Recent Advances in Analytical Methods for the Detection of Olive Oil Oxidation Status during Storage along with Chemometrics, Authenticity and Fraud Studies
Abstract
:1. Introduction
2. Lipid Stability Measurements
2.1. Evaluation of Primary Oxidation Products (“Lipid Hydroperoxides”)
2.1.1. Measurement of Peroxide Value (PV)
2.1.2. Determination of Lipid Hydroperoxides with the Ferric Thiocyanate Method
2.1.3. Measurement of Conjugated Dienes (CD) and Conjugated Trienes (CT)
2.2. Determination of Secondary Oxidation Products (“off-Flavour” Volatiles)
2.2.1. p-Anisidine Value Test (PAV)
2.2.2. Determination of Thiobarbituric Acid-Related Substances (TBARs)
2.2.3. Determination of Volatile “off-Flavour” Oxidation Products with Gas Chromatography
- (I)
- Headspace Techniques (HS-SPME extraction)
- (II)
- Non-headspace techniques
2.3. Other Methods for the Evaluation of Oxidative Rancidity
2.3.1. Measurement of the Induction Time of Oxidation (Rancimat, Swift Test)
2.3.2. Other Recent Oxidation Techniques
3. Chemometric Application in Studies Related to Lipid Changes due to Storage
4. Adulteration of Olive Oil Related to Its Oxidation with Modern Analytical Techniques
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- International Olive Oil Council. Available online: https://www.internationaloliveoil.org/ (accessed on 9 July 2022).
- D’Adamo, I.; Falcone, P.M.; Gastaldi, M.; Morone, P. A social analysis of the olive oil sector: The role of family business. Re-Sources 2019, 8, 151. [Google Scholar] [CrossRef]
- Meenu, M.; Cai, Q.; Xu, B. A critical review on analytical techniques to detect adulteration of extra virgin olive oil. Trends Food Sci. Technol. 2019, 91, 391–408. [Google Scholar] [CrossRef]
- Aparicio, R.; Conte, L.S.; Ad Fiebig, H.J. Olive Oil Authentication. In Handbook of Olive Oil; Aparicio, R., Harwood, J., Eds.; Springer: Boston, MA, USA, 2013; pp. 431–478. [Google Scholar]
- Skiada, V.; Agriopoulou, S.; Tsarouhas, P.; Katsaris, P.; Stamatelopoulou, E.; Varzakas, T. Evaluation and Origin Discrimina-tion of Two Monocultivar Extra Virgin Olive Oils, Cultivated in the Coastline Part of North-Western Greece. Appl. Sci. 2020, 10, 6733. [Google Scholar] [CrossRef]
- Tarapoulouzi, M.; Skiada, V.; Agriopoulou, S.; Psomiadis, D.; Rébufa, C.; Roussos, S.; Theocharis, C.R.; Katsaris, P.; Varzakas, T. Chemometric Discrimination of the Geographical Origin of Three Greek Cultivars of Olive Oils by Stable Isotope Ratio Analysis. Foods 2021, 10, 336. [Google Scholar] [CrossRef]
- El Yamani, M.; Boussakouran, A.; Rharrabti, Y. Effect of storage time and conditions on the quality characteristics of ‘Mo-roccan Picholine’ olive oil. Biocatal. Agric. Biotechnol. 2022, 39, 102244. [Google Scholar] [CrossRef]
- Mousavi, S.; Mariotti, R.; Stanzione, V.; Pandolfi, S.; Mastio, V.; Baldoni, L.; Cultrera, N.G.M. Evolution of Extra Virgin Olive Oil Quality under Different Storage Conditions. Foods 2021, 10, 1945. [Google Scholar] [CrossRef]
- Codex Alimentarius Commission. Standard for Olive Oils and Olive Pomace Oils; CXS 33-1981; FAO: Rome, Italia, 1981. [Google Scholar]
- Conte, L.; Bendini, A.; Valli, E.; Lucci, P.; Moret, S.; Maquet, A.; Lacoste, F.; Brereton, P.; García-González, D.L.; Moreda, W.; et al. Olive oil quality and authenticity: A review of current EU legislation, standards, relevant methods of analyses, their drawbacks and recommendations for the future. Trends Food Sci. Technol. 2020, 105, 483–493. [Google Scholar] [CrossRef]
- Boskou, D.; Tsimidou, M.; Blekas, G. Polar phenolic compounds. In Olive Oil; AOCS Press: Thessaloniki, Greece, 2006; pp. 73–92. [Google Scholar]
- García-González, D.L.; Tena, N.; Aparicio, R. Characterization of olive paste volatiles to predict the sensory quality of virgin olive oil. Eur. J. Lipid Sci. Technol. 2007, 109, 663–672. [Google Scholar] [CrossRef]
- Frankel, E.N. Recent advances in lipid oxidation. J. Sci. Food. Agric. 1991, 54, 495. [Google Scholar] [CrossRef]
- Hernández, M.L.; Velázquez-Palmero, D.; Sicardo, M.D.; Fernández, J.E.; Diaz-Espejo, A.; Martínez-Rivas, J.M. Effect of a regulated deficit irrigation strategy in a hedgerow ‘Arbequina’ olive orchard on the mesocarp fatty acid composition and desatuase gene expression with respect to olive oil quality. Agric. Water Manag. 2018, 204, 100–106. [Google Scholar] [CrossRef]
- Jimenez-Lopez, C.; Carpena, M.; Lourenço-Lopes, C.; Gallardo-Gomez, M.; Lorenzo, J.M.; Barba, F.J.; Prieto, M.A.; Simal-Gandara, J. Bioactive Compounds and Quality of Extra Virgin Olive Oil. Foods 2020, 9, 1014. [Google Scholar] [CrossRef] [PubMed]
