The Assembly of Super-Complexes in the Plant Chloroplast
Abstract
1. Introduction
2. Molecular Organization of Different Complexes
2.1. Photosynthesis Complex
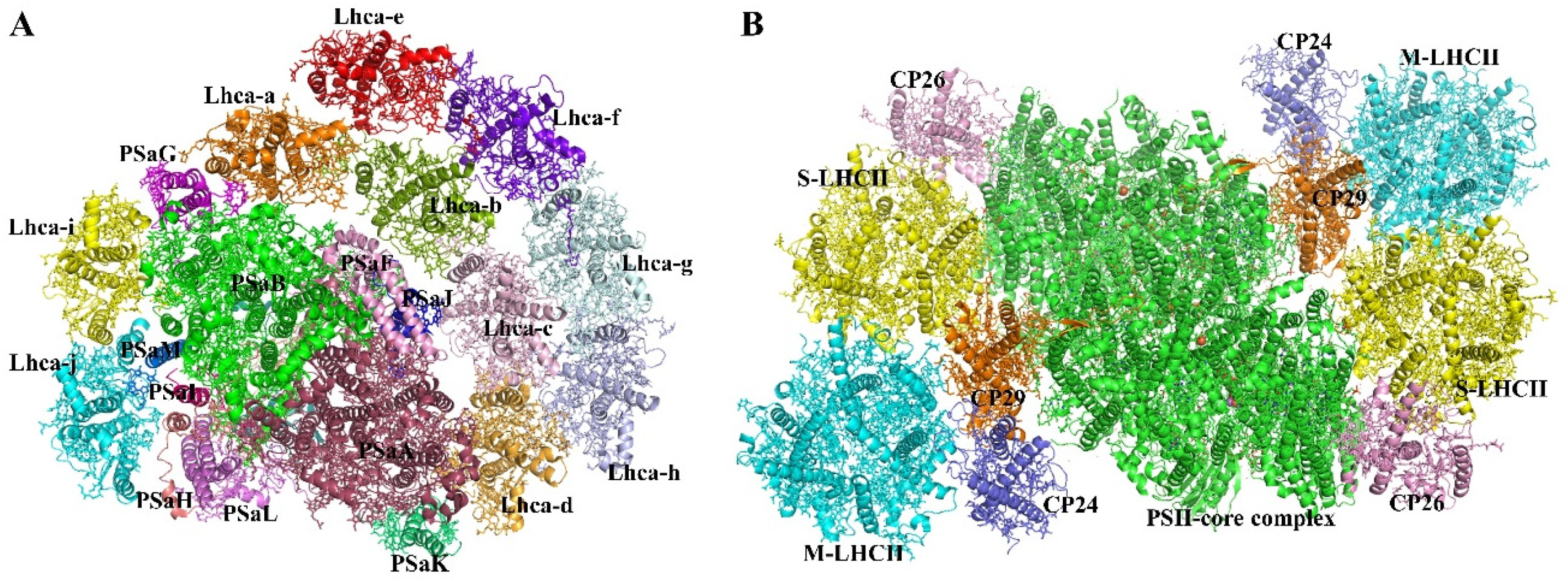
2.1.1. Photosystem I Core Complex
2.1.2. PSII Core Complex
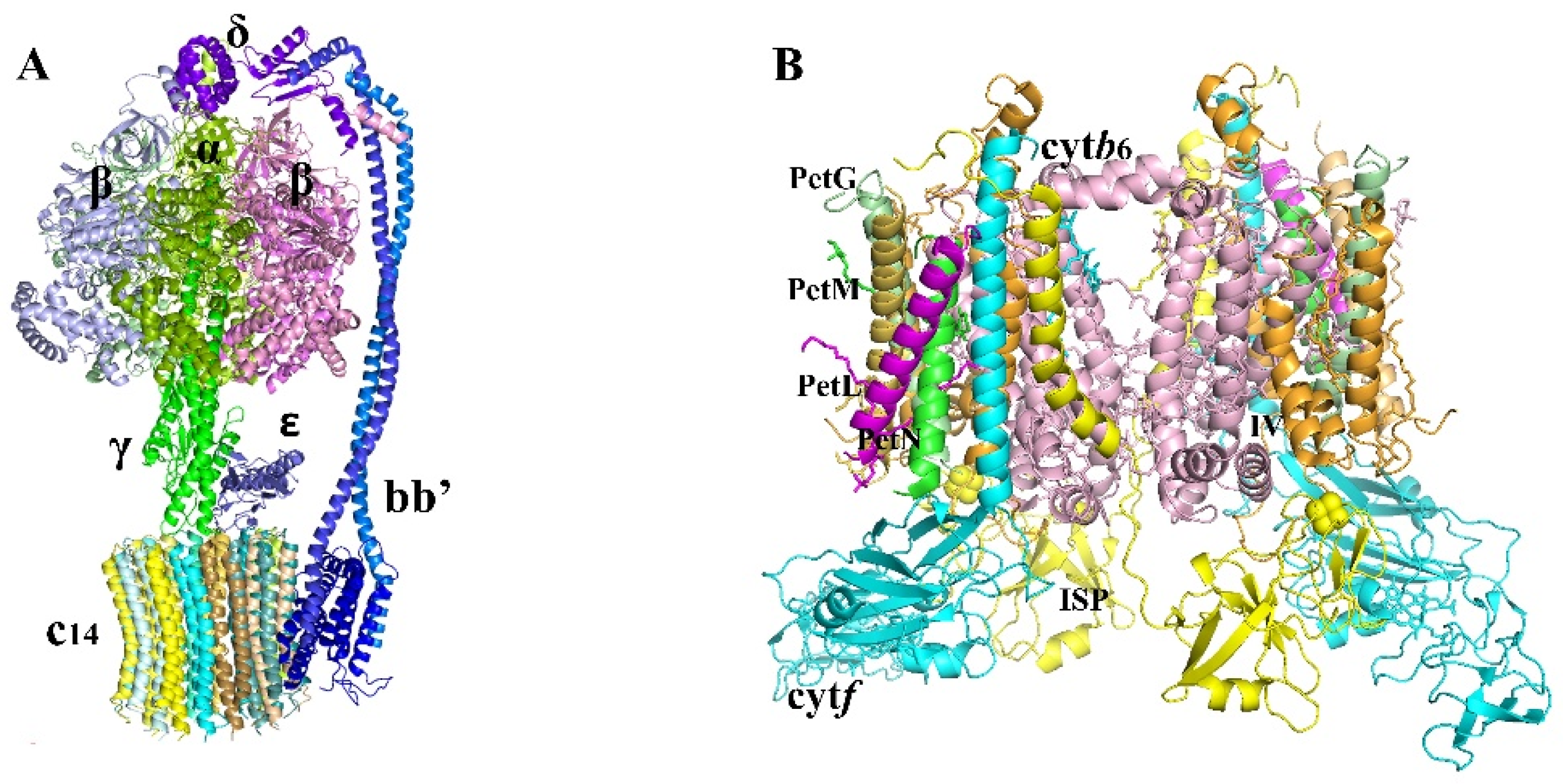
2.1.3. ATPase
2.1.4. Cytochrome b6/f
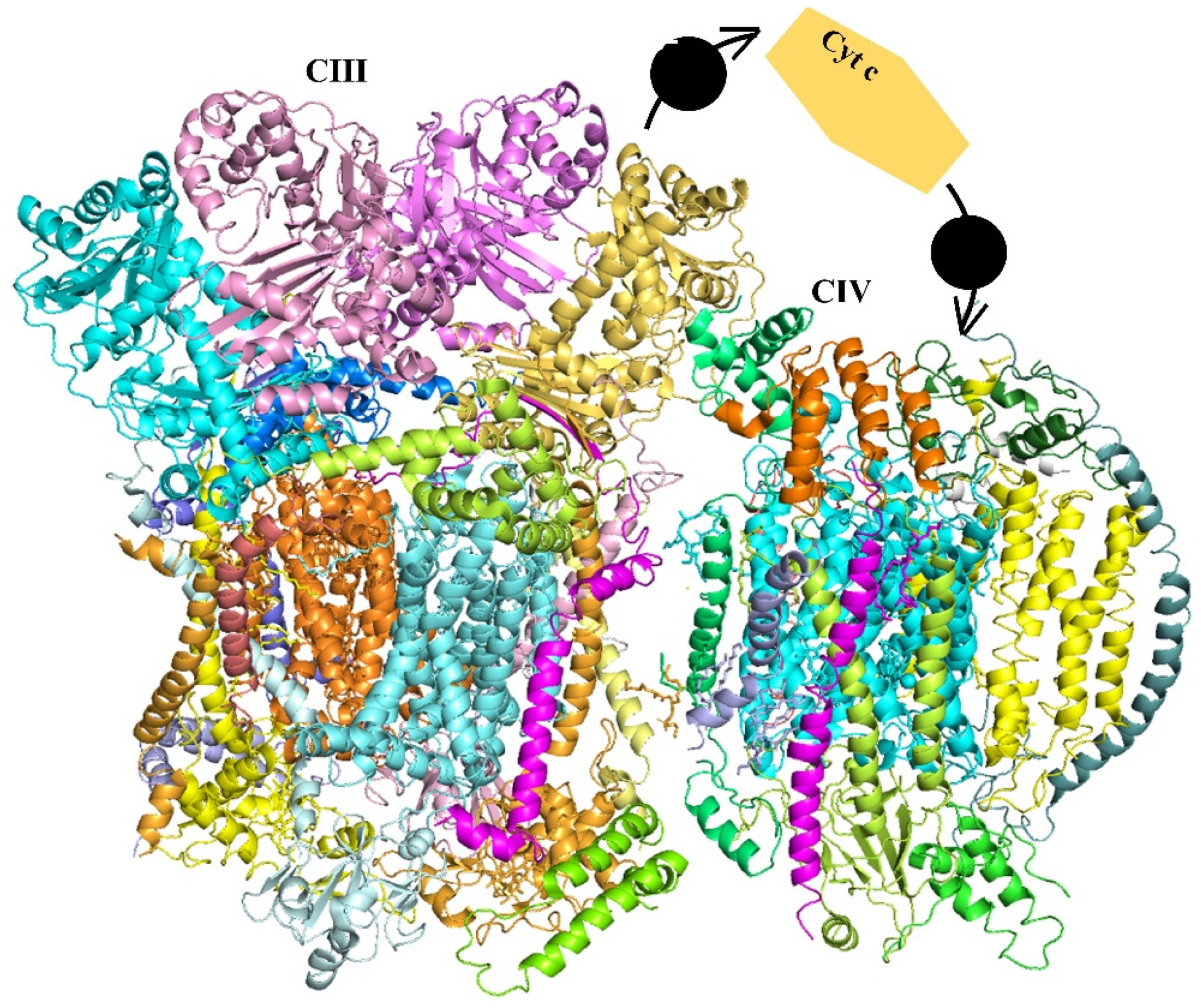
2.2. The Chloroplast Electron Transport Chain
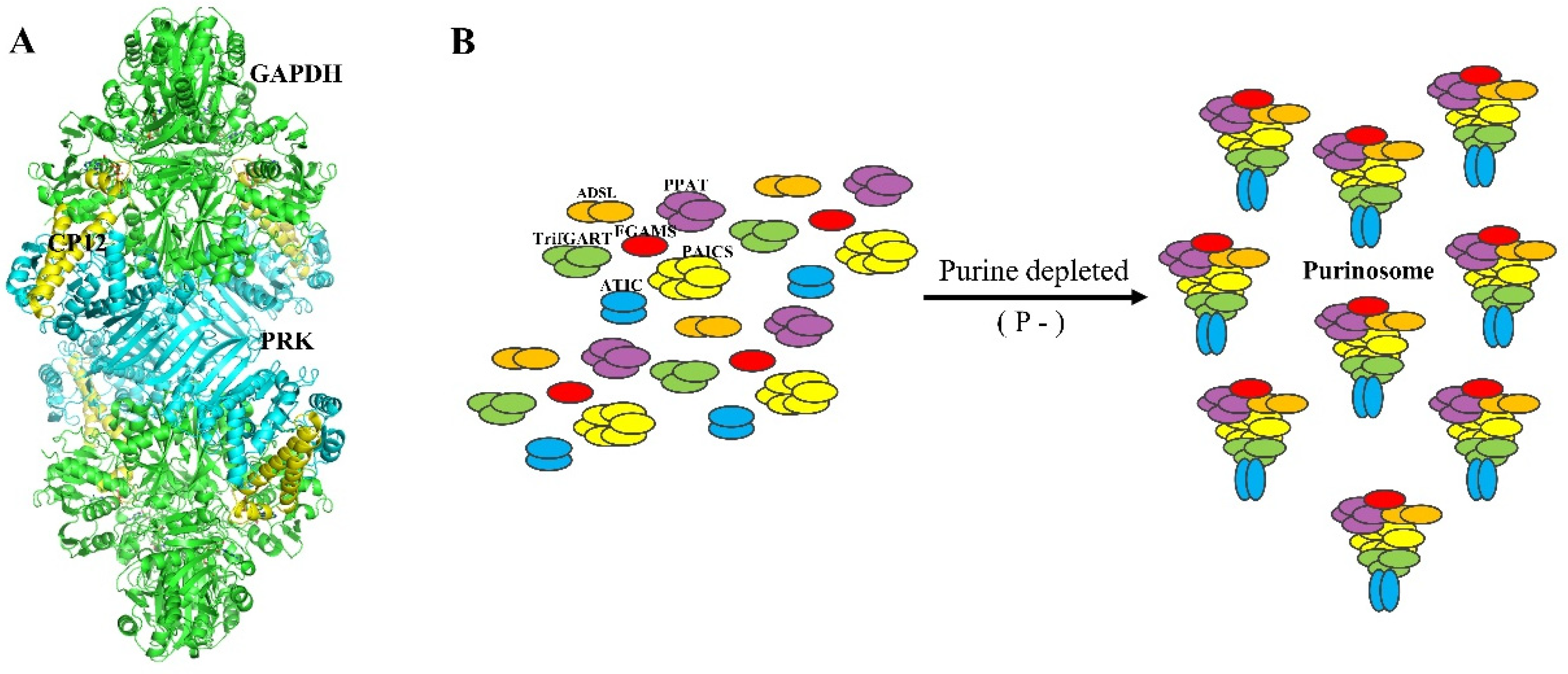
2.3. Calvin–Benson Cycle (CBC)
2.4. A Chloroplast Purinosome
3. Methods to Identify Super-Complexes
4. Substrate Channeling of the Super-Complex
5. Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Milenkovic, D.; Blaza, J.N.; Larsson, N.G.; Hirst, J. The enigma of the respiratory chain supercomplex. Cell Metab. 2017, 25, 765–776. [Google Scholar] [CrossRef]
- Lobo-Jarne, T.; Ugalde, C. Respiratory chain supercomplexes: Structures, function and biogenesis. Semin. Cell Dev. Biol. 2018, 76, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Toleco, M.R.; Naake, T.; Zhang, Y.; Heazlewood, J.L.; R Fernie, A. Plant mitochondrial carriers: Molecular gatekeepers that help to regulate plant central carbon metabolism. Plants 2020, 9, 117. [Google Scholar] [CrossRef] [PubMed]
- Letts, J.A.; Fiedorczuk, K.; Sazanov, L.A. The architecture of respiratory supercomplexes. Nature 2016, 537, 644–648. [Google Scholar] [CrossRef]
- Schäfer, E.; Seelert, H.; Reifschneider, N.H.; Krause, F.; Dencher, N.A.; Vonck, J. Architecture of active mammalian respiratory chain supercomplexes. J. Biol. Chem. 2006, 281, 15370–15375. [Google Scholar] [CrossRef] [PubMed]
- Lenaz, G.; Genova, M.L. Supramolecular organisation of the mitochondrial respiratory chain: A new challenge for the mechanism and control of oxidative phosphorylation. Mitochondrial Oxidative Phosphorylation 2012, 748, 107–144. [Google Scholar]
- Lapuente-Brun, E.; Moreno-Loshuertos, R.; Acín-Pérez, R.; Latorre-Pellicer, A.; Colás, C.; Balsa, E.; Perales-Clemente, E.; Quirós, P.M.; Calvo, E.; Rodríguez-Hernández, M. Supercomplex assembly determines electron flux in the mitochondrial electron transport chain. Science 2013, 340, 1567–1570. [Google Scholar] [CrossRef] [PubMed]
- Berndtsson, J.; Aufschnaiter, A.; Rathore, S.; Marin-Buera, L.; Dawitz, H.; Diessl, J.; Kohler, V.; Barrientos, A.; Büttner, S.; Fontanesi, F. Respiratory supercomplexes enhance electron transport by decreasing cytochrome c diffusion distance. EMBO Rep. 2020, 21, e51015. [Google Scholar] [CrossRef] [PubMed]
- Althoff, T.; Mills, D.J.; Popot, J.L.; Kuhlbrandt, W. Arrangement of electron transport chain components in bovine mitochondrial supercomplex i1iii2iv1. EMBO J. 2011, 30, 4652–4664. [Google Scholar] [CrossRef] [PubMed]
- Sousa, J.S.; Mills, D.J.; Vonck, J.; Kuhlbrandt, W. Functional asymmetry and electron flow in the bovine respirasome. Elife 2016, 5, e21290. [Google Scholar] [CrossRef] [PubMed]
- Dudkina, N.V.; Kudryashev, M.; Stahlberg, H.; Boekema, E.J. Interaction of complexes I, III, and IV within the bovine respirasome by single particle cryoelectron tomography. Proc. Natl. Acad. Sci. USA 2011, 108, 15196–15200. [Google Scholar] [CrossRef]
- Zhang, Y.; Fernie, A.R. Metabolons, enzyme–enzyme assemblies that mediate substrate channeling, and their roles in plant metabolism. Plant Commun. 2021, 2, 100081. [Google Scholar] [CrossRef]
- Maldonado, M.; Padavannil, A.; Zhou, L.; Guo, F.; Letts, J.A. Atomic structure of a mitochondrial complex i intermediate from vascular plants. Elife 2020, 9, e56664. [Google Scholar] [CrossRef] [PubMed]
