autosomal dominant disease gene with cleft palate as a clinical feature (NM_198216.2:c.686-136A>T). The number of genes with homozygous ultra-rare variants in Iranian cases did not differ significantly from Italian cases (8 SNVs in 7 samples versus 19 SNVs in 27 samples; Wilcoxon signed rank test p-value > 0.05) (Table S2), suggesting that no plausible recessive variant was implicated in nsCPO pathogenesis, despite the high consanguinity rate and the excess of genomic inbreeding of the Iranian cases compared to Italian samples (Figure S2).

Table 4.
Pathogenic and likely pathogenic variants of cases identified in genes associated with syndromes involving CPO.
Ultra-rare variants in clinically relevant genes were tested for segregation by Sanger sequencing in the cases and in their available relatives (Figures S3 and S4). Segregation analysis found that most of the ultra-rare variants were inherited from one of the healthy parents, suggesting incomplete penetrance. Interestingly, COL2A1 (NM_001844.5:c.2659C>T) missense P SNV was found to be de novo and the IRF6 (NM_006147.4:c.82T>G) missense LP SNV segregated with nsCPO in the family.
4. Discussion
During morphogenesis, cells need to divide, move, and interact with each other and the extracellular matrix (ECM). Different events must be coordinated in terms of timing and space in a succession of perfectly orchestrated steps. When the coordinated interplay of individual cell behavior, with regard to migration, adhesion, growth, differentiation, and apoptosis, is disrupted, it can lead to phenotypic alterations, such as orofacial clefts. During palatogenesis, palatine shelves modify their position from vertical to horizontal above the tongue, interface at the midline, adhere to each other, and fuse. Each step involves epithelial and mesenchymal cells that are subject to a dynamic remodeling that involves their cytoskeleton and cellular structures, named cell junctions.
Despite the huge efforts spent to clarify the causes leading to orofacial clefts, the genetic etiology of nsCPO has remained largely elusive. Recently, however, a large-scale sequencing study of 756 trio genomes implicated de novo mutations in the pathogenesis of orofacial clefts (OFC) [22]. Although only a minority of the cases in this study had nsCPO (58), they were found to have an overall significant excess of de novo mutations in protein-coding genes, and enrichment analysis pointed to gene sets involved in limb-bud formation and many craniofacial disease terms. Based on the hypothesis that ultra-rare variants might explain at least part of the heritability of nsCPO, we conducted a WES study to identify genes that individually and collectively contribute to nsCPO risk. Our results support this hypothesis in three ways:
- GSEA highlighted genes with ultra-rare variants cluster into gene ontology sets that are highly relevant to nsCPO pathobiology (cytoskeleton rearrangement, cell-adhesion, ECM-cell interaction);
- GCA identified two top-ranking genes (COL2A1 and GLI3) significantly overlapping in the list of genes relevant to nsCPO pathobiology;
- Pathogenic and likely pathogenic variants in genes, accounting for autosomal dominant orofacial cleft syndromes (CDON, COL2A1, IRF6, SNRPB), have been identified in four cases (10.8%).
Cytoskeleton, cell junctions, and ECM hold a network of proteins strictly involved in the dynamic morphogenetic events that characterize palate development. The transmembrane glycoproteins of the cell junctions are characterized by an extracellular binding site for transmembrane proteins of other cells, or ECM elements, as well as a cytosolic domain, that indirectly interacts with the cytoskeleton and is involved in the regulation of important signaling pathways regulation. Enrichment analysis strongly indicates that rare damaging variants preferably occur in genes coding for these groups of proteins. Some of them have been proposed as candidate genes for OFC malformations.
