Small Non-Coding RNAs as New Biomarkers to Evaluate the Quality of the Embryo in the IVF Process
Abstract
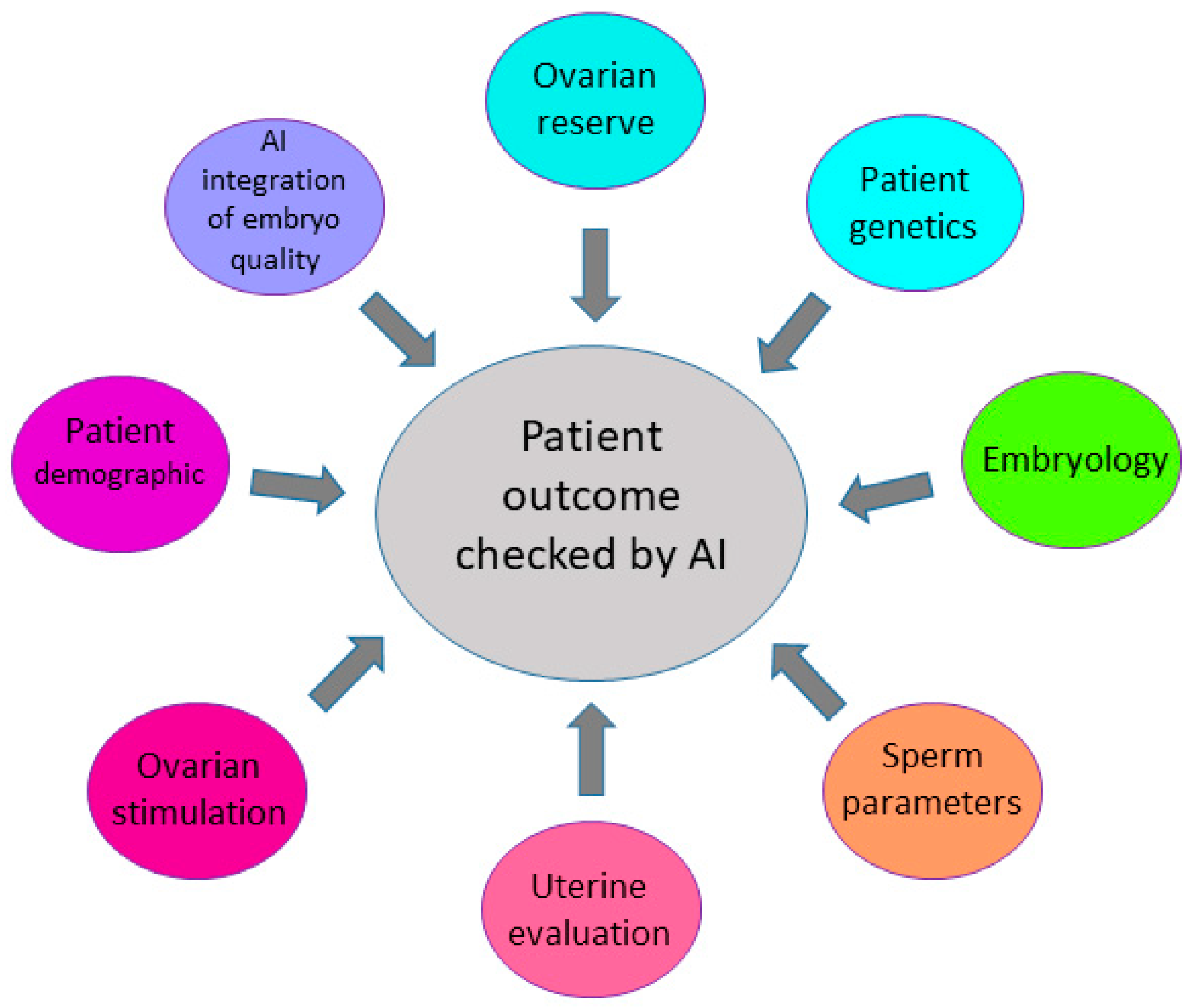
1. Introduction
2. Materials and Methods
2.1. Dataset Description
2.2. SncRNAs Sequencing
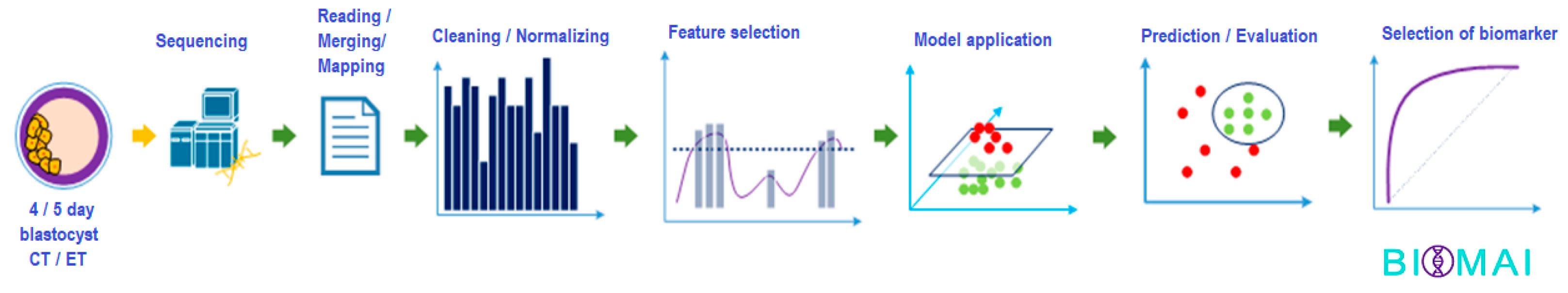
2.3. BioMAI Pipeline Predictor Model
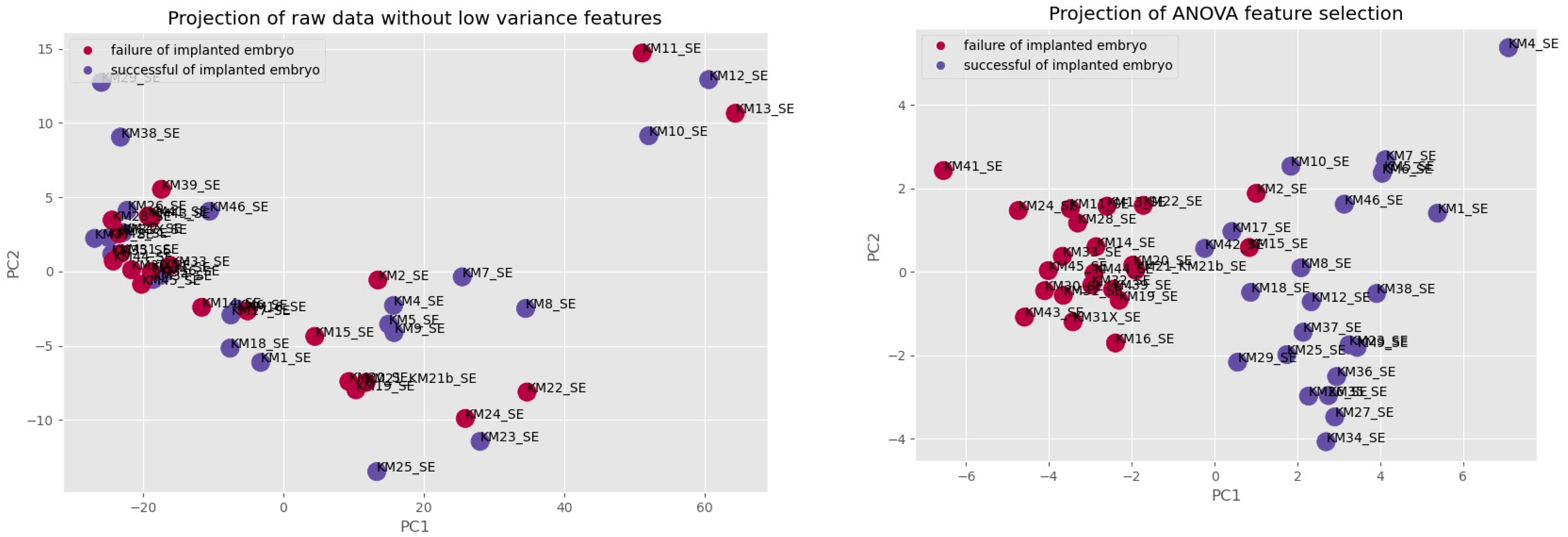
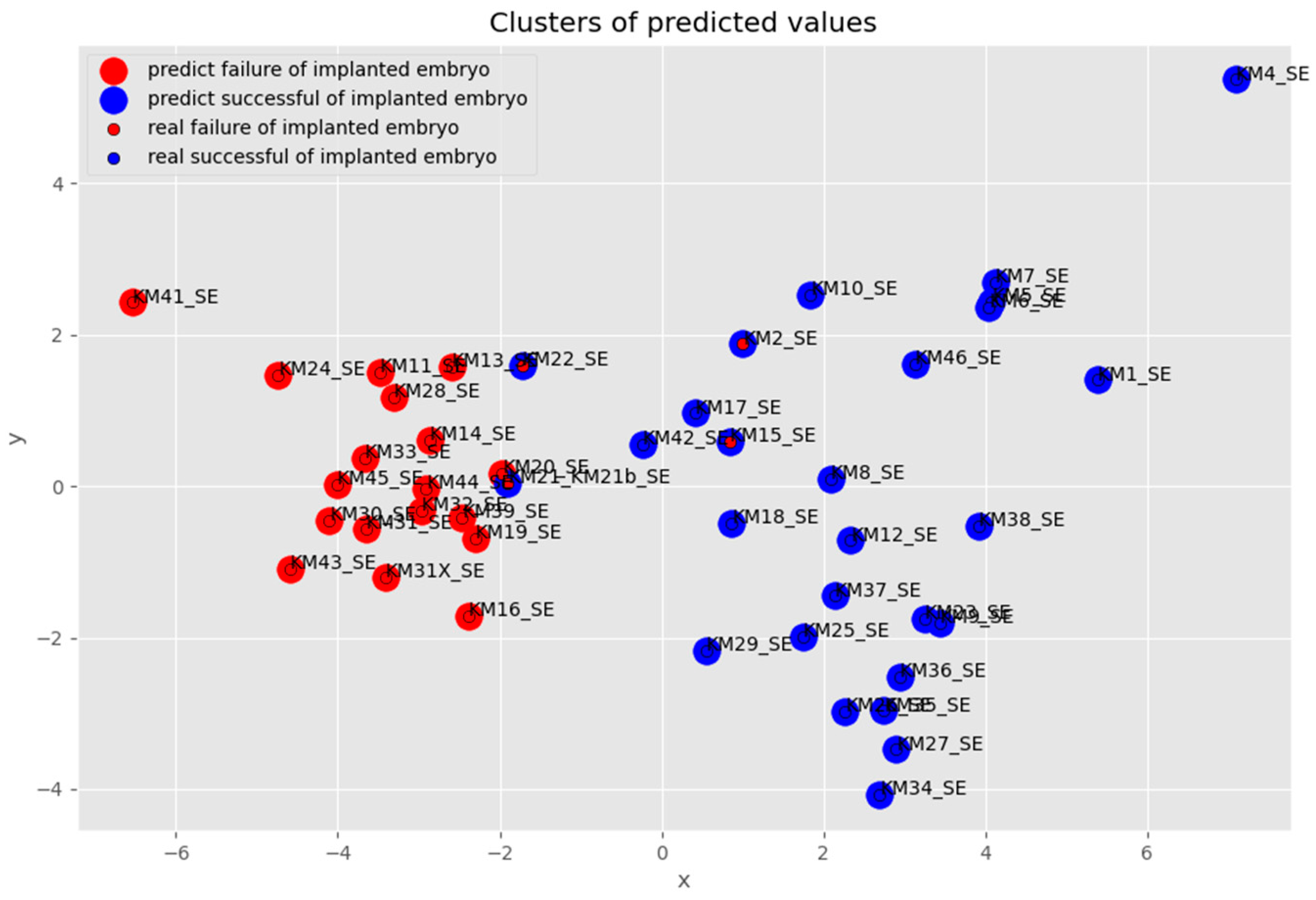
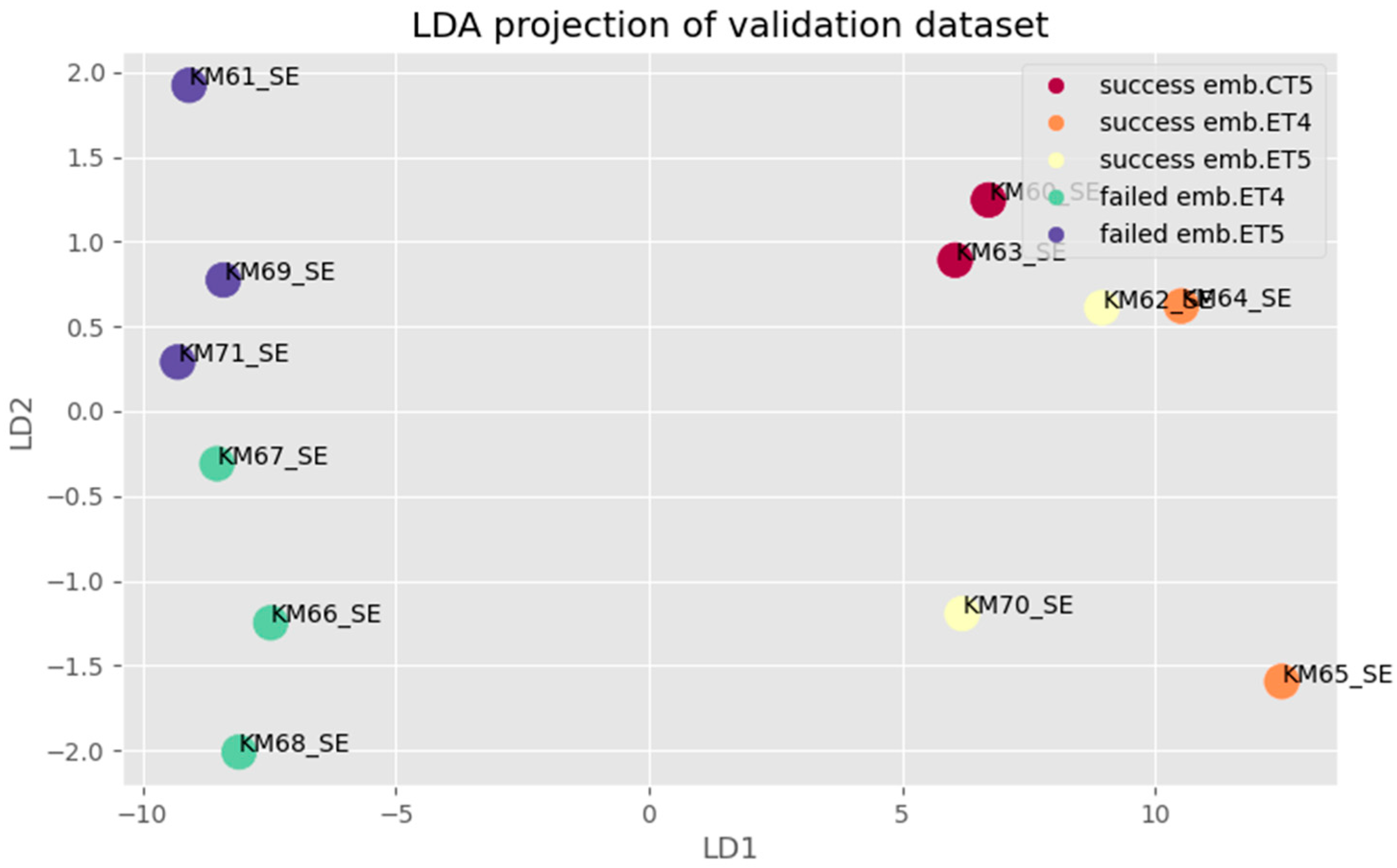
3. Results
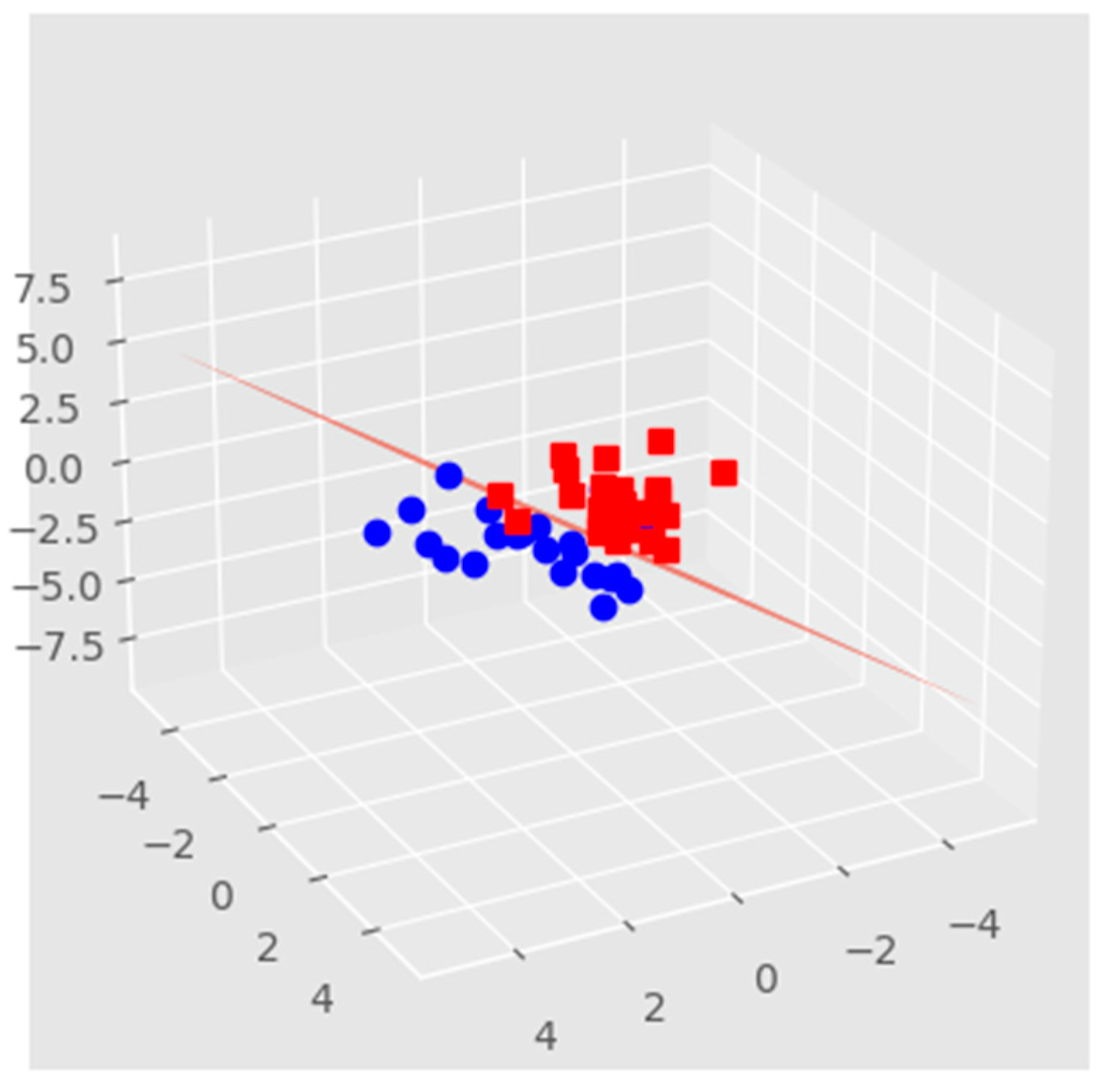
3.1. Ensemble Learning
3.2. Voting Technique
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Petraglia, F.; Serour, G.I.; Chapron, C. The changing prevalence of infertility. Int. J. Gynecol. Obstet. 2013, 123, S4–S8. [Google Scholar] [CrossRef] [PubMed]
- Vander Borght, M.; Wyns, C. Fertility and infertility: Definition and epidemiology. Clin. Biochem. 2018, 62, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Focus on Reproduction. ‘Dramatic’ Decline in Worldwide Total Fertility Rates Predicted. Available online: https://www.focusonreproduction.eu/article/News-in-Reproduction-Population?fbclid=IwAR3R3Icjm-LQC8zPLAKBDTLdLxCHKGAOqI1sYBe1eJnEvaAgSf2bUCoTkZ0 (accessed on 12 April 2022).
- Toporcerová, S.; Špaková, I.; Mareková, M.; Rabajdová, M. Biotechnology in Healthcare, 1st ed.; Barh, D., Ed.; Elsevier: London, UK, 2022; Volume 2. [Google Scholar]
- Ehsani, M.; Mohammadnia-Afrouzi, M.; Mirzakhami, M.; Esmaeilzadeh, S.; Shahbazi, M. Female Unexplained Infertility: A Disease with Imbalanced Adaptive Immunity. J. Hum. Reprod Sci 2019, 12, 274–282. [Google Scholar] [CrossRef] [PubMed]
- Bashiri, A.; Halper, K.I.; Orvieto, R. Recurrent Implantation Failure-update overview on etiology, diagnosis, treatment and future directions. Reprod. Biol. Endocrinol. 2018, 16, 121. [Google Scholar] [CrossRef]
- Rabajdová, M.; Urban, P.; Biščáková, Z.; Urdzík, P.; Mareková, M. Endometrial receptivity—A new challenge of infertility diagnostics in the In Vitro fertilization process. Lek. Obz. 2019, 68, 95–98. [Google Scholar]
- Li, L.; Wang, P.; Liu, S.; Bai, X.; Zou, B.; Li, Y. Transcriptome sequencing of endometrium revealed alterations in mRNAs and lncRNAs after ovarian stimulation. J. Assist. Reprod. Genet. 2020, 37, 21–32. [Google Scholar] [CrossRef]
