Protective Role of Spirulina platensis against Bifenthrin-Induced Reprotoxicity in Adult Male Mice by Reversing Expression of Altered Histological, Biochemical, and Molecular Markers Including MicroRNAs
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Evaluation of Physicochemical, Nutritional and Microbiological Qualities, and Antioxidant Activity of the Isolated SP
2.3. Animal Care
2.4. Experimental Protocol
2.5. Sperm Collection and Analysis
2.6. Determination of Oxidative Stress Markers
2.7. Determination of Non-enzymatic and Enzymatic Antioxidants in Testicular Tissues
2.8. Estimation of Testosterone in Sera
2.9. Analysis of DNA Fragmentation
2.10. Analysis of Gene Expression by Quantitative RT-PCR (qRT-PCR)
2.11. Protein Quantification
2.12. Testicular Histopathology
2.13. Statistical Analysis
3. Results
3.1. SP as a Source of Nutritional and Bioactive Compounds
3.2. Effect of BF and/or SP Treatment on General Health, Body Weight and Reproductive Organs Weight
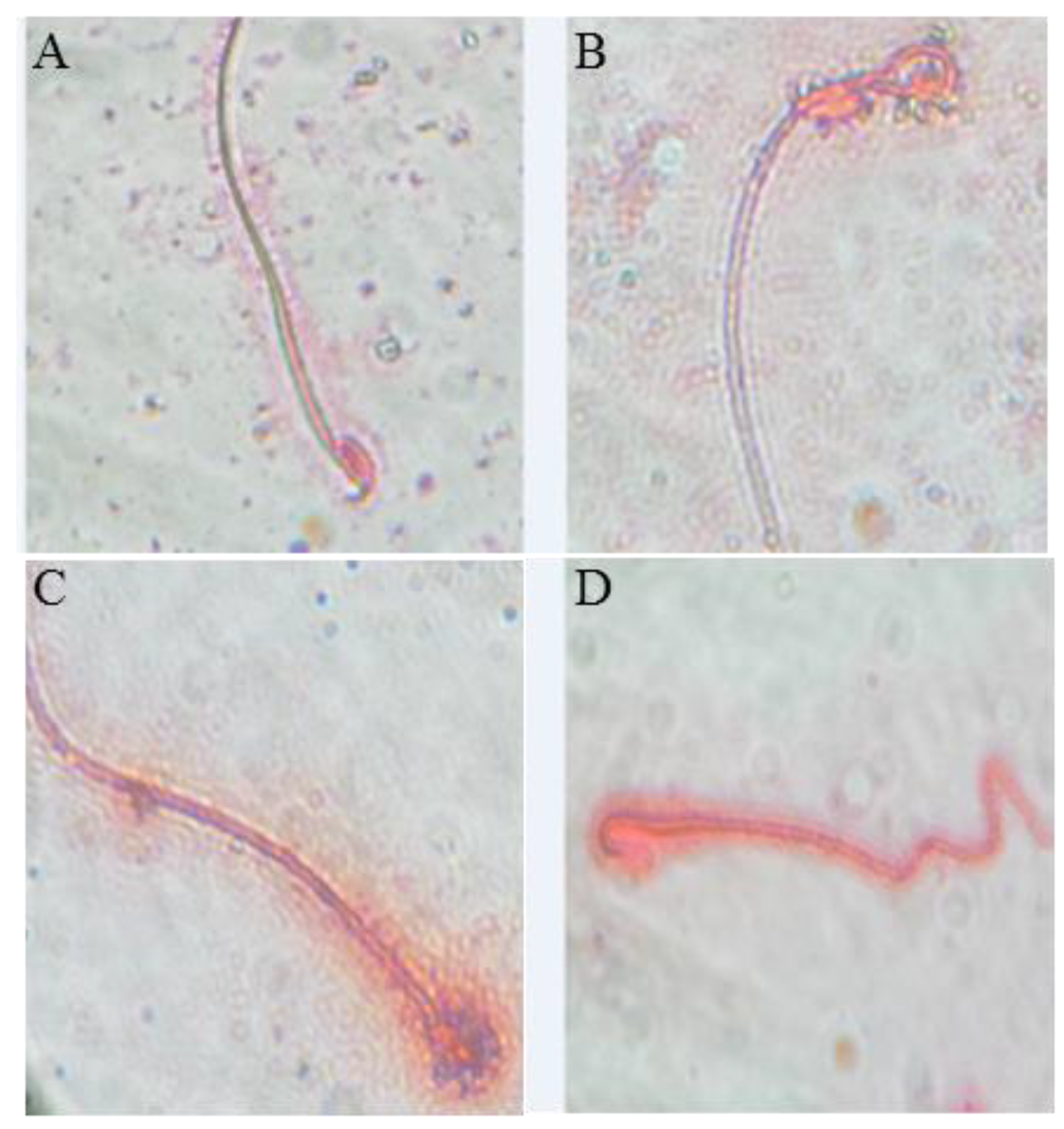
3.3. Effect of BF and/or SP Treatment on Seminal Picture
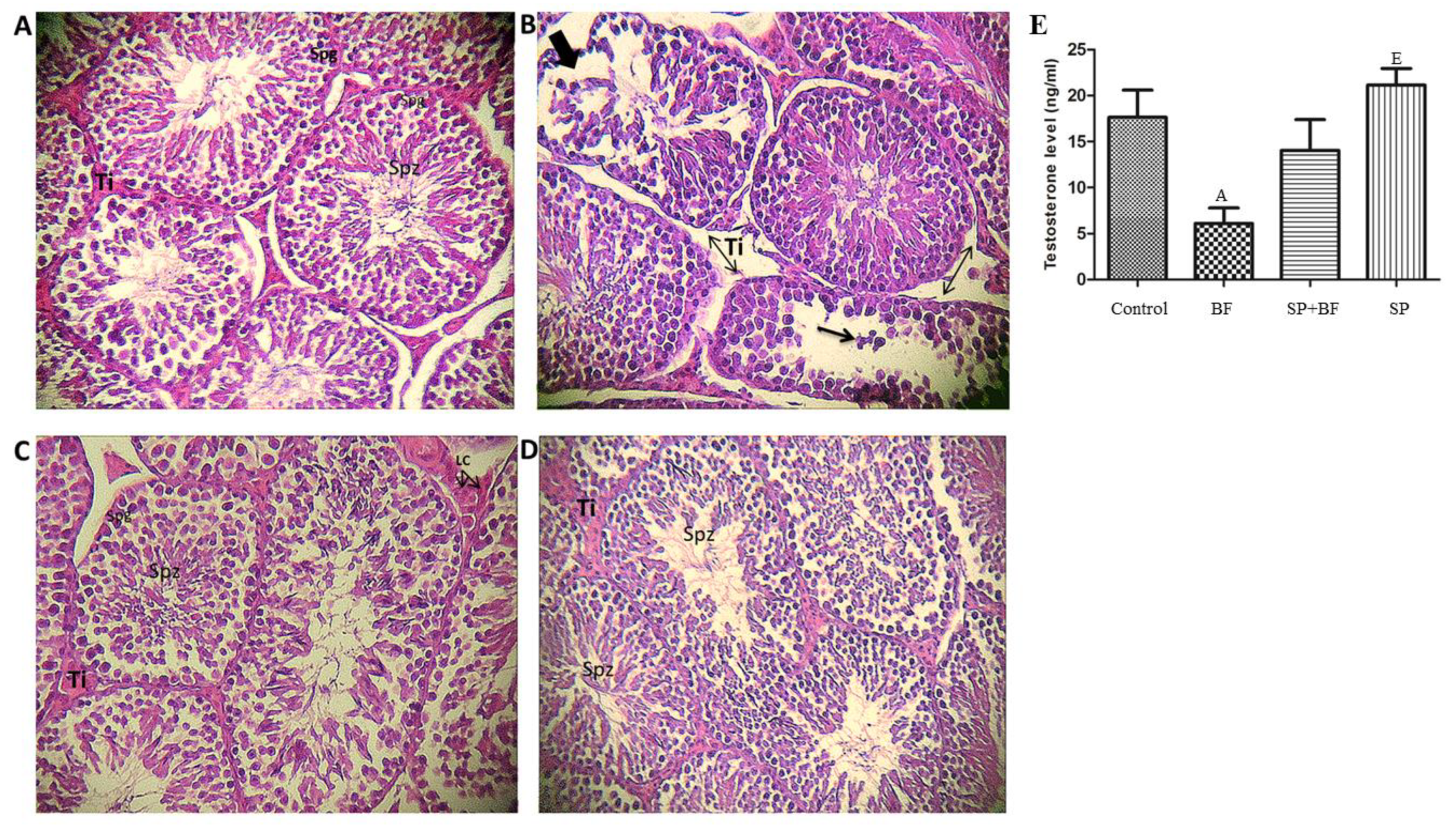
3.4. Histological Change in Mouse Testes
3.5. Effect of BF and/or SP Treatment on Plasma Testosterone
3.6. Effect of SP on Testicular Parameters of BF Intoxicated Male Mice
3.6.1. ROS Level and Oxidative Stress Markers (MDA, PCO, NO)
3.6.2. Enzymatic and Non-Enzymatic Antioxidants
3.6.3. Effect of SP on Oxidative DNA Fragmentation Induced by BF
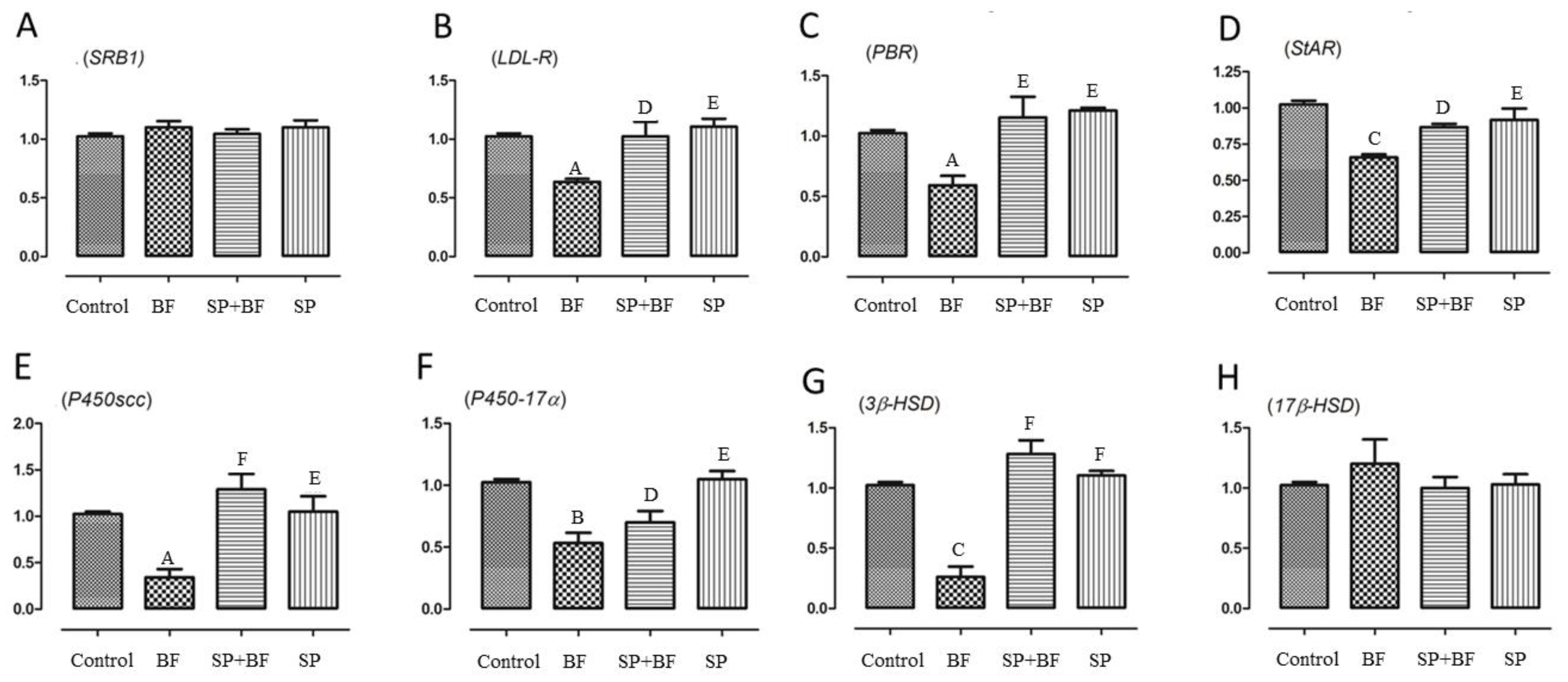
3.6.4. Changes in the Transcription of Genes Related to Cholesterol Transport and Testosterone Synthesis
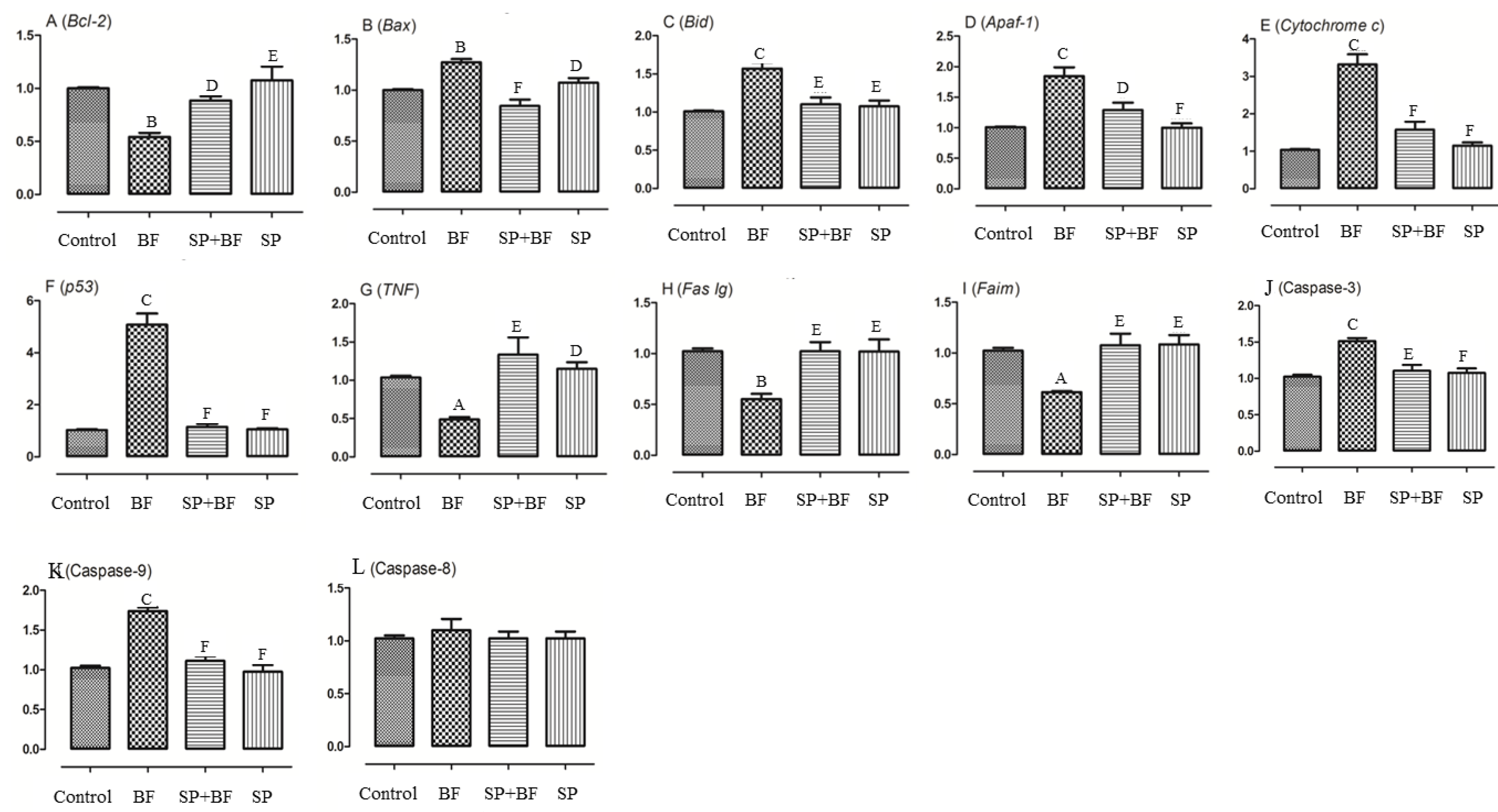
3.6.5. Changes in the Transcription of Genes on the Apoptosis-Inducing Pathway
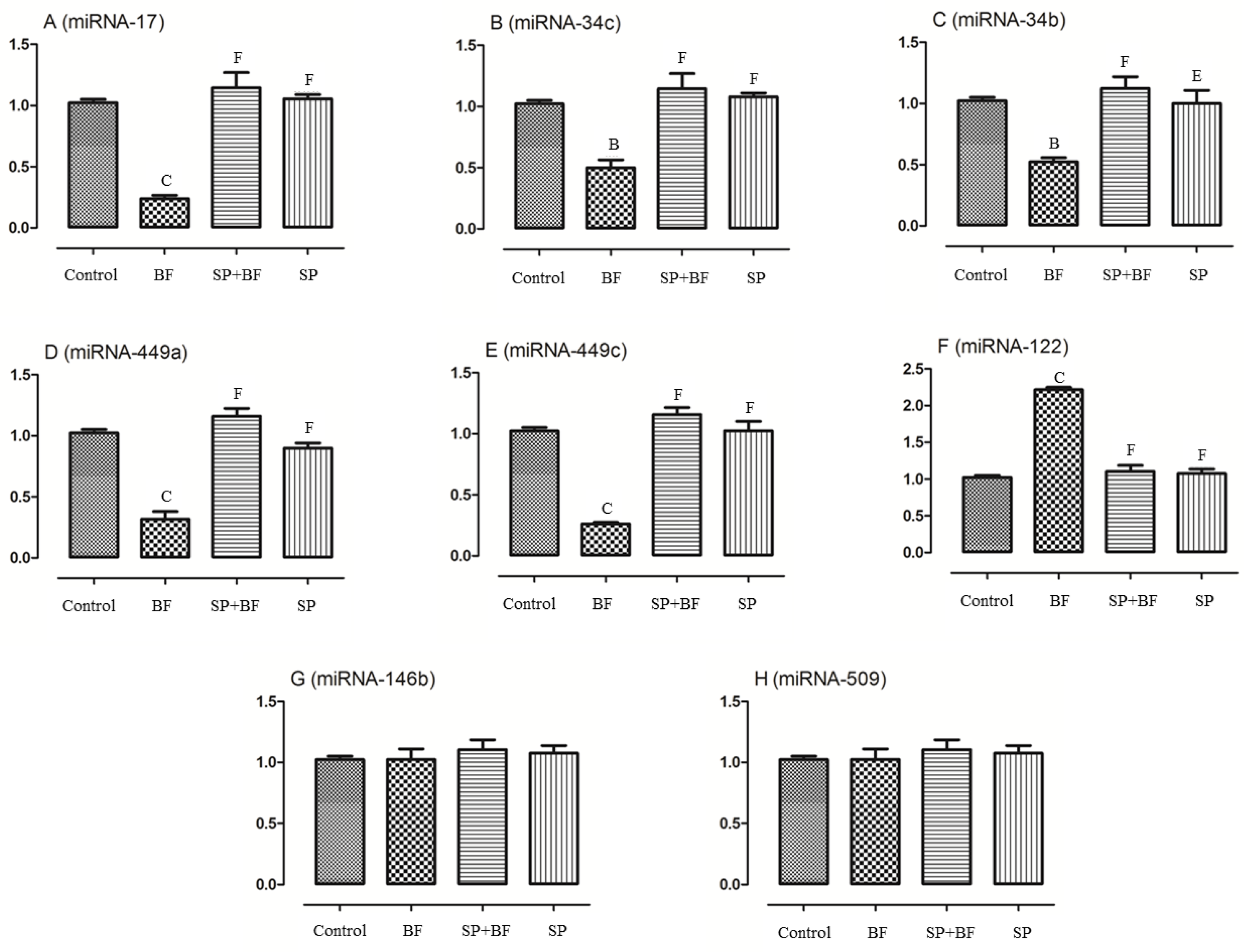
3.6.6. Changes in the Spermatogenesis and Apoptosis Related miRNA
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ben Halima, N.; Ben Slima, A.; Moalla, I.; Fetoui, H.; Pichon, C.; Gdoura, R.; Abdelkafi, S. Protective effects of oat oil on deltamethrin-induced reprotoxicity in male mice. Food Funct. 2014, 5, 2070–2077. [Google Scholar] [CrossRef]
