Jaw Periosteal Cells Seeded in Beta-Tricalcium Phosphate Inhibit Dendritic Cell Maturation
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolation of JPCs and PBMCs
2.2. Cell Seeding of β-TCP Constructs
2.3. Coculture of JPCs-Seeded Constructs and Monocytes
2.4. Analysis of Cell Viability Using Propidium Iodide
2.5. Flow Cytometric Analyses of Dendritic Marker Expression
2.6. RNA Isolation and Quantitative Gene Expression Analyses in DCs
2.7. Proteome Profiler Array
2.8. Statistical Analysis
3. Results
3.1. Cell Morphology of DCs
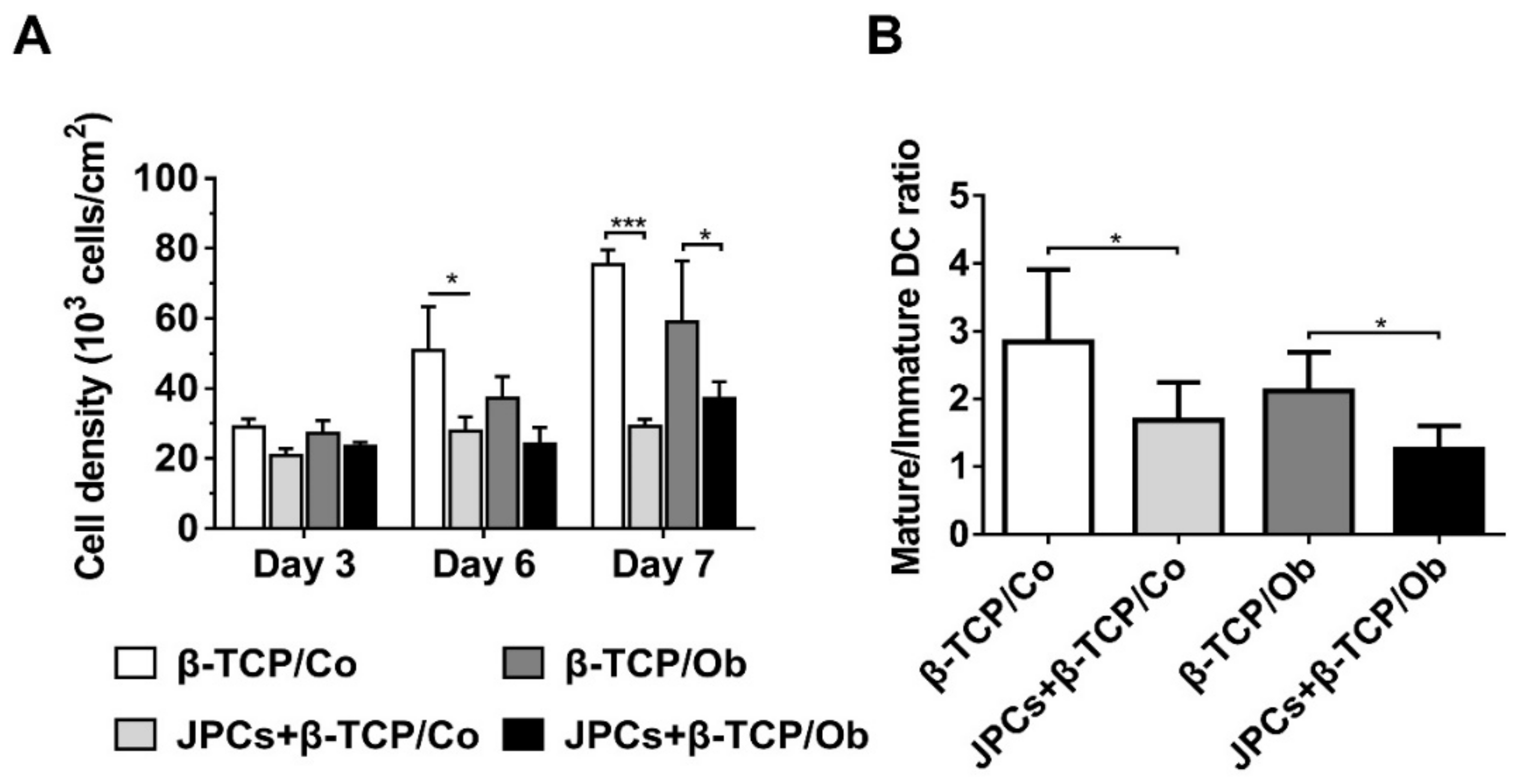
3.2. DC Densities in Mono- (β-TCP/Co and β-TCP/Ob) and Cocultures (JPCs + β-TCP/Co and Ob)
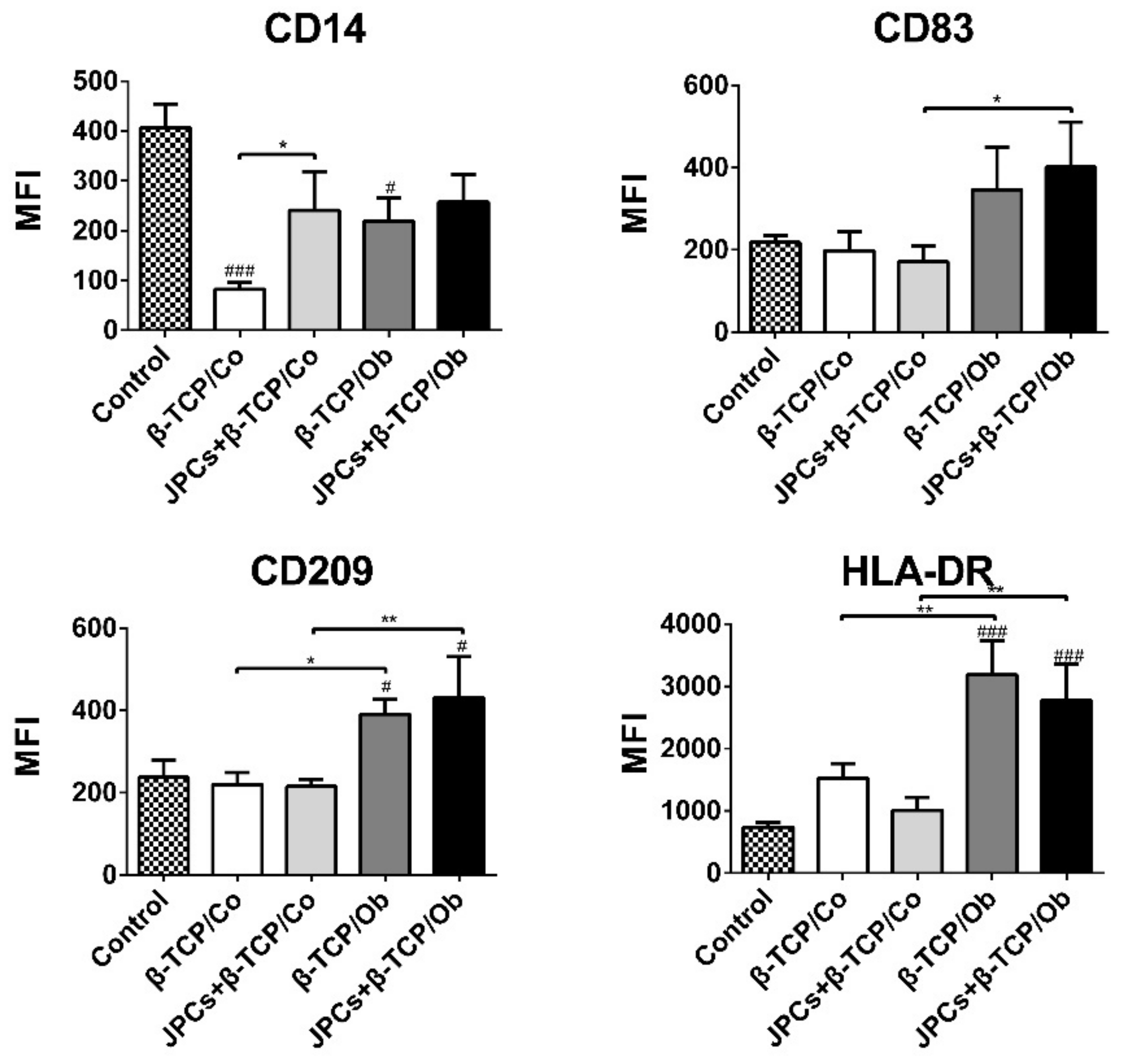
3.3. Flow Cytometric DC Characterization
3.4. DC Gene Expression Analysis
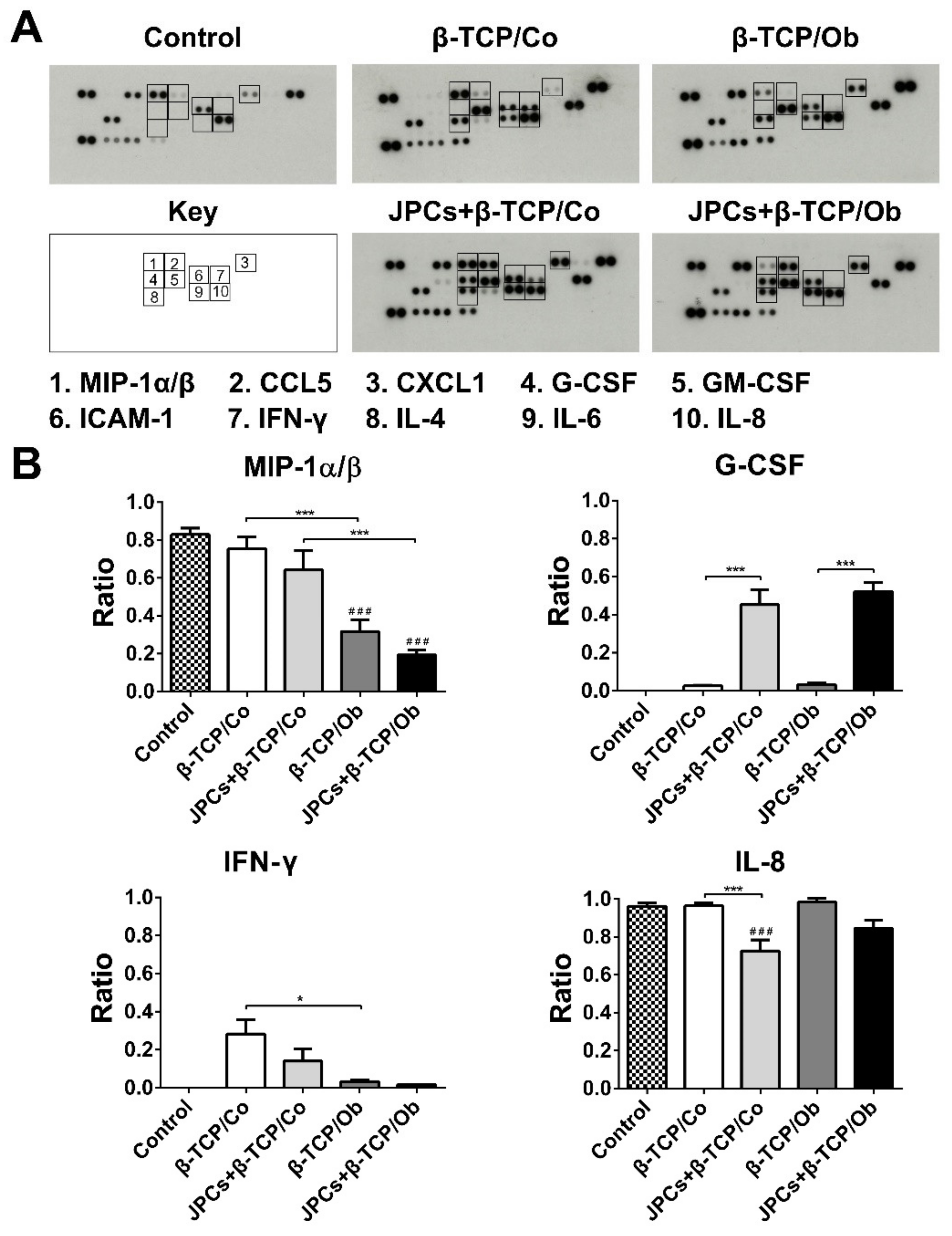
3.5. DC Protein Expression Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Paschos, N.K.; Brown, W.E.; Eswaramoorthy, R.; Hu, J.C.; Athanasiou, K.A. Advances in tissue engineering through stem cell-based co-culture. J. Tissue Eng. Regen. Med. 2014, 9, 488–503. [Google Scholar] [CrossRef] [PubMed]
- Pomeraniec, L.; Benayahu, D. Mesenchymal Cell Growth and Differentiation on a New Biocomposite Material: A Promising Model for Regeneration Therapy. Biomolecules 2020, 10, 458. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, M.; Nonomura, H.; Kamil, S.H.; Ignotz, R.A. Periosteal cell pellet culture system: A new technique for bone engineering. Cell Transplant. 2006, 15, 521–532. [Google Scholar] [CrossRef] [PubMed]
- Hutmacher, D.W.; Sittinger, M. Periosteal Cells in Bone Tissue Engineering. Tissue Eng. 2003, 9, 45–64. [Google Scholar] [CrossRef] [PubMed]
- Nakahara, H.; Bruder, S.; Haynesworth, S.; Holecek, J.; Baber, M.; Goldberg, V.; Caplan, A. Bone and cartilage formation in diffusion chambers by subcultured cells derived from the periosteum. Bone 1990, 11, 181–188. [Google Scholar] [CrossRef]
- Nakahara, H.; Goldberg, V.M.; Caplan, A. Culture-expanded human periosteal-derived cells exhibit osteochondral potential in vivo. J. Orthop. Res. 1991, 9, 465–476. [Google Scholar] [CrossRef] [PubMed]
