Amazonian Bacuri (Platonia insignis Mart.) Fruit Waste Valorisation Using Response Surface Methodology
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Biological Samples
2.3. Extraction Procedures and Analysis
2.4. Response Variable Determination
2.4.1. Total Phenolic Content
2.4.2. Total Flavonoids
2.4.3. ABTS Radical Scavenging Assay
2.4.4. DPPH Radical Scavenging Assay
3. Results and Discussion
3.1. Crude Extraction Yield
3.2. Chemical Characterization
3.3. Evaluation of Phenolic Composition and Antioxidant Capacity
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Lustosa, A.K.M.F.; Arcanjo, D.D.R.; Ribeiro, R.G.; Rodrigues, K.A.F.; Passos, F.F.B.; Piauilino, C.A.; Silva-Filho, J.C.; Araújo, B.Q.; Lima-Neto, J.S.; Costa-Júnior, J.S.; et al. Immunomodulatory and toxicological evaluation of the fruit seeds from Platonia insignis, a native species from Brazilian Amazon rainforest. Rev. Bras. Farmacogn. 2016, 26, 77–82. [Google Scholar] [CrossRef]
- Teixeira, N.; Melo, J.C.S.; Batista, L.F.; Paula-Souza, J.; Fronza, P.; Brandão, M.G.L. Edible fruits from Brazilian biodiversity: A review on their sensorial characteristics versus bioactivity as tool to select research. Food Res. Int. 2019, 119, 325–348. [Google Scholar] [CrossRef] [PubMed]
- Clerici, M.T.P.S.; Carvalho-Silva, L.B. Nutritional bioactive compounds and technological aspects of minor fruits grown in Brazil. Food Res. Int. 2011, 44, 1658–1670. [Google Scholar] [CrossRef]
- de Carvalho, J.E.U.; do Nascimento, W.M.O. Technological innovations in the propagation of Açaí palm and Bacuri. Rev. Bras. Frutic. 2018, 40. [Google Scholar] [CrossRef]
- Botelho, M.G.L.; Homma, A.K.O.; Furtado, L.G.; LIma, M.d.C.S.; Costa, M.d.S.S. Productive and market potential of bacuri fruit (Platonia insignis Mart.) in Pará, Brazil. Res. Soc. Dev. 2020, 9, 1–6. [Google Scholar] [CrossRef]
- Fontenele, M.A.; de Figueiredo, R.W.; Maia, G.A.; Alves, R.E.; de Sousa, P.H.M.; de Souza, V.A.B. Conservação pós-colheita de bacuri (Platonia insignis Mart.) sob refrigeração e embalado em PVC. Rev. Ceres 2010, 57, 292–296. [Google Scholar] [CrossRef][Green Version]
