Potential Effects of Natural H2S-Donors in Hypertension Management
Abstract
1. Introduction
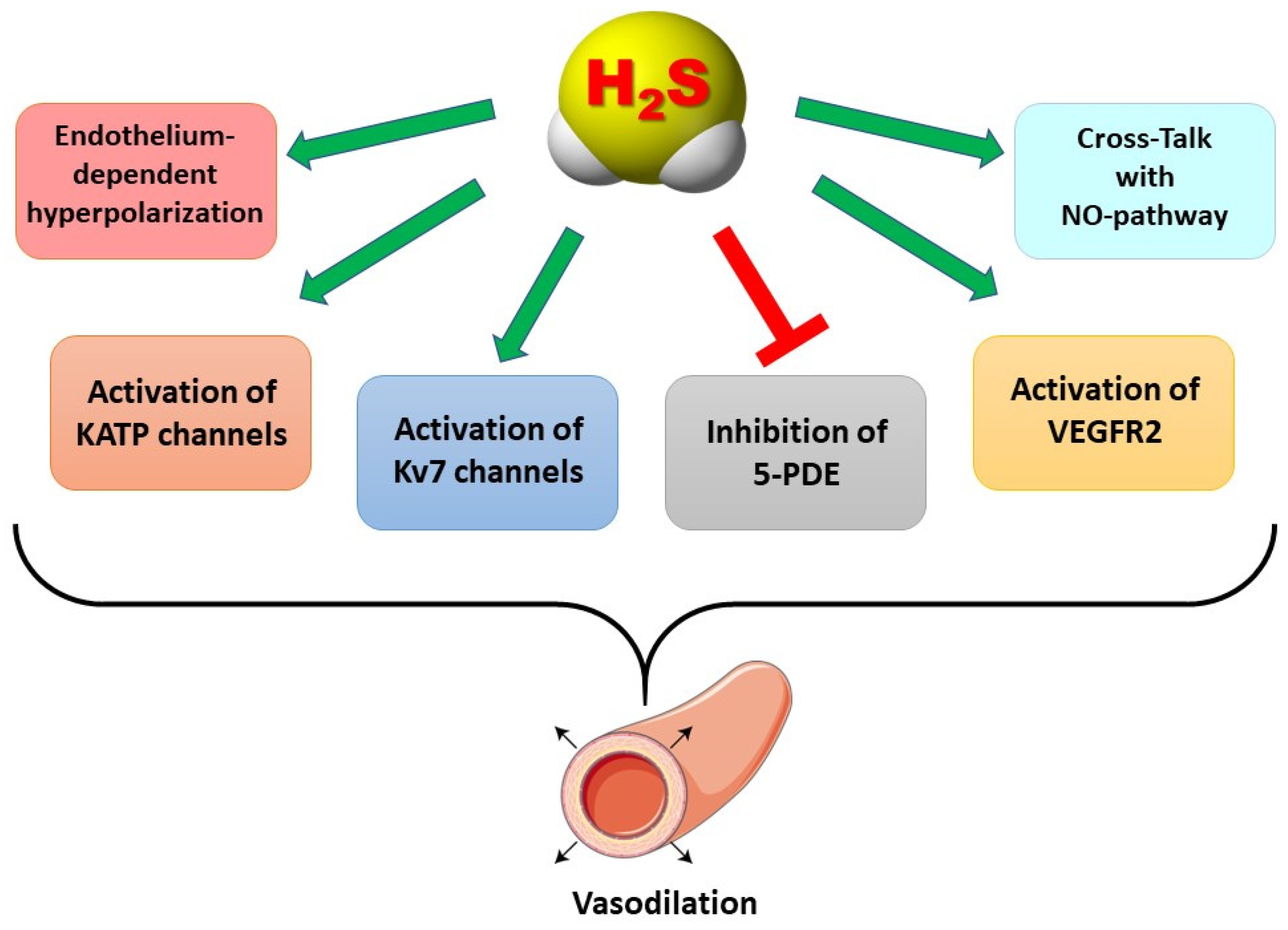
Mechanisms of Action Accounting for the Anti-Hypertensive Role of H2S
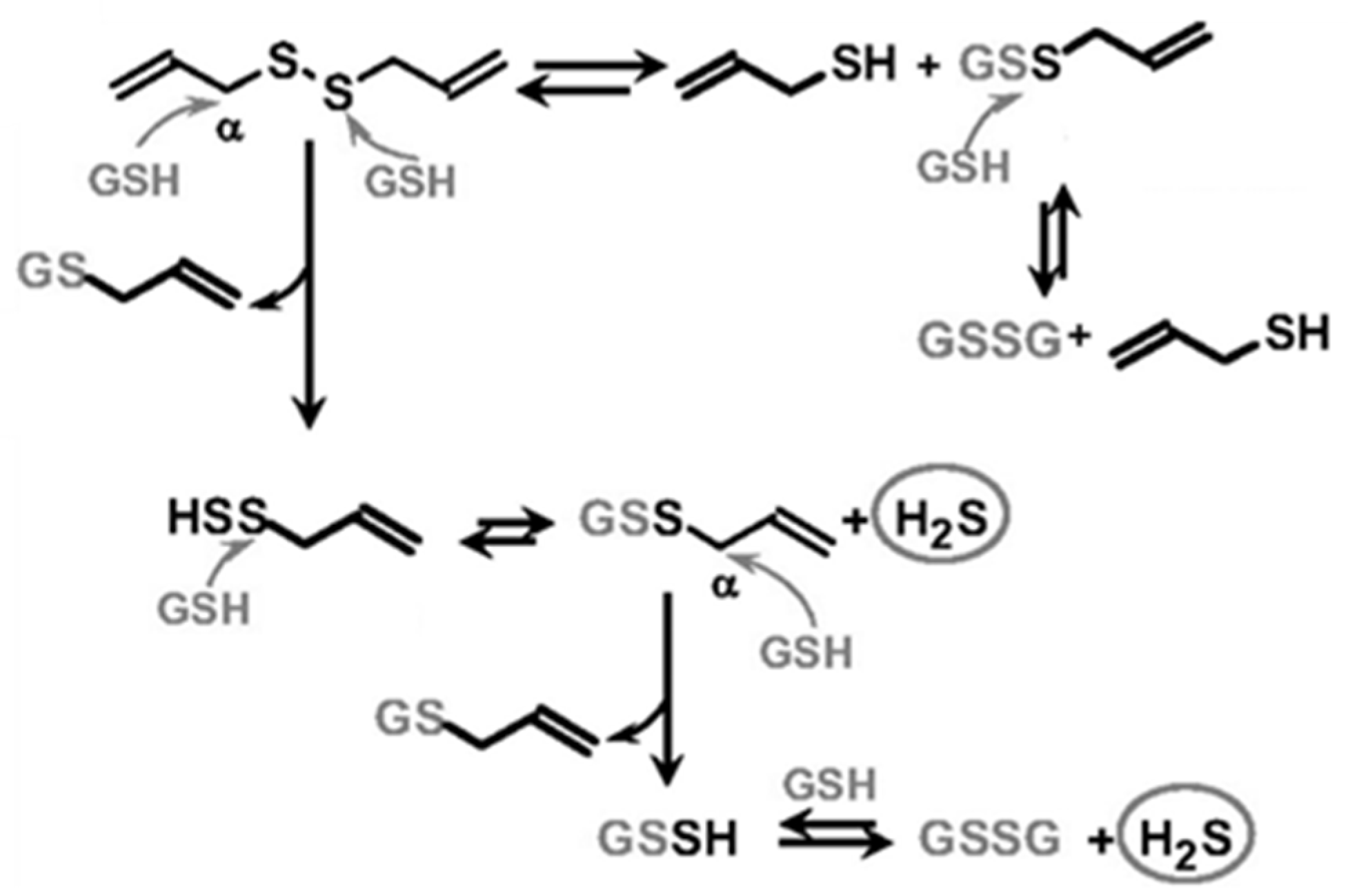
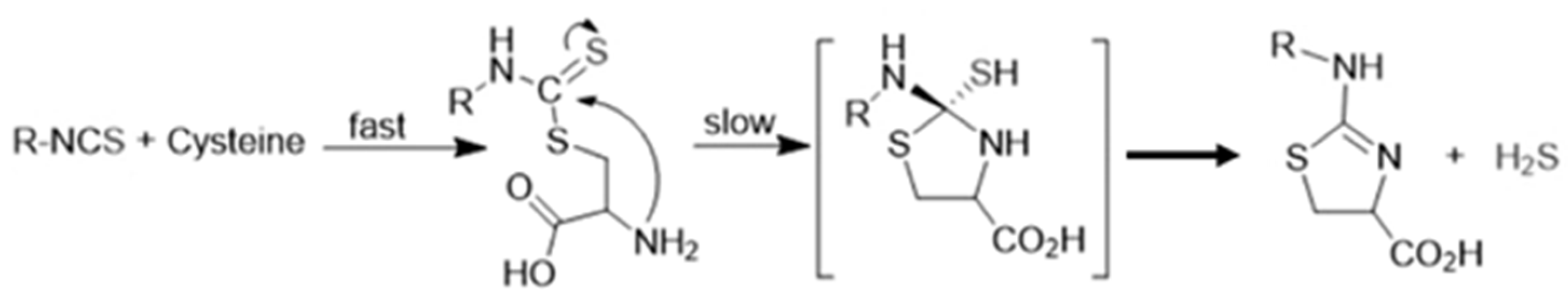
2. H2S Releasing Mechanism of Polysulfides and Isothiocyanates
3. Antihypertensive Effects of Garlic and Garlic Polysulfides in Preclinical Studies
4. Antihypertensive Effects of Isothiocyanates in Preclinical Studies
5. Antihypertensive Effects of Garlic in Humans
6. Antihypertensive Effects of Broccoli in Humans
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kimura, H. Hydrogen Sulfide (H2S) and Polysulfide (H2Sn) Signaling: The First 25 Years. Biomolecules 2021, 11, 896. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.H.; Lu, M.; Hu, L.F.; Wong, P.T.; Webb, G.D.; Bian, J.S. Hydrogen sulfide in the mammalian cardiovascular system. Antioxid. Redox Signal. 2012, 17, 141–185. [Google Scholar] [CrossRef] [PubMed]
- Martelli, A.; Testai, L.; Marino, A.; Breschi, M.C.; Da Settimo, F.; Calderone, V. Hydrogen sulphide: Biopharmacological roles in the cardiovascular system and pharmaceutical perspectives. Curr. Med. Chem. 2012, 19, 3325–3336. [Google Scholar] [CrossRef] [PubMed]
- Cacanyiova, S.; Berenyiova, A.; Kristek, F. The role of hydrogen sulphide in blood pressure regulation. Physiol. Res. 2016, 65, S273–S289. [Google Scholar] [CrossRef]
- Citi, V.; Piragine, E.; Testai, L.; Breschi, M.C.; Calderone, V.; Martelli, A. The Role of Hydrogen Sulfide and H2S-donors in Myocardial Protection Against Ischemia/Reperfusion Injury. Curr. Med. Chem. 2018, 25, 4380–4401. [Google Scholar] [CrossRef]
- Citi, V.; Martelli, A.; Gorica, E.; Brogi, S.; Testai, L.; Calderone, V. Role of hydrogen sulfide in endothelial dysfunction: Pathophysiology and therapeutic approaches. J. Adv. Res. 2021, 27, 99–113. [Google Scholar] [CrossRef]
- Testai, L.; Citi, V.; Martelli, A.; Brogi, S.; Calderone, V. Role of hydrogen sulfide in cardiovascular ageing. Pharmacol. Res. 2020, 160, 105125. [Google Scholar] [CrossRef]
- Ignarro, L.J.; Buga, G.M.; Wood, K.S.; Byrns, R.E.; Chaudhuri, G. Endothelium-derived relaxing factor produced and released from artery and vein is nitric oxide. Proc. Natl. Acad. Sci. USA 1987, 84, 9265–9269. [Google Scholar] [CrossRef]
- Mancardi, D.; Pla, A.F.; Moccia, F.; Tanzi, F.; Munaron, L. Old and new gasotransmitters in the cardiovascular system: Focus on the role of nitric oxide and hydrogen sulfide in endothelial cells and cardiomyocytes. Curr. Pharm. Biotechnol. 2011, 12, 1406–1415. [Google Scholar] [CrossRef]
- Testai, L.; D’Antongiovanni, V.; Piano, I.; Martelli, A.; Citi, V.; Duranti, E.; Virdis, A.; Blandizzi, C.; Gargini, C.; Breschi, M.C.; et al. Different patterns of H2S/NO activity and cross-talk in the control of the coronary vascular bed under normotensive or hypertensive conditions. Nitric Oxide 2015, 47, 25–33. [Google Scholar] [CrossRef]
- Nagpure, B.V.; Bian, J.S. Interaction of Hydrogen Sulfide with Nitric Oxide in the Cardiovascular System. Oxid. Med. Cell. Longev. 2016, 2016, 6904327. [Google Scholar] [CrossRef] [PubMed]
- Cirino, G.; Vellecco, V.; Bucci, M. Nitric oxide and hydrogen sulfide: The gasotransmitter paradigm of the vascular system. Br. J. Pharmacol. 2017, 174, 4021–4031. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Hu, Q.; Zhu, D. An Update on Hydrogen Sulfide and Nitric Oxide Interactions in the Cardiovascular System. Oxid. Med. Cell. Longev. 2018, 2018, 4579140. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Zhang, J.; Lu, Y.; Wang, R. The vasorelaxant effect of H2S as a novel endogenous gaseous K(ATP) channel opener. EMBO J. 2001, 20, 6008–6016. [Google Scholar] [CrossRef] [PubMed]
- Martelli, A.; Testai, L.; Breschi, M.C.; Lawson, K.; McKay, N.G.; Miceli, F.; Taglialatela, M.; Calderone, V. Vasorelaxation by hydrogen sulphide involves activation of Kv7 potassium channels. Pharmacol. Res. 2013, 70, 27–34. [Google Scholar] [CrossRef]
- Bucci, M.; Papapetropoulos, A.; Vellecco, V.; Zhou, Z.; Pyriochou, A.; Roussos, C.; Roviezzo, F.; Brancaleone, V.; Cirino, G. Hydrogen sulfide is an endogenous inhibitor of phosphodiesterase activity. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 1998–2004. [Google Scholar] [CrossRef]
