Spectrum of Kidney Injury Following COVID-19 Disease: Renal Biopsy Findings in a Single Italian Pathology Service
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Overall Population
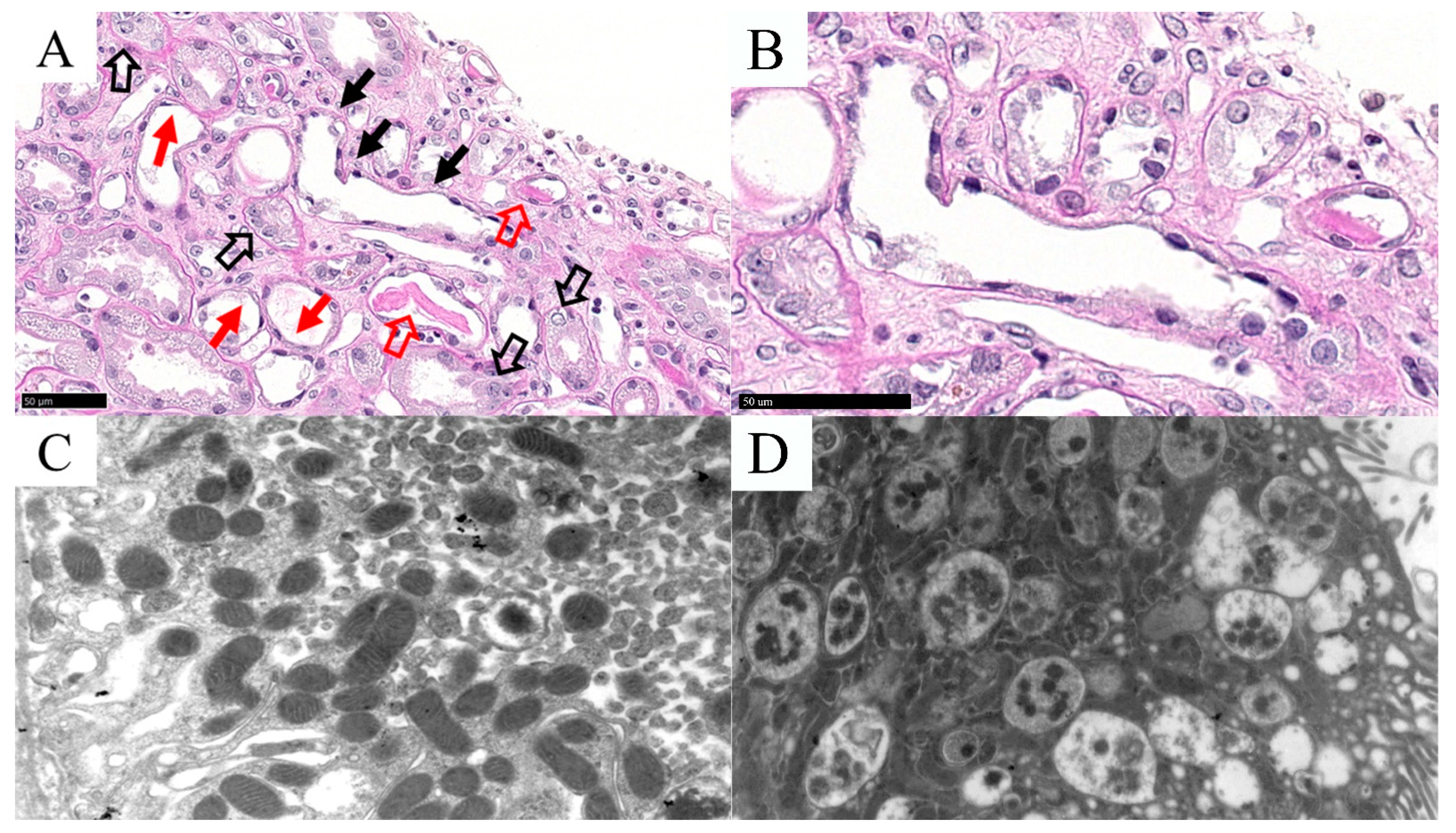
3.2. Acute Tubular Necrosis (ATN)
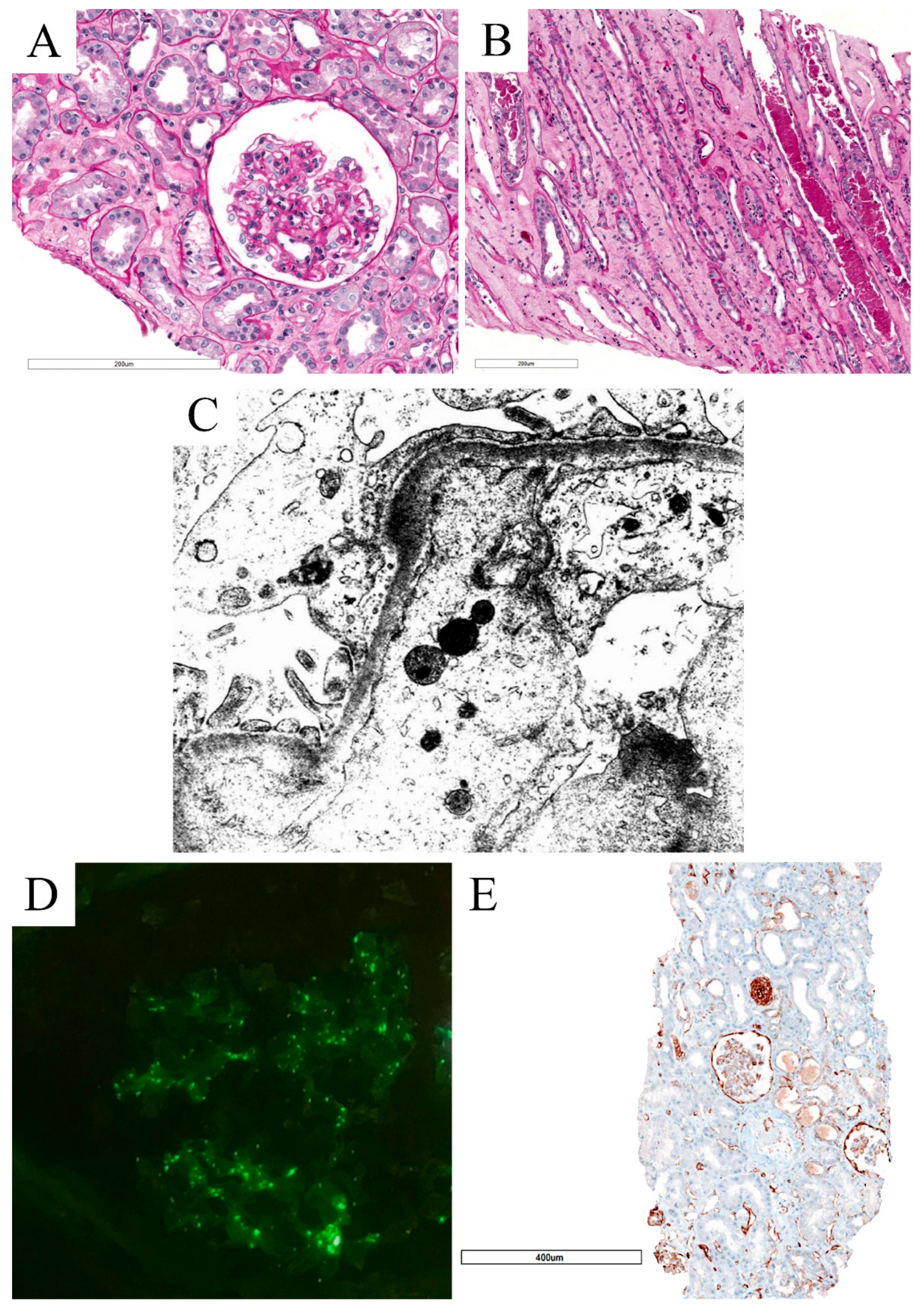
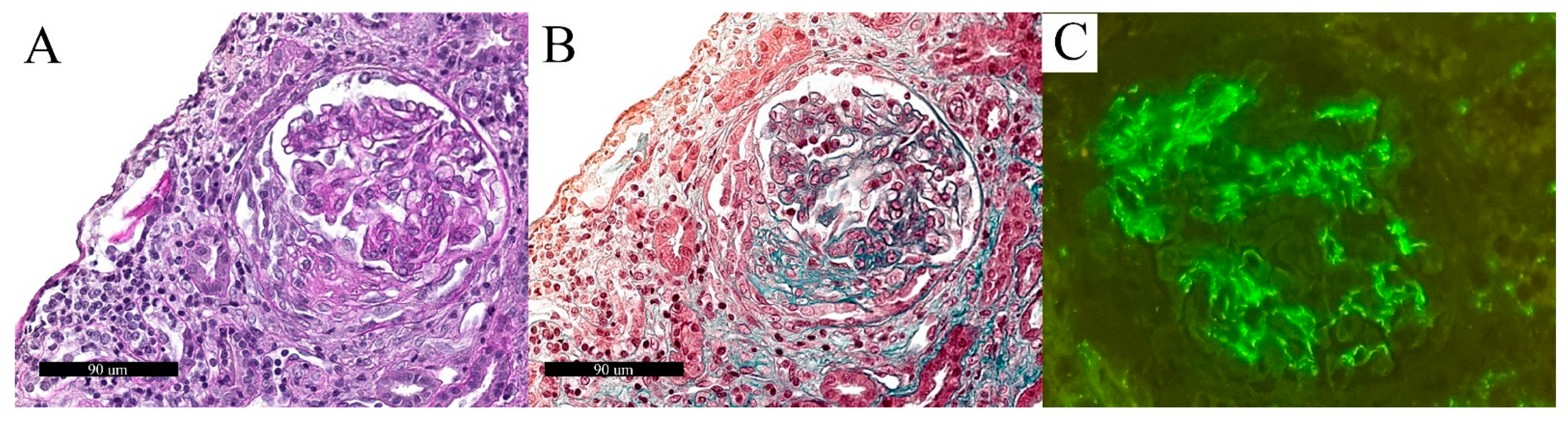
3.3. Collapsing Glomerulopathy (CG)
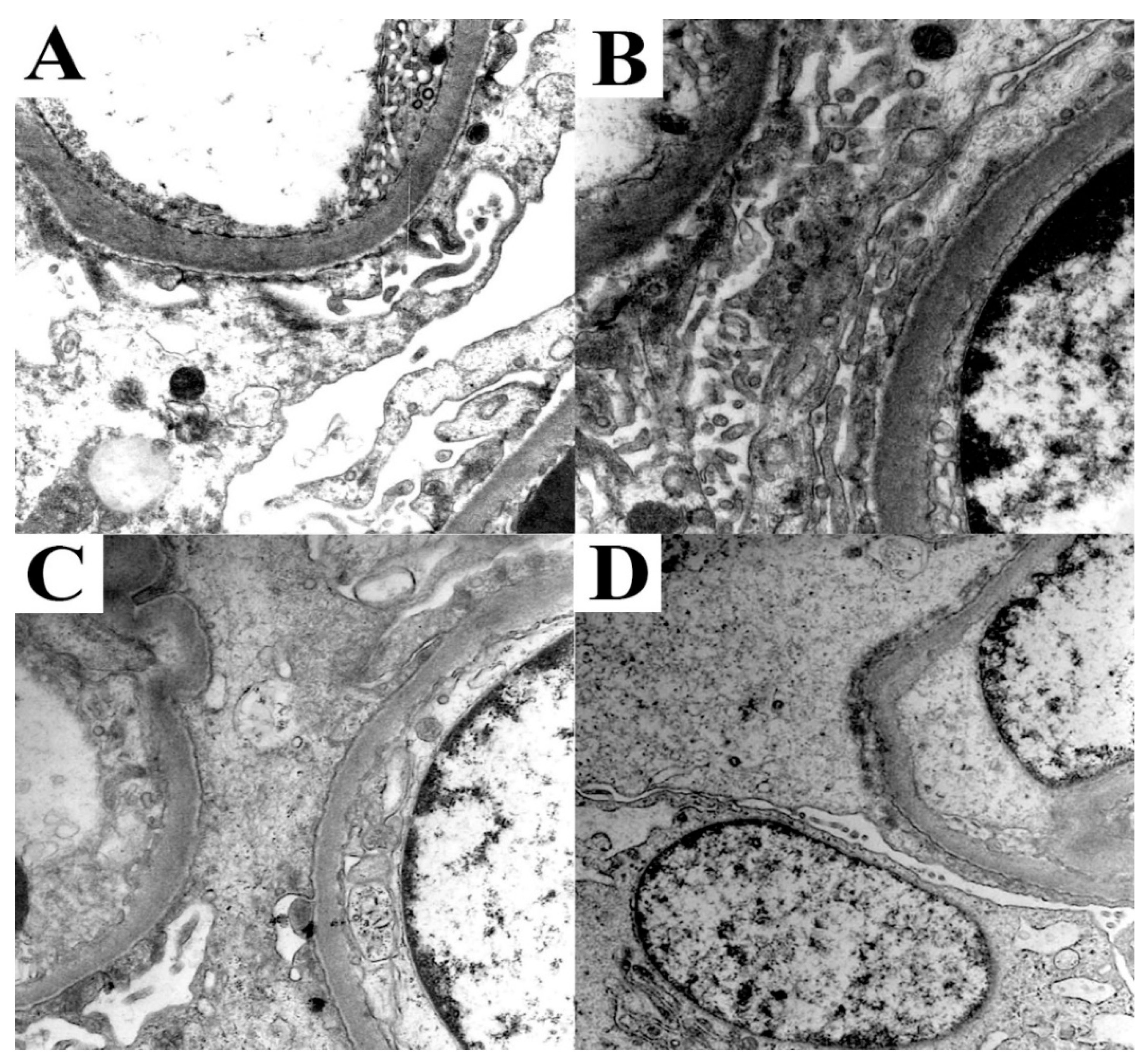
3.4. Minimal Change Disease (MCD)
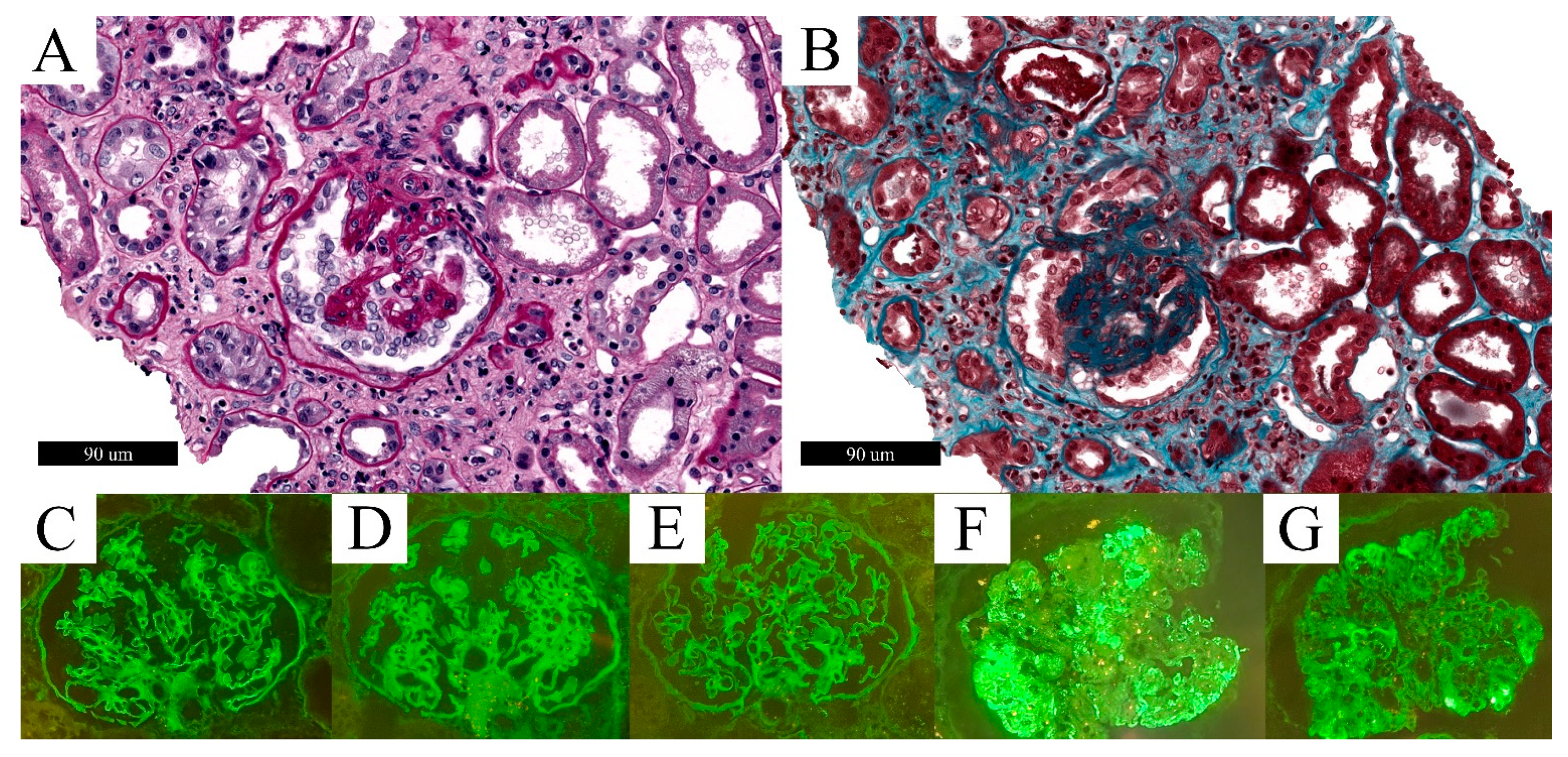
3.5. C3 Glomerulonephritis (C3G)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Saied, E.M.; El-Maradny, Y.A.; Osman, A.A.; Darwish, A.M.G.; Abo Nahas, H.H.; Niedbala, G.; Piekutowska, M.; Abdel-Rahman, M.A.; Balbool, B.A.; Abdel-Azeem, A.M. A Comprehensive Review about the Molecular Structure of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2): Insights into Natural Products against COVID-19. Pharmaceutics 2021, 13, 1759. [Google Scholar] [CrossRef] [PubMed]
- Kayode, A.J.; Banji-Onisile, F.O.; Olaniran, A.O.; Okoh, A.I. An Overview of the Pathogenesis, Transmission, Diagnosis, and Management of Endemic Human Coronaviruses: A Reflection on the Past and Present Episodes and Possible Future Outbreaks. Pathogens 2021, 10, 1108. [Google Scholar] [CrossRef]
- Barth, R.F.; Buja, L.M.; Barth, A.L.; Carpenter, D.E.; Parwani, A.V. A Comparison of the Clinical, Viral, Pathologic, and Immunologic Features of Severe Acute Respiratory Syndrome (SARS), Middle East Respiratory Syndrome (MERS), and Coronavirus 2019 (COVID-19) Diseases. Arch. Pathol. Lab. Med. 2021, 145, 1194–1211. [Google Scholar] [CrossRef] [PubMed]
- Churruca, M.; Martinez-Besteiro, E.; Counago, F.; Landete, P. COVID-19 pneumonia: A review of typical radiological characteristics. World J. Radiol. 2021, 13, 327–343. [Google Scholar] [CrossRef] [PubMed]
- Verma, H.K. Radiological and clinical spectrum of COVID-19: A major concern for public health. World J. Radiol. 2021, 13, 53–63. [Google Scholar] [CrossRef]
- AlSamman, M.; Caggiula, A.; Ganguli, S.; Misak, M.; Pourmand, A. Non-respiratory presentations of COVID-19, a clinical review. Am. J. Emerg. Med. 2020, 38, 2444–2454. [Google Scholar] [CrossRef]
- Enitan, A.O.; Olasode, O.A. The Cutaneous Manifestations of COVID-19: A Review of Emerging Literature. West. Afr. J. Med. 2020, 37, 569–573. [Google Scholar]
- Divani, A.A.; Andalib, S.; Biller, J.; Di Napoli, M.; Moghimi, N.; Rubinos, C.A.; Nobleza, C.O.; Sylaja, P.N.; Toledano, M.; Lattanzi, S.; et al. Central Nervous System Manifestations Associated with COVID-19. Curr. Neurol. Neurosci. Rep. 2020, 20, 60. [Google Scholar] [CrossRef]
- Wu, Z.H.; Yang, D.L. A meta-analysis of the impact of COVID-19 on liver dysfunction. Eur. J. Med. Res. 2020, 25, 54. [Google Scholar] [CrossRef]
- Lei, H.Y.; Ding, Y.H.; Nie, K.; Dong, Y.M.; Xu, J.H.; Yang, M.L.; Liu, M.Q.; Wei, L.; Nasser, M.I.; Xu, L.Y.; et al. Potential effects of SARS-CoV-2 on the gastrointestinal tract and liver. Biomed. Pharmacother. 2021, 133, 111064. [Google Scholar] [CrossRef]
- De Giorgio, M.R.; Di Noia, S.; Morciano, C.; Conte, D. The impact of SARS-CoV-2 on skeletal muscles. Acta Myol. 2020, 39, 307–312. [Google Scholar] [CrossRef]
- Harapan, B.N.; Yoo, H.J. Neurological symptoms, manifestations, and complications associated with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease 19 (COVID-19). J. Neurol. 2021, 268, 3059–3071. [Google Scholar] [CrossRef] [PubMed]
- Capaccione, K.M.; Yang, H.; West, E.; Patel, H.; Ma, H.; Patel, S.; Fruauff, A.; Loeb, G.; Maddocks, A.; Borowski, A.; et al. Pathophysiology and Imaging Findings of COVID-19 Infection: An Organ-system Based Review. Acad. Radiol. 2021, 28, 595–607. [Google Scholar] [CrossRef] [PubMed]
- Gattinoni, L.; Gattarello, S.; Steinberg, I.; Busana, M.; Palermo, P.; Lazzari, S.; Romitti, F.; Quintel, M.; Meissner, K.; Marini, J.J.; et al. COVID-19 pneumonia: Pathophysiology and management. Eur. Respir. Rev. 2021, 30, 210138. [Google Scholar] [CrossRef] [PubMed]
- Parker, A.M.; Brigham, E.; Connolly, B.; McPeake, J.; Agranovich, A.V.; Kenes, M.T.; Casey, K.; Reynolds, C.; Schmidt, K.F.R.; Kim, S.Y.; et al. Addressing the post-acute sequelae of SARS-CoV-2 infection: A multidisciplinary model of care. Lancet Respir. Med. 2021, 9, 1328–1341. [Google Scholar] [CrossRef]
- Neta, F.I.; Fernandes, A.C.L.; Vale, A.J.M.; Pinheiro, F.I.; Cobucci, R.N.; Azevedo, E.P.; Guzen, F.P. Pathophysiology and possible treatments for olfactory-gustatory disorders in patients affected by COVID-19. Curr. Res. Pharmacol. Drug Discov. 2021, 2, 100035. [Google Scholar] [CrossRef]
- Liang, F.; Wang, Y. COVID-19 Anosmia: High Prevalence, Plural Neuropathogenic Mechanisms, and Scarce Neurotropism of SARS-CoV-2? Viruses 2021, 13, 2225. [Google Scholar] [CrossRef]
