Eclipta prostrata (L.) L. (Asteraceae): Ethnomedicinal Uses, Chemical Constituents, and Biological Activities
Abstract
:1. Introduction
2. Methods
3. Ethnomedicinal Uses
4. Bioactive Chemical Constituents
5. Biological Activities
5.1. Antioxidant Activity
5.2. Antimicrobial Activity
5.3. Hepatoprotective Activities
5.4. Anti Hyperlipidemic Activities
5.5. Cerebroprotective and Nervous System Related Activities
5.6. Anti-Diabetic/Anti-Hyperglycemic Activities
5.7. Anticancer Activities
5.8. Hair Growth Promoting Activity
5.9. Immunomodulatory Activities
5.10. Others Activities
6. Toxicity Evaluation against Brine Shrimp and Mosquito
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CCl4 | Carbon Tetrachloride |
| DGAT | Diacylglycerol Acyltransferase |
| DPPH | 1,1-Diphenyl-2-Picrylhydrazyl |
| EAC | Ehrlich Ascites Carcinoma |
| GPT | Glutamine-Pyruvate Transaminase |
| GOT | Glutamic-Oxaloacetic Transaminase |
| HCV | Hepatitis C Virus |
| HRMCs | Human Renal Mesangial Cells |
| IC50 | Inhibition Concentration 50% |
| MBC | Minimum Bacterial Concentration |
| MIC | Minimum Inhibitory Concentration |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| NF-κB | Nuclear Factor κB |
| RBC | Red Blood Cells |
| TFC | Total Flavonoid Content |
| TG | Triglycerides |
| TPC | Total Phenolic Content |
| TBARS | Thiobarbituric Acid Reactive Substances |
| WBC | White Blood Cells |
References
- Ambu, G.; Chaudhary, R.P.; Mariotti, M.; Cornara, L. Traditional Uses of Medicinal Plants by Ethnic People in the Kavrepalanchok District, Central Nepal. Plants 2020, 9, 759. [Google Scholar] [CrossRef] [PubMed]
- Kunwar, R.M.; Bussmann, R.W. Ethnobotany in the Nepal Himalaya. J. Ethnobiol. Ethnomed. 2008, 4, 24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fitzgerald, M.; Heinrich, M.; Booker, A. Medicinal plant analysis: A historical and regional discussion of emergent complex techniques. Front. Pharmacol. 2020, 10, 1480. [Google Scholar] [CrossRef]
- David, B.; Wolfender, J.L.; Dias, D.A. The pharmaceutical industry and natural products: Historical status and new trends. Phytochem. Rev. 2015, 14, 299–315. [Google Scholar] [CrossRef]
- Khanal, A.; Devkota, H.P.; Kaundinnyayana, S.; Gyawali, P.; Ananda, R.; Adhikari, R. Culinary Herbs and Spices in Nepal: A Review of Their Traditional Uses, Chemical Constituents, and Pharmacological Activities. Ethnobot. Res. Appl. 2021, 21, 1–18. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol. Adv. 2015, 33, 1582–1614. [Google Scholar] [CrossRef] [Green Version]
- Manandhar, N.P. Plants and People of Nepal; Timber Press: Portland, OR, USA, 2002. [Google Scholar]
- Sherchan, J.; Poudel, P.; Sapkota, B.; Jan, H.A.; Bussmann, R.W. Eclipta prostrata (L.) L. Asteraceae. In Ethnobotany of the Himalayas; Kunwar, R.M., Sher, H., Bussmann, R.W., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–19. ISBN 978-3-030-45597-2. [Google Scholar]
- Uddin, M.N.; Rahman, M.A.; Ahmed, N.U.; Rana, M.S.; Akter, R.; Chowdhury, A.M.A. Antioxidant, Cytotoxic and Antimicrobial Properties of Eclipta alba Ethanol Extract. Int. J. Biol. Med. Res. 2010, 4, 341–346. [Google Scholar]
- Baskaran, P.; Jayabalan, N. An Efficient Micropropagation System for Eclipta alba—A Valuable Medicinal Herb. In Vitro Cell. Dev. Biol. Plant 2005, 41, 532–539. [Google Scholar] [CrossRef]
- Adhikari, M.; Thapa, R.; Kunwar, R.M.; Devkota, H.P.; Poudel, P. Ethnomedicinal Uses of Plant Resources in the Machhapuchchhre Rural Municipality of Kaski District, Nepal. Medicines 2019, 6, 69. [Google Scholar] [CrossRef] [Green Version]
- Mansoorali, K.P.; Prakash, T.; Kotresha, D.; Prabhu, K.; Rama Rao, N. Cerebroprotective Effect of Eclipta alba against Global Model of Cerebral Ischemia Induced Oxidative Stress in Rats. Phytomedicine 2012, 19, 1108–1116. [Google Scholar] [CrossRef]
- Gupta, A.; Kumar, A.; Kumar, D.; Nandan, S.; Shankar, K.; Varshney, S.; Rajan, S.; Srivastava, A.; Gupta, S.; Kanojiya, S.; et al. Ethyl Acetate Fraction of Eclipta alba: A Potential Phytopharmaceutical Targeting Adipocyte Differentiation. Biomed. Pharmacother. 2017, 96, 572–583. [Google Scholar] [CrossRef]
- Hussain, I.; Khan, N.; Ullah, R.; Ahmed, S.; Khan, F.A.; Yaz, S. Phytochemical, Physiochemical and Anti-Fungal Activity of Eclipta alba. Afr. J. Pharm. Pharmacol. 2011, 5, 2150–2155. [Google Scholar] [CrossRef]
- Global Biodiversity Information Facility Secretariat (GBIF). Eclipta prostrata (L.) L. Available online: https://www.gbif.org/species/5384950 (accessed on 1 November 2021).