- Lozano-Castellón, J.; López-Yerena, A.; Domínguez-López, I.; Siscart-Serra, A.; Fraga, N.; Sámano, S.; López-Sabater, C.; Lamuela-Raventós, R.M.; Vallverdú-Queralt, A.; Pérez, M. Extra virgin olive oil: A comprehensive review of efforts to ensure its authenticity, traceability, and safety. Compr. Rev. Food Sci. Food Saf. 2022, 21, 2639–2664. [Google Scholar] [CrossRef] [PubMed]
- Ben Hmida, R.; Gargouri, B.; Chtourou, F.; Sevim, D.; Bouaziz, M. Fatty acid and triacyglycerid as markers of virgin olive oil from mediterranean region: Traceability and chemometric authentication. Eur. Food Res. Technol. 2022, 248, 1–16. [Google Scholar] [CrossRef]
- Psomiadou, E.; Tsimidou, M. Stability of virgin olive oil. 2. Photo-oxidation studies. J. Agric. Food Chem. 2002, 50, 722–727. [Google Scholar] [CrossRef] [PubMed]
- Tsimidou, M.Z.; Georgiou, A.; Koidis, A.; Boskou, D. Loss of stability of “veiled” (cloudy) virgin olive oils in storage. Food Chem. 2005, 93, 377–383. [Google Scholar] [CrossRef]
- Cicerale, S.; Conlan, X.A.; Sinclair, A.J.; Keast, R.S. Chemistry and health of olive oil phenolics. Crit. Rev. Food Sci. Nutr. 2008, 49, 218–236. [Google Scholar] [CrossRef] [PubMed]
- Ambra, R.; Lucchetti, S.; Pastore, G. A Review of the Effects of Olive Oil-Cooking on Phenolic Compounds. Molecules 2022, 27, 661. [Google Scholar] [CrossRef]
- Di Stefano, V.; Melilli, M.G. Effect of storage on quality parameters and phenolic content of Italian extra-virgin olive oils. Nat. Prod. Res. 2020, 34, 78–86. [Google Scholar] [CrossRef]
- Bitsani, E.; Agriopoulou, S.; Athanasopoulou, C. The Cultural, Nutritional and the Socio-Economic Value of Greek Messinian Olive Oil. In Strategic Innovative Marketing and Tourism. Springer Proceedings in Business and Economics; Kavoura, A., Kefallonitis, E., Giovanis, A., Eds.; Springer: Boston, MA, USA, 2019; pp. 307–315. [Google Scholar] [CrossRef]
- Jukic Špika, M.; Liber, Z.; Montemurro, C.; Miazzi, M.M.; Ljubenkov, I.; Soldo, B.; Žanetic, M.; Vitanovic, E.; Politeo, O.; Škevin, D. Quantitatively Unraveling Hierarchy of Factors Impacting Virgin Olive Oil Phenolic Profile and Oxi-dative Stability. Antioxidants 2022, 11, 594. [Google Scholar] [CrossRef]
- Karkoula, E.; Skantzari, A.; Melliou, E.; Magiatis, P. Direct measurement of oleocanthal and oleacein levels in olive oil by quantitative 1H NMR. Establishment of a new index for the characterization of extra virgin olive oils. J. Agric. Food Chem. 2012, 60, 11696–11703. [Google Scholar] [CrossRef]
- Parthasarathy, S.; Soundararajan, P.; Krishnan, N.; Karuppiah, K.M.; Devadasan, V.; Prabhu, D.; Rajamanikandan, S.; Velusamy, P.; Gopinath, S.C.; Raman, P. Detection of adulterants from common edible oils by GC–MS. Biomass Convers. Biorefin. 2022, 1–21. [Google Scholar] [CrossRef]
- García Martín, J.F. Potential of Near-Infrared Spectroscopy for the Determination of Olive Oil Quality. Sensors 2022, 22, 2831. [Google Scholar] [CrossRef]
- Vichi, S.; Pizzale, L.; Conte, L.S.; Buxaderas, S.; López-Tamames, E. Solid-phase microextraction in the analysis of virgin olive oil volatile fraction: Modifications induced by oxidation and suitable markers of oxidative status. J. Agric. Food Chem. 2003, 51, 6564–6571. [Google Scholar] [CrossRef] [PubMed]
- Morales, M.T.; Rios, J.J.; Aparicio, R. Changes in the volatile composition of virgin olive oil during oxidation: Flavors and off-flavors. J. Agric. Food Chem. 1997, 45, 2666–2673. [Google Scholar] [CrossRef]
- Pasias, I.N.; Raptopoulou, K.G.; Proestos, C. Analytical Chemistry and Foodomics: Determination of Authenticity and Adulteration of Extra Virgin Oil as Case Study. In Comprehensive Foodomics, 1st ed.; Cifuentes, A., Ed.; Elsevier: Athens, Greece, 2020; pp. 494–500. [Google Scholar]
- Moore, K.; Roberts, L.S. Measurement of lipid peroxidation. Radic. Res. 1998, 28, 659. [Google Scholar] [CrossRef]
- Warner, K.; Eskin, N.M. Methods to Assess Quality and Stability of Oils and Fat Containing Foods, 1st ed.; Warner, K., Eskin, N.A.M., Eds.; AOCS Press: Champaign, IL, USA, 1995. [Google Scholar]
- Allen, J.C.; Hamilton, R.J. Rancidity in Foods, 3rd ed.; Chapman & Hall: London, UK, 1994; pp. 1–22. [Google Scholar]
- Gray, I.J. Measurement of lipid oxidation. J. Am. Oil Chem. Soc. 1978, 55, 53. [Google Scholar] [CrossRef]
- Ueda, S.; Hayashi, T.; Namiki, M. Effect of ascorbic acid on lipid autoxidation in a model food system. Agric. Biol. Chem. 1986, 50, 1. [Google Scholar]
