Treatment Options for Motor and Non-Motor Symptoms of Parkinson’s Disease
Abstract
1. Introduction
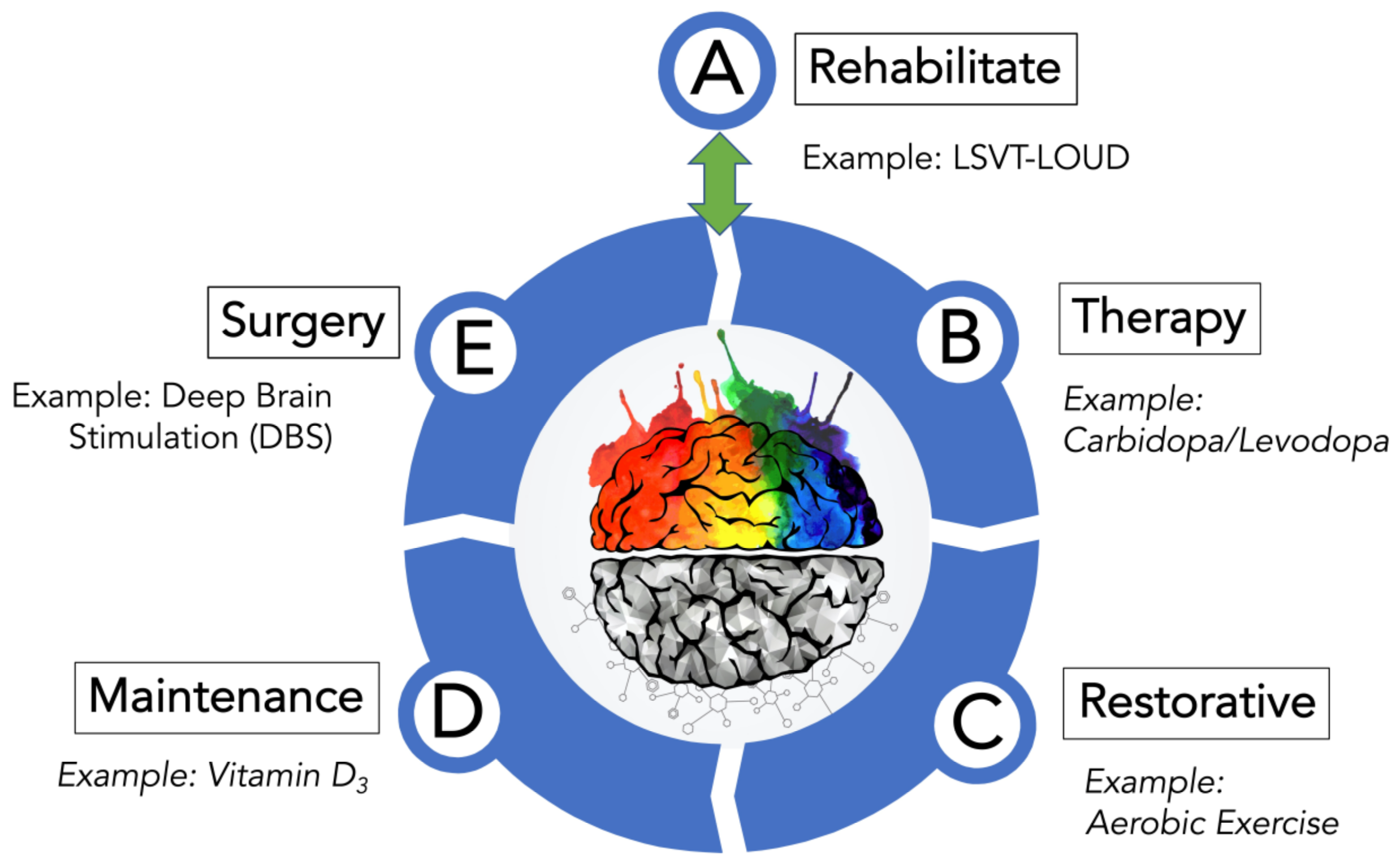
2. Treatment Plan for PD
2.1. Rehabilitate Options for Treating PD
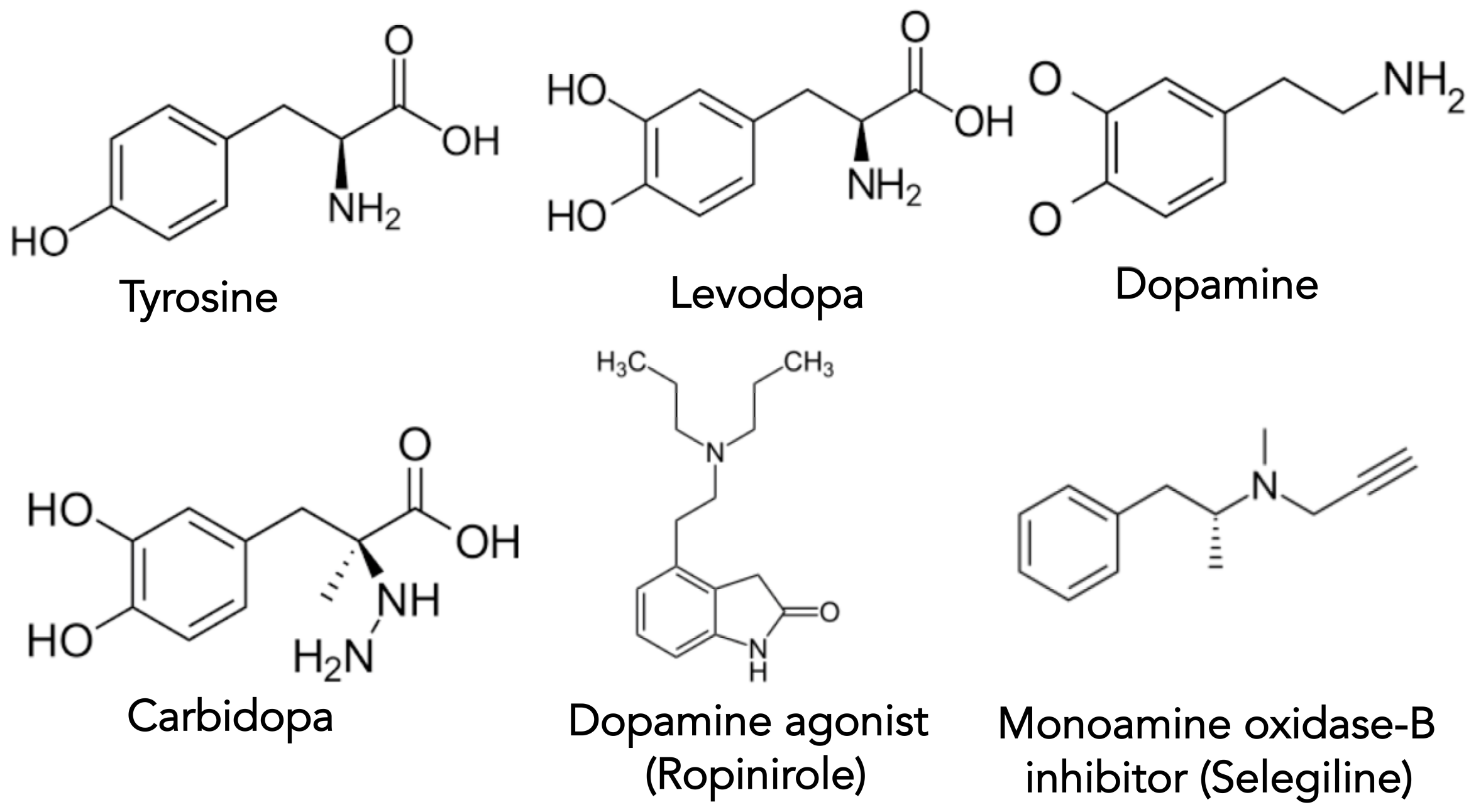
2.2. Therapy Options for Treating Motor Symptoms of PD
2.3. Therapy Options for Treating Non-Motor Symptoms of PD
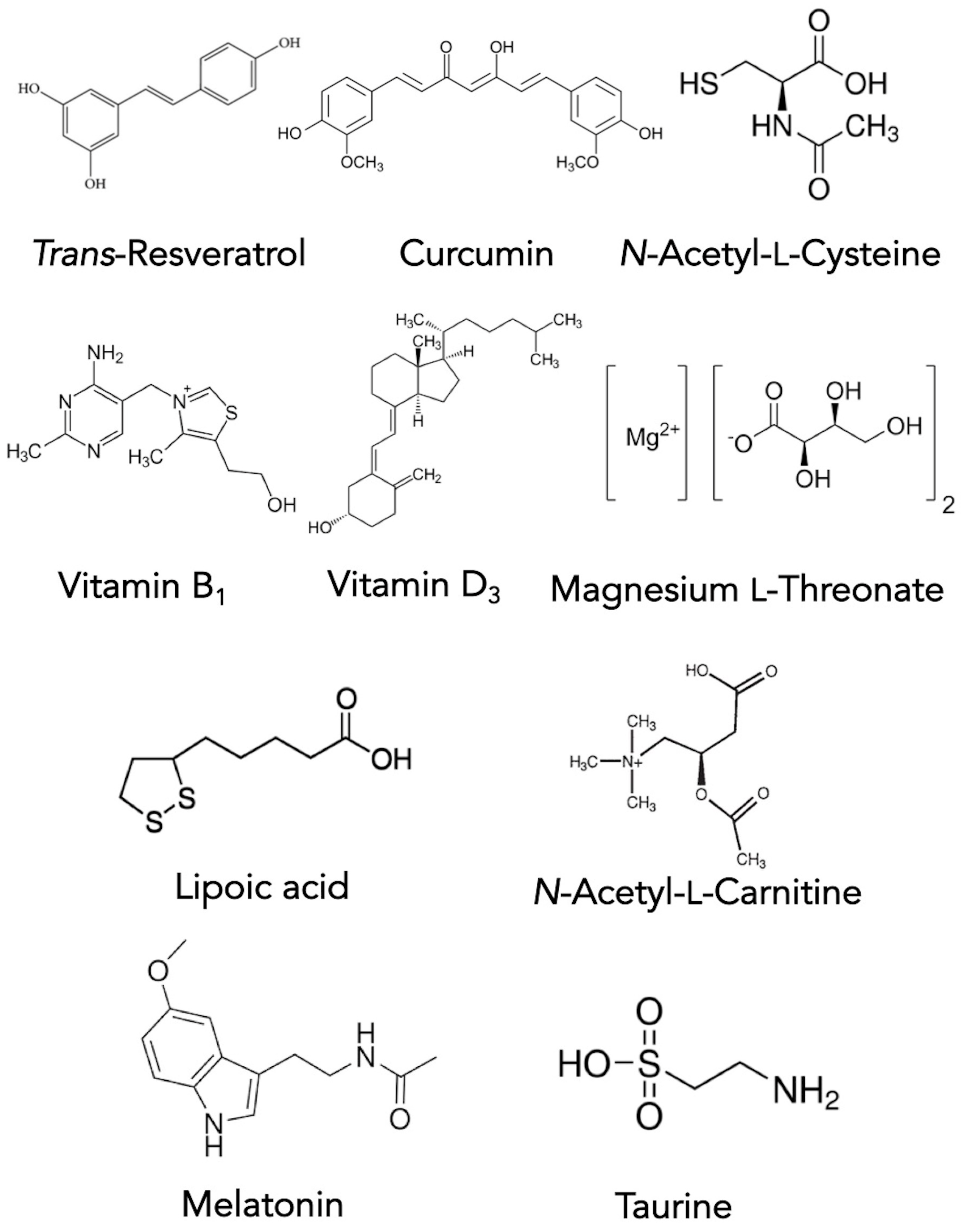
2.4. Restorative Options for Treating PD
2.5. Maintenance Options for Treating PD
2.6. Surgery Options for Treating PD
3. PD Clinical Trials in Progress and Novel (and Emerging) PD Therapies
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Armstrong, M.J.; Okun, M.S. Diagnosis and treatment of parkinson disease: A review. J. Am. Med. Assoc. 2020, 323, 548–560. [Google Scholar] [CrossRef] [PubMed]
- Simon, D.K.; Tanner, C.M.; Brundin, P. Parkinson disease epidemiology, pathology, genetics, and pathophysiology. Clin. Geriatr. Med. 2020, 36, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kalia, L.; Lang, A.E. Parkinson’s disease. Lancet 2015, 386, 896–912. [Google Scholar] [CrossRef]
- Werner, P.; Seppi, K.; Tanner, C.M.; Halliday, G.M.; Brundin, P.; Volkmann, J.; Schrag, A.E.; Lang, A.E. Parkinson disease. Nat. Rev. Dis. Primers 2017, 3, 17013. [Google Scholar]
- Ahlskog, J.E. The New Parkinson’s Disease Treatment Book: Partnering with Your Doctor to Get the Most from Your Medications; Oxford University Press: Oxford, UK, 2015. [Google Scholar]
- Santens, P.; Boon, P.; Van Roost, D.; Caemaert, J. The pathophysiology of motor symptoms in Parkinson’s disease. Acta Neurol. Belg. 2003, 103, 129–134. [Google Scholar]
- Jankovic, J. Motor fluctuations and dyskinesias in Parkinson’s disease: Clinical manifestations. Mov. Disord. 2005, 20, S11–S16. [Google Scholar] [CrossRef]
- Ferrazzoli, D.; Ortelli, P.; Cucca, A.; Bakdounes, L.; Canesi, M.; Volpe, D. Motor-cognitive approach and aerobic training: A synergism for rehabilitative intervention in Parkinson’s disease. Neurodegener. Dis. Manag. 2020, 10, 41–55. [Google Scholar] [CrossRef]
- Berganzo, K.; Tijero, B.; González-Eizaguirre, A.; Somme, J.; Lezcano, E.; Gabilondo, I.; Fernandez, M.; Zarranz, J.; Gómez-Esteban, J. Motor and non-motor symptoms of Parkinson’s disease and their impact on quality of life and on different clinical subgroups. Neurología 2016, 31, 585–591. [Google Scholar] [CrossRef]
- Crowley, E.K.; Nolan, Y.M.; Sullivan, A.M. Exercise as a therapeutic intervention for motor and non-motor symptoms in Parkinson’s disease: Evidence from rodent models. Prog. Neurobiol. 2019, 172, 2–22. [Google Scholar] [CrossRef]
- Carapellotti, A.M.; Stevenson, R.; Doumas, M. The efficacy of dance for improving motor impairments, non-motor symptoms, and quality of life in Parkinson’s disease: A systematic review and meta-analysis. PLoS ONE 2020, 15, e0236820. [Google Scholar] [CrossRef]
- Amara, A.W.; Memon, A.A. Effects of exercise on non-motor symptoms in Parkinson’s disease. Clin. Ther. 2018, 40, 8–15. [Google Scholar] [CrossRef]
- Bandres-Ciga, S.; Diez-Fairen, M.; Kim, J.J.; Singleton, A.B. Genetics of Parkinson’s disease: An introspection of its journey towards precision medicine. Neurobiol. Dis. 2020, 137, 104782. [Google Scholar] [CrossRef]
- Connolly, B.S.; Lang, A.E. Pharmacological treatment of Parkinson disease: A review. J. Am. Med. Assoc. 2014, 311, 1670–1683. [Google Scholar] [CrossRef]
- Ahlskog, J.E. Cheaper, simpler, and better: Tips for treating seniors with Parkinson disease. Mayo Clin. Proc. 2011, 86, 1211–1216. [Google Scholar] [CrossRef]
- Hall, M.-F.E.; Church, F.C. Integrative medicine and health therapy for Parkinson disease. Top. Geriatr. Rehabil. 2020, 36, 176–186. [Google Scholar] [CrossRef]
- HP-200 in Parkinson’s Disease Study Group. An alternative medicine treatment for Parkinson’s disease: Results of a multicenter clinical trial. J. Altern. Complement. Med. 1995, 1, 249–255. [Google Scholar] [CrossRef]
- Bishop, F.L.; Yardley, L.; Lewith, G.T. Systematic review of beliefs involved in the use of complementary and alternative medicine. J. Health Psychol. 2007, 12, 851–867. [Google Scholar] [CrossRef]
- Bega, D.; Gonzalez-Latapi, P.; Zadikoff, C.; Simuni, S. A review of the clinical evidence for complementary and alternative therapies in Parkinson’s disease. Curr. Treat. Options Neurol. 2014, 16, 314. [Google Scholar] [CrossRef]
- Seung-Nam, K.; Wang, X.; Park, H.-J. Integrative approach to Parkinson’s disease. Front. Aging Neurosci. 2019, 11, 339. [Google Scholar]
- Pickut, B.A.; Mischley, L.K.; Reversa, R.J. Integrative medicine and Parkinson’s disease. Integr. Neurol. 2020, 7, 141–183. [Google Scholar]
- Ahlskog, J.E. Does vigorous exercise have a neuroprotective effect in Parkinson disease? Neurology 2011, 77, 288–294. [Google Scholar] [CrossRef]
- Crowley, E.K.; Nolan, Y.M.; Sullivan, A.M. Exercise as therapy for Parkinson’s? Aging 2018, 10, 1536. [Google Scholar] [CrossRef]
- David, F.J.; Rafferty, M.R.; Robichaud, J.A.; Prodoehl, J.; Kohrt, W.M.; Vaillancourt, D.E.; Corcos, D.M. Progressive resistance exercise and Parkinson’s disease: A review of potential mechanisms. Parkinson’s Dis. 2012, 124527. [Google Scholar] [CrossRef]