- Bakht, J.; Islam, A.; Ali, H.; Tayyab, M.; Shafi, M. Antimicrobial Potentials of Eclipta alba by Disc Diffusion Method. Afr. J. Biotechnol. 2011, 10, 7658–7667. [Google Scholar] [CrossRef]
- Jayathirtha, M.G.; Mishra, S.H. Preliminary Immunomodulatory Activities of Methanol Extracts of Eclipta alba and Centella asiatica. Phytomedicine 2004, 11, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Datta, K.; Singh, A.T.; Mukherjee, A.; Bhat, B.; Ramesh, B.; Burman, A.C. Eclipta alba Extract with Potential for Hair Growth Promoting Activity. J. Ethnopharmacol. 2009, 124, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Jahan, R.; Al-Nahain, A.; Majumder, S.; Rahmatullah, M. Ethnopharmacological Significance of Eclipta alba (L.) Hassk. (Asteraceae). Int. Sch. Res. Not. 2014, 2014, 385969. [Google Scholar] [CrossRef] [Green Version]
- Dalal, S.; Kataria, S.; Sastry, K.; Rana, S.V.S. Phytochemical Screening of Methanolic Extract and Antibacterial Activity of Active Principles of Hepatoprotective Herb, Eclipta alba. Ethnobot. Leafl. 2010, 2010, 3. [Google Scholar]
- Gautam, T.P. Indigenous Uses of Some Medicinal Plants in Panchthar District, Nepal. Nepal. J. Biosci. 2011, 1, 125–130. [Google Scholar] [CrossRef]
- Diogo, L.C.; Fernandes, R.S.; Marcussi, S.; Menaldo, D.L.; Roberto, P.G.; Matrangulo, P.V.F.; Pereira, P.S.; França, S.C.; Giuliatti, S.; Soares, A.M.; et al. Inhibition of Snake Venoms and Phospholipases A2 by Extracts from Native and Genetically Modified Eclipta alba: Isolation of Active Coumestans. Basic Clin. Pharmacol. Toxicol. 2009, 104, 293–299. [Google Scholar] [CrossRef]
- Puri, H.S. Rasayana: Ayurvedic Herbs for Longevity and Rejuvenation. J. Altern. Complement. Med. 2003, 9, 331–332. [Google Scholar] [CrossRef]
- Rai, M.B. Medicinal Plants of Tehrathum District, Eastern Nepal. Our Nat. 2003, 1, 42–48. [Google Scholar] [CrossRef]
- Badgujar, S.B.; Patil, M.B. Ethnomedicines for Jaundice Used in Tribal Areas of North Maharashtra. Ind. J. Nat. Prod. Resour 2008, 7, 79–81. [Google Scholar]
- Govindarajan, M.; Karuppannan, P. Mosquito Larvicidal and Ovicidal Properties of Eclipta alba (L.) Hassk (Asteraceae) against Chikungunya Vector, Aedes aegypti (Linn.) (Diptera: Culicidae). Asian Pac. J. Trop. Med. 2011, 4, 24–28. [Google Scholar] [CrossRef] [Green Version]
- Rajith, N.P.; Ramachandran, V.S. Ethnomedicines of Kurichyas, Kannur District, Western Ghats, Kerala. Indian J. Nat. Prod. Resour. 2010, 1, 249–253. [Google Scholar]
- Roy, R.K.; Thakur, M.; Dixit, V.K. Hair Growth Promoting Activity of Eclipta alba in Male Albino Rats. Arch. Dermatol. Res. 2008, 300, 357–364. [Google Scholar] [CrossRef]
- Khan, A.V.; Khan, A.A. Ethnomedicinal Uses of Eclipta prostrata Linn. Indian J. Trad. Knowl. 2008, 7, 316–320. [Google Scholar]
- Kumari, C.S.; Govindasamy, S.; Sukumar, E. Lipid Lowering Activity of Eclipta prostrata in Experimental Hyperlipidemia. J. Ethnopharmacol. 2006, 105, 332–335. [Google Scholar] [CrossRef]
- Tewtrakul, S.; Subhadhirasakul, S.; Cheenpracha, S.; Karalai, C. HIV-1 Protease and HIV-1 Integrase Inhibitory Substances from Eclipta prostrata. Phytother. Res. 2007, 21, 1092–1095. [Google Scholar] [CrossRef]
- Neeraja, P.V.; Margaret, E. Eclipta alba (L.) Hassk: A Valuable Medicinal Herb. Int. J. Curr. Pharm. Rev. Res. 2011, 2, 188–197. [Google Scholar]
- Kaushik-Basu, N.; Bopda-Waffo, A.; Talele, T.T.; Basu, A.; Costa, P.R.R.; da Silva, A.J.M.; Sarafianos, S.G.; Noël, F. Identification and Characterization of Coumestans as Novel HCV NS5B Polymerase Inhibitors. Nucleic Acids Res. 2008, 36, 1482–1496. [Google Scholar] [CrossRef] [Green Version]
- Le, D.D.; Nguyen, D.H.; Ma, E.S.; Lee, J.H.; Min, B.S.; Choi, J.S.; Woo, M.H. PTP1B Inhibitory and Anti-Inflammatory Properties of Constituents from Eclipta prostrata L. Biol. Pharm. Bull. 2021, 44, 298–304. [Google Scholar] [CrossRef]