- Bezerra, G.D.; Maia, G.A.; de Figueiredo, R.W.; de Souza, M.D. Potencial agroeconômico do bacuri: Revisão. Bol. Cent. Pesqui. Process. Aliment. 2005, 23, 47–58. [Google Scholar] [CrossRef]
- Rufino, M.d.S.M.; Alves, R.E.; de Brito, E.S.; Pérez-Jiménez, J.; Saura-Calixto, F.; Mancini-Filho, J. Bioactive compounds and antioxidant capacities of 18 non-traditional tropical fruits from Brazil. Food Chem. 2010, 121, 996–1002. [Google Scholar] [CrossRef]
- Uekane, T.M.; Nicolotti, L.; Griglione, A.; Bizzo, H.R.; Rubiolo, P.; Bicchi, C.; Rocha-Leão, M.H.M.; Rezende, C.M. Studies on the volatile fraction composition of three native Amazonian-Brazilian fruits: Murici (Byrsonima crassifolia L., Malpighiaceae), bacuri (Platonia insignis M., Clusiaceae), and sapodilla (Manilkara sapota L., Sapotaceae). Food Chem. 2017, 219, 13–22. [Google Scholar] [CrossRef]
- do Nascimento Cavalcante, A.; Lima, L.K.F.; Araújo, C.M.; da Silva Santos, F.P.; do Nascimento, M.O.; de Castro e Sousa, J.M.; Rai, M.; Feitosa, C.M. Toxicity, cytotoxicity, mutagenicity and in vitro antioxidant models of 2-oleyl-1,3-dipalmitoyl-glycerol isolated from the hexane extract of Platonia insignis MART seeds. Toxicol. Rep. 2020, 7, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Faria, J.V.; Valido, I.H.; Paz, W.H.P.; da Silva, F.M.A.; de Souza, A.D.L.; Acho, L.R.D.; Lima, E.S.; Boleti, A.P.A.; Marinho, J.V.N.; Salvador, M.J.; et al. Comparative evaluation of chemical composition and biological activities of tropical fruits consumed in Manaus, central Amazonia, Brazil. Food Res. Int. 2021, 139, 109836. [Google Scholar] [CrossRef]
- Chendynski, L.T.; Cordeiro, T.; Messias, G.B.; Mantovani, A.C.G.; Spacino, K.R.; Zeraik, M.L.; Borsato, D. Evaluation and application of extracts of rosemary leaves, araçá pulp and peel of bacuri in the inhibition of the oxidation reaction of biodiesel. Fuel 2020, 261, 116379. [Google Scholar] [CrossRef]
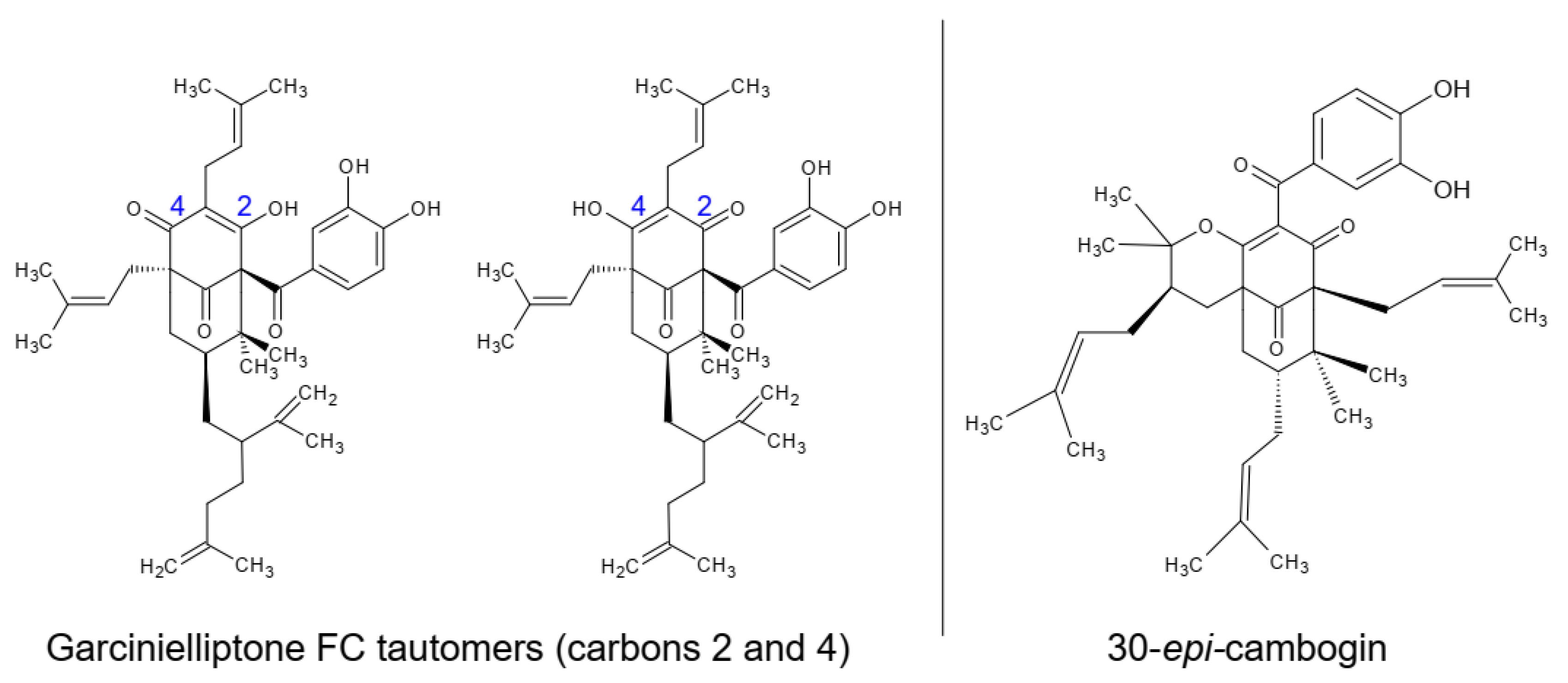
