Hyperbaric Oxygen Treatment—From Mechanisms to Cognitive Improvement
Abstract
1. Hyperbaric Oxygen Treatment (HBOT): The Concept
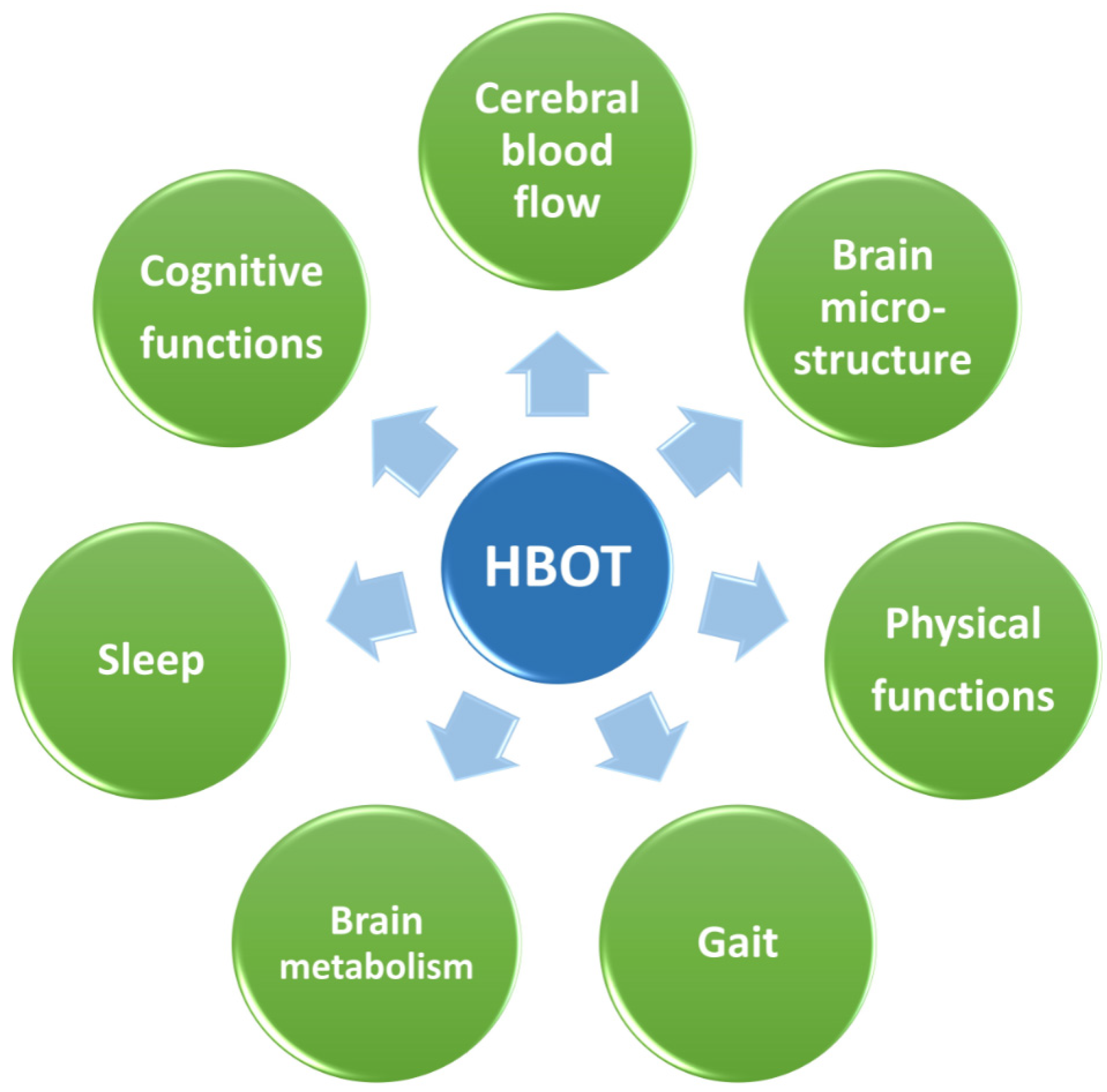
2. Cognitive Improvement
2.1. Cognitive Improvement Following Brain Injuries
2.2. Cognitive Improvement Following HBOT in AD and VD
2.3. Cognitive Improvement in Healthy Individuals
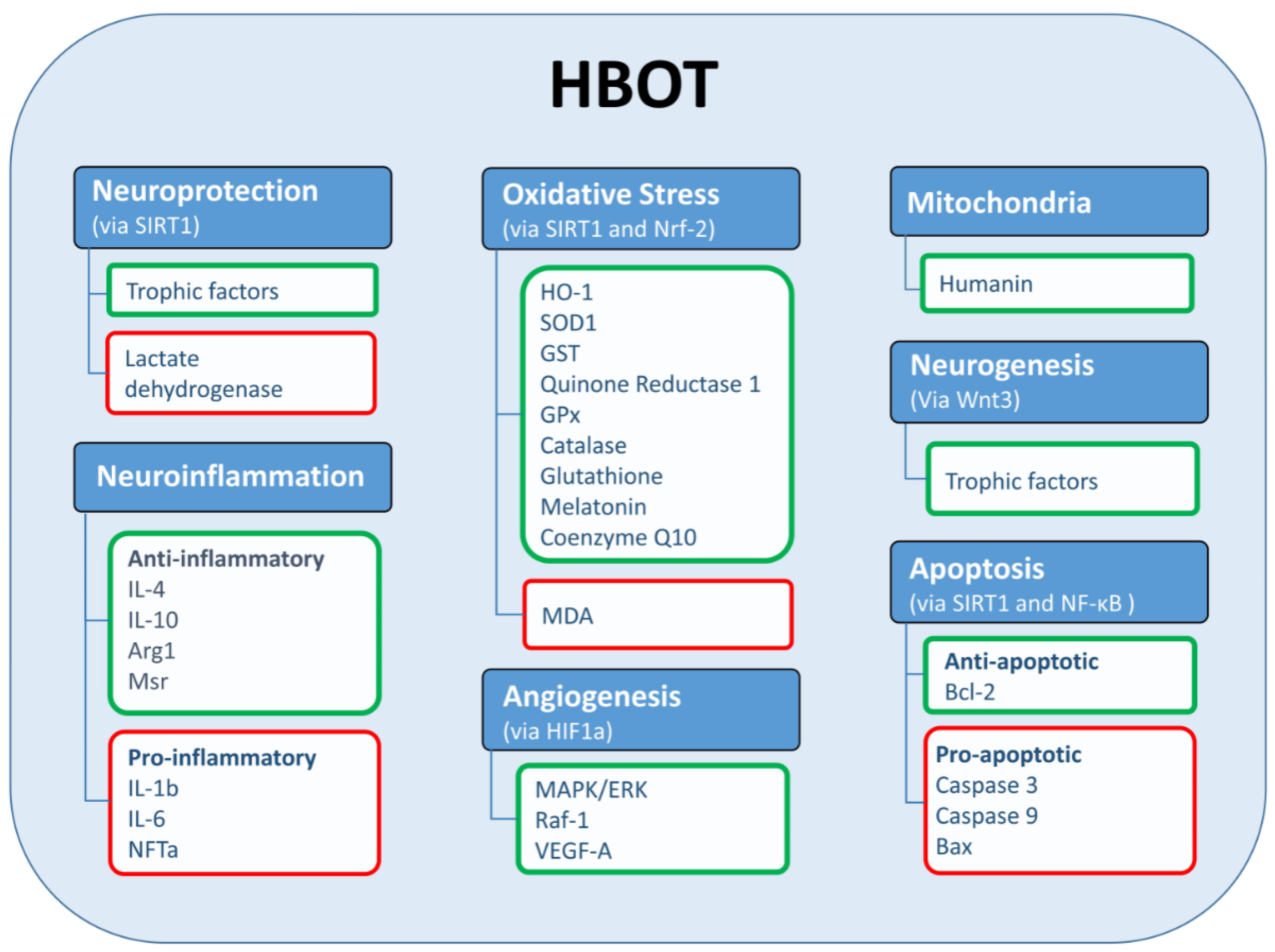
3. Mechanistic Explanation for the Effects of HBOT on Cognition
3.1. Mitochondrial Function
3.2. Neurogenesis and Angiogenesis
3.3. Neuroinflammation
3.4. Neuroprotective, Antioxidant, and Antiapoptotic Activities
4. HBOT—The Next Leap
Author Contributions
Funding
Conflicts of Interest
References
- Calvert, J.W.; Cahill, J.; Zhang, J.H. Hyperbaric oxygen and cerebral physiology. Neurol. Res. 2007, 29, 132–141. [Google Scholar] [CrossRef]
- Jones, M.W.; Brett, K.; Han, N.; Wyatt, H.A. Hyperbaric Physics; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Glik, J.; Cholewka, A.; Stanek, A.; Englisz, B.; Sieroń, K.; Mikuś-Zagórska, K.; Knefel, G.; Nowak, M.; Kawecki, M. Thermal imaging and planimetry evaluation of the results of chronic wounds treatment with hyperbaric oxygen therapy. Adv. Clin. Exp. Med. 2019, 28, 229–236. [Google Scholar] [CrossRef]
- Kasprzyk-Kucewicz, T.; Cholewka, A.; Englisz-Jurgielewicz, B.; Mucha, R.; Relich, M.; Kawecki, M.; Sieroń, K.; Onak, P.; Stanek, A. Thermal effects of topical hyperbaric oxygen therapy in hard-to-heal wounds—A pilot study. Int. J. Environ. Res. Public Health 2021, 18, 6737. [Google Scholar] [CrossRef]
- Stanek, A.; Gebala-Prajsnar, K.; Pasek, J.; Prajsnar, G.; Berszakiewicz, A.; Sieron, A.; Cholewka, A.; Zabrze, I. Selected physical medicine interventions in the treatment of diabetic foot syndrome. Acta Angiol. 2015, 21, 140–145. [Google Scholar] [CrossRef]
- Lin, P.-Y.; Sung, P.-H.; Chung, S.-Y.; Hsu, S.-L.; Chung, W.-J.; Sheu, J.-J.; Hsueh, S.-K.; Chen, K.-H.; Wu, R.-W.; Yip, H.-K. Clinical Medicine Hyperbaric Oxygen Therapy Enhanced Circulating Levels of Endothelial Progenitor Cells and Angiogenesis Biomarkers, Blood Flow, in Ischemic Areas in Patients with Peripheral Arterial Occlusive Disease. J. Clin. Med. 2018, 7, 548. [Google Scholar] [CrossRef]
- Carturan, D.; Boussuges, A.; Vanuxem, P.; Bar-Hen, A.; Burnet, H.; Gardette, B. Ascent rate, age, maximal oxygen uptake, adiposity, and circulating venous bubbles after diving. J. Appl. Physiol. 2002, 93, 1349–1356. [Google Scholar] [CrossRef][Green Version]
- Edwards, M.; Singh, M.; Selesny, S.; Cooper, J.S. Hyperbaric Treatment of Thermal Burns; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Cooper, J.S.; Hanley, M.E. Hyperbaric Treatment of Radiation Proctitis; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Hanley, M.E.; Manna, B. Hyperbaric Treatment of Diabetic Foot Ulcer; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Hyperbaric Oxygen Therapy: Get the Facts|FDA. Available online: https://www.fda.gov/consumers/consumer-updates/hyperbaric-oxygen-therapy-get-facts (accessed on 6 October 2021).