- Zhu, M.L.; Zhao, F.R.; Zhu, T.T.; Wang, Q.Q.; Wu, Z.Q.; Song, P.; Xu, J.; Wan, G.R.; Yin, Y.L.; Li, P. The antihypertension effect of hydrogen sulfide (H2S) is induced by activating VEGFR2 signaling pathway. Life Sci. 2021, 267, 118831. [Google Scholar] [CrossRef]
- Chen, J.; Ding, X.; Chen, W.; Chen, S.; Guan, Q.; Wen, J.; Chen, Z. VEGFR2 in vascular smooth muscle cells mediates H2S-induced dilation of the rat cerebral basilar artery. Microvasc. Res. 2022, 141, 104309. [Google Scholar] [CrossRef]
- Chen, L.; Ingrid, S.; Ding, Y.G.; Liu, Y.; Qi, J.G.; Tang, C.S.; Du, J.B. Imbalance of endogenous homocysteine and hydrogen sulfide metabolic pathway in essential hypertensive children. Chin. Med. J. 2007, 120, 389–393. [Google Scholar] [CrossRef]
- Weber, G.J.; Pushpakumar, S.; Tyagi, S.C.; Sen, U. Homocysteine and hydrogen sulfide in epigenetic, metabolic and microbiota related renovascular hypertension. Pharmacol. Res. 2016, 113, 300–312. [Google Scholar] [CrossRef]
- Calderone, V.; Martelli, A.; Testai, L.; Citi, V.; Breschi, M.C. Using hydrogen sulfide to design and develop drugs. Expert Opin. Drug Discov. 2016, 11, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Bibli, S.I.; Hu, J.; Looso, M.; Weigert, A.; Ratiu, C.; Wittig, J.; Drekolia, M.K.; Tombor, L.; Randriamboavonjy, V.; Leisegang, M.S.; et al. Mapping the Endothelial Cell S-Sulfhydrome Highlights the Crucial Role of Integrin Sulfhydration in Vascular Function. Circulation 2021, 143, 935–948. [Google Scholar] [CrossRef] [PubMed]
- Calderone, V. Large-conductance, Ca2+-activated K+ channels: Function, pharmacology and drugs. Curr. Med. Chem. 2002, 9, 1385–1395. [Google Scholar] [CrossRef] [PubMed]
- Cecchetti, V.; Calderone, V.; Tabarrini, O.; Sabatini, S.; Filipponi, E.; Testai, L.; Spogli, R.; Martinotti, E.; Fravolini, A. Highly potent 1,4-benzothiazine derivatives as K(ATP)-channel openers. J. Med. Chem. 2003, 46, 3670–3679. [Google Scholar] [CrossRef]
- Calderone, V.; Spogli, R.; Martelli, A.; Manfroni, G.; Testai, L.; Sabatini, S.; Tabarrini, O.; Cecchetti, V. Novel 1,4-benzothiazine derivatives as large conductance Ca2+-activated potassium channel openers. J. Med. Chem. 2008, 51, 5085–5092. [Google Scholar] [CrossRef]
- Calderone, V.; Testai, L.; Martelli, A.; Rapposelli, S.; Digiacomo, M.; Balsamo, A.; Breschi, M.C. Anti-ischemic properties of a new spiro-cyclic benzopyran activator of the cardiac mito-KATP channel. Biochem. Pharmacol. 2010, 79, 39–47. [Google Scholar] [CrossRef]
- Martelli, A.; Manfroni, G.; Sabbatini, P.; Barreca, M.L.; Testai, L.; Novelli, M.; Sabatini, S.; Massari, S.; Tabarrini, O.; Masiello, P.; et al. 1,4-Benzothiazine ATP-sensitive potassium channel openers: Modifications at the C-2 and C-6 positions. J. Med. Chem. 2013, 56, 4718–4728. [Google Scholar] [CrossRef] [PubMed]
- Tang, G.; Wu, L.; Liang, W.; Wang, R. Direct stimulation of K(ATP) channels by exogenous and endogenous hydrogen sulfide in vascular smooth muscle cells. Mol. Pharmacol. 2005, 68, 1757–1764. [Google Scholar] [CrossRef]
- Liang, G.H.; Adebiyi, A.; Leo, M.D.; McNally, E.M.; Leffler, C.W.; Jaggar, J.H. Hydrogen sulfide dilates cerebral arterioles by activating smooth muscle cell plasma membrane KATP channels. Am. J. Physiol. Heart Circ. Physiol. 2011, 300, H2088–H2095. [Google Scholar] [CrossRef]
- Siebert, N.; Cantre, D.; Eipel, C.; Vollmar, B. H2S contributes to the hepatic arterial buffer response and mediates vasorelaxation of the hepatic artery via activation of K(ATP) channels. Am. J. Physiol. Gastrointest Liver Physiol. 2008, 295, G1266–G1273. [Google Scholar] [CrossRef]
- Schleifenbaum, J.; Kohn, C.; Voblova, N.; Dubrovska, G.; Zavarirskaya, O.; Gloe, T.; Crean, C.S.; Luft, F.C.; Huang, Y.; Schubert, R.; et al. Systemic peripheral artery relaxation by KCNQ channel openers and hydrogen sulfide. J. Hypertens. 2010, 28, 1875–1882. [Google Scholar] [CrossRef] [PubMed]