- Kiokias, S. In Vitro and In Vivo Antioxidant Properties of Natural Carotenoid Mixtures. Ph.D. Thesis, School of Food Biosciences, The University of Reading, Berkshire, UK, 2002. [Google Scholar]
- Tsoukalas, V.; Grosch, W. Analysis of food deterioration-Comparison of some photometric tests. J. Am. Oil Chem. Soc. 1977, 54, 490. [Google Scholar] [CrossRef]
- Hu, M.; McClements, J.; Decker, E.A. Impact of whey protein emulsifiers on the oxidative stability of salmon oil-in-water emulsions. J. Agric. Food Chem. 2003, 51, 1435. [Google Scholar] [CrossRef]
- Koidis, A.; Boskou, B. Virgin Olive Oil: Losses of Antioxidant Polar Phenolic Compounds due to Storage, Packaging, and Cul-inary Uses. In Processing and Impact on Active Components in Food; Preedy, V., Ed.; Elsevier: London, UK, 2014; pp. 259–266. [Google Scholar]
- Bendini, A.; Cerretani, L.; Vecchi, S.; Carrasco-Pancorbo, A.; Lercker, G. Protective effects of extra virgin olive oil phenolics on oxidative stability in the presence or absence of copper ions. J. Agric. Food Chem. 2006, 54, 4880–4887. [Google Scholar] [CrossRef]
- Logani, K.M.; Davis, R.E. Lipid oxidation: Biological effects and antioxidants. Lipids 1980, 12, 485. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Frankel, E.N.; Schwarz, K.; German, B. Effect of pH on antioxidant activity of α-tocopherol and trolox in oil-in-water emulsions. J. Agric. Food. Chem. 1996, 42, 2496. [Google Scholar] [CrossRef]
- IUPAC. Standard Methods of Analysis of Oils Fats and Derivatives, 7th ed.; Paquot, C., Hautfenne, H., Eds.; Blackwell Scientific Publications: Oxford, UK, 1987. [Google Scholar]
- Wanasundara, N.U.; Shahidi, F.; Jablonski, C.R. Comparison of standard and NMR methodologies for assessment of oxidative stability of soybean oils. Food Chem. 1995, 52, 249. [Google Scholar] [CrossRef]
- Wheatley, A.R. Some recent trends in the analytical chemistry of lipid peroxidation. Trends Anal. Chem. 2000, 19, 620. [Google Scholar] [CrossRef]
- White, P.J. Conjugated diene, anisidine value and carbonyl value analysis. In Methods to Assess Quality and Stability of Oil and Fat Containing Foods; Warner, K., Eskin, M., Eds.; AOCS Publisher: Champaign, IL, USA, 1995; p. 159. [Google Scholar]
- Kasuga, A.; Aoyagi, Y.; Sugahara, T.J. Antioxidant activity of fungus Suihs bovinus (L: Fr.) O. Kuntze. Food Sci. 1995, 60, 1113. [Google Scholar] [CrossRef]
- Rossel, J.B. Rancidity in Foods, 3rd ed.; Allen, J.C., Hamilton, R.J., Eds.; Blackie Academie & Professional: Glasgow, Scotland, 1996. [Google Scholar]
- List, G.R.; Evans, C.D.; Kwolek, W.F.; Warner, K.; Boundy, B.K.; Cowan, J.C. Oxidation and quality of soybean oil: A prelim-inary study of the anisidine test. J. Am. Oil Chem. Soc. 1974, 51, 17. [Google Scholar] [CrossRef]
- Osborn, H.T.; Akoh, C.C. Effect of emulsifier type, droplet size, and oil concentration on lipid oxidation in structured lipid-based oil-in-water emulsions. Food Chem. 2004, 84, 451. [Google Scholar] [CrossRef]
- Shaidi, F.; Wanasadura, U.N. Methods for measuring oxidative stability in fats and oils. In Food Lipid Chemistry, Nutrition and Biotechnology; Akoh, C.C., Min, B.D., Eds.; Marcel Dekker: New York, NY, USA, 1997. [Google Scholar]
- Coupland, J.N.; Zhu, Z.; Wan, H.; McClements, D.J.; Nawar, W.W.; Chinachoti, P. Droplet composition affects the rate of oxidation of emulsified ethyl linoleate. J. Am. Oil Chem. Soc. 1996, 73, 795. [Google Scholar] [CrossRef]
- Tarladgis, B.G.; Pearson, A.M.; Dugan, R.L. Chemistry of the 2-TBA test for determination of oxidative stability in foods. J. Sci. Food Agric. 1964, 15, 602. [Google Scholar] [CrossRef]
- Mcdonald, R.E.; Hultin, H.O. Some characteristics of the enzymic lipid peroxidation system in the microsomal fraction of flounder skeletal muscle. J. Food Sci. 1987, 52, 15. [Google Scholar] [CrossRef]
- Hawrysh, J.Z. Stability of conola oil. In Canola and Rapeseed Oil. Production, Chemistry, Nutrition, and Processing; Shahidi, F., Ed.; Van Nortland Reinhlod: New York, NY, USA, 1990; p. 99. [Google Scholar]
- Schieberle, P. Characteristion of Food: Emerging Methods; Gaonkar, A.G., Ed.; Elsevier: London, UK, 1995. [Google Scholar]
- Snyder, J.M.; Frankel, E.N.; Selke, E. Capillary gas chromatographic analysis of headspace volatiles from vegetable oils. J. Am. Oil Chem. Soc. 1985, 62, 1675. [Google Scholar] [CrossRef]
- Zabaras, D.; Wyllie, S.G. Rearrangement of p-menthane terpenes by Carboxen during HS-SPME. J. Sep. Sci. 2002, 25, 685. [Google Scholar] [CrossRef]
