Potential Anticancer Activity of Pomegranate (Punica granatum L.) Fruits of Different Color: In Vitro and In Silico Evidence
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Pomegranates (PMG)
2.3. Chemical Analysis
2.4. Non-Phenolic Antioxidants
2.5. Phenolic Compounds (PC)
2.6. UPLC-DAD-MS2
2.7. Antioxidant Activity
2.8. Foodinformatics
2.9. In Vitro Cytotoxicity
2.10. Statistical Analysis
3. Results and Discussion
3.1. Chemical Profile of PMG Samples
3.2. Non-Phenolic Antioxidants
3.3. Phenolic Compounds (PC) and Organic Acids
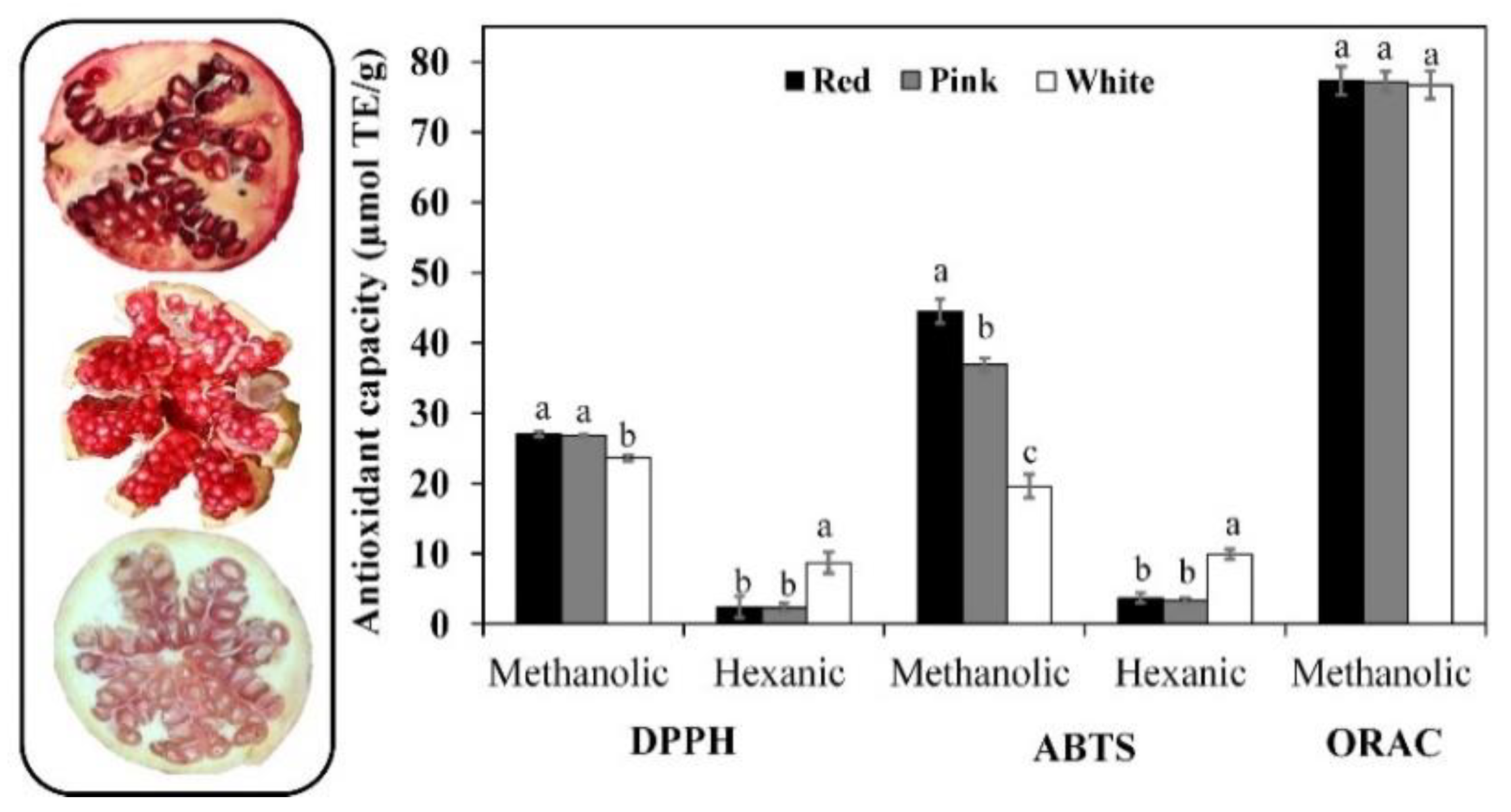
3.4. Antioxidant Capacity
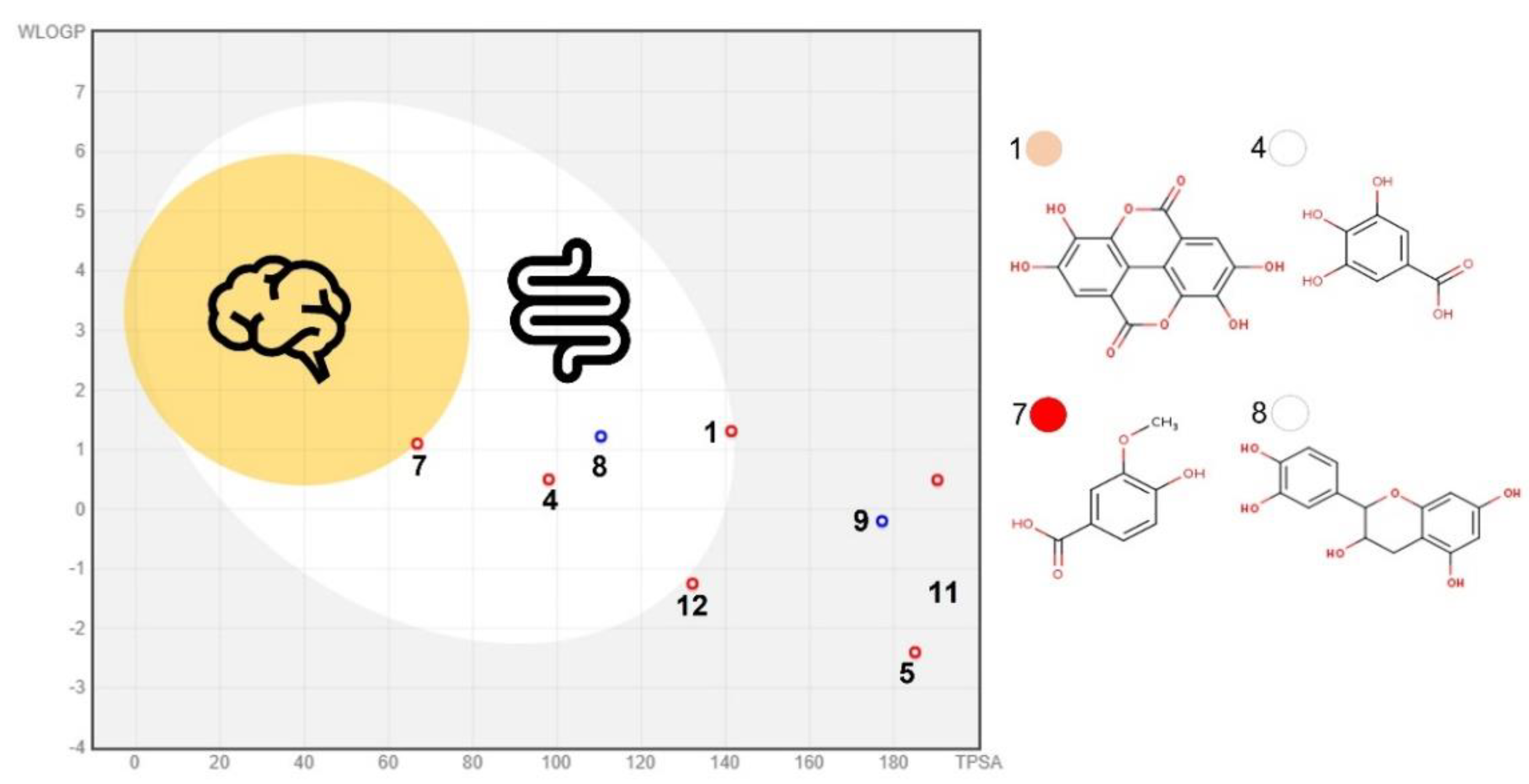
3.5. In Silico Gastrointestinal Fate Prediction
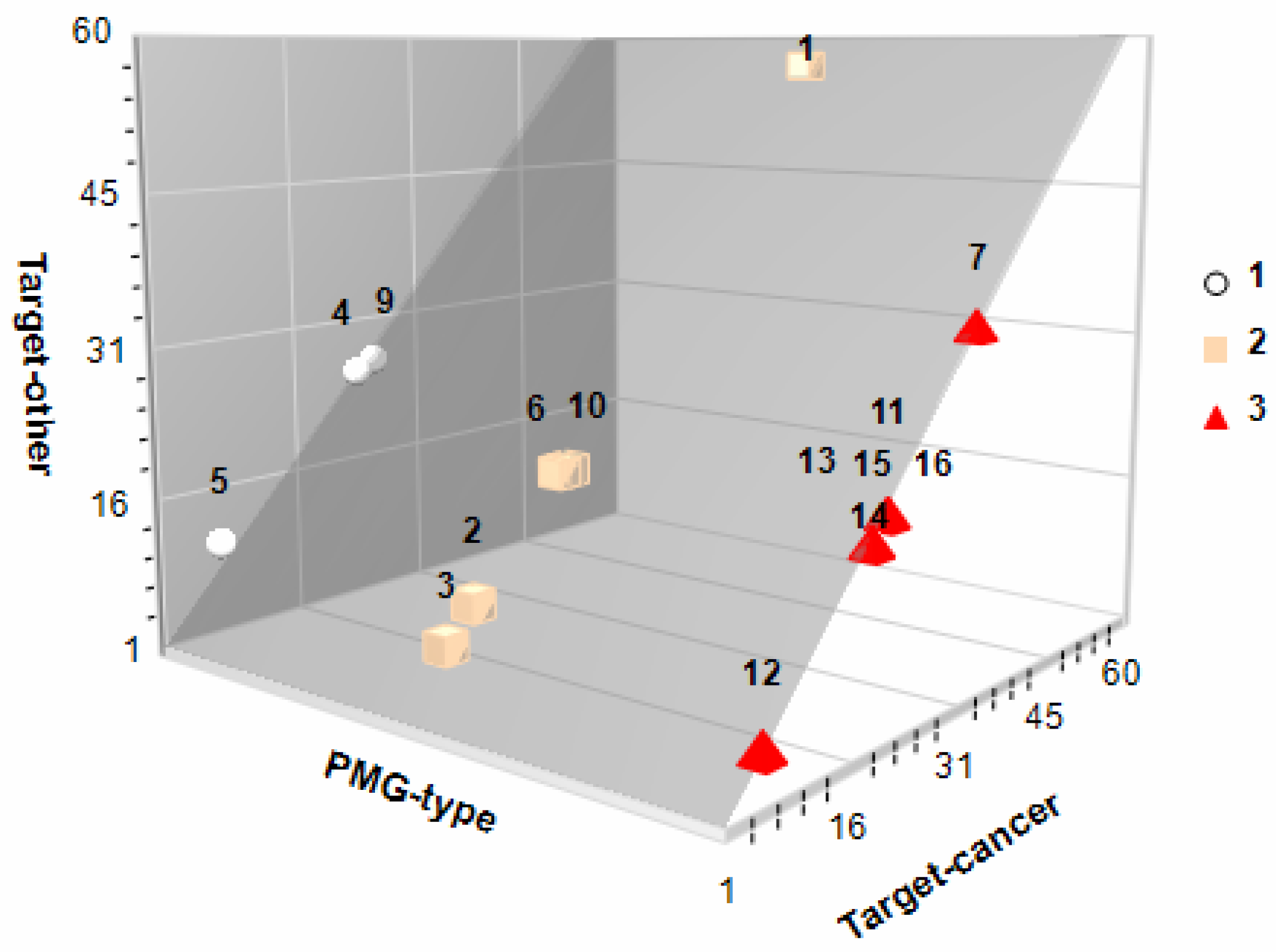
3.6. Protein-Targetting Prediction
3.7. In Vitro Citototoxicity Assays
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gopal, S.; Sharpless, N.E. Cancer as a global health priority. JAMA 2021, 326, 809–810. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Mattiuzzi, C.; Lippi, G. Current cancer epidemiology. J. Epidemiol. Glob. Health 2019, 9, 217–222. [Google Scholar] [CrossRef]
- Turanli, B.; Altay, O.; Borén, J.; Turkez, H.; Nielsen, J.; Uhlen, M.; Arga, K.Y.; Mardinoglu, A. Systems biology based drug repositioning for development of cancer therapy. In Seminars in Cancer Biology; Academic Press: Waltham, MA, USA, 2021; Volume 68, pp. 47–58. [Google Scholar] [CrossRef]
- Wall-Medrano, A.; Olivas-Aguirre, F.J. Antioxidant phytochemicals in cancer prevention and therapy—An update. In Functional Foods in Cancer Prevention and Therapy; Academic Press: Waltham, MA, USA, 2020; pp. 195–220. [Google Scholar] [CrossRef]
- Farvid, M.S.; Barnett, J.B.; Spence, N.D. Fruit and vegetable consumption and incident breast cancer: A systematic review and meta-analysis of prospective studies. Br. J. Cancer 2021, 125, 284–298. [Google Scholar] [CrossRef]
- Wigner, P.; Bijak, M.; Saluk-Bijak, J. Clinical potential of fruit in bladder cancer prevention and treatment. Nutrients 2022, 14, 1132. [Google Scholar] [CrossRef] [PubMed]
- Melgarejo-Sánchez, P.; Nunez-Gomez, D.; Martínez-Nicolás, J.J.; Hernández, F.; Legua, P.; Melgarejo, P. Pomegranate variety and pomegranate plant part, relevance from bioactive point of view: A review. Bioresour. Bioprocess. 2021, 8, 2. [Google Scholar] [CrossRef]
