Mitochondrial Uncoupling Proteins (UCP1-UCP3) and Adenine Nucleotide Translocase (ANT1) Enhance the Protonophoric Action of 2,4-Dinitrophenol in Mitochondria and Planar Bilayer Membranes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Cloning, Purification, and Reconstitution of Murine ANT1 and UCPs
2.3. Generation of the ANT1 R79S Mutant
2.4. Formation of Planar Lipid Bilayer Membranes and Membrane Conductance Measurements
2.5. Isolation of Rat Liver Mitochondria
2.6. Membrane Potential (Δψ) Measurements in Isolated Mitochondria
2.7. Molecular Dynamic Simulations
2.8. Statistics
3. Results
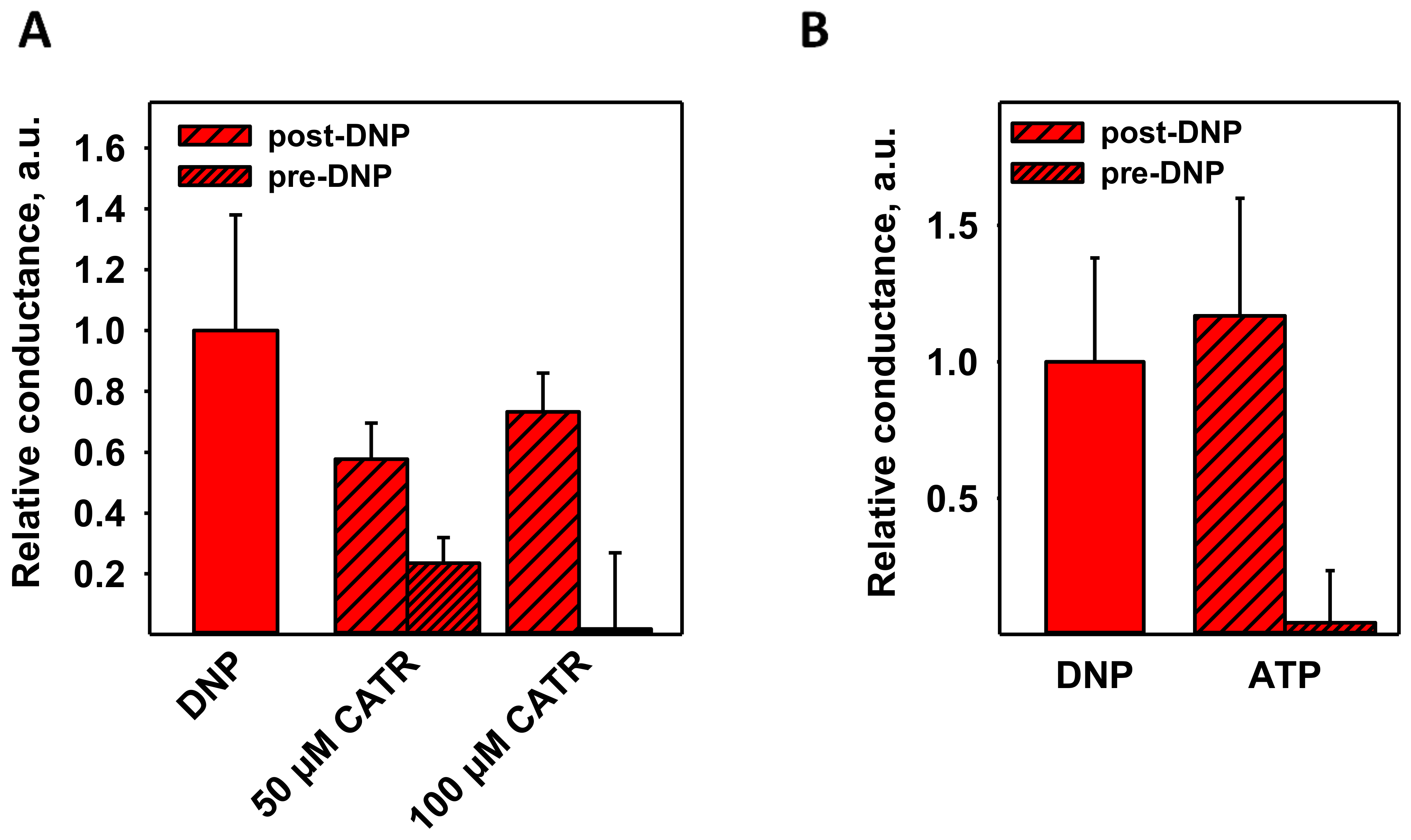
3.1. CATR Recouples Mitochondria Uncoupled by DNP
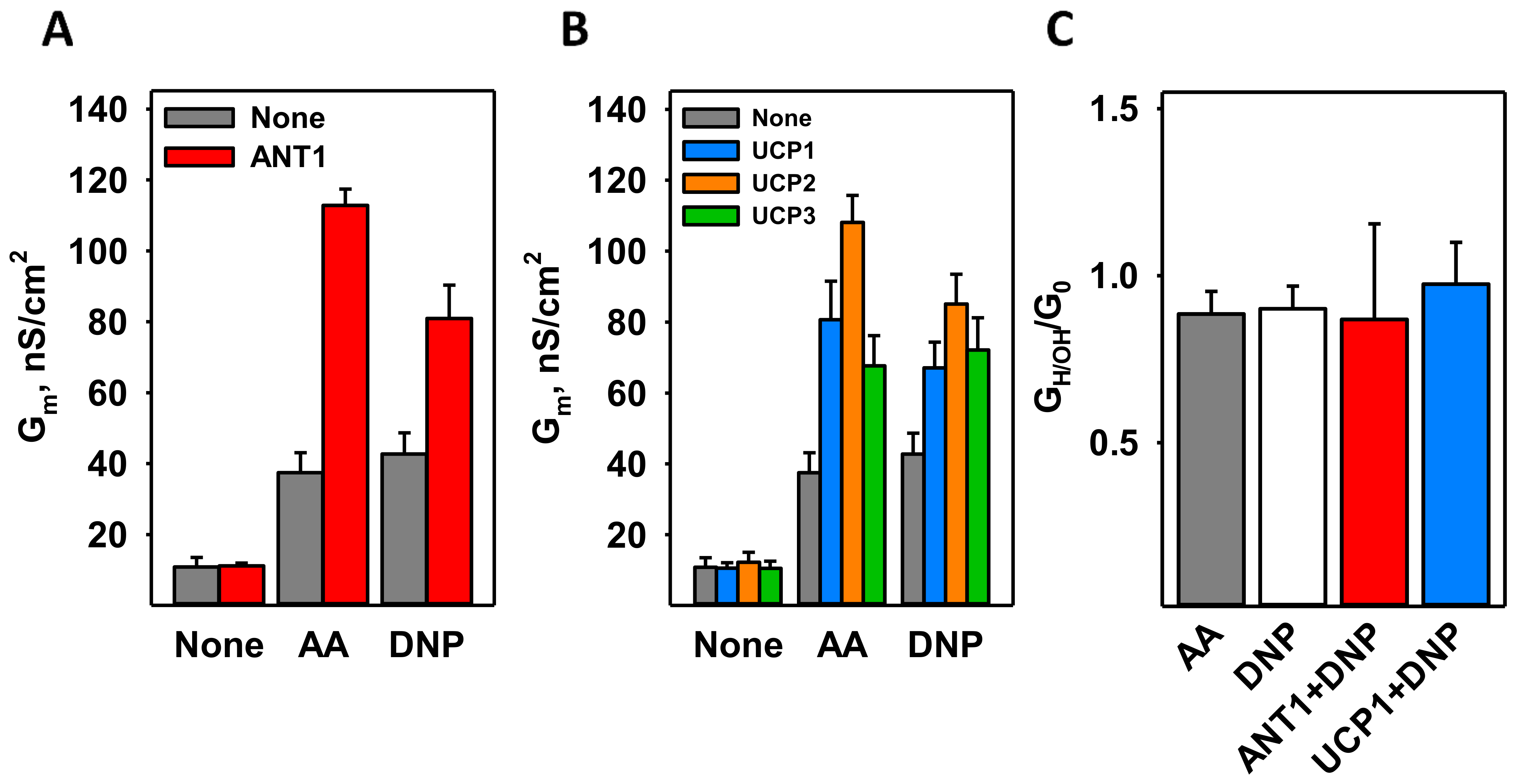
3.2. DNP Increases the Proton Conductance of the Membranes Reconstituted with Mitochondrial Membrane Proteins
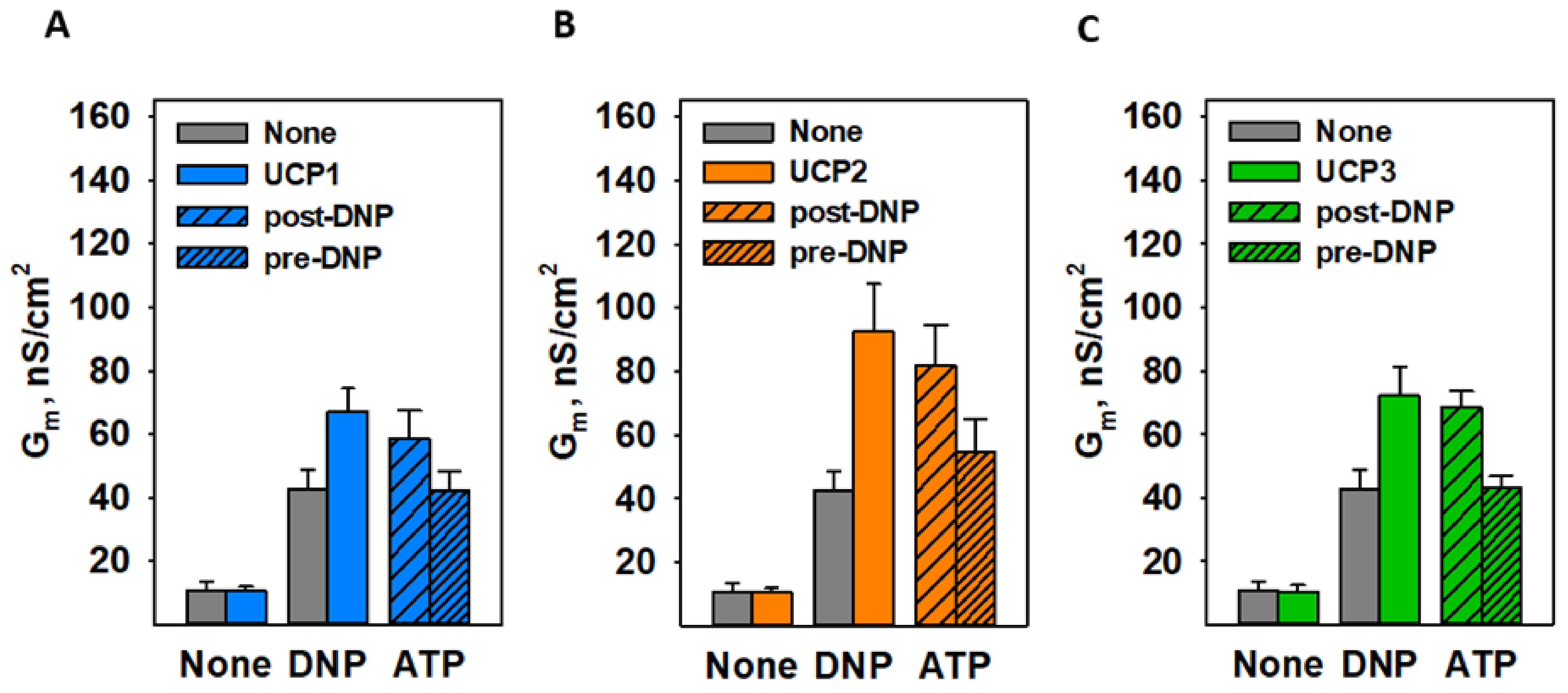
3.3. CATR and ATP Inhibit DNP-Mediated Activation of ANT1 Only If Added before DNP
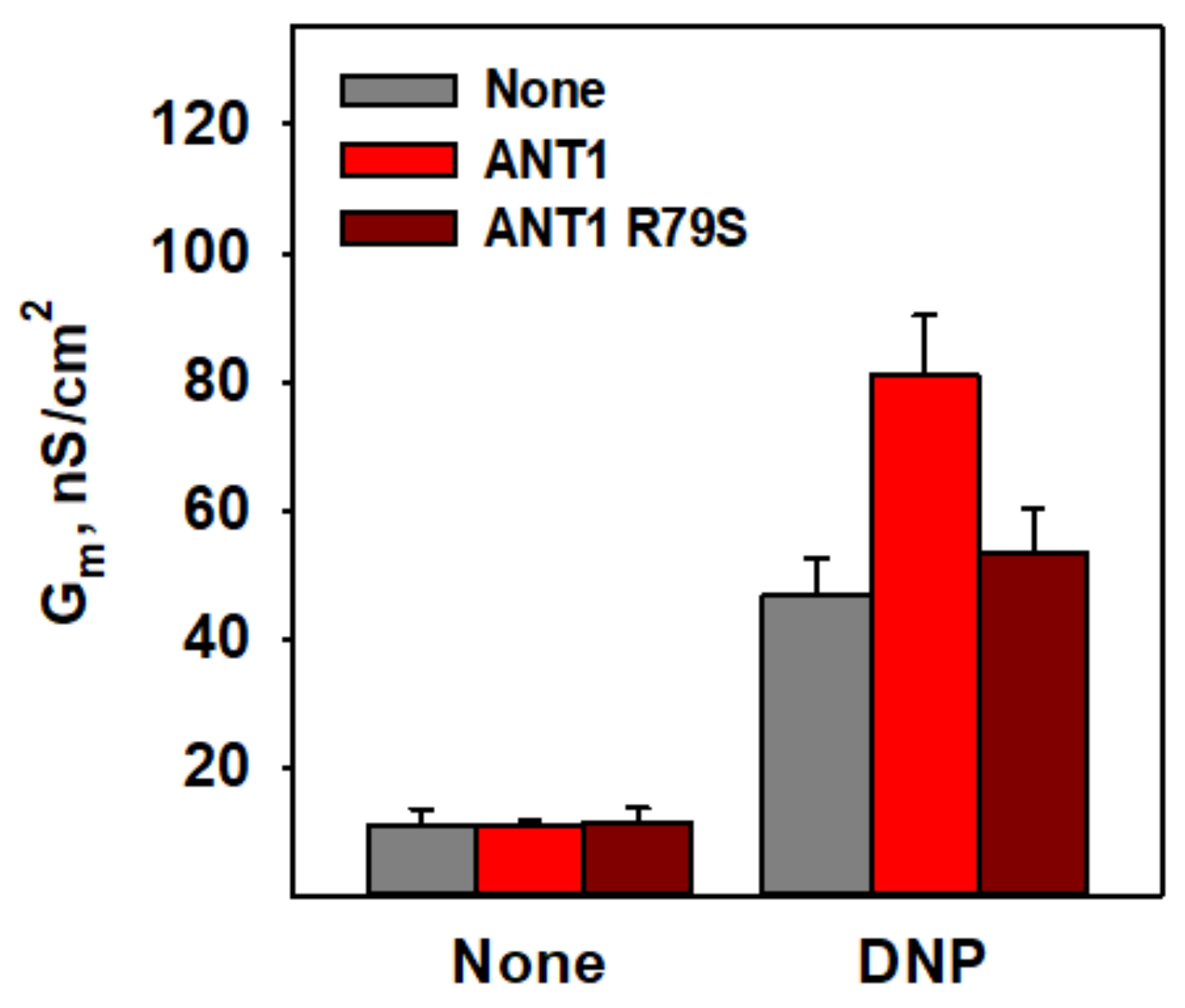
3.4. R79 Is Crucial for DNP–Protein Interaction
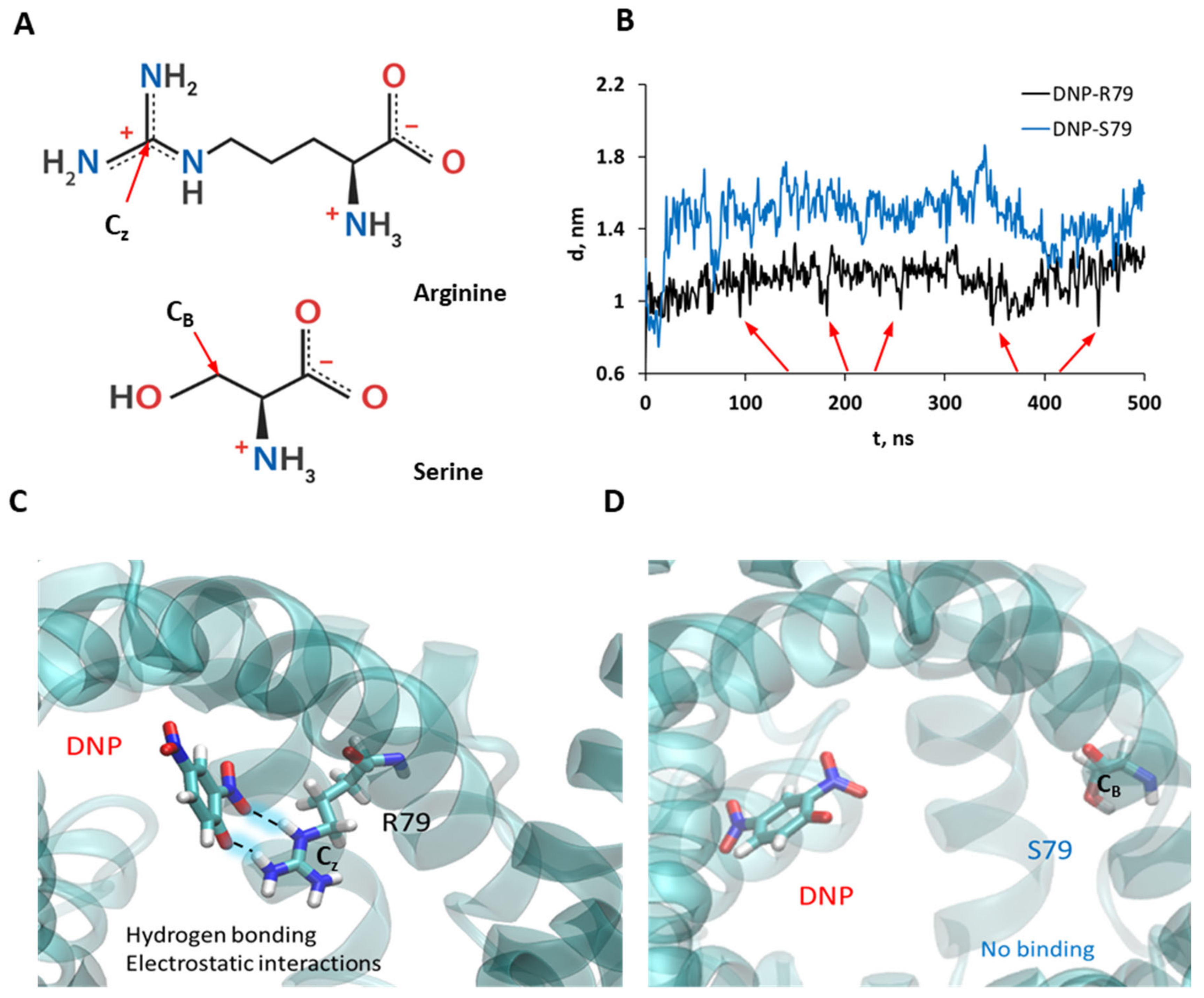
3.5. Molecular Dynamic Simulations Show the Binding of DNP to R79 but Not to S79
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cutting, W.; Mehrtens, H.; Tainter, M. Actions and uses of dinitrophenol: Promising metabolic applications. J. Am. Med. Assoc. 1933, 101, 193–195. [Google Scholar] [CrossRef]
- Tainter, M.L.; Cutting, W.C.; Stockton, A. Use of Dinitrophenol in Nutritional Disorders* A Critical Survey of Clinical Results. Am. J. Public Health Nations Health 1934, 24, 1045–1053. [Google Scholar] [CrossRef]
- Colman, E. Dinitrophenol and obesity: An early twentieth-century regulatory dilemma. Regul. Toxicol. Pharmacol. 2007, 48, 115–117. [Google Scholar] [CrossRef]
- Koncz, D.; Tóth, B.; Roza, O.; Csupor, D. A Systematic Review of the European Rapid Alert System for Food and Feed: Tendencies in Illegal Food Supplements for Weight Loss. Front. Pharmacol. 2021, 11, 611361. [Google Scholar] [CrossRef]
- Ebert, A.; Hannesschlaeger, C.; Goss, K.-U.; Pohl, P. Passive Permeability of Planar Lipid Bilayers to Organic Anions. Biophys. J. 2018, 115, 1931–1941. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hannesschlaeger, C.; Horner, A.; Pohl, P. Intrinsic Membrane Permeability to Small Molecules. Chem. Rev. 2019, 119, 5922–5953. [Google Scholar] [CrossRef] [PubMed]