- Liu, J.; Yang, Y.; Zhuang, S.; Yang, Y.; Li, F.; Liu, W. Enantioselective endocrine-disrupting effects of bifenthrin on hormone synthesis in rat ovarian cells. Toxicology 2011, 290, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Gao, X.; He, B.; Jin, Y.; Fu, Z. Cis-bifenthrin causes immunotoxicity in murine macrophages. Chemosphere 2017, 168, 1375–1382. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lu, M.; Zhou, P.; Wang, C.; Zhang, Q.; Zhao, M. Multilevel evaluations of potential liver injury of bifenthrin. Pestic. Biochem. Physiol. 2015, 122, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Wang, J.; Sun, X.; Ye, Y.; Xu, M.; Wang, J.; Chen, S.; Fu, Z. Exposure of maternal mice to cis-bifenthrin enantioselectively disrupts the transcription of genes related to testosterone synthesis in male offspring. Reprod. Toxicol. 2013, 42, 156–163. [Google Scholar] [CrossRef]
- Jin, Y.; Pan, X.; Fu, Z. Exposure to bifenthrin causes immunotoxicity and oxidative stress in male mice. Environ. Toxicol. 2015, 29, 991–999. [Google Scholar] [CrossRef]
- Gharagozloo, P.; Aitken, R.J. The role of sperm oxidative stress in male infertility and the significance of oral antioxidant therapy. Hum. Reprod. 2011, 26, 1628–1640. [Google Scholar] [CrossRef]
- Dammak, M.; Hadrich, B.; Miladi, R.; Barkallah, M.; Hentati, F.; Hachicha, R.; Laroche, C.; Michaud, P.; Fendri, I.; Abdelkafi, S. Effects of nutritional conditions on growth and biochemical composition of Tetraselmis sp. Lipids Health Dis. 2017, 16, 41. [Google Scholar] [CrossRef]
- Nehmé, R.; Atieh, C.; Fayad, S.; Claude, B.; Chartier, A.; Tannoury, M.; Elleuch, F.; Abdelkafi, S.; Pichon, C.; Morin, P. Microalgae amino acid extraction and analysis at nanomolar level using electroporation and capillary electrophoresis with laser-induced fluorescence detection. J. Sep. Sci. 2016, 40, 558–566. [Google Scholar] [CrossRef]
- Barkallah, M.; Dammak, M.; Louati, I.; Hentati, F.; Hadrich, B.; Mechichi, T.; Ayadi, M.A.; Fendri, I.; Attia, H.; Abdelkafi, S. Effect of Spirulina platensis fortification on physicochemical, textural, antioxidant and sensory properties of yogurt during fermentation and storage. LWT–Food Sci. Technol. 2017, 84, 323–330. [Google Scholar] [CrossRef]