- Wong, T.L.; Strandberg, K.R.; Croley, C.R.; Fraser, S.E.; Venkata, K.C.N.; Fimognari, C.; Sethi, G.; Bishayee, A. Pomegranate bioactive constituents target multiple oncogenic and oncosuppressive signaling for cancer prevention and intervention. In Seminars in Cancer Biology; Academic Press: Waltham, MA, USA, 2021; Volume 73, pp. 265–293. [Google Scholar] [CrossRef]
- Sharma, K.; Kesharwani, P.; Prajapati, S.K.; Jain, A.; Jain, D.; Mody, N.; Sharma, S. An insight into anticancer bioactives from Punica granatum (Pomegranate). Anti-Cancer Agents Med. Chem. 2022, 22, 694–702. [Google Scholar] [CrossRef] [PubMed]
- Maphetu, N.; Unuofin, J.O.; Masuku, N.P.; Olisah, C.; Lebelo, S.L. Medicinal uses, pharmacological activities, phytochemistry, and the molecular mechanisms of Punica granatum L.(pomegranate) plant extracts: A review. Biomed. Pharmacother. 2022, 153, 113256. [Google Scholar] [CrossRef]
- Escobar-Puentes, A.A.; Reyes-López, S.Y.; Baltazar, Á.D.J.R.; López-Teros, V.; Wall-Medrano, A. Molecular interaction of β-carotene with sweet potato starch: A bleaching-restitution assay. Food Hydrocoll. 2022, 127, 107522. [Google Scholar] [CrossRef]
- Doner, L.W.; Hicks, K.B. High-performance liquid chromatographic separation of ascorbic acid, erythorbic acid, dehydroascorbic acid, dehydroerythorbic acid, diketogulonic acid, and diketogluconic acid. Anal. Biochem. 1981, 115, 225–230. [Google Scholar] [CrossRef]
- Stevens-Barrón, J.C.; Wall-Medrano, A.; Álvarez-Parrilla, E.; Olivas-Armendáriz, I.; Astiazaran-García, H.; Robles-Zepeda, R.E.; De la Rosa, L.A. Synergistic interactions between tocol and phenolic extracts from different tree nut species against human cancer cell lines. Molecules 2022, 27, 3154. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Viticult. 1965, 16, 144–158. Available online: https://www.ajevonline.org/content/16/3/144.short (accessed on 6 October 2022).
- Kim, D.O.; Jeong, S.W.; Lee, C.Y. Antioxidant capacity of phenolic phytochemicals from various cultivars of plums. Food Chem. 2003, 81, 321–326. [Google Scholar] [CrossRef]
- Lee, J.; Durst, R.W.; Wrolstad, R.E.; Collaborators. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: Collaborative study. J. AOAC Int. 2005, 88, 1269–1278. [Google Scholar] [CrossRef]