- Kamat, S.M.; Mendelsohn, A.R.; Larrick, J.W. Rejuvenation through Oxygen, More or Less. Rejuvenation Res. 2021, 24, 158–163. [Google Scholar] [CrossRef]
- Somaa, F. A Review of the Application of Hyperbaric Oxygen Therapy in Alzheimer’s Disease. J. Alzheimer’s Dis. 2021, 81, 1361–1367. [Google Scholar] [CrossRef] [PubMed]
- Thibodeaux, K.; Speyrer, M.; Raza, A.; Yaakov, R.; Serena, T.E. Hyperbaric oxygen therapy in preventing mechanical ventilation in COVID-19 patients: A retrospective case series. J. Wound Care 2020, 29, S4–S8. [Google Scholar] [CrossRef] [PubMed]
- Abu El Hawa, A.A.; Charipova, K.; Bekeny, J.C.; Johnson-Arbor, K.K. The evolving use of hyperbaric oxygen therapy during the COVID-19 pandemic. J. Wound Care 2021, 30, S8–S11. [Google Scholar] [CrossRef] [PubMed]
- Paganini, M.; Bosco, G.; Perozzo, F.A.G.; Kohlscheen, E.; Sonda, R.; Bassetto, F.; Garetto, G.; Camporesi, E.M.; Thom, S.R. The Role of Hyperbaric Oxygen Treatment for COVID-19: A Review. In Advances in Experimental Medicine and Biology; Springer: Cham, Switzerland, 2021; Volume 1289, pp. 27–35. [Google Scholar]
- De Maio, A.; Hightower, L.E. COVID-19, acute respiratory distress syndrome (ARDS), and hyperbaric oxygen therapy (HBOT): What is the link? Cell Stress Chaperones 2020, 25, 717–720. [Google Scholar] [CrossRef]
- Guo, D.; Pan, S.; Wang, M.M.; Guo, Y. Hyperbaric oxygen therapy may be effective to improve hypoxemia in patients with severe COVID-2019 pneumonia: Two case reports. Undersea Hyperb. Med. 2020, 47, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Boet, S.; Etherington, C.; Djaiani, G.; Tricco, A.C.; Sikora, L.; Katznelson, R. Efficacy and safety of hyperbaric oxygen treatment in SARS-CoV-2 (COVID-19) pneumonia: A systematic review. Diving Hyperb. Med. 2021, 51, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Marcinkowska, A.B.; Mankowska, N.D.; Kot, J.; Winklewski, P.J. Impact of Hyperbaric Oxygen Therapy on Cognitive Functions: A Systematic Review. Neuropsychol. Rev. 2021. [Google Scholar] [CrossRef]
- Kim, H.-S.; Choi, M.-H.; Baek, J.-H.; Park, S.-J.; Lee, J.-C.; Jeong, U.-H.; Kim, S.-P.; Kim, H.-J.; Choi, Y.; Lim, D.-W.; et al. Effects of 92% oxygen administration on cognitive performance and physiological changes of intellectually and developmentally disabled people. J. Physiol. Anthropol. 2015, 34, 3. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hadanny, A.; Rittblat, M.; Bitterman, M.; May-Raz, I.; Suzin, G.; Boussi-Gross, R.; Zemel, Y.; Bechor, Y.; Catalogna, M.; Efrati, S. Hyperbaric oxygen therapy improves neurocognitive functions of post-stroke patients-a retrospective analysis. Restor. Neurol. Neurosci. 2020, 38, 93–107. [Google Scholar] [CrossRef] [PubMed]
- Efrati, S.; Fishlev, G.; Bechor, Y.; Volkov, O.; Bergan, J.; Kliakhandler, K.; Kamiager, I.; Gal, N.; Friedman, M.; Ben-Jacob, E.; et al. Hyperbaric oxygen induces late neuroplasticity in post stroke patients—Randomized, prospective trial. PLoS ONE 2013, 8, e53716. [Google Scholar] [CrossRef] [PubMed]