- Wagner, H.; Geyer, B.; Kiso, Y.; Hikino, H.; Rao, G. Coumestans as the Main Active Principles of the Liver Drugs Eclipta alba and Wedelia calendulacea. Planta Med. 1986, 52, 370–374. [Google Scholar] [CrossRef]
- Zhang, J.S.; Guo, Q.M. Studies on the chemical constituents of Eclipta prostrata (L). Yao Xue Xue Bao 2001, 36, 34–37. [Google Scholar]
- Feng, L.; Zhai, Y.-Y.; Xu, J.; Yao, W.-F.; Cao, Y.-D.; Cheng, F.-F.; Bao, B.-H.; Zhang, L. A Review on Traditional Uses, Phytochemistry and Pharmacology of Eclipta prostrata (L.) L. J. Ethnopharmacol. 2019, 245, 112109. [Google Scholar] [CrossRef]
- Yahara, S.; Ding, N.; Nohara, T.; Masuda, K.; Ageta, H. Taraxastane Glycosides from Eclipta alba. Phytochemistry 1997, 44, 131–135. [Google Scholar] [CrossRef]
- Yahara, S.; Ding, N.; Nohara, T. Oleanane Glycosides from Eclipta alba. Chem. Pharm. Bull. 1994, 42, 1336–1338. [Google Scholar] [CrossRef] [Green Version]
- Zhang, M.; Chen, Y.Y.; Di, X.H.; Liu, M. Isolation and identification of ecliptasaponin D from Eclipta alba (L.) Hassk. Yao Xue Xue Bao 1997, 32, 633–634. [Google Scholar]
- Kim, D.-I.; Lee, S.-H.; Choi, J.-H.; Lillehoj, H.S.; Yu, M.-H.; Lee, G.-S. The Butanol Fraction of Eclipta prostrata (Linn) Effectively Reduces Serum Lipid Levels and Improves Antioxidant Activities in CD Rats. Nutr. Res. 2008, 28, 550–554. [Google Scholar] [CrossRef]
- Khanna, V.G.; Kannabiran, K. Anticancer-Cytotoxic Activity of Saponins Isolated from the Leaves of Gymnema Sylvestre and Eclipta prostrata on HeLa Cells. Int. J. Green Pharm. 2009, 3, 227–229. [Google Scholar] [CrossRef]
- Upadhyay, R.K.; Pandey, M.B.; Jha, R.N.; Pandey, V.B. Eclalbatin, a Triterpene Saponin from Eclipta alba. J. Asian Nat. Prod. Res. 2001, 3, 213–217. [Google Scholar] [CrossRef]
- Yu, S.-J.; Yu, J.-H.; Yu, Z.-P.; Yan, X.; Zhang, J.-S.; Sun, J.; Zhang, H. Bioactive Terpenoid Constituents from Eclipta prostrata. Phytochemistry 2020, 170, 112192. [Google Scholar] [CrossRef]
- Xi, F.-M.; Li, C.-T.; Mi, J.-L.; Wu, Z.-J.; Chen, W.-S. Three New Olean-Type Triterpenoid Saponins from Aerial Parts of Eclipta prostrata (L.). Nat. Prod. Res. 2014, 28, 35–40. [Google Scholar] [CrossRef]
- Xi, F.-M.; Li, C.-T.; Han, J.; Yu, S.-S.; Wu, Z.-J.; Chen, W.-S. Thiophenes, Polyacetylenes and Terpenes from the Aerial Parts of Eclipta prostrata. Bioorg. Med. Chem. 2014, 22, 6515–6522. [Google Scholar] [CrossRef]
- Abdel-Kader, M.S.; Bahler, B.D.; Malone, S.; Werkhoven, M.C.M.; van Troon, F.; David, ⊥., II; Wisse, J.H.; Bursuker, I.; Neddermann, K.M.; Mamber, S.W.; et al. DNA-Damaging Steroidal Alkaloids from Eclipta alba from the Suriname Rainforest. J. Nat. Prod. 1998, 61, 1202–1208. [Google Scholar] [CrossRef]
- Sethiya, N.; Tomer, K.; Singh, V.; Kumar, M.; Jaiswal, D.; Yadav, I.; Singh, H.; Chandra, D.; Jain, D. Isolation and Characterization of New Lanosteriod from Ethanolic Extract of Eclipta alba Linn. J. Pharm. Res. 2009, 2, 1635–1637. [Google Scholar]
- Han, L.; Zhao, J.; Zhang, Y.; Kojo, A.; Liu, E.; Wang, T. Chemical Constituents from Dried Aerial Parts of Eclipta prostrata. Chin. Herb. Med. 2013, 5, 313–316. [Google Scholar] [CrossRef]
- Xiong, H.-P.; Xi, F.-M.; Chen, W.-S.; Lu, W.-Q.; Wu, Z.-J. Chemical Constituents of Eclipta prostrata. Chem. Nat. Compd. 2021, 57, 166–168. [Google Scholar] [CrossRef]
- Li, W.; Pang, X.; Han, L.-F.; Zhou, Y.; Cui, Y.-M. Chemical constituents of Eclipta prostrata. China J. Chin. Mater. Medica 2018, 43, 3498–3505. [Google Scholar] [CrossRef]
- Lee, M.K.; Ha, N.R.; Yang, H.; Sung, S.H.; Kim, Y.C. Stimulatory Constituents of Eclipta prostrata on Mouse Osteoblast Differentiation. Phytother. Res. 2009, 23, 129–131. [Google Scholar] [CrossRef]