- Da Silva, A.P.D.S.C.L.; Oliveira, G.L.D.S.; Medeiros, S.C.; Sousa, A.M.L.; Lopes, L.D.S.; David, J.M.; Junior, J.S.D.C.; De Freitas, R.M. Pre-clinical toxicology of garcinielliptone FC, a tautomeric pair of polyprenylated benzophenone, isolated from Platonia insignis Mart seeds. Phytomedicine 2016, 23, 477–482. [Google Scholar] [CrossRef]
- Barbosa, P.C.S.; Wiedemann, L.S.M.; Medeiros, R.S.; Da Veiga Junior, V.F. Phytochemical Fingerprints of Copaiba Oils (Copaifera multijuga HAYNE) Determined by Multivariate Analysis. Chem. Biodivers. 2013, 10, 1350–1360. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, C.V.; Dos Santos, A.O.; Ueda-Nakamura, T.; Dias Filho, B.P.; Da Veiga Junior, V.F. Copaiba oil: An alternative to development of new drugs against leishmaniasis. Evid.-Based Complement. Altern. Med. 2012, 2012. [Google Scholar] [CrossRef]
- Morais, B.D.M.; Silva Júnior, M.E.; Andrade, S.A.C.; Converti, A.; Silva, M.F.; Maciel, M.I.S. Optimization of Red Mombin Juice Spray Drying: Physicochemical and Sensory Analyses of Powders. Chem. Eng. Technol. 2020, 43, 1832–1840. [Google Scholar] [CrossRef]
- Koohi, S.; Nasernejad, B.; Zare, M.H.; Elahifard, M.; Shirazian, S.; Ghadiri, M. Extraction of Oxidative Enzymes from Green Tea Leaves and Optimization of Extraction Conditions. Chem. Eng. Technol. 2020, 43, 2548–2556. [Google Scholar] [CrossRef]
- Md Yusof, A.H.; Abd Gani, S.S.; Zaidan, U.H.; Halmi, M.I.E.; Zainudin, B.H. Optimization of an Ultrasound-Assisted Extraction Condition for Flavonoid Compounds from Cocoa Shells (Theobroma cacao) Using Response Surface Methodology. Molecules 2019, 24, 711. [Google Scholar] [CrossRef]
- Antonio, A.D.S.; Aguiar, A.T.C.; Santos, G.R.C.D.; Pereira, H.M.G.; Veiga-Junior, V.F.D.; Wiedemann, L.S.M. Phytochemistry by design: A case study of the chemical composition of: Ocotea guianensis optimized extracts focused on untargeted metabolomics analysis. RSC Adv. 2020, 10, 3459–3471. [Google Scholar] [CrossRef]
- Assefa, A.D.; Saini, R.K.; Keum, Y.S. Extraction of antioxidants and flavonoids from yuzu (Citrus junos Sieb ex Tanaka) peels: A response surface methodology study. J. Food Meas. Charact. 2017, 11, 364–379. [Google Scholar] [CrossRef]