- Liberman, E.A.; Topaly, V.P.; Silberstein, A.Y. Charged and neutral ion carriers through bimolecular phospholipid membranes. Biochim. Biophys. Acta 1970, 196, 221–234. [Google Scholar] [CrossRef]
- Bakker, E.; Heuvel, E.V.D.; Wiechmann, A.; Van Dam, K. A comparison between the effectiveness of uncouplers of oxidative phosphorylation in mitochondria and in different artificial membrane systems. Biochim. Biophys. Acta Bioenerg. 1973, 292, 78–87. [Google Scholar] [CrossRef]
- Bielawski, J.; Thompson, T.E.; Lehninger, A.L. The effect of 2,4-dinitrophenol on the electrical resistance of phospholipid bilayer membranes. Biochem. Biophys. Res. Commun. 1966, 24, 948–954. [Google Scholar] [CrossRef]
- Hopfer, U.; Lehninger, A.L.; Thompson, T.E. Protonic conductance across phospholipid bilayer membranes induced by uncoupling agents for oxidative phosphorylation. Proc. Natl. Acad. Sci. USA 1968, 59, 484–490. [Google Scholar] [CrossRef] [Green Version]
- Skulachbv, V.; Sharaf, A.; Yagujzinsky, L.; Jasaitis, A.; Liberman, E.; Topali, V. The effect of uncouplers on mitochondria, respiratory enzyme complexes and artificial phospholipid membranes. Biosystems 1968, 2, 98–105. [Google Scholar] [CrossRef]
- Liberman, E.A.; Topaly, V.P.; Tsofina, L.M.; Jasaitis, A.A.; Skulachev, V.P. Mechanism of coupling of oxidative phosphorylation and the membrane potential of mitochondria. Nature 1969, 222, 1076–1078. [Google Scholar] [CrossRef]
- Liberman, E.; Topaly, V. Selective transport of ions through bimolecular phospholipid membranes. Biochim. Biophys. Acta Biomembr. 1968, 163, 125–136. [Google Scholar] [CrossRef]
- Ting, H.; Wilson, D.F.; Chance, B. Effects of uncouplers of oxidative phosphorylation on the specific conductance of bimolecular lipid membranes. Arch. Biochem. Biophys. 1970, 141, 141–146. [Google Scholar] [CrossRef]
- Hanstein, W.G.; Hatefi, Y. Trinitrophenol: A Membrane-Impermeable Uncoupler of Oxidative Phosphorylation. Proc. Natl. Acad. Sci. USA 1974, 71, 288–292. [Google Scholar] [CrossRef] [Green Version]
- Cyboron, G.W.; Dryer, R. Uncoupling of hamster brown adipose and liver mitochondria by 2-azido-4-nitrophenol and binding properties of the reagent. Arch. Biochem. Biophys. 1977, 179, 141–146. [Google Scholar] [CrossRef]