- Sakata, Y.; Ueno, T.; Kagawa, T.; Kanou, M.; Fujii, T.; Yamachika, E.; Sugahara, T. Osteogenic potential of cultured human periosteum-derived cells—A pilot study of human cell transplantation into a rat calvarial defect model. J. Cranio Maxillofac. Surg. 2006, 34, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Alexander, D.; Hoffmann, J.; Munz, A.; Friedrich, B.; Geis-Gerstorfer, J.; Reinert, S. Analysis of OPLA scaffolds for bone engineering constructs using human jaw periosteal cells. J. Mater. Sci. Mater. Electron. 2007, 19, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Alexander, D.; Hoffmann, J.; Munz, A.; Friedrich, B.; Geis-Gerstorfer, J.; Reinert, S. Comparison of three dimensional scaffolds for bone engineering constructs using human jaw periosteal cells. J. Stem Cells Regen. Med. 2007, 2, 177. [Google Scholar] [PubMed]
- Brauchle, E.; Berrio, D.C.; Rieger, M.; Schenke-Layland, K.; Reinert, S.; Alexander, D. Raman Spectroscopic Analyses of Jaw Periosteal Cell Mineralization. Stem Cells Int. 2017, 2017, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Alexander, D.; Schäfer, F.; Olbrich, M.; Friedrich, B.; Bühring, H.-J.; Hoffmann, J.; Reinert, S. MSCA-1/TNAP selection of human jaw periosteal cells improves their mineralization capacity. Cell. Physiol. Biochem. 2010, 26, 1073–1080. [Google Scholar] [CrossRef] [PubMed]
- Alexander, D.; Rieger, M.; Klein, C.; Ardjomandi, N.; Reinert, S. Selection of Osteoprogenitors from the Jaw Periosteum by a Specific Animal-Free Culture Medium. PLoS ONE 2013, 8, e81674. [Google Scholar] [CrossRef] [PubMed]
- Ardjomandi, N.; Klein, C.; Köhler, K.; Maurer, A.; Kalbacher, H.; Niederländer, J.; Reinert, S.; Alexander, D. Indirect coating of RGD peptides using a poly-L-lysine spacer enhances jaw periosteal cell adhesion, proliferation, and differentiation into osteogenic tissue. J. Biomed. Mater. Res. Part A 2012, 100, 2034–2044. [Google Scholar] [CrossRef] [PubMed]
- Eggli, P.S.; Müller, W.; Schenk, R.K. Porous hydroxyapatite and tricalcium phosphate cylinders with two different pore size ranges implanted in the cancellous bone of rabbits. A comparative histomorphometric and histologic study of bony ingrowth and implant substitution. Clin. Orthop. Relat. Res. 1988, 232, 127–138. [Google Scholar] [CrossRef]
- Okuda, T.; Ioku, K.; Yonezawa, I.; Minagi, H.; Kawachi, G.; Gonda, Y.; Murayama, H.; Shibata, Y.; Minami, S.; Kamihira, S.; et al. The effect of the microstructure of β-tricalcium phosphate on the metabolism of subsequently formed bone tissue. Biomaterials 2007, 28, 2612–2621. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Lin, H.; Fang, T.; Li, X.; Dai, W.; Uemura, T.; Dong, J. The repair of large segmental bone defects in the rabbit with vascularized tissue engineered bone. Biomaterials 2010, 31, 1171–1179. [Google Scholar] [CrossRef] [PubMed]