- Amaya-Cruz, D.M.; Perez-Ramirez, I.F.; Ortega-Diaz, D.; Rodriguez-Garcia, M.E.; Reynoso-Camacho, R. Roselle (Hibiscus sabdariffa) by-product as functional ingredient: Effect of thermal processing and particle size reduction on bioactive constituents and functional, morphological, and structural properties. J. Food Meas. Charact. 2018, 12, 135–144. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Ou, B.; Hampsch-Woodill, M.; Prior, R.L. Development and validation of an improved oxygen radical absorbance capacity assay using fluorescein as the fluorescent probe. J. Agric. Food Chem. 2001, 49, 4619–4626. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Rad. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Christodouleas, D.C.; Fotakis, C.; Nikokavoura, A.; Papadopoulos, K.; Calokerinos, A.C. Modified DPPH and ABTS assays to assess the antioxidant profile of untreated oils. Food Anal. Met. 2015, 8, 1294–1302. [Google Scholar] [CrossRef]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness, and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef]
- López-López, E.; Naveja, J.J.; Medina-Franco, J.L. DataWarrior: An evaluation of the open-source drug discovery tool. Expert Opin. Drug Discov. 2019, 14, 335–341. [Google Scholar] [CrossRef] [PubMed]
- The UniProt Consortium. UniProt: The universal protein knowledgebase in 2021. Nucleic Acids Res. 2021, 49, D480–D489. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.-T.; Mathias, S.; Bologa, C.; Brunak, S.; Fernandez, N.; Gaulton, A.; Hersey, A.; Holmes, J.; Jensen, L.J.; Karlsson, A.; et al. Pharos: Collating protein information to shed light on the druggable genome. Nucleic Acids Res. 2021, 45, D995–D1002. [Google Scholar] [CrossRef] [PubMed]
- Olivas-Aguirre, F.J.; González-Aguilar, G.A.; Velderrain-Rodríguez, G.R.; Torres-Moreno, H.; Robles-Zepeda, R.E.; Vázquez-Flores, A.A.; de la Rosa, L.A.; Wall-Medrano, A. Radical scavenging and anti-proliferative capacity of three freeze-dried tropical fruits. Int. J. Food Sci. Technol. 2017, 52, 1699–1709. [Google Scholar] [CrossRef]
- Indrayanto, G.; Putra, G.S.; Suhud, F. Validation of in-vitro bioassay methods: Application in herbal drug research. Profiles Drug Subst. Excip. Relat. Methodol. 2021, 46, 273–307. [Google Scholar] [CrossRef] [PubMed]
- Sharma, K.; Akansha, C.E. Comparative studies of proximate, mineral, and phytochemical compositions of pomegranate (Punica granatum) in peel, seed and whole fruit powder. Int. J. Food Sci. Nutr. 2018, 3, 192–196. Available online: http://www.foodsciencejournal.com/archives/2018/vol3/issue2/3-2-49 (accessed on 15 August 2022).
- O’grady, L.; Sigge, G.; Caleb, O.J.; Opara, U.L. Effects of storage temperature and duration on chemical properties, proximate composition and selected bioactive components of pomegranate (Punica granatum L.) arils. LWT-Food Sci. Technol. 2014, 57, 508–515. [Google Scholar] [CrossRef]