- Hilali, S.; Fabiano-Tixier, A.S.; Ruiz, K.; Hejjaj, A.; Ait Nouh, F.; Idlimam, A.; Bily, A.; Mandi, L.; Chemat, F. Green Extraction of Essential Oils, Polyphenols, and Pectins from Orange Peel Employing Solar Energy: Toward a Zero-Waste Biorefinery. ACS Sustain. Chem. Eng. 2019, 7, 11815–11822. [Google Scholar] [CrossRef]
- Ciğeroğlu, Z.; Bayramoğlu, M.; Kırbaşlar, Ş.İ.; Şahin, S. Comparison of microwave-assisted techniques for the extraction of antioxidants from Citrus paradisi Macf. biowastes. J. Food Sci. Technol. 2020, 58, 1190–1198. [Google Scholar] [CrossRef]
- Alara, O.R.; Abdurahman, N.H.; Ukaegbu, C.I. Extraction of phenolic compounds: A review. Curr. Res. Food Sci. 2021, 4, 200–214. [Google Scholar] [CrossRef]
- Vagiri, M.; Ekholm, A.; Andersson, S.C.; Johansson, E.; Rumpunen, K. An optimized method for analysis of phenolic compounds in buds, leaves, and fruits of black currant (ribes nigrum l.). J. Agric. Food Chem. 2012, 60, 10501–10510. [Google Scholar] [CrossRef] [PubMed]
- Petreska Stanoeva, J.; Balshikevska, E.; Stefova, M.; Tusevski, O.; Simic, S.G. Comparison of the Effect of Acids in Solvent Mixtures for Extraction of Phenolic Compounds From Aronia melanocarpa. Nat. Prod. Commun. 2020, 15. [Google Scholar] [CrossRef]
- Johnson, R.A.; Wichern, D.W. Applied Multivariate Statistical Analysis, 6th ed.; Pearson Prentice Hall: Hoboken, NJ, USA, 2007. [Google Scholar]
- Singleton, V.L.; Rossi, J.A.J. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Chang, C.C.; Yang, M.H.; Wen, H.M.; Chern, J.C. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J. Food Drug Anal. 2002, 10, 178–182. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggent, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS+ radical cation decolorization assay. Free Radic. Biol. Med. 1998, 26, 1231–1237. [Google Scholar] [CrossRef]
- Molyneux, P. The use of the stable free radical diphenylpicrylhydrazyl (DPPH) for estimating antioxidant activity. J. Sci. Tech. 2004, 26, 211–219. [Google Scholar]
