A New Look at the Purported Health Benefits of Commercial and Natural Clays
Abstract
1. Introduction
2. Materials and Methods
2.1. Leachate Preparation
2.2. Inductively Coupled Plasma-Mass Spectroscopy
2.3. X-ray Diffraction
2.4. Electron Microscopy
2.5. Filamentous Actin Measurement
2.6. Confocal Laser Scanning Microscopy
2.7. Keratinocyte Proliferation and Viability
2.8. Keratinocyte Migration and Wound Repopulation
2.9. Minimum Inhibitory Concentration
2.10. Statistical Analysis
3. Results
3.1. Geochemical Characterisation
3.2. F-Actin Measurement
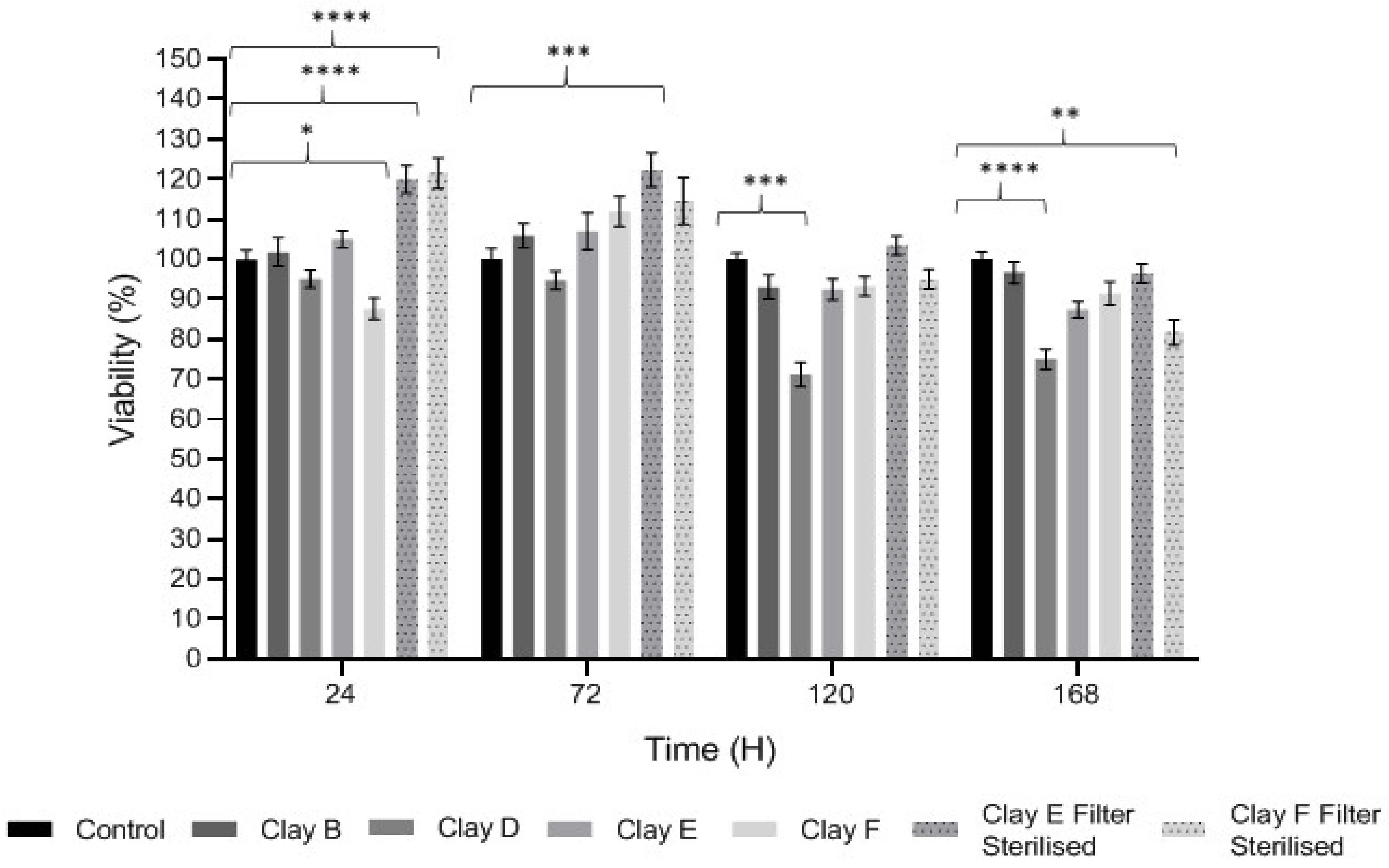
3.3. Keratinocyte Proliferation and Viability
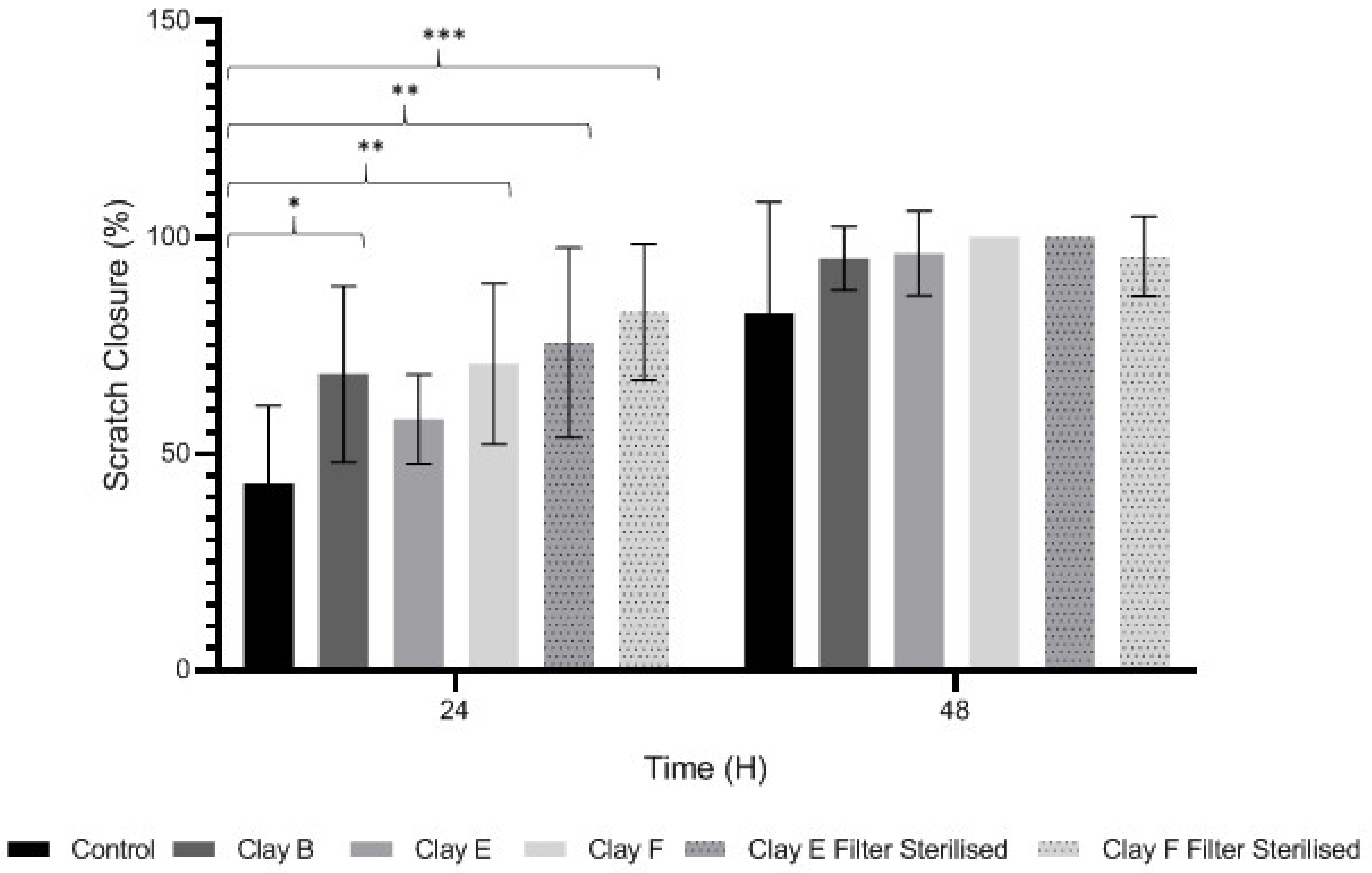
3.4. Keratinocyte Migration and Wound Repopulation
3.5. Antibacterial Efficacy
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bartlett, J.G.; Gilbert, D.N.; Spellberg, B. Seven ways to preserve the miracle of antibiotics. Clin. Infect. Dis. 2013, 56, 1445–1450. [Google Scholar] [CrossRef] [PubMed]
- CDC Antibiotic-Resistant Germs: New Threats. Available online: http://www.cdc.gov/drugresistance/threat-report-2013/index.html (accessed on 17 May 2018).