- Vacanti, C.A.; Bonassar, L.J.; Vacanti, M.P.; Shufflebarger, J. Replacement of an Avulsed Phalanx with Tissue-Engineered Bone. N. Engl. J. Med. 2001, 344, 1511–1514. [Google Scholar] [CrossRef] [PubMed]
- Ardjomandi, N.; Huth, J.; Stamov, D.; Henrich, A.; Klein, C.; Wendel, H.-P.; Reinert, S.; Alexander, D. Surface biofunctionalization of β-TCP blocks using aptamer 74 for bone tissue engineering. Mater. Sci. Eng. C 2016, 67, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Ardjomandi, N.; Henrich, A.; Huth, J.; Klein, C.; Schweizer, E.; Scheideler, L.; Rupp, F.; Reinert, S.; Alexander, D. Coating of ß-tricalcium phosphate scaffolds—A comparison between graphene oxide and poly-lactic-co-glycolic acid. Biomed. Mater. 2015, 10, 45018. [Google Scholar] [CrossRef] [PubMed]
- Heuze, M.; Vargas, P.; Chabaud, M.; Le Berre, M.; Liu, Y.-J.; Collin, O.; Solanes, P.; Voituriez, R.; Piel, M.; Lennon-Duménil, A.-M. Migration of dendritic cells: Physical principles, molecular mechanisms, and functional implications. Immunol. Rev. 2013, 256, 240–254. [Google Scholar] [CrossRef] [PubMed]
- Romani, N.; Gruner, S.; Brang, D.; Kämpgen, E.; Lenz, A.; Trockenbacher, B.; Konwalinka, G.; O Fritsch, P.; Steinman, R.M.; Schuler, G. Proliferating dendritic cell progenitors in human blood. J. Exp. Med. 1994, 180, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Sallusto, F.; Lanzavecchia, A. Efficient presentation of soluble antigen by cultured human dendritic cells is maintained by granulocyte/macrophage colony-stimulating factor plus interleukin 4 and downregulated by tumor necrosis factor alpha. J. Exp. Med. 1994, 179, 1109–1118. [Google Scholar] [CrossRef] [PubMed]
- Chapuis, F.; Rosenzwajg, M.; Yagello, M.; Ekman, M.; Biberfeld, P.; Gluckman, J.C. Differentiation of human dendritic cells from monocytesin vitro. Eur. J. Immunol. 1997, 27, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Steinman, R.M.; Hawiger, D.; Nussenzweig, M.C. Tolerogenic dendritic cells. Annu. Rev. Immunol. 2003, 21, 685–711. [Google Scholar] [CrossRef] [PubMed]
- E Macatonia, S.; A Hosken, N.; Litton, M.; Vieira, P.; Hsieh, C.S.; A Culpepper, J.; Wysocka, M.; Trinchieri, G.; Murphy, K.M.; O’Garra, A. Dendritic cells produce IL-12 and direct the development of Th1 cells from naive CD4+ T cells. J. Immunol. 1995, 154, 5071–5079. [Google Scholar]