- Zhao, Y.; Peng, L.; Lu, W.; Wang, Y.; Huang, X.; Gong, C.; He, L.; Hong, J.; Wu, S.; Jin, X. Effect of Eclipta prostrata on Lipid Metabolism in Hyperlipidemic Animals. Exp. Gerontol. 2015, 62, 37–44. [Google Scholar] [CrossRef]
- Meng, X.; Li, B.-B.; Lin, X.; Jiang, Y.-Y.; Zhang, L.; Li, H.-Z.; Cui, L. New Polyacetylenes Glycoside from Eclipta Prostrate with DGAT Inhibitory Activity. J. Asian Nat. Prod. Res. 2019, 21, 501–506. [Google Scholar] [CrossRef]
- Tewtrakul, S.; Subhadhirasakul, S.; Tansakul, P.; Cheenpracha, S.; Karalai, C. Antiinflammatory Constituents from Eclipta prostrata Using RAW264.7 Macrophage Cells. Phytother. Res. 2011, 25, 1313–1316. [Google Scholar] [CrossRef]
- Lee, J.-S.; Ahn, J.-H.; Cho, Y.-J.; Kim, H.-Y.; Yang, Y.-I.; Lee, K.-T.; Jang, D.-S.; Choi, J.-H. α-Terthienylmethanol, Isolated from Eclipta prostrata, Induces Apoptosis by Generating Reactive Oxygen Species via NADPH Oxidase in Human Endometrial Cancer Cells. J. Ethnopharmacol. 2015, 169, 426–434. [Google Scholar] [CrossRef]
- Yu, S.-J.; Zhang, J.-S.; He, H.; Yu, J.-H.; Bao, J.; Zhang, H. Thiophene Enantiomers from the Aerial Parts of Eclipta prostrata. J. Asian Nat. Prod. Res. 2021, 23, 745–753. [Google Scholar] [CrossRef]
- Yu, S.-J.; Yu, J.-H.; He, F.; Bao, J.; Zhang, J.-S.; Wang, Y.-Y.; Zhang, H. New Antibacterial Thiophenes from Eclipta prostrata. Fitoterapia 2020, 142, 104471. [Google Scholar] [CrossRef]
- Boregowda, R.S.; Murali, N.; Udayashankar, A.C.; Niranjana, S.R.; Lund, O.S.; Prakash, H.S. Antifungal Activity of Eclipta alba Metabolites against Sorghum Pathogens. Plants 2019, 8, 72. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.Y. Enhancement of Skin Anti-Inflammatory Activities of Eclipta prostrata L. from the Ultrasonic Extraction Process. Appl. Sci. 2017, 7, 1227. [Google Scholar] [CrossRef] [Green Version]
- Kang, E.Y.; Kim, H.K.; Jung, J.Y.; Kim, J.H.; Woo, T.K.; Choi, J.I.; Kim, J.H.; Ahn, C.; Lee, H.G.; Go, G.-W. Combined Extract of Leonurus Japonicus Houtt, Eclipta prostrata L., and Pueraria Lobata Ohwi Improved Hot Flashes and Depression in an Ovariectomized Rat Model of Menopause. Foods 2021, 10, 180. [Google Scholar] [CrossRef]
- Patel, M.; Verma, R.; Srivastav, P. Antioxidant Activity of Eclipta alba Extract. J. Med. Plants Stud. 2016, 4, 92–98. [Google Scholar]
- Gurrapu, S.; Mamidala, E. In Vitro Antibacterial Activity of Alkaloids Isolated from Leaves of Eclipta alba against Human Pathogenic Bacteria. Pharmacogn. J. 2017, 9, 573–577. [Google Scholar] [CrossRef] [Green Version]
- Thirumalai, T.; David, E.; Therasa, S.V.; Elumalai, E. Restorative Effect of Eclipta alba in CCl4 Induced Hepatotoxicity in Male Albino Rats. Asian Pac. J. Trop. Dis. 2011, 1, 304–307. [Google Scholar] [CrossRef]
- Lal, V.K.; Kumar, A.; Kumar, P.; Yadav, K.S. Screening of Leaves and Roots of Eclipta alba for Hepatoprotective Activity. Arch. Appl. Sci. Res. 2010, 2, 86–94. [Google Scholar]
- Saxena, A.K.; Singh, B.; Anand, K.K. Hepatoprotective Effects of Eclipta alba on Subcellular Levels in Rats. J. Ethnopharmacol. 1993, 40, 155–161. [Google Scholar] [CrossRef]
- Lee, M.K.; Ha, N.R.; Yang, H.; Sung, S.H.; Kim, G.H.; Kim, Y.C. Antiproliferative Activity of Triterpenoids from Eclipta prostrata on Hepatic Stellate Cells. Phytomed. Int. J. Phytother. Phytopharm. 2008, 15, 775–780. [Google Scholar] [CrossRef]
- Helmy, A.S.; Sherif, N.M.; Ghanem, H.Z.; Ibrahim, N.A.; El Gendy, A.N.G.; Hussein, N.S.; Abdel-Hamid, A.-H.Z. Targeted Metabolomics Reveals the Therapeutic Impact of Eclipta prostrata on Diet-Induced Non-Alcoholic Fatty Liver Disease in Rats. J. Appl. Pharm. Sci. 2019, 9, 77–90. [Google Scholar]
- Banji, O.; Banji, D.; Annamalai, A.; Manavalan, R. Investigation on the Effect of Eclipta alba on Animal Models of Learning and Memory. Indian J. Physiol. Pharmacol. 2007, 51, 274–278. [Google Scholar]