- Opara, L.U.; Al-Ani, M.R.; Al-Shuaibi, Y.S. Physico-chemical properties, vitamin C content, and antimicrobial properties of pomegranate fruit (Punica granatum L.). Food Bioprocess Technol. 2009, 2, 315–321. [Google Scholar] [CrossRef]
- Costa, A.M.M.; Silva, L.O.; Torres, A.G. Chemical composition of commercial cold-pressed pomegranate (Punica granatum) seed oil from Turkey and Israel, and the use of bioactive compounds for samples’ origin preliminary discrimination. J. Food Comp. Anal. 2019, 75, 8–16. [Google Scholar] [CrossRef]
- Guo, S.; Deng, Q.; Xiao, J.; Xie, B.; Sun, Z. Evaluation of antioxidant activity and preventing DNA damage effect of pomegranate extracts by chemiluminescence method. J. Agric. Food Chem. 2007, 55, 3134–3140. [Google Scholar] [CrossRef]
- van Gorkom, G.N.; Lookermans, E.L.; Van Elssen, C.H.; Bos, G.M. The effect of vitamin C (ascorbic acid) in the treatment of patients with cancer: A systematic review. Nutrients 2019, 11, 977. [Google Scholar] [CrossRef] [PubMed]
- Saini, R.K.; Keum, Y.S.; Daglia, M.; Rengasamy, K.R. Dietary carotenoids in cancer chemoprevention and chemotherapy: A review of emerging evidence. Pharmacol. Res. 2020, 157, 104830. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q. Natural forms of vitamin E and metabolites—Regulation of cancer cell death and underlying mechanisms. IUBMB Life 2019, 71, 495–506. [Google Scholar] [CrossRef] [PubMed]
- Balli, D.; Cecchi, L.; Khatib, M.; Bellumori, M.; Cairone, F.; Carradori, S.; Zengin, G.; Cesa, S.; Innocenti, M.; Mulinacci, N. Characterization of arils juice and peel decoction of fifteen varieties of Punica granatum L.: A Focus on anthocyanins, ellagitannins, and polysaccharides. Antioxidants 2020, 9, 238. [Google Scholar] [CrossRef] [PubMed]
- Di Stefano, V.; Pitonzo, R.; Novara, M.E.; Bongiorno, D.; Indelicato, S.; Gentile, C.; Avellone, G.; Bognanni, R.; Scandurra, S.; Melilli, M.G. Antioxidant activity and phenolic composition in pomegranate (Punica granatum L.) genotypes from south Italy by UPLC–Orbitrap-MS approach. J. Sci. Food Agric. 2019, 99, 1038–1045. [Google Scholar] [CrossRef]
- Skrt, M.; Albreht, A.; Vovk, I.; Constantin, O.E.; Râpeanu, G.; Sežun, M.; Črnivec, I.G.O.; Zalar, U.; Ulrih, N.P. Extraction of polyphenols and valorization of fibers from Istrian-grown pomegranate (Punica granatum L.). Foods 2022, 11, 2740. [Google Scholar] [CrossRef]