- Nauta, A.J.; Kruisselbrink, A.B.; Lurvink, E.; Willemze, R.; Fibbe, W.E. Mesenchymal stem cells inhibit generation and function of both CD34+-derived and monocyte-derived dendritic cells. J. Immunol. 2006, 177, 2080–2087. [Google Scholar] [CrossRef] [PubMed]
- Uccelli, A.; Moretta, L.; Pistoia, V. Immunoregulatory function of mesenchymal stem cells. Eur. J. Immunol. 2006, 36, 2566–2573. [Google Scholar] [CrossRef] [PubMed]
- Di Nicola, M.; Carlo-Stella, C.; Magni, M.; Milanesi, M.; Longoni, P.D.; Matteucci, P.; Grisanti, S.; Gianni, A.M. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood 2002, 99, 3838–3843. [Google Scholar] [CrossRef] [PubMed]
- Krampera, M.; Glennie, S.; Dyson, J.; Scott, D.; Laylor, R.; Simpson, E.; Dazzi, F. Bone marrow mesenchymal stem cells inhibit the response of naive and memory antigen-specific T cells to their cognate peptide. Blood 2003, 101, 3722–3729. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Rottau, D.; Kohler, F.; Reinert, S.; Alexander, D. Effects of Jaw Periosteal Cells on Dendritic Cell Maturation. J. Clin. Med. 2018, 7, 312. [Google Scholar] [CrossRef] [PubMed]
- Jensen, S.S.; Broggini, N.; Hjørting-Hansen, E.; Schenk, R.; Buser, D. Bone healing and graft resorption of autograft, anorganic bovine bone and β-tricalcium phosphate. A histologic and histomorphometric study in the mandibles of minipigs. Clin. Oral Implant. Res. 2006, 17, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Buser, D.; Hoffmann, B.; Bernard, J.-P.; Lussi, A.; Mettler, D.; Schenk, R.K. Evaluation of filling materials in membrane--protected bone defects. A comparative histomorphometric study in the mandible of miniature pigs. Clin. Oral Implant. Res. 1998, 9, 137–150. [Google Scholar] [CrossRef] [PubMed]
- Ding, T.; Sun, J.; Zhang, P. Immune evaluation of biomaterials in TNF-α and IL-1β at mRNA level. J. Mater. Sci. Mater. Electron. 2007, 18, 2233–2236. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, R.A.; Mazor, Z.; Foitzik, C.; Prasad, H.; Rohrer, M.; Palti, A. β-tricalcium phosphate as bone substitute material: Properties and clinical applications. J. Osseointegration 2010, 2, 61–68. [Google Scholar]