- De Carvalho, A.O.; Filho, A.S.S.; Murillo-Rodriguez, E.; Rocha, N.B.; Carta, M.G.; Machado, S. Physical exercise for Parkinson’s disease: Clinical and experimental evidence. Clin. Pract. Epidemiol. Ment. Health 2018, 14, 89. [Google Scholar]
- Ebersbach, G.; Grust, U.; Ebersbach, A.; Wegner, B.; Gandor, F.; Kühn, A.A. Amplitude-oriented exercise in Parkinson’s disease: A randomized study comparing LSVT-BIG and a short training protocol. J. Neural Transm. 2015, 122, 253–256. [Google Scholar] [CrossRef]
- El-Sayes, J.; Harasym, D.; Turco, C.V.; Locke, M.B.; Nelson, A.J. Exercise-induced neuroplasticity: A mechanistic model and prospects for promoting plasticity. Neuroscientist 2019, 25, 65–85. [Google Scholar] [CrossRef]
- Ellis, T.; Rochester, L. Mobilizing Parkinson’s disease: The future of exercise. J. Parkinson’s Dis. 2018, 8, S95–S100. [Google Scholar] [CrossRef]
- Fisher, B.E.; Petzinger, G.M.; Nixon, K.; Hogg, E.; Bremmer, S.; Meshul, C.K.; Jakowec, M.W. Exercise-induced behavioral recovery and neuroplasticity in the 1-Methyl-4-Phenyl-1, 2, 3, 6-tetrahydropyridine-lesioned mouse basal ganglia. J. Neurosci. Res. 2004, 77, 378–390. [Google Scholar] [CrossRef]
- Fisher, B.E.; Wu, A.D.; Salem, G.J.; Song, J.E.; Lin, C.-H.; Yip, J.; Cen, S.; Gordon, J.; Jakowec, M.; Petzinger, G. The effect of exercise training in improving motor performance and corticomotor excitability in people with early Parkinson’s disease. Arch. Phys. Med. Rehabil. 2008, 89, 1221–1229. [Google Scholar] [CrossRef]
- Hirsch, M.A.; Farley, B.G. Exercise and neuroplasticity in persons living with Parkinson’s disease. Eur. J. Phys. Rehab. Med. 2009, 45, 215–229. [Google Scholar]
- Jang, Y.; Koo, J.-H.; Kwon, I.; Kang, E.-B.; Um, H.-S.; Soya, H.; Lee, Y.; Cho, J.-Y. Neuroprotective effects of endurance exercise against neuroinflammation in MPTP-induced Parkinson’s disease mice. Brain Res. 2017, 1655, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Johansson, H.; Freidle, M.; Ekman, U.; Schalling, E.; Leavy, B.; Svenningsson, P.; Hagströmer, M.; Franzén, E. Feasibility aspects of exploring exercise-induced neuroplasticity in Parkinson’s disease: A pilot randomized controlled trial. Parkinson’s Dis. 2020, 2020. [Google Scholar] [CrossRef] [PubMed]
- Paillard, T.; Rolland, Y.; Barreto, P.D.S. Protective effects of physical exercise in Alzheimer’s disease and Parkinson’s disease: A narrative review. J. Clin. Neurol. 2015, 11, 212. [Google Scholar] [CrossRef] [PubMed]
- Palasz, E.; Niewiadomski, W.; Gasiorowska, A.; Wysocka, A.; Stepniewska, A.; Niewiadomska, G. Exercise-induced neuroprotection and recovery of motor function in animal models of Parkinson’s disease. Front. Neurol. 2019, 10, 1143. [Google Scholar] [CrossRef]
- Palmer, S.S.; A Mortimer, J.; Webster, D.D.; Bistevins, R.; Dickinson, G.L. Exercise therapy for Parkinson’s disease. Arch. Phys. Med. Rehabil. 1986, 67, 741–745. [Google Scholar] [CrossRef]
- Petzinger, G.M.; Fisher, B.E.; McEwen, S.; Beeler, J.A.; Walsh, J.P.; Jakowec, M.W. Exercise-enhanced neuroplasticity targeting motor and cognitive circuitry in Parkinson’s disease. Lancet Neurol. 2013, 12, 716–726. [Google Scholar] [CrossRef]
- Petzinger, G.M.; Fisher, B.E.; Van Leeuwen, J.-E.; Vukovic, M.; Akopian, G.; Meshul, C.K.; Holschneider, D.P.; Nacca, A.; Walsh, J.P.; Jakowec, M.W. Enhancing neuroplasticity in the basal ganglia: The role of exercise in Parkinson’s disease. Mov. Disord. 2010, 25, S141–S145. [Google Scholar] [CrossRef]
- Rafferty, M.R.; Prodoehl, J.; Robichaud, J.A.; David, F.J.; Poon, C.; Goelz, L.C.; Vaillancourt, D.E.; Kohrt, W.M.; Comella, C.L.; Corcos, D.M. Effects of two years of exercise on gait impairment in people with Parkinson’s Disease: The PRET-PD randomized trial. J. Neurol. Phys. 2017, 41, 21. [Google Scholar]
- Rafferty, M.R.; Schmidt, P.N.; Luo, S.T.; Li, K.; Marras, C.; Davis, T.L.; Guttman, M.; Cubillos, F.; Simuni, T.; on behalf of all NPF-QII Investigators. Regular exercise, quality of life, and mobility in Parkinson’s disease: A longitudinal analysis of national Parkinson foundation quality improvement initiative data. J. Parkinson’s Dis. 2017, 7, 193–202. [Google Scholar] [CrossRef]
- Schenkman, M.; Moore, C.G.; Kohrt, W.M.; Hall, D.A.; Delitto, A.; Comella, C.L.; Josbeno, D.A.; Christiansen, C.L.; Berman, B.D.; Kluger, B.M. Effect of high-intensity treadmill exercise on motor symptoms in patients with de novo Parkinson disease: A phase 2 randomized clinical trial. J. Am. Med. Assoc. Neurol. 2018, 75, 219–226. [Google Scholar] [CrossRef]
- Silveira, C.R.; Roy, E.A.; Intzandt, B.N.; Almeida, Q.J. Aerobic exercise is more effective than goal-based exercise for the treatment of cognition in Parkinson’s disease. Brain Cogn. 2018, 122, 1–8. [Google Scholar] [CrossRef]
- Bryans, L.A.; Palmer, A.D.; Anderson, S.; Schindler, J.; Graville, D.J. The impact of Lee Silverman Voice Treatment (Lsvt Loud®) on voice, communication, and participation: Findings from a prospective, longitudinal study. J. Commun. Disord. 2020, 89, 106031. [Google Scholar] [CrossRef]
- Foltynie, T.; Grover, T. Not only loud but also intelligible. EClinicalMedicine 2020, 24, 100456. [Google Scholar] [CrossRef]
- Yuan, F.; Guo, X.; Wei, X.; Xie, F.; Zheng, J.; Huang, Y.; Huang, Z.; Chang, Z.; Li, H.; Guo, Y.; et al. Lee Silverman Voice Treatment for dysarthria in patients with Parkinson’s disease: A systematic review and meta-analysis. Eur. J. Neurol. 2020, 27, 1957–1970. [Google Scholar] [CrossRef]