- Gross, M. Antibiotics in crisis. Curr. Biol. 2013, 23, R1063–R1065. [Google Scholar] [CrossRef] [PubMed]
- Michael, C.A.; Dominey-Howes, D.; Labbate, M. The antimicrobial resistance crisis: Causes, consequences, and management. Front. Public Health 2014, 2. [Google Scholar] [CrossRef] [PubMed]
- Spellberg, B.; Gilbert, D.N. The future of antibiotics and resistance: A tribute to a career of leadership by John Bartlett. Clin. Infect. Dis. 2014, 59, S71–S75. [Google Scholar] [CrossRef] [PubMed]
- Kirsner, R.S. The wound healing society chronic wound ulcer healing guidelines update of the 2006 guidelines-blending old with new. Wound Repair Regen. 2016, 24, 110–111. [Google Scholar] [CrossRef] [PubMed]
- Eming, S.A.; Krieg, T.; Davidson, J.M. Inflammation in wound repair: Molecular and cellular mechanisms. J. Investig. Dermatol. 2007, 127, 514–525. [Google Scholar] [CrossRef]
- Menke, N.B.; Ward, K.R.; Witten, T.M.; Bonchev, D.G.; Diegelmann, R.F. Impaired wound healing. Clin. Dermatol. 2007, 25, 19–25. [Google Scholar] [CrossRef]
- Pastar, I.; Stojadinovic, O.; Yin, N.C.; Ramirez, H.; Nusbaum, A.G.; Sawaya, A.; Patel, S.B.; Khalid, L.; Isseroff, R.R.; Tomic-Canic, M. Epithelialization in wound healing: A comprehensive review. Adv. Wound Care 2014, 3, 445–464. [Google Scholar] [CrossRef]
- Rahim, K.; Saleha, S.; Zhu, X.; Huo, L.; Basit, A.; Franco, O.L. Bacterial contribution in chronicity of wounds. Microb. Ecol. 2016, 73, 710–721. [Google Scholar] [CrossRef]
- Larouche, J.; Sheoran, S.; Maruyama, K.; Martino, M.M. Immune regulation of skin wound healing: Mechanisms and novel therapeutic targets. Adv. Wound Care 2018, 7, 209–231. [Google Scholar] [CrossRef]
- MacDonald, J. Global initiative for wound and lymphoedema care (GIWLC). Wounds UK 2009, 5, 172–175. [Google Scholar]
- Guest, J.F.; Gerrish, A.; Ayoub, N.; Vowden, K.; Vowden, P. Clinical outcomes and cost-effectiveness of three alternative compression systems used in the management of venous leg ulcers. J. Wound Care 2015, 24, 300–303. [Google Scholar] [CrossRef] [PubMed]
- Piddock, L.J. The crisis of no new antibiotics—What is the way forward? Lancet Infect. Dis. 2012, 12, 249–253. [Google Scholar] [CrossRef]
- O’Neill, J. Antimicrobial Resistance: Tackling a Crisis for the Future Health and Wealth of Nations; AMR Review: London, UK, 2014. [Google Scholar]
- Williams, L.B.; Holland, M.; Eberl, D.; De Courssou, L.B. Killer clays! Natural antibacterial clay minerals. Mineral. Soc. Bull. 2004, 139, 3–8. [Google Scholar]
- De Courssou, L.B. Study Group Report on Buruli Ulcer Treatment with Clay; WHO: Geneva, Switzerland, 2002. [Google Scholar]
- Williams, L.B.; Haydel, S.E.; Giese, R.F., Jr.; Eberl, D.D. Chemical and mineralogical characteristics of French Green clays used for healing. Clays Clay Miner. 2008, 56, 437–452. [Google Scholar] [CrossRef] [PubMed]
- Williams, L.B.; Metge, D.W.; Eberl, D.D.; Harvey, R.W.; Turner, A.G.; Prapaipong, P.; Poret-Peterson, A.T. What makes a natural clay antibacterial? Environ. Sci. Technol. 2011, 45, 3768–3773. [Google Scholar] [CrossRef] [PubMed]
