Tropoelastin Promotes the Formation of Dense, Interconnected Endothelial Networks
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture for HUVECs and MSCs
2.2. Cell Proliferation Assays
2.3. Endothelial Tube Formation Assay with Tropoelastin and Conditioned Media
2.4. Analysis and Statistics
3. Results
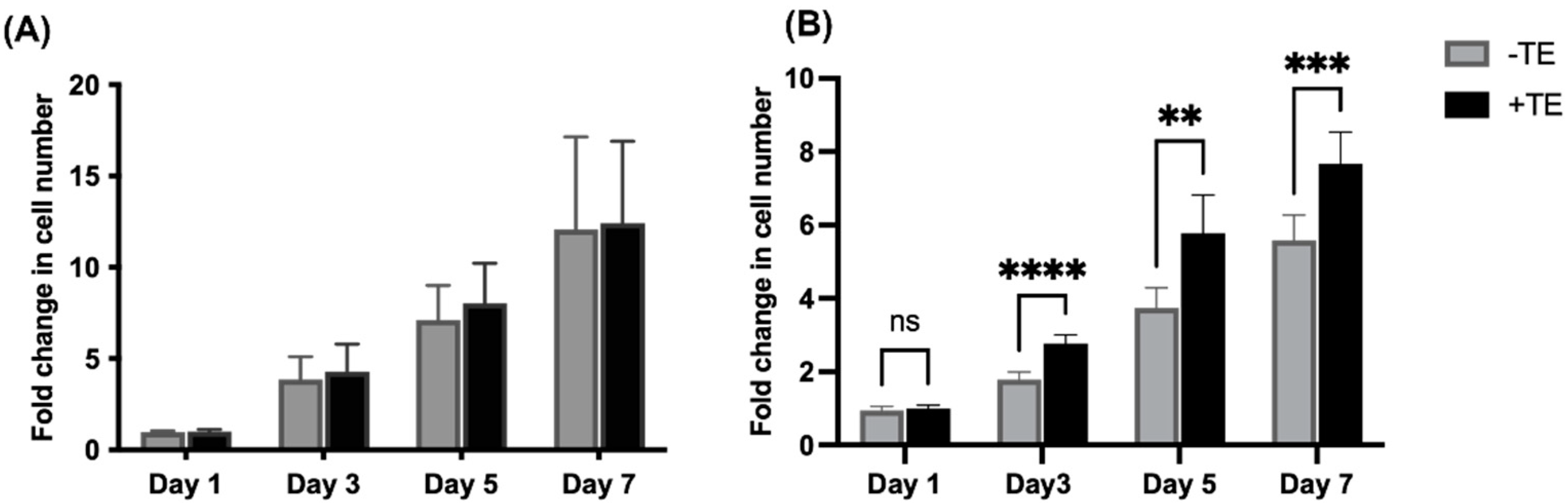
3.1. Endothelial Cell Proliferation Is Unaffected but Mesenchymal Stem Cell Proliferation Increases in the Presence of Tropoelastin
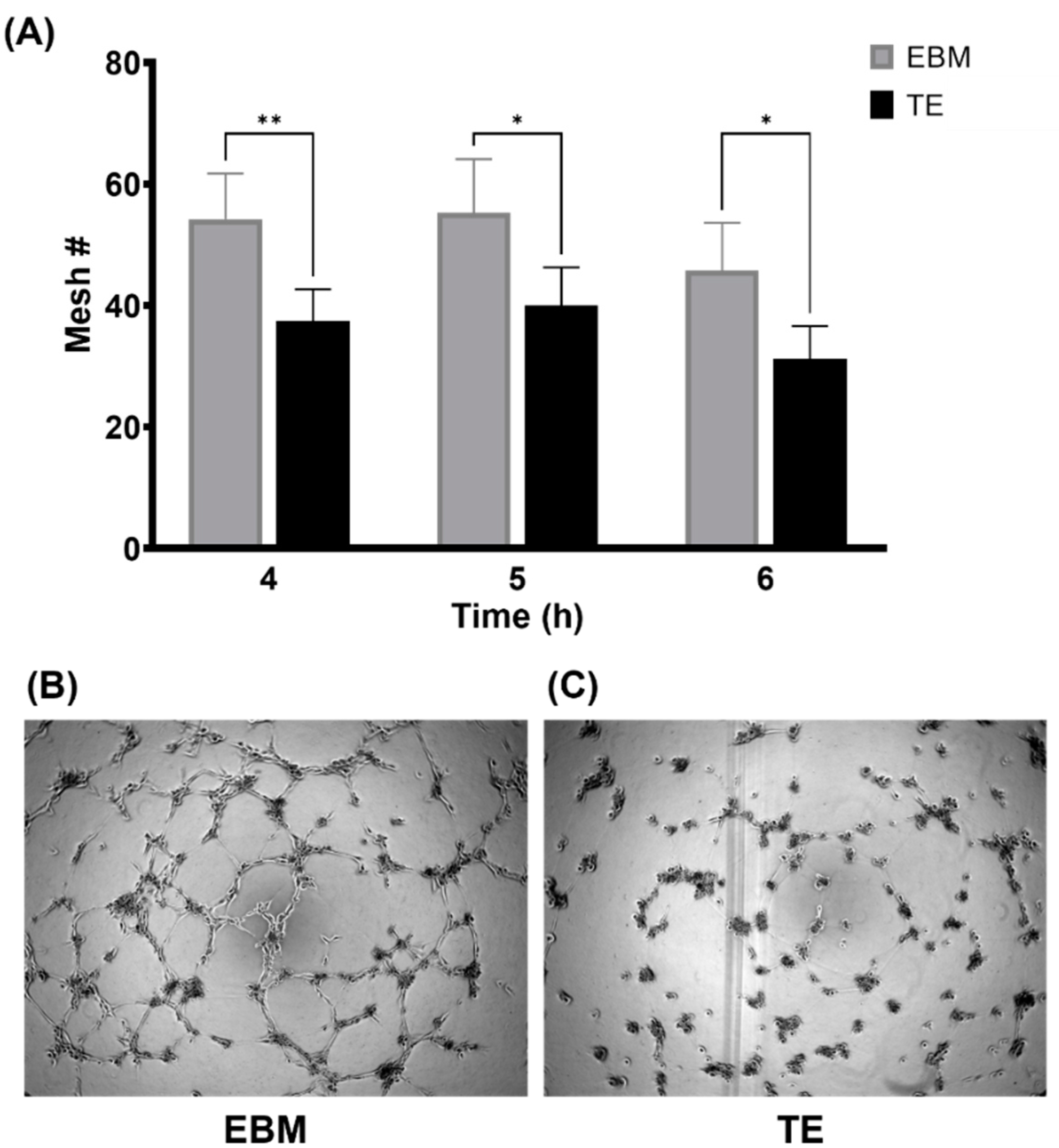
3.2. Tropoelastin Impedes Endothelial Network Formation
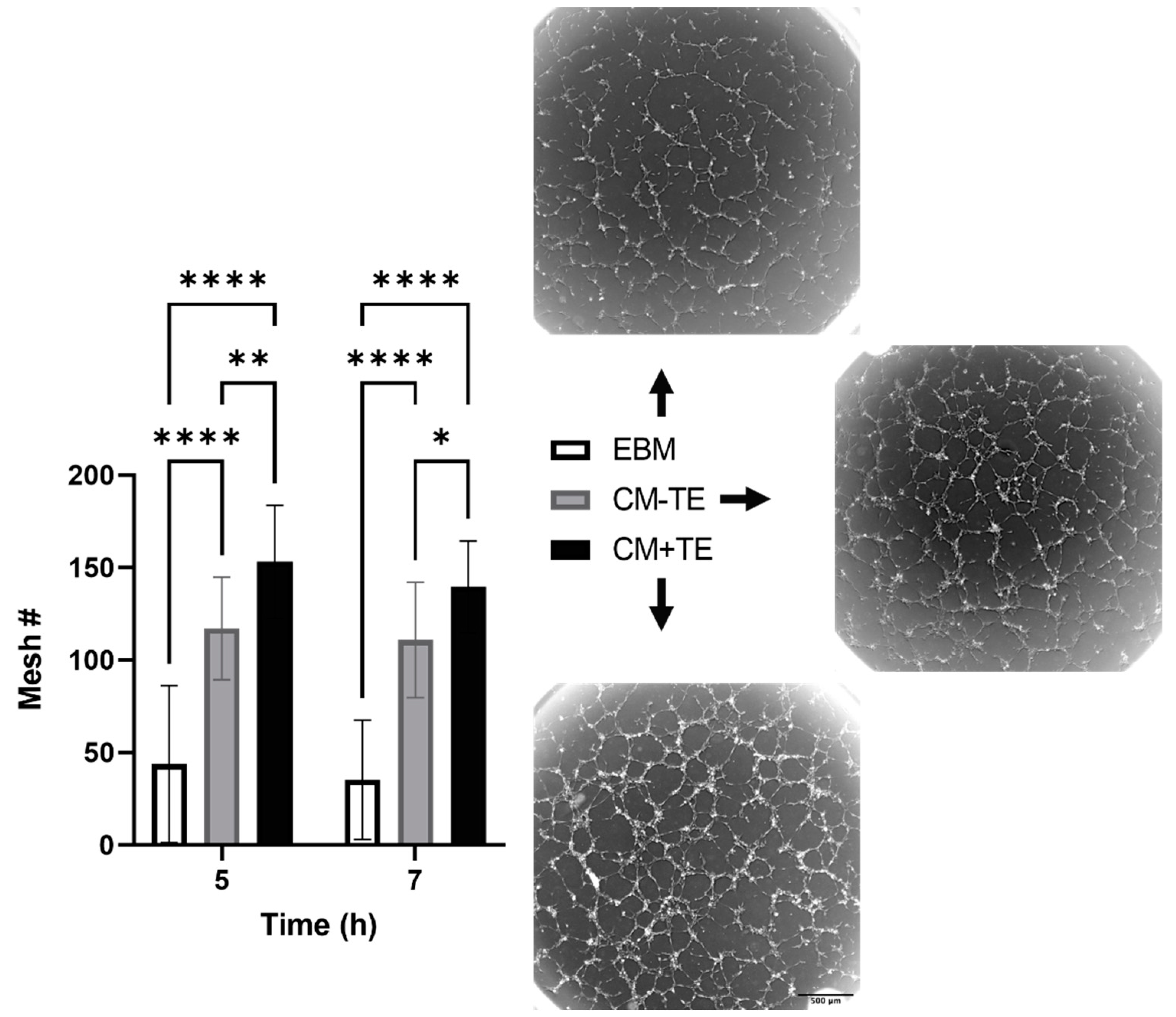
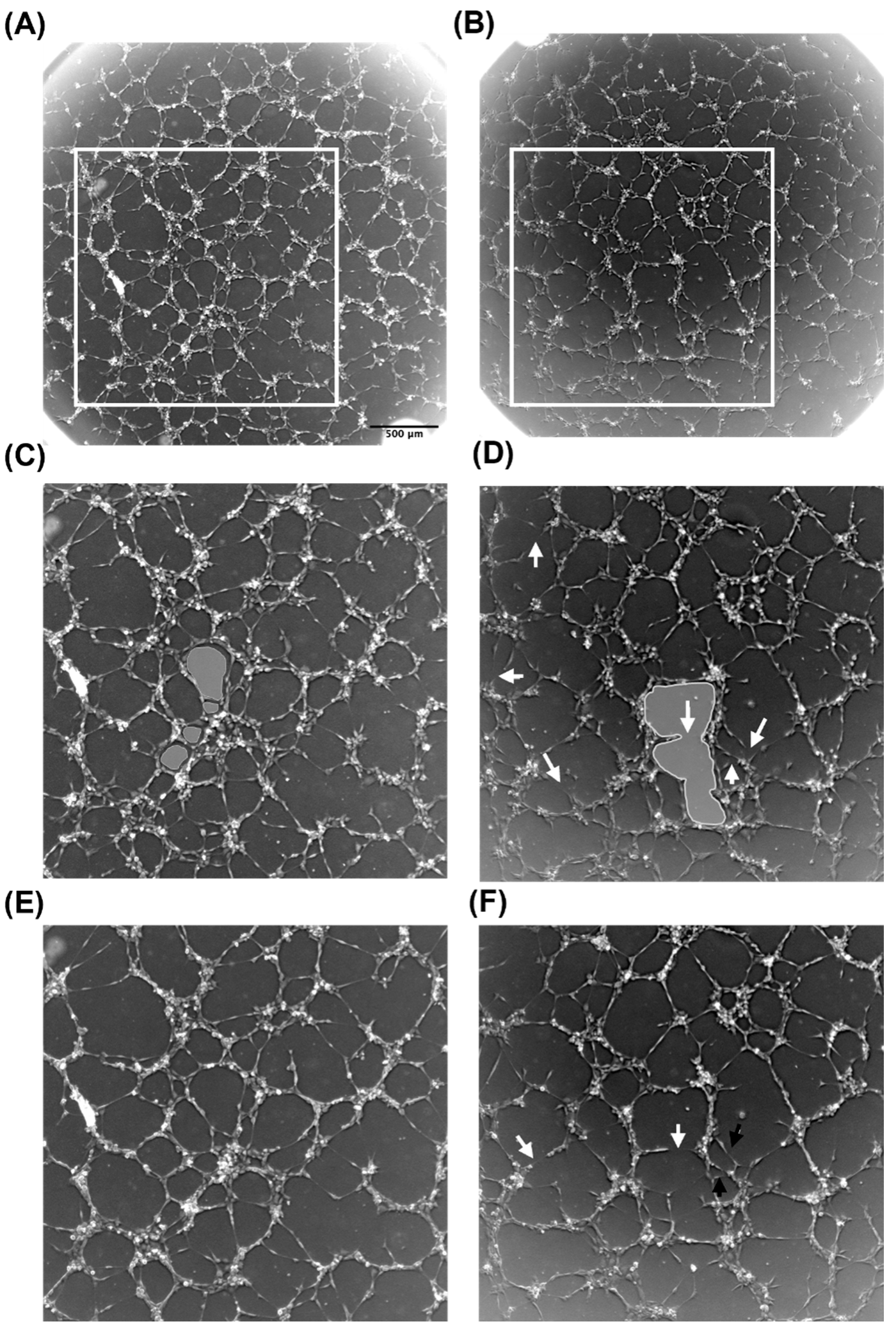
3.3. Conditioned Media from MSCs Cultured on Tropoelastin Results in Dense Interconnected Networks
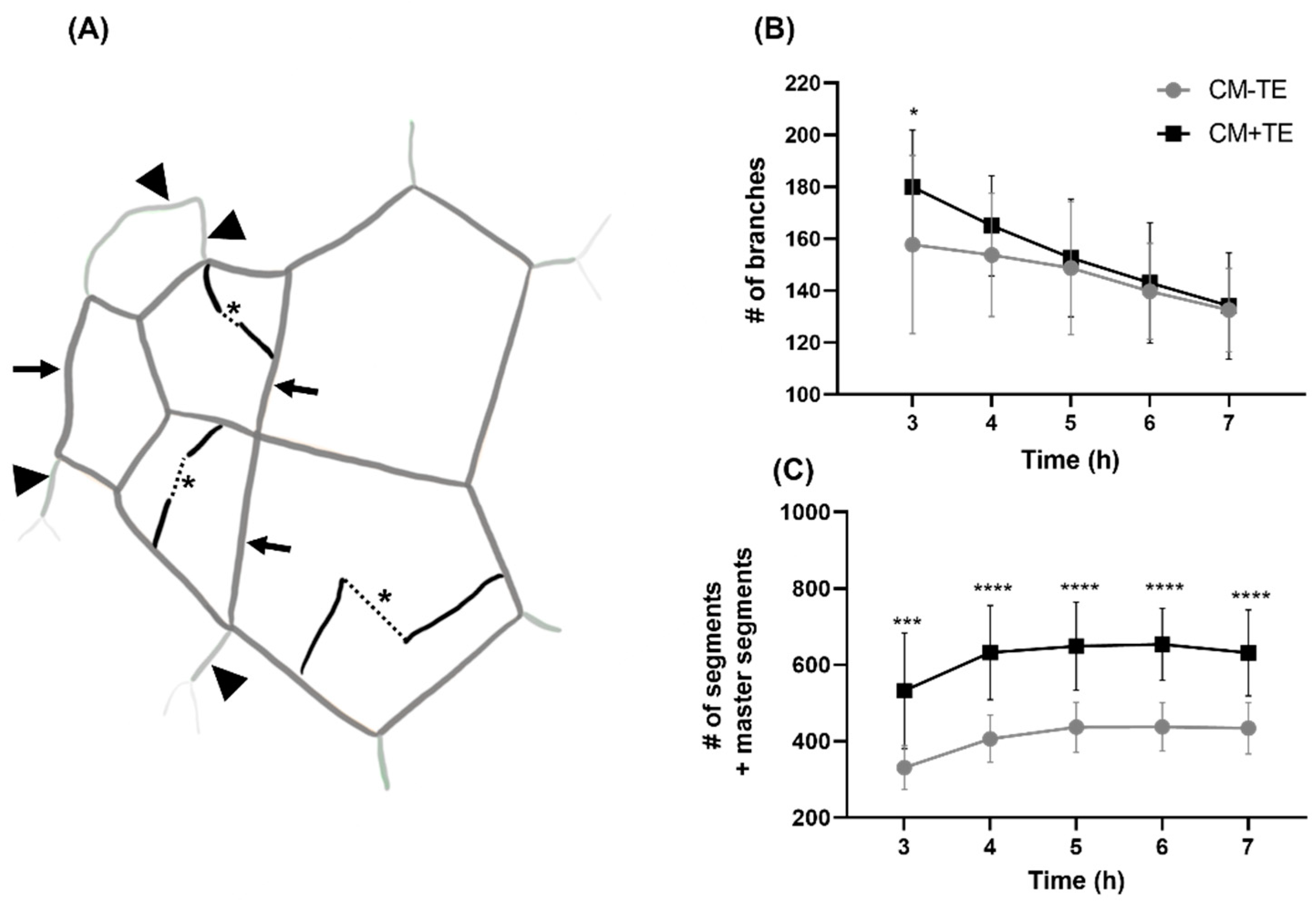
3.4. Endothelial Tube Branching and Anastomosis
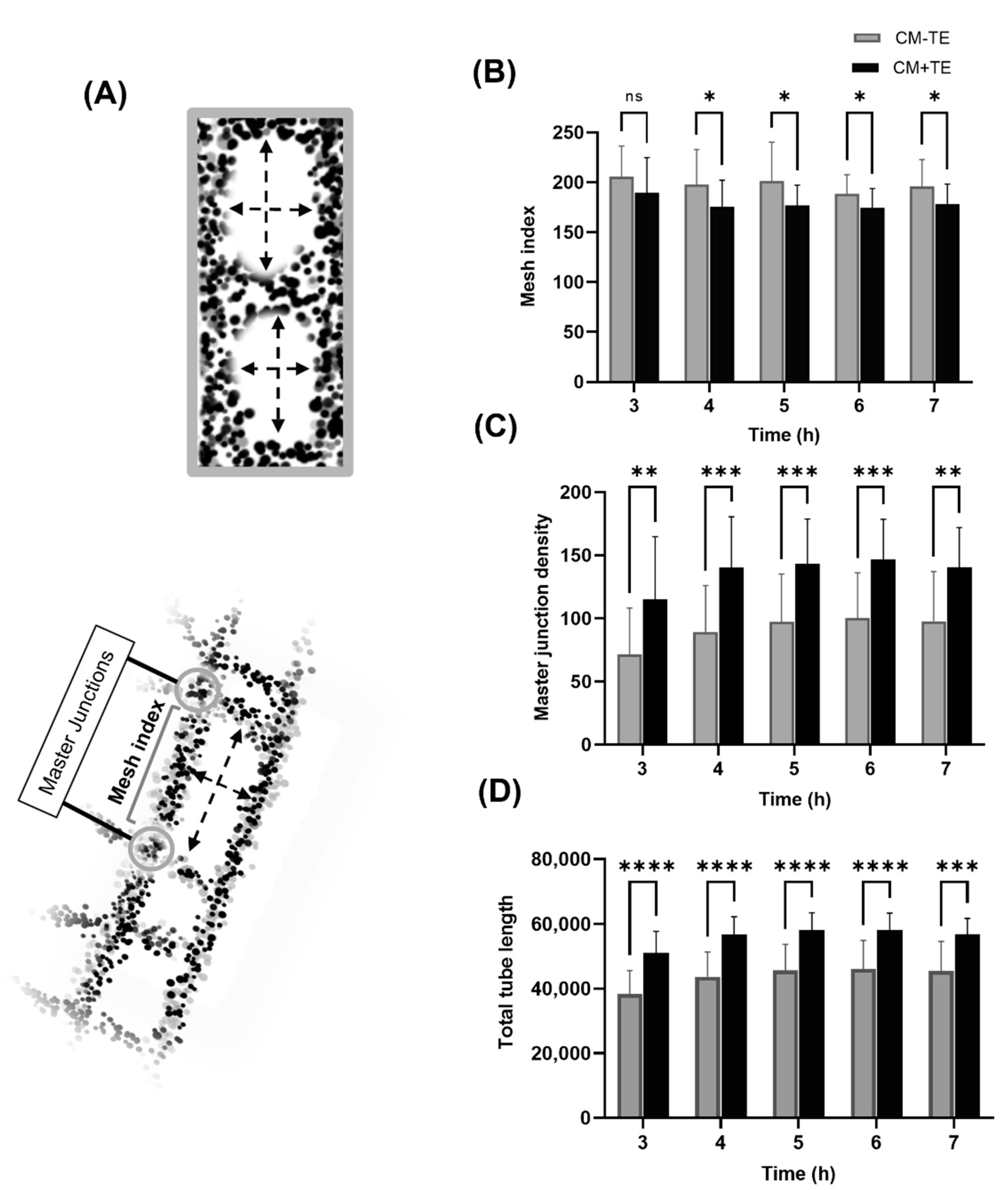
3.5. Network Complexity and Interconnectivity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