- Ben Amor, F.; Barkallah, M.; Elleuch, F.; Karkouch, N.; Dammak, M.; Baréa, B.; Villeneuve, P.; Abdelkafi, S.; Fendri, I. Cyanobacteria as source of marine bioactive compounds: Molecular specific detection based on Δ9 desaturase gene. Int. J. Biol. Macromol. 2017, 105, 1440–1445. [Google Scholar] [CrossRef] [PubMed]
- Ben Hlima, H.; Bohli, T.; Kraiem, M.; Ouederni, A.; Mellouli, L.; Michaud, P.; Abdelkafi, S.; Smaoui, S. Combined effect of Spirulina platensis and Punica granatum peel extacts: phytochemical content and antiphytophatogenic activity. Appl. Sci. 2019, 9, 5475. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, X. Inhibitory effects of small molecular peptides from Spirulina (Arthrospira) platensis on cancer cell growth. Food. Funct. 2016, 7, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Hashem, M.A.; Shoeeb, S.B.A.; Abd-Elhakim, Y.M.; Mohamed, W.A.M. The antitumor activity of Arthrospira platensis and/or cisplatin in a murine model of Ehrlich ascites carcinoma with hematinic and hepato-renal protective action. J. Funct. Food. 2020, 66, 103831. [Google Scholar] [CrossRef]
- Bashandy, S.A.; El Awdan, S.A.; Ebaid, H.; Alhazza, I.M. Antioxidant potential of Spirulina platensis mitigates oxidative stress and reprotoxicity induced by sodium arsenite in male rats. Oxid. Med. Cell. Longev. 2016, 7174351. [Google Scholar]
- Barkallah, M.; Ben Atitallah, A.; Hentat, F.; Dammak, M.; Hadrich, B.; Fendri, I.; Ayadi, M.-A.; Michaud, P.; Abdelkafi, S. Effect of Spirulina platensis biomass with high polysaccharides content on quality attributes of common Carp (Cyprinus carpio) and Common Barbel (Barbus barbus) fish burgers. App. Sci. 2019, 9, 2197. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of AOAC Internationa; AOAC International: Arlington, VA, USA, 2003. [Google Scholar]
- Bersuder, P.; Hole, M.; Smith, G. Antioxidants from a heated histidine glucose model system I: Investigation of the antioxidant role of histidine and isolation of antioxidants by high performance liquid chromatography. J. Am. Oil. Chem. Soc. 1998, 75, 181–187. [Google Scholar] [CrossRef]
- APHA. Compendium of Methods for the Microbiological Examination of Foods, 4th ed.; Downes, F.P., Ito, K., Eds.; American Public Health Association (APHA): Washington, DC, USA, 2001. [Google Scholar]