- Martelli, A.; Citi, V.; Calderone, V. Vascular Effects of H2S-Donors: Fluorimetric Detection of H2S Generation and Ion Channel Activation in Human Aortic Smooth Muscle Cells. Methods Mol. Biol. 2019, 2007, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Martelli, A.; Testai, L.; Citi, V.; Marino, A.; Bellagambi, F.G.; Ghimenti, S.; Breschi, M.C.; Calderone, V. Pharmacological characterization of the vascular effects of aryl isothiocyanates: Is hydrogen sulfide the real player? Vascul. Pharmacol. 2014, 60, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Citi, V.; Martelli, A.; Bucci, M.; Piragine, E.; Testai, L.; Vellecco, V.; Cirino, G.; Calderone, V. Searching for novel hydrogen sulfide donors: The vascular effects of two thiourea derivatives. Pharmacol. Res. 2020, 159, 105039. [Google Scholar] [CrossRef] [PubMed]
- Hedegaard, E.R.; Nielsen, B.D.; Kun, A.; Hughes, A.D.; Kroigaard, C.; Mogensen, S.; Matchkov, V.V.; Frobert, O.; Simonsen, U. KV 7 channels are involved in hypoxia-induced vasodilatation of porcine coronary arteries. Br. J. Pharmacol. 2014, 171, 69–82. [Google Scholar] [CrossRef]
- Abramavicius, S.; Petersen, A.G.; Renaltan, N.S.; Prat-Duran, J.; Torregrossa, R.; Stankevicius, E.; Whiteman, M.; Simonsen, U. GYY4137 and Sodium Hydrogen Sulfide Relaxations Are Inhibited by L-Cysteine and KV7 Channel Blockers in Rat Small Mesenteric Arteries. Front Pharmacol. 2021, 12, 613989. [Google Scholar] [CrossRef]
- Sun, Y.; Huang, Y.; Yu, W.; Chen, S.; Yao, Q.; Zhang, C.; Bu, D.; Tang, C.; Du, J.; Jin, H. Sulfhydration-associated phosphodiesterase 5A dimerization mediates vasorelaxant effect of hydrogen sulfide. Oncotarget 2017, 8, 31888–31900. [Google Scholar] [CrossRef]
- Tang, G.; Yang, G.; Jiang, B.; Ju, Y.; Wu, L.; Wang, R. H2S is an endothelium-derived hyperpolarizing factor. Antioxid. Redox Signal. 2013, 19, 1634–1646. [Google Scholar] [CrossRef]
- Dux, M.; Will, C.; Vogler, B.; Filipovic, M.R.; Messlinger, K. Meningeal blood flow is controlled by H2S-NO crosstalk activating a HNO-TRPA1-CGRP signalling pathway. Br. J. Pharmacol. 2016, 173, 431–445. [Google Scholar] [CrossRef]
- Zhou, Z.; Martin, E.; Sharina, I.; Esposito, I.; Szabo, C.; Bucci, M.; Cirino, G.; Papapetropoulos, A. Regulation of soluble guanylyl cyclase redox state by hydrogen sulfide. Pharmacol. Res. 2016, 111, 556–562. [Google Scholar] [CrossRef]
- Corvino, A.; Frecentese, F.; Magli, E.; Perissutti, E.; Santagada, V.; Scognamiglio, A.; Caliendo, G.; Fiorino, F.; Severino, B. Trends in H2S-Donors Chemistry and Their Effects in Cardiovascular Diseases. Antioxidants 2021, 10, 429. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Pacheco, A.; Xian, M. Medicinal Chemistry: Insights into the Development of Novel H2S Donors. Handb. Exp. Pharmacol. 2015, 230, 365–388. [Google Scholar] [CrossRef] [PubMed]
- Citi, V.; Corvino, A.; Fiorino, F.; Frecentese, F.; Magli, E.; Perissutti, E.; Santagada, V.; Brogi, S.; Flori, L.; Gorica, E.; et al. Structure-activity relationships study of isothiocyanates for H2S releasing properties: 3-Pyridyl-isothiocyanate as a new promising cardioprotective agent. J. Adv. Res. 2021, 27, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Severino, B.; Corvino, A.; Fiorino, F.; Luciano, P.; Frecentese, F.; Magli, E.; Saccone, I.; Di Vaio, P.; Citi, V.; Calderone, V.; et al. 1,2,4-Thiadiazolidin-3,5-diones as novel hydrogen sulfide donors. Eur. J. Med. Chem. 2018, 143, 1677–1686. [Google Scholar] [CrossRef] [PubMed]
- Corvino, A.; Citi, V.; Fiorino, F.; Frecentese, F.; Magli, E.; Perissutti, E.; Santagada, V.; Calderone, V.; Martelli, A.; Gorica, E.; et al. H2S donating corticosteroids: Design, synthesis and biological evaluation in a murine model of asthma. J. Adv. Res. 2022, 35, 267–277. [Google Scholar] [CrossRef]
- Brancaleone, V.; Esposito, I.; Gargiulo, A.; Vellecco, V.; Asimakopoulou, A.; Citi, V.; Calderone, V.; Gobbetti, T.; Perretti, M.; Papapetropoulos, A.; et al. D-Penicillamine modulates hydrogen sulfide (H2S) pathway through selective inhibition of cystathionine-gamma-lyase. Br. J. Pharmacol. 2016, 173, 1556–1565. [Google Scholar] [CrossRef]
- Mitidieri, E.; Tramontano, T.; Gurgone, D.; Citi, V.; Calderone, V.; Brancaleone, V.; Katsouda, A.; Nagahara, N.; Papapetropoulos, A.; Cirino, G.; et al. Mercaptopyruvate acts as endogenous vasodilator independently of 3-mercaptopyruvate sulfurtransferase activity. Nitric Oxide 2018, 75, 53–59. [Google Scholar] [CrossRef]