- Kim, D.-I.; Lee, S.-H.; Hong, J.-H.; Lillehoj, H.S.; Park, H.-J.; Rhie, S.-G.; Lee, G.-S. The Butanol Fraction of Eclipta prostrata (Linn) Increases the Formation of Brain Acetylcholine and Decreases Oxidative Stress in the Brain and Serum of Cesarean-Derived Rats. Nutr. Res. 2010, 30, 579–584. [Google Scholar] [CrossRef]
- Tambe, R.; Patil, A.; Jain, P.; Sancheti, J.; Somani, G.; Sathaye, S. Assessment of Luteolin Isolated from Eclipta alba Leaves in Animal Models of Epilepsy. Pharm. Biol. 2017, 55, 264–268. [Google Scholar] [CrossRef] [Green Version]
- Alam, N.; Sharma, K.r. Estimation of Phenolic Content, Flavonoid Content, Antioxidant, and Alpha Amylase Inhibitiory Activity of Some Selected Plant from Siraha District of Nepal. Asian J. Pharm. Clin. Res. 2020, 13, 18–23. [Google Scholar] [CrossRef]
- Shahab, U.; Faisal, M.; Alatar, A.A.; Ahmad, S. Impact of Wedelolactone in the Anti-Glycation and Anti-Diabetic Activity in Experimental Diabetic Animals. IUBMB Life 2018, 70, 547–552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jaiswal, N.; Bhatia, V.; Srivastava, S.P.; Srivastava, A.K.; Tamrakar, A.K. Antidiabetic Effect of Eclipta alba Associated with the Inhibition of Alpha-Glucosidase and Aldose Reductase. Nat. Prod. Res. 2012, 26, 2363–2367. [Google Scholar] [CrossRef]
- Hemalatha, S.; Ayyappan, T.; Shanmugan, S.; Nagavalli, D.; Shrivijaya Kirubha, T. Evaluation of Antidiabetic and Diuretic Activity of Polyherbal Formulation. Indian J. Tradit. Knowl. 2006, 5, 468–470. [Google Scholar]
- Ananthi, J.; Prakasam, A.; Pugalendi, K.V. Antihyperglycemic Activity of Eclipta alba Leaf on Alloxan-Induced Diabetic Rats. Yale J. Biol. Med. 2003, 76, 97–102. [Google Scholar]
- Singh, A.; Singh, A.; Dwivedi, V. Screening of Hydro-Alcoholic Extract of Eclipta alba for Its Anticancerous Efficacy. Int. J. Sci. Res. 2017, 6, 488–491. [Google Scholar]
- Arya, R.K.; Singh, A.; Yadav, N.K.; Cheruvu, S.H.; Hossain, Z.; Meena, S.; Maheshwari, S.; Singh, A.K.; Shahab, U.; Sharma, C.; et al. Anti-Breast Tumor Activity of Eclipta Extract in-Vitro and in-Vivo: Novel Evidence of Endoplasmic Reticulum Specific Localization of Hsp60 during Apoptosis. Sci. Rep. 2015, 5, 18457. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.-Y.; Kim, H.M.; Ryu, B.; Lee, J.-S.; Choi, J.-H.; Jang, D.S. Constituents of the Aerial Parts of Eclipta prostrata and Their Cytotoxicity on Human Ovarian Cancer Cells in Vitro. Arch. Pharm. Res. 2015, 38, 1963–1969. [Google Scholar] [CrossRef]
- Chaudhary, H.; Dhuna, V.; Singh, J.; Kamboj, S.S.; Seshadri, S. Evaluation of Hydro-Alcoholic Extract of Eclipta alba for Its Anticancer Potential: An in Vitro Study. J. Ethnopharmacol. 2011, 136, 363–367. [Google Scholar] [CrossRef]
- Mondal, S.; Ghosh, D.; Ganapaty, S.; Sushrutha, M. Preliminary Phytochemical Analysis and Evaluation of Hair Growth Stimulating Potential of Ethanol Extract from L. (Asteraceae) Leaves in Wistar Albino Eclipta alba Rats. Asian J. Pharm. Pharmacol. 2016, 2, 121–127. [Google Scholar]
- Begum, S.; Lee, M.R.; Gu, L.J.; Hossain, J.; Sung, C.K. Exogenous Stimulation with Eclipta alba Promotes Hair Matrix Keratinocyte Proliferation and Downregulates TGF-Β1 Expression in Nude Mice. Int. J. Mol. Med. 2015, 35, 496–502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roy, R.K.; Thakur, M.; Dixit, V.K. Development and Evaluation of Polyherbal Formulation for Hair Growth–Promoting Activity. J. Cosmet. Dermatol. 2007, 6, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Christybapita, D.; Divyagnaneswari, M.; Michael, R.D. Oral Administration of Eclipta alba Leaf Aqueous Extract Enhances the Non-Specific Immune Responses and Disease Resistance of Oreochromis mossambicus. Fish Shellfish Immunol. 2007, 23, 840–852. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Kang, W.; Zhao, J.; Liu, G.; Zhao, M. Osteoprotective Effect of Echinocystic Acid, a Triterpone Component from Eclipta prostrata, in Ovariectomy-Induced Osteoporotic Rats. PLoS ONE 2015, 10, e0136572. [Google Scholar] [CrossRef] [Green Version]