- Zhu, H.; Yang, F.; Tang, B.; Li, X.-M.; Chu, Y.-N.; Liu, Y.; Wang, S.-G.; Wu, D.; Zhang, Y. Mesenchymal stem cells attenuated PLGA-induced inflammatory responses by inhibiting host DC maturation and function. Biomaterials 2015, 53, 688–698. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Tang, H.; Guo, Z.; An, H.; Zhu, X.; Song, W.; Guo, J.; Huang, X.; Chen, T.; Wang, J.; et al. Splenic stroma drives mature dendritic cells to differentiate into regulatory dendritic cells. Nat. Immunol. 2004, 5, 1124–1133. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.-X.; Zhang, Y.; Liu, B.; Zhang, S.-X.; Wu, Y.; Yu, X.-D.; Mao, N. Human mesenchymal stem cells inhibit differentiation and function of monocyte-derived dendritic cells. Blood 2005, 105, 4120–4126. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-J.; Foon, K.A.; Mailliard, R.B.; Muthuswamy, R.; Kalinski, P. Type 1-polarized dendritic cells loaded with autologous tumor are a potent immunogen against chronic lymphocytic leukemia. J. Leukoc. Biol. 2008, 84, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Tiemessen, M.M.; Jagger, A.L.; Evans, H.G.; Herwijnen, M.J.; John, S.; Taams, L.S. CD4+CD25+Foxp3+ regulatory T cells induce alternative activation of human monocytes/macrophages. Proc. Natl. Acad. Sci. USA 2007, 104, 19446–19451. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Chen, K.; Du, W.; Han, Z.-B.; Ren, H.; Chi, Y.; Yang, S.G.; Bayard, F.; Zhu, D.; Han, Z.C. CD14+ monocytes promote the immunosuppressive effect of human umbilical cord matrix stem cells. Exp. Cell Res. 2010, 316, 2414–2423. [Google Scholar] [CrossRef] [PubMed]
- Hawrylowicz, C.M.; Guida, L.; Paleolog, E. Dexamethasone up-regulates granulocyte-macrophage colony-stimulating factor receptor expression on human monocytes. Immunology 1994, 83, 274–280. [Google Scholar] [PubMed]
- Oehler, L.; Majdic, O.; Pickl, W.F.; Stöckl, J.; Riedl, E.; Drach, J.; Rappersberger, K.; Geissler, K.; Knapp, W. Neutrophil Granulocyte–committed Cells Can Be Driven to Acquire Dendritic Cell Characteristics. J. Exp. Med. 1998, 187, 1019–1028. [Google Scholar] [CrossRef] [PubMed]
- Couper, K.N.; Blount, D.G.; Riley, E.M. IL-10: The master regulator of immunity to infection. J. Immunol. 2008, 180, 5771–5777. [Google Scholar] [CrossRef] [PubMed]