- Barresi, E.; Nesi, G.; Citi, V.; Piragine, E.; Piano, I.; Taliani, S.; Da Settimo, F.; Rapposelli, S.; Testai, L.; Breschi, M.C.; et al. Iminothioethers as Hydrogen Sulfide Donors: From the Gasotransmitter Release to the Vascular Effects. J. Med. Chem. 2017, 60, 7512–7523. [Google Scholar] [CrossRef]
- Martelli, A.; Testai, L.; Citi, V.; Marino, A.; Pugliesi, I.; Barresi, E.; Nesi, G.; Rapposelli, S.; Taliani, S.; Da Settimo, F.; et al. Arylthioamides as H2S Donors: L-Cysteine-Activated Releasing Properties and Vascular Effects in Vitro and in Vivo. ACS Med. Chem. Lett. 2013, 4, 904–908. [Google Scholar] [CrossRef]
- Benavides, G.A.; Squadrito, G.L.; Mills, R.W.; Patel, H.D.; Isbell, T.S.; Patel, R.P.; Darley-Usmar, V.M.; Doeller, J.E.; Kraus, D.W. Hydrogen sulfide mediates the vasoactivity of garlic. Proc. Natl. Acad. Sci. USA 2007, 104, 17977–17982. [Google Scholar] [CrossRef]
- Martelli, A.; Citi, V.; Testai, L.; Brogi, S.; Calderone, V. Organic Isothiocyanates as Hydrogen Sulfide Donors. Antioxid. Redox Signal. 2020, 32, 110–144. [Google Scholar] [CrossRef] [PubMed]
- Citi, V.; Martelli, A.; Testai, L.; Marino, A.; Breschi, M.C.; Calderone, V. Hydrogen sulfide releasing capacity of natural isothiocyanates: Is it a reliable explanation for the multiple biological effects of Brassicaceae? Planta Med. 2014, 80, 610–613. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, Y.; Liu, X.; Lin, Y.; Zheng, X.; Lu, Y. Hydrogen Sulfide (H2S) Releasing Capacity of Isothiocyanates from Moringa oleifera Lam. Molecules 2018, 23, 2809. [Google Scholar] [CrossRef]
- Lucarini, E.; Micheli, L.; Trallori, E.; Citi, V.; Martelli, A.; Testai, L.; De Nicola, G.R.; Iori, R.; Calderone, V.; Ghelardini, C.; et al. Effect of glucoraphanin and sulforaphane against chemotherapy-induced neuropathic pain: Kv7 potassium channels modulation by H2 S release in vivo. Phytother. Res. 2018, 32, 2226–2234. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Yang, X.; Lu, Y.; Liang, D.; Huang, D. Isothiocyanates as H2S Donors Triggered by Cysteine: Reaction Mechanism and Structure and Activity Relationship. Org. Lett. 2019, 21, 5977–5980. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, A.M.; Darabi, R.; Akbarloo, N. Investigation of antihypertensive mechanism of garlic in 2K1C hypertensive rat. J. Ethnopharmacol. 2003, 86, 219–224. [Google Scholar] [CrossRef]
- Laggner, H.; Hermann, M.; Esterbauer, H.; Muellner, M.K.; Exner, M.; Gmeiner, B.M.; Kapiotis, S. The novel gaseous vasorelaxant hydrogen sulfide inhibits angiotensin-converting enzyme activity of endothelial cells. J. Hypertens. 2007, 25, 2100–2104. [Google Scholar] [CrossRef]
- Nwokocha, C.R.; Ozolua, R.I.; Owu, D.U.; Nwokocha, M.I.; Ugwu, A.C. Antihypertensive properties of Allium sativum (garlic) on normotensive and two kidney one clip hypertensive rats. Niger. J. Physiol. Sci. 2011, 26, 213–218. [Google Scholar]
- Han, C.H.; Liu, J.C.; Chen, K.H.; Lin, Y.S.; Chen, C.T.; Fan, C.T.; Lee, H.L.; Liu, D.Z.; Hou, W.C. Antihypertensive activities of processed garlic on spontaneously hypertensive rats and hypertensive humans. Bot. Stud. 2011, 52, 277–283. [Google Scholar]
- Harauma, A.; Moriguchi, T. Aged garlic extract improves blood pressure in spontaneously hypertensive rats more safely than raw garlic. J. Nutr. 2006, 136, 769S–773S. [Google Scholar] [CrossRef]
- Chen, C.Y.; Tsai, T.Y.; Chen, B.H. Effects of Black Garlic Extract and Nanoemulsion on the Deoxy Corticosterone Acetate-Salt Induced Hypertension and Its Associated Mild Cognitive Impairment in Rats. Antioxidants 2021, 10, 1611. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.N.; Hou, C.Y.; Chang-Chien, G.P.; Lin, S.; Tain, Y.L. Maternal Garlic Oil Supplementation Prevents High-Fat Diet-Induced Hypertension in Adult Rat Offspring: Implications of H2S-Generating Pathway in the Gut and Kidneys. Mol. Nutr. Food Res. 2021, 65, e2001116. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M.Z.; Hussain, M.E.; Fahim, M. Endothelium mediated vasorelaxant response of garlic in isolated rat aorta: Role of nitric oxide. J. Ethnopharmacol. 2004, 90, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Wu, H.; Wong, M.W.; Huang, D. Diallyl Trisulfide Is a Fast H2S Donor, but Diallyl Disulfide Is a Slow One: The Reaction Pathways and Intermediates of Glutathione with Polysulfides. Org. Lett. 2015, 17, 4196–4199. [Google Scholar] [CrossRef] [PubMed]