- Farley, B.G.; Fox, C.M.; Ramig, L.O.; McFarland, D.H. Intensive amplitude-specific therapeutic approaches for Parkinson’s disease: Toward a neuroplasticity-principled rehabilitation model. Top. Geriatr. Rehabil. 2008, 24, 99–114. [Google Scholar] [CrossRef]
- Farley, B.G.; Koshland, G.F. Training BIG to move faster: The application of the speed–amplitude relation as a rehabilitation strategy for people with Parkinson’s disease. Exp. Brain Res. 2005, 167, 462–467. [Google Scholar] [CrossRef]
- Ebersbach, G.; Ebersbach, A.; Edler, D.; Kaufhold, O.; Kusch, M.; Kupsch, A.; Wissel, J. Comparing exercise in Parkinson’s disease—The Berlin LSVT®BIG study. Mov. Disord. 2010, 25, 1902–1908. [Google Scholar] [CrossRef]
- Fox, C.; Ebersbach, G.; Ramig, L.; Sapir, S. LSVT LOUD and LSVT BIG: Behavioral treatment programs for speech and body movement in Parkinson disease. Parkinson’s Dis. 2012, 2012. [Google Scholar] [CrossRef]
- LaMotte, G.; Rafferty, M.R.; Prodoehl, J.; Kohrt, W.M.; Comella, C.L.; Simuni, T.; Corcos, D.M. Effects of endurance exercise training on the motor and non-motor features of Parkinson’s disease: A review. J. Parkinson’s Dis. 2015, 5, 21–41. [Google Scholar] [CrossRef]
- van der Kolk, N.M.; de Vries, N.M.; Kessels, R.P.C.; Joosten, H.; Zwinderman, A.H.; Post, B.; Bloem, B.R. Effectiveness of home-based and remotely supervised aerobic exercise in Parkinson’s disease: A double-blind, randomised controlled trial. Lancet Neurol. 2019, 18, 998–1008. [Google Scholar] [CrossRef]
- Ridgel, A.L.; Vitek, J.L.; Alberts, J.L. Forced, not voluntary, exercise improves motor function in Parkinson’s disease patients. Neurorehabilit. Neural Repair 2009, 23, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Elsworth, J.D.; Roth, R.H. Dopamine synthesis, uptake, metabolism, and receptors: Relevance to gene therapy of Parkinson’s disease. Exp. Neurol. 1997, 144, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Cookson, M.R. The biochemistry of Parkinson’s disease. Annu. Rev. Biochem. 2005, 74, 29–52. [Google Scholar] [CrossRef] [PubMed]
- Fox, S.H.; Katzenschlager, R.; Lim, S.-Y.; Barton, B.; De Bie, R.M.A.; Seppi, K.; Coelho, M.; Sampaio, C.; on behalf of the Movement Disorder Society Evidence-Based Medicine Committee. International Parkinson and movement disorder society evidence-based medicine review: Update on treatments for the motor symptoms of Parkinson’s disease. Mov. Disord. 2018, 33, 1248–1266. [Google Scholar] [CrossRef]
- Abbott, A. Levodopa: The story so far. Nat. Cell Biol. 2010, 466, S6–S7. [Google Scholar] [CrossRef]
- Cotzias, G.C.; Papavasiliou, P.S.; Gellene, R. Modification of Parkinsonism—Chronic treatment with L-Dopa. N. Engl. J. Med. 1969, 280, 337–345. [Google Scholar] [CrossRef]
- Lees, A.J.; Tolosa, E.; Olanow, C.W. Four pioneers of L-dopa treatment: Arvid Carlsson, Oleh Hornykiewicz, George Cotzias, and Melvin Yahr. Mov. Disord. 2015, 30, 19–36. [Google Scholar] [CrossRef]
- Ciurleo, R.; Corallo, F.; Bonanno, L.; Buono, V.L.; Di Lorenzo, G.; Versaci, R.; Allone, C.; Palmeri, R.; Bramanti, P.; Marino, S. Assessment of Duodopa® effects on quality of life of patients with advanced Parkinson’s disease and their caregivers. J. Neurol. 2018, 265, 2005–2014. [Google Scholar] [CrossRef]
- Warren Olanow, C.; Espay, A.J.; Stocchi, F.; Ellenbogen, A.L.; Leinonen, M.; Adar, L.; Case, R.J.; Fuchs Orenbach, S.; Yardeni, T.; Oren, S.; et al. Continuous subcutaneous levodopa delivery for Parkinson’s disease: A randomized study. J. Parkinson’s Dis. 2020, 11, 177–186. [Google Scholar] [CrossRef]
- Ellenbogen, A.; Stocchi, F.; Espay, A.; Poewe, W.; Oren, S.; Case, R.; Olanow, C.W. Impact of Subcutaneous Levodopa Infusion with ND0612 on Patient Reported Outcomes (4506). Neurology 2021, 94, 4506. [Google Scholar]
- Braksick, S.A.; Nasr, D.M. Neurological emergencies from prescription drugs. In Neurological Emergencies in Clinical Practice; Springer: Cham, Switzerland, 2019; pp. 301–318. [Google Scholar]
- Ahlskog, J.E. Common myths and misconceptions that sidetrack Parkinson disease treatment, to the detriment of patients. Mayo Clin. Proc. 2020, 95, 2225–2234. [Google Scholar] [CrossRef]
- Stowe, R.; Ives, N.; Clarke, C.E.; Van Hilten, J.; Ferreira, J.; Hawker, R.J.; Shah, L.; Wheatley, K.; Gray, R. Dopamine agonist therapy in early Parkinson’s disease. Cochrane Database Syst. Rev. 2008, CD006564. [Google Scholar] [CrossRef]
- Carbone, F.; Djamshidian, A.; Seppi, K.; Poewe, W. Apomorphine for Parkinson’s disease: Efficacy and safety of current and new formulations. CNS Drugs 2019, 33, 905–918. [Google Scholar] [CrossRef]
- Foley, P.; Gerlach, M.; Youdim, M.; Riederer, P. MAO-B inhibitors: Multiple roles in the therapy of neurodegenerative disorders? Parkinsonism Relat. Disord. 2000, 6, 25–47. [Google Scholar] [CrossRef]
- Przuntek, H.; Conrad, B.; Dichgans, J.; Kraus, P.; Krauseneck, P.; Pergande, G.; Rinne, U.; Schimrigk, K.; Schnitker, J.; Vogel, H. SELEDO: A 5-year long-term trial on the effect of selegiline in early parkinsonian patients treated with levodopa. Eur. J. Neurol. 1999, 6, 141–150. [Google Scholar] [CrossRef]
- Männistö, P.T.; Kaakkola, S. Catechol-O-methyltransferase (COMT): Biochemistry, molecular biology, pharmacology, and clinical efficacy of the new selective COMT inhibitors. Pharmacol. Rev. 1999, 51, 593–628. [Google Scholar]