- Ferretti, C.; Bruni, L.; Dangles-Marie, V.; Pecking, A.; Bellet, D. Molecular circuits shared by placental and cancer cells, and their implications in the proliferative, invasive and migratory capacities of trophoblasts. Hum. Reprod. Updat. 2007, 13, 121–141. [Google Scholar] [CrossRef]
- Guo, X.; Yi, H.; Li, T.; Wang, Y.; Wang, H.; Chen, X. Role of Vascular Endothelial Growth Factor (VEGF) in Human Embryo Implantation: Clinical Implications. Biomolecules 2021, 11, 253. [Google Scholar] [CrossRef]
- Torry, D.S.; Leavenworth, J.; Chang, M.; Maheshwari, V.; Groesch, K.; Ball, E.R.; Torry, R. Angiogenesis in implantation. J. Assist. Reprod. Genet. 2007, 24, 303–315. [Google Scholar] [CrossRef]
- Varas-Godoy, M.; Acuña-Gallardo, S.; Venegas-Duarte, S.; Hill, C.; Caceres-Verschae, A.; Realini, O.; Monteiro, L.J.; Zavala, G.; Khoury, M.; Romero, R.; et al. Angiogenic Properties of Menstrual Stem Cells Are Impaired in Women with a History of Preeclampsia. Stem Cells Int. 2019, 2019, 1916542. [Google Scholar] [CrossRef]
- Rosenbluth, E.M.; Shelton, D.N.; Wells, L.M.; Sparks, A.E.; Van Voorhis, B.J. Human embryos secrete microRNAs into culture media—A potential biomarker for implantation. Fertil. Steril. 2014, 101, 1493–1500. [Google Scholar] [CrossRef] [PubMed]
- Ochoa-Bernal, M.A.; Fazleabas, A.T. Physiologic Events of Embryo Implantation and Decidualization in Human and Non-Human Primates. Int. J. Mol. Sci. 2020, 21, 1973. [Google Scholar] [CrossRef] [PubMed]
- Capalbo, A.; Ubaldi, F.M.; Cimadomo, D.; Noli, L.; Khalaf, Y.; Farcomeni, A.; Ilic, D.; Rienzi, L. MicroRNAs in spent blastocyst culture medium are derived from trophectoderm cells and can be explored for human embryo reproductive competence assessment. Fertil. Steril. 2015, 105, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Rødgaard, T.; Heegaard, P.M.; Callesen, H. Non-invasive assessment of in-vitro embryo quality to improve transfer success. Reprod. Biomed. Online 2015, 31, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Timofeeva, A.; Drapkina, Y.; Fedorov, I.; Chagovets, V.; Makarova, N.; Shamina, M.; Kalinina, E.; Sukhikh, G. Small Noncoding RNA Signatures for Determining the Developmental Potential of an Embryo at the Morula Stage. Int. J. Mol. Sci. 2020, 21, 9399. [Google Scholar] [CrossRef] [PubMed]
- Cimadomo, D.; Rienzi, L.; Giancani, A.; Alviggi, E.; Dusi, L.; Canipari, R.; Noli, L.; Ilic, D.; Khalaf, Y.; Ubaldi, F.M.; et al. Definition and validation of a custom protocol to detect miRNAs in the spent media after blastocyst culture: Searching for biomarkers of implantation. Hum. Reprod. 2019, 34, 1746–1761. [Google Scholar] [CrossRef]
