Xanthurenic Acid Is the Main Pigment of Trichonephila clavata Gold Dragline Silk
Abstract
1. Introduction
2. Materials and Methods
2.1. Pigment Extraction from Spider Silk
2.2. LC- ESI-MS and LC-APCI-MS Conditions
2.3. LC-UV Conditions
2.4. Quantitation of Xanthurenic Acid
2.5. Antibacterial Activity Test
2.6. UV Tolerance Assay
3. Results
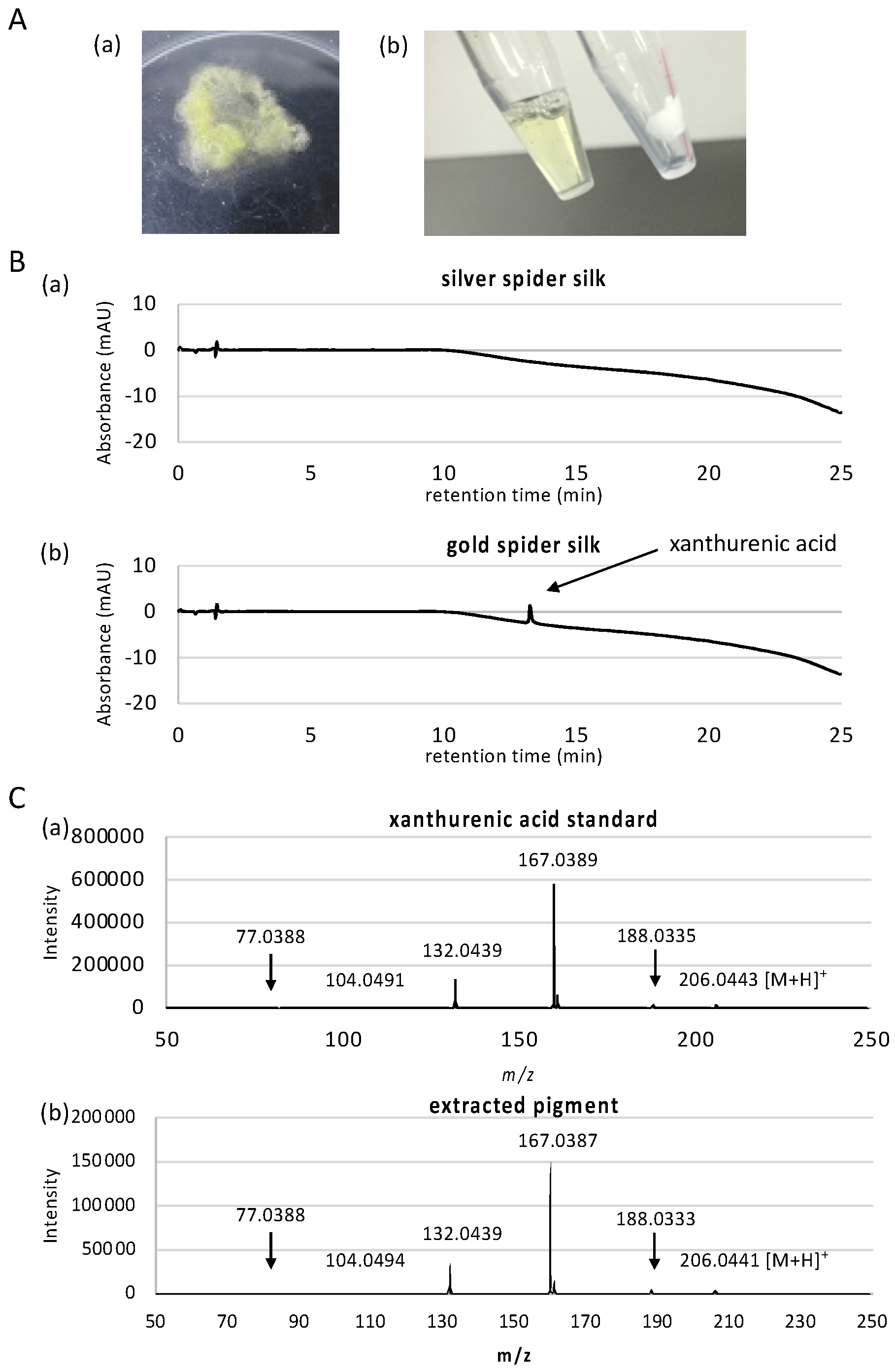
3.1. Characterization of the Gold Pigment
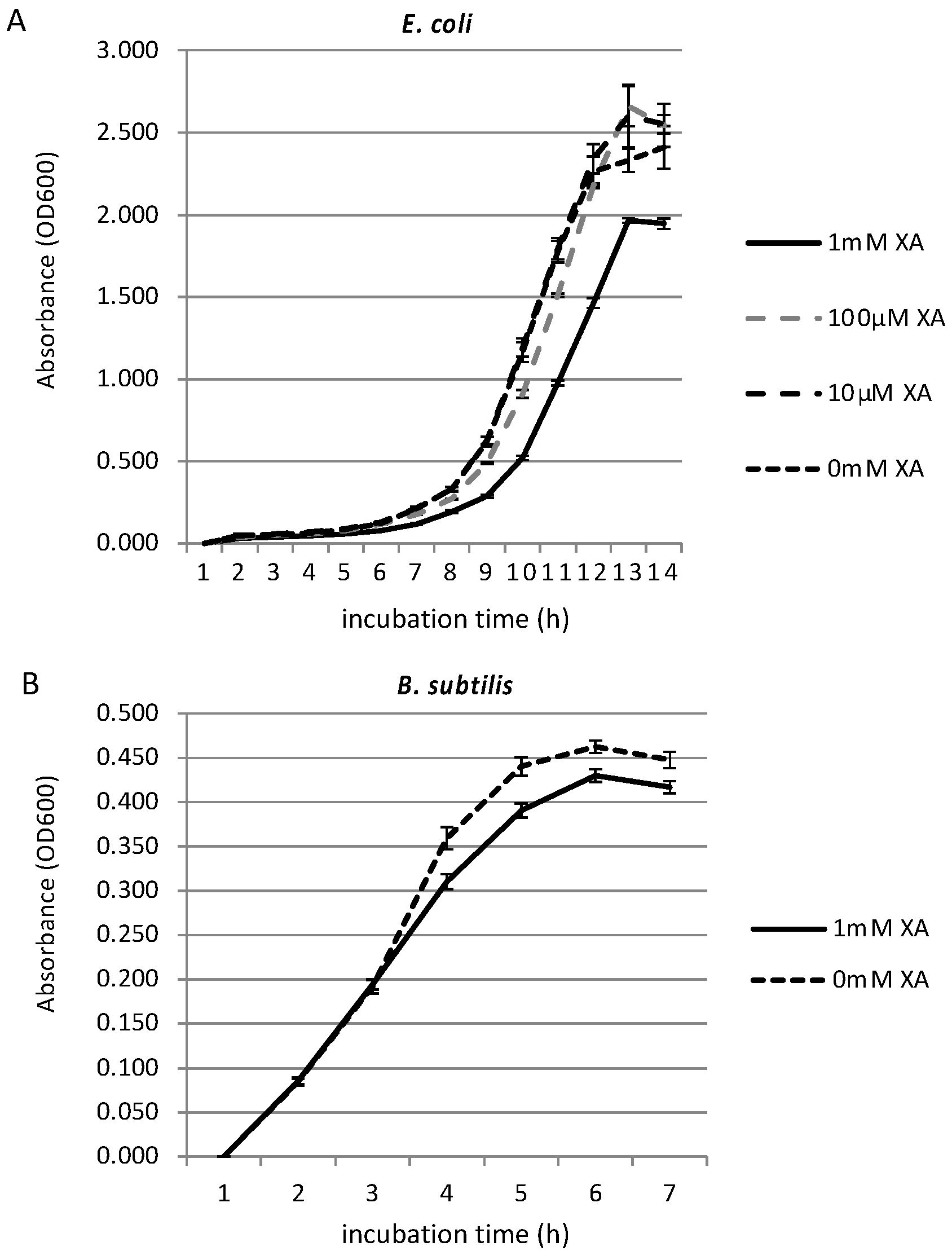
3.2. Growth Inhibition Effect of Xanthurenic Acid
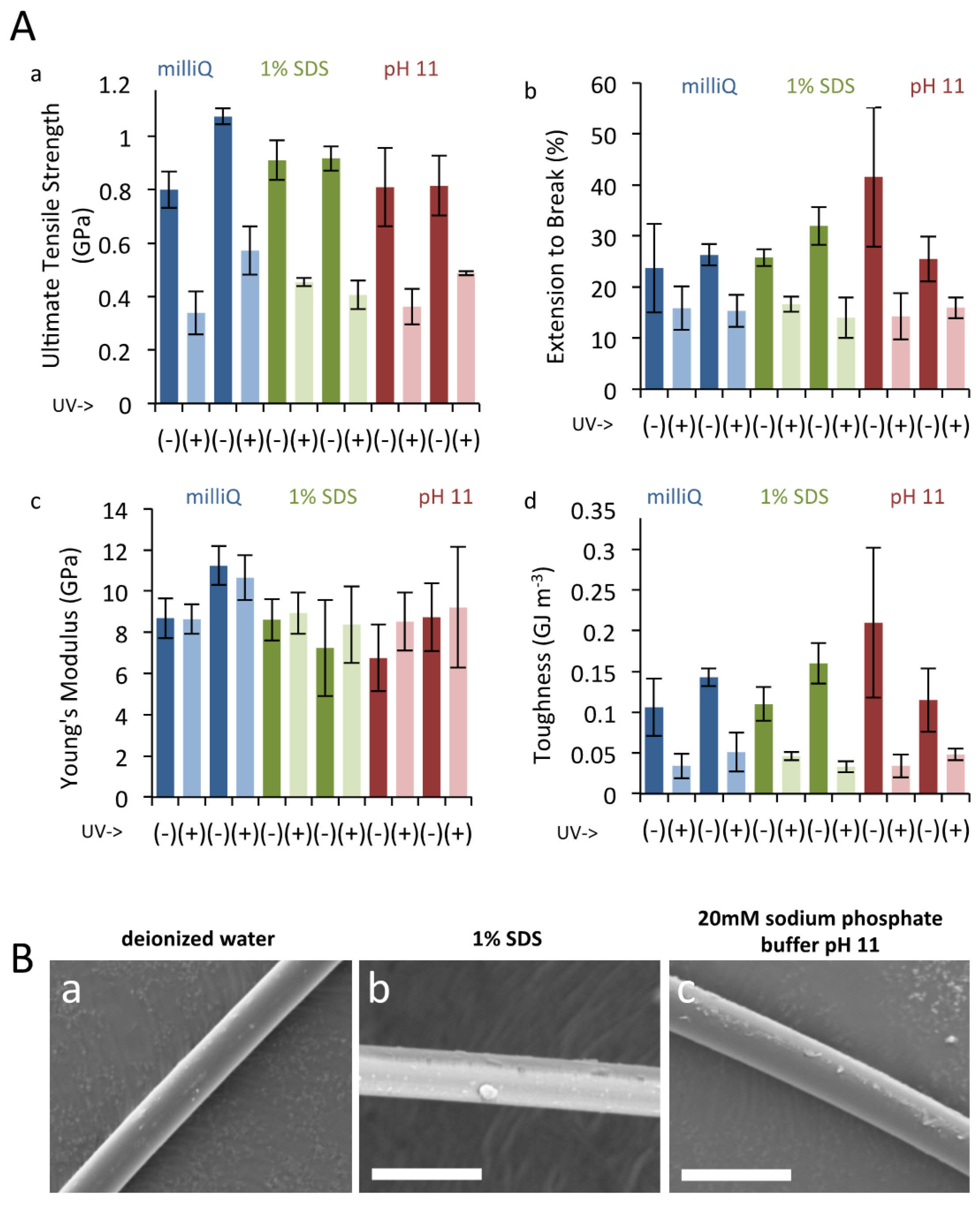
3.3. UV Irradiation to Dragline Silk
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Blamires, S.J.; Blackledge, T.A.; Tso, I.M. Physicochemical Property Variation in Spider Silk: Ecology, Evolution, and Synthetic Production. Annu. Rev. Entomol. 2017, 62, 443–460. [Google Scholar] [CrossRef]
- Copeland, C.G.; Bell, B.E.; Christensen, C.D.; Lewis, R.V. Development of a Process for the Spinning of Synthetic Spider Silk. ACS Biomater. Sci. Eng. 2015, 1, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Heidebrecht, A.; Eisoldt, L.; Diehl, J.; Schmidt, A.; Geffers, M.; Lang, G.; Scheibel, T. Biomimetic fibers made of recombinant spidroins with the same toughness as natural spider silk. Adv. Mater. 2015, 27, 2189–2194. [Google Scholar] [CrossRef]
- Rising, A.; Johansson, J. Toward spinning artificial spider silk. Nat. Chem. Biol. 2015, 11, 309–315. [Google Scholar] [CrossRef]
- Tokareva, O.; Michalczechen-Lacerda, V.A.; Rech, E.L.; Kaplan, D.L. Recombinant DNA production of spider silk proteins. Microb. Biotechnol. 2013, 6, 651–663. [Google Scholar] [CrossRef] [PubMed]
- Heppner, R.; Weichert, N.; Schierhorn, A.; Conrad, U.; Pietzsch, M. Low-Tech, Pilot Scale Purification of a Recombinant Spider Silk Protein Analog from Tobacco Leaves. Int. J. Mol. Sci. 2016, 17, 1687. [Google Scholar] [CrossRef]