- Xu, S.; Li, H.; Dong, P.; Wang, M.; Chen, C.P.; Feng, S.; Fan, J. High-throughput profiling volatiles in edible oils by cooling assisted solid-phase microextraction technique for sensitive discrimination of edible oils adulteration. Anal. Chim. Acta 2022, 1221, 340159. [Google Scholar] [CrossRef]
- Jimenez, A.; Beltran, G.; Aguilera, M.P. Application of SPME to the analysis of volatile compounds in virgin olive oils. J. Agric. Food Chem. 1998, 46, 2744. [Google Scholar]
- Brunton, P.N.; Cronin, A.D.; Durkan, R. A comparison of SPME fibres for measurement of hexanal, and pentanal in cooked turkey. Food Chem. 2000, 68, 339. [Google Scholar] [CrossRef]
- Steenson, D.F.; Lee, J.H.; Min, B.D. SPME microextraction of volatile soybean oil and corn oil compounds. J. Food Sci. 2002, 67, 71. [Google Scholar] [CrossRef]
- Yang, X.; Reppard, T. Solid phase microextraction for flavors analysis. J. Agric. Food Chem. 1994, 41, 1925. [Google Scholar] [CrossRef]
- Casadei, E.; Valli, E.; Aparicio-Ruiz, R.; Ortiz-Romero, C.; García-González, D.L.; Vichi, S.; Quintanilla-Casas, B.; Tres, A.; Bendini, A.; Toschi, T.G. Peer inter-laboratory validation study of a harmonized SPME-GC-FID method for the analysis of selected volatile compounds in virgin olive oils. Food Control 2021, 123, 107823. [Google Scholar] [CrossRef]
- Aparicio-Ruiz, R.; Morales, M.T.; Aparicio, R. Does authenticity of virgin olive oil sensory quality require input from chemis-try? Eur. J. Lipid Sci. Technol. 2019, 121, 1900202. [Google Scholar] [CrossRef]
- Aparicio-Ruiz, R.; Romero, C.O.; Casadei, E.; García-González, D.L.; Servili, M.; Selvaggini, R.; Lacoste, F.; Escobessa, J.; Vichi, S.; Quintanilla-Casas, B.; et al. Collaborative peer validation of a harmonized SPME-GC-MS method for analysis of selected volatile compounds in virgin olive oils. Food Control 2022, 135, 108756. [Google Scholar] [CrossRef]
- Morales, M.T.; Przybylski, R. Olive oil oxidation. In Handbook of Olive Oil; Aparicio, R., Harwood, J., Eds.; Springer: Boston, MA, USA, 2013; pp. 479–522. [Google Scholar]
- Beltran, G.; Aguilera, M.P.; Gordon, M.H. Solid phase microextraction of volatile oxidation compounds in oil-in-water emul-sions. Food Chem. 2005, 92, 401–406. [Google Scholar] [CrossRef]
- Kiokias, S.; Gordon, M. Antioxidant properties of annatto carotenoids. Food Chem. 2003, 83, 523. [Google Scholar] [CrossRef]
- Zegarska, Z.; Amarowicz, R.; Karamac, M.; Rafalowski, R. Antioxidative effect of rosmary ethanolic extract on but-ter. Milchwissenschaft 1996, 51, 195. [Google Scholar]
- Karahadian, C.; Lindsay, R.C. Action of tocopherol-type compounds in directing reactions forming flavor compounds in au-tooxidizing fish oils. In Flavour Chemistry: Trends and Developments; Teranishi, R., Buttery, R.G., Shahidi, F., Eds.; ACS Sympo-sium Series: Madison; American Chemical Society: Washington, DC, USA, 1989; pp. 1302–1308. [Google Scholar]
- Akoh, C.C.; Min, B.D. Food Lipid Chemistry, Nutrition and Biotechnology; Marcel Dekker: New York, NY, USA, 1997. [Google Scholar]
- Slater, F.T. Overview of methods used for detecting lipid peroxidation. Meth. Enzymol. 1995, 300, 283. [Google Scholar]
- Oreopoulou, V. Extraction of Natural Antioxidants. In Extraction Optimization in Food Engineering; Tzia, C., Liadiakis, G., Eds.; Marcel Dekker: New York, NY, USA, 2003; pp. 329–346. [Google Scholar]
- Porter, L.W.; Black, D.E.; Drolet, M.A. Use of polyamide oxidative fluorescence test on lipid emulsions: Contrast in relative effectiveness of antioxidants in bulk versus dispersed systems. J. Agric. Food. Chem. 1980, 37, 61. [Google Scholar] [CrossRef]
- Alonso-Salces, R.M.; Gallo, B.; Collado, M.I.; Sasía-Arriba, A.; Viacava, G.E.; García-González, D.L.; Toschi, T.G.; Servili, M.; Berrueta, L.Á. 1H–NMR fingerprinting and supervised pattern recognition to evaluate the stability of virgin olive oil during storage. Food Control 2021, 123, 107831. [Google Scholar] [CrossRef]
- Facino, R.M.; Carini, M.; Aldini, G.; Colombo, L. Characterization of the intermediate products of lipid peroxidation in phos-phatidylcholine liposomes by fast-atom bombardment mass spectrometry and tandem mass spectrometry techniques. Rap. Commun. Mass Spectrom. 1996, 10, 1148. [Google Scholar] [CrossRef]
- Kiokias, S.; Varzakas, T.; Arvanitoyannis, I.; Labropoulos, A. Lipid oxidation and control of oxidation. In Advances in Food Biochemistry; Yildiz, F., Ed.; CRC Press, Taylor and Francis Group: New York, NY, USA, 2010; pp. 383–409. [Google Scholar]