- Cuman, C.; Van Sinderen, M.; Gantier, M.P.; Rainczuk, K.; Sorby, K.; Rombauts, L.; Osianlis, T.; Dimitriadis, E. Human Blastocyst Secreted microRNA Regulate Endometrial Epithelial Cell Adhesion. eBioMedicine 2015, 2, 1528–1535. [Google Scholar] [CrossRef]
- Borges, E., Jr.; Setti, A.S.; Braga, D.P.A.F.; Geraldo, M.V.; Figueira, R.d.C.S.; Iaconelli, A., Jr. miR-142-3p as a biomarker of blastocyst implantation failure—A pilot study. JBRA Assist. Reprod. 2016, 20, 200–205. [Google Scholar] [CrossRef]
- Abu-Halima, M.; Häusler, S.; Backes, C.; Fehlmann, T.; Staib, C.; Nestel, S.; Nazarenko, I.; Meese, E.; Keller, A. Micro-ribonucleic acids and extracellular vesicles repertoire in the spent culture media is altered in women undergoing In Vitro Fertilization. Sci. Rep. 2017, 7, 13525. [Google Scholar] [CrossRef]
- Kim, J.; Lee, J.; Jun, J.H. Identification of differentially expressed microRNAs in outgrowth embryos compared with blastocysts and non-outgrowth embryos in mice. Reprod. Fertil. Dev. 2019, 31, 645–657. [Google Scholar] [CrossRef]
- Timofeeva, A.V.; Chagovets, V.V.; Drapkina, Y.S.; Makarova, N.P.; Kalinina, E.A.; Sukhikh, G.T. Cell-Free, Embryo-Specific sncRNA as a Molecular Biological Bridge between Patient Fertility and IVF Efficiency. Int. J. Mol. Sci. 2019, 20, 2912. [Google Scholar] [CrossRef] [PubMed]
- Cimadomo, D.; Capalbo, A.; Ubaldi, F.M.; Scarica, C.; Palagiano, A.; Canipari, R.; Rienzi, L. The Impact of Biopsy on Human Embryo Developmental Potential during Preimplantation Genetic Diagnosis. BioMed Res. Int. 2016, 2016, 7193075. [Google Scholar] [CrossRef] [PubMed]
- Manna, C.; Nanni, L.; Lumini, A.; Pappalardo, S. Artificial intelligence techniques for embryo and oocyte classification. Reprod. Biomed. Online 2013, 26, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, E.I.; Ferreira, A.S.; Cecílio, M.H.M.; Chéles, D.; De Souza, R.C.M.; Nogueira, M.F.G.; Rocha, J.C. Artificial intelligence in the IVF laboratory: Overview through the application of different types of algorithms for the classification of reproductive data. J. Assist. Reprod. Genet. 2020, 37, 2359–2376. [Google Scholar] [CrossRef] [PubMed]
- Raimundo, J.; Cabrita, P. Artificial intelligence at assisted reproductive technology. Procedia Comput. Sci. 2021, 181, 442–447. [Google Scholar] [CrossRef]