- Kvist, U.; Björndahl, L. Manual on Basic Semen Analysis. ESHRE Mono-graphs 2; Oxford University Press: Oxford, UK, 2002. [Google Scholar]
- WHO (World Health Organization). Laboratory Manual for Examination of the Human Semen and Sperm-Cervical Mucus interaction, 5th ed.; Lars, B., Trevor, G.C., Eds.; WHO (World Health Organization): Geneva, Switzerland, 2010; pp. 7–129. [Google Scholar]
- Wyrobek, A.J.; Bruce, W.R. Chemical induction of sperm abnormalities in mice. Proc. Nat. Acad. Sci. USA. 1975, 72, 4425–4429. [Google Scholar] [CrossRef]
- Draper, H.H.; Hadley, M. Malondialdehyde determination as index of lipid peroxidation. Methods. Enzymol. 1990, 86, 421–431. [Google Scholar]
- Levine, R.L.; Garland, D.; Oliver, C.N.; Amici, A.; Climent, I.; Lenz, A.G.; Ahn, B.W.; Shaltiel, S.; Stadtman, E.R. Determination of carbonyl content in oxidative modified proteins. Methods. Enzymol. 1990, 186, 464–478. [Google Scholar]
- Driver, A.S.; Kodavanti, P.S.; Mundy, W.R. Age-related changes in reactive oxygen species production in rat brain homogenates. Neurotox. Teratol. 2000, 22, 175–181. [Google Scholar] [CrossRef]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite and [I5N] nitrate in biological fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar] [CrossRef]
- Ellman, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 1959, 82, 70–77. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. Methods. Enzymol. 1984, 105, 121–126. [Google Scholar] [PubMed]
- Beauchamp, C.; Fridovich, I. Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 1971, 44, 276–287. [Google Scholar] [CrossRef]
- Flohe, L.; Gunzler, W.A. Assay of glutathione peroxidase. Methods. Enzymol. 1984, 105, 115–121. [Google Scholar]
- Kanno, S.; Ishikawsa, M.; Takayanagi, M.; Takayanagi, Y.; Sasaki, K. Potentiation of acetaminophen hepatotoxicity by doxapram in mouse primary cultured hepatocytes. Biol. Pharm. Bull. 2000, 23, 446–450. [Google Scholar] [CrossRef][Green Version]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–270. [Google Scholar]