- Otto, C.C.; Haydel, S.E. Exchangeable ions are responsible for the in vitro antibacterial properties of natural clay mixtures. PLoS ONE 2013, 8, e64068. [Google Scholar] [CrossRef]
- Morrison, K.D.; Misra, R.; Williams, L.B. Unearthing the antibacterial mechanism of medicinal clay: A geochemical approach to combating antibiotic resistance. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Londono, S.C.; Hartnett, H.E.; Williams, L.B. Antibacterial activity of aluminum in clay from the Colombian Amazon. Environ. Sci. Technol. 2017, 51, 2401–2408. [Google Scholar] [CrossRef]
- Kong, B.; Seog, J.H.; Graham, L.M.; Lee, S.B. Experimental considerations on the cytotoxicity of nanoparticles. Nanomedicine (Lond) 2011, 6, 929–941. [Google Scholar] [CrossRef]
- Holder, A.L.; Goth-Goldstein, R.; Lucas, D.; Koshland, C.P. Particle-induced artifacts in the MTT and LDH viability assays. Chem. Res. Toxicol. 2012, 25, 1885–1892. [Google Scholar] [CrossRef]
- Kroll, A.; Pillukat, M.H.; Hahn, D.; Schnekenburger, J. Interference of engineered nanoparticles with in vitro toxicity assays. Arch. Toxicol. 2012, 86, 1123–1136. [Google Scholar] [CrossRef]
- Aslantürk, O.S. In vitro cytotoxicity and cell viability assays: Principles, advantages, and disadvantages. In Genotoxicity—A Predictable Risk to Our Actual World, 1st ed.; Larramendy, M.L., Soloneski, S., Eds.; IntechOpen: London, UK, 2017. [Google Scholar] [CrossRef]
- Scarcello, E.; Lambremont, A.; Vanbever, R.; Jacques, P.J.; Lison, D. Mind your assays: Misleading cytotoxicty with the WST-1 assay in the presence of manganese. PLoS ONE 2020. [Google Scholar] [CrossRef]
- Vrček, I.V.; Pavičić, I.; Crnković, T.; Jurašin, D.; Babič, M.; Horák, D.; Lovrić, M.; Ferhatović, L.; Ćurlin, M.; Gajović, S. Does surface coating of metallic nanoparticles modulate their interference with in vitro assays? Rsc. Adv. 2015, 5, 70787–70807. [Google Scholar] [CrossRef]
- Zarate-Reyes, L.; Lopez-Pacheco, C.; Nieto-Camacho, A.; Palacios, E.; Gómez-Vidales, V.; Kaufhold, S.; Ufer, K.; Zepeda, E.G.; Cervini-Silva, J. Antibacterial clay against gram-negative antibiotic resistant bacteria. J. Hazard. Mater. 2018, 342, 625–632. [Google Scholar] [CrossRef]
- Londono, S.C.; Williams, L.B. Unraveling the antibacterial mode of action of a clay from the Colombian Amazon. Environ. Geochem. Health 2015, 38, 363–379. [Google Scholar] [CrossRef]
- Dextreit, R. Une Merveille, L’argile Qui Guérit: Memento de Medecine Naturelle; Editions de la Revue; Vivre En Harmonie: Paris, France, 1989; ISBN 9782715500136. [Google Scholar]
- Pough, F.H. A Field Guide to Rocks and Minerals; Houghton Mifflin Co.: Boston, MA, USA, 1988; ISBN 9780395910979. [Google Scholar]
- BSI (British Standards Institution). Characterisation of Waste. Leaching. Compliance Test for Leaching of Granular Waste Materials and Sludges. One Stage Batch Test at a Liquid to Solid Ratio of 10 l/kg for Materials with Particle Size below 4 mm (Without or with Size Reduction); BSI: London, UK, 2002.
- AIST (National Institute of Industrial Science and Technology). Geological Survey of Japan; AIST: Tsukuba, Japan, 2018.