The active role of actin filaments in mesenchymal reorganization during palatogenesis has already been evidenced [26], as has actomyosin-dependent cell contractility [27]. Based on these observations, genetic association between variants in the MYH9 gene and orofacial cleft has been demonstrated in several studies [28,29,30]. Interestingly, GSEA of genes carrying mutations in our dataset identified higher concordance with the cytoskeletal protein binding gene ontology set (GO:0008092). Among these genes, additional myosin family members are included (MYH15, MYH2, MYH3, MYH4, MYO18B, MYO15A). In Tbx1-knockout palatal shelves, a gene profile analysis showed that myosin heavy chain 3 (Myh3) and nebulin (Neb) were downregulated [31]. Other genes included in the GO:0008092 list have been related in some way to the OFC onset. Alpha-parvin (PARVA), considered a candidate gene for nsCL/P, was demonstrated to be involved in cell proliferation and migration of human oral keratinocytes by RNAi silencing experiments [32]. Filamin B (FLNB) is an actin-binding protein that interacts with receptors and intracellular proteins that regulate cytoskeleton-dependent cell proliferation, differentiation, and migration [33]. Pathogenic variants in FLNB cause disorders presenting a spectrum of phenotypes, which sometimes includes cleft palate [34,35]. Maternal mutations at FLNB were suggested to influence the risk of cleft in the offspring based on an interaction between maternal gene and specific teratogens or fetal genes [36]. A study of environmental exposures and the effect of parent-of-origin on a CL/P cohort evidenced a possible association between SNPs in ANK3 and maternal smoking [37].
In our patients, we found a significant enrichment of ultra-rare mutations in genes encoding protein-containing complex binding (GO:0044877), which is a process crucial for several morphogenetic steps. Among these genes, the most studied was GAD1, which encodes glutamate decarboxylase-67 (GAD67), an enzyme with a main role in the γ-aminobutyric acid (GABA) metabolism. In mouse and rat models, Gad1 played a role in the normal development of the palate [38,39], while a study on the role of miRNAs in the regulation of CP genes found that variants in GAD1 significantly contributed to the human cleft palate phenotype [40]. An association study carried out on non-Hispanic white families revealed a significant association between nsCL/P and rs1046117 mapping on Fos proto-oncogene (FOS) [41]. In addition, the association of noggin (NOG) with nsOFC was confirmed by GWASs and confirmed in a recent meta-analysis [42,43,44].
Two genes were among the genes with nominal enrichment in the ultra-rare variant collapsing analysis and are also included in the list of genes relevant to nsCPO pathobiology: type II collagen alpha-1 chain (COL2A1) and glioma-associated oncogene family zinc finger 3 (GLI3). Interestingly, COL2A1 is listed in three enriched pathways identified by GSEA, whereas the GLI3 gene is found in the protein-containing complex-binding gene category of the ontology. COL2A1 is a fibrillar collagen typical of cartilaginous tissues and essential for normal embryonic skeletal development. A variety of mutations in the gene sequence can lead to a range of COL2A1-related disorders exhibiting a phenotypic spectrum that includes cleft palate [45]. The first evidence for COL2A1 involvement in the occurrence of nsCPO was provided by Nikopensius and colleagues, who found multiple haplotypes in COL2A1 (and COL11A2), associated with nsCPO, in an association study carried out on a cohort composed of 104 cases and 606 controls from the Baltic region [46]. GLI3 is one among the three members of the GLI family of zinc finger transcription effectors of the sonic hedgehog signaling pathway. GLI family members regulate gene expression and repression through the transduction operated by primary cilia in many tissues at various phases of embryonic morphogenesis. In particular, GLI3 mostly acts as a repressor. In 2008, Huang’s group correlated GLI3 deficiency with a high incidence of cleft palate, associated with abnormal tongue development, in a murine model [47]. In an association study conducted on 504 Chinese cases and 455 healthy controls, risk variants of GLI3 were identified as significantly associated with nsCL/P susceptibility [48]. The pivotal role played by primary cilium during the development of the orofacial region could be confirmed by the fact that more than 30% of ciliopathies are mainly defined by craniofacial defects, including lip/palate clefting.
Finally, we identified LP/P variants in genes associated with AD syndromes with cleft palate in their clinical synopsis. The nonsense variant in COL2A1 (NM_001844.5:c.2659C>T; p.Arg887Ter) was previously reported in Clinvar (Table 4), as implicated in Stickler syndrome [49,50]. In addition, the missense variant in IRF6 (NM_006147.4:c.82T>G; p.Trp28Gly) has been reported in ClinVar as variant of uncertain significance. In contrast, the stop-loss variant in SNRPB and the nonsense variant in CDON are novel findings and are not observed in the general population (gnomAD), nor have they been previously identified among patients.