- Chemat, F.; Rombaut, N.; Sicaire, A.G.; Meullemiestre, A.; Fabiano-Tixier, A.S.; Abert-Vian, M. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar] [CrossRef]
- Silva, A.P.; Silva, M.P.; Oliveira, C.G.; Monteiro, D.C.; Pinto, P.L.; Mendonça, R.Z.; Costa Júnior, J.S.; Freitas, R.M.; de Moraes, J. Garcinielliptone FC: Antiparasitic activity without cytotoxicity to mammalian cells. Toxicol. Vitr. 2015, 29, 681–687. [Google Scholar] [CrossRef]
- Costa Junior, J.S.; de Almeida, A.A.; Ferraz, A.D.; Rossatto, R.R.; Silva, T.G.; Silva, P.B.; Militão, G.C.; Citó, A.M.; Santana, L.C.; Carvalho, F.A.; et al. Cytotoxic and leishmanicidal properties of garcinielliptone FC, a prenylated benzophenone from Platonia insignis. Nat. Prod. Res. 2013, 27, 470–474. [Google Scholar] [CrossRef]
- Piccinelli, A.L.; Campone, L.; Dal Piaz, F.; Cuesta-Rubio, O.; Rastrelli, L. Fragmentation Pathways of Polycyclic Polyisoprenylated Benzophenones and Degradation Profile of Nemorosone by Multiple-Stage Tandem Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2009, 20, 1688–1698. [Google Scholar] [CrossRef][Green Version]
- Fuller, R.W.; Blunt, J.W.; Boswell, J.L.; Cardellina, J.H.; Boyd, M.R. Guttiferone F, the first prenylated benzophenone from Allanblackia stuhlmannii. J. Nat. Prod. 1999, 62, 130–132. [Google Scholar] [CrossRef] [PubMed]
- Chemat, F.; Abert-Vian, M.; Fabiano-Tixier, A.S.; Strube, J.; Uhlenbrock, L.; Gunjevic, V.; Cravotto, G. Green extraction of natural products. Origins, current status, and future challenges. TrAC—Trends Anal. Chem. 2019, 118, 248–263. [Google Scholar] [CrossRef]
- Chaves, J.O.; de Souza, M.C.; da Silva, L.C.; Lachos-Perez, D.; Torres-Mayanga, P.C.; Machado, A.P.d.F.; Forster-Carneiro, T.; Vázquez-Espinosa, M.; González-de-Peredo, A.V.; Barbero, G.F.; et al. Extraction of Flavonoids From Natural Sources Using Modern Techniques. Front. Chem. 2020, 8, 507887. [Google Scholar] [CrossRef] [PubMed]