- Biscaye, P.E. Distinction between chlorite and kaolinite in recent sediments by X-ray diffraction. Am. Mineral. 1964, 49, 1281–1289. [Google Scholar]
- Cook, H.E.; Johnson, P.D.; Matti, J.C.; Zemells, I. Methods of sample preparation and X-ray diffraction data analysis, X-ray mineralogy laboratory. In Initial Reports of the Deep Sea Drilling Project; Hayes, D.E., Frakes, L.A., Barrett, P.J., Burns, D.A., Chen, P., Ford, A.B., Kaneps, A.G., Kemp, E.M., McCollum, D.W., Piper, D.J.W., et al., Eds.; U.S Government Printing Office: Washington, DC, USA, 1975; Volume 28, pp. 999–1007. [Google Scholar] [CrossRef]
- Johns, W.D.; Grim, R.E.; Bradley, W.F. Quantitative estimations of clay minerals by diffraction methods. J. Sediment. Pet. 1954, 24, 242–251. [Google Scholar]
- Sampath, V. Bacterial endotoxin-lipopolysaccharide; structure, function and its role in immunity in vertebrates and invertebrates. Agric. Nat. Resour. 2018, 52, 115–120. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Moses, R.L.; Boyle, G.M.; Howard-Jones, R.A.; Errington, R.J.; Johns, J.P.; Gordon, V.; Reddell, P.; Steadman, R.; Moseley, R. Novel epoxy-tiglianes stimulate skin keratinocyte wound healing responses and re-epithelialization via protein kinase C activation. Biochem. Pharmacol. 2020, 178, 114048. [Google Scholar] [CrossRef]
- Jorgensen, J.H.; Turnidge, J.D.; Washington, J.A. Antibacterial susceptibility test: Dilution and disk diffusion methods. In Manual of Clinical Microbiology, 7th ed.; Muray, P.R., Baron, E.J., Pfaller, M.A., Tenover, F.C., Yolken, R.H., Eds.; ASM Press: Washington, DC, USA, 1999; pp. 1526–1543. [Google Scholar]
- CLSI. Methods for Antimicrobial Susceptibility Testing of Anaerobic Bacteria, 9th ed.; CLSI Standard M11; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012. [Google Scholar]
- Munsell, A.H. A pigment color system and notation. Am. J. Psychol. 1912, 23, 236. [Google Scholar] [CrossRef]
- Adams, R.A.; Al-Mosawi, A.; Bérubé, K.; Jones, T.; Higgins, T.; Evans, S.-A. Increased monocyte actin polymerization in rat blood after intratracheal instillation of air pollution particles. Biorheology 2014, 51, 329–338. [Google Scholar] [CrossRef]
- Lambers, H.; Piessens, S.; Bloem, A.; Pronk, H.; Finkel, P. Natural skin surface pH is on average below 5, which is beneficial for its resident flora. Int. J. Cosmet. Sci. 2006, 28, 359–370. [Google Scholar] [CrossRef]
- Tarun, J.; Susan, V.; Susan, J.; Suria, J.; Criton, S. Evaluation of pH of bathing soaps and shampoos for skin and hair care. Indial J. Dermatol. 2014, 59, 442–444. [Google Scholar] [CrossRef]
- Prakash, C.; Bhargava, P.; Tiwari, S.; Majumdar, B.; Bhargava, R. Skin surface pH in acne vulgaris: Insights from an observational study and review of the literature. J. Aesthetic Clin. Dermatol. 2017, 3310, 33–39. [Google Scholar]
- Gethin, G. The significance of surface pH in chronic wounds. Wounds UK 2007, 3, 52–56. [Google Scholar]
- Bergey, D.; Boone, D.; Garrity, G. Bergey’s Manual of Systemic Bacteriology, 2nd ed.; Springer: New York, NY, USA, 2005. [Google Scholar]