- Jeremic, J.N.; Jakovljevic, V.L.; Zivkovic, V.I.; Srejovic, I.M.; Bradic, J.V.; Milosavljevic, I.M.; Mitrovic, S.L.; Jovicic, N.U.; Bolevich, S.B.; Svistunov, A.A.; et al. Garlic Derived Diallyl Trisulfide in Experimental Metabolic Syndrome: Metabolic Effects and Cardioprotective Role. Int. J. Mol. Sci 2020, 21, 9100. [Google Scholar] [CrossRef]
- Yang, Q.; He, G.W. Imbalance of Homocysteine and H2S: Significance, Mechanisms, and Therapeutic Promise in Vascular Injury. Oxid Med. Cell. Longev. 2019, 2019, 7629673. [Google Scholar] [CrossRef]
- Cui, T.; Liu, W.; Chen, S.; Yu, C.; Li, Y.; Zhang, J.Y. Antihypertensive effects of allicin on spontaneously hypertensive rats via vasorelaxation and hydrogen sulfide mechanisms. Biomed. Pharmacother. 2020, 128, 110240. [Google Scholar] [CrossRef]
- Cortese-Krott, M.M.; Kuhnle, G.G.; Dyson, A.; Fernandez, B.O.; Grman, M.; DuMond, J.F.; Barrow, M.P.; McLeod, G.; Nakagawa, H.; Ondrias, K.; et al. Key bioactive reaction products of the NO/H2S interaction are S/N-hybrid species, polysulfides, and nitroxyl. Proc. Natl. Acad. Sci. USA 2015, 112, E4651–E4660. [Google Scholar] [CrossRef]
- Dubey, H.; Singh, A.; Patole, A.M.; Tenpe, C.R. Antihypertensive effect of allicin in dexamethasone-induced hypertensive rats. Integr. Med. Res. 2017, 6, 60–65. [Google Scholar] [CrossRef]
- Elkayam, A.; Mirelman, D.; Peleg, E.; Wilchek, M.; Miron, T.; Rabinkov, A.; Sadetzki, S.; Rosenthal, T. The effects of allicin and enalapril in fructose-induced hyperinsulinemic hyperlipidemic hypertensive rats. Am. J. Hypertens. 2001, 14, 377–381. [Google Scholar] [CrossRef]
- Garcia-Trejo, E.M.; Arellano-Buendia, A.S.; Arguello-Garcia, R.; Loredo-Mendoza, M.L.; Garcia-Arroyo, F.E.; Arellano-Mendoza, M.G.; Castillo-Hernandez, M.C.; Guevara-Balcazar, G.; Tapia, E.; Sanchez-Lozada, L.G.; et al. Effects of Allicin on Hypertension and Cardiac Function in Chronic Kidney Disease. Oxid. Med. Cell. Longev. 2016, 2016, 3850402. [Google Scholar] [CrossRef] [PubMed]
- Martelli, A.; Piragine, E.; Citi, V.; Testai, L.; Pagnotta, E.; Ugolini, L.; Lazzeri, L.; Di Cesare Mannelli, L.; Manzo, O.L.; Bucci, M.; et al. Erucin exhibits vasorelaxing effects and antihypertensive activity by H2 S-releasing properties. Br. J. Pharmacol. 2020, 177, 824–835. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.K.; Kwan, T.K.; Kwan, C.Y.; Sorger, G.J. Effects of papaya seed extract and benzyl isothiocyanate on vascular contraction. Life Sci. 2002, 71, 497–507. [Google Scholar] [CrossRef]
- Salma, U.; Khan, T.; Shah, A.J. Antihypertensive effect of the methanolic extract from Eruca sativa Mill., (Brassicaceae) in rats: Muscarinic receptor-linked vasorelaxant and cardiotonic effects. J. Ethnopharmacol. 2018, 224, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Martelli, A.; Piragine, E.; Gorica, E.; Citi, V.; Testai, L.; Pagnotta, E.; Lazzeri, L.; Pecchioni, N.; Ciccone, V.; Montanaro, R.; et al. The H2S-Donor Erucin Exhibits Protective Effects against Vascular Inflammation in Human Endothelial and Smooth Muscle Cells. Antioxidants 2021, 10, 961. [Google Scholar] [CrossRef] [PubMed]
- Lin, F.; Huang, X.; Xing, F.; Xu, L.; Zhang, W.; Chen, Z.; Ke, X.; Song, Y.; Zeng, Z. Semen Brassicae reduces thoracic aortic remodeling, inflammation, and oxidative damage in spontaneously hypertensive rats. Biomed. Pharmacother. 2020, 129, 110400. [Google Scholar] [CrossRef]
- Aekthammarat, D.; Pannangpetch, P.; Tangsucharit, P. Moringa oleifera leaf extract lowers high blood pressure by alleviating vascular dysfunction and decreasing oxidative stress in L-NAME hypertensive rats. Phytomedicine 2019, 54, 9–16. [Google Scholar] [CrossRef]
- Ried, K. Garlic lowers blood pressure in hypertensive subjects, improves arterial stiffness and gut microbiota: A review and meta-analysis. Exp. Ther. Med. 2020, 19, 1472–1478. [Google Scholar] [CrossRef]
- Ried, K. Garlic Lowers Blood Pressure in Hypertensive Individuals, Regulates Serum Cholesterol, and Stimulates Immunity: An Updated Meta-analysis and Review. J. Nutr. 2016, 146, 389S–396S. [Google Scholar] [CrossRef]