- Jansson, R.; Lau, C.H.; Ishida, T.; Ramstrom, M.; Sandgren, M.; Hedhammar, M. Functionalized silk assembled from a recombinant spider silk fusion protein (Z-4RepCT) produced in the methylotrophic yeast Pichia pastoris. Biotechnol. J. 2016, 11, 687–699. [Google Scholar] [CrossRef] [PubMed]
- Lazaris, A.; Arcidiacono, S.; Huang, Y.; Zhou, J.F.; Duguay, F.; Chretien, N.; Welsh, E.A.; Soares, J.W.; Karatzas, C.N. Spider silk fibers spun from soluble recombinant silk produced in mammalian cells. Science 2002, 295, 472–476. [Google Scholar] [CrossRef]
- Peng, C.A.; Russo, J.; Gravgaard, C.; McCartney, H.; Gaines, W.; Marcotte, W.R., Jr. Spider silk-like proteins derived from transgenic Nicotiana tabacum. Transgenic Res. 2016, 25, 517–526. [Google Scholar] [CrossRef]
- Schmidt, M.; Romer, L.; Strehle, M.; Scheibel, T. Conquering isoleucine auxotrophy of Escherichia coli BLR(DE3) to recombinantly produce spider silk proteins in minimal media. Biotechnol. Lett. 2007, 29, 1741–1744. [Google Scholar] [CrossRef]
- Teule, F.; Miao, Y.G.; Sohn, B.H.; Kim, Y.S.; Hull, J.J.; Fraser, M.J., Jr.; Lewis, R.V.; Jarvis, D.L. Silkworms transformed with chimeric silkworm/spider silk genes spin composite silk fibers with improved mechanical properties. Proc. Natl. Acad. Sci. USA 2012, 109, 923–928. [Google Scholar] [CrossRef]
- Dos Santos-Pinto, J.R.; Garcia, A.M.; Arcuri, H.A.; Esteves, F.G.; Salles, H.C.; Lubec, G.; Palma, M.S. Silkomics: Insight into the Silk Spinning Process of Spiders. J. Proteome Res. 2016, 15, 1179–1193. [Google Scholar] [CrossRef] [PubMed]
- Larracas, C.; Hekman, R.; Dyrness, S.; Arata, A.; Williams, C.; Crawford, T.; Vierra, C.A. Comprehensive Proteomic Analysis of Spider Dragline Silk from Black Widows: A Recipe to Build Synthetic Silk Fibers. Int. J. Mol. Sci. 2016, 17, 1537. [Google Scholar] [CrossRef]