- Kurup, C.K.R.; Sanadi, D.R. Photoaffinity labeling of uncoupler binding sites on mitochondrial membrane. J. Bioenerg. Biomembr. 1977, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Andreyev, A.; Bondareva, T.; Dedukhova, V.; Mokhova, E.; Skulachev, V.; Volkov, N. Carboxyatractylate inhibits the uncoupling effect of free fatty acids. FEBS Lett. 1988, 226, 265–269. [Google Scholar] [CrossRef] [Green Version]
- Lou, P.-H.; Hansen, B.S.; Olsen, P.H.; Tullin, S.; Murphy, M.P.; Brand, M.D. Mitochondrial uncouplers with an extraordinary dynamic range. Biochem. J. 2007, 407, 129–140. [Google Scholar] [CrossRef] [Green Version]
- Kopustinskiene, D.M.; Jovaisiene, J.; Liobikas, J.; Toleikis, A. Diazoxide and Pinacidil Uncouple Pyruvate–Malate-Induced Mitochondrial Respiration. J. Bioenerg. Biomembr. 2002, 34, 49–53. [Google Scholar] [CrossRef]
- Ortega, R.; Garcia, N. The flavonoid quercetin induces changes in mitochondrial permeability by inhibiting adenine nu-cleotide translocase. J. Bioenerg. Biomembr. 2009, 41, 41–47. [Google Scholar] [CrossRef]
- Lu, Y.; Liu, S.; Wang, Y.; Wang, D.; Gao, J.; Zhu, L. Asiatic acid uncouples respiration in isolated mouse liver mitochondria and induces HepG2 cells death. Eur. J. Pharmacol. 2016, 786, 212–223. [Google Scholar] [CrossRef]
- Antonenko, Y.N.; Denisov, S.; Khailova, L.S.; Nazarov, P.A.; Rokitskaya, T.; Tashlitsky, V.N.; Firsov, A.M.; Korshunova, G.A.; Kotova, E.A. Alkyl-substituted phenylamino derivatives of 7-nitrobenz-2-oxa-1,3-diazole as uncouplers of oxidative phosphorylation and antibacterial agents: Involvement of membrane proteins in the uncoupling action. Biochim. Biophys. Acta Biomembr. 2017, 1859, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Firsov, A.M.; Popova, L.B.; Khailova, L.S.; Nazarov, P.A.; Kotova, E.A.; Antonenko, Y.N. Protonophoric action of BAM15 on planar bilayers, liposomes, mitochondria, bacteria and neurons. Bioelectrochemistry 2021, 137, 107673. [Google Scholar] [CrossRef]
- Skulachev, V.P. Uncoupling: New approaches to an old problem of bioenergetics. Biochim. Biophys. Acta Bioenerg. 1998, 1363, 100–124. [Google Scholar] [CrossRef] [Green Version]
- Kreiter, J.; Rupprecht, A.; Škulj, S.; Brkljača, Z.; Žuna, K.; Knyazev, D.; Bardakji, S.; Vazdar, M.; Pohl, E. ANT1 Activation and Inhibition Patterns Support the Fatty Acid Cycling Mechanism for Proton Transport. Int. J. Mol. Sci. 2021, 22, 2490. [Google Scholar] [CrossRef]