- Fernandes, L.; Pereira, J.A.; Lopéz-Cortés, I.; Salazar, D.M.; González-Álvarez, J.; Ramalhosa, E. Physicochemical composition and antioxidant activity of several pomegranate (Punica granatum L.) cultivars grown in Spain. Eur. Foood Res. Technol. 2017, 243, 1799–1814. [Google Scholar] [CrossRef]
- Ambigaipalan, P.; de Camargo, A.C.; Shahidi, F. Identification of phenolic antioxidants and bioactives of pomegranate seeds following juice extraction using HPLC-DAD-ESI-MSn. Food Chem. 2017, 221, 1883–1894. [Google Scholar] [CrossRef]
- Lavoro, A.; Falzone, L.; Gattuso, G.; Salemi, R.; Cultrera, G.; Leone, G.M.; Scandurra, G.; Candido, S.; Libra, M. Pomegranate: A promising avenue against the most common chronic diseases and their associated risk factors. Int. J. Funct. Nutr. 2021, 2, 6. [Google Scholar] [CrossRef]
- Ismail, T.; Calcabrini, C.; Diaz, A.R.; Fimognari, C.; Turrini, E.; Catanzaro, E.; Akhtar, S.; Sestili, P. Ellagitannins in cancer chemoprevention and therapy. Toxins 2016, 8, 151. [Google Scholar] [CrossRef]
- Sahin, T.K.; Bilir, B.; Kucuk, O. Modulation of inflammation by phytochemicals to enhance efficacy and reduce toxicity of cancer chemotherapy. Crit. Rev. Food Sci. Nutr. 2021, 16, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Ozkan, E.E.; Seyhan, M.F.; Sirin, O.K.; Ozden, T.Y.; Ersoy, E.; Cakmar, S.D.H.; Goren, A.C.; Aydogan, H.Y.; Ozturk, O. Antiproliferative effects of Turkish pomegranate (Punica granatum L.) extracts on MCF-7 human breast cancer cell lines with focus on antioxidant potential and bioactive compounds analyzed by LC-MS/MS. J. Food Biochem. 2021, 45, e13904. [Google Scholar] [CrossRef]
- Velderrain-Rodríguez, G.R.; Torres-Moreno, H.; Villegas-Ochoa, M.A.; Ayala-Zavala, J.F.; Robles-Zepeda, R.E.; Wall-Medrano, A.; González-Aguilar, G.A. Gallic acid content and an antioxidant mechanism are responsible for the antiproliferative activity of ‘Ataulfo’mango peel on LS180 cells. Molecules 2018, 23, 695. [Google Scholar] [CrossRef] [PubMed]
- Nicolson, G.L.; Veljkovic, V.; Glisic, S.; Perovic, V.; Veljkovic, N. Pomegranate (Punica granatum): A natural source for the development of therapeutic compositions of food supplements with anticancer activities based on electron acceptor molecular characteristics. Funct. Foods Health Dis. 2016, 6, 769–787. [Google Scholar] [CrossRef]
- Forni, C.; Rossi, M.; Borromeo, I.; Feriotto, G.; Platamone, G.; Tabolacci, C.; Mischiati, C.; Beninati, S. Flavonoids: A myth or a reality for cancer therapy? Molecules 2021, 26, 3583. [Google Scholar] [CrossRef]