- Zhang, J.; Garrett, S.; Sun, J. Gastrointestinal symptoms, pathophysiology, and treatment in COVID-19. Genes Dis. 2021, 8, 385–400. [Google Scholar] [CrossRef]
- Groff, A.; Kavanaugh, M.; Ramgobin, D.; McClafferty, B.; Aggarwal, C.S.; Golamari, R.; Jain, R. Gastrointestinal Manifestations of COVID-19: A Review of What We Know. Ochsner J. 2021, 21, 177–180. [Google Scholar] [CrossRef]
- Bodro, M.; Compta, Y.; Sanchez-Valle, R. Presentations and mechanisms of CNS disorders related to COVID-19. Neurol. Neuroimmunol. Neuroinflamm. 2021, 8, e923. [Google Scholar] [CrossRef]
- Li, M.; Chen, S.; Xiang, X.; Wang, Q.; Liu, X. Effects of SARS-CoV-2 and its functional receptor ACE2 on the cardiovascular system. Herz 2020, 45, 659–662. [Google Scholar] [CrossRef] [PubMed]
- Dhakal, B.P.; Sweitzer, N.K.; Indik, J.H.; Acharya, D.; William, P. SARS-CoV-2 Infection and Cardiovascular Disease: COVID-19 Heart. Heart Lung Circ. 2020, 29, 973–987. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Chen, X.; Ren, X.; Wu, Y.; Ren, S.; Qin, C. SARS-CoV-2 effects in the genitourinary system and prospects of sex hormone therapy. Asian J. Urol. 2021, 8, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Madhavan, M.V.; Sehgal, K.; Nair, N.; Mahajan, S.; Sehrawat, T.S.; Bikdeli, B.; Ahluwalia, N.; Ausiello, J.C.; Wan, E.Y.; et al. Extrapulmonary manifestations of COVID-19. Nat. Med. 2020, 26, 1017–1032. [Google Scholar] [CrossRef] [PubMed]
- Mokhtari, T.; Hassani, F.; Ghaffari, N.; Ebrahimi, B.; Yarahmadi, A.; Hassanzadeh, G. COVID-19 and multiorgan failure: A narrative review on potential mechanisms. J. Mol. Histol. 2020, 51, 613–628. [Google Scholar] [CrossRef] [PubMed]
- Hamming, I.; Timens, W.; Bulthuis, M.L.; Lely, A.T.; Navis, G.; van Goor, H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004, 203, 631–637. [Google Scholar] [CrossRef]
- Benton, D.J.; Wrobel, A.G.; Xu, P.; Roustan, C.; Martin, S.R.; Rosenthal, P.B.; Skehel, J.J.; Gamblin, S.J. Receptor binding and priming of the spike protein of SARS-CoV-2 for membrane fusion. Nature 2020, 588, 327–330. [Google Scholar] [CrossRef]
- Lan, J.; Ge, J.; Yu, J.; Shan, S.; Zhou, H.; Fan, S.; Zhang, Q.; Shi, X.; Wang, Q.; Zhang, L.; et al. Structure of the SARS-CoV-2 spike receptor-binding domain bound to the ACE2 receptor. Nature 2020, 581, 215–220. [Google Scholar] [CrossRef] [Green Version]
- Shang, J.; Ye, G.; Shi, K.; Wan, Y.; Luo, C.; Aihara, H.; Geng, Q.; Auerbach, A.; Li, F. Structural basis of receptor recognition by SARS-CoV-2. Nature 2020, 581, 221–224. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Moore, M.J.; Vasilieva, N.; Sui, J.; Wong, S.K.; Berne, M.A.; Somasundaran, M.; Sullivan, J.L.; Luzuriaga, K.; Greenough, T.C.; et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 2003, 426, 450–454. [Google Scholar] [CrossRef] [Green Version]
- Soler, M.J.; Wysocki, J.; Batlle, D. Angiotensin-converting enzyme 2 and the kidney. Exp. Physiol. 2008, 93, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Koitka, A.; Cooper, M.E.; Thomas, M.C.; Tikellis, C. Angiotensin converting enzyme 2 in the kidney. Clin. Exp. Pharmacol. Physiol. 2008, 35, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.W.; Xu, D.; Zhang, H.; Zhou, W.; Wang, L.H.; Cui, X.G. Identification of a potential mechanism of acute kidney injury during the COVID-19 outbreak: A study based on single-cell transcriptome analysis. Intensive Care Med. 2020, 46, 1114–1116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Varga, Z.; Flammer, A.J.; Steiger, P.; Haberecker, M.; Andermatt, R.; Zinkernagel, A.S.; Mehra, M.R.; Schuepbach, R.A.; Ruschitzka, F.; Moch, H. Endothelial cell infection and endotheliitis in COVID-19. Lancet 2020, 395, 1417–1418. [Google Scholar] [CrossRef]
- Contributors to the C4 Article. C4 article: Implications of COVID-19 in transplantation. Am. J. Transplant. 2021, 21, 1801–1815. [Google Scholar] [CrossRef]
- Glowacka, M.; Lipka, S.; Mlynarska, E.; Franczyk, B.; Rysz, J. Acute Kidney Injury in COVID-19. Int. J. Mol. Sci. 2021, 22, 8081. [Google Scholar] [CrossRef] [PubMed]
- Kaye, A.D.; Okeagu, C.N.; Tortorich, G.; Pham, A.D.; Ly, E.I.; Brondeel, K.C.; Eng, M.R.; Luedi, M.M.; Urman, R.D.; Cornett, E.M. COVID-19 impact on the renal system: Pathophysiology and clinical outcomes. Best Pract. Res. Clin. Anaesthesiol. 2021, 35, 449–459. [Google Scholar] [CrossRef]
- Bruchfeld, A. The COVID-19 pandemic: Consequences for nephrology. Nat. Rev. Nephrol. 2021, 17, 81–82. [Google Scholar] [CrossRef]
- Caillard, S.; Thaunat, O. COVID-19 vaccination in kidney transplant recipients. Nat. Rev. Nephrol. 2021, 17, 785–787. [Google Scholar] [CrossRef]
- Windpessl, M.; Bruchfeld, A.; Anders, H.J.; Kramer, H.; Waldman, M.; Renia, L.; Ng, L.F.P.; Xing, Z.; Kronbichler, A. COVID-19 vaccines and kidney disease. Nat. Rev. Nephrol. 2021, 17, 291–293. [Google Scholar] [CrossRef]