- Mileykovskaya, E.; Dowhan, W. Cardiolipin-dependent formation of mitochondrial respiratory supercomplexes. Chem. Phys. Lipids 2014, 179, 42–48. [Google Scholar] [CrossRef]
- Wenz, T.; Hielscher, R.; Hellwig, P.; Schägger, H.; Richers, S.; Hunte, C. Role of phospholipids in respiratory cytochrome bc1 complex catalysis and supercomplex formation. Biochim. Biophys. Acta (BBA)-Bioenerg. 2009, 1787, 609–616. [Google Scholar] [CrossRef] [PubMed]
- Bazan, J.; Całkosiński, I.; Gamian, A. Phage display—A powerful technique for immunotherapy: 1. Introduction and potential of therapeutic applications. Hum. Vaccines Immunother. 2012, 8, 1817–1828. [Google Scholar] [CrossRef]
- Ariga, K.; Hill, J.P.; Lee, M.V.; Vinu, A.; Charvet, R.; Acharya, S. Challenges and breakthroughs in recent research on self-assembly. Sci. Technol. Adv. Mater. 2008, 9, 014109. [Google Scholar] [CrossRef]
- Gao, J.; Wang, H.; Yuan, Q.; Feng, Y. Structure and function of the photosystem supercomplexes. Front. Plant Sci. 2018, 9, 357. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Suga, M.; Kuang, T.; Shen, J.-R. Structural basis for energy transfer pathways in the plant PSI–LHCI supercomplex. Science 2015, 348, 989–995. [Google Scholar] [CrossRef]
- Pan, X.; Liu, Z.; Li, M.; Chang, W. Architecture and function of plant light-harvesting complexes ii. Curr. Opin. Struct. Biol. 2013, 23, 515–525. [Google Scholar] [CrossRef]
- Nelson, N.; Junge, W. Structure and energy transfer in photosystems of oxygenic photosynthesis. Annu. Rev. Biochem. 2015, 84, 659–683. [Google Scholar] [CrossRef]
- Suga, M.; Qin, X.; Kuang, T.; Shen, J.-R. Structure and energy transfer pathways of the plant photosystem I-LHCI supercomplex. Curr. Opin. Struct. Biol. 2016, 39, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Croce, R.; van Amerongen, H. Light-harvesting in photosystem I. Photosynth. Res. 2013, 116, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Amunts, A.; Drory, O.; Nelson, N. The structure of a plant photosystem I supercomplex at 3.4 å resolution. Nature 2007, 447, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Mazor, Y.; Borovikova, A.; Caspy, I.; Nelson, N. Structure of the plant photosystem I supercomplex at 2.6 å resolution. Nat. Plants 2017, 3, 1–9. [Google Scholar] [CrossRef]
- Antoshvili, M.; Caspy, I.; Hippler, M.; Nelson, N. Structure and function of photosystem I in cyanidioschyzon merolae. Photosynth. Res. 2019, 139, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Mazor, Y.; Borovikova, A.; Nelson, N. The structure of plant photosystem I super-complex at 2.8 å resolution. Elife 2015, 4, e07433. [Google Scholar] [CrossRef]
- Pan, X.; Ma, J.; Su, X.; Cao, P.; Chang, W.; Liu, Z.; Zhang, X.; Li, M. Structure of the maize photosystem I supercomplex with light-harvesting complexes I and II. Science 2018, 360, 1109–1113. [Google Scholar] [CrossRef]
- Busch, A.; Hippler, M. The structure and function of eukaryotic photosystem I. Biochim. Biophys. Acta (BBA)-Bioenerg. 2011, 1807, 864–877. [Google Scholar] [CrossRef]
- Jansson, S. A guide to the lhc genes and their relatives in arabidopsis. Trends Plant Sci. 1999, 4, 236–240. [Google Scholar] [CrossRef]
- Matsuzaki, M.; Misumi, O.; Shin-i, T.; Maruyama, S.; Takahara, M.; Miyagishima, S.-Y.; Mori, T.; Nishida, K.; Yagisawa, F.; Nishida, K. Genome sequence of the ultrasmall unicellular red alga cyanidioschyzon merolae 10d. Nature 2004, 428, 653–657. [Google Scholar] [CrossRef] [PubMed]
- Crepin, A.; Caffarri, S. The specific localizations of phosphorylated lhcb1 and lhcb2 isoforms reveal the role of lhcb2 in the formation of the PSI–LHCIi supercomplex in arabidopsis during state transitions. Biochim. Biophys. Acta (BBA)-Bioenerg. 2015, 1847, 1539–1548. [Google Scholar] [CrossRef] [PubMed]