- Zhao, Y.; Sun, H.; Feng, M.; Zhao, J.; Zhao, X.; Wan, Q.; Cai, D. Metformin is associated with reduced cell proliferation in human endometrial cancer by inbibiting PI3K/AKT/mTOR signaling. Gynecol. Endocrinol. 2017, 34, 428–432. [Google Scholar] [CrossRef]
- Amjadi, F.S.; Salehi, E.; Zandieh, Z.; Rashidi, M.; Taleahmad, S.; Masrour, M.J.; Aflatoonian, R.; Mehdizadeh, M. Comparative evaluation of NOTCH signaling molecules in the endometrium of women with various gynecological diseases during the window of implantation. Iran. J. Basic. Med. Sci. 2019, 22, 426–431. [Google Scholar] [CrossRef]
- Ashary, N.; Tiwari, A.; Modi, D. Embryo Implantation: War in Times of Love. Endocrinology 2018, 159, 1188–1198. [Google Scholar] [CrossRef]
- Zaninovic, N.; Rosenwaks, Z. Artificial intelligence in human in vitro fertilization and embryology. Fertil. Steril. 2020, 114, 914–920. [Google Scholar] [CrossRef]
- Nasiri, N.; Eftekhari-Yazdi, P. An Overview of The Available Methods for Morphological Scoring of Pre-Implantation Embryos in In Vitro Fertilization. Cell J. 2015, 16, 392–405. [Google Scholar] [CrossRef]
- Milewski, R.; Ajduk, A. Time-lapse imaging of cleavage divisions in embryo quality assessment. Reproduction 2017, 154, R37–R53. [Google Scholar] [CrossRef] [PubMed]
- Irani, M.; Reichman, D.; Robles, A.; Melnick, A.; Davis, O.; Zaninovic, N.; Xu, K.; Rosenwaks, Z. Morphologic grading of euploid blastocysts influences implantation and ongoing pregnancy rates. Fertil. Steril. 2017, 107, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Gardner, D.K.; Lane, M.; Stevens, J.; Schlenker, T.; Schoolcraft, W.B. Blastocyst score affects implantation and pregnancy outcome: Towards a single blastocyst transfer. Fertil. Steril. 2000, 73, 1155–1158. [Google Scholar] [CrossRef]
- Moayeri, M.; Saeidi, H.; Modarresi, M.H.; Hashemi, M. The Effect of Preimplantation Genetic Screening on Implantation Rate in Women over 35 Years of Age. Cell J. 2016, 18, 13–20. [Google Scholar] [CrossRef]
- Qiu, J.; Li, P.; Dong, M.; Xin, X.; Tan, J. Personalized prediction of live birth prior to the first in vitro fertilization treatment: A machine learning method. J. Transl. Med. 2019, 17, 317. [Google Scholar] [CrossRef]
- Liu, R.; Bai, S.; Jiang, X.; Luo, L.; Tong, X.; Zheng, S.; Wang, Y.; Xu, B. Multifactor Prediction of Embryo Transfer Outcomes Based on a Machine Learning Algorithm. Front. Endocrinol. 2021, 12, 745039. [Google Scholar] [CrossRef]