Some interesting lines of discussion emerge from the points above. First, the present study confirms that nsCPO etiology is complex and also suggests that ultra-rare variants can act as susceptibility factors in nsCPO onset. Nonetheless, a portion of cases, albeit small, is attributable to monogenic causes of disease, being carriers of pathogenic or likely-pathogenic variants, strongly suggesting that ultra-rare variants contribute substantially to risk of nsCPO. Interestingly, the GCA and GSEA also support this view. The main limitation of this study was the small sample size, considering that nsCPO is a complex disease. In principle, the lack of Iranian controls may have affected the results of GCA analysis, but this is unlikely to have a tremendous impact because Iranian samples are a clear minority of the total cases. Nonetheless, indications of specific genes emerge quite clearly. COL2A1 is emblematic in this sense. Indeed, it is not only found in a case with a de novo pathogenic variant, but it is also among the genes with the lowest p-values in GCA and is represented in four significantly enriched gene sets. This is consistent with a previous report describing COL2A1 variants as a risk factor for nsCPO [46]. Similarly, MACF1 emerged as a potential candidate gene, in line with previous findings by Bishop et al. [22], as it is present in three of our significantly enriched gene sets. These gene sets are all implicated in biological functions or processes that are relevant to nsCPO etiology, such as cytoskeletal organization, cellular adhesion, and interaction with ECM, which are all crucial during palate morphogenesis.
Another limitation of this study is that Copy Number Variants (CNVs) were not included in the analysis. Although it is unlikely that, with the present small sample size, we could identify a CNV contribution, it has been recently suggested [51] that their role in orofacial clefts has been relatively overlooked.
In conclusion, WES analysis in patients with isolated cleft palate led to the discovery of ultra-rare variants in a number of genes already in the spotlight as candidate genes for orofacial cleft. We suggest that the approach is effective and allows us to identify relevant pathways in nsCPO pathobiology. Identification or confirmation of genes likely to contribute to the risk of nsCPO expands our knowledge of the genetic architecture and etiopathogenesis of this very common craniofacial birth defect. However, further investigations are needed in order to confirm the role of these genes in the development of the palate.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/biom13020236/s1, Figure S1: Principal component analysis (PCA) depicting the comparison between nsCPO and controls; Figure S2: Number and size of ROHs detected in Italian and Iranian samples; Figure S3: Sanger sequencing of the rare damaging variants of COL2A1 gene identified by WES; Figure S4: Sanger sequencing of the rare damaging variants of GLI3 gene identified by WES; Table S1: Sample coverage metrics for cases and controls; Table S2: Summary of homozygous ultra-rare variants in Iranian and Italian cases.
Author Contributions
Conceptualization, L.S., E.I., T.P. and M.M.; enrollment of patients and interpretation of data results, R.S., N.N., F.C. and M.S.; handling of biological samples, A.P. (Annalisa Palmieri) and A.P. (Agnese Pellati); acquisition and analysis of data, L.S., E.I., T.P. and M.M.; writing—original draft preparation, L.S., E.I., T.P. and M.M.; writing—review and editing, M.S. and M.M.; supervision, M.S. and M.M.; funding acquisition, M.M. and F.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Association Interethnos-Interplast Italy (to M.M.), from Ricerca Fondamentale Orientata (RFO, University of Bologna to M.M.) and from the University of Ferrara (FIR2019 to F.C.). The materials presented, and views expressed herein are the responsibility of the authors only. The sponsor takes no responsibility for any use of the information presented herein.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Human Research Ethics Committee of Area Vasta Emilia Centro (Cod. CE: 14020/2017) and Ethics Committee of Isfahan University of Medical Sciences (IR.MUI.MED.REC.1399.178).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The datasets presented in this study can be found in an online repository. The name of the repository and accession number can be found below: https://www.ncbi.nlm.nih.gov/sra/PRJNA870708.