- Männistö, P.T.; Ulmanen, I.; Lundström, K.; Taskinen, J.; Tenhunen, J.; Tilgmann, C.; Kaakkola, S. Characteristics of catechol O-methyltransferase (COMT) and properties of selective COMT inhibitors. In Progress in Drug Research/Fortschritte der Arzneimittelforschung/Progrès des Recherches Pharmaceutiques; Springer: Berlin/Heidelberg, Germany, 1992; Volume 39, pp. 291–350. [Google Scholar]
- Schwab, R.S.; England, A.C.; Poskanzer, D.C.; Young, R.R. Amantadine in the treatment of Parkinson’s disease. J. Am. Med. Assoc. 1969, 208, 1168–1170. [Google Scholar] [CrossRef]
- Santiago, P.L.; Rascol, R. Efficacy and safety of Amantadine for the treatment of L-Dopa-induced Dyskinesia. J. Neural Transm. 2018, 125, 1237–1250. [Google Scholar]
- Agid, Y.; Arnulf, I.; Bejjani, P.; Bloch, F.; Bonnet, A.M.; Damier, P.; Dubois, B.; François, C.; Houeto, J.L.; Iacono, D.; et al. Parkinson’s disease is a neuropsychiatric disorder. Adv. Neurol. 2003, 91, 365–370. [Google Scholar]
- Castrioto, A.; Thobois, S.; Carnicella, S.; Maillet, A.; Krack, P. Emotional manifestations of PD: Neurobiological basis. Mov. Disord. 2016, 31, 1103–1113. [Google Scholar] [CrossRef]
- Rieu, I.; Martinez-Martin, P.; Pereira, B.; De Chazeron, I.; Metman, L.V.; Jahanshahi, M.; Ardouin, C.; Chéreau, I.; Brefel-Courbon, C.; Ory-Magne, F.; et al. International validation of a behavioral scale in Parkinson’s disease without dementia. Mov. Disord. 2015, 30, 705–713. [Google Scholar] [CrossRef] [PubMed]
- Rieu, I.; Houeto, J.L.; Pereira, B.; De Chazeron, I.; Bichon, A.; Chéreau, I.; Ulla, M.; Brefel-Courbon, C.; Ory-Magne, F.; Dujardin, K.; et al. Impact of Mood and Behavioral Disorders on Quality of Life in Parkinson’s disease. J. Parkinson’s Dis. 2016, 6, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Weintraub, D.; David, A.S.; Evans, A.H.; Grant, J.E.; Stacy, M. Clinical spectrum of impulse control disorders in Parkinson’s disease. Mov. Disord. 2015, 30, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Sierra, M.; Carnicella, S.; Strafella, A.P.; Bichon, A.; Lhommée, E.; Castrioto, A.; Chabardes, S.; Thobois, S.; Krack, P. Apathy and Impulse Control Disorders: Yin & Yang of Dopamine Dependent Behaviors. J. Parkinson’s Dis. 2015, 5, 625–636. [Google Scholar] [CrossRef]
- Zhang, J.-F.; Wang, X.-X.; Feng, Y.; Fekete, R.; Jankovic, J.; Wu, Y.-C. Impulse Control Disorders in Parkinson’s disease: Epidemiology, Pathogenesis and Therapeutic Strategies. Front. Psychiatry 2021, 12, 97. [Google Scholar] [CrossRef]
- Seppi, K.; Ray Chaudhuri, K.; Coelho, M.; Fox, S.H.; Katzenschlager, R.; Perez Lloret, S.; Weintraub, D.; Sampaio, C.; Collaborators of the Parkinson’s disease Update on Non-Motor Symptoms Study Group on behalf of the Movement Disorders Society Evidence-Based Medicine Committee. Update on treatments for nonmotor symptoms of Parkinson’s disease—An evidence-based medicine review. Mov. Disord. 2019, 34, 180–198. [Google Scholar] [CrossRef]
- Chaudhuri, K.R.; Healy, D.G.; Schapira, A.H. Non-motor symptoms of Parkinson’s disease: Diagnosis and management. Lancet Neurol. 2006, 5, 235–245. [Google Scholar] [CrossRef]
- Chaudhuri, K.R.; Schapira, A.H.V. Non-motor symptoms of Parkinson’s disease: Dopaminergic pathophysiology and treatment. Lancet Neurol. 2009, 8, 464–474. [Google Scholar] [CrossRef]
- Schapira, A.H.V.; Chaudhuri, K.R.; Jenner, P. Non-motor features of Parkinson disease. Nat. Rev. Neurosci. 2017, 18, 435–450. [Google Scholar] [CrossRef]
- Vuletić, V. Non-motor symptoms in Parkinson’s disease. In Mind and Brain; Springer International Publishing: Cham, Switzerland, 2020; pp. 109–118. [Google Scholar] [CrossRef]
- Pontone, G.M.; Mills, K.A. Optimal treatment of depression and anxiety in Parkinson’s disease. Am. J. Geriatr. Psychiatry 2021, in press. [Google Scholar] [CrossRef]
- Martimbianco, A.L.C.; Prosdocimi, F.C.; Anauate-Netto, C.; dos Santos, E.M.; Mendes, G.D.; Fragoso, Y.D. Evidence-based recommendations for the oral health of patients with Parkinson’s disease. Neurology 2021, 1–10. [Google Scholar] [CrossRef]
- Skjærbæk, C.; Knudsen, K.; Horsager, J.; Borghammer, P. Gastrointestinal dysfunction in Parkinson’s disease. J. Clin. Med. 2021, 10, 493. [Google Scholar] [CrossRef]
- Rashid, N.; Shim, A.; Andes, S.; Quale, S.; Abler, V. Treatment patterns with antipsychotics in long-term care patients with Parkinson’s disease psychosis. J. Appl. Gerontol. 2021, 0733464820987032. [Google Scholar] [CrossRef]
- Naasan, G.; Shdo, S.M.; Rodriguez, E.M.; Spina, S.; Grinberg, L.; Lopez, L.; Karydas, A.; Seeley, W.W.; Miller, B.L.; Rankin, K.P. Psychosis in neurodegenerative disease: Differential patterns of hallucination and delusion symptoms. Brain 2021, 144, 999–1012. [Google Scholar] [CrossRef]
- Pakkenberg, H.; Pakkenberg, B. Clozapine in the treatment of tremor. Acta Neurol. Scand. 1986, 73, 295–297. [Google Scholar] [CrossRef]
- Friedman, J.H.; Lannon, M.C. Clozapine-responsive tremor in Parkinson’s disease. Mov. Disord. 1990, 5, 225–229. [Google Scholar] [CrossRef]
- Keir, L.H.M.; Breen, D.P. New awakenings: Current understanding of sleep dysfunction and its treatment in Parkinson’s disease. J. Neurol. 2020, 267, 288–294. [Google Scholar] [CrossRef]