- Bae, I.K.; Ham, H.M.; Jeong, M.H.; Kim, D.H.; Kim, H.J. Simultaneous determination of 15 phenolic compounds and caffeine in teas and mate using RP-HPLC/UV detection: Method development and optimization of extraction process. Food Chem. 2015, 172, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Irakli, M.; Chatzopoulou, P.; Ekateriniadou, L. Optimization of ultrasound-assisted extraction of phenolic compounds: Oleuropein, phenolic acids, phenolic alcohols and flavonoids from olive leaves and evaluation of its antioxidant activities. Ind. Crops Prod. 2018, 124, 382–388. [Google Scholar] [CrossRef]
- Li, H.-Z.; Zhang, Z.-J.; Xue, J.; Cui, L.-X.; Hou, T.; Li, X.-J.; Chen, T. Optimization of ultrasound-assisted extraction of phenolic compounds, antioxidants and rosmarinic acid from perilla leaves using response surface methodology. Food Sci. Technol. 2016, 36, 686–693. [Google Scholar] [CrossRef]
- Xu, D.P.; Zheng, J.; Zhou, Y.; Li, Y.; Li, S.; Li, H. Bin Ultrasound-assisted extraction of natural antioxidants from the flower of Limonium sinuatum: Optimization and comparison with conventional methods. Food Chem. 2017, 217, 552–559. [Google Scholar] [CrossRef]

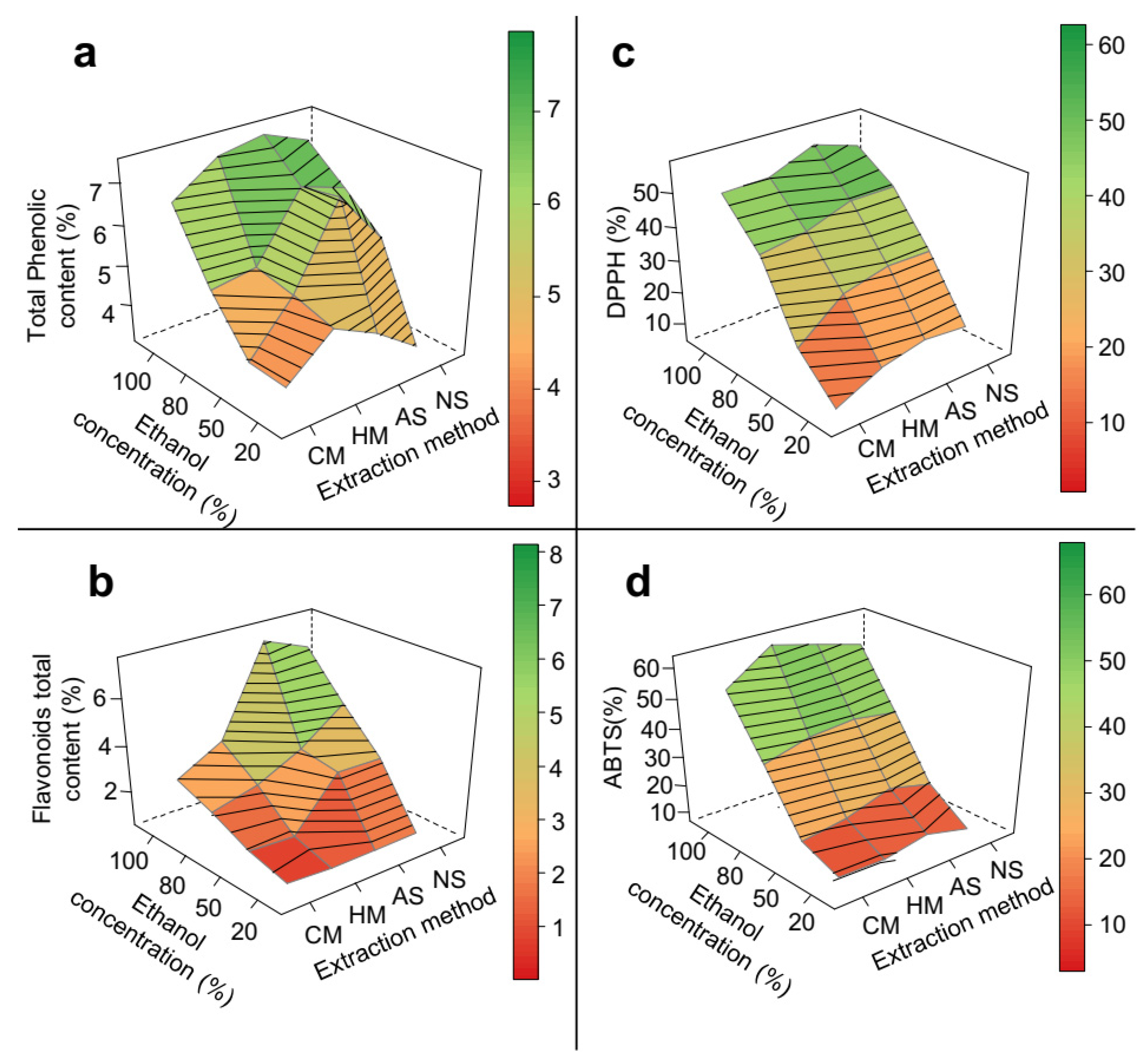
| Sample | Extraction Method | % of Ethanol | Crude Extraction Yield (%) ± SD | Total Phenolic Compounds (% w/w ± SD) | Total Flavonoids (% w/w ± SD) | Antioxidant Capacity Assays (% of Inhibition) | |
|---|---|---|---|---|---|---|---|
| DPPH | ABTS | ||||||
| Mesocarp | Cold maceration | 100 | 30.18 ± 1.45 | 14.60 ± 0.42 | 14.51 ± 0.24 | 77.34 ± 2.13 | 74.09 ± 1.48 ** |
| 80 | 28.22 ± 1.28 | 9.59 ± 0.50 | 10.06 ± 0.36 | 58.16 ± 2.29 | 44.12 ± 3.21 | ||
| 50 | 22.16 ± 1.52 | 5.23 ± 0.30 | 2.54 ± 0.19 | 33.91 ± 1.78 | 25.80 ± 1.47 | ||
| 20 | 13.49 ± 0.61 | 2.64 ± 0.16 | 1.12 ± 0.08 | 15.14 ± 0.76 | 10.06 ± 0.42 | ||
| Hot maceration | 100 | 36.63 ± 0.55 | 15.80 ± 0.83 | 15.05 ± 0.80 | 74.81 ± 1.25 | 73.31 ± 1.21 ** | |
| 80 | 31.95 ± 2.03 | 12.34 ± 0.04 | 12.50 ± 0.42 | 61.10 ± 1.17 | 48.91 ± 3.37 | ||
| 50 | 27.62 ± 0.26 | 6.86 ± 0.30 | 3.72 ± 0.19 | 33.75 ± 1.72 | 24.26 ± 1.18 | ||
| 20 | 20.49 ± 0.85 | 3.64 ± 0.14 | 1.64 ± 0.10 | 16.72 ± 0.78 | 9.95 ± 0.31 | ||