Acknowledgments
We thank the patients and their families for supporting this study.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Ferguson, M.W. Palate development. Development 1988, 103, 41–60. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.; Bian, Z.; Torensma, R.; Von den Hoff, J.W. Biological mechanisms in palatogenesis and cleft palate. J. Dent. Res. 2009, 88, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Grosen, D.; Chevrier, C.; Skytthe, A.; Bille, C.; Molsted, K.; Sivertsen, A.; Murray, J.C.; Christensen, K. A cohort study of recurrence patterns among more than 54,000 relatives of oral cleft cases in Denmark: Support for the multifactorial threshold model of inheritance. J. Med. Genet. 2010, 47, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Martinelli, M.; Palmieri, A.; Carinci, F.; Scapoli, L. Non-syndromic Cleft Palate: An Overview on Human Genetic and Environmental Risk Factors. Front Cell Dev. Biol. 2020, 8, 592271. [Google Scholar] [CrossRef]
- Beaty, T.H.; Ruczinski, I.; Murray, J.C.; Marazita, M.L.; Munger, R.G.; Hetmanski, J.B.; Murray, T.; Redett, R.J.; Fallin, M.D.; Liang, K.Y.; et al. Evidence for gene-environment interaction in a genome wide study of nonsyndromic cleft palate. Genet Epidemiol. 2011, 35, 469–478. [Google Scholar] [CrossRef]
- Leslie, E.J.; Liu, H.; Carlson, J.C.; Shaffer, J.R.; Feingold, E.; Wehby, G.; Laurie, C.A.; Jain, D.; Laurie, C.C.; Doheny, K.F.; et al. A Genome-wide Association Study of Nonsyndromic Cleft Palate Identifies an Etiologic Missense Variant in GRHL3. Am. J. Hum. Genet. 2016, 98, 744–754. [Google Scholar] [CrossRef]
- Tam, V.; Patel, N.; Turcotte, M.; Bosse, Y.; Pare, G.; Meyre, D. Benefits and limitations of genome-wide association studies. Nat. Rev. Genet. 2019, 20, 467–484. [Google Scholar] [CrossRef]
- Gudmundsson, S.; Singer-Berk, M.; Watts, N.A.; Phu, W.; Goodrich, J.K.; Solomonson, M.; Genome Aggregation Database, C.; Rehm, H.L.; MacArthur, D.G.; O’Donnell-Luria, A. Variant interpretation using population databases: Lessons from gnomAD. Hum. Mutat. 2022, 43, 1012–1030. [Google Scholar] [CrossRef]
- Bonnefond, A.; Froguel, P. Rare and common genetic events in type 2 diabetes: What should biologists know? Cell Metab. 2015, 21, 357–368. [Google Scholar] [CrossRef]
- Hoebel, A.K.; Drichel, D.; van de Vorst, M.; Bohmer, A.C.; Sivalingam, S.; Ishorst, N.; Klamt, J.; Golz, L.; Alblas, M.; Maaser, A.; et al. Candidate Genes for Nonsyndromic Cleft Palate Detected by Exome Sequencing. J. Dent. Res. 2017, 96, 1314–1321. [Google Scholar] [CrossRef]
- Liu, H.; Busch, T.; Eliason, S.; Anand, D.; Bullard, S.; Gowans, L.J.J.; Nidey, N.; Petrin, A.; Augustine-Akpan, E.A.; Saadi, I.; et al. Exome sequencing provides additional evidence for the involvement of ARHGAP29 in Mendelian orofacial clefting and extends the phenotypic spectrum to isolated cleft palate. Birth Defects Res. 2017, 109, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Magini, P.; Pippucci, T.; Tsai, I.C.; Coppola, S.; Stellacci, E.; Bartoletti-Stella, A.; Turchetti, D.; Graziano, C.; Cenacchi, G.; Neri, I.; et al. A mutation in PAK3 with a dual molecular effect deregulates the RAS/MAPK pathway and drives an X-linked syndromic phenotype. Hum. Mol. Genet. 2014, 23, 3607–3617. [Google Scholar] [CrossRef] [PubMed]