- Bhalke, R.D.; Giri, M.A.; Anil, R.Y.; Balasaheb, N.M.; Nanasaheb, P.A.; Pande, V.V. Hypotension: A comprehensive review. J. Pharmacogn. Phytochem. 2021, 10, 1945–1947. [Google Scholar]
- Idiaquez, J.F.; Casar, J.C.; Biaggioni, I. Neurogenic orthostatic hypotension. Lessons from synucleinopathies. Am. J. Hypertens. 2021, 34, 125–133. [Google Scholar] [CrossRef]
- Gupta, A.; LaFaver, K.; Duque, K.R.; Lingaiah, A.; Meriwether, K.V.; Gaskins, J.; Gomes, J.; Espay, A.J.; Mahajan, A. Pelvic floor health in women with Parkinson’s disease. J. Parkinson’s Dis. 2021, 11, 857–864. [Google Scholar] [CrossRef]
- Osaki, Y.; Morita, Y.; Miyamoto, Y.; Furushima, T.; Furuta, K.; Furuya, H. Disease progression and phenotypes of non-motor symptoms in Parkinson’s disease. Neurol. Clin. Neurosci. 2021, 9, 83–90. [Google Scholar] [CrossRef]
- Oldfield, V.; Keating, G.M.; Perry, C.M. Rasagiline. Drugs 2007, 67, 1725–1747. [Google Scholar] [CrossRef] [PubMed]
- Parkinson Study Group. DATATOP: A multicenter controlled clinical trial in early Parkinson’s disease. Arch. Neurol. 1989, 46, 1052–1060. [Google Scholar] [CrossRef] [PubMed]
- Shoulson, I.; Parkinson Study Group. DATATOP: A decade of neuroprotective inquiry. Ann. Neurol. 1998, 44, S160–S166. [Google Scholar] [CrossRef]
- Rascol, O.; Fitzer-Attas, C.J.; Hauser, R.; Jankovic, J.; Lang, A.; Langston, J.W.; Melamed, E.; Poewe, W.; Stocchi, F.; Tolosa, E.; et al. A double-blind, delayed-start trial of rasagiline in Parkinson’s disease (the ADAGIO study): Prespecified and post-hoc analyses of the need for additional therapies, changes in UPDRS scores, and non-motor outcomes. Lancet Neurol. 2011, 10, 415–423. [Google Scholar] [CrossRef]
- Siderowf, A.; Stern, M.; Shoulson, I.; Kieburtz, K.; Oakes, D.; Day, D.; Shinaman, A.; Plumb, S.; Fahn, S.; Blindauer, K. A controlled trial of rasagiline in early Parkinson disease: The tempo study. Arch. Neurol. 2002, 59, 1937–1943. [Google Scholar]
- Reish, A.; Heather, E.; Standaert, D. Role of α-synuclein in inducing innate and adaptive immunity in Parkinson disease. J. Parkinson’s Dis. 2005, 5, 1–19. [Google Scholar]
- Kannarkat, G.T.; Boss, J.M.; Tansey, M.G. The role of innate and adaptive immunity in Parkinson’s disease. J. Parkinson’s Dis. 2013, 3, 493–514. [Google Scholar] [CrossRef]
- Salim, S.; Sarraj, N.; Taneja, M.; Saha, K.; Tejada-Simon, M.V.; Chugh, G. Moderate treadmill exercise prevents oxidative stress-induced anxiety-like behavior in rats. Behav. Brain Res. 2010, 208, 545–552. [Google Scholar] [CrossRef]
- Chen, W.; Wang, H.J.; Shang, N.N.; Liu, J.; Li, J.; Tang, D.H.; Li, Q. Moderate intensity treadmill exercise alters food preference via dopaminergic plasticity of ventral tegmental area-nucleus accumbens in obese mice. Neurosci. Lett. 2017, 641, 56–61. [Google Scholar] [CrossRef]
- Zhou, W.; Barkow, J.C.; Freed, C.R. Running wheel exercise reduces α-synuclein aggregation and improves motor and cognitive function in a transgenic mouse model of Parkinson’s disease. PLoS ONE 2017, 12, e0190160. [Google Scholar] [CrossRef]
- Sacheli, M.A.; Murray, D.K.; Vafai, N.; Cherkasova, M.V.; Dinelle, K.; Shahinfard, E.; Neilson, N.; McKenzie, J.; Schulzer, M.; Appel-Cresswell, S. Habitual exercisers versus sedentary subjects with Parkinson’s disease: Multimodal Pet and fMRI study. Mov. Disord. 2018, 33, 1945–1950. [Google Scholar] [CrossRef]
- Hall, M.-F.E.; Church, F.C. Exercise for older adults improves the quality of life in Parkinson’s disease and potentially enhances the immune response to COVID-19. Brain Sci. 2020, 10, 612. [Google Scholar] [CrossRef]
- Van der Heide, A.; Speckens, A.E.; Meinders, M.J.; Rosenthal, L.S.; Bloem, B.R.; Helmich, R.C. Stress and mindfulness in Parkinson’s disease—A survey in 5000 patients. NPJ Parkinson’s Dis. 2021, 7, 1–10. [Google Scholar]
- Van der Heide, A.; Meinders, M.J.; Speckens, A.E.M.; Peerbolte, T.F.; Bloem, B.R.; Helmich, R.C. Stress and mindfulness in Parkinson’s disease: Clinical effects and potential underlying mechanisms. Mov. Disord. 2021, 36, 64–70. [Google Scholar] [CrossRef]
- Rodríguez, M.Á.; Albillos-Almaraz, L.; López-Aguado, I.; Crespo, I.; Del Valle, M.; Olmedillas, H. Vigorous aerobic exercise in the management of Parkinson disease: A systematic review. PM&R 2020. [Google Scholar] [CrossRef]
- Gronek, P.; Haas, A.N.; Czarny, W.; Podstawski, R.; Delabary, M.D.S.; Clark, C.C.; Boraczyński, M.; Tarnas, M.; Wycichowska, P.; Pawlaczyk, M.; et al. The mechanism of physical activity-induced amelioration of Parkinson’s disease: A narrative review. Aging Dis. 2021, 12, 192–202. [Google Scholar] [CrossRef]
- Giudice, J.; Taylor, J.M. Muscle as a paracrine and endocrine organ. Curr. Opin. Pharmacol. 2017, 34, 49–55. [Google Scholar] [CrossRef]
- Taylor, J.M. Editorial overview: Muscle and bone are highly effective communicators. Curr. Opin. Pharmacol. 2017, 34. [Google Scholar] [CrossRef]
- Severinsen, M.C.K.; Pedersen, B.K. Muscle–Organ Crosstalk: The Emerging Roles of Myokines. Endocr. Rev. 2020, 41, 594–609. [Google Scholar] [CrossRef]
- Bente Klarlund, P. Physical activity and muscle–brain crosstalk. Nat. Rev. Endocrinol. 2019, 15, 383–392. [Google Scholar]