- Yu, M.; Zhang, L.; Qiao, P.; Chang, B. Testicular oxidative stress and downregulation of CYP17a1 indused by di (n-butyl) phthalate inhibit synthesis of testosterone. Wei Sheng Yan Jiu 2015, 44, 364–370. [Google Scholar]
- Hossain, M.M.; Richardson, J.R. Mechanism of pyrethroid pesticide-induced apoptosis: role of calpain and the ER stress pathway. Toxicol. Sci. 2011, 122, 512–525. [Google Scholar] [CrossRef] [PubMed]
- Ogaly, H.A.; Khalaf, A.A.; Ibrahim, M.A.; Galal, M.K.; Abd-Elsalam, R.M. Influence of green tea extract on oxidative damage and apoptosis induced by deltamethrin in rat brain. Neurotoxicol. Teratol. 2015, 50, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Bao, J.; Kim, M.; Yuan, S.; Tang, C.; Zheng, H.; Mastick, G.S.; Xu, C.; Yan, W. Two miRNA clusters, miR-34b/c and miR-449, are essential for normal brain development, motile ciliogenesis, and spermatogenesis. Proc. Natl. Acad. Sci. USA 2014, 111, E2851–E2857. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, K.; Chuva de Sousa Lopes, S.M.; Kaneda, M.; Tang, F.; Hajkova, P.; Lao, K.; O’Carroll, D.; Das, P.P.; Tarakhovsky, A.; Miska, E.A.; et al. MicroRNA biogenesis is required for mouse primordial germ cell development and spermatogenesis. PLoS ONE 2008, 3, e1738. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Tang, C.; Zhang, Y.; Wu, J.; Bao, J.; Zheng, H.; Xu, C.; Yan, W. mir-34b/c and mir-449a/b/c are required for spermatogenesis, but not for the first cleavage division in mice. Biol. Open 2015, 4, 212–223. [Google Scholar] [CrossRef]
- Wang, C.; Yang, C.; Chen, X.; Yao, B.; Yang, C.; Zhu, C.; Li, L.; Wang, J.; Li, X.; Shao, Y.; et al. Altered profile of seminal plasma microRNAs in the molecular diagnosis of male infertility. Clin. Chem. 2011, 57, 1722–1731. [Google Scholar] [CrossRef]
- Nah, W.H.; Koh, I.K.; Ahn, H.S.; Kim, M.J.; Kang, H.G.; Gye, M.C. Effect of Spirulina maxima on spermatogenesis and steroidogenesis in streptozotocin-induced type I diabetic male rats. Food. Chem. 2012, 134, 173–179. [Google Scholar] [CrossRef]
- Sharma, R.S.; Pal, P.C.; Griffin, P.D.; Waites, G.M.; Rajalakshmi, M. Effects of levonorgestrel butanoate alone and in combination with testosterone buciclate on spermatogenesis in the bonnet monkey. Andrologia 2007, 39, 117–123. [Google Scholar] [CrossRef]