- Pippucci, T.; Maresca, A.; Magini, P.; Cenacchi, G.; Donadio, V.; Palombo, F.; Papa, V.; Incensi, A.; Gasparre, G.; Valentino, M.L.; et al. Homozygous NOTCH3 null mutation and impaired NOTCH3 signaling in recessive early-onset arteriopathy and cavitating leukoencephalopathy. EMBO Mol. Med. 2015, 7, 848–858. [Google Scholar] [CrossRef] [PubMed]
- Pippucci, T.; Licchetta, L.; Baldassari, S.; Marconi, C.; De Luise, M.; Myers, C.; Nardi, E.; Provini, F.; Cameli, C.; Minardi, R.; et al. Contribution of ultrarare variants in mTOR pathway genes to sporadic focal epilepsies. Ann. Clin. Transl. Neurol. 2019, 6, 475–485. [Google Scholar] [CrossRef]
- Kendig, K.I.; Baheti, S.; Bockol, M.A.; Drucker, T.M.; Hart, S.N.; Heldenbrand, J.R.; Hernaez, M.; Hudson, M.E.; Kalmbach, M.T.; Klee, E.W.; et al. Sentieon DNASeq Variant Calling Workflow Demonstrates Strong Computational Performance and Accuracy. Front Genet. 2019, 10, 736. [Google Scholar] [CrossRef] [PubMed]
- McLaren, W.; Gil, L.; Hunt, S.E.; Riat, H.S.; Ritchie, G.R.; Thormann, A.; Flicek, P.; Cunningham, F. The Ensembl Variant Effect Predictor. Genome Biol. 2016, 17, 122. [Google Scholar] [CrossRef]
- Harrow, J.; Frankish, A.; Gonzalez, J.M.; Tapanari, E.; Diekhans, M.; Kokocinski, F.; Aken, B.L.; Barrell, D.; Zadissa, A.; Searle, S.; et al. GENCODE: The reference human genome annotation for The ENCODE Project. Genome Res. 2012, 22, 1760–1774. [Google Scholar] [CrossRef]
- Liu, X.; Li, C.; Mou, C.; Dong, Y.; Tu, Y. dbNSFP v4: A comprehensive database of transcript-specific functional predictions and annotations for human nonsynonymous and splice-site SNVs. Genome Med. 2020, 12, 103. [Google Scholar] [CrossRef]
- Rentzsch, P.; Schubach, M.; Shendure, J.; Kircher, M. CADD-Splice-improving genome-wide variant effect prediction using deep learning-derived splice scores. Genome Med. 2021, 13, 31. [Google Scholar] [CrossRef]
- Magi, A.; Giangregorio, T.; Semeraro, R.; Carangelo, G.; Palombo, F.; Romeo, G.; Seri, M.; Pippucci, T. AUDACITY: A comprehensive approach for the detection and classification of Runs of Homozygosity in medical and population genomics. Comput Struct Biotechnol. J. 2020, 18, 1956–1967. [Google Scholar] [CrossRef]
- Chen, J.; Xu, H.; Aronow, B.J.; Jegga, A.G. Improved human disease candidate gene prioritization using mouse phenotype. BMC Bioinform. 2007, 8, 392. [Google Scholar] [CrossRef] [PubMed]
- Bishop, M.R.; Diaz Perez, K.K.; Sun, M.; Ho, S.; Chopra, P.; Mukhopadhyay, N.; Hetmanski, J.B.; Taub, M.A.; Moreno-Uribe, L.M.; Valencia-Ramirez, L.C.; et al. Genome-wide Enrichment of De Novo Coding Mutations in Orofacial Cleft Trios. Am. J. Hum. Genet. 2020, 107, 124–136. [Google Scholar] [CrossRef] [PubMed]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015, 17, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Shi, G. On rare variants in principal component analysis of population stratification. BMC Genet. 2020, 21, 34. [Google Scholar] [CrossRef] [PubMed]
- Raghavan, N.S.; Brickman, A.M.; Andrews, H.; Manly, J.J.; Schupf, N.; Lantigua, R.; Wolock, C.J.; Kamalakaran, S.; Petrovski, S.; Tosto, G.; et al. Whole-exome sequencing in 20,197 persons for rare variants in Alzheimer’s disease. Ann. Clin. Transl. Neurol. 2018, 5, 832–842. [Google Scholar] [CrossRef] [PubMed]
- Lessard, J.L.; Wee, E.L.; Zimmerman, E.F. Presence of contractile proteins in mouse fetal palate prior to shelf elevation. Teratology 1974, 9, 113–125. [Google Scholar] [CrossRef]
- Kim, S.; Lewis, A.E.; Singh, V.; Ma, X.; Adelstein, R.; Bush, J.O. Convergence and extrusion are required for normal fusion of the mammalian secondary palate. PLoS Biol. 2015, 13, e1002122. [Google Scholar] [CrossRef]