- Kreiter, J.; Beitz, E.; Pohl, E.E. A Fluorescence-Based Method to Measure ADP/ATP Exchange of Recombinant Adenine Nucleotide Translocase in Liposomes. Biomolecules 2020, 10, 685. [Google Scholar] [CrossRef] [PubMed]
- Rupprecht, A.; Sokolenko, E.A.; Beck, V.; Ninnemann, O.; Jaburek, M.; Trimbuch, T.; Klishin, S.S.; Jezek, P.; Skulachev, V.; Pohl, E.E. Role of the Transmembrane Potential in the Membrane Proton Leak. Biophys. J. 2010, 98, 1503–1511. [Google Scholar] [CrossRef] [Green Version]
- Macher, G.; Koehler, M.; Rupprecht, A.; Kreiter, J.; Hinterdorfer, P.; Pohl, E.E. Inhibition of mitochondrial UCP1 and UCP3 by purine nucleotides and phosphate. Biochim. Biophys. Acta Biomembr. 2018, 1860, 664–672. [Google Scholar] [CrossRef] [PubMed]
- Kreiter, J.; Pohl, E.E. A Micro-agar Salt Bridge Electrode for Analyzing the Proton Turnover Rate of Recombinant Membrane Proteins. J. Vis. Exp. 2019, 143, e58552. [Google Scholar] [CrossRef] [PubMed]
- Beck, V.; Jabůrek, M.; Breen, E.P.; Porter, R.K.; Ježek, P.; Pohl, E.E. A new automated technique for the reconstitution of hydrophobic proteins into planar bilayer membranes. Studies of human recombinant uncoupling protein 1. Biochim. Biophys. Acta Bioenerg. 2006, 1757, 474–479. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kreiter, J.; Rupprecht, A.; Zimmermann, L.; Moschinger, M.; Rokitskaya, T.; Antonenko, Y.N.; Gille, L.; Fedorova, M.; Pohl, E.E. Molecular Mechanisms Responsible for Pharmacological Effects of Genipin on Mitochondrial Proteins. Biophys. J. 2019, 117, 1845–1857. [Google Scholar] [CrossRef] [Green Version]
- Johnson, D.; Lardy, H. Isolation of liver or kidney mitochondria. Anal. Biochem. 1967, 94–96. [Google Scholar] [CrossRef]
- Åkerman, K.E.; Wikström, M.K. Safranine as a probe of the mitochondrial membrane potential. FEBS Lett. 1976, 68, 191–197. [Google Scholar] [CrossRef] [Green Version]
- Škulj, S.; Brkljača, Z.; Vazdar, M. Molecular Dynamics Simulations of the Elusive Matrix-Open State of Mitochondrial ADP/ATP Carrier. Isr. J. Chem. 2020, 60, 735–743. [Google Scholar] [CrossRef]
- Jo, S.; Lim, J.B.; Klauda, J.B.; Im, W. CHARMM-GUI Membrane Builder for mixed bilayers and its application to yeast membranes. Biophys. J. 2009, 97, 50–58. [Google Scholar] [CrossRef] [Green Version]