- Hrenovic, J.; Durn, G.; Goic-Barisic, I.; Kovacic, A. Occurrence of an environmental acinetobacter baumannii strain similar to a clinical isolate in paleosol from Croatia. Appl. Environ. Microbiol. 2014, 80, 2860–2866. [Google Scholar] [CrossRef]
- Tsuji, A.; Kaneko, Y.; Takahashi, K.; Ogawa, M.; Goto, S. The effects of temperature and ph on the growth of eight enteric and nine glucose non-fermenting species of Gram-Negative rods. Microbiol. Immunol. 1982, 26, 15–24. [Google Scholar] [CrossRef]
- Klein, S.; Lorenzo, C.; Hoffmann, S.; Walther, J.M.; Storbeck, S.; Piekarski, T.; Tindall, B.J.; Wray, V.; Nimtz, M.; Moser, J. Adaptation of Pseudomonas aeruginosato various conditions includes tRNA-dependent formation of alanyl-phosphatidylglycerol. Mol. Microbiol. 2009, 71, 551–565. [Google Scholar] [CrossRef]
- Medvedova, A.; Valik, L. Staphylococcus aureus: Characterisation and quantitative growth description in milk and artisanal raw milk cheese production. In Structure and Function of Food Engineering, 1st ed.; Eissa, A.A., Ed.; IntechOpen: London, UK, 2012; pp. 71–102. [Google Scholar]
- Albrecht, C.; Becker, A.; Schins, R.P.F.; Höhr, D.; Unfried, K.; Knaapen, A.D.M.; Borm, P.J.A. Importance of surface characteristics of quartz DQ12 for acute inflammation. Ann. Occup. Hyg. 2002, 46, 22–26. [Google Scholar] [CrossRef]
- Brown, D.M.; Danielsen, P.H.; Derr, R.; Moelijker, N.; Fowler, P.; Stone, V.; Hendriks, G.; Møller, P.; Kermanizadeh, A. The mechanism-based toxicity screening of particles with use in the food and nutrition sector via the ToxTracker reporter system. Toxicol. Vitr. 2019, 61, 104594. [Google Scholar] [CrossRef]
- Price, E.W. The association of endemic elephantiasis of the lower legs in East Africa with soil derived from volcanic rocks. Trans. R. Soc. Trop. Med. Hyg. 1976, 70, 288–295. [Google Scholar] [CrossRef]
- Li, Y.; Boraschi, D. Endotoxin contamination: A key element in the interpretation of nanosafety studies. Nanomedicine (Lond) 2016, 11, 269–287. [Google Scholar] [CrossRef]
- Luo, C.; Li, Y.; Yang, L.; Wang, X.; Long, J.; Liu, J. Superparamagnetic iron oxide nanoparticles exacerbate the risks of reactive oxygen species-mediated external stresses. Arch. Toxicol. 2014, 89, 357–369. [Google Scholar] [CrossRef]
- Soderberg, T.A. Trace elements in normal and impaired wound healing. In Wound Healing and Skin Physiology, 1st ed.; Altemeyer, P., Hoffman, K., el Gammal, S., Hutchinson, J., Eds.; Springer: Berlin, Germany, 1995; pp. 183–199. [Google Scholar]
- Wright, J.A.; Richards, T.; Srai, S.K.S. The role of iron in the skin and cutaneous wound healing. Front. Pharmacol. 2014, 5. [Google Scholar] [CrossRef]
- Williams, L.B.; Haydel, S.E. Evaluation of the medicinal use of clay minerals as antibacterial agents. Int. Geol. Rev. 2010, 52, 745–770. [Google Scholar] [CrossRef]
- Williams, L.B. Geomimicry: Harnessing the antibacterial action of clays. Clay Miner. 2017, 52, 1–24. [Google Scholar] [CrossRef][Green Version]