- Martinelli, M.; Di Stazio, M.; Scapoli, L.; Marchesini, J.; Di Bari, F.; Pezzetti, F.; Carinci, F.; Palmieri, A.; Carinci, P.; Savoia, A. Cleft lip with or without cleft palate: Implication of the heavy chain of non-muscle myosin IIA. J. Med. Genet. 2007, 44, 387–392. [Google Scholar] [CrossRef]
- Birnbaum, S.; Reutter, H.; Mende, M.; de Assis, N.A.; Diaz-Lacava, A.; Herms, S.; Scheer, M.; Lauster, C.; Braumann, B.; Schmidt, G.; et al. Further evidence for the involvement of MYH9 in the etiology of non-syndromic cleft lip with or without cleft palate. Eur. J. Oral. Sci. 2009, 117, 200–203. [Google Scholar] [CrossRef]
- Chiquet, B.T.; Hashmi, S.S.; Henry, R.; Burt, A.; Mulliken, J.B.; Stal, S.; Bray, M.; Blanton, S.H.; Hecht, J.T. Genomic screening identifies novel linkages and provides further evidence for a role of MYH9 in nonsyndromic cleft lip and palate. Eur. J. Hum. Genet. 2009, 17, 195–204. [Google Scholar] [CrossRef]
- Funato, N.; Yanagisawa, H. Deletion of the T-box transcription factor gene, Tbx1, in mice induces differential expression of genes associated with cleft palate in humans. Arch. Oral. Biol. 2018, 95, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Li, M.X.; Li, Z.; Zhang, R.; Yu, Y.; Wang, L.S.; Wang, Q.; Ding, Z.; Zhang, J.P.; Zhang, M.R.; Xu, L.C. Effects of small interfering RNA-mediated silencing of susceptibility genes of non-syndromic cleft lip with or without cleft palate on cell proliferation and migration. Int. J. Pediatr. Otorhinolaryngol. 2020, 138, 110382. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Lu, J.; Lian, G.; Ferland, R.J.; Dettenhofer, M.; Sheen, V.L. Formin 1 and filamin B physically interact to coordinate chondrocyte proliferation and differentiation in the growth plate. Hum. Mol. Genet. 2014, 23, 4663–4673. [Google Scholar] [CrossRef] [PubMed]
- Robertson, S. FLNB Disorders. In GeneReviews((R)); Adam, M.P., Mirzaa, G.M., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Gripp, K.W., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Li, B.C.; Hogue, J.; Eilers, M.; Mehrotra, P.; Hyland, J.; Holm, T.; Prosen, T.; Slavotinek, A.M. Clinical report: Two patients with atelosteogenesis type I caused by missense mutations affecting the same FLNB residue. Am. J. Med. Genet A 2013, 161A, 619–625. [Google Scholar] [CrossRef]
- Jugessur, A.; Shi, M.; Gjessing, H.K.; Lie, R.T.; Wilcox, A.J.; Weinberg, C.R.; Christensen, K.; Boyles, A.L.; Daack-Hirsch, S.; Nguyen, T.T.; et al. Maternal genes and facial clefts in offspring: A comprehensive search for genetic associations in two population-based cleft studies from Scandinavia. PLoS ONE 2010, 5, e11493. [Google Scholar] [CrossRef]
- Haaland, O.A.; Romanowska, J.; Gjerdevik, M.; Lie, R.T.; Gjessing, H.K.; Jugessur, A. A genome-wide scan of cleft lip triads identifies parent-of-origin interaction effects between ANK3 and maternal smoking, and between ARHGEF10 and alcohol consumption. F1000Res 2019, 8, 960. [Google Scholar] [CrossRef]
- Oh, W.J.; Westmoreland, J.J.; Summers, R.; Condie, B.G. Cleft palate is caused by CNS dysfunction in Gad1 and Viaat knockout mice. PLoS ONE 2010, 5, e9758. [Google Scholar] [CrossRef]
- Jiang, W.; Kakizaki, T.; Fujihara, K.; Miyata, S.; Zhang, Y.; Suto, T.; Kato, D.; Saito, S.; Shibasaki, K.; Ishizaki, Y.; et al. Impact of GAD65 and/or GAD67 deficiency on perinatal development in rats. FASEB J. 2022, 36, e22123. [Google Scholar] [CrossRef]