- Dungca, N.T.P. Protective Effect of the Methanolic Leaf Extract of Eclipta alba (L.) Hassk. (Asteraceae) against Gentamicin-Induced Nephrotoxicity in Sprague Dawley Rats. J. Ethnopharmacol. 2016, 184, 18–21. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Huang, B.; Li, C.; Yang, B.; Jia, X.; Feng, L. The Combination of HPLC and Biological Analysis to Determine the Quality Markers and Its Structural Composition of Eclipta prostrata L. Phytochem. Anal. 2020, 31, 968–981. [Google Scholar] [CrossRef]
- Shen, P.; Yang, X.; Jiang, J.; Wang, X.; Liang, T.; He, L. Wedelolactone from Eclipta alba Inhibits Lipopolysaccharide-Enhanced Cell Proliferation of Human Renal Mesangial Cells via NF-ΚB Signaling Pathway. Am. J. Transl. Res. 2017, 9, 2132–2142. [Google Scholar]

| Chemical Compounds (Class/Constituents) | Plant Parts | References |
|---|---|---|
| Coumestan derivatives | ||
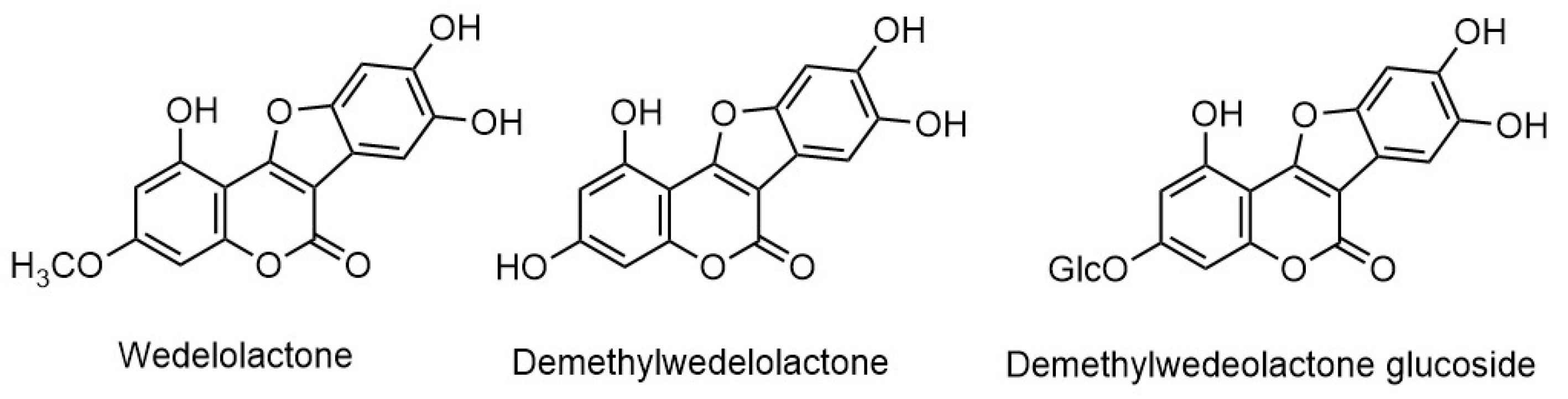
| Wedelolactone | Leaves | [21,33,34,35,36] |
| Demethylwedelolactone | Leaves | [35,36] |
| Isodemethylwedelolactone | Whole plant | [36] |
| Strychnolactone | Whole plant | [36] |
| Demethylwedelolactone glucoside | Aerial Parts | [37] |
| Steroidal and triterpene saponins, steroidal alkaloids, steroids and triterpenoids | ||
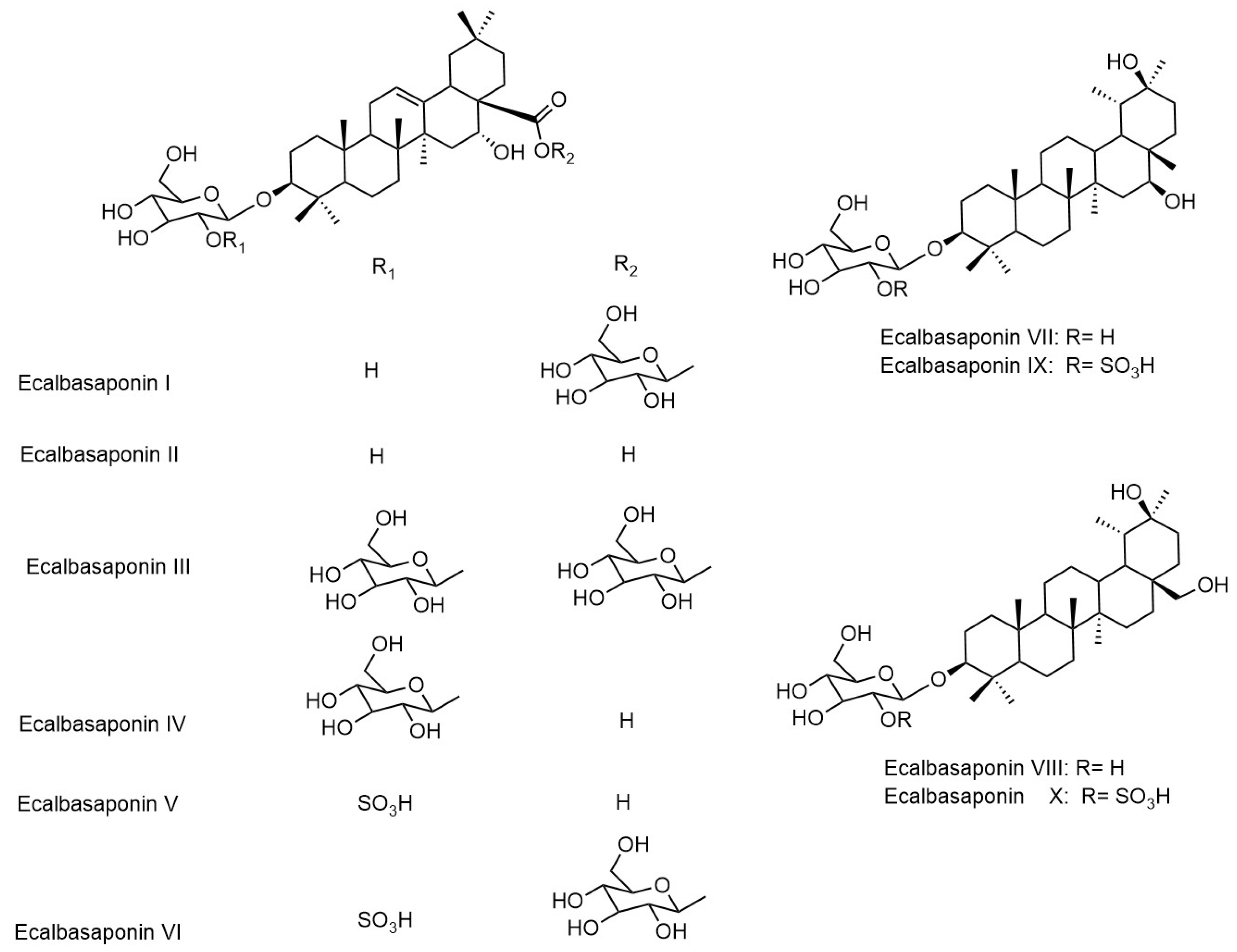
| Eclalbasaponins I | Whole plant | [34,38,39] |
| Eclalbasaponins II | Whole plant | [34,38,39] |
| Eclalbasaponins III | Whole plant | [34,38,39] |
| Eclalbasaponins IV | Whole plant | [38,39] |
| Eclalbasaponins V | Whole plant | [34,38,39] |
| Eclalbasaponins VI | Whole plant | [38,39] |
| Eclalbasaponins VII | Whole plant | [38] |
| Eclalbasaponins VIII | Whole plant | [38] |
| Eclalbasaponins IX | Whole plant | [38] |
| Eclalbasaponins X | Whole plant | [38] |
| Eclalbasaponin XI | Whole plant | [38] |
| Eclalbasaponin XII | Whole plant | [38] |
| Eclalbasaponin XIII | Whole plant | [38] |
| Eclalbasaponin A | Whole plant | [40] |
| Eclalbasaponin B | Whole plant | [40] |
| Eclalbasaponin C | Whole plant | [40] |
| Eclalbasaponin D | Whole plant | [40] |
| Echinocystic acid | Whole plant | [40] |
| Echinocystic acid-3-O-(6-O-acetyl)-β-D-glucopyranoside | Aerial parts | [41] |
| Eclalbatin | Aerial Parts | [42,43] |
| 3β,25-Dihydroxy-23E-lemmaphyll-8,23-diene | Whole plant | [44] |
| 16α-Hydroxy-olean-12-en-3-on-28,21β-olide | Whole plant | [44] |
| 3β-Hydroxy-17-epi-28-norolean-12-en-16-one 3-O-β-D-glucopyranoside | Whole plant | [44] |
| 3β-O-(6-O-Crotonyl-β-D-glucopyranosyl)-16α-hydroxy-olean-12-en-28-oic acid 28-O-β-D-glucopyranosyl ester | Whole plant | [44] |
| 3-O-(2-O-Acetyl-β-D-glucopyranosyl) oleanolic acid-28-O-(β-D-glucopyranosyl) ester | Aerial parts | [45] |
| 3-O-(6-O-Acetyl-β-D-glucopyranosyl) oleanolic acid-28-O-(β-D-glucopyranosyl) ester | Aerial parts | [45] |
| 3-O-(β-D-Glucopyranosyl) oleanolic acid-28-O-(6-O-acetyl-β-D-glucopyranosyl) ester | Aerial parts | [45] |
| 3β,16β,29-Trihydroxy oleanane-12-ene-3-O-β-D-glucopyranoside | Aerial parts | [46] |
| 3,28-di-O-β-D-Glucopyranosyl-3β,16β-dihydroxy oleanane-12-ene-28-oleanlic acid | Aerial parts | [46] |
| 3-O-β-D-Glucopyranosyl-(1-2)-β-D-glucopyranosyl oleanlic-18- ene acid-28-O-β-D-glucopyranoside | Aerial parts | [46] |
| (20S)(25S)-22,26-Imino-cholesta-5,22(N)-dien-3β-ol (Verazine) | Leaves | [47] |
| 20-epi-3-Dehydroxy-3-oxo-5,6-dihydro-4,5-dehydroverazine | Leaves | [32,47] |
| (20R)-20-Pyridyl-cholesta-5-ene-3β,23-diol (Ecliptalbine) | Leaves | [47] |
| (20R)-25β-Hydroxyverazine | Leaves | [47] |
| 20-epi-4β-Hydroxyverazine | Leaves | [47] |
| 20-epi-25β-Hydroxyverazin | Leaves | [47] |