- Kronbichler, A.; Gauckler, P.; Windpessl, M.; Il Shin, J.; Jha, V.; Rovin, B.H.; Oberbauer, R. COVID-19: Implications for immunosuppression in kidney disease and transplantation. Nat. Rev. Nephrol. 2020, 16, 365–367. [Google Scholar] [CrossRef]
- Nadim, M.K.; Forni, L.G.; Mehta, R.L.; Connor, M.J., Jr.; Liu, K.D.; Ostermann, M.; Rimmele, T.; Zarbock, A.; Bell, S.; Bihorac, A.; et al. COVID-19-associated acute kidney injury: Consensus report of the 25th Acute Disease Quality Initiative (ADQI) Workgroup. Nat. Rev. Nephrol. 2020, 16, 747–764. [Google Scholar] [CrossRef]
- Zaman, M.S.; Sizemore, R.C. Diverse Manifestations of COVID-19: Some Suggested Mechanisms. Int. J. Environ. Res. Public Health 2021, 18, 9785. [Google Scholar] [CrossRef]
- Perico, L.; Benigni, A.; Casiraghi, F.; Ng, L.F.P.; Renia, L.; Remuzzi, G. Immunity, endothelial injury and complement-induced coagulopathy in COVID-19. Nat. Rev. Nephrol. 2021, 17, 46–64. [Google Scholar] [CrossRef] [PubMed]
- Alasfar, S.; Avery, R.K. The impact of COVID-19 on kidney transplantation. Nat. Rev. Nephrol. 2020, 16, 568–569. [Google Scholar] [CrossRef]
- Cheng, Y.; Luo, R.; Wang, K.; Zhang, M.; Wang, Z.; Dong, L.; Li, J.; Yao, Y.; Ge, S.; Xu, G. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020, 97, 829–838. [Google Scholar] [CrossRef] [PubMed]
- Allison, S. Kidney involvement in COVID-19. Nat. Rev. Nephrol. 2021, 17, 572. [Google Scholar] [CrossRef]
- Ronco, C.; Reis, T. Kidney involvement in COVID-19 and rationale for extracorporeal therapies. Nat. Rev. Nephrol. 2020, 16, 308–310. [Google Scholar] [CrossRef] [Green Version]
- Yende, S.; Parikh, C.R. Long COVID and kidney disease. Nat. Rev. Nephrol. 2021, 17, 792–793. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.D.; Sumeh, A.S.; Sheraz, M.; Kavitha, M.S.; Venmathi Maran, B.A.; Rodrigues, K.F. A mini-review on the impact of COVID 19 on vital organs. Biomed. Pharmacother. 2021, 143, 112158. [Google Scholar] [CrossRef]
- Legrand, M.; Bell, S.; Forni, L.; Joannidis, M.; Koyner, J.L.; Liu, K.; Cantaluppi, V. Pathophysiology of COVID-19-associated acute kidney injury. Nat. Rev. Nephrol. 2021, 17, 751–764. [Google Scholar] [CrossRef]
- George, J.A.; Khoza, S. SARS-CoV-2 Infection and the Kidneys: An Evolving Picture. Adv. Exp. Med. Biol. 2021, 1327, 107–118. [Google Scholar] [CrossRef]
- Chan, L.; Chaudhary, K.; Saha, A.; Chauhan, K.; Vaid, A.; Zhao, S.; Paranjpe, I.; Somani, S.; Richter, F.; Miotto, R.; et al. AKI in Hospitalized Patients with COVID-19. J. Am. Soc. Nephrol. 2021, 32, 151–160. [Google Scholar] [CrossRef]
- Karagiannidis, C.; Mostert, C.; Hentschker, C.; Voshaar, T.; Malzahn, J.; Schillinger, G.; Klauber, J.; Janssens, U.; Marx, G.; Weber-Carstens, S.; et al. Case characteristics, resource use, and outcomes of 10 021 patients with COVID-19 admitted to 920 German hospitals: An observational study. Lancet Respir. Med. 2020, 8, 853–862. [Google Scholar] [CrossRef]
- Kesiena, O.; Papadopoulos, P.; Amakye, D.; Hama, E.; Mackay, R. COVID-19 associated collapsing glomerulopathy presenting as acute kidney injury on chronic kidney disease: A case report and review of the literature. CEN Case Rep. 2021. [Google Scholar] [CrossRef] [PubMed]
- Kudose, S.; Santoriello, D.; Bomback, A.S.; Sekulic, M.; Batal, I.; Stokes, M.B.; Ghavami, I.A.; Kim, J.S.; Marasa, M.; Xu, K.; et al. Longitudinal Outcomes of COVID-19-Associated Collapsing Glomerulopathy and Other Podocytopathies. J. Am. Soc. Nephrol. 2021, 32, 2958–2969. [Google Scholar] [CrossRef]
- Sharma, Y.; Nasr, S.H.; Larsen, C.P.; Kemper, A.; Ormsby, A.H.; Williamson, S.R. COVID-19-Associated Collapsing Focal Segmental Glomerulosclerosis: A Report of 2 Cases. Kidney Med. 2020, 2, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Uppal, N.N.; Wanchoo, R.; Shah, H.H.; Yang, Y.; Parikh, R.; Khanin, Y.; Madireddy, V.; Larsen, C.P.; Jhaveri, K.D.; et al. COVID-19-Associated Kidney Injury: A Case Series of Kidney Biopsy Findings. J. Am. Soc. Nephrol. 2020, 31, 1948–1958. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Ng, J.H.; Bijol, V.; Jhaveri, K.D.; Wanchoo, R. Pathology of COVID-19-associated acute kidney injury. Clin. Kidney J. 2021, 14, i30–i39. [Google Scholar] [CrossRef]
- Smith, K.D.; Akilesh, S. Pathogenesis of coronavirus disease 2019-associated kidney injury. Curr. Opin. Nephrol. Hypertens 2021, 30, 324–331. [Google Scholar] [CrossRef]
- Izzedine, H.; Brocheriou, I.; Arzouk, N.; Seilhean, D.; Couvert, P.; Cluzel, P.; Pha, M.; Le Monnier, O.; Varnous, S.; Andreelli, F.; et al. COVID-19-associated collapsing glomerulopathy: A report of two cases and literature review. Intern. Med. J. 2020, 50, 1551–1558. [Google Scholar] [CrossRef] [PubMed]