- Cozene, B.; Sadanandan, N.; Gonzales-Portillo, B.; Saft, M.; Cho, J.; Park, Y.J.; Borlongan, C.V. An extra breath of fresh air: Hyperbaric oxygenation as a stroke therapeutic. Biomolecules 2020, 10, 1279. [Google Scholar] [CrossRef]
- Lee, Y.S.; Chio, C.C.; Chang, C.P.; Wang, L.C.; Chiang, P.M.; Niu, K.C.; Tsai, K.J. Long course hyperbaric oxygen stimulates neurogenesis and attenuates inflammation after ischemic stroke. Mediat. Inflamm. 2013, 2013, 512978. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Li, F.; Gu, D. Hyperbaric oxygen therapy for cerebral blood flow and electroencephalogram in patients with acute cerebral infarction: Choice for therapeutic occasion. Neural Regen. Res. 2007, 2, 171–174. [Google Scholar] [CrossRef]
- Rosario, E.R.; Kaplan, S.E.; Khonsari, S.; Vazquez, G.; Solanki, N.; Lane, M.; Brownell, H.; Rosenberg, S.S. The Effect of Hyperbaric Oxygen Therapy on Functional Impairments Caused by Ischemic Stroke. Neurol. Res. Int. 2018, 2018, 3172679. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Hong, G.; Ha, E.; Hong, H.; Kim, J.; Joo, Y.; Yoon, S.; Lyoo, I.K.; Kim, J. Hippocampal cerebral blood flow increased following low-pressure hyperbaric oxygenation in firefighters with mild traumatic brain injury and emotional distress. Neurol. Sci. 2021, 42, 4131–4138. [Google Scholar] [CrossRef] [PubMed]
- Skiba, M.; Rękas-Dudziak, A.; Bekała, A.; Płotek, W. Late application of hyperbaric oxygen therapy during the rehabilitation of a patient with severe cognitive impairment after a traumatic brain injury. Clin. Case Rep. 2021, 9, 960–965. [Google Scholar] [CrossRef] [PubMed]
- Tal, S.; Hadanny, A.; Berkovitz, N.; Sasson, E.; Ben-Jacob, E.; Efrati, S. Hyperbaric oxygen may induce angiogenesis in patients suffering from prolonged post-concussion syndrome due to traumatic brain injury. Restor. Neurol. Neurosci. 2015, 33, 943–951. [Google Scholar] [CrossRef] [PubMed]
- Boussi-Gross, R.; Golan, H.; Fishlev, G.; Bechor, Y.; Volkov, O.; Bergan, J.; Friedman, M.; Hoofien, D.; Shlamkovitch, N.; Ben-Jacob, E.; et al. Hyperbaric oxygen therapy can improve post concussion syndrome years after mild traumatic brain injury—Randomized prospective trial. PLoS ONE 2013, 8, e79995. [Google Scholar] [CrossRef]
- Harch, P.; Andrews, S.; Rowe, C.; Lischka, J.; Townsend, M.; Yu, Q.; Mercante, D. Hyperbaric oxygen therapy for mild traumatic brain injury persistent postconcussion syndrome: A randomized controlled trial. Med. Gas Res. 2020, 10, 8–20. [Google Scholar] [CrossRef] [PubMed]
- Tal, S.; Hadanny, A.; Sasson, E.; Suzin, G.; Efrati, S. Hyperbaric Oxygen Therapy Can Induce Angiogenesis and Regeneration of Nerve Fibers in Traumatic Brain Injury Patients. Front. Hum. Neurosci. 2017, 11, 508. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zhang, F.; Zhao, L.; Cheng, C.; Zhong, R.; Dong, C.; Le, W. Hyperbaric oxygen ameliorates cognitive impairment in patients with Alzheimer’s disease and amnestic mild cognitive impairment. Alzheimer’s Dement. 2020, 6, e12030. [Google Scholar] [CrossRef]