- Wedel, N.; Soll, J.; Paap, B.K. Cp12 provides a new mode of light regulation of calvin cycle activity in higher plants. Proc. Natl. Acad. Sci. USA 1997, 94, 10479–10484. [Google Scholar] [CrossRef]
- Wei, X.; Su, X.; Cao, P.; Liu, X.; Chang, W.; Li, M.; Zhang, X.; Liu, Z. Structure of spinach photosystem II–LHCII supercomplex at 3.2 å resolution. Nature 2016, 534, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Jackowski, G.; Kacprzak, K.; Jansson, S. Identification of lhcb1/lhcb2/lhcb3 heterotrimers of the main light-harvesting chlorophyll a/b–protein complex of photosystem II (LHC II). Biochim. Biophys. Acta (BBA)-Bioenerg. 2001, 1504, 340–345. [Google Scholar] [CrossRef][Green Version]
- Caffarri, S.; Croce, R.; Cattivelli, L.; Bassi, R. A look within LHCII: Differential analysis of the Lhcb1−3 complexes building the major trimeric antenna complex of higher-plant photosynthesis. Biochemistry 2004, 43, 9467–9476. [Google Scholar] [CrossRef] [PubMed]
- Standfuss, J.; Kühlbrandt, W. The three isoforms of the light-harvesting complex II: Spectroscopic features, trimer formation, and functional roles. J. Biol. Chem. 2004, 279, 36884–36891. [Google Scholar] [CrossRef] [PubMed]
- Camm, E.L.; Green, B.R. How the chlorophyll-proteins got their names. Photosynth. Res. 2004, 80, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Boekema, E.J.; van Roon, H.; Calkoen, F.; Bassi, R.; Dekker, J.P. Multiple types of association of photosystem II and its light-harvesting antenna in partially solubilized photosystem ii membranes. Biochemistry 1999, 38, 2233–2239. [Google Scholar] [CrossRef] [PubMed]
- Kouřil, R.; Wientjes, E.; Bultema, J.B.; Croce, R.; Boekema, E.J. High-light vs. Low-light: Effect of light acclimation on photosystem ii composition and organization in arabidopsis thaliana. Biochim. Biophys. Acta (BBA)-Bioenerg. 2013, 1827, 411–419. [Google Scholar] [CrossRef]
- Bielczynski, L.W.; Schansker, G.; Croce, R. Effect of light acclimation on the organization of photosystem II super-and sub-complexes in arabidopsis thaliana. Front. Plant Sci. 2016, 7, 105. [Google Scholar] [CrossRef]
- Albanese, P.; Manfredi, M.; Meneghesso, A.; Marengo, E.; Saracco, G.; Barber, J.; Morosinotto, T.; Pagliano, C. Dynamic reorganization of photosystem ii supercomplexes in response to variations in light intensities. Biochim. Biophys. Acta (BBA)-Bioenerg. 2016, 1857, 1651–1660. [Google Scholar] [CrossRef]
- Goldschmidt-Clermont, M.; Bassi, R. Sharing light between two photosystems: Mechanism of state transitions. Curr. Opin. Plant Biol. 2015, 25, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Galka, P.; Santabarbara, S.; Khuong, T.T.H.; Degand, H.; Morsomme, P.; Jennings, R.C.; Boekema, E.J.; Caffarri, S. Functional analyses of the plant photosystem I–light-harvesting complex ii supercomplex reveal that light-harvesting complex II loosely bound to photosystem II is a very efficient antenna for photosystem I in state II. Plant Cell 2012, 24, 2963–2978. [Google Scholar] [CrossRef] [PubMed]
- Crepin, A.; Caffarri, S. Functions and evolution of lhcb isoforms composing LHCII, the major light harvesting complex of photosystem II of green eukaryotic organisms. Curr. Protein Pept. Sci. 2018, 19, 699–713. [Google Scholar] [CrossRef]
- Peter, G.F.; Thornber, J.P. Biochemical composition and organization of higher plant photosystem II light-harvesting pigment-proteins. J. Biol. Chem. 1991, 266, 16745–16754. [Google Scholar] [CrossRef]