- Patil, G.R.; Rao, M.V. Role of ascorbic acid on mercuric chloride toxicity in vital organs of mice. Indian J. Environ. Toxicol. 1999, 9, 53–55. [Google Scholar]
- Gabr, G.A.; El-Sayed, S.M.; Hikal, M.S. Antioxidant Activities of Phycocyanin: A Bioactive Compound from Spirulina platensis. J. Pharm. Res. Int. 2020, 32, 73–85. [Google Scholar] [CrossRef]
- Eleiwa, N.Z.H.; Galal, A.A.A.; Abd El-Aziz, R.M.; Hussin, E.M. Antioxidant activity of Spirulina platensis alleviates doxorubicin-induced oxidative stress and reprotoxicity in male rats. Orient. Pharm. Exp. Med. 2018, 18, 87–95. [Google Scholar] [CrossRef]
- Feriani, A.; Hachani, R.; Tir, M.; Ghazouani, L.; Mufti, A.; Borgi, M.A.; Allagui, M.S. Bifenthrin exerts proatherogenic effects via arterial accumulation of native and oxidized LDL in rats: the beneficial role of vitamin E and selenium. Environ. Sci. Pollut. Res. Int. 2018. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, Y.L.; Abd El-Ghffar, E.A. Spirulina ameliorates aspirin-induced gastric ulcer in albino mice by alleviating oxidative stress and inflammation. Biomed. Pharmacother. 2019, 109, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Hakim, Y.M.; Mohamed, W.A.; El-Metwally, A.E. Spirulina platensis attenuates furan reprotoxicity by regulating oxidative stress, inflammation, and apoptosis in testis of rats. Ecotox. Environ. Saf. 2018, 161, 25–33. [Google Scholar] [CrossRef]
- Yang, Y.H.; Du, L.; Hosokawa, M.; Miyashita, K. Spirulina Lipids Alleviate Oxidative Stress and Inflammation in Mice Fed a High-Fat and High-Sucrose Diet. Mar. Drugs 2020, 18, 148. [Google Scholar] [CrossRef]
- Lee, J.; Park, A.; Kim, M.J.; Lim, H.J.; Rha, Y.A.; Kang, H.G. Spirulina Extract Enhanced a Protective Effect in Type 1 Diabetes by Anti-Apoptosis and Anti-ROS Production. Nutrients 2017, 9, 1363. [Google Scholar] [CrossRef] [PubMed]
- Śmieszek, A.; Giezek, E.; Chrapiec, M.; Murat, M.; Mucha, A.; Michalak, I.; Marycz, K. The Influence of Spirulina platensis Filtrates on Caco-2 Proliferative Activity and Expression of Apoptosis-Related microRNAs and mRNA. Mar. Drugs 2017, 15. [Google Scholar]

| Composition | Values | Physical Properties | Values |
|---|---|---|---|