- Xi, Q.; Yang, Q.; Wang, M.; Huang, B.; Zhang, B.; Li, Z.; Liu, S.; Yang, L.; Zhu, L.; Jin, L. Individualized embryo selection strategy developed by stacking machine learning model for better in vitro fertilization outcomes: An application study. Reprod. Biol. Endocrinol. 2021, 19, 53. [Google Scholar] [CrossRef]
- Khosravi, P.; Kazemi, E.; Zhan, Q.; Malmsten, J.E.; Toschi, M.; Zisimopoulos, P.; Sigaras, A.; Lavery, S.; Cooper, L.A.D.; Hickman, C.; et al. Deep learning enables robust assessment and selection of human blastocysts after in vitro fertilization. NPJ Digit. Med. 2019, 2, 21. [Google Scholar] [CrossRef]
- Liang, P.; Yang, W.; Chen, X.; Long, C.; Zheng, L.; Li, H.; Zuo, Y. Machine Learning of Single-Cell Transcriptome Highly Identifies mRNA Signature by Comparing F-Score Selection with DGE Analysis. Mol. Ther. Nucleic Acids 2020, 20, 155–163. [Google Scholar] [CrossRef]
- Yang, Q.; Li, R.; Lyu, Q.; Hou, L.; Liu, Z.; Sun, Q.; Liu, M.; Shi, H.; Xu, B.; Yin, M.; et al. Single-cell CAS-seq reveals a class of short PIWI-interacting RNAs in human oocytes. Nat. Commun. 2019, 10, 3389. [Google Scholar] [CrossRef]
- Ahmad, J.; Hasnain, S.E.; Siddiqui, M.A.; Ahamed, M.; Musarrat, J.; Al-Khedhairy, A.A. MicroRNA in Carcinogenesis & Cancer Diagnostics: A New Paradigm. Indian J. Med. Res. 2013, 137, 680–694. [Google Scholar] [PubMed]
- Ha, M.; Kim, V.N. Regulation of microRNA biogenesis. Nat. Rev. Mol. Cell Biol. 2014, 15, 509–524. [Google Scholar] [CrossRef] [PubMed]
- Ding, D.; Liu, J.; Dong, K.; Midic, U.; Hess, R.; Xie, H.; Demireva, E.Y.; Chen, C. PNLDC1 is essential for piRNA 3′ end trimming and transposon silencing during spermatogenesis in mice. Nat. Commun. 2017, 8, 819. [Google Scholar] [CrossRef] [PubMed]
- Battaglia, R.; Musumeci, P.; Ragusa, M.; Barbagallo, D.; Scalia, M.; Zimbone, M.; Faro, J.M.L.; Borzì, P.; Scollo, P.; Purrello, M.; et al. Ovarian aging increases small extracellular vesicle CD81+ release in human follicular fluid and influences miRNA profiles. Aging 2020, 12, 12324–12341. [Google Scholar] [CrossRef] [PubMed]