- Harch, P.G.; Fogarty, E.F. Hyperbaric oxygen therapy for Alzheimer’s dementia with positron emission tomography imaging: A case report. Med. Gas Res. 2018, 8, 181–184. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, Q.; Qu, Z.; Yang, J.; Zhang, X.; Zhao, Y. Protective Effect of Hyperbaric Oxygen Therapy on Cognitive Function in Patients with Vascular Dementia. Cell Transpl. 2019, 28, 1071–1075. [Google Scholar] [CrossRef]
- You, Q.; Li, L.; Xiong, S.-Q.; Yan, Y.-F.; Li, D.; Yan, N.-N.; Chen, H.-P.; Liu, Y.-P. Meta-Analysis on the Efficacy and Safety of Hyperbaric Oxygen as Adjunctive Therapy for Vascular Dementia. Front. Aging Neurosci. 2019, 11, 86. [Google Scholar] [CrossRef]
- Vila, J.F.; Balcarce, P.E.; Abiusi, G.R.; Domínguez, R.O.; Pisarello, J.B. Improvement in motor and cognitive impairment after hyperbaric oxygen therapy in a selected group of patients with cerebrovascular disease: A prospective single-blind controlled trial. Undersea Hyperb. Med. 2005, 32, 341–349. [Google Scholar]
- Denis, P.A. Alzheimer’s disease: A gas model. The NADPH oxidase-Nitric Oxide system as an antibubble biomachinery. Med. Hypotheses 2013, 81, 976–987. [Google Scholar] [CrossRef]
- Zárate, S.C.; Traetta, M.E.; Codagnone, M.G.; Seilicovich, A.; Reinés, A.G. Humanin, a mitochondrial-derived peptide released by astrocytes, prevents synapse loss in hippocampal neurons. Front. Aging Neurosci. 2019, 11, 123. [Google Scholar] [CrossRef] [PubMed]
- Hazafa, A.; Batool, A.; Ahmad, S.; Amjad, M.; Chaudhry, S.N.; Asad, J.; Ghuman, H.F.; Khan, H.M.; Naeem, M.; Ghani, U. Humanin: A mitochondrial-derived peptide in the treatment of apoptosis-related diseases. Life Sci. 2021, 264, 118679. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Ito, Y.; Niikura, T.; Shao, Z.; Hata, M.; Oyama, F.; Nishimoto, I. Mechanisms of neuroprotection by a novel rescue factor humanin from swedish mutant amyloid precursor protein. Biochem. Biophys. Res. Commun. 2001, 283, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, E.A.; Winter, P.M.; Alvis, H.J.; Small, S.M. Hyperoxygenation Effect on Cognitive Functioning in the Aged. N. Engl. J. Med. 1969, 281, 753–757. [Google Scholar] [CrossRef]
- Yu, R.; Wang, B.; Li, S.; Wang, J.; Zhou, F.; Chu, S.; He, X.; Wen, X.; Ni, X.; Liu, L.; et al. Cognitive enhancement of healthy young adults with hyperbaric oxygen: A preliminary resting-state fMRI study. Clin. Neurophysiol. 2015, 126, 2058–2067. [Google Scholar] [CrossRef] [PubMed]
- Vadas, D.; Kalichman, L.; Hadanny, A.; Efrati, S. Hyperbaric Oxygen Environment Can Enhance Brain Activity and Multitasking Performance. Front. Integr. Neurosci. 2017, 11, 25. [Google Scholar] [CrossRef]