| 4β-Hydroxyverazine | Leaves | [47] |
| 25β-Hydroxyverazine | Leaves | [47] |
| Lanost-5,24-dien-3β-ol-18, 21-olide -3β- yl tetradecanoate | Whole plant | [48] |
| α-Amyrin | Whole plant | [43] |
| Ursolic acid | Whole plant | [43] |
| Oleanolic acid | Whole plant | [43] |
| 3-Oxo-16α-hydroxy-olean-12-en-28-oic acid | Aerial parts | [49] |
| Machaeroceric acid | Aerial parts | [34] |
| Silphioside C | Whole plant | [50] |
| β-Sitosterol | Whole plant | [36] |
| Stigmasterol | Leaves/Stems | [40] |
| Stigmasterol-3-O-glucoside | Aerial parts/leaves/Stems | [19,34,40] |
| 3-O-(6′-O-Palmitoyl-β-D-glucopyranosyl) stigmasterol | Whole plant | [50] |
| Daucosterol | Leaves/Stems | [40] |
| Flavonoids | ||
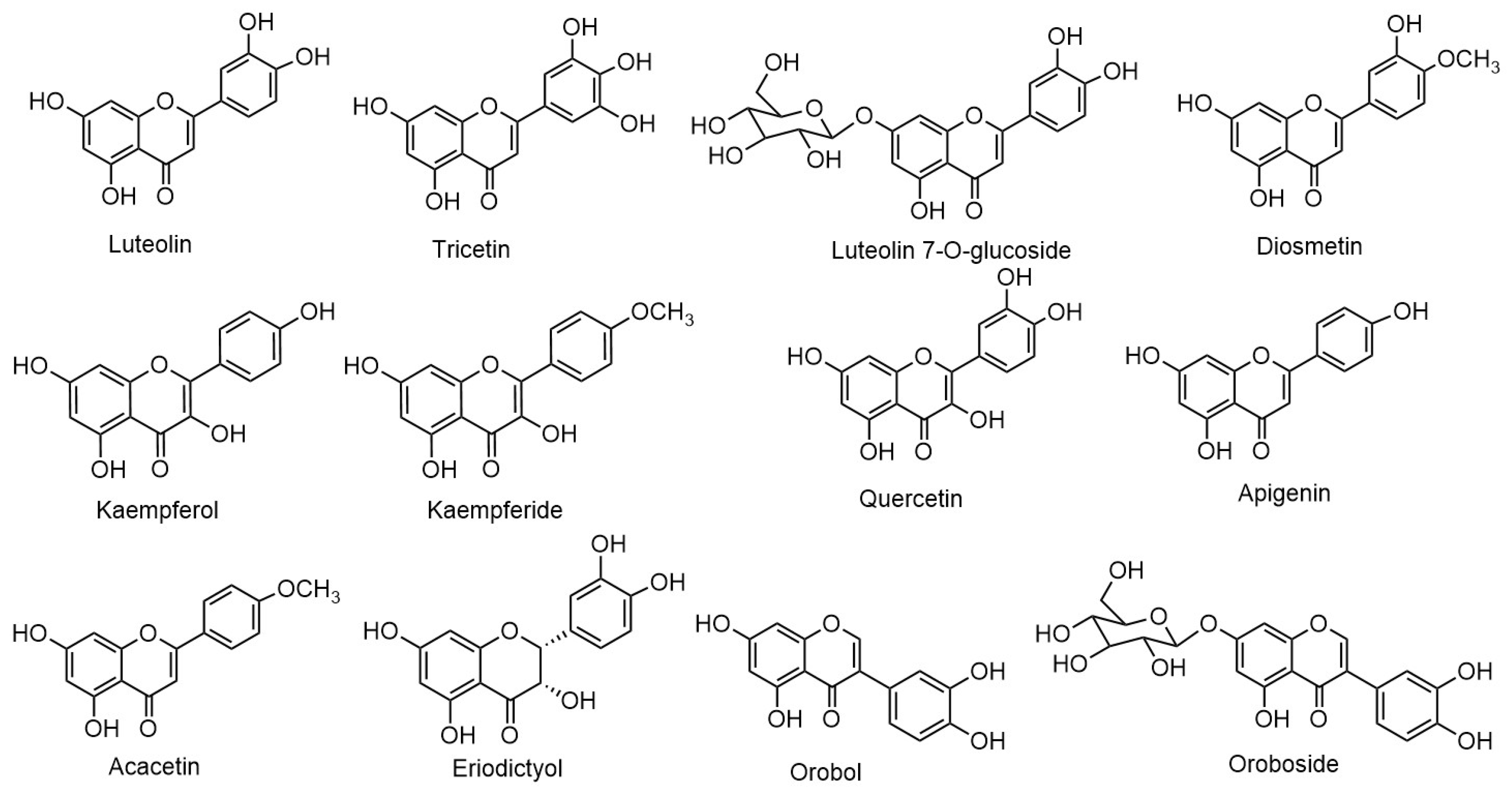
| Luteolin | Aerial parts | [34,41,51] |
| Tricetin | Aerial parts | [34] |
| Luteolin-7-O-β-D-glucoside | Aerial parts | [34,41,51] |
| Diosmetin | Aerial parts | [52] |
| Skullcapflavone Ⅱ | Whole plant | [51] |
| Kaempferol | Whole plant | [51] |
| Kaempferol-7-O-α-D-rhamnoside | Aerial parts | [34] |
| Kaempferide | Whole plant | [51] |
| Quercetin | Aerial parts | [51,53] |
| Quercetin-3-O-β-D-glucoside | Aerial parts | [34] |
| Apigenin | Aerial parts | [34,41,51] |
| Acacetin | Whole plant | [51] |
| Acacetin-7-O-rutinoside | Whole plant | [51] |
| Eriodictyol | Whole plant | [50] |
| Pyracanthoside | Whole plant | [50] |
| Hesperetin-7-O-β-D-glucoside | Aerial parts | [34] |
| 3′-Hydroxybiochanin A | Aerial Parts | [37,49] |
| Orobol (isoluteolin) | Whole plant | [31,34] |
| 7-O-Methylorobol-4′-O-β-D-glucopyranoside | Aerial Parts | [34,49,50] |
| 7-Dihydroxyl-3′, 6′-dimethoxylisoflavone-7-O-glucoside | Whole plant | [51] |
| 3′-O-Methylorobol | Aerial parts | [50,52] |
| Pratensein | Aerial parts | [37,49,50] |
| Pratensein-7-O-β-D-glucopyranoside | Aerial parts | [41,50] |
| Oroboside (Orobol-7-O-β-D-glucoside) | Whole plant | [34,37,50,51] |
| Phenolic acids | ||
| Protocatechuic acid | Leaves/Steam/Whole plant | [34,36,40,43] |
| 4-Hydroxybenzoic acid | Leaves/Steam | [34,40,43] |
| Vanillic acid | Aerial parts | [34] |
| Syringic acid | Aerial parts | [34] |
| Chlorogenic acid | Aerial parts | [34] |
| Syringic acid | Aerial parts | [34] |
| Tachinoside | Whole plant | [50] |
| Coniferylaldehyde | Whole plant | [50] |
| Leonuriside A | Whole plant | [50] |
| Caffeic acid | Whole plant | [50] |
| Ferulic acid ethyl ester | Whole plant | [50] |
| Caffeic acid ethyl ester | Whole plant | [50] |
| Lignin | ||
| Ecliptalignin A | ||
| Coumarins | ||
| Psoralen | Whole plant | [51] |
| Isopsoralen | Whole plant | [51] |
| Polyacetylinic compounds | ||
| (5E)-Hendeca-1,5- dien-7,9-diyne-diol-4-O-β-D-glucopyranoside | Stem | [54] |
| (5E)-Trideca-1,5-dien-7,9,11-triyne-3,4-diol-4-O-β-D-glucopyranoside | Stem | [46,54] |
| 3-O-β-D-Glucopyranosyl1-hydroxy-4E,6E-tetradecene,8,10,12-triyne | Stem | [46,54] |
| 2-O-β-D-Glucosyltrideca-3E,11E-dien5,7,9-triyne-1,2,13-triol | Stem | [54] |
| 2-O-β-D-Glucosyltrideca-3E,11E-dien-5,7,9-triyne-1,2-diol | Stem | [54] |
| 2-O-β-D-Glucosyltrideca-3E,11Z-dien-5,7,9-triyne3–1,2-diol | Stem | [54] |
| Substituted thiophenes | ||
| 5-Hydroxymethyl-(2,2′:5′,2″)-terthienyl tiglate | Whole plant | [55] |
| 5-Hydroxymethyl-(2,2′:5′,2″)-terthienyl agelate | Whole plant | [55] |
| 5-Hydroxymethyl-(2,2′:5′,2″)-terthienyl acetate | Whole plant | [55] |
| 5-Formyl-(2, 2:5, 2″)-terthiophene (Ecliptal) | Whole plant | [56] |
| 5-Hydroxymethyl-(2, 2: 5, 2″)-terthiophene (α-terthienylmethanol) | Whole plant | [56] |
| 5-Methoxy-(2, 2:5, 2″)-terthiophene | Whole plant | [56] |
| 3′-Methoxy-2,2′:5′,2″-terthiophene | Aerial parts | [41] |
| 5-(3″,4″-Dihydroxy-1″-butynyl)-2,2′-bithiophene | Aerial parts | [41] |
| α-Terthienyl | Aerial parts | [41] |
| α-Formylterthienyl | Whole plant | [54] |