- Ried, K.; Frank, O.R.; Stocks, N.P. Aged garlic extract lowers blood pressure in patients with treated but uncontrolled hypertension: A randomised controlled trial. Maturitas 2010, 67, 144–150. [Google Scholar] [CrossRef]
- Ried, K.; Frank, O.R.; Stocks, N.P. Aged garlic extract reduces blood pressure in hypertensives: A dose-response trial. Eur. J. Clin. Nutr. 2013, 67, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, F.; Capasso, R.; Izzo, A.A. Garlic (Allium sativum L.): Adverse effects and drug interactions in humans. Mol. Nutr. Food Res. 2007, 51, 1386–1397. [Google Scholar] [CrossRef] [PubMed]
- Kravchuk, O.M.; Goshovska, Y.V.; Korkach, Y.P.; Sagach, V.F. Garlic supplement lowers blood pressure in 40-60 years old hypertensive individuals, regulates oxidative stress, plasma cholesterol and protrombin index. J. Cardiovasc. Med. Cardiol. 2021, 8, 41–47. [Google Scholar] [CrossRef]
- Ried, K.; Travica, N.; Sali, A. The Effect of Kyolic Aged Garlic Extract on Gut Microbiota, Inflammation, and Cardiovascular Markers in Hypertensives: The GarGIC Trial. Front. Nutr. 2018, 5, 122. [Google Scholar] [CrossRef] [PubMed]
- Soleimani, D.; Parisa Moosavian, S.; Zolfaghari, H.; Paknahad, Z. Effect of garlic powder supplementation on blood pressure and hs-C-reactive protein among nonalcoholic fatty liver disease patients: A randomized, double-blind, placebo-controlled trial. Food Sci. Nutr. 2021, 9, 3556–3562. [Google Scholar] [CrossRef]
- Ashraf, R.; Khan, R.A.; Ashraf, I.; Qureshi, A.A. Effects of Allium sativum (garlic) on systolic and diastolic blood pressure in patients with essential hypertension. Pak. J. Pharm. Sci. 2013, 26, 859–863. [Google Scholar]
- Auer, W.; Eiber, A.; Hertkorn, E.; Hoehfeld, E.; Koehrle, U.; Lorenz, A.; Mader, F.; Merx, W.; Otto, G.; Schmid-Otto, B.; et al. Hypertension and hyperlipidaemia: Garlic helps in mild cases. Br. J. Clin. Pract. Suppl. 1990, 69, 3–6. [Google Scholar]
- Holzgartner, H.; Schmidt, U.; Kuhn, U. Comparison of the efficacy and tolerance of a garlic preparation vs. bezafibrate. Arzneimittelforschung 1992, 42, 1473–1477. [Google Scholar]
- Kandziora, J. Blood pressure and lipid reducing effect of a garlic supplement in combination with a diuretic. Arztl. Forsch. 1988, 35, 3–8. [Google Scholar]
- Nakasone, Y.; Nakamura, Y.; Yamamoto, T.; Yamaguchi, H. Effect of a traditional Japanese garlic preparation on blood pressure in prehypertensive and mildly hypertensive adults. Exp. Ther. Med. 2013, 5, 399–405. [Google Scholar] [CrossRef]
- Ried, K.; Travica, N.; Sali, A. The effect of aged garlic extract on blood pressure and other cardiovascular risk factors in uncontrolled hypertensives: The AGE at Heart trial. Integr. Blood Press Control. 2016, 9, 9–21. [Google Scholar] [CrossRef] [PubMed]
- De Santos, O.; Johns, R. Effects of garlic powder and garlic oil preparations on blood lipids, blood pressure and well-being. Br. J. Clin. Res. 1995, 6, 91–100. [Google Scholar]
- Sobenin, I.A.; Andrianova, I.V.; Demidova, O.N.; Gorchakova, T.; Orekhov, A.N. Lipid-lowering effects of time-released garlic powder tablets in double-blinded placebo-controlled randomized study. J. Atheroscler. Thromb. 2008, 15, 334–338. [Google Scholar] [CrossRef] [PubMed]
- Sobenin, I.A.; Andrianova, I.V.; Fomchenkov, I.V.; Gorchakova, T.V.; Orekhov, A.N. Time-released garlic powder tablets lower systolic and diastolic blood pressure in men with mild and moderate arterial hypertension. Hypertens. Res. 2009, 32, 433–437. [Google Scholar] [CrossRef] [PubMed]
- Vorberg, G.; Schneider, B. Therapy with garlic: Results of a placebo-controlled, double-blind study. Br. J. Clin. Pract. Suppl. 1990, 69, 7–11. [Google Scholar]
- Christiansen, B.; Bellostas Muguerza, N.; Petersen, A.M.; Kveiborg, B.; Madsen, C.R.; Thomas, H.; Ihlemann, N.; Sorensen, J.C.; Kober, L.; Sorensen, H.; et al. Ingestion of broccoli sprouts does not improve endothelial function in humans with hypertension. PLoS ONE 2010, 5, e12461. [Google Scholar] [CrossRef]
- Langston-Cox, A.G.; Anderson, D.; Creek, D.J.; Palmer, K.R.; Marshall, S.A.; Wallace, E.M. Sulforaphane Bioavailability and Effects on Blood Pressure in Women with Pregnancy Hypertension. Reprod. Sci. 2021, 28, 1489–1497. [Google Scholar] [CrossRef]