- Suzin, G.; Frolinger, T.H.; Yogev, D.; Hadanny, A.; Catalogna, M.; Rassovsky, Y.; Efrati, S. Oxygen: The rate-limiting factor for episodic memory performance, even in healthy young individuals. Biomolecules 2020, 10, 1328. [Google Scholar] [CrossRef]
- Hadanny, A.; Malka, D.K.; Gil, S.; Rahav, B.G.; Merav, C.; Kobi, D.; Yafit, H.; Ramzia, A.H.; Efrat, S.; Gregory, F.; et al. Cognitive enhancement of healthy older adults using hyperbaric oxygen: A randomized controlled trial. Aging 2020, 12, 13740–13761. [Google Scholar] [CrossRef]
- Shapira, R.; Gdalyahu, A.; Gottfried, I.; Sasson, E.; Hadanny, A.; Efrati, S.; Blinder, P.; Ashery, U. Hyperbaric oxygen therapy alleviates vascular dysfunction and amyloid burden in an Alzheimer’s disease mouse model and in elderly patients. Aging 2021, 13, 20935–20961. [Google Scholar] [CrossRef]
- Raskin, A.; Gershon, S.; Crook, T.H.; Sathananthan, G.; Ferris, S. The effects of hyperbaric and normobaric oxygen on cognitive impairment in the elderly. Arch. Gen. Psychiatry 1978, 35, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Hadanny, A.; Efrati, S. The hyperoxic-hypoxic paradox. Biomolecules 2020, 10, 958. [Google Scholar] [CrossRef] [PubMed]
- Fratantonio, D.; Virgili, F.; Zucchi, A.; Lambrechts, K.; Latronico, T.; Lafère, P.; Germonpré, P.; Balestra, C. Increasing oxygen partial pressures induce a distinct transcriptional response in human pbmc: A pilot study on the “normobaric oxygen paradox”. Int. J. Mol. Sci. 2021, 22, 458. [Google Scholar] [CrossRef] [PubMed]
- Balestra, C.; Lambrechts, K.; Mrakic-Sposta, S.; Vezzoli, A.; Levenez, M.; Germonpré, P.; Virgili, F.; Bosco, G.; Lafère, P. Hypoxic and Hyperoxic Breathing as a Complement to Low-Intensity Physical Exercise Programs: A Proof-of-Principle Study. Int. J. Mol. Sci. 2021, 22, 9600. [Google Scholar] [CrossRef] [PubMed]
- Van Vliet, T.; Casciaro, F.; Demaria, M. To breathe or not to breathe: Understanding how oxygen sensing contributes to age-related phenotypes. Ageing Res. Rev. 2021, 67, 101267. [Google Scholar] [CrossRef] [PubMed]
- Greer, S.N.; Metcalf, J.L.; Wang, Y.; Ohh, M. The updated biology of hypoxia-inducible factor. EMBO J. 2012, 31, 2448–2460. [Google Scholar] [CrossRef] [PubMed]
- Fratantonio, D.; Cimino, F.; Speciale, A.; Virgili, F. Need (more than) two to Tango: Multiple tools to adapt to changes in oxygen availability. BioFactors 2018, 44, 207–218. [Google Scholar] [CrossRef]
- Cimino, F.; Speciale, A.; Anwar, S.; Canali, R.; Ricciardi, E.; Virgili, F.; Trombetta, D.; Saija, A. Anthocyanins protect human endothelial cells from mild hyperoxia damage through modulation of Nrf2 pathway. Genes Nutr. 2013, 8, 391–399. [Google Scholar] [CrossRef]
- Tonelli, C.; Chio, I.I.C.; Tuveson, D.A. Transcriptional Regulation by Nrf2. Antioxid. Redox Signal. 2018, 29, 1727–1745. [Google Scholar] [CrossRef]
- Mattson, M.P.; Meffert, M.K. Roles for NF-κB in nerve cell survival, plasticity, and disease. Cell Death Differ. 2006, 13, 852–860. [Google Scholar] [CrossRef] [PubMed]