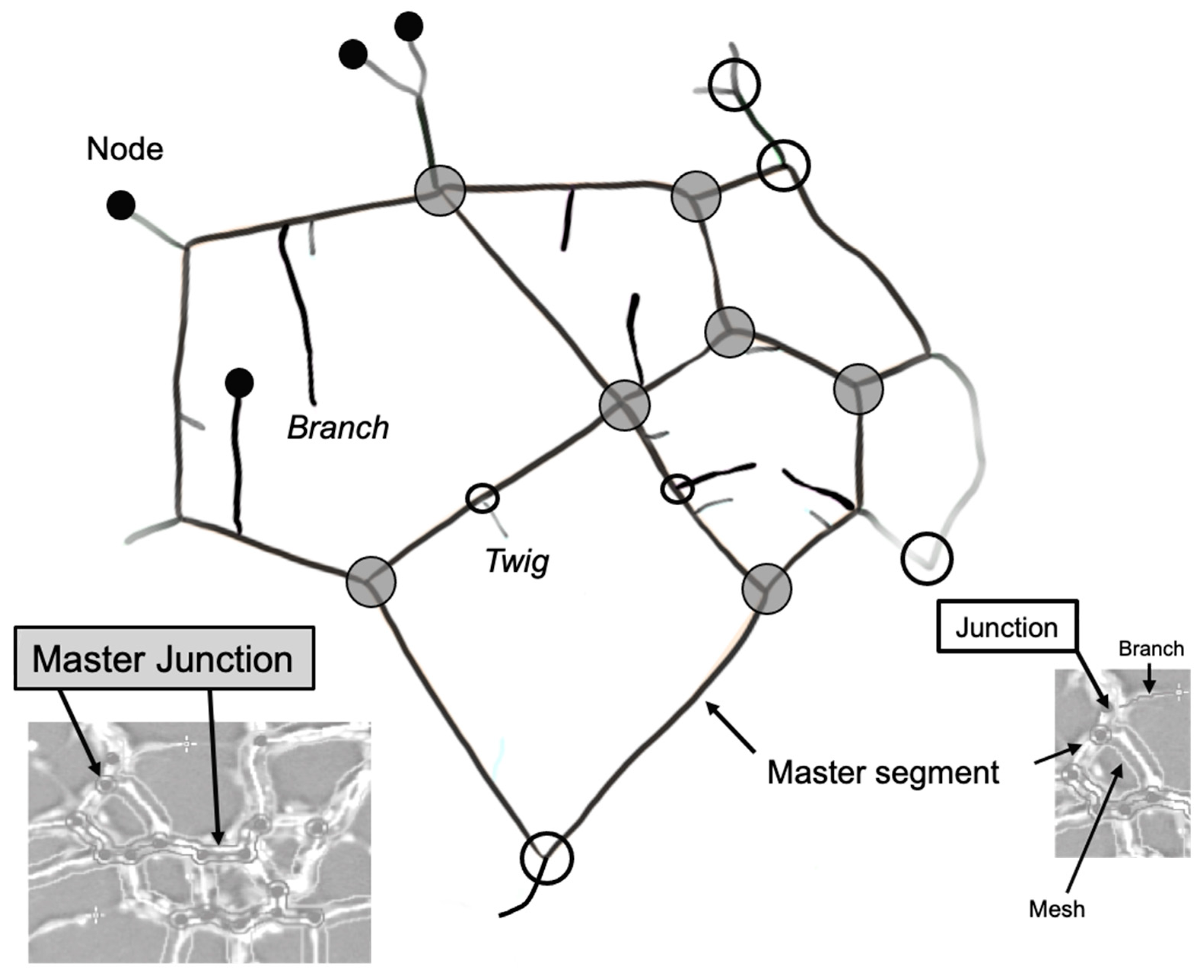
| Master Segment * | Tubes delimited by two junctions or master junctions that are further implicated with other segments and/or branches. |
| Master Junction * | Junction linking at least three master segments. |
| Segment * | Delimited by two junctions. |
| Junction * | Links two master segments as well as segments. Contribute to the non-free end of a branch or a twig. |
| Branch * | Tube delimited by a junction and a node. A branch is identified by the plugin as so if its length meets a set threshold value. |
| Twig | A short branch that is below the threshold cut-off for what the plugin quantifies as a branch. |
| Node | Free end of a branch or a twig. Also present where master junctions and junctions are detected. |
References
- Olczyk, P.; Mencner, L.; Komosińska-Vassev, K. The Role of the Extracellular Matrix Components in Cutaneous Wound Healing. BioMed Res. Int. 2014, 