- Mirmiran, P.; Bahadoran, Z.; Golzarand, M.; Zojaji, H.; Azizi, F. A comparative study of broccoli sprouts powder and standard triple therapy on cardiovascular risk factors following H.pylori eradication: A randomized clinical trial in patients with type 2 diabetes. J. Diabetes Metab. Disord. 2014, 13, 64. [Google Scholar] [CrossRef]
- Borgi, L.; Muraki, I.; Satija, A.; Willett, W.C.; Rimm, E.B.; Forman, J.P. Fruit and Vegetable Consumption and the Incidence of Hypertension in Three Prospective Cohort Studies. Hypertension 2016, 67, 288–293. [Google Scholar] [CrossRef]
- Zhao, C.; Liu, Y.Y.; Lai, S.S.; Cao, H.; Guan, Y.; Cheang, W.S.; Liu, B.; Zhao, K.W.; Miao, S.; Riviere, C.; et al. Effects of domestic cooking process on the chemical and biological properties of dietary phytochemicals. Trends Food Sci. Tech. 2019, 85, 55–66. [Google Scholar] [CrossRef]
- Oliviero, T.; Verkerk, R.; Dekker, M. Isothiocyanates from Brassica Vegetables-Effects of Processing, Cooking, Mastication, and Digestion. Mol. Nutr. Food Res. 2018, 62, e1701069. [Google Scholar] [CrossRef] [PubMed]
| First Author, Year | Experimental Model | Treatment | Daily Dose (mg/kg) | Time | BP in the Control Group at the End of Treatment | BP in the Treated Group at the End of Treatment | BP Change (%) from the Baseline |
|---|---|---|---|---|---|---|---|
| Chen, 2021 [61] | Deoxycorticosterone acetate salt-induced hypertensive rats | Black garlic extract | 50, orally | 7 weeks | 173.4 ± 1.8 | 155.0 ± 3.2 | - |
| Chen, 2021 [61] | Deoxycorticosterone acetate salt-induced hypertensive rats | Black garlic extract | 100, orally | 7 weeks | 173.4 ± 1.8 | 150.0 ± 3.0 | - |
| Cui, 2020 [67] | Spontaneously hypertensive rats | Allicin | 7, orally | 4 weeks | 194.20 ± 8.6 | 168.22 ± 2.6 | - |
| Cui, 2020 [67] | Spontaneously hypertensive rats | Allicin | 14, orally | 4 weeks | 194.20 ± 8.6 | 141.01 ± 2.5 | - |
| Dubey, 2017 [69] | Dexamethasone-induced hypertensive rats | Allicin | 8, orally | 8 weeks | 133.6 ± 0.8 | 103.8 ± 1.9 | - |
| Elkayam, 2001 [70] | High-fructose diet-induced hypertensive rats | Allicin | 8, orally | 2 weeks | 152.4 ± 3.9 | 139.7 ± 12.0 | −8.9 ± 7.8 |
| Garcia-Trejo, 2016 [71] | Hypertensive rats with chronic kidney disease | Allicin | 40, orally | 6 weeks | Significant antihypertensive effects * | ||
| Han, 2011 [59] | Spontaneously hypertensive rats | Processed garlic | 30–50, orally | 8 weeks | Significant antihypertensive effects * | ||
| Harauma, 2006 [60] | Spontaneously hypertensive rats | Aged garlic extract/raw garlic | Unknown | 10 weeks | Significant antihypertensive effects * | ||
| Hsu, 2021 [62] | High-fat diet-induced hypertensive rats | Garlic oil | 100, orally (maternal supplementation) | During pregnancy and lactation | 153.0 ± 1.0 | 139.0 ± 1.0 | - |
| Jeremic, 2020 [65] | High-fat diet-induced hypertensive rats | Diallyl trisulfide | 40, orally | 3 weeks | Significant antihypertensive effects * | ||
| Nwokocha, 2011 [58] | Two-kidney-one-clip hypertensive rats | Garlic extract | 20, intravenously | Acute administration (30 min) | - | - | 16.7 ± 2.0 |
| Sharifi, 2003 [56] | Two-kidney-one-clip hypertensive rats | Garlic extract | 50, orally | 4 weeks | Significant antihypertensive effects * | ||
| First Author, Year | Experimental Model | Treatment | Daily Dose | Time | BP in the Control Group at the End of Treatment | BP in the Treated Group at the End of Treatment | BP Change (%) from the Baseline |
|---|---|---|---|---|---|---|---|
| Aekthammarat, 2019 [77] | L-NAME-induced hypertensive rats | Moringa oleifera leaf extract | 30 mg/kg, orally | 3 weeks | 189.9 ± 2.1 | 177.0 ± 2.7 | - |
| Aekthammarat, 2019 [77] | L-NAME-induced hypertensive rats | Moringa oleifera leaf extract | 60 mg/kg, orally | 3 weeks | 189.9 ± 2.1 | 152.0 ± 0.7 | - |
| Lin, 2020 [76] | Spontaneously hypertensive rats | WD solution from Semen Brassicae | 0.5 g/kg, orally | 8 weeks | 192.2 ± 2.6 | 128.7 ± 2.3 | - |
| Lin, 2020 [76] | Spontaneously hypertensive rats | WD solution from Semen Brassicae | 1 g/kg, orally | 8 weeks | 192.2 ± 2.6 | 118.7 ± 2.6 | - |
| Lin, 2020 [76] | Spontaneously hypertensive rats | WD solution from Semen Brassicae | 1 g/kg, orally | 8 weeks | 192.2 ± 2.6 | 104.6 ± 1.8 | - |