- Lippert, T.; Borlongan, C.V. Prophylactic treatment of hyperbaric oxygen treatment mitigates inflammatory response via mitochondria transfer. CNS Neurosci. Ther. 2019, 25, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Daugherty, W.P.; Sun, D.; Levasseur, J.E.; Altememi, N.; Hamm, R.J.; Rockswold, G.L.; Bullock, M.R. Protection of mitochondrial function and improvement in cognitive recovery in rats treated with hyperbaric oxygen following lateral fluid-percussion injury. J. Neurosurg. 2007, 106, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Zhang, L.; Wang, J.; Dai, J.; Shen, S.; Yang, L.; Huang, P. The protective effect of hyperbaric oxygen and Ginkgo biloba extract on Aβ25–35-induced oxidative stress and neuronal apoptosis in rats. Behav. Brain Res. 2013, 242, 1–8. [Google Scholar] [CrossRef]
- Thom, S.R.; Milovanova, T.N.; Yang, M.; Bhopale, V.M.; Sorokina, E.M.; Uzun, G.; Malay, D.S.; Troiano, M.A.; Hardy, K.R.; Lambert, D.S.; et al. Vasculogenic stem cell mobilization and wound recruitment in diabetic patients: Increased cell number and intracellular regulatory protein content associated with hyperbaric oxygen therapy. Wound Repair Regen. 2011, 19, 149–161. [Google Scholar] [CrossRef]
- Goldstein, L.J.; Gallagher, K.A.; Bauer, S.M.; Bauer, R.J.; Baireddy, V.; Liu, Z.-J.; Buerk, D.G.; Thom, S.R.; Velazquez, O.C. Endothelial Progenitor Cell Release into Circulation Is Triggered by Hyperoxia-Induced Increases in Bone Marrow Nitric Oxide. Stem Cells 2006, 24, 2309–2318. [Google Scholar] [CrossRef]
- Milovanova, T.N.; Bhopale, V.M.; Sorokina, E.M.; Moore, J.S.; Hunt, T.K.; Hauer-Jensen, M.; Velazquez, O.C.; Thom, S.R. Hyperbaric oxygen stimulates vasculogenic stem cell growth and differentiation in vivo. J. Appl. Physiol. 2009, 106, 711–728. [Google Scholar] [CrossRef]
- Yang, Y.J.; Wang, X.L.; Yu, X.H.; Wang, X.; Xie, M.; Liu, C.T. Hyperbaric oxygen induces endogenous neural stem cells to proliferate and differentiate in hypoxic-ischemic brain damage in neonatal rats. Undersea Hyperb. Med. 2008, 35, 113–129. [Google Scholar]
- Liu, S.; Shen, G.; Deng, S.; Wang, X.; Wu, Q.; Guo, A. Hyperbaric oxygen therapy improves cognitive functioning after brain injury. Neural Regen. Res. 2013, 8, 3334–3343. [Google Scholar] [CrossRef]
- Wei, L.; Wang, J.; Cao, Y.; Ren, Q.; Zhao, L.; Li, X.; Wang, J. Hyperbaric oxygenation promotes neural stem cell proliferation and protects the learning and memory ability in neonatal hypoxic-ischemic brain damage. Int. J. Clin. Exp. Pathol. 2015, 8, 1752–1759. [Google Scholar]
- Wang, X.L.; Yang, Y.J.; Xie, M.; Yu, X.H.; Liu, C.T.; Wang, X. Proliferation of neural stem cells correlates with Wnt-3 protein in hypoxic-ischemic neonate rats after hyperbaric oxygen therapy. Neuroreport 2007, 18, 1753–1756. [Google Scholar] [CrossRef]