| Protein (%) | 64.35 ± 1.2 | Appearance | Uniform powder |
| Fat (%) | 7.46 ± 0.42 | color | Blue green |
| Saturated acids | 42.5 ± 1.5 | Odor and taste | Mild like algae |
| Lauric (C12:0) | 0.9 ± 0.1 | Consistency | Powder |
| Myristic (C14:0) | 0.53 ± 0.04 | Particles size | 60 mesh (> 98%) |
| Palmitic (16:0) | 39.22 ± 0.8 | pH | 6.9 ± 0.2 |
| Stearic (C18:0) | 1.85 ± 0.2 | ||
| Monounsaturated | 14.78 ± 0.22 | ||
| Palmitoleic (C16:1) | 6.53 ± 0.3 | ||
| Oleic (C18:1) | 8.25 ± 0.4 | ||
| Polyunsaturated | 41.55 ± 0.78 | ||
| Linoleic (C18:2) | 18.02 ± 0.24 | ||
| Gamma-linolenic (C18:3) | 22.27 ± 0.1 | ||
| Dihomo-γ-linolenic (C20:3) | 1.26 ± 0.08 | ||
| Carbohydrate (%) | 21.9 ± 1.1 | ||
| Total dietary fiber (%) | 8.85 ± 0.36 | ||
| Sugar (%) | 2.9 ± 0.5 | ||
| Starch | 3.7 ± 0.1 | ||
| Ash (%) | 6.8 ± 0.05 | ||
| Iron (mg/100 g on dry weight basis) | 336 ± 19.8 | ||
| Calcium (mg/100 g on dry weight basis) | 998 ± 14.5 | ||
| Magnesium (mg/100 g on dry weight basis) | 1.35 ± 0.03 | ||
| Potassium (mg/100 g on dry weight basis) | 2150 ± 37.5 | ||
| Sodium (mg/100 g on dry weight basis) | 1380 ± 28.4 | ||
| Vitamins (mg/ 100 g on dry weight basis) | |||
| Vitamin B1 | 5.53 ± 0.9 | ||
| Vitamin B2 | 4.99 ± 0.78 | ||
| Vitamin B7 | 46 ± 4.5 | ||
| Vitamin B9 | 9.88 ± 1.23 | ||
| Vitamin E | 8.98 ± 0.9 | ||
| Phytopigments (mg/100 g on dry weight basis) | |||
| Β-carotene | 1480 ± 10.2 | ||
| Chlorophylls | 2350 ± 29.7 | ||
| Phycocyanin | 52 ± 2.3 | ||
| Microbiological quality (CFU g−1) | |||
| Total plate count | < 2 × 102 | ||
| Total coliforms | < 10 | ||
| Yeasts and molds | Negative | ||
| Escherichia coli | Negative | ||
| Salmonella spp. | Negative | ||
| Listeria spp. | Negative | ||
| Staphylococcus aureus | Negative | ||
| Specific contaminants (ppm) | |||
| Arsenic | < 0.5 | ||
| Mercury | < 0.05 | ||
| Cadmium | < 0.2 | ||
| Lead | < 0.5 | ||
| Pesticides | Negative | ||
| DPPH radical-scavenging activity (%) at 100 µg mL−1 | 42 ± 0.54 |
| Parameters | C | BF | SP + BF | SP |
|---|---|---|---|---|
| Initial body weight (g) | 31.5 ± 3.00 | 30.25 ± 4.92 | 27 ± 1.00 | 27 ± 2.83 |
| Final body weight (g) | 35 ± 4.69 | 29.75 ± 3.30 | 30 ± 2.63 | 30.75 ± 4.27 |