- Stobie, L.; Gurunathan, S.; Prussin, C.; Sacks, D.L.; Glaichenhaus, N.; Wu, C.-Y.; Seder, R.A. The role of antigen and IL-12 in sustaining Th1 memory cells in vivo: IL-12 is required to maintain memory/effector Th1 cells sufficient to mediate protection to an infectious parasite challenge. Proc. Natl. Acad. Sci. USA 2000, 97, 8427–8432. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ge, X.-H.; Guo, X.; Guan, S.; Li, X.-M.; Gu, W.; Xu, W. Bone Marrow Mesenchymal Stem Cells Inhibit the Function of Dendritic Cells by Secreting Galectin-1. BioMed Res. Int. 2017, 2017, 1–19. [Google Scholar] [CrossRef] [PubMed]
- McLemore, M.L.; Grewal, S.; Liu, F.; Archambault, A.; Poursine-Laurent, J.; Haug, J.; Link, D.C. STAT-3 activation is required for normal G-CSF-dependent proliferation and granulocytic differentiation. Immunity 2001, 14, 193–204. [Google Scholar] [CrossRef]
- Wang, T.; Niu, G.; Kortylewski, M.; Burdelya, L.; Shain, K.; Zhang, S.; Bhattacharya, R.; Gabrilovich, D.; Heller, R.; Coppola, M.; et al. Regulation of the innate and adaptive immune responses by Stat-3 signaling in tumor cells. Nat. Med. 2003, 10, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Bharadwaj, U.; Li, M.; Zhang, R.; Chen, C.; Yao, Q.C. Elevated Interleukin-6 and G-CSF in Human Pancreatic Cancer Cell Conditioned Medium Suppress Dendritic Cell Differentiation and Activation. Cancer Res. 2007, 67, 5479–5488. [Google Scholar] [CrossRef] [PubMed]
- Xia, C.-Q.; Peng, R.; Beato, F.; Clare-Salzler, M.J. Dexamethasone Induces IL-10-Producing Monocyte-Derived Dendritic Cells with Durable Immaturity. Scand. J. Immunol. 2005, 62, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Lange, T.; Schilling, A.F.; Peters, F.; Haag, F.; Morlock, M.M.; Rueger, J.M.; Amling, M. Proinflammatory and osteoclastogenic effects of beta-tricalciumphosphate and hydroxyapatite particles on human mononuclear cells in vitro. Biomaterials 2009, 30, 5312–5318. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Uemura, T.; Shirasaki, Y.; Tateishi, T. Promotion of bone formation using highly pure porous β-TCP combined with bone marrow-derived osteoprogenitor cells. Biomaterials 2002, 23, 4493–4502. [Google Scholar] [CrossRef]
- Tai, S.; Cheng, J.-Y.; Ishii, H.; Shimono, K.; Zangiacomi, V.; Satoh, T.; Hosono, T.; Suzuki, E.; Yamaguchi, K.; Maruyama, K. Effects of beta-tricalcium phosphate particles on primary cultured murine dendritic cells and macrophages. Int. Immunopharmacol. 2016, 40, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Solaroglu, I.; Cahill, J.; Tsubokawa, T.; Beskonakli, E.; Zhang, J.H. Granulocyte colony-stimulating factor protects the brain against experimental stroke via inhibition of apoptosis and inflammation. Neurol. Res. 2009, 31, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Park, C.W.; Kim, K.