| Martelli, 2020 [72] | Spontaneously hypertensive rats | Erucin | 10 mg/kg, intraperitoneally | Acute administration (2 h) | - | - | −23.9 ± 3.8 |
| Salma, 2018 [74] | High salt (NaCl)-induced hypertensive rats | Crude extract of Eruca sativa Mill. | 1 mg/kg, intravenously | Acute administration | - | - | −25.4 ± 3.9 |
| Salma, 2018 [74] | High salt (NaCl)-induced hypertensive rats | Crude extract of Eruca sativa Mill. | 3 mg/kg, intravenously | Acute administration | - | - | −39.2 ± 1.8 |
| Salma, 2018 [74] | High salt (NaCl)-induced hypertensive rats | Crude extract of Eruca sativa Mill. | 10 mg/kg, intravenously | Acute administration | - | - | −46.8 ± 3.6 |
| Salma, 2018 [74] | High salt (NaCl)-induced hypertensive rats | Crude extract of Eruca sativa Mill. | 30 mg/kg, intravenously | Acute administration | - | - | −58.3 ± 0.9 |
| Salma, 2018 [74] | High salt (NaCl)-induced hypertensive rats | Crude extract of Eruca sativa Mill. | 30 mg/kg, orally | Acute administration | - | - | −40.3 ± 1.2 |
| Salma, 2018 [74] | High salt (NaCl)-induced hypertensive rats | Crude extract of Eruca sativa Mill. | 100 mg/kg orally | Acute administration | - | - | −59.4 ± 0.8 |
| First Author, Year | No. of Subjects in the Experimental Group | Treatment | Daily Dose (mg) | Time (Weeks) | Change in BP from the Baseline |
|---|---|---|---|---|---|
| Ashraf, 2013 [86] | 30 | Garlic tablets | 300 | 24 | −2.3 ± 0.9 |
| Ashraf, 2013 [86] | 30 | Garlic tablets | 600 | 24 | −4.3 ± 1.0 |
| Ashraf, 2013 [86] | 30 | Garlic tablets | 900 | 24 | −6.1 ± 1.0 |
| Ashraf, 2013 [86] | 30 | Garlic tablets | 1200 | 24 | −6.7 ± 1.2 |
| Ashraf, 2013 [86] | 30 | Garlic tablets | 1500 | 24 | −7.6 ± 0.9 |
| Auer, 1990 * [87] | 20 | Garlic powder | 600 | 12 | −19.0 ± 3.5 |
| Han, 2011 [59] | 23 | Processed garlic | 500 | 8 | −8.1 ± 2.9 |
| Holzgartner, 1992 * [88] | 47 | Garlic powder | 900 | 12 | −8.0 ± 1.7 |
| Kandziora, 1988 * [89] | 20 | Garlic powder | 600 | 12 | −16.0 ± 1.7 |
| Kravchuk, 2021 [83] | 10 | Garlic powder | 400 | 4 | −16.5 ± 2.6 |
| Nakasone, 2013 * [90] | 23 | Garlic powder | 188 | 12 | −6.6 ± 1.8 |
| Ried, 2010 * [80] | 6 | Aged garlic extract | 960 | 12 | −15.2 ± 2.6 |
| Ried, 2013 * [81] | 20 | Aged garlic extract | 480 | 12 | −2.5 ± 3.7 |
| Ried, 2016 * [91] | 38 | Aged garlic extract | 1200 | 12 | −10.0 ± 1.8 |
| Ried, 2018 * [84] | 23 | Aged garlic extract | 1200 | 12 | −14.3 ± 2.9 |
| De Santos, 1993 * [92] | 27 | Garlic powder | 900 | 24 | −25.0 ± 4.2 |
| Sobenin, 2008 * [93] | 23 | Garlic powder | 600 | 12 | −6.6 ± 1.4 |
| Sobenin, 2009 * [94] | 18 | Garlic powder | 2400 | 8 | −9.3 ± 0.7 |
| Soleimani, 2021 [85] | 47 | Garlic powder | 800 | 15 | −6.7 ± 1.3 |
| Vorberg, 1990 * [95] | 20 | Garlic powder | 900 | 16 | −6.0 ± 2.4 |
| First Author, Year | No. of Subjects in the Experimental Group | Treatment | Daily Dose (g) | Time | Change in BP from the Baseline |
|---|---|---|---|---|---|
| Christiansen, 2010 [96] | 20 | Dried broccoli sprouts | 10 | 4 weeks | −7.8 [−19.13; 3.53] |
| Langston-Cox, 2021 [97] | 12 | Myrosinase-activated broccoli seed extract (BroccoMax®) | 1–2 | 8 h | Antihypertensive effects not observed |
| Mirmiran, 2014 [98] | 14 | Broccoli sprouts powder | 6 | 4 weeks | −6.0 ± 8.3 |
| Mirmiran, 2014 [98] | 22 | Broccoli sprouts powder + STT | 6 | 4 weeks | −14.0 ± 5.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piragine, E.; Citi, V.; Lawson, K.; Calderone, V.; Martelli, A. Potential Effects of Natural H2S-Donors in Hypertension Management. Biomolecules 2022, 12, 581. https://doi.org/10.3390/biom12040581
Piragine E, Citi V, Lawson K, Calderone V, Martelli A. Potential Effects of Natural H2S-Donors in Hypertension Management. Biomolecules. 2022; 12(4):581. https://doi.org/10.3390/biom12040581
Chicago/Turabian StylePiragine, Eugenia, Valentina Citi, Kim Lawson, Vincenzo Calderone, and Alma Martelli. 2022. "Potential Effects of Natural H2S-Donors in Hypertension Management" Biomolecules 12, no. 4: 581. https://doi.org/10.3390/biom12040581
APA StylePiragine, E., Citi, V., Lawson, K., Calderone, V., & Martelli, A. (2022). Potential Effects of Natural H2S-Donors in Hypertension Management. Biomolecules, 12(4), 581. https://doi.org/10.3390/biom12040581