| α-Terthienyl methanol | Whole plant | [41,54,56] |
| 3′-Methoxy-2,2′:5′,2″-terthiophene | Aerial parts | [41] |
| 4-(2,2′-Bithiophen-5-yl)but-3-yne-1,2-diol | Aerial parts | [57] |
| Arctinol B | Aerial parts | [57] |
| 2-(Penta-1,3-diynyl)-5-(3,4-dihydroxy-but-1-ynyl)-thiophene | Aerial parts | [57] |
| 6-Methoxy-arctinol-b | Aerial parts | [57] |
| 5-[l-(4-Hydroxybut-l-ynyl)]-2,20 -bithiophene-50 -carbaldehyde | Aerial parts | [57] |
| 5-Hydroxymethyl- (2,2′:5′,2′’-terthienyl) | Aerial parts | [57] |
| 5′-Hydroxymethyl-5-(3-butene-1-ynyl)-2,2′ -bithiophene | Aerial parts | [46,57] |
| 3′-Hydroxy-2,2′:5′,2′’ terthiophene-3′-O-β-D-glucopyranoside | Aerial parts | [57] |
| Ecliprostin A | Aerial parts | [58] |
| Ecliprostin B | Aerial parts | [58] |
| Ecliprostin C | Aerial parts | [58] |
| Alkaloids | ||
| Crinumaquine | Whole plant | [51] |
| 2,3,9,12-Tetramethoxyprotoberberine | Whole plant | [51] |
| Lignans | ||
| Pinoresinol-4-O-β-D-glucopyranoside | Whole plant | [50] |
| 4,4′-Dimethoxy-3′-hydroxy-7,9′:7′,9-diepoxylignan-3-O-β-D-glucopyranoside | Whole plant | [50] |
| Syringaresinol-4′-O-β-D-glucopyroside | Whole plant | [50] |
| Lanicepside A | Whole plant | [50] |
| Longifloroside | Whole plant | [50] |
| Other compounds | ||
| 1-O-Octadecanoyl-2-O-(9Z,12Z-octadecadienoyl)-3-O-[α-D-galactopyranosyl- (1′′→6′)-O-β-D-galactopyranosyl]glycerol | Whole plant | [50] |
| (2S)-3-O-α-D-Galactopyranosyl-(1′′→6′)-β-D-galactopyranosyl-1,2-di-O-[(9Z,12Z,15Z)-octadeca-9,12,15-trienoyl]-sn-glycerol | Whole plant | [50] |
| 1-O-(9Z,12Z,15Z-Octadecatrienoyl)-2-O-hexadecanoyl-3-O-[α-D-galactopyranosyl-(1′′→6′)-O-β-D-galactopyranosyl]glycerol | Whole plant | [50] |
| 1-O-(β-D-glucopyranosyl)- (2S,3S,4R,8Z)-2N-[(2′R)-2′-hydroxytetracosanoyl]-8-(Z)-octadecene-1,3,4-triol | Whole plant | [50] |
| (2S,3S,4R,10E)-2-[(2′R)-2′- Hydroxytetracosanoylamino]-10-octadecene-1,3,4-triol | Whole plant | [50] |
| (3S,5R,6S,7E,9R)-3-Hydroxy-5,6-epoxy-β-ionyl-3-O-β-D-glucopyranoside | Whole plant | [50] |
| Euodionoside A | Whole plant | [50] |
| Junipeionoloside | Whole plant | [50] |
| Calaliukiuenoside | Whole plant | [50] |
| rel-(1S,2S,3S,4R,6R)-1,6-Epoxy-menthane-2,3-diol-3-O-β-D-glucopyranoside | Aerial parts | [46] |
| rel-(1S,2S,3S,4R,6R)-3-O-(6-O-caffeoyl-β-D-glucopyranosyl)-1,6-epoxy menthane-2,3-diol | Aerial parts | [46] |
| Siliphioside E | Aerial parts | [46] |
| (2E,6E)- 2,6,10-trimethyl-2,6,11-dodecatriene-1,10-diol-1-O-β-D-glucopyranoside | Aerial parts | [46] |
| (2S)-1-O-Stearoyl-3-O-β-D-galactopyranosyl-sn-glycerol | Aerial parts | [34] |
| (2S)-3-O-(9Z,12Z-Octadecadienoyl) glyceryl-O-β-D-galactopyranoside | Aerial parts | [34] |
| Bidensmenthoside A | Whole plant | [50] |
| Bidensmenthoside B | Whole plant | [50] |
| 11β,17-Dihydroxy-beyer-15-ene | Whole plant | [44] |
| 4β- Hydroxy-guai-10(14),11(13)-dien-12-oic acid | Whole plant | [44] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Timalsina, D.; Devkota, H.P. Eclipta prostrata (L.) L. (Asteraceae): Ethnomedicinal Uses, Chemical Constituents, and Biological Activities. Biomolecules 2021, 11, 1738. https://doi.org/10.3390/biom11111738
Timalsina D, Devkota HP. Eclipta prostrata (L.) L. (Asteraceae): Ethnomedicinal Uses, Chemical Constituents, and Biological Activities. Biomolecules. 2021; 11(11):1738. https://doi.org/10.3390/biom11111738
Chicago/Turabian StyleTimalsina, Deepak, and Hari Prasad Devkota. 2021. "Eclipta prostrata (L.) L. (Asteraceae): Ethnomedicinal Uses, Chemical Constituents, and Biological Activities" Biomolecules 11, no. 11: 1738. https://doi.org/10.3390/biom11111738
APA StyleTimalsina, D., & Devkota, H. P. (2021). Eclipta prostrata (L.) L. (Asteraceae): Ethnomedicinal Uses, Chemical Constituents, and Biological Activities. Biomolecules, 11(11), 1738. https://doi.org/10.3390/biom11111738