- Yang, Y.; Wei, H.; Zhou, X.; Zhang, F.; Wang, C. Hyperbaric oxygen promotes neural stem cell proliferation by activating vascular endothelial growth factor/extracellular signal-regulated kinase signaling after traumatic brain injury. Neuroreport 2017, 28, 1232–1238. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Yang, Q.-W.W.; Wang, S.-N.N.; Wang, J.-Z.Z.; Wang, Q.; Wang, Y.; Luo, Y.-J.J. Hyperbaric oxygen therapy improves neurogenesis and brain blood supply in piriform cortex in rats with vascular dementia. Brain Inj. 2010, 24, 1350–1357. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Sun, Q.; Xin, Q.; Qin, J.; Zhang, L.; Wu, D.; Gao, G.; Xia, Y. Hyperbaric oxygen therapy mobilized circulating stem cells and improved delayed encephalopathy after acute carbon monoxide poisoning with up-regulation of brain-derived neurotrophic factor. Am. J. Emerg. Med. 2021, 42, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.-C.C.; Niu, K.-C.C.; Tsai, K.-J.J.; Kuo, J.-R.R.; Wang, L.-C.C.; Chio, C.-C.C.; Chang, C.-P.P. Attenuating inflammation but stimulating both angiogenesis and neurogenesis using hyperbaric oxygen in rats with traumatic brain injury. J. Trauma Acute Care Surg. 2012, 72, 650–659. [Google Scholar] [CrossRef] [PubMed]
- Buckley, C.J.; Cooper, J.S. Hyperbaric Affects on Angiogenesis; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Shapira, R.; Solomon, B.; Efrati, S.; Frenkel, D.; Ashery, U. Hyperbaric oxygen therapy ameliorates pathophysiology of 3xTg-AD mouse model by attenuating neuroinflammation. Neurobiol. Aging 2018, 62, 105–119. [Google Scholar] [CrossRef]
- Shapira, R.; Efrati, S.; Ashery, U. Hyperbaric oxygen therapy as a new treatment approach for Alzheimer’s disease. Neural Regen. Res. 2018, 13. [Google Scholar] [CrossRef]
- Halbach, J.L.; Prieto, J.M.; Wang, A.W.; Hawisher, D.; Cauvi, D.M.; Reyes, T.; Okerblom, J.; Ramirez-Sanchez, I.; Villarreal, F.; Patel, H.H.; et al. Early hyperbaric oxygen therapy improves survival in a model of severe sepsis. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2019, 317, R160–R168. [Google Scholar] [CrossRef]
- Lin, Y.; Lin, X.; Zheng, X.; Liu, F.; Ye, C.; Huang, L.; Zhou, Q.; Chen, T.; Lin, L. Hyperbaric oxygen therapy cognitive function in a rat model of mild cognitive impairment via ERK signaling. Ann. Cardiothorac. Surg. 2020, 9, 3472–3480. [Google Scholar] [CrossRef]
- Zhao, B.; Pan, Y.; Wang, Z.; Xu, H.; Song, X. Hyperbaric oxygen pretreatment improves cognition and reduces hippocampal damage via p38 mitogen-activated protein kinase in a rat model. Yonsei Med. J. 2017, 58, 131–138. [Google Scholar] [CrossRef]
- Corrêa, S.A.L.; Eales, K.L. The Role of p38 MAPK and Its Substrates in Neuronal Plasticity and Neurodegenerative Disease. J. Signal Transduct. 2012, 2012, 1–12. [Google Scholar] [CrossRef]
- Sun, A.; Liu, M.; Nguyen, X.V.; Bing, G. p38 MAP kinase is activated at early stages in Alzheimer’s disease brain. Exp. Neurol. 2003, 183, 394–405. [Google Scholar] [CrossRef]
- Gamdzyk, M.; Małek, M.; Bratek, E.; Koks, A.; Kaminski, K.; Ziembowicz, A.; Salinska, E. Hyperbaric oxygen and hyperbaric air preconditioning induces ischemic tolerance to transient forebrain ischemia in the gerbil. Brain Res. 2016, 1648, 257–265. [Google Scholar] [CrossRef]