- Hocaoglu, M.; Demirer, S.; Karaalp, I.L.; Kaynak, E.; Attar, E.; Turgut, A.; Karateke, A.; Komurcu-Bayrak, E. Identification of miR-16-5p and miR-155-5p microRNAs differentially expressed in circulating leukocytes of pregnant women with polycystic ovary syndrome and gestational diabetes. Gynecol. Endocrinol. 2021, 37, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Tagliaferri, S.; Cepparulo, P.; Vinciguerra, A.; Campanile, M.; Esposito, G.; Maruotti, G.M.; Zullo, F.; Annunziato, L.; Pignataro, G. miR-16-5p, miR-103-3p, and miR-27b-3p as Early Peripheral Biomarkers of Fetal Growth Restriction. Front. Pediatr. 2021, 9, 611112. [Google Scholar] [CrossRef]
- Russell, S.J.; Menezes, K.; Balakier, H.; Librach, C. Comprehensive profiling of Small RNAs in human embryo-conditioned culture media by improved sequencing and quantitative PCR methods. Syst. Biol. Reprod. Med. 2020, 66, 129–139. [Google Scholar] [CrossRef]
- Calleja-Agius, J.; Jauniaux, E.; Pizzey, A.; Muttukrishna, S. Investigation of systemic inflammatory response in first trimester pregnancy failure. Hum. Reprod. 2012, 27, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Zahedkalaei, A.T.; Kazemi, M.; Zolfaghari, P.; Rashidan, M.; Sohrabi, M.B. Association Between Urinary Tract Infection in the First Trimester and Risk of Preeclampsia: A Case–Control Study. Int. J. Womens Health 2020, 12, 521–526. [Google Scholar] [CrossRef]
- Giakoumelou, S.; Wheelhouse, N.; Cuschieri, K.; Entrican, G.; Howie, S.E.; Horne, A.W. The role of infection in miscarriage. Hum. Reprod. Update 2016, 22, 116–133. [Google Scholar] [CrossRef]
- Ibarra, A.; Vega-Guedes, B.; Brito-Casillas, Y.; Wägner, A.M. Diabetes in Pregnancy and MicroRNAs: Promises and Limitations in Their Clinical Application. Non Coding RNA 2018, 4, 32. [Google Scholar] [CrossRef] [PubMed]
- Winger, E.E.; Reed, J.L.; Ji, X. First-trimester maternal cell microRNA is a superior pregnancy marker to immunological testing for predicting adverse pregnancy outcome. J. Reprod. Immunol. 2015, 110, 22–35. [Google Scholar] [CrossRef] [PubMed]
- Kaczmarek, M.M.; Najmula, J.; Guzewska, M.M.; Przygrodzka, E. MiRNAs in the Peri-Implantation Period: Contribution to Embryo–Maternal Communication in Pigs. Int. J. Mol. Sci. 2020, 21, 2229. [Google Scholar] [CrossRef] [PubMed]
- Bahramy, A.; Zafari, N.; Izadi, P.; Soleymani, F.; Kavousi, S.; Noruzinia, M. The Role of miRNAs 340-5p, 92a-3p, and 381-3p in Patients with Endometriosis: A Plasma and Mesenchymal Stem-Like Cell Study. BioMed Res. Int. 2021, 2021, 1–15. [Google Scholar] [CrossRef]