- Ren, J.-G.; Seth, P.; Ye, H.; Jian-Guo, R.; Hanai, J.-I.; Husain, Z.; Sukhatme, V.P. Citrate suppresses tumor growth in multiple models through inhibition of glycolysis, the tricarboxylic acid cycle and the IGF-1R pathway. Sci. Rep. 2017, 7, 4537. [Google Scholar] [CrossRef]
- Yan, Z.; Qinyan, Z.; Shuai, W.; Zunle, Z.; Xin, Y.; Lihua, Z.; Wei, T. Comparison of phenolic content and antioxidant capacity in fruit parts of different pomegranate varieties. J. Agric. 2020, 10, 57. [Google Scholar] [CrossRef]
- Reczek, C.R.; Chandel, N.S. The two faces of reactive oxygen species in cancer. Ann. Rev. Cancer Biol. 2017, 1, 79–98. [Google Scholar] [CrossRef]
- Saldívar-González, F.I.; Aldas-Bulos, V.D.; Medina-Franco, J.L.; Plisson, F. Natural product drug discovery in the artificial intelligence era. Chem. Sci. 2022, 13, 1526–1546. [Google Scholar] [CrossRef]
- Kumar, N.; Goel, N. Phenolic acids: Natural versatile molecules with promising therapeutic applications. Biotechnol. Rep. 2019, 24, e00370. [Google Scholar] [CrossRef]
- Sengul, H.; Surek, E.; Nilufer-Erdil, D. Investigating the effects of food matrix and food components on bioaccessibility of pomegranate (Punica granatum) phenolics and anthocyanins using an in-vitro gastrointestinal digestion model. Food Res. Int. 2014, 62, 1069–1079. [Google Scholar] [CrossRef]
- Heleno, S.A.; Martins, A.; Queiroz, M.J.R.; Ferreira, I.C. Bioactivity of phenolic acids: Metabolites versus parent compounds: A review. Food Chem. 2015, 173, 501–513. [Google Scholar] [CrossRef] [PubMed]
- Qie, X.; Wu, Y.; Chen, Y.; Liu, C.; Zeng, M.; Qin, F.; Wang, Z.; Chen, J.; He, Z. Competitive interactions among tea catechins, proteins, and digestive enzymes modulate in vitro protein digestibility, catechin bioaccessibility, and antioxidant activity of milk tea beverage model systems. Food Res. Int. 2021, 140, 110050. [Google Scholar] [CrossRef]
- Bansal, T.; Jaggi, M.; Khar, R.; Talegaonkar, S. Emerging significance of flavonoids as P-glycoprotein inhibitors in cancer chemotherapy. J. Pharm. Pharm. Sci. 2009, 12, 46–78. [Google Scholar] [CrossRef] [PubMed]
- Kumari, B.N.; Mahalakshmi, G.S. A cloud-based knowledge discovery framework, for medicinal plants from PubMed literature. Inform. Med. Unlocked 2019, 16, 100226. [Google Scholar] [CrossRef]
- Daina, A.; Michielin, O.; Zoete, V. SwissTargetPrediction: Updated data and new features for efficient prediction of protein targets of small molecules. Nucleic acids Res. 2019, 47, W357–W364. [Google Scholar] [CrossRef]