- Tari, A.R.; Norevik, C.S.; Scrimgeour, N.R.; Kobro-Flatmoen, A.; Storm-Mathisen, J.; Bergersen, L.H.; Wrann, D.C.; Selbæk, G.; Kivipelto, M.; Moreira, J.B.N. Are the neuroprotective effects of exercise training systemically mediated? Prog. Cardiovasc. Dis. 2019, 62, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, N.H.; Stanya, K.J.; Hyde, A.L.; Chalom, M.M.; Alexander, R.K.; Liou, Y.-H.; Starost, K.A.; Gangl, M.R.; Jacobi, D.; Liu, S.; et al. Interleukin-13 drives metabolic conditioning of muscle to endurance exercise. Science 2020, 368. [Google Scholar] [CrossRef] [PubMed]
- Correia, J.C.; Ruas, J.L. Exercised cytokines promote endurance. Science 2020, 368, 470–471. [Google Scholar] [CrossRef]
- Hribar, C.A.; Cobbold, P.H.; Church, F.C. Potential role of vitamin D in the elderly to resist COVID-19 and to slow progression of Parkinson’s disease. Brain Sci. 2020, 10, 284. [Google Scholar] [CrossRef]
- Ding, H.; Dhima, K.; Lockhart, K.C.; Locascio, J.J.; Hoesing, A.N.; Duong, K.; Trisini-Lipsanopoulos, A.; Hayes, M.T.; Sohur, U.S.; Wills, A.-M.; et al. Unrecognized vitamin D3 deficiency is common in Parkinson disease: Harvard Biomarker Study. Neurology 2013, 81, 1531–1537. [Google Scholar] [CrossRef]
- Peterson, A.L.; Murchison, C.; Zabetian, C.; Leverenz, J.B.; Watson, G.S.; Montine, T.; Carney, N.; Bowman, G.L.; Edwards, K.; Quinn, J.F. Memory, mood, and vitamin D in persons with Parkinson’s disease. J. Parkinson’s Dis. 2013, 3, 547–555. [Google Scholar] [CrossRef]
- Sleeman, I.; Aspray, T.; Lawson, R.; Coleman, S.; Duncan, G.; Khoo, T.K.; Schoenmakers, I.; Rochester, L.; Burn, D.; Yarnall, A. The role of vitamin D in disease progression in early Parkinson’s disease. J. Parkinson’s Dis. 2017, 7, 669–675. [Google Scholar] [CrossRef]
- Ebel, H.; Günther, T. Magnesium metabolism: A review. Clin. Chem. Lab. Med. 1980, 18, 257–270. [Google Scholar] [CrossRef]
- Vink, R. Magnesium in the CNS: Recent advances and developments. Magnes. Res. 2016, 29, 95–101. [Google Scholar]
- Shen, Y.; Dai, L.; Tian, H.; Xu, R.; Li, F.; Li, Z.; Zhou, J.; Wang, L.; Dong, J.; Sun, L. Treatment of magnesium-L-threonate elevates the magnesium level in the cerebrospinal fluid and attenuates motor deficits and dopamine neuron loss in a mouse model of Parkinson’s disease. Neuropsychiatr. Dis. Treat. 2019, 15, 3143–3153. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Stojanovska, L.; Tangalakis, K.; Bosevski, M.; Apostolopoulos, V. Cognitive decline: A vitamin B perspective. Maturitas 2016, 93, 108–113. [Google Scholar] [CrossRef]
- Lu’O’ng, K.V.Q.; Nguyê∼n, L.T.H. Thiamine and Parkinson’s disease. J. Neurol. Sci. 2012, 316, 1–8. [Google Scholar] [CrossRef]
- Parkhomenko, Y.M.; Pavlova, A.S.; Mezhenskaya, O.A. Mechanisms responsible for the high sensitivity of neural cells to vitamin B1 deficiency. Neurophysiology 2016, 48, 429–448. [Google Scholar] [CrossRef]
- Wright, C.E.; Tallan, H.H.; Lin, Y.Y.; Gaull, G.E. Taurine: Biological update. Annu. Rev. Biochem. 1986, 55, 427–453. [Google Scholar] [CrossRef]
- Che, Y.; Hou, L.; Sun, F.; Zhang, C.; Liu, X.; Piao, F.; Zhang, D.; Li, H.; Wang, Q. Taurine protects dopaminergic neurons in a mouse Parkinson’s disease model through inhibition of microglial M1 polarization. Cell Death Dis. 2018, 9, 435. [Google Scholar] [CrossRef]
- Hou, L.; Che, Y.; Sun, F.; Wang, Q. Taurine protects noradrenergic locus coeruleus neurons in a mouse Parkinson’s disease model by inhibiting microglial M1 polarization. Amino Acids 2018, 50, 547–556. [Google Scholar] [CrossRef]
- Hu, S.; Maiti, P.; Ma, Q.; Zuo, X.; Jones, M.R.; Cole, G.M.; Frautschy, S.A. Clinical development of curcumin in neuro-degenerative disease. Expert Rev. Neurother. 2015, 15, 629–637. [Google Scholar] [CrossRef]
- Jiang, T.-F.; Zhang, Y.-J.; Zhou, H.-Y.; Wang, H.-M.; Tian, L.-P.; Liu, J.; Ding, J.-Q.; Chen, S.-D. Curcumin ameliorates the neurodegenerative pathology in A53T α-synuclein cell model of Parkinson’s disease through the downregulation of mTOR/p70S6K signaling and the recovery of macroautophagy. J. Neuroimmune Pharmacol. 2013, 8, 356–369. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-S.; Zhang, Z.-R.; Zhang, M.-M.; Sun, M.-X.; Wang, W.-W.; Xie, C.-L. Neuroprotective properties of curcumin in toxin-base animal models of Parkinson’s disease: A systematic experiment literatures review. BMC Complement. Altern. Med. 2017, 17, 412. [Google Scholar] [CrossRef] [PubMed]
- Phillipson, O.T. Management of the aging risk factor for Parkinson’s disease. Neurobiol. Aging 2014, 35, 847–857. [Google Scholar] [CrossRef] [PubMed]
- Phillipson, O.T. Inhibition of aging in Parkinson’s disease: A case study. J. Altern. Complement. Med. 2013, 19, 851. [Google Scholar] [CrossRef] [PubMed]
- Hagen, T.M.; Ingersoll, R.T.; Wehr, C.M.; Lykkesfeldt, J.; Vinarsky, V.; Bartholomew, J.C.; Song, M.-H.; Ames, B.N. Acetyl-l-carnitine fed to old rats partially restores mitochondrial function and ambulatory activity. Proc. Natl. Acad. Sci. USA 1998, 95, 9562–9566. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Atamna, H.; Kuratsune, H.; Ames, B.N. Delaying brain mitochondrial decay and aging with mitochondrial antioxidants and metabolites. Ann. N. Y. Acad. Sci. 2002, 959, 133–166. [Google Scholar] [CrossRef]
- Katz, M.; Won, S.J.; Park, Y.; Orr, A.; Jones, D.P.; Swanson, R.A.; Glass, G.A. Cerebrospinal fluid concentrations of N-Acetylcysteine after oral administration in Parkinson’s disease. Parkinsonism Relat. Disord. 2015, 21, 500–503. [Google Scholar] [CrossRef]