- Caffarri, S.; Kouřil, R.; Kereïche, S.; Boekema, E.J.; Croce, R. Functional architecture of higher plant photosystem II supercomplexes. EMBO J. 2009, 28, 3052–3063. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Ma, J.; Wei, X.; Cao, P.; Zhu, D.; Chang, W.; Liu, Z.; Zhang, X.; Li, M. Structure and assembly mechanism of plant c2s2m2-type PSII–LHCII supercomplex. Science 2017, 357, 815–820. [Google Scholar] [CrossRef]
- Allen, J.F.; Nield, J.; Krauß, N. Molecular recognition: How photosynthesis anchors the mobile antenna. Trends Plant Sci. 2019, 24, 388–392. [Google Scholar] [CrossRef] [PubMed]
- Longoni, P.; Douchi, D.; Cariti, F.; Fucile, G.; Goldschmidt-Clermont, M. Phosphorylation of the light-harvesting complex II isoform lhcb2 is central to state transitions. Plant Physiol. 2015, 169, 2874–2883. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yan, H.; Wang, K.; Kuang, T.; Zhang, J.; Gui, L.; An, X.; Chang, W. Crystal structure of spinach major light-harvesting complex at 2.72 å resolution. Nature 2004, 428, 287–292. [Google Scholar] [CrossRef]
- Pan, X.; Li, M.; Wan, T.; Wang, L.; Jia, C.; Hou, Z.; Zhao, X.; Zhang, J.; Chang, W. Structural insights into energy regulation of light-harvesting complex cp29 from spinach. Nat. Struct. Mol. Biol. 2011, 18, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Standfuss, J.; Terwisscha van Scheltinga, A.C.; Lamborghini, M.; Kühlbrandt, W. Mechanisms of photoprotection and nonphotochemical quenching in pea light-harvesting complex at 2.5 å resolution. EMBO J. 2005, 24, 919–928. [Google Scholar] [CrossRef] [PubMed]
- Nield, J.; Orlova, E.V.; Morris, E.P.; Gowen, B.; van Heel, M.; Barber, J. 3d map of the plant photosystem II supercomplex obtained by cryoelectron microscopy and single particle analysis. Nat. Struct. Biol. 2000, 7, 44–47. [Google Scholar] [PubMed]
- van Bezouwen, L.S.; Caffarri, S.; Kale, R.S.; Kouřil, R.; Thunnissen, A.-M.W.; Oostergetel, G.T.; Boekema, E.J. Subunit and chlorophyll organization of the plant photosystem II supercomplex. Nat. Plants 2017, 3, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Alboresi, A.; Caffarri, S.; Nogue, F.; Bassi, R.; Morosinotto, T. In silico and biochemical analysis of physcomitrella patens photosynthetic antenna: Identification of subunits which evolved upon land adaptation. PLoS ONE 2008, 3, e2033. [Google Scholar] [CrossRef]
- Kovacs, L.; Damkjær, J.; Kereïche, S.; Ilioaia, C.; Ruban, A.V.; Boekema, E.J.; Jansson, S.; Horton, P. Lack of the light-harvesting complex cp24 affects the structure and function of the grana membranes of higher plant chloroplasts. Plant Cell 2006, 18, 3106–3120. [Google Scholar] [CrossRef]
- Nosek, L.; Semchonok, D.; Boekema, E.J.; Ilík, P.; Kouřil, R. Structural variability of plant photosystem II megacomplexes in thylakoid membranes. Plant J. 2017, 89, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Pietrzykowska, M.; Suorsa, M.; Semchonok, D.A.; Tikkanen, M.; Boekema, E.J.; Aro, E.-M.; Jansson, S. The light-harvesting chlorophyll a/b binding proteins lhcb1 and lhcb2 play complementary roles during state transitions in arabidopsis. Plant Cell 2014, 26, 3646–3660. [Google Scholar] [CrossRef] [PubMed]
- Jenny, A.; Mark, W.; Robin, G.W.; Caroline, A.H.; Alexander, V.R.; Peter, H.; Stefan, J. Absence of the lhcb1 and lhcb2 proteins of the light-harvesting complex of photosystem II–effects on photosynthesis, grana stacking and fitness. Plant J. 2003, 35, 350–361. [Google Scholar] [CrossRef] [PubMed]
- Ruban, A.V.; Wentworth, M.; Yakushevska, A.E.; Andersson, J.; Lee, P.; Keegstra, W.; Dekker, J.; Boekema, E.; Jansson, S.; Horton, P. Plants lacking the main light-harvesting complex retain photosystem ii macro-organization. Nature 2003, 421, 648–652. [Google Scholar] [CrossRef]