- Üçüncüoğlu, D.; Küçük, V.A. A multivariate data approach for FTIR-ATR monitoring of virgin olive oils: Lipid structure during autoxidation. OCL 2019, 26, 6. [Google Scholar] [CrossRef]
- Rodrigues, N.; Dias, L.G.; Veloso, A.C.; Pereira, J.A.; Peres, A.M. Evaluation of extra-virgin olive oils shelf life using an electronic tongue—Chemometric approach. Eur. Food Res. Technol. 2017, 243, 597–607. [Google Scholar] [CrossRef]
- Gonçalves, T.R.; Rosa, L.N.; Torquato, A.S.; Da Silva, L.F.; Março, P.H.; Gomes, S.T.M.; Matsushita, M.; Valderrama, P. Assessment of brazilian monovarietal olive oil in two different package systems by using data fusion and chemometrics. Food Anal. Methods 2020, 13, 86–96. [Google Scholar] [CrossRef]
- Gargouri, B.; Zribi, A.; Bouaziz, M. Effect of containers on the quality of Chemlali olive oil during storage. J. Food Sci. Technol. 2015, 52, 1948–1959. [Google Scholar] [CrossRef] [PubMed]
- Raffo, A.; Bucci, R.; D’Aloise, A.; Pastore, G. Combined effects of reduced malaxation oxygen levels and storage time on extra-virgin olive oil volatiles investigated by a novel chemometric approach. Food Chem. 2015, 182, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Lukić, M.; Lukić, I.; Krapac, M.; Sladonja, B.; Piližota, V. Sterols and triterpene diols in olive oil as indicators of variety and degree of ripening. Food Chem. 2013, 136, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.F.; Anjos, C.A.R.; Cavalcanti, R.N.; dos Santos Celeghini, R.M. Evaluation of extra virgin olive oil stability by artificial neural network. Food Chem. 2015, 179, 35–43. [Google Scholar] [CrossRef]
- Arabameri, M.; Nazari, R.R.; Abdolshahi, A.; Abdollahzadeh, M.; Mirzamohammadi, S.; Shariatifar, N.; Barba, F.J.; Mousavi Khaneghah, A. Oxidative stability of virgin olive oil: Evaluation and prediction with an adaptive neuro-fuzzy inference system (ANFIS). J. Sci. Food Agric. 2019, 99, 5358–5367. [Google Scholar] [CrossRef]
- Uncu, O.; Ozen, B. Prediction of various chemical parameters of olive oils with Fourier transform infrared spectroscopy. LWT-Food Sci. Technol. 2015, 63, 978–984. [Google Scholar] [CrossRef]
- Esposto, S.; Taticchi, A.; Servili, M.; Urbani, S.; Sordini, B.; Veneziani, G.; Daidone, L.; Selvaggini, R. Overall quality evolution of extra virgin olive oil exposed to light for 10 months in different containers. Food Chem. 2021, 351, 129297. [Google Scholar] [CrossRef]
- Botosoa, E.P.; Chèné, C.; Karoui, R. Front face fluorescence spectroscopy combined with PLS-DA allows to monitor chemical changes of edible vegetable oils during storage at 60 °C. Eur. J. Lipid Sci. Technol. 2021, 123, 2000088. [Google Scholar] [CrossRef]
- Cosio, M.S.; Ballabio, D.; Benedetti, S.; Gigliotti, C. Evaluation of different storage conditions of extra virgin olive oils with an innovative recognition tool built by means of electronic nose and electronic tongue. Food Chem. 2007, 101, 485–491. [Google Scholar] [CrossRef]
- Sinelli, N.; Cosio, M.S.; Gigliotti, C.; Casiraghi, E. Preliminary study on application of mid infrared spectroscopy for the evaluation of the virgin olive oil “freshness”. Anal. Chim. Acta 2007, 598, 128–134. [Google Scholar] [CrossRef]
- Maggio, R.M.; Kaufman, T.S.; Del Carlo, M.; Cerretani, L.; Bendini, A.; Cichelli, A.; Compagnone, D. Monitoring of fatty acid composition in virgin olive oil by Fourier transformed infrared spectroscopy coupled with partial least squares. Food Chem. 2009, 114, 1549–1554. [Google Scholar] [CrossRef]
- Buratti, S.; Benedetti, S.; Cosio, M.S. An electronic nose to evaluate olive oil oxidation during storage. Ital. J. Food Sci. 2005, 17, 203–210. [Google Scholar]
- Mahesar, S.A.; Bendini, A.; Cerretani, L.; Bonoli-Carbognin, M.; Sherazi, S.T.H. Application of a spectroscopic method to estimate the olive oil oxidative status. Eur. J. Lipid Sci. Technol. 2010, 112, 1356–1362. [Google Scholar] [CrossRef]
- Sanmartin, C.; Venturi, F.; Sgherri, C.; Nari, A.; Macaluso, M.; Flamini, G.; Quartacci, M.F.; Taglieri, I.; Andrich, G.; Zinnai, A. The effects of packaging and storage temperature on the shelf-life of extra virgin olive oil. Heliyon 2018, 4, e0088. [Google Scholar] [CrossRef] [Green Version]
- Georgouli, K.; Del Rincon, J.M.; Koidis, A. Continuous statistical modelling for rapid detection of adulteration of extra virgin olive oil using mid infrared and Raman spectroscopic data. Food Chem. 2017, 217, 735–742. [Google Scholar] [CrossRef] [PubMed]