- Abotaleb, M.; Liskova, A.; Kubatka, P.; Büsselberg, D. Therapeutic potential of plant phenolic acids in the treatment of cancer. Biomolecules 2020, 10, 221. [Google Scholar] [CrossRef]
- Baradaran Rahimi, V.; Ghadiri, M.; Ramezani, M.; Askari, V.R. Antiinflammatory and anti-cancer activities of pomegranate and its constituent, ellagic acid: Evidence from cellular, animal, and clinical studies. Phytother. Res. 2020, 34, 685–720. [Google Scholar] [CrossRef]
- Angeli, A.; Carta, F.; Nocentini, A.; Winum, J.-Y.; Zalubovskis, R.; Akdemir, A.; Onnis, V.; Eldehna, W.; Capasso, C.; Simone, G.; et al. Carbonic anhydrase inhibitors targeting metabolism and tumor microenvironment. Metabolites 2020, 10, 412. [Google Scholar] [CrossRef]
- Varela-Rodríguez, L.; Sánchez-Ramírez, B.; Hernández-Ramírez, V.I.; Varela-Rodríguez, H.; Castellanos-Mijangos, R.D.; González-Horta, C.; Chávez-Munguía, B.; Talamás-Rohana, P. Effect of Gallic acid and Myricetin on ovarian cancer models: A possible alternative antitumoral treatment. BMC Compl. Med. Ther. 2020, 20, 110–116. [Google Scholar] [CrossRef]
- Karioti, A.; Ceruso, M.; Carta, F.; Bilia, A.-R.; Supuran, C.T. New natural product carbonic anhydrase inhibitors incorporating phenol moieties. Bioorg. Med. Chem. 2015, 23, 7219–7225. [Google Scholar] [CrossRef] [PubMed]
- Flores-Estrada, R.A.; Gámez-Meza, N.; Medina-Juárez, L.A.; Castillón-Campaña, L.G.; Molina-Domínguez, C.C.; Rascón-Valenzuela, L.A.; García-Galaz, A. Chemical composition, antioxidant, antimicrobial and antiproliferative activities of wastes from pecan nut [Carya illinoinensis (Wagenh) K. Koch]. Waste Biomass Valorization 2020, 11, 3419–3432. [Google Scholar] [CrossRef]
- Kim, N.D.; Mehta, R.; Yu, W.; Neeman, I.; Livney, T.; Amichay, A.; Poirier, D.; Nicholls, P.; Kirby, A.; Jiang, W.; et al. Chemopreventive and adjuvant therapeutic potential of pomegranate (Punica granatum) for human breast cancer. Breast Cancer Res. Treat. 2002, 71, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Santhini, E.; Ramji, B.; Viswanadha Vijaya, P. Gallic acid isolated from pomegranate peel extract induces reactive oxygen species mediated apoptosis in A549 cell line. J. Cancer Ther. 2011, 2, 16582. [Google Scholar] [CrossRef][Green Version]


| Red | Pink | White | |
|---|---|---|---|
| Moisture (g) | 11.8 ± 0.0 c | 17.8 ± 0.1 a | 14.0 ± 0.7 b |
| Protein (g) | 6.5 ± 0.3 b | 7.4 ± 0.4 a | 7.3 ± 0.4 a |