- Van Oort, B.; Murali, S.; Wientjes, E.; Koehorst, R.B.; Spruijt, R.B.; van Hoek, A.; Croce, R.; van Amerongen, H. Ultrafast resonance energy transfer from a site-specifically attached fluorescent chromophore reveals the folding of the n-terminal domain of cp29. Chem. Phys. 2009, 357, 113–119. [Google Scholar] [CrossRef]
- Lolkema, J.S.; Boekema, E.J. The a-type atp synthase subunit k of methanopyrus kandleri is deduced from its sequence to form a monomeric rotor comprising 13 hairpin domains. FEBS Lett. 2003, 543, 47–50. [Google Scholar] [CrossRef]
- Seelert, H.; Poetsch, A.; Dencher, N.A.; Engel, A.; Stahlberg, H.; Müller, D.J. Proton-powered turbine of a plant motor. Nature 2000, 405, 418–419. [Google Scholar] [CrossRef] [PubMed]
- Abrahams, J.P.; Leslie, A.; Walker, J. Structure at 2.8 a resolution of f1-atpasem. Nature 1994, 370, 25. [Google Scholar] [CrossRef] [PubMed]
- Groth, G.; Pohl, E. The structure of the chloroplast f1-atpase at 3.2 å resolution. J. Biol. Chem. 2001, 276, 1345–1352. [Google Scholar] [CrossRef]
- Arnold, I.; Pfeiffer, K.; Neupert, W.; Stuart, R.A.; Schägger, H. Yeast mitochondrial f1f0-atp synthase exists as a dimer: Identification of three dimer-specific subunits. EMBO J. 1998, 17, 7170–7178. [Google Scholar] [CrossRef]
- Eubel, H.; Jansch, L.; Braun, H.-P. New insights into the respiratory chain of plant mitochondria. Supercomplexes and a unique composition of complex ii. Plant Physiol. 2003, 133, 274–286. [Google Scholar] [CrossRef]
- Zhang, H.; Whitelegge, J.P.; Cramer, W.A. Ferredoxin: Nadp+ oxidoreductase is a subunit of the chloroplast cytochrome b6fcomplex. J. Biol. Chem. 2001, 276, 38159–38165. [Google Scholar] [CrossRef] [PubMed]
- Kurisu, G.; Zhang, H.; Smith, J.L.; Cramer, W.A. Structure of the cytochrome b6f complex of oxygenic photosynthesis: Tuning the cavity. Science 2003, 302, 1009–1014. [Google Scholar] [CrossRef]
- Stroebel, D.; Choquet, Y.; Popot, J.-L.; Picot, D. An atypical haem in the cytochrome b 6 f complex. Nature 2003, 426, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Iwata, S.; Lee, J.W.; Okada, K.; Lee, J.K.; Iwata, M.; Rasmussen, B.; Link, T.A.; Ramaswamy, S.; Jap, B.K. Complete structure of the 11-subunit bovine mitochondrial cytochrome bc1 complex. Science 1998, 281, 64–71. [Google Scholar] [CrossRef]
- Allen, J.F. Cytochrome b6f: Structure for signalling and vectorial metabolism. Trends Plant Sci. 2004, 9, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Depège, N.; Bellafiore, S.; Rochaix, J.-D. Role of chloroplast protein kinase stt7 in lhcii phosphorylation and state transition in chlamydomonas. Science 2003, 299, 1572–1575. [Google Scholar] [CrossRef] [PubMed]
- Wollman, F.A. State transitions reveal the dynamics and flexibility of the photosynthetic apparatus. EMBO J. 2001, 20, 3623–3630. [Google Scholar] [CrossRef]
- Heinemeyer, J.; Eubel, H.; Wehmhöner, D.; Jänsch, L.; Braun, H.-P. Proteomic approach to characterize the supramolecular organization of photosystems in higher plants. Phytochemistry 2004, 65, 1683–1692. [Google Scholar] [CrossRef] [PubMed]
- Joliot, P.; Béal, D.; Joliot, A. Cyclic electron flow under saturating excitation of dark-adapted arabidopsis leaves. Biochim. Biophys. Acta (BBA)-Bioenerg. 2004, 1656, 166–176. [Google Scholar] [CrossRef]
- Joliot, P.; Lavergne, J.; Béal, D. Plastoquinone compartmentation in chloroplasts. I. Evidence for domains with different rates of photo-reduction. Biochim. Biophys. Acta (BBA)-Bioenerg. 1992, 1101, 1–12. [Google Scholar] [CrossRef]