- Blamires, S.J.; Wu, C.L.; Blackledge, T.A.; Tso, I.M. Post-secretion processing influences spider silk performance. J. R. Soc. Interface 2012, 9, 2479–2487. [Google Scholar] [CrossRef]
- Chen, X.; Shao, Z.; Vollrath, F. The spinning processes for spider silk. Soft Matter 2006, 2, 448–451. [Google Scholar] [CrossRef] [PubMed]
- Porter, D.; Vollrath, F. Silk as a Biomimetic Ideal for Structural Polymers. Adv. Mater. 2009, 21, 487–492. [Google Scholar] [CrossRef]
- Su, I.; Buehler, M.J. Spider silk: Dynamic mechanics. Nat. Mater. 2016, 15, 1054–1055. [Google Scholar] [CrossRef]
- Schulz, S. The Chemistry of Spider Toxins and Spider Silk. Angew. Chem. Int. Ed. Engl. 1997, 36, 314–326. [Google Scholar] [CrossRef]
- Amarpuri, G.; Chaurasia, V.; Jain, D.; Blackledge, T.A.; Dhinojwala, A. Ubiquitous distribution of salts and proteins in spider glue enhances spider silk adhesion. Sci. Rep. 2015, 5, 9030. [Google Scholar] [CrossRef]
- Townley, M.A.; Bernstein, D.T.; Gallagher, K.S.; Tillinghast, E.K. Comparative study of orb web hygroscopicity and adhesive spiral composition in three araneid spiders. J. Exp. Zool. 1991, 259, 154–165. [Google Scholar] [CrossRef]
- Vollrath, F.; Fairbrother, W.J.; Williams, R.J.P.; Tillinghast, E.K.; Bernstein, D.T.; Gallagher, K.S.; Townley, M.A. Compounds in the droplets of the orb spider’s viscid spiral. Nature 1990, 345, 526–528. [Google Scholar] [CrossRef]
- Townley, M.A.; Tillinghast, E.K.; Neefus, C.D. Changes in composition of spider orb web sticky droplets with starvation and web removal, and synthesis of sticky droplet compounds. J. Exp. Biol. 2006, 209, 1463–1486. [Google Scholar] [CrossRef]
- Craig, C.L.; Weber, R.S.; Bernard, G.D. Evolution of Predator-Prey Systems: Spider Foraging Plasticity in Response to the Visual Ecology of Prey. Am. Nat. 1996, 147, 205–229. [Google Scholar] [CrossRef]