- Wu, E.L.; Cheng, X.; Jo, S.; Rui, H.; Song, K.C.; Davila-Contreras, E.M.; Qi, Y.; Lee, J.; Monje-Galvan, V.; Venable, R.M.; et al. CHARMM-GUI Membrane Builder toward realistic biological membrane simulations. J. Comput. Chem. 2014, 35, 1997–2004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.; Cheng, X.; Swails, J.M.; Yeom, M.S.; Eastman, P.K.; Lemkul, J.A.; Wei, S.; Buckner, J.; Jeong, J.C.; Qi, Y.; et al. CHARMM-GUI Input Generator for NAMD, GROMACS, AMBER, OpenMM, and CHARMM/OpenMM Simulations Using the CHARMM36 Additive Force Field. J. Chem. Theory Comput. 2016, 12, 405–413. [Google Scholar] [CrossRef]
- Huang, J.; Rauscher, S.; Nawrocki, G.; Ran, T.; Feig, M.; de Groot, B.L.; Grubmuller, H.; MacKerell, A.D., Jr. CHARMM36m: An improved force field for folded and intrinsically disordered proteins. Nat. Methods 2017, 14, 71–73. [Google Scholar] [CrossRef] [Green Version]
- Vanommeslaeghe, K.; Hatcher, E.; Acharya, C.; Kundu, S.; Zhong, S.; Shim, J.; Darian, E.; Guvench, O.; Lopes, P.E.M.; Vorobyov, I.; et al. CHARMM general force field: A force field for drug-like molecules compatible with the CHARMM all-atom additive biological force fields. J. Comput. Chem. 2009, 31, 671–690. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jo, S.; Kim, T.; Im, W. Automated Builder and Database of Protein/Membrane Complexes for Molecular Dynamics Simulations. PLoS ONE 2007, 2, e880. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nosé, S. A molecular dynamics method for simulations in the canonical ensemble. Mol. Phys. 1984, 52, 255–268. [Google Scholar] [CrossRef]
- Parrinello, M.; Rahman, A. Polymorphic transitions in single crystals: A new molecular dynamics method. J. Appl. Phys. 1981, 52, 7182–7190. [Google Scholar] [CrossRef]
- Essmann, U.; Perera, L.; Berkowitz, M.L.; Darden, T.; Lee, H.; Pedersen, L.G. A smooth particle mesh Ewald method. J. Chem. Phys. 1995, 103, 8577–8593. [Google Scholar] [CrossRef] [Green Version]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual molecular dynamics. J. Mol. Graph. 1996, 14, 33–38. [Google Scholar] [CrossRef]
- Abraham, M.J.; Murtola, T.; Schulz, R.; Páll, S.; Smith, J.; Hess, B.; Lindahl, E. GROMACS: High performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 2015, 1-2, 19–25. [Google Scholar] [CrossRef] [Green Version]