- Oueslati, I.; Djebali, H.M.; Mhamdi, R. Use of Phytosterols as a Tool for the Authenticity Assessment of Virgin Olive Oil: Protection of the Olive Oil Market. In Sterols: Types, Classification and Structure; Jimenez, S., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2020. [Google Scholar]
- González-Pereira, A.; Otero, P.; Fraga-Corral, M.; Garcia-Oliveira, P.; Carpena, M.; Prieto, M.A.; Simal-Gandara, J. State-of-the-Art of Analytical Techniques to Determine Food Fraud in Olive Oils. Foods 2021, 10, 484. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Xue, L.; Zhang, L.; Wang, X.; Qi, X.; Jiang, J.; Yu, L.; Wang, X.; Zhang, W.; Zhang, Q.; et al. Phytosterol contents of edible oils and their contributions to estimated phytosterol intake in the Chinese diet. Foods 2019, 8, 334. [Google Scholar] [CrossRef]
- Azadmard-Damirchi, S.; Torbati, M. Adulterations in some edible oils and fats and their detection methods. J. Food Qual. Hazards Control 2015, 2, 38–44. [Google Scholar]
- Pasias, I.N.; Theodorou, K.; Raptopoulou, K.G.; Evaggelaras, C.; Floros, G.; Ladavos, A.; Asimakopoulos, A.G.; Calokerinos, A.C.; Proestos, C. Rapid, low-cost spectrophotometric characterization of olive oil quality to meet newly implemented compliance requirements. Anal. Lett. 2022, 55, 281–291. [Google Scholar] [CrossRef]
- Cecchi, L.; Migliorini, M.; Mulinacci, N. Virgin olive oil volatile compounds: Composition, sensory characteristics, analytical approaches, quality control, and authentication. J. Agric. Food Chem. 2021, 69, 2013–2040. [Google Scholar] [CrossRef]
- Navratilova, K.; Hurkova, K.; Hrbek, V.; Uttl, L.; Tomaniova, M.; Valli, E.; Hajslova, J. Metabolic fingerprint-ing strategy: Investigation of markers for the detection of extra virgin olive oil adulteration with soft-deodorized olive oils. Food Control 2022, 134, 108649. [Google Scholar] [CrossRef]
- Cavanna, D.; Hurkova, K.; Džuman, Z.; Serani, A.; Serani, M.; Dall’Asta, C.; Tomaniova, M.; Hajslova, J. Suman, M. A non-targeted high-resolution mass spectrometry study for extra virgin olive oil adulteration with soft refined oils: Preliminary findings from two different laboratories. ACS Omega 2020, 5, 24169–24178. [Google Scholar] [CrossRef]
- Damiani, T.; Cavanna, D.; Serani, A.; Dall’Asta, C.; Suman, M. GC-IMS and FGC-Enose fingerprint as screening tools for revealing extra virgin olive oil blendingwith soft-refined olive oils: A feasibility study. Microchem. J. 2020, 159, 105374. [Google Scholar] [CrossRef]
- Drira, M.; Guclu, G.; Portolés, T.; Jabeur, H.; Kelebek, H.; Selli, S.; Bouaziz, M. Safe and fast fingerprint aroma detection in adulterated extra virgin olive oil using gas chromatography—Olfactometry-mass spectrometry com-bined with chemometrics. Food Anal. Methods 2021, 14, 2121–2135. [Google Scholar] [CrossRef]
- Violino, S.; Benincasa, C.; Taiti, C.; Ortenzi, L.; Pallottino, F.; Marone, E.; Mancuso, S.; Costa, C. AI-based hyper-spectral and VOCs assessment approach to identify adulterated extra virgin olive oil. Eur. Food Res. Technol. 2021, 247, 1013–1022. [Google Scholar] [CrossRef]
- Zhou, X.; Zhang, Q.; Chen, X.; Li, X.; Han, C. In-situ assessment of olive oil adulteration with soybean oil based on thermogravimetric-gas chromatography/mass spectrometry combined with chemometrics. Food Control 2021, 130, 108251. [Google Scholar] [CrossRef]
- Giuffrè, A.M.; Capocasale, M.; Macrì, R.; Caracciolo, M.; Zappia, C.; Poiana, M. Volatile profiles of extra virgin olive oil, olive pomace oil, soybean oil and palm oil in different heating conditions. LWT—Food Sci. Technol. 2020, 117, 108631–108637. [Google Scholar] [CrossRef]
- Van Durme, J.; Vandamme, J. Non-thermal plasma as preparative technique to evaluate olive oil adulteration. Food Chem. 2016, 208, 185–191. [Google Scholar] [CrossRef]
- Ozcan-Sinir, G. Detection of adulteration in extra virgin olive oil by selected ion flow tube mass spectrometry (SIFT-MS) and chemometrics. Food Control 2020, 118, 107433. [Google Scholar] [CrossRef]
- Quintanilla-Casas, B.; Strocchi, G.; Bustamante, J.; Torres-Cobos, B.; Guardiola, F.; Moreda, W.; Martínez-Rivas, J.M.; Valli, E.; Bendini, A.; Toschi, T.G.; et al. Large-scale evaluation of shotgun triacylglycerol profiling for the fast detection of olive oil adulteration. Food Control 2021, 123, 107851. [Google Scholar] [CrossRef]
- Green, H.S.; Li, X.; De Pra, M.; Lovejoy, K.S.; Steiner, F.; Acworth, I.N.; Wang, S.C. A rapid method for the detection of extra virgin olive oil adulteration using UHPLC-CAD profiling of triacylglycerols and PCA. Food Control 2020, 107, 106773. [Google Scholar] [CrossRef]