- Suzuki, A.; Abdallah, N.; Gajera, M.; Jun, G.; Jia, P.; Zhao, Z.; Iwata, J. Genes and microRNAs associated with mouse cleft palate: A systematic review and bioinformatics analysis. Mech. Dev. 2018, 150, 21–27. [Google Scholar] [CrossRef]
- Chiquet, B.T.; Yuan, Q.; Swindell, E.C.; Maili, L.; Plant, R.; Dyke, J.; Boyer, R.; Teichgraeber, J.F.; Greives, M.R.; Mulliken, J.B.; et al. Knockdown of Crispld2 in zebrafish identifies a novel network for nonsyndromic cleft lip with or without cleft palate candidate genes. Eur. J. Hum. Genet. 2018, 26, 1441–1450. [Google Scholar] [CrossRef]
- Figueiredo, J.C.; Ly, S.; Raimondi, H.; Magee, K.; Baurley, J.W.; Sanchez-Lara, P.A.; Ihenacho, U.; Yao, C.; Edlund, C.K.; van den Berg, D.; et al. Genetic risk factors for orofacial clefts in Central Africans and Southeast Asians. Am. J. Med. Genet. A 2014, 164A, 2572–2580. [Google Scholar] [CrossRef]
- Ray, D.; Venkataraghavan, S.; Zhang, W.; Leslie, E.J.; Hetmanski, J.B.; Weinberg, S.M.; Murray, J.C.; Marazita, M.L.; Ruczinski, I.; Taub, M.A.; et al. Pleiotropy method reveals genetic overlap between orofacial clefts at multiple novel loci from GWAS of multi-ethnic trios. PLoS Genet. 2021, 17, e1009584. [Google Scholar] [CrossRef] [PubMed]
- Slavec, L.; Karas Kuzelicki, N.; Locatelli, I.; Gersak, K. Genetic markers for non-syndromic orofacial clefts in populations of European ancestry: A meta-analysis. Sci. Rep. 2022, 12, 1214. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Huang, X.; Yuan, L. Molecular genetics of the COL2A1-related disorders. Mutat Res. Rev. Mutat Res. 2016, 768, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Nikopensius, T.; Jagomagi, T.; Krjutskov, K.; Tammekivi, V.; Saag, M.; Prane, I.; Piekuse, L.; Akota, I.; Barkane, B.; Krumina, A.; et al. Genetic variants in COL2A1, COL11A2, and IRF6 contribute risk to nonsyndromic cleft palate. Birth Defects Res. A Clin. Mol. Teratol. 2010, 88, 748–756. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Goudy, S.L.; Ketova, T.; Litingtung, Y.; Chiang, C. Gli3-deficient mice exhibit cleft palate associated with abnormal tongue development. Dev. Dyn. 2008, 237, 3079–3087. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, Y.; Huang, Y.; Pan, Y.; Shi, B.; Ma, J.; Ma, L.; Lan, F.; Zhou, Y.; Shi, J.; et al. The association study of nonsyndromic cleft lip with or without cleft palate identified risk variants of the GLI3 gene in a Chinese population. J. Genet. 2017, 96, 687–693. [Google Scholar] [CrossRef]
- Xiong, H.Y.; Alipanahi, B.; Lee, L.J.; Bretschneider, H.; Merico, D.; Yuen, R.K.; Hua, Y.; Gueroussov, S.; Najafabadi, H.S.; Hughes, T.R.; et al. RNA splicing. The human splicing code reveals new insights into the genetic determinants of disease. Science 2015, 347, 1254806. [Google Scholar] [CrossRef]
- Hoornaert, K.P.; Vereecke, I.; Dewinter, C.; Rosenberg, T.; Beemer, F.A.; Leroy, J.G.; Bendix, L.; Bjorck, E.; Bonduelle, M.; Boute, O.; et al. Stickler syndrome caused by COL2A1 mutations: Genotype-phenotype correlation in a series of 100 patients. Eur. J. Hum. Genet. 2010, 18, 872–880. [Google Scholar] [CrossRef]
- Lansdon, L.A.; Dickinson, A.; Arlis, S.; Liu, H.; Hlas, A.; Hahn, A.; Bonde, G.; Long, A.; Standley, J.; Tyryshkina, A.; et al. Genome-wide analysis of copy number variation in humans with cleft lip and/or cleft palate identifies COBLL1, RIC1, and ARHGEF38 as clefting genes. Am. J. Hum. Genet. 2022, 110, 71–91. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).