- Shetty, A.A.; Tawhari, I.; Safar-Boueri, L.; Seif, N.; Alahmadi, A.; Gargiulo, R.; Aggarwal, V.; Usman, I.; Kisselev, S.; Gharavi, A.G.; et al. COVID-19-Associated Glomerular Disease. J. Am. Soc. Nephrol. 2021, 32, 33–40. [Google Scholar] [CrossRef]
- Nasr, S.H.; Kopp, J.B. COVID-19-Associated Collapsing Glomerulopathy: An Emerging Entity. Kidney Int. Rep. 2020, 5, 759–761. [Google Scholar] [CrossRef]
- Akilesh, S.; Nast, C.C.; Yamashita, M.; Henriksen, K.; Charu, V.; Troxell, M.L.; Kambham, N.; Bracamonte, E.; Houghton, D.; Ahmed, N.I.; et al. Multicenter Clinicopathologic Correlation of Kidney Biopsies Performed in COVID-19 Patients Presenting With Acute Kidney Injury or Proteinuria. Am. J. Kidney Dis. 2021, 77, 82–93 e81. [Google Scholar] [CrossRef] [PubMed]
- Elsoukkary, S.S.; Mostyka, M.; Dillard, A.; Berman, D.R.; Ma, L.X.; Chadburn, A.; Yantiss, R.K.; Jessurun, J.; Seshan, S.V.; Borczuk, A.C.; et al. Autopsy Findings in 32 Patients with COVID-19: A Single-Institution Experience. Pathobiology 2021, 88, 56–68. [Google Scholar] [CrossRef]
- Ferlicot, S.; Jamme, M.; Gaillard, F.; Oniszczuk, J.; Couturier, A.; May, O.; Grunenwald, A.; Sannier, A.; Moktefi, A.; Le Monnier, O.; et al. The spectrum of kidney biopsies in hospitalized patients with COVID-19, acute kidney injury, and/or proteinuria. Nephrol. Dial. Transplant. 2021, 36, 1253–1262. [Google Scholar] [CrossRef]
- Jhaveri, K.D.; Meir, L.R.; Flores Chang, B.S.; Parikh, R.; Wanchoo, R.; Barilla-LaBarca, M.L.; Bijol, V.; Hajizadeh, N. Thrombotic microangiopathy in a patient with COVID-19. Kidney Int. 2020, 98, 509–512. [Google Scholar] [CrossRef] [PubMed]
- Golmai, P.; Larsen, C.P.; DeVita, M.V.; Wahl, S.J.; Weins, A.; Rennke, H.G.; Bijol, V.; Rosenstock, J.L. Histopathologic and Ultrastructural Findings in Postmortem Kidney Biopsy Material in 12 Patients with AKI and COVID-19. J. Am. Soc. Nephrol. 2020, 31, 1944–1947. [Google Scholar] [CrossRef]
- May, R.M.; Cassol, C.; Hannoudi, A.; Larsen, C.P.; Lerma, E.V.; Haun, R.S.; Braga, J.R.; Hassen, S.I.; Wilson, J.; VanBeek, C.; et al. A multi-center retrospective cohort study defines the spectrum of kidney pathology in Coronavirus 2019 Disease (COVID-19). Kidney Int. 2021, 100, 1303–1315. [Google Scholar] [CrossRef]
- Peleg, Y.; Kudose, S.; D'Agati, V.; Siddall, E.; Ahmad, S.; Nickolas, T.; Kisselev, S.; Gharavi, A.; Canetta, P. Acute Kidney Injury Due to Collapsing Glomerulopathy Following COVID-19 Infection. Kidney Int. Rep. 2020, 5, 940–945. [Google Scholar] [CrossRef] [PubMed]
- Kudose, S.; Batal, I.; Santoriello, D.; Xu, K.; Barasch, J.; Peleg, Y.; Canetta, P.; Ratner, L.E.; Marasa, M.; Gharavi, A.G.; et al. Kidney Biopsy Findings in Patients with COVID-19. J. Am. Soc. Nephrol. 2020, 31, 1959–1968. [Google Scholar] [CrossRef] [PubMed]
- Kissling, S.; Rotman, S.; Gerber, C.; Halfon, M.; Lamoth, F.; Comte, D.; Lhopitallier, L.; Sadallah, S.; Fakhouri, F. Collapsing glomerulopathy in a COVID-19 patient. Kidney Int. 2020, 98, 228–231. [Google Scholar] [CrossRef] [PubMed]
- Larsen, C.P.; Bourne, T.D.; Wilson, J.D.; Saqqa, O.; Sharshir, M.A. Collapsing Glomerulopathy in a Patient With COVID-19. Kidney Int. Rep. 2020, 5, 935–939. [Google Scholar] [CrossRef]
- Magoon, S.; Bichu, P.; Malhotra, V.; Alhashimi, F.; Hu, Y.; Khanna, S.; Berhanu, K. COVID-19-Related Glomerulopathy: A Report of 2 Cases of Collapsing Focal Segmental Glomerulosclerosis. Kidney Med. 2020, 2, 488–492. [Google Scholar] [CrossRef] [PubMed]
- Noble, R.; Tan, M.Y.; McCulloch, T.; Shantier, M.; Byrne, C.; Hall, M.; Jesky, M. Collapsing Glomerulopathy Affecting Native and Transplant Kidneys in Individuals with COVID-19. Nephron 2020, 144, 589–594. [Google Scholar] [CrossRef]
- Wu, H.; Larsen, C.P.; Hernandez-Arroyo, C.F.; Mohamed, M.M.B.; Caza, T.; Sharshir, M.; Chughtai, A.; Xie, L.; Gimenez, J.M.; Sandow, T.A.; et al. AKI and Collapsing Glomerulopathy Associated with COVID-19 and APOL 1 High-Risk Genotype. J. Am. Soc. Nephrol. 2020, 31, 1688–1695. [Google Scholar] [CrossRef]
- Kadosh, B.S.; Pavone, J.; Wu, M.; Reyentovich, A.; Gidea, C. Collapsing glomerulopathy associated with COVID-19 infection in a heart transplant recipient. J. Heart Lung Transplant. 2020, 39, 855–857. [Google Scholar] [CrossRef]
- Couturier, A.; Ferlicot, S.; Chevalier, K.; Guillet, M.; Essig, M.; Jaureguiberry, S.; Collarino, R.; Dargelos, M.; Michaut, A.; Geri, G.; et al. Indirect effects of severe acute respiratory syndrome coronavirus 2 on the kidney in coronavirus disease patients. Clin. Kidney J. 2020, 13, 347–353. [Google Scholar] [CrossRef]
- Nlandu, Y.M.; Makulo, J.R.; Pakasa, N.M.; Sumaili, E.K.; Nkondi, C.N.; Bukabau, J.B.; Beya, F.K.; Nseka, N.M.; Lepira, F.B. First Case of COVID-19-Associated Collapsing Glomerulopathy in Sub-Saharan Africa. Case Rep. Nephrol. 2020, 2020, 8820713. [Google Scholar] [CrossRef]