- Lin, L.; Du, T.; Huang, J.; Huang, L.-L.; Yang, D.-Z. Identification of Differentially Expressed MicroRNAs in the Ovary of Polycystic Ovary Syndrome with Hyperandrogenism and Insulin Resistance. Chin. Med. J. 2015, 128, 169–174. [Google Scholar] [CrossRef]
- Zhong, C.; Gao, L.; Shu, L.; Hou, Z.; Cai, L.; Huang, J.; Liu, J.; Mao, Y. Analysis of IVF/ICSI Outcomes in Endometriosis Patients with Recurrent Implantation Failure: Influence on Cumulative Live Birth Rate. Front. Endocrinol. 2021, 12, 640288. [Google Scholar] [CrossRef]
- Butler, A.E.; Ramachandran, V.; Sathyapalan, T.; David, R.; Gooderham, N.; Benurwar, M.; Dargham, S.; Hayat, S.; Najafi-Shoushtari, S.H.; Atkin, S.L. microRNA Expression in Women with and without Polycystic Ovarian Syndrome Matched for Body Mass Index. Front. Endocrinol. 2020, 11, 206. [Google Scholar] [CrossRef]
- Gu, T.; Elgin, S.C.R. Maternal Depletion of Piwi, a Component of the RNAi System, Impacts Heterochromatin Formation in Drosophila. PLoS Genet. 2013, 9, e1003780. [Google Scholar] [CrossRef]
- Ding, D.; Chen, C. Cracking the egg: A breakthrough in piRNA function in mammalian oocytes and embryos. Biol. Reprod. 2022, 106, 6–8. [Google Scholar] [CrossRef]
- Russell, S.; Stalker, L.; LaMarre, J. PIWIs, piRNAs and Retrotransposons: Complex battles during reprogramming in gametes and early embryos. Reprod. Domest. Anim. 2017, 52, 28–38. [Google Scholar] [CrossRef]
- Kamalidehghan, B.; Habibi, M.; Afjeh, S.S.; Shoai, M.; Alidoost, S.; Ghale, R.A.; Eshghifar, N.; Pouresmaeili, F. The Importance of Small Non-Coding RNAs in Human Reproduction: A Review Article. Appl. Clin. Genet. 2020, 13, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Du, W.W.; Yang, W.; Xuan, J.; Gupta, S.; Krylov, S.N.; Ma, X.; Yang, Q.; Yang, B.B. Reciprocal regulation of miRNAs and piRNAs in embryonic development. Cell Death Differ. 2016, 23, 1458–1470. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, F.; Chen, Q.; Li, M.; Lv, X.; Xiao, Y.; Zhang, Z.; Hou, L.; Lai, Y.; Zhang, Y.; et al. The piRNA pathway is essential for generating functional oocytes in golden hamsters. Nature 2021, 23, 1013–1022. [Google Scholar] [CrossRef] [PubMed]
- Jia, S.-Z.; Yang, Y.; Lang, J.; Sun, P.; Leng, J. Plasma miR-17-5p, miR-20a and miR-22 are down-regulated in women with endometriosis. Hum. Reprod. 2013, 28, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Lau, N.L.; Ohsumi, T.; Borowsky, M.; Kingston, R.E.; Blower, M.D. Systematic and Single Cell Analysis of Xenopus Piwi-Interacting RNAs and Xiwi. EMBO J. 2009, 28, 2945–2958. [Google Scholar] [CrossRef] [PubMed]