2014, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davis, G.E.; Saunders, W.B. Molecular Balance of Capillary Tube Formation versus Regression in Wound Repair: Role of Matrix Metalloproteinases and Their Inhibitors. J. Investig. Dermatol. Symp. Proc. 2006, 11, 44–56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davis, G.E.; Stratman, A.N.; Sacharidou, A. Molecular Control of Vascular Tube Morphogenesis and Stabilization: Regulation by Extracellular Matrix, Matrix Metalloproteinases, and Endothelial Cell–Pericyte Interactions. In Biophysical Regulation of Vascular Differentiation and Assembly; Gerecht, S., Ed.; Springer: New York, NY, USA, 2011; pp. 17–47. [Google Scholar]
- Davis, G.E.; Senger, D.R. Endothelial Extracellular Matrix: Biosynthesis, Remodeling, and Functions During Vascular Morphogenesis and Neovessel Stabilization. Circ. Res. 2005, 97, 1093–1107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arnaoutova, I.; George, J.; Kleinman, H.K.; Benton, G. The endothelial cell tube formation assay on basement membrane turns 20: State of the science and the art. Angiogenesis 2009, 12, 267–274. [Google Scholar] [CrossRef] [PubMed]
- DeCicco-Skinner, K.L.; Henry, G.; Cataisson, C.; Tabib, T.; Gwilliam, J.C.; Watson, N.J.; Bullwinkle, E.M.; Falkenburg, L.; O’Neill, R.C.; Morin, A.; et al. Endothelial Cell Tube Formation Assay for the In Vitro Study of Angiogenesis. J. Vis. Exp. 2014, e51312. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Kang, K.-T. Advanced tube formation assay using human endothelial colony forming cells for in vitro evaluation of angiogenesis. Korean J. Physiol. Pharmacol. 2018, 22, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Manoussaki, D.; Lubkin, S.R.; Vernon, R.B.; Murray, J.D. A mechanical model for the formation of vascular networks in vitro. Acta Biotheor. 