- Andreyev, A.Y.; Bondareva, T.O.; Dedukhova, V.I.; Mokhova, E.N.; Skulachev, V.P.; Tsofina, L.M.; Volkov, N.I.; Vygodina, T.V. The ATP/ADP-antiporter is involved in the uncoupling effect of fatty acids on mitochondria. Eur. J. Biochem. 1989, 182, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Schonfeld, P. Does the function of adenine nucleotide translocase in fatty acid uncoupling depend on the type of mitochondria? FEBS Lett. 1990, 264, 246–248. [Google Scholar] [CrossRef] [Green Version]
- Hilse, K.E.; Rupprecht, A.; Egerbacher, M.; Bardakji, S.; Zimmermann, L.; Wulczyn, A.; Pohl, E.E. The Expression of Un-coupling Protein 3 Coincides with the Fatty Acid Oxidation Type of Metabolism in Adult Murine Heart. Front. Physiol. 2018, 9, 747. [Google Scholar] [CrossRef]
- Skárka, L.; Bardová, K.; Brauner, P.; Flachs, P.; Jarkovská, D.; Kopecký, J.; Ostádal, B. Expression of mitochondrial uncoupling protein 3 and adenine nucleotide translocase 1 genes in developing rat heart: Putative involvement in control of mitochondrial membrane potential. J. Mol. Cell. Cardiol. 2003, 35. [Google Scholar]
- Bertholet, A.M.; Chouchani, E.T.; Kazak, L.; Angelin, A.; Fedorenko, A.; Long, J.Z.; Vidoni, S.; Garrity, R.; Cho, J.; Terada, N.; et al. H+ transport is an integral function of the mitochondrial ADP/ATP carrier. Nat. Cell Biol. 2019, 571, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Brustovetsky, N.; Klingenberg, M. The reconstituted ADP/ATP carrier can mediate H+ transport by free fatty acids, which is further stimulated by mersalyl. J. Biol. Chem. 1994, 269, 27329–27336. [Google Scholar] [CrossRef]
- Nelson, D.; Lawson, J.E.; Klingenberg, M.; Douglas, M.G. Site-directed Mutagenesis of the Yeast Mitochondrial ADP/ATP Translocator: Six Arginines and One Lysine are Essential. J. Mol. Biol. 1993, 230, 1159–1170. [Google Scholar] [CrossRef] [PubMed]
- Heidkämper, D.; Müller, V.; Nelson, D.; Klingenberg, M. Probing the Role of Positive Residues in the ADP/ATP Carrier from Yeast. The Effect of Six Arginine Mutations on Transport and the Four ATP versus ADP Exchange Modes. Biochemistry 1996, 35, 16144–16152. [Google Scholar] [CrossRef] [PubMed]
- Pebay-Peyroula, E.; Dahout-Gonzalez, C.; Kahn, R.; Trézéguet, V.; Lauquin, G.J.-M.; Brandolin, G. Structure of mitochondrial ADP/ATP carrier in complex with carboxyatractyloside. Nat. Cell Biol. 2003, 426, 39–44. [Google Scholar] [CrossRef]