- Kim, V.N. Small RNAs Just Got Bigger: Piwi-Interacting RNAs (PiRNAs) in Mammalian Testes. Genes Dev. 2006, 20, 1993–1997. [Google Scholar] [CrossRef]
- Gou, L.-T.; Kang, J.-Y.; Dai, P.; Wang, X.; Li, F.; Zhao, S.; Zhang, M.; Hua, M.-M.; Lu, Y.; Zhu, Y.; et al. Ubiquitination-Deficient Mutations in Human Piwi Cause Male Infertility by Impairing Histone-to-Protamine Exchange during Spermiogenesis. Cell 2017, 169, 1090–1104.e13. [Google Scholar] [CrossRef]



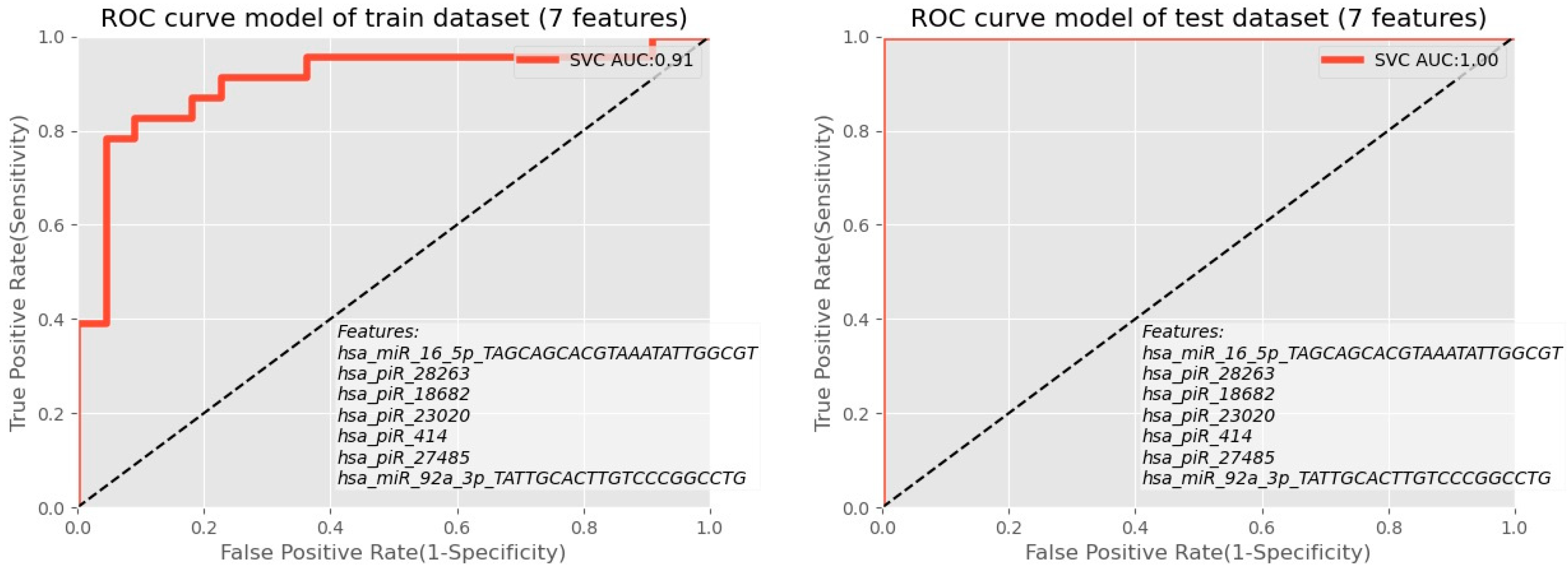

| F-Value ANOVA Coefficient of Importance | p-Value | |
|---|---|---|
| hsa_miR_16_5p | 8,853,048 | 0.004786 |
| hsa_piR_28263 | 6,154,501 | 0.017098 |
| hsa_piR_18682 | 5,506,564 | 0.023622 |
| hsa_piR_23020 | 5,341,872 | 0.025678 |
| hsa_piR_414 | 5,103,818 | 0.028998 |
| hsa_piR_27485 | 5,028,244 | 0.030147 |
| hsa_miR_92a_3p | 499,102 | 0.030731 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toporcerová, S.; Špaková, I.; Šoltys, K.; Klepcová, Z.; Kľoc, M.; Bohošová, J.; Trachtová, K.; Peterová, L.; Mičková, H.; Urdzík, P.; et al. Small Non-Coding RNAs as New Biomarkers to Evaluate the Quality of the Embryo in the IVF Process. Biomolecules 2022, 12, 1687. https://doi.org/10.3390/biom12111687
Toporcerová S, Špaková I, Šoltys K, Klepcová Z, Kľoc M, Bohošová J, Trachtová K, Peterová L, Mičková H, Urdzík P, et al. Small Non-Coding RNAs as New Biomarkers to Evaluate the Quality of the Embryo in the IVF Process. Biomolecules. 2022; 12(11):1687. https://doi.org/10.3390/biom12111687
Chicago/Turabian StyleToporcerová, Silvia, Ivana Špaková, Katarína Šoltys, Zuzana Klepcová, Marek Kľoc, Júlia Bohošová, Karolína Trachtová, Lucia Peterová, Helena Mičková, Peter Urdzík, and et al. 2022. "Small Non-Coding RNAs as New Biomarkers to Evaluate the Quality of the Embryo in the IVF Process" Biomolecules 12, no. 11: 1687. https://doi.org/10.3390/biom12111687
APA StyleToporcerová, S., Špaková, I., Šoltys, K., Klepcová, Z., Kľoc, M., Bohošová, J., Trachtová, K., Peterová, L., Mičková, H., Urdzík, P., Mareková, M., Slabý, O., & Rabajdová, M. (2022). Small Non-Coding RNAs as New Biomarkers to Evaluate the Quality of the Embryo in the IVF Process. Biomolecules, 12(11), 1687. https://doi.org/10.3390/biom12111687