- Gupta, R.K.; Bhargava, R.; Shaukat, A.A.; Albert, E.; Leggat, J. Spectrum of podocytopathies in new-onset nephrotic syndrome following COVID-19 disease: A report of 2 cases. BMC Nephrol. 2020, 21, 326. [Google Scholar] [CrossRef]
- Nasr, S.H.; Alexander, M.P.; Cornell, L.D.; Herrera, L.H.; Fidler, M.E.; Said, S.M.; Zhang, P.; Larsen, C.P.; Sethi, S. Kidney Biopsy Findings in Patients With COVID-19, Kidney Injury, and Proteinuria. Am. J. Kidney Dis. 2021, 77, 465–468. [Google Scholar] [CrossRef]
- Lazareth, H.; Pere, H.; Binois, Y.; Chabannes, M.; Schurder, J.; Bruneau, T.; Karras, A.; Thervet, E.; Rabant, M.; Veyer, D.; et al. COVID-19-Related Collapsing Glomerulopathy in a Kidney Transplant Recipient. Am. J. Kidney Dis. 2020, 76, 590–594. [Google Scholar] [CrossRef]
- Rossi, G.M.; Delsante, M.; Pilato, F.P.; Gnetti, L.; Gabrielli, L.; Rossini, G.; Re, M.C.; Cenacchi, G.; Affanni, P.; Colucci, M.E.; et al. Kidney Biopsy Findings in a Critically Ill COVID-19 Patient With Dialysis-Dependent Acute Kidney Injury: A Case Against “SARS-CoV-2 Nephropathy”. Kidney Int. Rep. 2020, 5, 1100–1105. [Google Scholar] [CrossRef]
- Papadimitriou, J.C.; Drachenberg, C.B.; Kleiner, D.; Choudhri, N.; Haririan, A.; Cebotaru, V. Tubular Epithelial and Peritubular Capillary Endothelial Injury in COVID-19 AKI. Kidney Int. Rep. 2021, 6, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Li, X.J.; Li, Y.Q.; Dai, W.; Shao, T.; Liu, W.Y.; Han, M.; Xu, G.; Liu, L. Clinical and pathological findings of SARS-CoV-2 infection and concurrent IgA nephropathy: A case report. BMC Nephrol. 2020, 21, 504. [Google Scholar] [CrossRef] [PubMed]
- Sandhu, S.; Chand, S.; Bhatnagar, A.; Dabas, R.; Bhat, S.; Kumar, H.; Dixit, P.K. Possible association between IgA vasculitis and COVID-19. Dermatol. Ther. 2021, 34, e14551. [Google Scholar] [CrossRef] [PubMed]
- Suso, A.S.; Mon, C.; Onate Alonso, I.; Galindo Romo, K.; Juarez, R.C.; Ramirez, C.L.; Sanchez Sanchez, M.; Mercado Valdivia, V.; Ortiz Librero, M.; Oliet Pala, A.; et al. IgA Vasculitis With Nephritis (Henoch-Schonlein Purpura) in a COVID-19 Patient. Kidney Int. Rep. 2020, 5, 2074–2078. [Google Scholar] [CrossRef]
- Moeinzadeh, F.; Dezfouli, M.; Naimi, A.; Shahidi, S.; Moradi, H. Newly Diagnosed Glomerulonephritis During COVID-19 Infection Undergoing Immunosuppression Therapy, a Case Report. Iran. J. Kidney Dis. 2020, 14, 239–242. [Google Scholar]
- Uppal, N.N.; Kello, N.; Shah, H.H.; Khanin, Y.; De Oleo, I.R.; Epstein, E.; Sharma, P.; Larsen, C.P.; Bijol, V.; Jhaveri, K.D. De Novo ANCA-Associated Vasculitis With Glomerulonephritis in COVID-19. Kidney Int. Rep. 2020, 5, 2079–2083. [Google Scholar] [CrossRef]
- Brix, S.R.; Jones, R.B.; Jayne, D.R.W. Glomerular basement membrane nephritis: Crescentic renal inflammation and immunosuppressive intervention in the time of the severe acute respiratory syndrome coronavirus 2 pandemic. Kidney Int. 2021, 99, 1234–1235. [Google Scholar] [CrossRef]
- Koc, N.S.; Yildirim, T.; Saglam, A.; Arici, M.; Erdem, Y. A patient with COVID-19 and anti-glomerular basement membrane disease. Nefrologia 2021, 41, 471–473. [Google Scholar] [CrossRef]
- Fontana, F.; Cazzato, S.; Giovanella, S.; Ballestri, M.; Leonelli, M.; Mori, G.; Alfano, G.; Ligabue, G.; Magistroni, R.; Cenacchi, G.; et al. Oxalate Nephropathy Caused by Excessive Vitamin C Administration in 2 Patients With COVID-19. Kidney Int. Rep. 2020, 5, 1815–1822. [Google Scholar] [CrossRef] [PubMed]
- Szajek, K.; Kajdi, M.E.; Luyckx, V.A.; Fehr, T.H.; Gaspert, A.; Cusini, A.; Hohloch, K.; Grosse, P. Granulomatous interstitial nephritis in a patient with SARS-CoV-2 infection. BMC Nephrol. 2021, 22, 19. [Google Scholar] [CrossRef]
- Sethi, S.; D'Costa, M.R.; Hermann, S.M.; Nasr, S.H.; Fervenza, F.C. Immune-Complex Glomerulonephritis After COVID-19 Infection. Kidney Int Rep. 2021, 6, 1170–1173. [Google Scholar] [CrossRef]
- Goldsmith, C.S.; Miller, S.E.; Martines, R.B.; Bullock, H.A.; Zaki, S.R. Electron microscopy of SARS-CoV-2: A challenging task. Lancet 2020, 395, e99. [Google Scholar] [CrossRef]
- Roufosse, C.; Curtis, E.; Moran, L.; Hollinshead, M.; Cook, T.; Hanley, B.; Horsfield, C.; Neil, D. Electron microscopic investigations in COVID-19: Not all crowns are coronas. Kidney Int. 2020, 98, 505–506. [Google Scholar] [CrossRef] [PubMed]
- Oshiro, L.S.; Schieble, J.H.; Lennette, E.H. Electron microscopic studies of coronavirus. J. Gen. Virol. 1971, 12, 161–168. [Google Scholar] [CrossRef]
- Gambella, A.; Barreca, A.; Osella-Abate, S.; Bottasso, E.; Giarin, M.M.; Papotti, M.; Biancone, L.; Metovic, J.; Collemi, G.; Cassoni, P.; et al. Caveolin-1 in Kidney Chronic Antibody-Mediated Rejection: An Integrated Immunohistochemical and Transcriptomic Analysis Based on the Banff Human Organ Transplant (B-HOT) Gene Panel. Biomedicines 2021, 9, 1318. [Google Scholar] [CrossRef]