- Kirchhoff, H.; Horstmann, S.; Weis, E. Control of the photosynthetic electron transport by pq diffusion microdomains in thylakoids of higher plants. Biochim. Biophys. Acta (BBA)-Bioenerg. 2000, 1459, 148–168. [Google Scholar] [CrossRef]
- Kirchhoff, H.; Mukherjee, U.; Galla, H.-J. Molecular architecture of the thylakoid membrane: Lipid diffusion space for plastoquinone. Biochemistry 2002, 41, 4872–4882. [Google Scholar] [CrossRef]
- Lavergne, J.; Bouchaud, J.-P.; Joliot, P. Plastoquinone compartmentation in chloroplasts. II. Theoretical aspects. Biochim. Biophys. Acta (BBA)-Bioenerg. 1992, 1101, 13–22. [Google Scholar] [CrossRef]
- Johnson, G.N. Physiology of PSI Cyclic Electron Transport in Higher Plants Bioenergetics. Biochim. Biophys. Acta (BBA)-Bioenerg. 2011, 1807, 384–389. [Google Scholar] [CrossRef]
- Rochaix, J.-D. Reprint of: Regulation of photosynthetic electron transport. Biochim. Biophys. Acta (BBA)-Bioenerg. 2011, 1807, 878–886. [Google Scholar] [CrossRef] [PubMed]
- DalCorso, G.; Pesaresi, P.; Masiero, S.; Aseeva, E.; Schünemann, D.; Finazzi, G.; Joliot, P.; Barbato, R.; Leister, D. A complex containing pgrl1 and pgr5 is involved in the switch between linear and cyclic electron flow in arabidopsis. Cell 2008, 132, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Burrows, P.A.; Sazanov, L.A.; Svab, Z.; Maliga, P.; Nixon, P.J. Identification of a functional respiratory complex in chloroplasts through analysis of tobacco mutants containing disrupted plastid ndh genes. EMBO J. 1998, 17, 868–876. [Google Scholar] [CrossRef] [PubMed]
- Shikanai, T.; Endo, T.; Hashimoto, T.; Yamada, Y.; Asada, K.; Yokota, A. Directed disruption of the tobacco ndhB gene impairs cyclic electron flow around photosystem I. Proc. Natl. Acad. Sci. USA 1998, 95, 9705–9709. [Google Scholar] [CrossRef]
- Munekage, Y.; Hashimoto, M.; Miyake, C.; Tomizawa, K.-I.; Endo, T.; Tasaka, M.; Shikanai, T. Cyclic electron flow around photosystem I is essential for photosynthesis. Nature 2004, 429, 579–582. [Google Scholar] [CrossRef]
- Livingston, A.K.; Cruz, J.A.; Kohzuma, K.; Dhingra, A.; Kramer, D.M. An arabidopsis mutant with high cyclic electron flow around photosystem I (hcef) involving the nadph dehydrogenase complex. Plant Cell 2010, 22, 221–233. [Google Scholar] [CrossRef]
- Iwai, M.; Takizawa, K.; Tokutsu, R.; Okamuro, A.; Takahashi, Y.; Minagawa, J. Isolation of the elusive supercomplex that drives cyclic electron flow in photosynthesis. Nature 2010, 464, 1210–1213. [Google Scholar] [CrossRef]
- Hertle, A.P.; Blunder, T.; Wunder, T.; Pesaresi, P.; Pribil, M.; Armbruster, U.; Leister, D. Pgrl1 is the elusive ferredoxin-plastoquinone reductase in photosynthetic cyclic electron flow. Mol. Cell 2013, 49, 511–523. [Google Scholar] [CrossRef] [PubMed]
- Johnson, X.; Steinbeck, J.; Dent, R.M.; Takahashi, H.; Richaud, P.; Ozawa, S.-I.; Houille-Vernes, L.; Petroutsos, D.; Rappaport, F.; Grossman, A.R. Proton gradient regulation 5-mediated cyclic electron flow under atp- or redox-limited conditions: A study of ɗ atpase pgr5 and ɗ rbcl pgr5 mutants in the green alga chlamydomonas reinhardtii. Plant Physiol. 2014, 165, 438–452. [Google Scholar] [CrossRef]
- Boekema, E.J.; Folea, M.; Kouřil, R. Single particle electron microscopy. Photosynth. Res. 2009, 102, 189–196. [Google Scholar] [CrossRef][Green Version]
- Johnson, M.P. Correction: Photosynthesis. Essays Biochem. 2017, 61, 429. [Google Scholar] [CrossRef]
- Buchanan, B.B. Regulation of co2 assimilation in oxygenic photosynthesis: The ferredoxin/thioredoxin system: Perspective on its discovery, present status, and future development. Arch. Biochem. Biophys. 1991, 288, 1–9. [Google Scholar] [CrossRef]