| Body weight gain (g) | 3.5 | −0.5 B | 3 E | 3.75 E |
| Absolute organs weights (g) | ||||
| Testes | 0.3 ± 0.09 | 0.17 ± 0.004 B | 0.23 ± 0.005 D | 0.24 ± 0.004 D |
| Epididymides | 0.12 ± 0.008 | 0.07 ± 0.016 | 0.08 ± 0.008 | 0.10 ± 0.012 |
| Sperm parameters | Control | BF | SP + BF | SP |
|---|---|---|---|---|
| Spermatozoa count per epididymis (×106) | 5.82 ± 0.28 | 2.3 ± 2.45 C | 5.45 ± 0.82 F | 5.58 ± 0.91 F |
| Motility (%) | 76.75 ± 15.56 | 45 ± 7.07 B | 81.25 ± 7.5 E | 91.75 ± 7.89 F |
| Viability (%) | 97.5 ± 1.00 | 95 ± 2.45 A | 96.75 ± 2.06 D | 98 ± 0.82 D |
| Abnormal forms (%) | 5.75 ± 2.06 | 12.75 ± 2.87 B | 7.5 ± 0.02 E | 3.5 ± 1.00 F |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barkallah, M.; Ben Slima, A.; Elleuch, F.; Fendri, I.; Pichon, C.; Abdelkafi, S.; Baril, P. Protective Role of Spirulina platensis against Bifenthrin-Induced Reprotoxicity in Adult Male Mice by Reversing Expression of Altered Histological, Biochemical, and Molecular Markers Including MicroRNAs. Biomolecules 2020, 10, 753. https://doi.org/10.3390/biom10050753
Barkallah M, Ben Slima A, Elleuch F, Fendri I, Pichon C, Abdelkafi S, Baril P. Protective Role of Spirulina platensis against Bifenthrin-Induced Reprotoxicity in Adult Male Mice by Reversing Expression of Altered Histological, Biochemical, and Molecular Markers Including MicroRNAs. Biomolecules. 2020; 10(5):753. https://doi.org/10.3390/biom10050753
Chicago/Turabian StyleBarkallah, Mohamed, Ahlem Ben Slima, Fatma Elleuch, Imen Fendri, Chantal Pichon, Slim Abdelkafi, and Patrick Baril. 2020. "Protective Role of Spirulina platensis against Bifenthrin-Induced Reprotoxicity in Adult Male Mice by Reversing Expression of Altered Histological, Biochemical, and Molecular Markers Including MicroRNAs" Biomolecules 10, no. 5: 753. https://doi.org/10.3390/biom10050753
APA StyleBarkallah, M., Ben Slima, A., Elleuch, F., Fendri, I., Pichon, C., Abdelkafi, S., & Baril, P. (2020). Protective Role of Spirulina platensis against Bifenthrin-Induced Reprotoxicity in Adult Male Mice by Reversing Expression of Altered Histological, Biochemical, and Molecular Markers Including MicroRNAs. Biomolecules, 10(5), 753. https://doi.org/10.3390/biom10050753