- Qian, Y.; Rudzińska, M.; Grygier, A.; Przybylski, R. Determination of triacylglycerols by HTGC-FID as a sensi-tive tool for the identification of rapeseed and olive oil adulteration. Molecules 2020, 25, 3881. [Google Scholar] [CrossRef]
- Zhang, Y.; Han, Y.; Hu, W.; Pan, Q.; Liu, Z.; Ling, G.; Shi, Q.; Weng, R. Diacylglycerols ions as novel mark-er indicators for the classification of edible oils using ultrahigh resolution mass spectrometry. Food Res. Int. 2021, 145, 110422. [Google Scholar] [CrossRef] [PubMed]
- Salah, W.A.; Nofal, M. Review of some adulteration detection techniques of edible oils. J. Sci. Food Agric. 2021, 101, 811–819. [Google Scholar] [CrossRef] [PubMed]
- Beneito-Cambra, M.; Moreno-González, D.; García-Reyes, J.F.; Bouza, M.; Gilbert-López, B.; Molina-Díaz, A. Direct analysis of olive oil and other vegetable oils by mass spectrometry: A review. TrAC-Trends Anal. Chem. 2020, 132, 116046. [Google Scholar] [CrossRef]
- Zaroual, H.; Chénè, C.; El Hadrami, E.M.; Karoui, R. Application of new emerging techniques in combination with classical methods for the determination of the quality and authenticity of olive oil: A review. Crit. Rev. Food Sci. Nutr. 2022, 62, 4526–4549. [Google Scholar] [CrossRef]
- Calvini, R.; Pigani, L. Toward the development of combined artificial sensing systems for food quality evaluation: A review on the application of data fusion of electronic noses, electronic tongues and electronic eyes. Sensors 2022, 22, 577. [Google Scholar] [CrossRef]
- Rifna, E.J.; Pandiselvam, R.; Kothakota, A.; Rao, K.S.; Dwivedi, M.; Kumar, M.; Thirumdas, R.; Ramesh, S.V. Advanced process analytical tools for identification of adulterants in edible oils—A review. Food Chem. 2022, 369, 130898. [Google Scholar] [CrossRef]
- Buratti, S.; Malegori, C.; Benedetti, S.; Oliveri, P.; Giovanelli, G. E-nose, e-tongue and e-eye for edible olive oil characterization and shelf life assessment: A powerful data fusion approach. Talanta 2018, 182, 131–141. [Google Scholar] [CrossRef]
- Harzalli, U.; Rodrigues, N.; Veloso, A.C.A.; Dias, L.G.; Pereira, J.A.; Oueslati, S.; Peres, A.M. A taste sensor device for unmasking admixing of rancid or winey-vinegary olive oil to extra virgin olive oil. Comput. Electron. Agric. 2018, 144, 222–231. [Google Scholar] [CrossRef]
- de Melo Milanez, K.D.T.; Pontes, M.J.C. Classification of extra virgin olive oil and verification of adulteration using digital images and discriminant analysis. Anal. Methods 2015, 7, 8839–8846. [Google Scholar] [CrossRef]
| Component | Concentration | |
|---|---|---|
| Fatty acids (%) | ||
| Myristic acid | C14:0 | 0.05 |
| Palmitic acid | C16:0 | 9.4–19.5 |
| Palmitoleic acid | C16:1 | 0.6–3.2 |
| Heptadecanoic acid | C17:0 | 0.07–0.13 |
| Heptadecenoic acid | C17:1 | 0.17–0.24 |
| Stearic acid | C18:0 | 1.4–3.0 |
| Oleic acid | C18:1 | 63.1–79.7 |
| Linoleic acid | C18:2 | 6.6–14.8 |
| α-Linolenic acid | C18:3 | 0.46–0.69 |
| Arachidic acid | C20:0 | 0.3–0.4 |
| Eicosenoic acid | C20:1 | 0.2–0.3 |
| Docosanoic acid | C22:0 | 0.09–0.12 |
| Lignoceric acid | C24:0 | 0.04–0.05 |
| MUFA | 65.2–80.8 | |
| PUFA | 7.0–15.5 | |
| Other lipids | ||
| Diacylglycerols (%) | 1–2.8 | |
| Monoacylglycerols (%) | 0.25 | |
| Total sterol content (mg/kg) | 1000–3040 | |
| Type of Olive Oil | Storage Conditions/Period | Findings | Analytical Instrumentation | Chemometric Method(s) | References |
|---|---|---|---|---|---|
| VOOs | 12 months | 1488–924 cm−1 band: important for the beginning of storage, symmetric and asymmetric stretching vibration of aldehydes, ketones, alcohols and hydroperoxides; 3008, 2924, 1745 cm−1: greater intensity at the end of the storage time; 3080–2790 cm−1 band: important at the end of storage | ATR-FTIR | PCA | [79] |
| EVOOs | 0, 3, 6 and 9 months, protected from light vs. exposed to light conditions | Decreased quality (reduction in shelf life), because of storage exposed to light | E-tongue | LDA | [80] |
| EVOOs | 1, 3, 6 and 12 months, room temperature, no light exposure | Glass bottles provide more protection to olive oil. The high content of unsaturated fatty acids was attributed to the high resistance of some varieties to oxidation; however, other varieties showed less resistance to oxidation since they had a lower amount of tocopherol and phenolic compounds | Data fusion: physicochemical parameters (peroxide value, iodine value, free acidity, refraction index), GC analysis of fatty acid methyl esters, UV-Vis and NIR spectroscopies | PCA | [81] |