- Dai, S.; Johansson, K.; Miginiac-Maslow, M.; Schürmann, P.; Eklund, H. Structural basis of redox signaling in photosynthesis: Structure and function of ferredoxin: Thioredoxin reductase and target enzymes. Photosynth. Res. 2004, 79, 233–248. [Google Scholar] [CrossRef]
- Howard, T.P.; Metodiev, M.; Lloyd, J.C.; Raines, C.A. Thioredoxin-mediated reversible dissociation of a stromal multiprotein complex in response to changes in light availability. Proc. Natl. Acad. Sci. USA 2008, 105, 4056–4061. [Google Scholar] [CrossRef]
- Lopez-Calcagno, P.E.; Howard, T.P.; Raines, C.A. The cp12 protein family: A thioredoxin-mediated metabolic switch? Front. Plant Sci. 2014, 5, 9. [Google Scholar] [CrossRef] [PubMed]
- Graciet, E.; Gans, P.; Wedel, N.; Lebreton, S.; Camadro, J.-M.; Gontero, B. The small protein cp12: A protein linker for supramolecular complex assembly. Biochemistry 2003, 42, 8163–8170. [Google Scholar] [CrossRef] [PubMed]
- Marri, L.; Trost, P.; Pupillo, P.; Sparla, F. Reconstitution and properties of the recombinant glyceraldehyde-3-phosphate dehydrogenase/cp12/phosphoribulokinase supramolecular complex of arabidopsis. Plant Physiol. 2005, 139, 1433–1443. [Google Scholar] [CrossRef]
- Marri, L.; Trost, P.; Trivelli, X.; Gonnelli, L.; Pupillo, P.; Sparla, F. Spontaneous assembly of photosynthetic supramolecular complexes as mediated by the intrinsically unstructured protein cp12. J. Biol. Chem. 2008, 283, 1831–1838. [Google Scholar] [CrossRef] [PubMed]
- Wedel, N.; Soll, J. Evolutionary conserved light regulation of calvin cycle activity by nadph-mediated reversible phosphoribulokinase/cp12/glyceraldehyde-3-phosphate dehydrogenase complex dissociation. Proc. Natl. Acad. Sci. USA 1998, 95, 9699–9704. [Google Scholar] [CrossRef]
- Marri, L.; Zaffagnini, M.; Collin, V.; Issakidis-Bourguet, E.; Lemaire, S.D.; Pupillo, P.; Sparla, F.; Miginiac-Maslow, M.; Trost, P. Prompt and easy activation by specific thioredoxins of calvin cycle enzymes of arabidopsis thaliana associated in the gapdh/cp12/prk supramolecular complex. Mol. Plant 2009, 2, 259–269. [Google Scholar] [CrossRef] [PubMed]
- Marri, L.; Thieulin-Pardo, G.; Lebrun, R.; Puppo, R.; Zaffagnini, M.; Trost, P.; Gontero, B.; Sparla, F. Cp12-mediated protection of calvin–benson cycle enzymes from oxidative stress. Biochimie 2014, 97, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Elena López-Calcagno, P.; Omar Abuzaid, A.; Lawson, T.; Anne Raines, C. Arabidopsis cp12 mutants have reduced levels of phosphoribulokinase and impaired function of the calvin–benson cycle. J. Exp. Bot. 2017, 68, 2285–2298. [Google Scholar] [CrossRef] [PubMed]
- Del Giudice, A.; Pavel, N.V.; Galantini, L.; Falini, G.; Trost, P.; Fermani, S.; Sparla, F. Unravelling the shape and structural assembly of the photosynthetic gapdh–cp12–prk complex from arabidopsis thaliana by small-angle x-ray scattering analysis. Acta Crystallogr. Sect. D Biol. Crystallogr. 2015, 71, 2372–2385. [Google Scholar] [CrossRef] [PubMed]
- McFarlane, C.R.; Shah, N.R.; Kabasakal, B.V.; Echeverria, B.; Cotton, C.A.; Bubeck, D.; Murray, J.W. Structural basis of light-induced redox regulation in the calvin–benson cycle in cyanobacteria. Proc. Natl. Acad. Sci. USA 2019, 116, 20984–20990. [Google Scholar] [CrossRef]
- Chan, C.Y.; Zhao, H.; Pugh, R.J.; Pedley, A.M.; French, J.; Jones, S.A.; Zhuang, X.; Jinnah, H.; Huang, T.J.; Benkovic, S.J. Purinosome formation as a function of the cell cycle. Proc. Natl. Acad. Sci. USA 2015, 112, 1368–1373. [Google Scholar] [CrossRef]
- Zhao, H.; French, J.B.; Fang, Y.; Benkovic, S.J. The purinosome, a multi-protein complex involved in the de novo biosynthesis of purines in humans. Chem. Commun. 2013, 49, 4444–4452. [Google Scholar] [CrossRef]