- Hénaut, Y.; Machkour-M’Rabet, S.; Winterton, P.; Calmé, S. Insect attraction by webs of Nephila clavipes (Araneae: Nephilidae). J. Arachnol. 2010, 38, 135–138. [Google Scholar] [CrossRef]
- White, T.E.; Dalrymple, R.L.; Herberstein, M.E.; Kemp, D.J. The perceptual similarity of orb-spider prey lures and flower colours. Evol. Ecol. 2017, 31, 1–20. [Google Scholar] [CrossRef]
- Craig, C. Spiderwebs and Silk: Tracing Evolution from Molecules to Genes to Phenotypes; Oxford University Press: New York, NY, USA, 2003. [Google Scholar]
- Putthanarat, S.; Tapadia, P.; Zarkoob, S.; Miller, L.; Eby, R.; Adams, W. The color of dragline silk produced in captivity by the spider Nephila clavipes. Polymer 2004, 45, 1933–1937. [Google Scholar] [CrossRef]
- Holl, A.; Henze, M. Pigmentary constituents of yellow threads of Nephila webs. In Proceedings of the XI. Europäisches Arachnologisches Colloquium, Berlin, Germany, 28 August–2 September 1988; p. 350. [Google Scholar]
- Hsiung, B.K.; Justyn, N.M.; Blackledge, T.A.; Shawkey, M.D. Spiders have rich pigmentary and structural colour palettes. J. Exp. Biol. 2017, 220, 1975–1983. [Google Scholar] [CrossRef]
- Siva, R. Status of natural dyes and dye-yielding plants in India. Curr. Sci. 2007, 92, 916–925. [Google Scholar]
- Singh, R.; Jain, A.; Panwar, S.; Gupta, D.; Khare, S.K. Antimicrobial activity of some natural dyes. Dye. Pigment. 2005, 66, 99–102. [Google Scholar] [CrossRef]
- Umadevi, K.; Krishnaveni, M. Antibacterial activity of pigment produced from Micrococcus luteus KF532949. Int. J. Chem. Anal. Sci. 2013, 4, 149–152. [Google Scholar] [CrossRef]
- Venil, C.K.; Zakaria, Z.A.; Ahmad, W.A. Bacterial pigments and their applications. Process. Biochem. 2013, 48, 1065–1079. [Google Scholar] [CrossRef]