1996, 44, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Vernon, R.B.; Stephanie, L.L.; Christopher, J.D.; Iruela-Arispe, M.L.; John, C.A.; Charles, D.L.; Thomas, N.W.; Sage, E.H. Organized Type I Collagen Influences Endothelial Patterns During Spontaneous Angiogenesis in Vitro: Planar Cultures as Models of Vascular Development. In vitro cellular & developmental biology. Animal 1995, 31, 120–131. [Google Scholar]
- Shen, C.; Lie, P.; Miao, T.; Tianyu, M.; Lu, Q.; Feng, T.; Li, J.; Zu, T.; Liu, X.; Li, H. Conditioned medium from umbilical cord mesenchymal stem cells induces migration and angiogenesis. Mol. Med. Rep. 2015, 12, 20–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maacha, S.; Sidahmed, H.; Jacob, S.; Gentilcore, G.; Calzone, R.; Grivel, J.-C.; Cugno, C. Paracrine Mechanisms of Mesenchymal Stromal Cells in Angiogenesis. Stem Cells Int. 2020, 2020, 4356359. [Google Scholar] [CrossRef]
- Yeo, G.C.; Weiss, A.S. Soluble matrix protein is a potent modulator of mesenchymal stem cell performance. Proc. Natl. Acad. Sci. USA 2019, 116, 2042–2051. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, P.; Yeo, G.; Weiss, A.S. A cell adhesive peptide from tropoelastin promotes sequential cell attachment and spreading via distinct receptors. FEBS J. 2017, 284, 2216–2230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bochicchio, B.; Yeo, G.C.; Lee, P.; Emul, D.; Pepe, A.; Laezza, A.; Ciarfaglia, N.; Quaglino, D.; Weiss, A.S. Domains 12 to 16 of tropoelastin promote cell attachment and spreading through interactions with glycosaminoglycan and integrins alphaV and alpha5beta1. FEBS J. 2021, 288, 4024–4038. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Mithieux, S.M.; Kong, Y.; Wang, X.Q.; Chong, C.; Fathi, A.; Dehghani, F.; Panas, E.; Kemnitzer, J.; Daniels, R.; et al. Tropoelastin Incorporation into a Dermal Regeneration Template Promotes Wound Angiogenesis. Adv. Healthc. Mater. 2015, 4, 577–584. [Google Scholar] [CrossRef]