- Pedley, A.M.; Benkovic, S.J. A new view into the regulation of purine metabolism: The purinosome. Trends Biochem. Sci. 2017, 42, 141–154. [Google Scholar] [CrossRef]
- Sweetlove, L.J.; Fernie, A.R. The role of dynamic enzyme assemblies and substrate channelling in metabolic regulation. Nat. Commun. 2018, 9, 2136. [Google Scholar] [CrossRef] [PubMed]
- Baresova, V.; Skopova, V.; Sikora, J.; Patterson, D.; Sovova, J.; Zikanova, M.; Kmoch, S. Mutations of atic and adsl affect purinosome assembly in cultured skin fibroblasts from patients with aica-ribosiduria and ADSL deficiency. Hum. Mol. Genet. 2011, 21, 1534–1543. [Google Scholar] [CrossRef]
- Zhang, Y.; Skirycz, A.; Fernie, A.R. An abundance and interaction encyclopedia of plant protein function. Trends Plant Sci. 2020, 25, 627–630. [Google Scholar] [CrossRef]
- McWhite, C.D.; Papoulas, O.; Drew, K.; Cox, R.M.; June, V.; Dong, O.X.; Kwon, T.; Wan, C.; Salmi, M.L.; Roux, S.J. A pan-plant protein complex map reveals deep conservation and novel assemblies. Cell 2020, 181, 460–474. [Google Scholar] [CrossRef] [PubMed]
- Srere, P.A. The metabolon. Trends Biochem. Sci. 1985, 10, 109–110. [Google Scholar] [CrossRef]
- Spivey, H.O.; Ovadi, J. Substrate channeling. Methods-A Companion Methods Enzymol. 1999, 19, 306–321. [Google Scholar] [CrossRef] [PubMed]
- Winkel, B.S. Metabolic channeling in plants. Annu. Rev. Plant Biol. 2004, 55, 85–107. [Google Scholar] [CrossRef]
- Srere, P.A. Complexes of sequential metabolic enzymes. Annu. Rev. Biochem. 1987, 56, 89–124. [Google Scholar] [CrossRef]
- Fernie, A.R.; Zhang, Y.; Sweetlove, L.J. Passing the baton: Substrate channelling in respiratory metabolism. Research 2018, 2018, 1539325. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sampathkumar, A.; Kerber, S.M.-L.; Swart, C.; Hille, C.; Seerangan, K.; Graf, A.; Sweetlove, L.; Fernie, A.R. A moonlighting role for enzymes of glycolysis in the co-localization of mitochondria and chloroplasts. Nat. Commun. 2020, 11, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.J.; Beard, K.F.M.; Swart, C.; Bergmann, S.; Krahnert, I.; Nikoloski, Z.; Graf, A.; Ratcliffe, R.G.; Sweetlove, L.J.; Fernie, A.R.; et al. Protein-protein interactions and metabolite channelling in the plant tricarboxylic acid cycle. Nat. Commun. 2017, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Achnine, L.; Blancaflor, E.B.; Rasmussen, S.; Dixon, R.A. Colocalization of l-phenylalanine ammonia-lyase and cinnamate 4-hydroxylase for metabolic channeling in phenylpropanoid biosynthesis. Plant Cell 2004, 16, 3098–3109. [Google Scholar] [CrossRef] [PubMed]
- Laursen, T.; Borch, J.; Knudsen, C.; Bavishi, K.; Torta, F.; Martens, H.J.; Silvestro, D.; Hatzakis, N.S.; Wenk, M.R.; Dafforn, T.R.; et al. Characterization of a dynamic metabolon producing the defense compound dhurrin in sorghum. Science 2016, 354, 890–893. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qin, K.; Fernie, A.R.; Zhang, Y. The Assembly of Super-Complexes in the Plant Chloroplast. Biomolecules 2021, 11, 1839. https://doi.org/10.3390/biom11121839
Qin K, Fernie AR, Zhang Y. The Assembly of Super-Complexes in the Plant Chloroplast. Biomolecules. 2021; 11(12):1839. https://doi.org/10.3390/biom11121839
Chicago/Turabian StyleQin, Kezhen, Alisdair R. Fernie, and Youjun Zhang. 2021. "The Assembly of Super-Complexes in the Plant Chloroplast" Biomolecules 11, no. 12: 1839. https://doi.org/10.3390/biom11121839
APA StyleQin, K., Fernie, A. R., & Zhang, Y. (2021). The Assembly of Super-Complexes in the Plant Chloroplast. Biomolecules, 11(12), 1839. https://doi.org/10.3390/biom11121839