- Wang, Y.; Tajkhorshid, E. Electrostatic funneling of substrate in mitochondrial inner membrane carriers. Proc. Natl. Acad. Sci. USA 2008, 105, 9598–9603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dehez, F.; Pebay-Peyroula, E.; Chipot, C. Binding of ADP in the Mitochondrial ADP/ATP Carrier Is Driven by an Electrostatic Funnel. J. Am. Chem. Soc. 2008, 130, 12725–12733. [Google Scholar] [CrossRef]
- Parker, V.H. Uncouplers of rat-liver mitochondrial oxidative phosphorylation. Biochem. J. 1965, 97, 658–662. [Google Scholar] [CrossRef] [PubMed]
- Pohl, E.E.; Rupprecht, A.; Macher, G.; Hilse, K.E. Important Trends in UCP3 Investigation. Front. Physiol. 2019, 10, 470. [Google Scholar] [CrossRef]
- Skulachev, V.P. Fatty acid circuit as a physiological mechanism of uncoupling of oxidative phosphorylation. FEBS Lett. 1991, 294, 158–162. [Google Scholar] [CrossRef] [Green Version]
- Klingenberg, M. Wanderings in bioenergetics and biomembranes. Biochim. Biophys. Acta 2010, 1797, 579–594. [Google Scholar] [CrossRef] [Green Version]
- Kreiter, J.; Skulj, S.; Brkljaca, Z.; Zuna, K.; Vazdar, M.; Pohl, E.E. The transport of fatty acid anions across the inner mitochondrial membrane by the adenine nucleotide translocase. Eur. Biophys. J. 2021, 50 (Suppl. 1), S57. [Google Scholar]
- Langdown, M.L.; Smith, N.D.; Sugden, M.C.; Holness, M.J. Excessive glucocorticoid exposure during late intrauterine development modulates the expression of cardiac uncoupling proteins in adult hypertensive male offspring. Pflügers Archiv. 2001, 442, 248–255. [Google Scholar] [CrossRef]
- Hilse, K.E.; Kalinovich, A.V.; Rupprecht, A.; Smorodchenko, A.; Zeitz, U.; Staniek, K.; Erben, R.G.; Pohl, E.E. The expression of UCP3 directly correlates to UCP1 abundance in brown adipose tissue. Biochim. Biophys. Acta Bioenerg. 2016, 1857, 72–78. [Google Scholar] [CrossRef] [Green Version]
- Samartsev, V.N.; Smirnov, A.V.; Zeldi, I.P.; Markova, O.V.; Mokhova, E.N.; Skulachev, V.P. Involvement of aspartate/glutamate antiporter in fatty acid-induced uncoupling of liver mitochondria. Biochim. Biophys. Acta 1997, 1319, 251–257. [Google Scholar] [CrossRef] [Green Version]
- Wieckowski, M.; Wojtczak, L. Involvement of the Dicarboxylate Carrier in the Protonophoric Action of Long-Chain Fatty Acids in Mitochondria. Biochem. Biophys. Res. Commun. 1997, 232, 414–417. [Google Scholar] [CrossRef] [PubMed]