| Neutral sonication | 100 | 27.88 ± 1.36 | 14.12 ± 0.72 | 12.93 ± 0.42 | 72.78 ± 1.16 | 70.38 ± 1.55 | |
| 80 | 25.62 ± 0.62 | 11.50 ± 0.48 | 11.26 ± 0.99 | 54.30 ± 1.12 | 51.05 ± 2.50 | ||
| 50 | 24.26 ± 0.25 | 6.42 ± 0.59 | 4.29 ± 0.15 | 34.26 ± 1.71 | 24.27 ± 1.23 | ||
| 20 | 19.84 ± 0.73 | 2.86 ± 0.23 | 0.97 ± 0.04 | 10.18 ± 0.94 | 8.59 ± 0.66 | ||
| Acid sonication | 100 | 16.98 ± 0.69 | 14.31 ± 0.53 | 13.95 ± 0.62 | 74.14 ± 1.31 | 73.49 ± 2.82 ** | |
| 80 | 18.25 ± 1.34 | 10.22 ± 0.54 | 10.87 ± 0.48 | 54.61 ± 4.20 | 39.18 ± 1.80 | ||
| 50 | 14.95 ± 0.61 | 6.38 ± 0.46 | 6.18 ± 0.41 | 35.02 ± 1.42 | 20.48 ± 1.10 | ||
| 20 | 11.70 ± 0.49 | 3.58 ± 0.19 | 1.06 ± 0.03 | 14.96 ± 1.02 | 10.19 ± 0.41 | ||
| Epicarp | Cold maceration | 100 | 10.90 ± 0.26 | 6.51 ± 0.41 | 2.85 ± 0.15 | 52.14 ± 2.23 | 54.89 ± 1.92 |
| 80 | 17.32 ± 0.56 | 5.47 ± 0.25 | 2.03 ± 0.11 | 34.56 ± 1.78 | 32.90 ± 1.08 | ||
| 50 | 15.79 ± 0.76 | 3.56 ± 0.22 | 1.04 ± 0.09 | 15.47 ± 1.00 | 14.69 ± 0.96 | ||
| 20 | 8.85 ± 0.29 | 3.57 ± 0.18 | 0.68 ± 0.04 | 5.07 ± 0.39 | 9.08 ± 0.59 | ||
| Hot maceration | 100 | 12.69 ± 0.53 | 7.02 ± 0.31 | 3.97 ± 0.23 | 53.36 ± 2.37 | 58.58 ± 2.62 | |
| 80 | 14.47 ± 0.83 | 5.15 ± 0.37 | 2.37 ± 0.16 | 38.84 ± 1.94 | 38.87 ± 0.42 | ||
| 50 | 13.38 ± 0.58 | 4.70 ± 0.26 | 1.09 ± 0.09 | 25.05 ± 1.54 | 17.15 ± 1.06 | ||
| 20 | 10.66 ± 0.59 | 4.40 ± 0.27 | 0.61 ± 0.04 | 13.08 ± 1.03 | 9.12 ± 0.44 | ||
| Neutral sonication | 100 | 4.84 ± 0.23 | 7.14 ± 0.39 | 6.95 ± 0.30 | 53.77 ± 1.75 | 56.44 ± 0.36 | |
| 80 | 7.80 ± 0.38 | 6.24 ± 0.23 | 4.92 ± 0.20 | 46.00 ± 2.33 | 39.99 ± 1.59 | ||
| 50 | 10.94 ± 0.69 | 5.35 ± 0.29 | 2.99 ± 0.16 | 30.06 ± 2.35 | 18.17 ± 0.68 | ||
| 20 | 13.73 ± 0.74 | 3.27 ± 0.15 | 0.57 ± 0.04 | 12.23 ± 0.71 | 7.81 ± 0.54 | ||
| Acid sonication | 100 | 6.40 ± 0.39 | 7.22 ± 0.16 | 6.51 ± 0.39 | 55.91 ± 0.86 | 60.23 ± 2.52 | |
| 80 | 7.97 ± 0.07 | 6.62 ± 0.31 | 3.58 ± 0.25 | 42.63 ± 2.89 | 41.75 ± 2.48 | ||
| 50 | 10.72 ± 0.32 | 6.58 ± 0.32 | 2.92 ± 0.25 | 30.52 ± 0.75 | 22.08 ± 1.23 | ||
| 20 | 13.19 ± 0.87 | 4.02 ± 0.08 | 0.59 ± 0.03 | 14.65 ± 0.93 | 11.00 ± 0.84 | ||