- Otto, C.C.; Kilbourne, J.; Haydel, S.E. Natural and ion-exchanged illite clays reduce bacterial burden and inflammation in cutaneous meticillin-resistant Staphylococcus aureus infections in mice. J. Med Microbiol. 2016, 65, 19–27. [Google Scholar] [CrossRef]
- Wesolowski, D.J. Aluminum speciation and equilibria in aqueous solution: I. the solubility of gibbsite in the system Na-K-Cl-OH-Al(OH)4 from 0 to 100 °C. Geochim. Cosmochim. Acta 2002, 66, 3519, Erratum in 1992, 56, 1065–1091. [Google Scholar] [CrossRef]
| Source, Grain Size, and Munsell Color | TEM | SEM | Mineralogy |
|---|---|---|---|
| Clay A |  |  | 40% Cristobalite 20% Montmorillonite 16% Calcite 8% Illite 5% Quartz |
| “Redmond Healing” clay purchased online. <200 nm Munsell 10R 7/1 | |||
| Clay B |  |  | 87% Kaolinite 10% Quartz 3% Illite |
| Kaolinite sourced from Sigma Aldrich (CAS 1318-74-7). 2–5 µm Munsell 2.5YR 8/1 | |||
| Clay C |  |  | 32% Montmorillonite 25.5% Quartz 20% Calcite 16% Dolomite 6.5% Kaolinite |
| “Red Clay” from “The Clay Cure Company”. 500 nm Munsell 2.5YR 5/6 | |||
| Clay D |  |  | 55% Montmorillonite 31% Anorthoclase 8% Cristobalite 6% Calcite |
| “Premium Nutri Clay” from the “The Clay Cure Company”. <200 nm Munsell 5YR 7/1 | |||
| Clay E |  |  | 91% Quartz 3% Gypsum 3% Kaolinite 2% Illite-Smectite |
| Green from the Argiletz company. 500 nm Munsell 5Y 6/3 | |||
| Clay F |  |  | 37% Kaolinite (halloysite) 23% Hematite 17% Magnetite 17% Illite 6% Anorthoclase |
| From Mirador Chimaque, Tenerife. <200 nm Munsell 2.5YR 4/6 |
| Clay | Na (mg/L) | Mg (mg/L) | Al (mg/L) | P (mg/L) | K (mg/L) | Ca (mg/L) | Mn (mg/L) | Fe (mg/L) | Total (mg/L) |
|---|---|---|---|---|---|---|---|---|---|
| A | 31.50 | 0.30 | 0.33 | 0.00 | 0.68 | 0.43 | 0.01 | 0.10 | 33.39 |
| 94.32% | 0.89% | 0.98% | 0.00% | 2.05% | 1.30% | 0.03% | 0.30% | 99.86% | |
| B | 0.13 | 0.07 | 0.03 | 0.05 | 0.20 | 0.01 | 0.00 | 0.00 | 0.51 |
| 25.90% | 14.74% | 5.55% | 10.17% | 40.20% | 1.96% | 0.30% | 0.25% | 99.08% | |
| C | 0.57 | 2.21 | 0.23 | 0.02 | 1.01 | 1.49 | 0.00 | 0.12 | 5.68 |
| 10.05% | 38.85% | 4.12% | 0.34% | 17.77% | 26.23% | 0.01% | 2.03% | 99.39% | |
| D | 3.17 | 1.00 | 0.27 | 0.02 | 1.43 | 1.46 | 0.01 | 0.19 | 7.56 |
| 41.98% | 13.25% | 3.54% | 0.24% | 18.92% | 19.30% | 0.07% | 2.53% | 99.82% | |
| E | 0.03 | 0.90 | 0.68 | 0.01 | 0.03 | 2.87 | 0.21 | 1.65 | 6.48 |
| 0.51% | 13.92% | 10.45% | 0.15% | 0.43% | 44.35% | 3.22% | 25.41% | 98.43% | |
| F | 0.41 | 0.11 | 0.54 | 0.01 | 0.49 | 0.00 | 0.02 | 0.36 | 1.93 |
| 21.22% | 5.54% | 27.75% | 0.75% | 25.14% | 0.00% | 0.94% | 18.76% | 99.49% |
| Clay | Leachates | MFI | Suspensions | MFI |
|---|---|---|---|---|
| A | 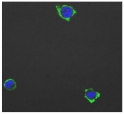 | 1,227,394 | 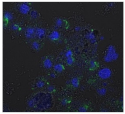 | 3519 |
| B | 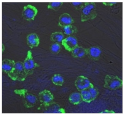 | 640,023 | 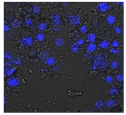 | 397 |
| C | 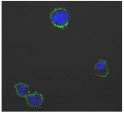 | 911,285 | 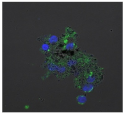 | 40,541 |
| D | 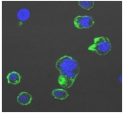 | 535,868 | 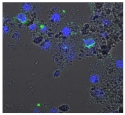 | 5441 |