- Wright, S.; Goodacre, S.L. Evidence for antimicrobial activity associated with common house spider silk. BMC Res. Notes 2012, 5, 326. [Google Scholar] [CrossRef]
- Jacobs, J.L.; Carroll, T.L.; Sundin, G.W. The role of pigmentation, ultraviolet radiation tolerance, and leaf colonization strategies in the epiphytic survival of phyllosphere bacteria. Microb. Ecol. 2005, 49, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Craig, C.L.; Bernard, G.D. Insect Attraction to Ultraviolet-Reflecting Spider Webs and Web Decorations. Ecology 1990, 71, 616–623. [Google Scholar] [CrossRef]
- Blamires, S.J.; Cerexhe, G.; White, T.E.; Herberstein, M.E.; Kasumovic, M.M. Spider silk colour covaries with thermal properties but not protein structure. J. R. Soc. Interface 2019, 16, 20190199. [Google Scholar] [CrossRef] [PubMed]
- Kumar, J.P.; Alam, S.; Jain, A.K.; Ansari, K.M.; Mandal, B.B. Protective Activity of Silk Sericin against UV Radiation-Induced Skin Damage by Downregulating Oxidative Stress. ACS Appl. Bio Mater. 2018, 1, 2120–2132. [Google Scholar] [CrossRef]
- Dontsov, A.E.; Sakina, N.L.; Yakovleva, M.A.; Bastrakov, A.I.; Bastrakova, I.G.; Zagorinsky, A.A.; Ushakova, N.A.; Feldman, T.B.; Ostrovsky, M.A. Ommochromes from the Compound Eyes of Insects: Physicochemical Properties and Antioxidant Activity. Biochemistry 2020, 85, 668–678. [Google Scholar] [CrossRef] [PubMed]
- Spizizen, J. Transformation of Biochemically Deficient Strains of Bacillus Subtilis by Deoxyribonucleate. Proc. Natl. Acad. Sci. USA 1958, 44, 1072–1078. [Google Scholar] [CrossRef]
- Daniels, E.V.; Reed, R.D. Xanthurenic acid is a pigment in Junonia coenia butterfly wings. Biochem. Syst. Ecol. 2012, 44, 161–163. [Google Scholar] [CrossRef]
- Linzen, B. The Tryptophan → Omrnochrome Pathway in Insects. In Advances in Insect Physiology; Treherne, J.E., Wigglesworth, V.B., Berridge, M.J., Eds.; Academic Press: Cambridge, MA, USA, 1974; Volume 10, pp. 117–246. [Google Scholar]