| EVOOs | 35 days after extraction, and 1, 3 and 6 months | Long storage times influence the formation of octane, hexanal, C10 hydrocarbons and the level of volatiles of possible microbial origin | HS-SPME/GC-MS | ASCA | [82] |
| EVOO | 6 months, room temperature, under light exposure | The physicochemical and organoleptic criteria of EVOO are best retained in tin containers and dark glass bottles, compared to clear glass bottles and PE containers (degradation of the antioxidant contents, i.e., carotenes, chlorophylls and total phenols) | GC | HCA | [83] |
| VOOs | 12 months, at three different temperatures (variable room, refrigeration and freezing) | Variety identification and degree of ripening after storage can be achieved through sterols and triterpenediols | GC-FID | MANOVA, PCA, SLDA | [84] |
| EVOOs | 6 months under dark and light conditions, in different packaging materials | Package material and light exposure had influence on the stability of the oil | HPLC, UV-Vis | ANN | [85] |
| VOOs | 36 months at different temperatures (25 and 37 °C), in darkness | Diacylglycerols found to be good indicators of oil oxidative stability | GC-FID, GC-MS, HPLC, UV-Vis | ANFIS, PCA, MLR | [86] |
| OOs | Darkness, at refrigeration temperature | The oxidative stability of OOs is significantly dependent on palmitic, vanillic and cinnamic acids and hydroxytyrosol | FTIR, GC, HPLC, UV | PLS | [87] |
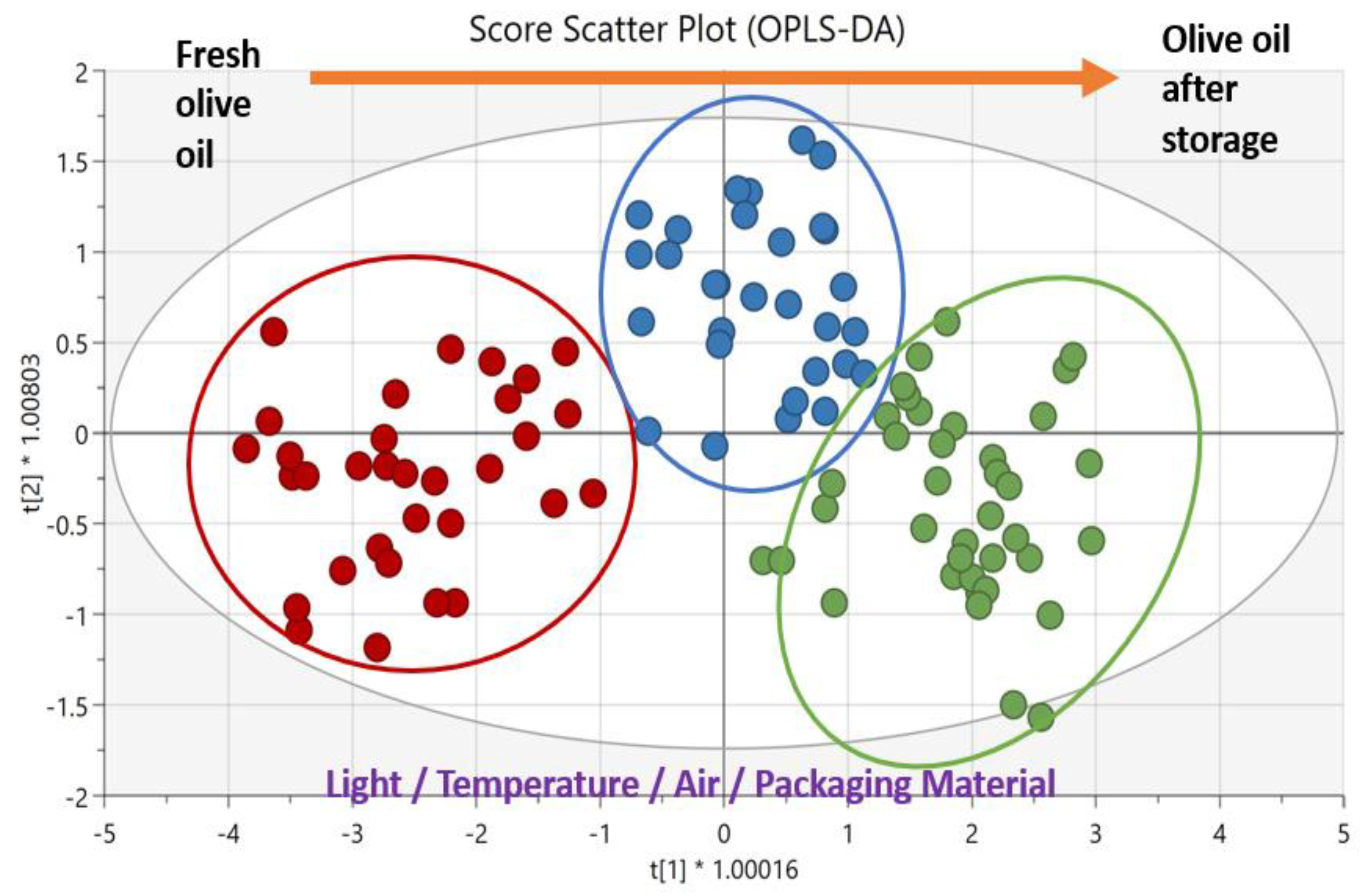
| EVOOs | 10 months exposed to light, in different packaging materials | MLP found to be the best material against oxidation, since EVOOs retained their initial quality within the regulatory limits since more antioxidants and fewer ‘rancid’ defects by related volatile compounds were identified | GC-FID, HPLC, HPLC-DAD-FLD, HS-SPME/GC-MS, sensory analysis | PCA, OPLS-DA | [88] |
| EVOOs and three other oils | Heating at 60 °C for up to 15 days | Different trends due to the different composition were obtained from rapeseed, sunflower, extra virgin olive and linseed oils | Front-face fluorescence spectroscopy | PLS-DA | [89] |
| VOOs | One year under dark conditions, one year under normal light, two years under dark | The electronic nose achieved the determination of the oxidation of the extra virgin olive oil as well as the descriptions of the different storage conditions | E-tongue | LDA | [90] |
| VOOs | In the light for 1 year and in the dark for 1 or 2 years | Fresh and oxidized oils were discriminated using FTIR PCA | ATR-MIR | PLS-DA, LDA, SIMCA | [91] |
| VOOs | 1 week and 2 months after production | Monitoring of fatty acid composition was optimum in the spectral range from 3033 to 700 cm−1 for oleic acid, linoleic acid, MUFA, PUFA and SFA | ATR-FTIR | PLS | [92] |
| OOs | 64 days for dark | Discrimination of olive oil samples based on aging time | E- nose | PCA | [93] |
| EVOO EVOP, EVOTP | 60 °C for 20 days | Monitoring of CD and CT was optimum in the spectral range of 2935–715 cm−1 | ATR-FTIR | PLS | [30] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tarapoulouzi, M.; Agriopoulou, S.; Koidis, A.; Proestos, C.; Enshasy, H.A.E.; Varzakas, T. Recent Advances in Analytical Methods for the Detection of Olive Oil Oxidation Status during Storage along with Chemometrics, Authenticity and Fraud Studies. Biomolecules 2022, 12, 1180. https://doi.org/10.3390/biom12091180
Tarapoulouzi M, Agriopoulou S, Koidis A, Proestos C, Enshasy HAE, Varzakas T. Recent Advances in Analytical Methods for the Detection of Olive Oil Oxidation Status during Storage along with Chemometrics, Authenticity and Fraud Studies. Biomolecules. 2022; 12(9):1180. https://doi.org/10.3390/biom12091180
Chicago/Turabian StyleTarapoulouzi, Maria, Sofia Agriopoulou, Anastasios Koidis, Charalampos Proestos, Hesham Ali El Enshasy, and Theodoros Varzakas. 2022. "Recent Advances in Analytical Methods for the Detection of Olive Oil Oxidation Status during Storage along with Chemometrics, Authenticity and Fraud Studies" Biomolecules 12, no. 9: 1180. https://doi.org/10.3390/biom12091180
APA StyleTarapoulouzi, M., Agriopoulou, S., Koidis, A., Proestos, C., Enshasy, H. A. E., & Varzakas, T. (2022). Recent Advances in Analytical Methods for the Detection of Olive Oil Oxidation Status during Storage along with Chemometrics, Authenticity and Fraud Studies. Biomolecules, 12(9), 1180. https://doi.org/10.3390/biom12091180