| E | 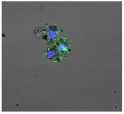 | 172,740 | 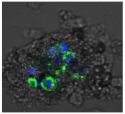 | 21,127 |
| F | 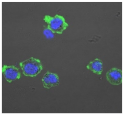 | 453,096 | 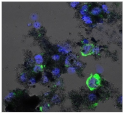 | 71,004 |
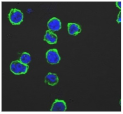 | 10,206 (Negative control) | 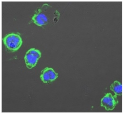 | 1,273,184 (Positive control) |
| Clay | 0 h | 24 h | 48 h |
|---|---|---|---|
| Control | 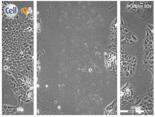 | 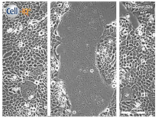 | 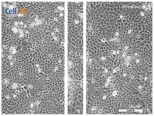 |
| Untreated | |||
| Clay B | 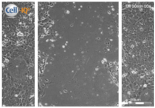 | 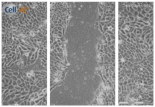 | 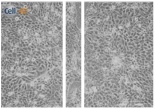 |
| A 0.05 g/L Kaolinite clay suspension | |||
| Clay E | 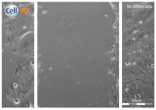 | 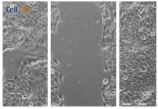 | 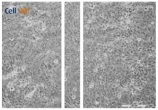 |
| A 0.05 g/L “Green” clay suspension | |||
| Clay F | 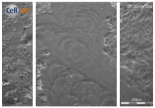 | 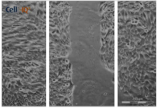 |  |
| A 0.05 g/L “Mirador Chimaque, Tenerife” clay suspension | |||
| Clay E | 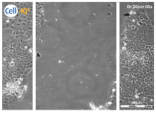 | 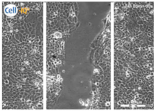 | 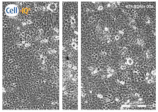 |
| Filter Sterilized Filtered from a “Green” clay suspension. | |||
| Clay F | 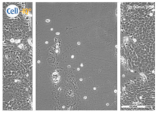 | 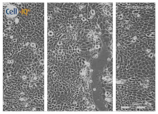 | 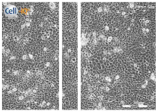 |
| Filter Sterilized Filtered from a “Mirador Chimaque, Tenerife” clay suspension. |
| pH | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | Reference | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Normal Skin | [45,46,47] | ||||||||||||||||
| Chronic Wounds | [48] | ||||||||||||||||
| A. baumannii | [49,50] | ||||||||||||||||
| E. coli | [51] | ||||||||||||||||
| K. pneumoniae | [51] | ||||||||||||||||
| P. aeruginosa | [52] | ||||||||||||||||
| MRSA | [53] | ||||||||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Incledion, A.; Boseley, M.; Moses, R.L.; Moseley, R.; Hill, K.E.; Thomas, D.W.; Adams, R.A.; Jones, T.P.; BéruBé, K.A. A New Look at the Purported Health Benefits of Commercial and Natural Clays. Biomolecules 2021, 11, 58. https://doi.org/10.3390/biom11010058
Incledion A, Boseley M, Moses RL, Moseley R, Hill KE, Thomas DW, Adams RA, Jones TP, BéruBé KA. A New Look at the Purported Health Benefits of Commercial and Natural Clays. Biomolecules. 2021; 11(1):58. https://doi.org/10.3390/biom11010058
Chicago/Turabian StyleIncledion, Alexander, Megan Boseley, Rachael L. Moses, Ryan Moseley, Katja E. Hill, David W. Thomas, Rachel A. Adams, Tim P. Jones, and Kelly A. BéruBé. 2021. "A New Look at the Purported Health Benefits of Commercial and Natural Clays" Biomolecules 11, no. 1: 58. https://doi.org/10.3390/biom11010058
APA StyleIncledion, A., Boseley, M., Moses, R. L., Moseley, R., Hill, K. E., Thomas, D. W., Adams, R. A., Jones, T. P., & BéruBé, K. A. (2021). A New Look at the Purported Health Benefits of Commercial and Natural Clays. Biomolecules, 11(1), 58. https://doi.org/10.3390/biom11010058