- Oxford, G.S.; Gillespie, R.G. Evolution and Ecology of Spider Coloration. Annu. Rev. Entomol. 1998, 43, 619–643. [Google Scholar] [CrossRef]
- Seligy, V.L. Ommochrome pigments of spiders. Comp. Biochem. Physiol. Part A Physiol. 1972, 42, 699–709. [Google Scholar] [CrossRef]
- Han, Q.; Beerntsen, B.T.; Li, J. The tryptophan oxidation pathway in mosquitoes with emphasis on xanthurenic acid biosynthesis. J. Insect Physiol. 2007, 53, 254–263. [Google Scholar] [CrossRef] [PubMed]
- Thery, M.; Casas, J. The multiple disguises of spiders: Web colour and decorations, body colour and movement. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 471–480. [Google Scholar] [CrossRef]
- Chen, C.F.; Liu, T.Z.; Lan, W.H.; Wu, L.A.; Tsai, C.H.; Chiou, J.F.; Tsai, L.Y. Novel spectrophotometric method for the quantitation of urinary xanthurenic acid and its application in identifying individuals with hyperhomocysteinemia associated with Vitamin B(6) deficiency. Biomed. Res. Int. 2013, 2013, 678476. [Google Scholar] [CrossRef]
- Nayak, B.N.; Buttar, H.S. Evaluation of the antioxidant properties of tryptophan and its metabolites in in vitro assay. J. Complement. Integr. Med. 2016, 13, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Tew, E.R.; Adamson, A.; Hesselberg, T. The web repair behaviour of an orb spider. Anim. Behav. 2015, 103, 137–146. [Google Scholar] [CrossRef]
- Perea, G.B.; Solanas, C.; Plaza, G.R.; Guinea, G.V.; Jorge, I.; Vazquez, J.; Perez Mateos, J.M.; Mari-Buye, N.; Elices, M.; Perez-Rigueiro, J. Unexpected behavior of irradiated spider silk links conformational freedom to mechanical performance. Soft Matter 2015, 11, 4868–4878. [Google Scholar] [CrossRef]
| ID | Migration Time | Average m/z | Average Area | Formula | Comment | ||
|---|---|---|---|---|---|---|---|