| Mesocarp Samples | Epicarp Samples | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Response-variables | TFC | TPC | CEY | DPPH | ABTS | TFC | TPC | CEY | DPPH | ABTS |
| TFC | 1.0000 | −0.0104 | 0.1124 | 0.2391 | −0.0035 | 1.0000 | −0.0021 | 0.1067 | 0.1312 | 0.2697 |
| TPC | −0.0104 | 1.0000 | −0.3430 | −0.0510 | 0.0334 | −0.0021 | 1.0000 | 0.1552 | −0.1947 | 0.3763 |
| CEY | 0.1124 | −0.3430 | 1.0000 | −0.0862 | 0.0992 | 0.1067 | 0.1552 | 1.0000 | 0.0352 | 0.1225 |
| DPPH | 0.2391 | −0.0510 | −0.0862 | 1.0000 | −0.0684 | 0.1312 | −0.1947 | 0.0352 | 1.0000 | 0.0299 |
| ABTS | −0.0035 | 0.0334 | 0.0992 | −0.0684 | 1.0000 | 0.2697 | 0.3763 | 0.1225 | 0.0299 | 1.0000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamaguchi, K.K.L.; Dias, D.S.; Lamarão, C.V.; Castelo, K.F.A.; Lima, M.S.; Antonio, A.S.; Converti, A.; Lima, E.S.; Veiga-Junior, V.F. Amazonian Bacuri (Platonia insignis Mart.) Fruit Waste Valorisation Using Response Surface Methodology. Biomolecules 2021, 11, 1767. https://doi.org/10.3390/biom11121767
Yamaguchi KKL, Dias DS, Lamarão CV, Castelo KFA, Lima MS, Antonio AS, Converti A, Lima ES, Veiga-Junior VF. Amazonian Bacuri (Platonia insignis Mart.) Fruit Waste Valorisation Using Response Surface Methodology. Biomolecules. 2021; 11(12):1767. https://doi.org/10.3390/biom11121767
Chicago/Turabian StyleYamaguchi, Klenicy K. L., David S. Dias, Carlos Victor Lamarão, Karen F. A. Castelo, Max S. Lima, Ananda S. Antonio, Attilio Converti, Emerson S. Lima, and Valdir F. Veiga-Junior. 2021. "Amazonian Bacuri (Platonia insignis Mart.) Fruit Waste Valorisation Using Response Surface Methodology" Biomolecules 11, no. 12: 1767. https://doi.org/10.3390/biom11121767
APA StyleYamaguchi, K. K. L., Dias, D. S., Lamarão, C. V., Castelo, K. F. A., Lima, M. S., Antonio, A. S., Converti, A., Lima, E. S., & Veiga-Junior, V. F. (2021). Amazonian Bacuri (Platonia insignis Mart.) Fruit Waste Valorisation Using Response Surface Methodology. Biomolecules, 11(12), 1767. https://doi.org/10.3390/biom11121767