- Tervaert, T.W.; Mooyaart, A.L.; Amann, K.; Cohen, A.H.; Cook, H.T.; Drachenberg, C.B.; Ferrario, F.; Fogo, A.B.; Haas, M.; de Heer, E.; et al. Pathologic classification of diabetic nephropathy. J. Am. Soc. Nephrol. 2010, 21, 556–563. [Google Scholar] [CrossRef] [Green Version]
- Su, H.; Yang, M.; Wan, C.; Yi, L.X.; Tang, F.; Zhu, H.Y.; Yi, F.; Yang, H.C.; Fogo, A.B.; Nie, X.; et al. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int. 2020, 98, 219–227. [Google Scholar] [CrossRef]
- Punj, S.; Eng, E.; Shetty, A.A. Coronavirus disease 2019 and kidney injury. Curr. Opin. Nephrol. Hypertens 2021, 30, 444–449. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.E.; Brealey, J.K. Visualization of putative coronavirus in kidney. Kidney Int. 2020, 98, 231–232. [Google Scholar] [CrossRef]
- Calomeni, E.; Satoskar, A.; Ayoub, I.; Brodsky, S.; Rovin, B.H.; Nadasdy, T. Multivesicular bodies mimicking SARS-CoV-2 in patients without COVID-19. Kidney Int. 2020, 98, 233–234. [Google Scholar] [CrossRef] [PubMed]
- Farouk, S.S.; Fiaccadori, E.; Cravedi, P.; Campbell, K.N. COVID-19 and the kidney: What we think we know so far and what we don’t. J. Nephrol. 2020, 33, 1213–1218. [Google Scholar] [CrossRef] [PubMed]
- Hassler, L.; Reyes, F.; Sparks, M.A.; Welling, P.; Batlle, D. Evidence For and Against Direct Kidney Infection by SARS-CoV-2 in Patients with COVID-19. Clin. J. Am. Soc. Nephrol. 2021, 16, 1755–1765. [Google Scholar] [CrossRef]
- Shah, S.A.; Carter, H.P. New-Onset Nephrotic Syndrome in a Child Associated With COVID-19 Infection. Front. Pediatr. 2020, 8, 471. [Google Scholar] [CrossRef]
- Alvarado, A.; Franceschi, G.; Resplandor, E.; Sumba, J.; Orta, N. COVID-19 associated with onset nephrotic syndrome in a pediatric patient: Coincidence or related conditions? Pediatr. Nephrol. 2021, 36, 205–207. [Google Scholar] [CrossRef]
- Bjornstad, E.C.; Seifert, M.E.; Sanderson, K.; Feig, D.I. Kidney implications of SARS-CoV2 infection in children. Pediatr. Nephrol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Morello, W.; Vianello, F.A.; Proverbio, E.; Peruzzi, L.; Pasini, A.; Montini, G. COVID-19 and idiopathic nephrotic syndrome in children: Systematic review of the literature and recommendations from a highly affected area. Pediatr. Nephrol. 2021. [Google Scholar] [CrossRef]
- Stewart, D.J.; Hartley, J.C.; Johnson, M.; Marks, S.D.; du Pre, P.; Stojanovic, J. Renal dysfunction in hospitalised children with COVID-19. Lancet Child. Adolesc. Health 2020, 4, e28–e29. [Google Scholar] [CrossRef]
- Serafinelli, J.; Mastrangelo, A.; Morello, W.; Cerioni, V.F.; Salim, A.; Nebuloni, M.; Montini, G. Kidney involvement and histological findings in two pediatric COVID-19 patients. Pediatr. Nephrol. 2021, 36, 3789–3793. [Google Scholar] [CrossRef]
- Basiratnia, M.; Derakhshan, D.; Yeganeh, B.S.; Derakhshan, A. Acute necrotizing glomerulonephritis associated with COVID-19 infection: Report of two pediatric cases. Pediatr. Nephrol. 2021, 36, 1019–1023. [Google Scholar] [CrossRef]
- Zamora, G.; Pearson-Shaver, A.L. Minimal Change Disease. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Yamada, M.; Rastogi, P.; Ince, D.; Thayyil, A.; Adela Mansilla, M.; Smith, R.J.H.; Kuppachi, S.; Thomas, C.P. Minimal Change Disease With Nephrotic Syndrome Associated With Coronavirus Disease 2019 After Apolipoprotein L1 Risk Variant Kidney Transplant: A Case Report. Transplant. Proc. 2020, 52, 2693–2697. [Google Scholar] [CrossRef] [PubMed]
- Shimada, M.; Araya, C.; Rivard, C.; Ishimoto, T.; Johnson, R.J.; Garin, E.H. Minimal change disease: A “two-hit” podocyte immune disorder? Pediatr. Nephrol. 2011, 26, 645–649. [Google Scholar] [CrossRef]
- Ishimoto, T.; Shimada, M.; Araya, C.E.; Huskey, J.; Garin, E.H.; Johnson, R.J. Minimal change disease: A CD80 podocytopathy? Semin. Nephrol. 2011, 31, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.R.; Lee, S.B.; Kim, I.Y.; Lee, D.W.; Rhee, H.; Seong, E.Y.; Song, S.H.; Kwak, I.S. Relapse of minimal change disease following infection with the 2009 pandemic influenza (H1N1) virus. Clin. Exp. Nephrol. 2012, 16, 329–332. [Google Scholar] [CrossRef]
- Bomback, A.S.; Appel, G.B. Pathogenesis of the C3 glomerulopathies and reclassification of MPGN. Nat. Rev. Nephrol. 2012, 8, 634–642. [Google Scholar] [CrossRef] [PubMed]
- Lebreton, C.; Bacchetta, J.; Dijoud, F.; Bessenay, L.; Fremeaux-Bacchi, V.; Sellier-Leclerc, A.L. C3 glomerulopathy and eculizumab: A report on four paediatric cases. Pediatr. Nephrol. 2017, 32, 1023–1028. [Google Scholar] [CrossRef]
- Servais, A.; Noel, L.H.; Roumenina, L.T.; Le Quintrec, M.; Ngo, S.; Dragon-Durey, M.A.; Macher, M.A.; Zuber, J.; Karras, A.; Provot, F.; et al. Acquired and genetic complement abnormalities play a critical role in dense deposit disease and other C3 glomerulopathies. Kidney Int. 2012, 82, 454–464. [Google Scholar] [CrossRef] [Green Version]