- Engstová, H.; Z̆ác̆ková, M.; Růz̆ic̆ka, M.; Meinhardt, A.; Hanuš, J.; Krämer, R.; Jezek, P. Natural and Azido Fatty Acids Inhibit Phosphate Transport and Activate Fatty Acid Anion Uniport Mediated by the Mitochondrial Phosphate Carrier. J. Biol. Chem. 2001, 276, 4683–4691. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, X.X.; Lewin, D.A.; Zhong, A.; Brush, J.; Schow, P.W.; Sherwood, S.W.; Pan, G.; Adams, S.H. Overexpression of the human 2-oxoglutarate carrier lowers mitochondrial membrane potential in HEK-293 cells: Contrast with the unique cold-induced mito-chondrial carrier CGI-69. Biochem. J. 2001, 353, 369–375. [Google Scholar] [CrossRef] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Žuna, K.; Jovanović, O.; Khailova, L.S.; Škulj, S.; Brkljača, Z.; Kreiter, J.; Kotova, E.A.; Vazdar, M.; Antonenko, Y.N.; Pohl, E.E. Mitochondrial Uncoupling Proteins (UCP1-UCP3) and Adenine Nucleotide Translocase (ANT1) Enhance the Protonophoric Action of 2,4-Dinitrophenol in Mitochondria and Planar Bilayer Membranes. Biomolecules 2021, 11, 1178. https://doi.org/10.3390/biom11081178
Žuna K, Jovanović O, Khailova LS, Škulj S, Brkljača Z, Kreiter J, Kotova EA, Vazdar M, Antonenko YN, Pohl EE. Mitochondrial Uncoupling Proteins (UCP1-UCP3) and Adenine Nucleotide Translocase (ANT1) Enhance the Protonophoric Action of 2,4-Dinitrophenol in Mitochondria and Planar Bilayer Membranes. Biomolecules. 2021; 11(8):1178. https://doi.org/10.3390/biom11081178
Chicago/Turabian StyleŽuna, Kristina, Olga Jovanović, Ljudmila S. Khailova, Sanja Škulj, Zlatko Brkljača, Jürgen Kreiter, Elena A. Kotova, Mario Vazdar, Yuri N. Antonenko, and Elena E. Pohl. 2021. "Mitochondrial Uncoupling Proteins (UCP1-UCP3) and Adenine Nucleotide Translocase (ANT1) Enhance the Protonophoric Action of 2,4-Dinitrophenol in Mitochondria and Planar Bilayer Membranes" Biomolecules 11, no. 8: 1178. https://doi.org/10.3390/biom11081178
APA StyleŽuna, K., Jovanović, O., Khailova, L. S., Škulj, S., Brkljača, Z., Kreiter, J., Kotova, E. A., Vazdar, M., Antonenko, Y. N., & Pohl, E. E. (2021). Mitochondrial Uncoupling Proteins (UCP1-UCP3) and Adenine Nucleotide Translocase (ANT1) Enhance the Protonophoric Action of 2,4-Dinitrophenol in Mitochondria and Planar Bilayer Membranes. Biomolecules, 11(8), 1178. https://doi.org/10.3390/biom11081178