-S.; Bae, S.; Son, H.K.; Myung, P.-K.; Hong, H.J.; Kim, H. Cytokine Secretion Profiling of Human Mesenchymal Stem Cells by Antibody Array. Int. J. Stem Cells 2009, 2, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.H.; Yoo, K.H.; Choi, K.S.; Choi, J.; Choi, S.-Y.; Yang, S.-E.; Yang, Y.-S.; Im, H.J.; Kim, K.H.; Jung, H.L.; et al. Gene expression profile of cytokine and growth factor during differentiation of bone marrow-derived mesenchymal stem cell. Cytokine 2005, 31, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Ben Azouna, N.; Jenhani, F.; Regaya, Z.; Berraeis, L.; Ben Othman, T.; Ducrocq, E.; Domenech, J. Phenotypical and functional characteristics of mesenchymal stem cells from bone marrow: Comparison of culture using different media supplemented with human platelet lysate or fetal bovine serum. Stem Cell Res. Ther. 2012, 3, 6. [Google Scholar] [CrossRef] [PubMed]
- Talarn, C.; Urbano-Ispizua, A.; Martino, R.; Batlle, M.; Fernández-Avilés, F.; Herrera, C.; A Pérez-Simón, J.; Gaya, A.; Aymerich, M.; Pétriz, J.; et al. G-CSF increases the number of peripheral blood dendritic cells CD16+ and modifies the expression of the costimulatory molecule CD86+. Bone Marrow Transplant. 2006, 37, 873–879. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Reddy, V.; Hill, G.R.; Pan, L.; Gerbitz, A.; Teshima, T.; Brinson, Y.; Ferrara, J.L.M. G-CSF modulates cytokine profile of dendritic cells and decreases acute graft-versus-host disease through effects on the donor rather than the recipient. Transplantation 2000, 69, 691–693. [Google Scholar] [CrossRef] [PubMed]
- Kondo, M.; Wagers, A.J.; Manz, M.G.; Prohaska, S.S.; Scherer, D.C.; Beilhack, G.F.; Shizuru, J.A.; Weissman, I.L. Biology of hematopoietic stem cells and progenitors: Implications for Clinical Application. Annu. Rev. Immunol. 2003, 21, 759–806. [Google Scholar] [CrossRef] [PubMed]
- Souza, L.M.; Boone, T.C.; Gabrilove, J.; Lai, P.H.; Zsebo, K.M.; Murdock, D.C.; Chazin, V.R.; Bruszewski, J.; Lu, H.; Chen, K.K. Recombinant human granulocyte colony-stimulating factor: Effects on normal and leukemic myeloid cells. Science 1986, 232, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Sheridan, W.; Fox, R.; Begley, C.; Maher, D.; McGrath, K.; Juttner, C.; To, L.; Szer, J.; Mostyn, G. Effect of peripheral-blood progenitor cells mobilised by filgrastim (G-CSF) on platelet recovery after high-dose chemotherapy. Lancet 1992, 339, 640–644. [Google Scholar] [CrossRef]