| Fats (g) | 0.4 ± 0.0 b | 0.9 ± 0.0 a | 0.4 ± 0.1 b |
| Ash (g) | 2.4 ± 0.0 b | 3.1 ± 0.1 a | 2.9 ± 0.0 a |
| Carbohydrates (g) 2 | 79.0 ± 0.4 a | 70.8 ± 0.4 c | 75.4 ± 0.1 b |
| Phenolic compounds (mg GAE) | 2450 ± 90 a | 2240 ± 30 b | 1820 ± 30 c |
| Flavonoids (mg QE) | 320 ± 30 a | 250 ± 20 b | 170 ± 10 c |
| Anthocyanins (mg Cy3GE) 3 | 8070 ± 20 a | 460 ± 20 b | -- |
| Ascorbic acid (μg) | 54.7 ± 12.5 b | 18.5 ± 5.4 c | 105.2 ± 5.3 a |
| Total carotenoids (μg) | 31.0 ± 1.0 a | 32.0 ± 1.2 a | 21.0 ± 0.3 a |
| Lutein | 31.0 ± 1.0 a | 32.0 ± 1.2 a | 9.8 ± 0.3 b |
| α-Carotene | -- | -- | 1.5 ± 0.2 |
| β-Carotene | -- | -- | 9.7 ± 0.4 |
| Total tocols (μg) | 32.1 ± 0.2 c | 69.4 ± 1.0 b | 110.0 ± 0.2 a |
| α-tocopherol | 3.5 ± 0.0 a | 3.2 ± 0.0 b | 3.4 ± 0.0 a |
| γ-tocopherol | 24.7 ± 0.1 c | 63.7 ± 2.9 b | 103.1 ± 0.1 a |
| δ-tocopherol | 3.2 ± 0.2 | -- | -- |
| β-tocotrienol | 0.44 ± 0.1 c | 2.5 ± 0.1 b | 3.1 ± 0.0 a |
| γ-tocotrienol | 0.24 ± 0.0 a | -- | 0.3 ± 0.0 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cortez-Trejo, M.C.; Olivas-Aguirre, F.J.; Dufoo-Hurtado, E.; Castañeda-Moreno, R.; Villegas-Quintero, H.; Medina-Franco, J.L.; Mendoza, S.; Wall-Medrano, A. Potential Anticancer Activity of Pomegranate (Punica granatum L.) Fruits of Different Color: In Vitro and In Silico Evidence. Biomolecules 2022, 12, 1649. https://doi.org/10.3390/biom12111649
Cortez-Trejo MC, Olivas-Aguirre FJ, Dufoo-Hurtado E, Castañeda-Moreno R, Villegas-Quintero H, Medina-Franco JL, Mendoza S, Wall-Medrano A. Potential Anticancer Activity of Pomegranate (Punica granatum L.) Fruits of Different Color: In Vitro and In Silico Evidence. Biomolecules. 2022; 12(11):1649. https://doi.org/10.3390/biom12111649
Chicago/Turabian StyleCortez-Trejo, Maria C., Francisco J. Olivas-Aguirre, Elisa Dufoo-Hurtado, Raquel Castañeda-Moreno, Hassan Villegas-Quintero, José L. Medina-Franco, Sandra Mendoza, and Abraham Wall-Medrano. 2022. "Potential Anticancer Activity of Pomegranate (Punica granatum L.) Fruits of Different Color: In Vitro and In Silico Evidence" Biomolecules 12, no. 11: 1649. https://doi.org/10.3390/biom12111649
APA StyleCortez-Trejo, M. C., Olivas-Aguirre, F. J., Dufoo-Hurtado, E., Castañeda-Moreno, R., Villegas-Quintero, H., Medina-Franco, J. L., Mendoza, S., & Wall-Medrano, A. (2022). Potential Anticancer Activity of Pomegranate (Punica granatum L.) Fruits of Different Color: In Vitro and In Silico Evidence. Biomolecules, 12(11), 1649. https://doi.org/10.3390/biom12111649