- Yan, W.; Fang, Z.; Yang, Q.; Dong, H.; Lu, Y.; Lei, C.; Xiong, L. SirT1 mediates hyperbaric oxygen preconditioning-induced ischemic tolerance in rat brain. J. Cereb. Blood Flow Metab. 2013, 33, 396–406. [Google Scholar] [CrossRef] [PubMed]
- Xue, F.; Huang, J.W.; Ding, P.Y.; Zang, H.G.; Kou, Z.J.; Li, T.; Fan, J.; Peng, Z.W.; Yan, W.J. Nrf2/antioxidant defense pathway is involved in the neuroprotective effects of Sirt1 against focal cerebral ischemia in rats after hyperbaric oxygen preconditioning. Behav. Brain Res. 2016, 309, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hong-qiang, H.; Mang-qiao, S.; Fen, X.; Shan-shan, L.; Hui-juan, C.; Wu-gang, H.; Wen-jun, Y.; Zheng-wu, P. Sirt1 mediates improvement of isoflurane-induced memory impairment following hyperbaric oxygen preconditioning in middle-aged mice. Physiol. Behav. 2018, 195, 1–8. [Google Scholar] [CrossRef]
- Hu, Q.; Manaenko, A.; Bian, H.; Guo, Z.; Huang, J.L.; Guo, Z.N.; Yang, P.; Tang, J.; Zhang, J.H. Hyperbaric Oxygen Reduces Infarction Volume and Hemorrhagic Transformation Through ATP/NAD+/Sirt1 Pathway in Hyperglycemic Middle Cerebral Artery Occlusion Rats. Stroke 2017, 48, 1655–1664. [Google Scholar] [CrossRef]
- Tian, X.; Wang, J.; Dai, J.; Yang, L.; Zhang, L.; Shen, S.; Huang, P. Hyperbaric Oxygen and Ginkgo Biloba Extract Inhibit Aβ25-35-induced Toxicity and Oxidative Stress in vivo: A Potential Role in Alzheimer’s Disease. Int. J. Neurosci. 2012, 122, 563–569. [Google Scholar] [CrossRef]
- Baratz-Goldstein, R.; Toussia-Cohen, S.; Elpaz, A.; Rubovitch, V.; Pick, C.G. Immediate and delayed hyperbaric oxygen therapy as a neuroprotective treatment for traumatic brain injury in mice. Mol. Cell. Neurosci. 2017, 83, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.D.; Ma, L.; Zhang, L.; Dai, J.G.; Chang, L.G.; Huang, P.L.; Tian, X.Q. Hyperbaric oxygen and ginkgo biloba extract ameliorate cognitive and memory impairment via nuclear factor Kappa-B pathway in rat model of alzheimer’s disease. Chin. Med. J. 2015, 128, 3088–3093. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gottfried, I.; Schottlender, N.; Ashery, U. Hyperbaric Oxygen Treatment—From Mechanisms to Cognitive Improvement. Biomolecules 2021, 11, 1520. https://doi.org/10.3390/biom11101520
Gottfried I, Schottlender N, Ashery U. Hyperbaric Oxygen Treatment—From Mechanisms to Cognitive Improvement. Biomolecules. 2021; 11(10):1520. https://doi.org/10.3390/biom11101520
Chicago/Turabian StyleGottfried, Irit, Nofar Schottlender, and Uri Ashery. 2021. "Hyperbaric Oxygen Treatment—From Mechanisms to Cognitive Improvement" Biomolecules 11, no. 10: 1520. https://doi.org/10.3390/biom11101520
APA StyleGottfried, I., Schottlender, N., & Ashery, U. (2021). Hyperbaric Oxygen Treatment—From Mechanisms to Cognitive Improvement. Biomolecules, 11(10), 1520. https://doi.org/10.3390/biom11101520