| ESI_POS | 25 | 13.25 | 81.98547 | 10,800.3 | Silver only | ID416-related | |
| 32 | 13.23 | 86.99120 | 10,614.0 | Silver only | ID416-related | ||
| 39 | 13.26 | 89.05266 | 24,523.0 | Silver only | ID416-related | ||
| 61 | 13.25 | 96.98379 | 19,043.3 | Silver only | ID416-related | ||
| 64 | 12.37 | 99.04526 | 31,315.7 | Silver only | |||
| 67 | 4.71 | 100.07710 | 22,357.3 | Silver only | |||
| 74 | 8.62 | 103.07697 | 23,533.3 | Silver only | |||
| 82 | 13.24 | 105.03710 | 11,356.7 | Silver only | |||
| 121 | 2.88 | 117.09238 | 12,281.3 | Silver only | |||
| 145 | 13.24 | 129.98900 | 51,675.7 | Silver only | ID416-related | ||
| 229 | 9.49 | 160.04129 | 36,029.7 | Gold only | xanthurenic acid-HCOOH | ||
| 252 | 8.62 | 165.11397 | 20,5405.3 | Silver only | C7H16O4 | ||
| 258 | 8.62 | 166.11690 | 17,431.0 | Silver only | ID252 isotope | ||
| 323 | 8.63 | 182.14073 | 74,372.3 | Silver only | ID252 plus NH4+ | ||
| 346 | 8.62 | 187.09587 | 11,368.7 | Silver only | |||
| 353 | 9.49 | 188.03569 | 11,801.0 | Gold only | xanthurenic acid-H2O | ||
| 380 | 10.81 | 193.14507 | 20,329.3 | Silver only | |||
| 416 | 13.24 | 201.12398 | 26,460.7 | Silver only | |||
| 457 | 9.49 | 206.04450 | 677,591.0 | Gold only | C10H7NO4 | xanthurenic acid | |
| 470 | 9.49 | 207.05003 | 72,818.0 | Gold only | xanthurenic acid 13C-isotope | ||
| 479 | 9.49 | 208.05075 | 9736.3 | Gold only | xanthurenic acid 13C*2-isotope | ||
| 501 | 8.62 | 210.17206 | 14,782.7 | Silver only | |||
| 532 | 10.83 | 215.12811 | 13,143.3 | Silver only | |||
| 547 | 12.37 | 219.12465 | 32,967.3 | Silver only | ID643-NH3 | ||
| 604 | 9.49 | 228.02776 | 9588.7 | Gold only | |||
| 643 | 12.37 | 236.15169 | 88,752.7 | Silver only | C10H21NO5 | ||
| 652 | 12.37 | 237.15506 | 11,120.0 | Silver only | ID643 isotope | ||
| 679 | 12.37 | 241.10661 | 10,978.3 | Silver only | |||