- Monti, D.A.; Zabrecky, G.; Kremens, D.; Liang, T.W.; Wintering, N.A.; Cai, J.; Wei, X.; Bazzan, A.J.; Zhong, L.; Bowen, B.; et al. N-Acetyl cysteine may support dopamine neurons in Parkinson’s disease: Preliminary clinical and cell line data. PLoS ONE 2016, 11, e0157602. [Google Scholar] [CrossRef]
- Ferretta, A.; Gaballo, A.; Tanzarella, P.; Piccoli, C.; Capitanio, N.; Nico, B.; Annese, T.; Di Paola, M.; Dell’Aquila, C.; De Mari, M.; et al. Effect of resveratrol on mitochondrial function: Implications in parkin-associated familiar Parkinson’s disease. Biochim. Biophys. Acta Mol. Basis Dis. 2014, 1842, 902–915. [Google Scholar] [CrossRef]
- Kulkarni, S.S.; Cantó, C. The molecular targets of resveratrol. Biochim. Biophys. Acta Mol. Basis Dis. 2015, 1852, 1114–1123. [Google Scholar] [CrossRef]
- Mayo, J.C.; Sainz, R.M.; Tan, D.X.; Antolín, I.; Rodríguez, C.; Reiter, R.J. Melatonin and Parkinson’s disease. Endocrine 2005, 27, 169–178. [Google Scholar] [CrossRef]
- Cernera, S.; Eisinger, R.S.; Wong, J.K.; Ho, K.W.D.; Lopes, J.L.; To, K.; Carbunaru, S.; Ramirez-Zamora, A.; Almeida, L.; Foote, K.D.; et al. Long-term Parkinson’s disease quality of life after staged DBS: STN vs GPi and first vs. second lead. NPJ Parkinson’s Dis. 2020, 6, 1–10. [Google Scholar] [CrossRef]
- Sharma, V.D.; Patel, M.; Miocinovic, S. Surgical treatment of Parkinson’s disease: Devices and lesion approaches. Neurotherapeutics 2020, 17, 1525–1538. [Google Scholar] [CrossRef]
- Artusi, C.A.; Lopiano, L.; Morgante, F. Deep brain stimulation selection criteria for Parkinson’s disease: Time to go beyond CAPSIT-PD. J. Clin. Med. 2020, 9, 3931. [Google Scholar] [CrossRef]
- Lozano, C.S.; Tam, J.; Lozano, A.M. The changing landscape of surgery for Parkinson’s disease. Mov. Disord. 2018, 33, 36–47. [Google Scholar] [CrossRef]
- Hariz, M. My 25 Stimulating years with DBS in Parkinson’s disease. J. Parkinson’s Dis. 2017, 7, S33–S41. [Google Scholar] [CrossRef]
- Dallapiazza, R.F.; De Vloo, P.; Fomenko, A.; Lee, D.J.; Hamani, C.; Munhoz, R.P.; Hodaie, M.; Lozano, A.M.; Fasano, A.; Kalia, S.K. Considerations for patient and target selection in deep brain stimulation surgery for Parkinson’s disease. Exon Publ. 2018, 145–160. [Google Scholar]
- Hitti, F.L.; Ramayya, A.G.; McShane, B.J.; Yang, A.I.; Vaughan, K.A.; Baltuch, G.H. Long-term outcomes following deep brain stimulation for Parkinson’s disease. J. Neurosurg. 2020, 132, 205–210. [Google Scholar] [CrossRef]
- Kurtis, M.M.; Rajah, T.; Delgado, L.F.; Dafsari, H.S. The effect of deep brain stimulation on the non-motor symptoms of Parkinson’s disease: A critical review of the current evidence. NPJ Parkinson’s Dis. 2017, 3, 1–12. [Google Scholar] [CrossRef]
- McFarthing, K.; Buff, S.; Rafaloff, G.; Dominey, T.; Wyse, R.K.; Stott, S.R. Parkinson’s disease drug therapies in the clinical trial pipeline: 2020. J. Parkinson’s Dis. 2020, 10, 757–774. [Google Scholar] [CrossRef]
- Kordower, J.H.; Freeman, T.B.; Snow, B.J.; Vingerhoets, F.J.; Mufson, E.J.; Sanberg, P.R.; Hauser, R.A.; Smith, D.A.; Nauert, G.M.; Perl, D.P.; et al. Neuropathological evidence of graft survival and striatal reinnervation after the transplantation of fetal mesencephalic tissue in a patient with Parkinson’s disease. N. Engl. J. Med. 1995, 332, 1118–1124. [Google Scholar] [CrossRef]
- Freed, C.R.; Greene, P.E.; Breeze, R.E.; Tsai, W.-Y.; DuMouchel, W.; Kao, R.; Dillon, S.; Winfield, H.; Culver, S.; Trojanowski, J.Q.; et al. Transplantation of embryonic dopamine neurons for severe Parkinson’s disease. N. Engl. J. Med. 2001, 344, 710–719. [Google Scholar] [CrossRef]
- Doi, D.; Magotani, H.; Kikuchi, T.; Ikeda, M.; Hiramatsu, S.; Yoshida, K.; Amano, N.; Nomura, M.; Umekage, M.; Morizane, A. Pre-clinical study of induced pluripotent stem cell-derived dopaminergic progenitor cells for Parkinson’s disease. Nat. Commun. 2020, 11, 1–14. [Google Scholar] [CrossRef]
- Stone, J.; Johnstone, D.; Mitrofanis, J. The helmet experiment in Parkinson’s disease: An observation of the mechanism of neuroprotection by near infra-red light. In Proceedings of the 9th WALT Congress, Gold Coast, QLD, Australia, 28–30 September 2012. [Google Scholar]
- Hamilton, C.L.; El Khoury, H.; Hamilton, D.; Nicklason, F.; Mitrofanis, J. Buckets: Early observations on the use of red and infrared light helmets in Parkinson’s disease patients. Photobiomodul. Photomed. Laser Surg. 2019, 37, 615–622. [Google Scholar] [CrossRef]
- Johnstone, D.M.; Emoro, C.; Estone, J.; Benabid, A.-L.; Emitrofanis, J. Turning on lights to stop neurodegeneration: The potential of near infrared light therapy in Alzheimer’s and Parkinson’s disease. Front. Neurosci. 2016, 9, 500. [Google Scholar] [CrossRef]
| Type Compound | Drug Name | Brand Name | Additional Description |
|---|---|---|---|
| Dopamine replacement | Carbidopa/Levodopa | Sinemet IR, Sinemet CR, Rytary, Duopa | Sustained-release capsules |
| Rytary | |||
| Duopa | Enteral suspension in jejunum | ||
| Inbrija | Enhaled powder of Levodopa alone (no carbidopa) | ||
| Dopamine agonist (DA) | Apomorphine | Apokyn | Nonergoline receptor |
| Kynmobi | antagonist | ||
| Pramipexole | Mirapex | Stimulates D2 dopamine receptors | |
| Mirapex ER | |||