- Mithieux, S.M.; Aghaei-Ghareh-Bolagh, B.; Yan, L.; Kuppan, K.V.; Wang, Y.; Garces-Suarez, F.; Li, Z.; Maitz, P.K.; Carter, E.A.; Limantoro, C. Tropoelastin Implants That Accelerate Wound Repair. Adv. Healthc. Mater. 2018, 7, 1701206. [Google Scholar] [CrossRef] [Green Version]
- Landau, S.; Szklanny, A.A.; Yeo, G.; Shandalov, Y.; Kosobrodova, E.; Weiss, A.; Levenberg, S. Tropoelastin coated PLLA-PLGA scaffolds promote vascular network formation. Biomaterials 2017, 122, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Carpentier, G.; Martinelli, M.; Courty, J.; Cascone, I. Angiogenesis Analyzer for Imagej. In Proceedings of the 4th ImageJ User and Developer Conference Proceedings, Mondorf-les-Bains, Luxembourg, 24–26 October 2012; pp. 198–201. [Google Scholar]
- Pittman, R.N. The Circulatory System and Oxygen Transport. In Regulation of Tissue Oxygenation, 2nd ed.; Granger, D.N., Granger, J.P., Eds.; Morgan & Claypool Life Sciences: San Rafael, CA, USA, 2016; pp. 3–7. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al Halawani, A.; Abdulkhalek, L.; Mithieux, S.M.; Weiss, A.S. Tropoelastin Promotes the Formation of Dense, Interconnected Endothelial Networks. Biomolecules 2021, 11, 1318. https://doi.org/10.3390/biom11091318
Al Halawani A, Abdulkhalek L, Mithieux SM, Weiss AS. Tropoelastin Promotes the Formation of Dense, Interconnected Endothelial Networks. Biomolecules. 2021; 11(9):1318. https://doi.org/10.3390/biom11091318
Chicago/Turabian StyleAl Halawani, Aleen, Lea Abdulkhalek, Suzanne M. Mithieux, and Anthony S. Weiss. 2021. "Tropoelastin Promotes the Formation of Dense, Interconnected Endothelial Networks" Biomolecules 11, no. 9: 1318. https://doi.org/10.3390/biom11091318
APA StyleAl Halawani, A., Abdulkhalek, L., Mithieux, S. M., & Weiss, A. S. (2021). Tropoelastin Promotes the Formation of Dense, Interconnected Endothelial Networks. Biomolecules, 11(9), 1318. https://doi.org/10.3390/biom11091318