- Castagnoli, R.; Votto, M.; Licari, A.; Brambilla, I.; Bruno, R.; Perlini, S.; Rovida, F.; Baldanti, F.; Marseglia, G.L. Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection in Children and Adolescents: A Systematic Review. JAMA Pediatr. 2020, 174, 882–889. [Google Scholar] [CrossRef] [Green Version]
- de Souza, T.H.; Nadal, J.A.; Nogueira, R.J.N.; Pereira, R.M.; Brandao, M.B. Clinical manifestations of children with COVID-19: A systematic review. Pediatr. Pulmonol. 2020, 55, 1892–1899. [Google Scholar] [CrossRef]
- Panahi, L.; Amiri, M.; Pouy, S. Clinical Characteristics of COVID-19 Infection in Newborns and Pediatrics: A Systematic Review. Arch. Acad. Emerg. Med. 2020, 8, e50. [Google Scholar] [PubMed]
- Mustafa, N.M.; Selim, L.A. Characterisation of COVID-19 Pandemic in Paediatric Age Group: A Systematic Review and Meta-Analysis. J. Clin. Virol. 2020, 128, 104395. [Google Scholar] [CrossRef]
- Hoang, A.; Chorath, K.; Moreira, A.; Evans, M.; Burmeister-Morton, F.; Burmeister, F.; Naqvi, R.; Petershack, M.; Moreira, A. COVID-19 in 7780 pediatric patients: A systematic review. EClinicalMedicine 2020, 24, 100433. [Google Scholar] [CrossRef] [PubMed]
- Mastrangelo, A.; Morello, W.; Vidal, E.; Guzzo, I.; Annicchiarico Petruzzelli, L.; Benetti, E.; Materassi, M.; Giordano, M.; Pasini, A.; Corrado, C.; et al. Impact of COVID-19 Pandemic in Children with CKD or Immunosuppression. Clin. J. Am. Soc. Nephrol. 2021, 16, 449–451. [Google Scholar] [CrossRef]
- Nicastro, E.; Verdoni, L.; Bettini, L.R.; Zuin, G.; Balduzzi, A.; Montini, G.; Biondi, A.; D'Antiga, L. COVID-19 in Immunosuppressed Children. Front. Pediatr. 2021, 9, 629240. [Google Scholar] [CrossRef]
- Liakopoulos, V.; Roumeliotis, S.; Papachristou, S.; Papanas, N. COVID-19 and the kidney: Time to take a closer look. Int. Urol. Nephrol. 2021. [Google Scholar] [CrossRef]
- Bibbo, C. Reconstruction of COVID-19-Related Compartment Syndrome With Massive Soft Tissue Necrosis. Wounds 2021, 33, 99–105. [Google Scholar] [PubMed]
- Maritati, F.; Moretti, M.I.; Nastasi, V.; Mazzucchelli, R.; Morroni, M.; Bagnarelli, P.; Rupoli, S.; Tavio, M.; Galiotta, P.; Bisello, W.; et al. ANCA-Associated Glomerulonephritis and Anti-Phospholipid Syndrome in a Patient with SARS-CoV-2 Infection: Just a Coincidence? Case Rep. Nephrol. Dial. 2021, 11, 214–220. [Google Scholar] [CrossRef]
- Miao, J.; Fidler, M.E.; Nasr, S.H.; Larsen, C.P.; Zoghby, Z.M. Membranous nephropathy in a patient with coronavirus disease 2019 (COVID-19): A case report. Clin. Nephrol. Case Stud. 2021, 9, 11–18. [Google Scholar] [CrossRef]
- Tarragon, B.; Valdenebro, M.; Serrano, M.L.; Maroto, A.; Llopez-Carratala, M.R.; Ramos, A.; Rubio, E.; Huerta, A.; Marques, M.; Portoles, J. Acute kidney failure in patients admitted due to COVID-19. Nefrologia 2021, 41, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Chueh, T.I.; Zheng, C.M.; Hou, Y.C.; Lu, K.C. Novel Evidence of Acute Kidney Injury in COVID-19. J. Clin. Med. 2020, 9, 3547. [Google Scholar] [CrossRef] [PubMed]
- Taghizadieh, A.; Mikaeili, H.; Ahmadi, M.; Valizadeh, H. Acute kidney injury in pregnant women following SARS-CoV-2 infection: A case report from Iran. Respir Med. Case Rep. 2020, 30, 101090. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, S.X.; Zhao, L.F.; Kong, D.L.; Guo, Z.Y. Management recommendations for patients with chronic kidney disease during the novel coronavirus disease 2019 (COVID-19) epidemic. Chronic. Dis. Transl. Med. 2020, 6, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Basic-Jukic, N.; Coric, M.; Bulimbasic, S.; Dika, Z.; Juric, I.; Furic-Cunko, V.; Katalinic, L.; Kos, J.; Fistrek, M.; Kastelan, Z.; et al. Histopathologic findings on indication renal allograft biopsies after recovery from acute COVID-19. Clin. Transplant. 2021, 35, e14486. [Google Scholar] [CrossRef] [PubMed]
- Akalin, E.; Azzi, Y.; Bartash, R.; Seethamraju, H.; Parides, M.; Hemmige, V.; Ross, M.; Forest, S.; Goldstein, Y.D.; Ajaimy, M.; et al. Covid-19 and Kidney Transplantation. N. Engl. J. Med. 2020, 382, 2475–2477. [Google Scholar] [CrossRef] [PubMed]
- Daniel, E.; Sekulic, M.; Kudose, S.; Kubin, C.; Ye, X.; Shayan, K.; Patel, A.; Cohen, D.J.; ERatner, L.; Santoriello, D.; et al. Kidney allograft biopsy findings after COVID-19. Am. J. Transplant. 2021, 21, 4032–4042. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, A.Z.; Naicker, S.; Winkler, C.A.; Kopp, J.B. HIV-associated nephropathies: Epidemiology, pathology, mechanisms and treatment. Nat. Rev. Nephrol. 2015, 11, 150–160. [Google Scholar] [CrossRef]
- Velez, J.C.Q.; Caza, T.; Larsen, C.P. COVAN is the new HIVAN: The re-emergence of collapsing glomerulopathy with COVID-19. Nat. Rev. Nephrol. 2020, 16, 565–567. [Google Scholar] [CrossRef]