| 697 | 13.58 | 245.15811 | 9542.3 | Silver only | |||
| 737 | 11.22 | 251.03191 | 117,229.7 | Gold only | C10H6N2O6 | peak tailing | |
| 1076 | 4.98 | 301.05057 | 11,205.0 | Gold only | |||
| 3407 | 17.29 | 904.76282 | 10,659.7 | Gold only | |||
| ESI_NEG | 304 | 9.52 | 160.04257 | 105,782.3 | Gold only | xanthurenic acid-CO2 | |
| 332 | 9.53 | 161.04552 | 10,576.7 | Gold only | ID304 isotope | ||
| 894 | 11.23 | 205.02788 | 34,258.3 | Gold only | ID1209-related | ||
| 895 | 9.52 | 205.03566 | 36,281.3 | Gold only | xanthurenic acid isotope | ||
| 1209 | 11.22 | 249.01787 | 177,504.3 | Gold only | C10H6N2O6 | same as pos ID737 | |
| 1789 | 9.52 | 272.01950 | 9950.7 | Gold only |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fujiwara, M.; Kono, N.; Hirayama, A.; Malay, A.D.; Nakamura, H.; Ohtoshi, R.; Numata, K.; Tomita, M.; Arakawa, K. Xanthurenic Acid Is the Main Pigment of Trichonephila clavata Gold Dragline Silk. Biomolecules 2021, 11, 563. https://doi.org/10.3390/biom11040563
Fujiwara M, Kono N, Hirayama A, Malay AD, Nakamura H, Ohtoshi R, Numata K, Tomita M, Arakawa K. Xanthurenic Acid Is the Main Pigment of Trichonephila clavata Gold Dragline Silk. Biomolecules. 2021; 11(4):563. https://doi.org/10.3390/biom11040563
Chicago/Turabian StyleFujiwara, Masayuki, Nobuaki Kono, Akiyoshi Hirayama, Ali D. Malay, Hiroyuki Nakamura, Rintaro Ohtoshi, Keiji Numata, Masaru Tomita, and Kazuharu Arakawa. 2021. "Xanthurenic Acid Is the Main Pigment of Trichonephila clavata Gold Dragline Silk" Biomolecules 11, no. 4: 563. https://doi.org/10.3390/biom11040563
APA StyleFujiwara, M., Kono, N., Hirayama, A., Malay, A. D., Nakamura, H., Ohtoshi, R., Numata, K., Tomita, M., & Arakawa, K. (2021). Xanthurenic Acid Is the Main Pigment of Trichonephila clavata Gold Dragline Silk. Biomolecules, 11(4), 563. https://doi.org/10.3390/biom11040563