| Ropinirole | Requip | Stimulates D2 dopamine receptor | |
| Requip XL | |||
| Rotigotine | Neupro | Transdermal patch | |
| MAO-B Inhibitors | Selegiline | Eldepryl | Blocks the breakdown of dopamine |
| Zelapar | |||
| Rasagiline | Azilect | ||
| Safinamide | Xadago | ||
| COMT Inhibitors | Opicapone | Ongentys | Catechol-O-methyl- transferase (COMT) inhibitors make more levodopa available for transport across the BBB |
| Entacapone | Comtan | ||
| Carbidopa, levodopa, and Entacapone | Stalevo | ||
| Tolcapone | Tasmar | ||
| Anticholinergic | Trihexyphenidyl | Cogentin | Provides relief from tremor |
| Benztropine mesylate | |||
| Anti-influenza drug | Amantadine | Symmetrel | Provides relief for most motor symptoms, but effect is short-term. |
| Gocovri | |||
| Symadine |
| Symptom/Type Compound | Drug Name | Brand Name | Additional Description |
|---|---|---|---|
| Depression and Anxiety | |||
| Benzodiazepine | Alprazolam | Xanax, Xanax XR, Niravam | Anxiety and panic |
| Clonazepam | Klonopin | Anxiety and panic | |
| Diazepam | Valium | Anxiety and panic | |
| Lorazepam | Ativan | Anxiety and panic | |
| Selective Serotonin Reuptake Inhibitors (SSRI) | Fluoxetine | Prozac | Depression, panic, anxiety |
| Sertraline | Zoloft | Depression, panic, anxiety | |
| Serotonin/Norepinephrine Reuptake Inhibitors (SNRI) | Duloxetine | Cymbalta | Depression and anxiety |
| Desvenlafaxin | Pristiq | Depression and anxiety | |
| Milnacipran | Savella | Depression and anxiety | |
| Venlafaxine | Mirapex | Depression and anxiety | |
| Effexor/Effexor XR | |||
| Tricylic compounds | Amitrytiline | Elavil | Depression, anxiety |
| Imipramine | Tofranil/Tofranil PM | Depression, anxiety | |
| Nortriptyline | Pamelor | Depression, anxiety | |
| Additional anti-anxiety | Buspirone | BuSpar | General anxiety |
| Propanolol | Inderal/Inderal LA | Panic attack/anxiety | |
| Quetiapine | Seroquel | Depression/anxiety | |
| Trazodone | Desyrel, Oleptro | Depression/anxiety | |
| Other anti-depressants | Bupropion | Wellbutrin SR/XL/SR/XL | Depression |
| Zyban | |||
| Mirtazapine | Remeron/SolTab | Depression | |
| Excessive Drooling | Atropine drops | Unwarranted drooling | |
| Botulinum toxin A | Xeomin | Unwarranted drooling | |
| Botulinum toxin B | Myobloc | Unwarranted drooling | |
| Glycopyrrolate | Unwarranted drooling | ||
| Scopolamine patch | Unwarranted drooling | ||
| Gastrointestinal Problems | |||
| Constipation | Lubiprostone | Amitiza | Constipation |
| Polyethylenene glycol | MiraLax | Constipation | |
| Nausea and Vomiting | Ondansetron | Zofran | Nausea, vomiting |
| Trimethobenzamide | Tigan | Nausea, vomiting |
| Symptom/Type Compound | Drug Name | Brand Name | Additional Description |
|---|---|---|---|
| Dementia | |||
| Acetylcholinesterase Inhibitors | Donepezil | Aricept | Dementia |
| Galantamine | Razadyne/ER | Dementia | |
| Rivastigmine | Exelon, Exelon Patch | Dementia | |
| Psychosis | |||
| Clozapine | Clozaril, FazoClo | Hallucinations/Psychosis | |
| Pimavanserin | Nuplazid | Hallucinations/Delusions | |
| Quetiapine | Seroquel, Seroquel SR | Hallucinations/Psychosis | |
| Sleeping Disorders | |||
| Amitriptyline | Elavil | Insomnia | |
| Clonazepam | Klonopin | REM Sleep Behavior Disorder | |
| Doxepin | Silenor | Insomnia | |
| Eszopicione | Lunestra | Insomnia | |
| Melatonin | Insomnia | ||
| Mirtazapine | Remeron | Insomnia | |
| Trazadone | Desyrel | Insomnia | |
| Cognition and Daytime Sleepiness | |||
| Methylphenidate | Concerta | Unable to focus, overly sleepy during the day, fatigue | |
| Daytrana Patch | |||
| Metadate CD | |||
| Methylin | |||
| Ritalin, Ritalin LA/SRF | |||
| Memantine | Namenda | PD related dementia | |
| Modafinil | Provigil | Unable to focus, sleepy during the day | |
| Orthostatic Hypotension | |||
| Fludrocortisone | Florinef | Neurogenic Orthostatic Hypotension | |
| Pyridostigmine | Mestinon | ||
| Droxidopa | Northera | ||
| Urinary Incontinence | |||
| Anticholinergics | Darifenacin | Enoblex | Overactive bladder |
| Oxybutynin | Ditropan/XL, Glenique, Oxytrol | Overactive bladder +/− Incontinence | |
| Solifenacin | Vesicare | Overactive bladder | |
| Tolterodine | Detrol/LA | Overactive bladder | |
| Beta-3-Agonist | Mirabegron | Mybetriq | Overactive bladder |
| Alpha-1A blockers | Alfuzosin | Uroxatral | Overactive bladder, benign prostatic hyperplasia (BPH) |
| Silodosin | Rapaflo | Overactive bladder, BPH | |
| Tamsulosin | Flomax | ||
| Terazosin | |||
| Serotonin, Norepinephrine Reuptake Inhibitors (SNRI) | Duloxetine | Cymbalta | Urinary incontinence from stress |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Church, F.C. Treatment Options for Motor and Non-Motor Symptoms of Parkinson’s Disease. Biomolecules 2021, 11, 612. https://doi.org/10.3390/biom11040612
Church FC. Treatment Options for Motor and Non-Motor Symptoms of Parkinson’s Disease. Biomolecules. 2021; 11(4):612. https://doi.org/10.3390/biom11040612
Chicago/Turabian StyleChurch, Frank C. 2021. "Treatment Options for Motor and Non-Motor Symptoms of Parkinson’s Disease" Biomolecules 11, no. 4: 612. https://doi.org/10.3390/biom11040612
APA StyleChurch, F. C. (2021). Treatment Options for Motor and Non-Motor Symptoms of Parkinson’s Disease. Biomolecules, 11(4), 612. https://doi.org/10.3390/biom11040612