| Pathology Features | Reference |
|---|---|
| Collapsing glomerulopathy (now defined as COVID-19–associated nephropathy (COVAN)) | [56,57,58,61,62,64,66,69,70,71,72,73,74,75,76,77,78,79,80,81,82] |
| Acute tubular necrosis | [58,64,66,69,71,72,76,80,82,83,84] |
| IgA nephropathy | [66,69,81,85,86,87] |
| Thrombotic microangiopathy | [58,64,66,67,69] |
| Crescentic glomerulonephritis | [58,66,69,88,89] |
| Non collapsing FSGS | [64,66,69,84] |
| Minimal change disease | [64,69,71,80] |
| Membranous nephropathy | [66,69,71,81] |
| Anti-glomerular basement membrane disease | [69,71,90,91] |
| Diabetic nephropathy | [64,69,81] |
| Myoglobin cast nephropathy | [58,69] |
| Post-infectious glomerulonephritis | [64,69] |
| Antibody-mediated rejection | [64,69] |
| Light chain cast nephropathy | [66,69] |
| Calcineurin inhibitor nephrotoxicity | [66] |
| Amyloidosis | [66,69] |
| Cortical infarction | [69,71] |
| Lupus nephritis | [69,71] |
| T-cell mediated rejection | [69,71] |
| Acute interstitial nephritis | [69,91] |
| Oxalate nephropathy | [92] |
| Granulomatous tubulointerstitial nephritis | [93] |
| Membranoproliferative glomerulonephritis | [94] |
| Cases | Sex/Age | Risk Factor/Clinical History | Clinical Background on Admission * | Kidney Impairment | Laboratory Data § | Analysis Performed | Pathology Features |
|---|---|---|---|---|---|---|---|
| Case 1 | M/73 | Kidney transplant; hypertension; metabolic syndrome | Elevated fever | AKI | Creatininemia: 4.0 mg/dL Proteinuria: 0.64–1 gr/die | LM; IHC; IF; EM | ATN |
| Case 2 | F/77 | Hypertension; obesity; rheumatoid arthritis | Pneumonia | AKI | Creatininemia: 3.2 mg/dL Proteinuria: 0.17 gr/die | LM; IF; EM | ATN and TMA |
| Case 3 | F/61 | Hypertension; obesity; type 2 diabetes | Acute respiratory distress syndrome | Nephrotic syndrome | Proteinuria: 14 gr/die Albuminemia: 2.2 g/dL | LM; IF; EM | CG |
| Case 4 | M/45 | Hypertension; type 2 diabetes | Fatigue; dyspnea | Nephrotic syndrome | Proteinuria: 30 gr/die | LM; IF | ATN and CG |
| Case 5 | M/15 | None | Fever; fatigue; peripheral edema | Nephrotic syndrome | Proteinuria: 1.83 gr/die/1.73 m2 Albuminemia: 3.9 g/dL | LM; IF; EM | MCD |
| Case 6 | M/71 | Hypertension; obesity; | Pneumonia; peripheral edema | Nephrotic syndrome | Proteinuria: 15.3 gr/die Albuminemia: 2.3 g/dL | LM; IF; EM | MCD and ATN |
| Case 7 | F/80 | Hypertension | Peripheral edema | Nephrotic syndrome | Proteinuria: 3.8 gr/die Albuminemia: 2.5 g/dL | LM; IF; EM | MCD and ATN |
| Case 8 | M/64 | Hypertension | Peripheral edema | AKI and Nephrotic syndrome | Creatininemia: 2.8 mg/dL Proteinuria: 22 gr/die Albuminemia: 2.7 g/dL | LM; IF; EM | MCD |
| Case 9 | F/15 | None | Fever | Microhematuria; leukocyturia | Creatininemia: 1.1 mg/dL eGFR 60 mL/min/1.73 m2 Proteinuria: 1.9 gr/die/1.73 m2 Albuminemia: 3.7 g/dL ANCA negative | LM; IHC; IF; EM | C3G and ATN |
| Cases | Histological Findings | IF Pattern | EM Findings— Percentage of Foot Process Effacement (FPE) |
|---|---|---|---|
| Case 5 | No evident injury | Unspecific faint mesangial IgM positivity | 80% |
| Case 6 | Focal mild ischemic global glomerulosclerosis age-correlated and occasional nuclear tubular atypia | Negative | 77% |
| Case 7 | Minimal and focal glomerular basement membrane wrinkling and occasional nuclear tubular atypia | Negative | 75% |
| Case 8 | No evident injury | Negative | 70–75% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gambella, A.; Barreca, A.; Biancone, L.; Roccatello, D.; Peruzzi, L.; Besso, L.; Licata, C.; Attanasio, A.; Papotti, M.; Cassoni, P. Spectrum of Kidney Injury Following COVID-19 Disease: Renal Biopsy Findings in a Single Italian Pathology Service. Biomolecules 2022, 12, 298. https://doi.org/10.3390/biom12020298
Gambella A, Barreca A, Biancone L, Roccatello D, Peruzzi L, Besso L, Licata C, Attanasio A, Papotti M, Cassoni P. Spectrum of Kidney Injury Following COVID-19 Disease: Renal Biopsy Findings in a Single Italian Pathology Service. Biomolecules. 2022; 12(2):298. https://doi.org/10.3390/biom12020298
Chicago/Turabian StyleGambella, Alessandro, Antonella Barreca, Luigi Biancone, Dario Roccatello, Licia Peruzzi, Luca Besso, Carolina Licata, Angelo Attanasio, Mauro Papotti, and Paola Cassoni. 2022. "Spectrum of Kidney Injury Following COVID-19 Disease: Renal Biopsy Findings in a Single Italian Pathology Service" Biomolecules 12, no. 2: 298. https://doi.org/10.3390/biom12020298
APA StyleGambella, A., Barreca, A., Biancone, L., Roccatello, D., Peruzzi, L., Besso, L., Licata, C., Attanasio, A., Papotti, M., & Cassoni, P. (2022). Spectrum of Kidney Injury Following COVID-19 Disease: Renal Biopsy Findings in a Single Italian Pathology Service. Biomolecules, 12(2), 298. https://doi.org/10.3390/biom12020298