- Zhang, N.; Rogers, T.J.; Caterina, M.; Oppenheim, J.J. Proinflammatory Chemokines, Such as C-C Chemokine Ligand 3, Desensitize μ-Opioid Receptors on Dorsal Root Ganglia Neurons. J. Immunol. 2004, 173, 594–599. [Google Scholar] [CrossRef] [PubMed]
- Tedla, N.; Wang, H.-W.; McNeil, H.P.; Di Girolamo, N.; Hampartzoumian, T.; Wakefield, D.; Lloyd, A. Regulation of T lymphocyte trafficking into lymph nodes during an immune response by the chemokines macrophage inflammatory protein (MIP)-1α and MIP-1β. J. Immunol. 1998, 161, 5663–5672. [Google Scholar] [PubMed]
- Nehmé, A.; Edelman, J. Dexamethasone Inhibits High Glucose-, TNF-α-, and IL-1β–induced Secretion of Inflammatory and Angiogenic Mediators from Retinal Microvascular Pericytes. Investig. Opthalmology Vis. Sci. 2008, 49, 2030–2038. [Google Scholar] [CrossRef] [PubMed]


| Surface Markers | Control | Experimental Groups | |||
|---|---|---|---|---|---|
| β-TCP/Co | β-TCP/Ob | JPCs + β-TCP/Co | JPCs + β-TCP/Ob | ||
| CD14 | 11.06 ± 6.39 | 20.85 ± 10.68 a | 72.45 ± 3.44 a* | 14.27 ± 2.37 A | 66.67 ± 1.39 A* |
| CD83 | 9.21 ± 4.76 | 40.85 ± 21.36 | 38.64 ± 26.19 | 48.48 ± 9.91 | 41.38 ± 15.69 |
| CD209 | 1.03 ± 0.59 | 14.35 ± 7.32 a | 69.50 ± 13.02 a* | 28.56 ± 6.19 | 62.77 ± 13.54 * |
| HLA-DR | 26.74 ± 5.68 | 91.55 ± 5.86 * | 95.83 ± 1.57 * | 87.85 ± 6.25 * | 93.73 ± 3.59 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dai, J.; Umrath, F.; Reinert, S.; Alexander, D. Jaw Periosteal Cells Seeded in Beta-Tricalcium Phosphate Inhibit Dendritic Cell Maturation. Biomolecules 2020, 10, 887. https://doi.org/10.3390/biom10060887
Dai J, Umrath F, Reinert S, Alexander D. Jaw Periosteal Cells Seeded in Beta-Tricalcium Phosphate Inhibit Dendritic Cell Maturation. Biomolecules. 2020; 10(6):887. https://doi.org/10.3390/biom10060887
Chicago/Turabian StyleDai, Jingtao, Felix Umrath, Siegmar Reinert, and Dorothea Alexander. 2020. "Jaw Periosteal Cells Seeded in Beta-Tricalcium Phosphate Inhibit Dendritic Cell Maturation" Biomolecules 10, no. 6: 887. https://doi.org/10.3390/biom10060887
APA StyleDai, J., Umrath, F., Reinert, S., & Alexander, D. (2020). Jaw Periosteal Cells Seeded in Beta-Tricalcium Phosphate Inhibit Dendritic Cell Maturation. Biomolecules, 10(6), 887. https://doi.org/10.3390/biom10060887




